Abstract
Bariatric surgery is an effective method to rapidly induce weight loss in severely obese people, however its impact on brain functional connectivity after longer periods of follow-up is yet to be assessed. We investigated changes in connectivity in 16 severely obese women one month before, one month after and one year after Roux-en-Y gastric bypass surgery (RYGB). 12 lean controls were also enrolled. Resting-state fMRI was acquired for all participants following an overnight fast and after a 260 kcal load. Connectivity between regions involved in food-related saliency attribution and reward-driven eating behavior was stronger in presurgery patients compared to controls, but progressively weakened after follow-up. At one year, changes in networks related to cognitive control over eating and bodily perception also occurred. Connectivity between regions involved in emotional control and social cognition had a temporary reduction early after treatment but had increased again after one year of follow-up. Furthermore, we could predict the BMI loss by presurgery connectivity in areas linked to emotional control and social interaction. RYGBP seems to reshape brain functional connectivity, early affecting cognitive control over eating, and these changes could be an important part of the therapeutic effect of bariatric surgery.
Introduction
Bariatric surgery can be considered as an effective method to rapidly induce weight loss in severely obese people. Patients exceeding 40 kg/m2 in body mass index (BMI) are usually eligible for weight loss surgery; however, lower BMI (35–40 kg/m2) can be considered if obesity comorbidities are also present1. Different procedures are currently available for bariatric surgery, all performed laparoscopically. Roux-en-Y gastric bypass (RYGB) is an irreversible procedure, which excludes the major part of the stomach by connecting a small gastric pouch directly to the proximal part of the small bowel (first limb of the Y). The body of the stomach and the duodenum (second limb of the Y) are reattached further down the small intestine2. RYGB induces weight loss by combining the effect of gastric reduction, hormonal changes when bypassing the duodenum and malabsorption2. However, a problem in bariatric surgery is the long-term maintenance of low weight, and educational and psychological support must be granted to the patients during follow-up2.
Psychological and cognitive changes in patients undergoing bariatric surgery procedures can be explored using advanced neuroimaging techniques, such as functional magnetic resonance imaging (fMRI), which registers the neuronal activity of the brain by measuring the blood-oxygenation level dependent (BOLD) response. fMRI can be performed with a block-design, when the participant has to perform a task or is passively exposed to a stimulus, or during resting-state, when the participant is simply laying still in the MRI scanner and is instructed not to focus his/her thoughts on anything specific. Most of the studies that have been performed on patients undergoing bariatric surgery have used a block-design to compare operated patients with lean controls, showing changes in reward-related circuitry activity3, 4. Others have used a cross-sectional design, comparing obese patients with a different group of operated patients5. Some studies have not included lean controls, but either compared the effect of surgical versus behavioral weight loss6, or only included patients studied before and after RYGB7–9 with a short-term follow-up (4 to 12 weeks). Finally one study, performed on patients before and after RYGB, specifically focused on taste perception modifications after surgery10.
Resting-state connectivity, on the other hand, is reflective of brain activity when the brain is not engaged in any task, and has been considered as a “baseline” for brain activity11. As the individual is not performing any task, bias due to differences in protocols design can be avoided. However, very few studies have used resting-state fMRI to investigate functional changes in brain activity or connectivity in bariatric surgery patients. One study had a cross-sectional design5, while another study, which had a longitudinal design with eight months of follow-up and also included lean controls, only focused on the functional connectivity of the hypothalamus before and after the ingestion of a glucose solution12. There are only two studies that have explored whether neural activity relates to BMI loss, despite long-term BMI loss is the primary end-point for bariatric surgery. One study specifically focused on the executive control network activity and was performed on patients after bariatric surgery and did not include any baseline presurgery data13. The other study tested BMI loss at six months follow-up against presurgery cerebral activity during the viewing of food versus non food pictures, showing that the activity in areas involved in cognitive control was related to low BMI maintenance14.
We have previously investigated the changes in resting-state neural activity in the fasted and sated state, measured as fractional amplitude of low frequency fluctuations (fALFF), in severely obese women before and one month after RYGB, showing that bariatric surgery rapidly alters brain activity15. With the present study, we aimed to investigate whether and to what extent bariatric surgery could reshape brain functional connectivity, with a specific focus on connectivity changes occurring in the long-term. We have assessed modifications in the whole-brain resting-state connectivity of 16 severely obese patients before RYGB, after a short-term follow-up of one month and after a long-term follow-up of one year. We explored the effect of the feeding condition, studying both patients and lean controls in the fasted and sated states. Moreover, we tested whether presurgery connectivity could predict BMI loss after one year of follow-up.
Results
Main effect of group
Lean controls vs presurgery patients
A main effect of group was found on the connectivity of precuneus (p < 0.000), left cerebellum (0.001), right superior lateral occipital cortex (SLOC) (p < 0.001), medial frontal cortex (MedFC) (p < 0.001), posterior cingulate cortex (PCC) (p < 0.002), left amygdala (p < 0.002), right planum temporale (p < 0.006) and right anterior parahippocampal gyrus (p < 0.01) (Table 1). No effect of the feeding condition was found.
Table 1.
Lean controls vs presurgery patients: effect of group on connectivity.
| F-test | Seed ROI | F | p uncorr |
|---|---|---|---|
| Main Effect of Group | Precuneus | 29.48 | 0.000 |
| Cerebellum 9 L | 22.84 | 0.001 | |
| Superior Lateral Occipital Cortex R | 22.82 | 0.001 | |
| Medial Frontal Cortex | 21.8 | 0.001 | |
| Posterior Cingulate Cortex | 21.4 | 0.002 | |
| Amygdala L | 21.31 | 0.002 | |
| Planum Temporale R | 18.05 | 0.006 | |
| Anterior Parahippocampal Gyrus R | 16.89 | 0.010 |
Within-patients: presurgery vs one month vs one year follow-up
A main effect of time on connectivity was found in the PCC (p < 0.002), in the precuneus (p < 0.003), in the left supplementary motor area (SMA) (p < 0.004) and in the right posterior middle temporal gyrus (p < 0.005), the right amygdala (p < 0.006) and the vermis (p < 0.01) (Table 2). A main effect of the feeding condition was found in the left occipital fusiform gyrus (p < 0.000) and in the left posterior temporal fusiform gyrus (p < 0.001) (Table 2).
Table 2.
