Abstract
Lipoxin A4 (LXA4) is involved in the resolution of inflammation and wound healing; however, it is extremely unstable. Thus, to preserve its biological activities and confer stability, we encapsulated LXA4 in poly-lactic-co-glycolic acid (PLGA) microparticles (LXA4-MS) and assessed its application in treating dorsal rat skin lesions. Ulcers were sealed with fibrin adhesive and treated with either LXA4-MS, unloaded microparticles (Un-MS), soluble LXA4, or PBS/glue (vehicle). All groups were compared at 0, 2, 7, and 14 days post-lesions. Our results revealed that LXA4-MS accelerated wound healing from day 7 and reduced initial ulcer diameters by 80%. Soluble LXA4, Un-MS, or PBS closed wounds by 60%, 45%, and 39%, respectively. LXA4-MS reduced IL-1β and TNF-α, but increased TGF-β, collagen deposition, and the number of blood vessels. Compared to other treatments, LXA4-MS reduced inflammatory cell numbers, myeloperoxidase (MPO) concentration, and metalloproteinase-8 (MMP8) mRNA in scar tissue, indicating decreased neutrophil chemotaxis. In addition, LXA4-MS treatment increased macrophages and IL-4, suggesting a positive impact on wound healing. Finally, we demonstrated that WRW4, a selective LXA4 receptor (ALX) antagonist, reversed healing by 50%, indicating that LXA4 must interact with ALX to induce wound healing. Our results show that LXA4-MS could be used as a pharmaceutical formulation for the treatment of skin ulcers.
Introduction
Wound healing is a complex process in which tissues are repaired post injury. Wound healing is divided into inflammatory, proliferative, and remodeling phases [1] that result in scar formation. Scarring involves recruitment of cells, growth factors, cytokines, and eicosanoids along with the release of enzymes to elicit extracellular matrix formation [1,2]. Pathological alterations of each phase are related to wound exacerbation or an inefficient healing process [3]. The aforementioned phases are potentially ideal pharmacological targets that may enhance wound healing [4]. Identifying effective drugs and/or therapies is critically important since complications in wound healing incurs a cost of more than $25 billion annually [5].
Lipid mediator lipoxins (LXs) are eicosanoids derived from arachidonic acid (AA) metabolism. Lipoxygenases 5, 12, and 15-lipoxygenase (LO) generate LXs via interactions with leukocytes and other cells, like platelets and epithelial cells [6,7]. LXs were described in 1984 when 15-hydroxyeicosatetraenoic acid (H(p)ETE), an AA metabolite, was added to human leukocytes. The most abundant compound contained four double conjugated bonds and was subsequently named LXA4 [8]. It was later shown that LXA4 binds to a specific receptor named ALX, which is a G protein-coupled receptor and is expressed in a variety of cells [7].
After receptor binding to cell types such as neutrophils and macrophages, LXA4 induces various responses, including the resolution of inflammation and wound healing [9–13]. In polymorphonuclear cells (PMN), LXA4 reduces pro-inflammatory functions by inhibiting reactive oxygen species (ROS) production and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and activator protein-1 (AP-1) activation as well as impairing PMN transmigration across the vessel [7]. In monocytes, LXA4 enhances macrophage phagocytosis of apoptotic PMN [14], creating a wound healing microenvironment at inflammation sites [7]. Furthermore, LXA4 is endogenously produced in the eye to induce a faster wound healing response in the cornea [15], and reduces the release of inflammatory mediators by human epidermal keratinocytes [16]. Based on the abovementioned reasons, LXA4 is heralded as a critical resolutive and wound healing mediator [9,10,12,13,15]. To this end, we hypothesized that LXA4 also promotes wound healing in the skin. We employed PLGA (poly-lactic-co-glycolic acid) microparticles (MS) as a means to preserve LXA4 biological activities and to promote controlled release, taking into consideration that LXA4 is easily oxidized [17] and PLGA MS have the ability to preserve the biological activity of lipid mediators [18–23]. We demonstrated that LXA4-MS applied to rat skin ulcers were more effective than soluble LXA4 in accelerating the wound healing process. We propose LXA4-MS as an innovative and viable option to treat skin ulcers.
Materials and methods
Microparticle preparation
LXA4-MS and Un-MS were prepared using the single emulsion oil/water (o/w) method [18]. Briefly, 250 μl of LXA4 solution (initial concentration: 100 μg/ml) (Cayman Chemical Company, USA) was added to a solution containing 10 ml of methylene dichloride and 100 mg of PLGA 50:50 (lactic/glycolic acid) (Boehringer Ingelheim, Germany). This organic phase was afterwards mixed into an aqueous phase containing surfactant (poly (vinyl alcohol) PVA 3% w/v (Aldrich Chemicals, USA). After they were stirred for 4 h at 22°C in an RW20 IKA homogenizer (IKA Labortechnik, Germany), the microparticles were washed two times with deionized water, suspended in 1 ml of PBS, and stored at -20°C. After, LXA4-MS were lyophilized for 24 h and then stored at -80°C. Un-MS were prepared with the same protocol without the addition of LXA4.