Within patients main effect of time and feeding condition on connectivity.
| Test | Seed ROI | F | p uncorr |
|---|---|---|---|
| Main Effect of Time | Posterior Cingulate Cortex | 25.54 | 0.002 |
| Precuneus | 20.38 | 0.003 | |
| Supplementary Motor Area L | 16.91 | 0.004 | |
| Posterior Middle Temporal Gyrus R | 16.32 | 0.005 | |
| Amygdala R | 14.77 | 0.006 | |
| Vermis 10 | 11.42 | 0.010 | |
| Main Effect of Feeding | Occipital Fusiform Gyrus L | 46.30 | 0.000 |
| Posterior Temporal Fusiform Gyrus L | 20.70 | 0.001 |
Lean controls vs patients at one year follow-up
A main effect of group was found on the connectivity of the left amygdala (p < 0.001), not surviving the threshold for multiple comparisons at the post-hoc test. No effect of the feeding condition was detected.
Post-hoc analyses and seed-to-target connectivity over time across groups
For a full list of specific seed-to-target found to be significantly different between groups, see Supplementary Materials (Tables S1–S3). Some seed-to-ROI connections, which were stronger in patients presurgery when compared with controls, appeared to have weakened after surgery (Table 3). Specifically, connectivity between the precuneus and the left anterior STG was stronger in presurgery patients compared with controls, but was weaker in patients after one month and one year follow-up, compared with presurgery (Fig. 1). Moreover, no differences were found in the connectivity between these regions when comparing controls and patients after one year follow-up. The same pattern was seen for connectivity from the PCC to the left aSTG and the right anterior temporal fusiform gyrus (Fig. 1), suggesting that these connections are the most prominently involved in determining obesity in these patients and the most affected by the surgery.
Table 3.
Seed-to-target changes in connectivity over time.
| Seed ROI | Target ROI | T-values | p FDR-corr | ||||
|---|---|---|---|---|---|---|---|
| PS > LC | PS > 1 m | PS > 1 y | PS > LC | PS > 1 m | PS > 1 y | ||
| Precuneus | Anterior Superior Temporal Gyrus L | 7.9 | 4.9 | 4.5 | 0.00 | 0.02 | 0.04 |
| Posterior Supramarginal Gyrus L | 3.7 | 4.2 | 0.04 | 0.04 | |||
| Temporo-occipital Middle Temporal Gyrus R | 3.5 | 3.7 | 0.04 | 0.05 | |||
| Amygdala R | 3.3 | 8.1 | 0.05 | 0.00 | |||
| Posterior Cingulate Cortex | Posterior Temporal Fusiform Gyrus R | 5.7 | 5.8 | 0.00 | 0.02 | ||
| Anterior Superior Temporal Gyrus L | 4.1 | 5.1 | 6.2 | 0.02 | 0.02 | 0.02 | |
| Inferior Lateral Occipital Cortex R | 3.7 | 3.4 | 0.03 | 0.05 | |||
| Amygdala R | 3.5 | 5.3 | 0.05 | 0.02 | |||
| Anterior Middle Temporal Gyrus R | 3.4 | 4.1 | 0.05 | 0.04 | |||
| Precentral Gyrus L | 3.3 | 4.0 | 0.05 | 0.03 | |||
| Anterior Temporal Fusiform Gyrus R | 3.3 | 4.4 | 4.6 | 0.05 | 0.03 | 0.04 | |
PS = presurgery. 1 m = 1-month follow-up. 1 y = 1-year follow-up.
Figure 1.
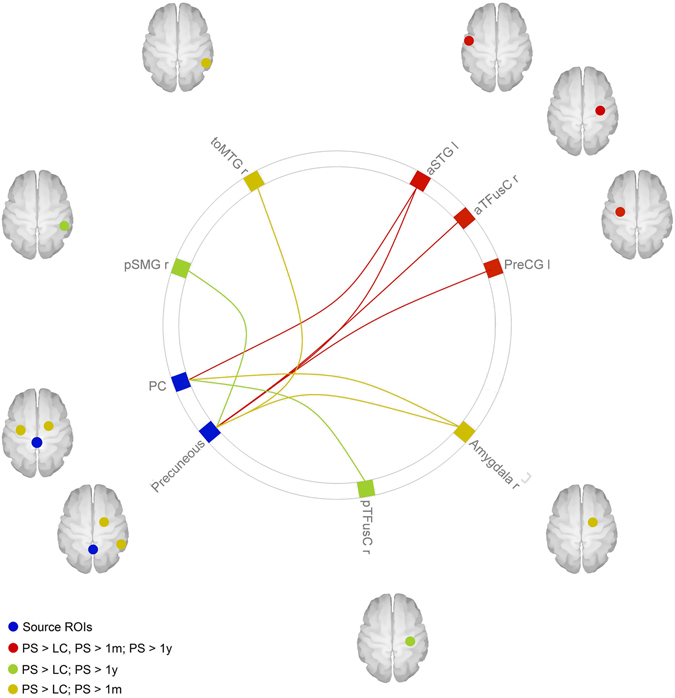
Seed-to-target connectivity changes over time. The figure shows the seed-to-ROI connectivity found to be significantly different between groups. The red lines represent connections found to be stronger in presurgery patients (PS) compared to all other groups (lean controls, 1 month follow-up and 1 year follow-up). The yellow lines represent connections where a minor change occurred, as they were found to be stronger in presurgery patients compared with both lean controls (LS) and 1 month follow-up (1 m), but not to 1 year follow-up (1 y) nor between lean controls and 1 year follow-up. The green lines represent the connections where a change in connectivity was observed more slowly, as their connectivity was stronger in presurgery patients compared with lean controls and 1 year follow-up, but not compared with 1 month follow-up. All results were significant with a p < 0.05, corrected for multiple comparisons with a FDR approach. The image was generated with the CONN software used for the analysis.
While the abovementioned connections showed changes in their strength already in the short-term, other connectivity changes required a longer period of follow-up to be appreciated (Table 3). In particular, the connectivity from the precuneus to the left posterior supramarginal gyrus (SMG) and from the PCC to the right posterior temporal fusiform gyrus and right anterior MTG, which was greater in presurgery patients compared with controls, at 1-moth follow-up showed no significant changes, but appeared to have normalized after one year of follow-up (Fig. 1). Indeed, the reduction in connectivity between these ROIs was only found in patients after one year follow-up compared to presurgery, and previously observed differences compared with controls were no longer detectable.
Other connections also appeared to be involved, but with a more peculiar pattern. For example, the connectivity from the precuneus to the right MTG and right amygdala, as well as the connectivity from the PCC to the right amygdala, left precentral gyrus and right inferior lateral occipital cortex, was greater in presurgery patients compared with controls, but it had reduced after one month follow-up (Table 3; Fig. 1). However, no differences could be detected after one year follow-up when compared to either presurgery patients or controls, suggesting that after a temporary reduction at one month follow-up, the connectivity between these regions had somewhat settled halfway between controls and presurgery patients - still at a higher set-point compared with controls, but not as high as before surgery. A longer follow-up period would then be required to investigate further changes, and whether the connectivity between these areas might return to presurgery levels over time.
Main effect of the feeding condition across groups
At the post-hoc analysis, within-patients connectivity from the left occipital fusiform gyrus was found to be greater in the fasted, rather than the sated state, to the left posterior temporal fusiform gyrus (p FDR-corr < 0.01), to the right cerebellar first and second divisions (p FDR-corr < 0.02 and p FDR-corr < 0.05 respectively), to the left occipital pole (p FDR-corr 0.05) and to the left SLOC (p FDR-corr < 0.05) (Table 4, Fig. 2). Connectivity from the left posterior temporal fusiform gyrus to the left and right occipital fusiform gyrus (p FDR-corr < 0.01) and to the right precentral gyrus (p FDR-corr < 0.05) was also greater in the fasted rather than the sated state (Table 4; Fig. 2).
Table 4.
Within-patients: connections with greater seed-to-target connectivity in the sated compared with the fasted state.
| Seed ROI | Target ROI | T | p FDR-corr analyses-level |
|---|---|---|---|
| Occipital Fusiform Gyrus L | Posterior Temporal Fusiform Gyrus L | 6.21 | 0.01 |
| Cerebellum 1 R | 6.06 | 0.01 | |
| Occipital Pole L | 4.53 | 0.05 | |
| Cerebellum 2 R | 4.24 | 0.05 | |
| Superior Lateral Occipital Cortex L | 4.21 | 0.05 | |
| Posterior Temporal Fusiform Gyrus L | Occipital Fusiform Gyrus L | 6.21 | 0.01 |
| Occipital Fusiform Gyrus R | 6.12 | 0.01 | |
| Precentral Gyrus R | 4.35 | 0.05 |
Figure 2.
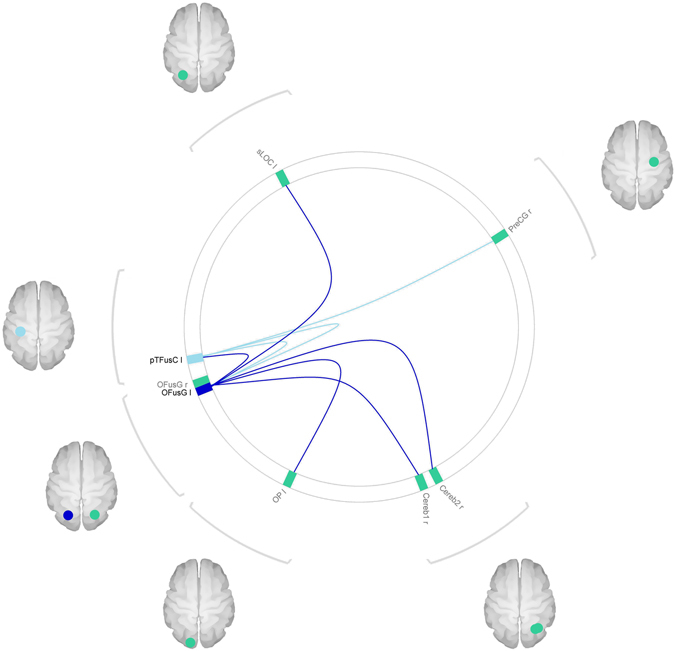
Within-patients effect of the feeding condition on connectivity. The figure shows connections where connectivity was found to be greater in the sated state compared to the fasted state, in patients. Blue circles represent seeds regions; green circles represent target ROIs. The image was generated with the CONN software used for the analysis.
Effect of the interaction between group and feeding status on connectivity
Lean controls vs presurgery patients
An effect of the interaction between group and feeding condition was found on the connectivity of the left anterior inferior temporal gyrus (ITG) (p < 0.007) and of the right hippocampus (p < 0.009) (Table S4, Supplementary Materials). At the post-hoc t-test, however, only the differences in the hippocampal connectivity survived the correction for multiple comparisons. In particular, the right hippocampus showed a greater connectivity with the right parahippocampal gyrus (p FDR-corr < 0.04) in pre-surgery patients compared with controls, only in the sated state (Table S4, Supplementary Materials).
Within-patients: presurgery vs one month vs one year follow-up
No effect of the interaction between time and feeding condition on connectivity was detected.
Lean controls vs patients at one year follow-up
An effect of the group*condition interaction was also found on the connectivity of the temporo-occipital fusiform gyrus (p < 0.003) and of the vermis (p < 0.005). However, no specific seed-to-target connectivity differences survived the FDR correction for multiple comparisons at the post-hoc t-tests.
Correlation between presurgery connectivity and BMI reduction at one year
An effect of BMI reduction at one year was found on presurgery connectivity of the right paracingulate gyrus (p < 0.000) in the sated state. Specifically, a positive correlation was found at the post-hoc analysis between BMI loss and connectivity between the right paracingulate gyrus and the right amygdala (p FDR-corr < 0.00), in the sated state (Fig. 3).
Figure 3.
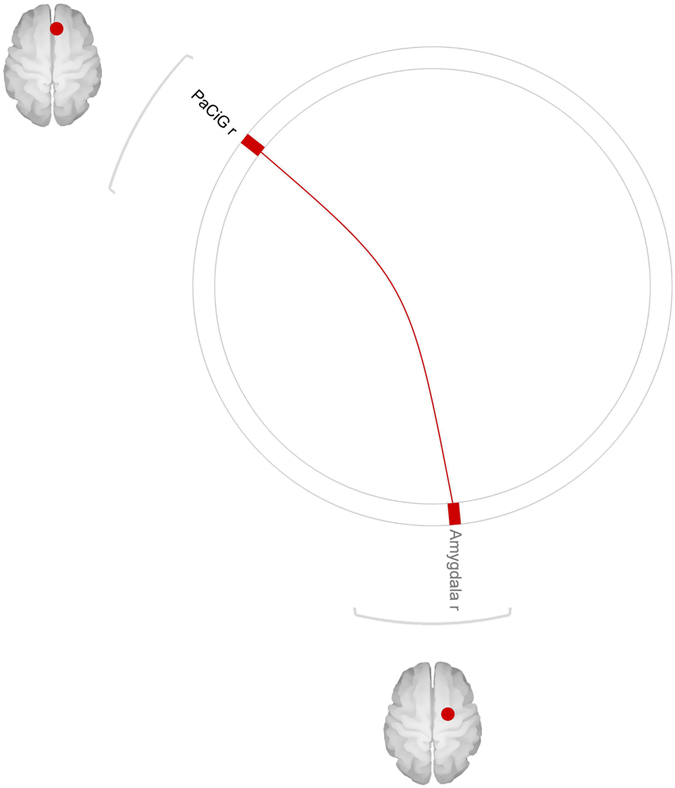
Positive correlation between baseline connectivity and BMI loss at 1 year follow-up. The figures shows seed (black font) to target (grey font) connections where a positive correlations was found between presurgery connectivity and BMI loss over 1 year of follow-up. Results are corrected for multiple comparisons with a FDR approach. The image was generated with the CONN software used for the analysis.
Discussion
We investigated the longitudinal changes in brain functional connectivity in 11 severely obese patients one month before, and one month and one year after bariatric surgery, and compared them to 12 lean controls. We focused on seed-to-target connections which were found to be importantly changed over time. Some connections, which were stronger in presurgery patients compared with controls, showed a trend for progressive weakening in connectivity, suggesting their involvement in obesity as well as in surgery-induced modifications. Moreover, we found that presurgery connectivity between the right paracingulate gyrus and the right amygdala in the sated state could predict one-year BMI loss, which is a primary end-point of bariatric surgery.
The connectivity from the PCC to the left aSTG and to the right anterior temporal fusiform gyrus, and from the precuneus to the left aSTG, was stronger in presurgery patients compared with controls, but appeared to have progressively weakened at one month and one year follow-up. The PCC is a hub of the default-mode network (DMN), the activity of which is elicited by internally-focused attention16. Both the PCC and the STG have been found to activate in response to food commercials rather than non-food commercials in adolescents17, 18, and to high energy versus low energy food cues in obese individuals19, 20, suggesting their role in a reward-driven eating behavior20. Moreover, the activity in the STG has been found to correlate with insulin resistance levels19. On the other hand, the anterior fusiform gyrus is involved in integrating relevant sensory cues along the visual stream21. Thus, the reduction in connectivity from the PCC to the STG and anterior fusiform gyrus might be indicative of a blunted drive toward rewards. Specifically, connectivity between the PCC and the left STG was greater, in presurgery patients compared with one year follow-up, in the fasted rather than the sated state (group vs condition interaction), suggesting that the reduction in the drive for reward induced by surgery is particularly evident when the individual is fasting (i.e. hungry). In line with this hypothesis, the connectivity from the precuneus to the left aSTG was reduced after surgery as well. The precuneus is involved in salience attribution22 and is associated with the processing of introspective information23, such as hunger sensation, as well as with the representation of the self24. Thus, the reduced connectivity from the precuneus to the STG after surgery, is also suggestive of a reduced drive toward salient and rewarding stimuli, e.g. food, due to a decreased sensitivity to the hunger sensation. Indeed, resting-state connectivity strength in the precuneus had been previously reported to be greater in obese versus lean individuals25, and its activity in response to food-related cues has been found to decrease in obese patients undergoing different types of bariatric surgery, such as laparoscopic gastric banding6 or RYGB intervention26. However, precuneus resting-state activity had been also found to decrease after non-surgical weight-loss27, suggesting that weight-loss in general, rather than bariatric surgery itself, might be responsible for such changes. Still, the surgical intervention might lead to more pronounced connectivity modifications after weight reduction following RYGB intervention, compared with non-surgical weight reduction3. The reduction in connectivity with the anterior fusiform gyrus might also be due to the massive weight loss induced by surgery, as functional connectivity of the anterior fusiform gyrus has been found to be positively associated with weight status28.
Other connections, such as those from the precuneus to the left posterior SMG and from the PCC to the right posterior temporal fusiform gyrus and to the right aMTG, showed the same pattern but with a slower adjustment, as the reduction in their connectivity could only be appreciated at one year follow-up. SMG is involved in the processing of bodily parts29, as well as in saliency detection30 and self regulation31. Connectivity between the precuneus and the supramarginal gyrus has been found to correlate with hypocondriac traits in patients with irritable bowel syndrome32. The reduced connectivity from the precuneus to the SMG in our patients might therefore indicate a lesser tendency to worry about own body sensations. The fusiform gyrus GM volume is associated to waist circumference and obesity33. The posterior fusiform gyrus has been reported to be more active, in response to food cues, in obesity-risk FTO allele rather than in non-risk34. It is also more active in the sated than in fasted state in response to food imagery35. The right MTG shows greater activity in response to food cues, in the sated state, in bariatric surgery patients versus behavioral dieters6. Interestingly, resting-state amplitude of low frequency fluctuations (ALFF), which is a measure of neural activity, in the right MTG and right fusiform gyrus has been found to positively correlate with dietary restraint (i.e. cognitive control over eating)28, but at the same time greater ALFF in the right MTG resulted in a higher, rather than lower, BMI28. We can therefore speculate that a more effective semantic attribution to food, in which the MTG is involved, might improve self-consciousness about the consequences of eating. Thus, MTG connectivity might be more important than isolated activity in determining an effective dietary restraint, particularly through its connections with frontal regions, prominently involved in inhibitory control. On the other hand, the activity of the MTG per se might result in a greater semantic value of food, and in a higher BMI. In this frame, the post-surgery decrease in connectivity from the PCC to the right MTG and fusiform gyrus could indicated, in our sample, a reduced tendency to focus own attention on food imagery and on highly semantic thoughts, such as food-related thoughts, during mind-wandering. Interestingly, WM in both the fusiform and middle temporal gyri has been reported to correlate with BMI36. This corroborates that the reduction in their connectivity might be involved – or be subsequent to – weight reduction after surgery. Moreover, the changes in functional connectivity might result from the reshaping of structural connections, explaining the necessity for a longer period of follow-up in order to detect a decrease.
A different pattern was observed for the connectivity from the precuneus to the right MTG and the right amygdala, as well as from the PCC to the right amygdala, left precentral gyrus and right inferior lateral occipital cortex. The connectivity between these regions was in fact greater in presurgery patients compared with controls, and though it had reduced after one month follow-up, after one year of follow-up it appeared to have settled halfway between controls and presurgery patients levels. The amygdala is involved in emotional attribution to stimuli37, particularly when evaluating threats, and in social anxiety38. The right amygdala is involved in affective information retrieval relative to visual stimuli39 and in stress-sensitivity40. Moreover, precuneus and amygdala have both a role in the perception of negative feelings, such as the feeling of guilt (precuneus) and embarassment (amygdala)41. The reduction in connectivity from the precuneus, involved in the representation of the self, to the amygdala might indicate a lesser rumination on negative feelings of concern about the body shape and appearance, as also suggested by the reduced connectivity between the PCC and the amygdala. However, such reduction in connectivity was more prominent in the fed state, rather than in the fasted state, suggesting that hunger might still cause anxiety, but even a low caloric load might be enough to subdue it in the immediate follow-up. The connectivity between the PCC to the lateral occipital cortex and to the left precentral gyrus was also found to be greater in presurgery patients compared with lean controls, and to be reduced only at one month follow-up. The lateral occipital cortex cortical thickness has been reported to be associated to the visceral adiposity in obesity42. As for the connectivity toward the precentral gyrus, it has been hypothesized that the greater connectivity between these two regions in unoperated obese people is reflective of a compensatory increase in control-related connectivity during volitional appetite43. This need for an overactive control might have decreased after surgery in our sample, as indicated by the decrease in connectivity.
We also found a positive correlation between presurgery connectivity from the right paracingulate to right amygdala in sated state, and BMI loss over one year of follow-up. The paracingulate cortex is involved in social interactions and in predicting the consequences of social behavior44. It has also been associated with expressive suppression (i.e. the hiding of an emotion)45, whereas the right amygdala has been associated with the use of cognitive reappraisal (i.e. re-interpreting a situation with the aim of changing its emotional impact)45. This might suggest that a better control over emotions and social interactions might help achieving a more sustained reduction in BMI after surgery.
Three patients included in our studiy had been treated with citalopram for past depression. Changes in brain volume as well as in connectivity have been reported to exist in depressed patients46, involving areas such as the PCC and the amygdala. However, the few studies performed on drug-naïve depressed patients undergoing treatment with selective serotonine re-uptake inhibitor (SSRI) have shown a normalization of functional activity after treatment with citalopram47 and escitalopram48. Given the small size of our sample, and given the absence of a current diagnosis of depression, we have therefore decided to retain the abovementioned three patients in our study. However it must be noted that only few studies have been performed using fMRI on patients undergoing antidepressant treatment, thus the issue of brain activity changes induced by SSRI should be clarified, and caution must be used in interpreting our results.
This is the first long-term longitudinal study of resting-state connectivity in patients undergoing bariatric surgery. Overall, our results suggest that, after surgery, there is a progressive reduction of food-related thinking which appears to be an important part of the therapeutic progress of the patients, while the emotional aspect related to the external changes of own body seems to be more complex. Some limitations, however, have to be considered. Though our results are in line with previous knowledge and studies on bariatric surgery patients, the small sample size calls nonetheless for a careful interpretation of our findings, and future studies on larger samples will have to be performed in order to validate our results. A longer period of follow-up could also help to further exploring the tendency for some seed-to-target connections to restore their previous level of connectivity over time. Future studies might be designed to also include some detailed emotional and psychological evaluation of the patients, as well as their socio-economic status, in order to investigate the impact of bariatric surgery on the relationship between social/emotional cognition and neural connectivity. Finally, it must be noticed that our study only included females participants. We focused on females as there is a well-documented disparity in the number of females versus men seeking bariatric surgery, with percentages ranging from 70–80% versus 20–30%49, 50. However, gender differences have been reported in brain functional connectivity51, specifically in the PCC52 and in the default mode network connectivity53, though this finding is not constant54. thus a comparison with a sample of male participants would is needed in order to validate and generalize our results.
Conclusions
We investigated connectivity changes in 11 severely obese woman undergoing Roux-en-Y gastric bypass surgery one month before, one month after and one year after follow-up, and twelve lean controls. We found that changes in hunger perception and reward-seeking behavior occur early after surgery and persist in the long-term, while changes in networks related to cognitive control over eating and bodily perception require a longer time to manifest, probably due to the need of prior reshaping of the underlying structural connections. Emotional control and social cognition related circuitry has a temporary reduction in connectivity early after treatment but increased again after one year follow-up, posing the need for further studies to clarify its role in postsurgical neural modifications. Furthermore, presurgery connectivity in areas linked to emotional control and social interaction was predictive of postsurgical BMI loss, suggesting that a better capacity in controlling own emotions might play a role in maintaining a lower BMI.
Our study suggests that Roux-en-Y gastric bypass surgery might reshape brain functional connectivity, early affecting cognitive control over eating, and that the connections between brain regions related to emotional control might play an important role in the maintenance of a reduced BMI.
Methods
All participants gave written informed consent prior to any experimental testing. The present study was approved by the Regional Ethical Review Board in Uppsala, and the procedures followed were in accordance with the Helsinki Declaration. Participants were sixteen severely-obese, female adult patients seeking bariatric surgery at the University Hospital of Uppsala, and twelve female lean controls (LC). All participants were Northern Europeans, right-handed and non-smokers. None of the participants had contraindications for MRI scanning procedures.
Recruitment and screening were performed by the doctor consulting the surgery. Participants’ self-reported their mental health medical history, and the use of major psychoactive drugs (such as opioid, benzodiazepines, psychotropic substances) was considered an exclusion criteria, as well as previous bariatric surgery and diabetes. Patients were psychologically evaluated by the General Practitioner (GP) before being referred to the surgery clinic. At the surgery clinic, patients were further evaluated by a psychologist in order to confirm the absence of psychiatric conditions, which is a requirement for the surgery. Three patients using a maintenance dose of citalopram (a selective, serotonin-reuptake inhibitor; 20 mg, once per day) for past depression were included. A full list of inclusion/exclusion criteria is reported in Table 5. Patients had a mean ± sd BMI of 42.9 ± 4.7 kg/m2, and were aged 39 ± 11 (min = 19, max = 55) years. Patients were studied one month before surgery, one month after surgery and one year after surgery. After drop-outs, fifteen patients were included at one month, 11 of which with MRI scanning; at one year, thirteen patients were still in the study (80%), 11 of which with MRI scanning (see Table S5, Supplementary Materials). Six of the patients in the longitudinal study had more than 12 years of education; the remaining 5 had between 9 and 12 years of education. LC had a mean ± sd BMI of 22.7 ± 1.7 kg/m2, and were aged 36 ± 12 (min = 22, max = 55) years.
Table 5.
Inclusion and exclusion criteria from the study.
| Inclusion Criteria: | Exclusion Criteria: |
|---|---|
| 1 Patients completing bariatric surgery | 1 Claustrophobia, metallic implants, or other contraindications for MRI scanning |
| 2 Sex = female | 2 Previous bariatric surgery |
| 3 BMI ≥ 35 kg/m2 | 3 Illicit drug use |
| 4 Age = 18–60 years | 4 Diabetic or use of insulin |
| 5 Right-handed | 5 History of alcohol, drug, or medication abuse in last 2 years |
| 6 Non-smoking | 6 Current psychiatric diagnosis |
| 7 Use of psychoactive drugs | |
| 8 Use of opiates or benzodiazapines | |
| 9 Severe medical impairment | |
| 10 Unstable social situation/unlikely to comply |
Surgical procedure
All patients had laparoscopic RYGB surgery at the University Hospital of Uppsala. In this procedure, the majority of the stomach and the duodenum was excluded from the passage of food by connecting the proximal small bowel to a small gastric pouch directly below the esophagus55. No complications occurred. The first day after surgery, only liquids were administered to the patients. Patients were started on their new diet, six small meals daily, on the second postoperative day and returned home. A sick-leave of 2–3 weeks was initiated as well as a follow-up visit at four weeks.
Experimental procedure
Resting state-activity was measured in patients one month before surgery, one month after surgery and one year after surgery; LC were scanned once. Patients were instructed to fast from the 18.00 of the day before the MRI acquisition. Scanning was performed in the morning. Before scanning, participants reported the number of hours they had slept the night before and rated their feelings of hunger, fullness, tiredness and stress on a 100-mm visual analogue scale (VAS). After the scanning, participants consumed a caloric load that consisted of 250 ml of a milk-based, vanilla-flavored drink fortified with protein and carbohydrates (carbohydrates/protein/fat = 64/32/4 energy %) and provided 260 kcal (Gainomax Recovery Vanille; Norrmejerier Umeå, Sweden). Twenty-five minutes after this caloric load, subjective appetite ratings were repeated and a second rsfMRI scanning session was acquired (see Table S6, Supplementary Material, for the timing of the experimental procedures).
MRI acquisition
Structural and functional brain images were acquired with a Philips 3-Tesla (Achieva, Philips Healthcare, Best, Netherlands) using a 32-channel head coil. 180 resting-state volumes were registered during the T2*-weighed echo-planar imaging (EPI) sequence (TR = 2000 ms; TE = 30 ms; flip angle: 90°; slice thickness = 3 mm; slice spacing = 3.9 mm; slices number = 32). Participants were reminded not to focus their thoughts on anything and to lay still with their eyes closed, without falling asleep. Structural images were acquired with a T1-weighted turbo-field-echo (TFE) sequence (TR = 8100 ms; TE = 3.7 ms; flip angle: 8°; slice thickness = 1 mm; slice spacing = 1 mm).
Pre-processing
Structural and functional images were pre-processed using DPARSFA (Data Processing Assistant for Resting-State fMRI Advanced; http://rfmri.org/DPARSF) and CONN toolbox (https://www.nitrc.org/projects/conn/), running in a Matlab environment. Functional images were realigned to correct for head movements using DPARSFA, after having removed the first 10 time-points to allow for signal equilibration. A cut-off of 3 mm of head movements was an exclusion criterion; all participants had moved less than 2 mm. Slice timing was performed. Structural images were coregistered to the functional images and segmented into grey matter (GM), white matter (WM) and cerebrospinal fluid (CSF) probability maps, using DARTEL (Diffeomorphic Anatomical Registration through Exponentiated Lie Algebra; ref. 56) and normalized to the MNI space. Functional images were also normalized to the MNI space using the flow fields derived from the DARTEL procedure, and smoothed with a 4 mm FWHM (Full Width Half Maximum) Gaussian kernel. Preprocessed images were then loaded in CONN. The effects of WM, CSF and motion were regressed out. A band-pass filter of 0.008–0.09 Hz was applied to reduce the interference of physiological noise, such as respiratory and cardiac artifacts, and linear detrending was performed. Motion parameters were entered as confound covariates in the first level analysis.
Statistical analyses
Statistical analyses were performed with CONN. Each LC was assigned three conditions: rest, fasted and sated. Patients were assigned the same conditions for each scanning session they had, prior and after surgery. 132 ROIs provided by the CONN toolbox - which include the FSL Harvard-Oxford atlas for cortical and subcortical areas and AAL atlas for cerebellar areas – were investigated for between-groups differences in connectivity.
We first investigated the effects of group (for LC vs patients assessment) or time (for within-patients assessment), feeding condition and the effect of the interaction between the two on connectivity, by testing whether a given seed showed a between-groups difference in connectivity with any of the targets ROIs. Seeds showing a significant effect of group/surgery, condition or interaction on connectivity were then selected as sources for the post-hoc analysis. In the post-hoc analysis, specific seed-to-target connectivity was investigated, as well as the direction of any effect previously found.
For the preliminary analysis of effect no correction for multiple comparisons was performed, but a stricter threshold of p < 0.01 was applied. Age was entered as covariate of no interest when comparing controls and patients. At the post-hoc analysis, we applied a correction for multiple comparisons with a false discovery rate (FDR) approach at analysis level (i.e. correcting across the multiple comparisons arising from having multiple seed and target ROIs), a more conservative correction than the more commonly used FDR at seed-level (i.e. correcting across multipel target areas, separately for each seed ROI). The FDR-corrected significance threshold was set at p < 0.05.
A correlation analysis was carried out to test whether presurgery connectivity could be predictive of long-term BMI loss, expressed as percentage of BMI reduction after 1-year follow-up. The threshold for significance were the same as detailed above, and age was entered as covariate of no interest.
Electronic supplementary material
Acknowledgements
The studies were supported by the Swedish Research Council, the Swedish Brain Research Foundation and the Swedish Society of Medical Research.
Author Contributions
G.O. and W.Z. contributed equally to this work. G.O. wrote the manuscript and performed with a majority of the analysis; W.Z. wrote the manuscript and assisted with the analysis. M.S. performed human data collection and quality control. C.Z. performed human data collection and quality control and assisted with scanning acquisition. P.H., L.N., L.W. and J.S. assisted with the analysis and in writing the manuscript. E.-M.L. directed imaging procedures and screened the scans for safety. C.B., M.S. and H.B.S. were involved in the initial design of the study and inspiration for the project. H.B.S. oversaw the project and was involved in the planning/organization of the experimental procedures and writing of paper. All authors have added insight to the manuscript and have approved this version.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Gaia Olivo and Wei Zhou contributed equally to this work.
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-06663-5
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Colquitt, J. L., Pickett, K., Loveman, E. & Frampton, G. K. Surgery for weight loss in adults. Cochrane Database Syst Rev CD003641, doi:10.1002/14651858.CD003641.pub4 (2014). [DOI] [PMC free article] [PubMed]
- 2.McGrice M, Don Paul K. Interventions to improve long-term weight loss in patients following bariatric surgery: challenges and solutions. Diabetes Metab Syndr Obes. 2015;8:263–274. doi: 10.2147/DMSO.S57054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Frank S, et al. Neuronal Food Reward Activity in Patients With Type 2 Diabetes With Improved Glycemic Control After Bariatric Surgery. Diabetes Care. 2016;39:1311–1317. doi: 10.2337/dc16-0094. [DOI] [PubMed] [Google Scholar]
- 4.Scholtz S, et al. Obese patients after gastric bypass surgery have lower brain-hedonic responses to food than after gastric banding. Gut. 2014;63:891–902. doi: 10.1136/gutjnl-2013-305008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Frank S, et al. Altered brain activity in severely obese women may recover after Roux-en Y gastric bypass surgery. Int J Obes (Lond) 2014;38:341–348. doi: 10.1038/ijo.2013.60. [DOI] [PubMed] [Google Scholar]
- 6.Bruce AS, et al. A comparison of functional brain changes associated with surgical versus behavioral weight loss. Obesity (Silver Spring) 2014;22:337–343. doi: 10.1002/oby.20630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bruce JM, et al. Changes in brain activation to food pictures after adjustable gastric banding. Surg Obes Relat Dis. 2012;8:602–608. doi: 10.1016/j.soard.2011.07.006. [DOI] [PubMed] [Google Scholar]
- 8.Ochner CN, et al. Selective reduction in neural responses to high calorie foods following gastric bypass surgery. Ann Surg. 2011;253:502–507. doi: 10.1097/SLA.0b013e318203a289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ochner CN, et al. Neural responsivity to food cues in fasted and fed states pre and post gastric bypass surgery. Neurosci Res. 2012;74:138–143. doi: 10.1016/j.neures.2012.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang JL, Yang Q, Hajnal A, Rogers AM. A pilot functional MRI study in Roux-en-Y gastric bypass patients to study alteration in taste functions after surgery. Surg Endosc. 2016;30:892–898. doi: 10.1007/s00464-015-4288-5. [DOI] [PubMed] [Google Scholar]
- 11.Gusnard DA, Raichle ME, Raichle ME. Searching for a baseline: functional imaging and the resting human brain. Nature reviews. Neuroscience. 2001;2:685–694. doi: 10.1038/35094500. [DOI] [PubMed] [Google Scholar]
- 12.van de Sande-Lee S, et al. Partial reversibility of hypothalamic dysfunction and changes in brain activity after body mass reduction in obese subjects. Diabetes. 2011;60:1699–1704. doi: 10.2337/db10-1614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Goldman RL, et al. Executive control circuitry differentiates degree of success in weight loss following gastric-bypass surgery. Obesity (Silver Spring) 2013;21:2189–2196. doi: 10.1002/oby.20575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ness A, et al. Pre-surgical cortical activation to food pictures is associated with weight loss following bariatric surgery. Surg Obes Relat Dis. 2014;10:1188–1195. doi: 10.1016/j.soard.2014.06.005. [DOI] [PubMed] [Google Scholar]
- 15.Wiemerslage, L. et al. A resting-state fMRI study of obese females between pre- and postprandial states before and after bariatric surgery. Eur J Neurosci, doi:10.1111/ejn.13428 (2016). [DOI] [PubMed]
- 16.Leech R, Sharp DJ. The role of the posterior cingulate cortex in cognition and disease. Brain. 2014;137:12–32. doi: 10.1093/brain/awt162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gearhardt AN, Yokum S, Stice E, Harris JL, Brownell KD. Relation of obesity to neural activation in response to food commercials. Soc Cogn Affect Neurosci. 2014;9:932–938. doi: 10.1093/scan/nst059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rapuano KM, Huckins JF, Sargent JD, Heatherton TF, Kelley WM. Individual Differences in Reward and Somatosensory-Motor Brain Regions Correlate with Adiposity in Adolescents. Cereb Cortex. 2016;26:2602–2611. doi: 10.1093/cercor/bhv097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Feldstein Ewing, S. W. et al. Overweight adolescents’ brain response to sweetened beverages mirrors addiction pathways. Brain Imaging Behav, doi:10.1007/s11682-016-9564-z (2016). [DOI] [PMC free article] [PubMed]
- 20.Geliebter A. Neuroimaging of gastric distension and gastric bypass surgery. Appetite. 2013;71:459–465. doi: 10.1016/j.appet.2013.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.LaBar KS, et al. Hunger selectively modulates corticolimbic activation to food stimuli in humans. Behav Neurosci. 2001;115:493–500. doi: 10.1037/0735-7044.115.2.493. [DOI] [PubMed] [Google Scholar]
- 22.Tang DW, Fellows LK, Small DM, Dagher A. Food and drug cues activate similar brain regions: a meta-analysis of functional MRI studies. Physiol Behav. 2012;106:317–324. doi: 10.1016/j.physbeh.2012.03.009. [DOI] [PubMed] [Google Scholar]
- 23.Stawarczyk D, Majerus S, Maquet P, D’Argembeau A. Neural correlates of ongoing conscious experience: both task-unrelatedness and stimulus-independence are related to default network activity. PLoS One. 2011;6:e16997. doi: 10.1371/journal.pone.0016997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lamm C, Decety J, Singer T. Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. Neuroimage. 2011;54:2492–2502. doi: 10.1016/j.neuroimage.2010.10.014. [DOI] [PubMed] [Google Scholar]
- 25.Kullmann S, et al. The obese brain: association of body mass index and insulin sensitivity with resting state network functional connectivity. Hum Brain Mapp. 2012;33:1052–1061. doi: 10.1002/hbm.21268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Murdaugh DL, Cox JE, Cook EW, 3rd, Weller RE. fMRI reactivity to high-calorie food pictures predicts short- and long-term outcome in a weight-loss program. Neuroimage. 2012;59:2709–2721. doi: 10.1016/j.neuroimage.2011.10.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McFadden KL, Cornier MA, Melanson EL, Bechtell JL, Tregellas JR. Effects of exercise on resting-state default mode and salience network activity in overweight/obese adults. Neuroreport. 2013;24:866–871. doi: 10.1097/WNR.0000000000000013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhao, J. et al. Intrinsic brain subsystem associated with dietary restraint, disinhibition and hunger: an fMRI study. Brain Imaging Behav, doi:10.1007/s11682-015-9491-4 (2016). [DOI] [PubMed]
- 29.Corradi-Dell’acqua C, et al. Effects of shifting perspective of the self: an fMRI study. Neuroimage. 2008;40:1902–1911. doi: 10.1016/j.neuroimage.2007.12.062. [DOI] [PubMed] [Google Scholar]
- 30.Kucyi A, Hodaie M, Davis KD. Lateralization in intrinsic functional connectivity of the temporoparietal junction with salience- and attention-related brain networks. J Neurophysiol. 2012;108:3382–3392. doi: 10.1152/jn.00674.2012. [DOI] [PubMed] [Google Scholar]
- 31.Mitchell JP, Heatherton TF, Macrae CN. Distinct neural systems subserve person and object knowledge. Proc Natl Acad Sci USA. 2002;99:15238–15243. doi: 10.1073/pnas.232395699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Longarzo, M. et al. The influence of interoceptive awareness on functional connectivity in patients with irritable bowel syndrome. Brain Imaging Behav, doi:10.1007/s11682-016-9595-5 (2016). [DOI] [PubMed]
- 33.Janowitz D, et al. Association between waist circumference and gray matter volume in 2344 individuals from two adult community-based samples. Neuroimage. 2015;122:149–157. doi: 10.1016/j.neuroimage.2015.07.086. [DOI] [PubMed] [Google Scholar]
- 34.Kuhn AB, et al. FTO gene variant modulates the neural correlates of visual food perception. Neuroimage. 2016;128:21–31. doi: 10.1016/j.neuroimage.2015.12.049. [DOI] [PubMed] [Google Scholar]
- 35.Hinton EC, et al. Neural contributions to the motivational control of appetite in humans. Eur J Neurosci. 2004;20:1411–1418. doi: 10.1111/j.1460-9568.2004.03589.x. [DOI] [PubMed] [Google Scholar]
- 36.Yokum S, Ng J, Stice E. Relation of regional gray and white matter volumes to current BMI and future increases in BMI: a prospective MRI study. Int J Obes (Lond) 2012;36:656–664. doi: 10.1038/ijo.2011.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vytal K, Hamann S. Neuroimaging support for discrete neural correlates of basic emotions: a voxel-based meta-analysis. J Cogn Neurosci. 2010;22:2864–2885. doi: 10.1162/jocn.2009.21366. [DOI] [PubMed] [Google Scholar]
- 38.Bas-Hoogendam JM, et al. Neurobiological candidate endophenotypes of social anxiety disorder. Neurosci Biobehav Rev. 2016;71:362–378. doi: 10.1016/j.neubiorev.2016.08.040. [DOI] [PubMed] [Google Scholar]
- 39.Markowitsch HJ. Differential contribution of right and left amygdala to affective information processing. Behav Neurol. 1998;11:233–244. doi: 10.1155/1999/180434. [DOI] [PubMed] [Google Scholar]
- 40.Baeken C, et al. Left and right amygdala - mediofrontal cortical functional connectivity is differentially modulated by harm avoidance. PLoS One. 2014;9:e95740. doi: 10.1371/journal.pone.0095740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bastin C, Harrison BJ, Davey CG, Moll J, Whittle S. Feelings of shame, embarrassment and guilt and their neural correlates: A systematic review. Neurosci Biobehav Rev. 2016;71:455–471. doi: 10.1016/j.neubiorev.2016.09.019. [DOI] [PubMed] [Google Scholar]
- 42.Veit R, et al. Reduced cortical thickness associated with visceral fat and BMI. Neuroimage Clin. 2014;6:307–311. doi: 10.1016/j.nicl.2014.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tuulari JJ, et al. Neural circuits for cognitive appetite control in healthy and obese individuals: an fMRI study. PLoS One. 2015;10:e0116640. doi: 10.1371/journal.pone.0116640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Walter H, et al. Understanding intentions in social interaction: the role of the anterior paracingulate cortex. J Cogn Neurosci. 2004;16:1854–1863. doi: 10.1162/0898929042947838. [DOI] [PubMed] [Google Scholar]
- 45.Hermann A, Bieber A, Keck T, Vaitl D, Stark R. Brain structural basis of cognitive reappraisal and expressive suppression. Soc Cogn Affect Neurosci. 2014;9:1435–1442. doi: 10.1093/scan/nst130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gudayol-Ferre E, Pero-Cebollero M, Gonzalez-Garrido AA, Guardia-Olmos J. Changes in brain connectivity related to the treatment of depression measured through fMRI: a systematic review. Frontiers in human neuroscience. 2015;9:582. doi: 10.3389/fnhum.2015.00582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wagner G, et al. Differential effects of serotonergic and noradrenergic antidepressants on brain activity during a cognitive control task and neurofunctional prediction of treatment outcome in patients with depression. Journal of psychiatry & neuroscience: JPN. 2010;35:247–257. doi: 10.1503/jpn.090081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Godlewska BR, Norbury R, Selvaraj S, Cowen PJ, Harmer CJ. Short-term SSRI treatment normalises amygdala hyperactivity in depressed patients. Psychological medicine. 2012;42:2609–2617. doi: 10.1017/S0033291712000591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fuchs HF, et al. Benefits of bariatric surgery do not reach obese men. Journal of laparoendoscopic & advanced surgical techniques. Part A. 2015;25:196–201. doi: 10.1089/lap.2014.0639. [DOI] [PubMed] [Google Scholar]
- 50.Sjostrom L, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. The New England journal of medicine. 2007;357:741–752. doi: 10.1056/NEJMoa066254. [DOI] [PubMed] [Google Scholar]
- 51.Gong G, He Y, Evans AC. Brain connectivity: gender makes a difference. The Neuroscientist: a review journal bringing neurobiology, neurology and psychiatry. 2011;17:575–591. doi: 10.1177/1073858410386492. [DOI] [PubMed] [Google Scholar]
- 52.Biswal BB, et al. Toward discovery science of human brain function. Proc Natl Acad Sci USA. 2010;107:4734–4739. doi: 10.1073/pnas.0911855107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Liu H, Stufflebeam SM, Sepulcre J, Hedden T, Buckner RL. Evidence from intrinsic activity that asymmetry of the human brain is controlled by multiple factors. Proc Natl Acad Sci USA. 2009;106:20499–20503. doi: 10.1073/pnas.0908073106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Weissman-Fogel I, Moayedi M, Taylor KS, Pope G, Davis KD. Cognitive and default-mode resting state networks: do male and female brains “rest” differently? Hum Brain Mapp. 2010;31:1713–1726. doi: 10.1002/hbm.20968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Olbers T, Lonroth H, Fagevik-Olsen M, Lundell L. Laparoscopic gastric bypass: development of technique, respiratory function, and long-term outcome. Obes Surg. 2003;13:364–370. doi: 10.1381/096089203765887679. [DOI] [PubMed] [Google Scholar]
- 56.Ashburner J. A fast diffeomorphic image registration algorithm. Neuroimage. 2007;38:95–113. doi: 10.1016/j.neuroimage.2007.07.007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