Microparticle characterization
Particle diameter was characterized using the LS 13 320 Laser Diffraction Particle Size Analyzer (Beckman Counter, USA), and zeta potential was determined using Nano Zeta Sizer (Malvern Instruments, England), employing 5 mg of the MS samples suspended in 1 ml of PBS. MS covered with gold were used to analyze morphology by scanning electron microscopy (SEM) using the Evo 50 (Zeiss, England). To determine encapsulation efficiency, 10 mg of LXA4-MS was dissolved in 1 ml of acetonitrile, vortexed to complete dissolution, evaporated for 2 h using Speed Vacuum (Eppendorf, Germany), and suspended in EIA buffer for LXA4 quantification using enzyme immunoassay EIA Kit EA45 (Oxford Biomedical Research, USA). Encapsulation efficiency was calculated as described [18]. To determine whether MS were contaminated by LPS, a limulus amebocyte lysate test (LAL) (Pierce Biotechnology, USA) was performed, which showed an insignificant amount of LPS in the MS. The release kinetics of LXA4 was monitored in vitro. LXA4 release from MS was assessed using a modified Franz-type diffusion cell (Microette; Hanson Research, USA) [18] and cellulose acetate membrane with a 0.45-μm pore size (Fisher, USA) that was placed between the sample and the receptor chamber. LXA4-MS were suspended in 300 μl of saline buffer (PBS; pH 7.4) and placed on top of the membrane that is the donor compartment. Samples (1 ml) were collected at 0.5, 1, 3, 6, 9, 12, 24, and 48 h from a receiving compartment containing PBS/ethanol (50:50, v/v). The samples were analyzed using a reverse phase C18 Ascentis Express column 10 cm x 2.1 mm, 2.7 μm (Sigma-Aldrich, USA) and binary gradient of 0.002% acetic acid in water/acetonitrile 7:3 solution (A) and acetonitrile/2-propanol solution (B) at a constant flow rate of 0.6 ml/min. Concentration of lipids mediators were calculated (Triple TOF 5600+ LC-MS/MS System; Sciex, USA) from linear calibration curves by MRMHR experiments. The experimental data were evaluated with MultQuant 3.0 (Sciex, USA).
Fibrin sealant (glue) preparation
Production of fibrin sealant was adapted from the Skin Cell Culture Laboratory, Universidade Estadual de Campinas (UNICAMP), Campinas, São Paulo, Brazil, as previously proposed [24–26].
Animals and wound injury
Wistar rats (Rattus norvegicus) (105 adult male, weight: 180–220 g; age: 6–7 weeks old) from the Bioterium of Campus of Ribeirão Preto, Ribeirão Preto, São Paulo, Brazil were used. Animals were singly housed and maintained on a light/dark cycle with free access to food and water. Rats were divided into four groups (n = 15): untreated (Vehicle—PBS/glue); treated with Un-MS; soluble LXA4; LXA4-MS. Rats were anesthetized intraperitoneally (i.p.) with ketamine (80 mg/kg) and xylazine (15 mg/kg), and thereafter shaved and cleaned with 70% ethanol. Two excisions were made on the dorsal cervical region of the rats with a sterile histological (biopsy) punch of 1.5 cm diameter (Stiefel Laboratories, Germany). For the treatment, wounds were treated with either 10 mg of MS (Un-MS or LXA4-MS) or soluble LXA4, and the wounds were sealed with fibrin glue every 3 days, beginning on the day of wound induction (day 0). For the untreated group, PBS and fibrin glue were used. Rats were euthanized in a CO2 chamber and wounds plus surrounding areas were cut with a sterile histological punch. On the day of collection, one ulcer was stored at -80°C for later use in cytokine analyses, MPO measurements, and qRT-PCR. The other ulcer was used for histological analysis (H/E and Picro Sirius Red). In the experiment using WRW4 (specific antagonist of ALX receptor) (WRW4, Formyl Peptide Receptor-Like 1 (FPRL1) antagonist (Aaspec EGT Group, USA), the following groups were included: skin ulcers treated with WRW4 (25 μg of the compound in 25 μl of PBS per wound), skin ulcers treated with WRW4 + LXA4-MS (WRW4 applied 10 minutes before MS application, 10 mg of LXA4-MS), and skin ulcers treated with LXA4-MS (10 mg). The animals did not receive anti-inflammatory drugs commonly used to promote analgesia because these compounds may interfere with wound healing and LXA4 actions. All animal experiments were conducted in accordance with the Ethical Principles in Animal Research adopted by the National Council for the Control of Animal Experimentation (CONCEUA), Brazil (Process n° 016/2014-1). This study was approved by Animal Care and Use Committee of the School of Pharmaceutical Sciences of Ribeirão Preto (CEUA-FCFRP) (Process: n° 016/2014-1).
Wound-healing index
Wound-healing was analyzed using microscopic images of skin ulcers at days 0, 2, 7, and 14 for all groups. Diameters of wound areas were calculated using ImageJ (NIH, USA). The wound healing index was calculated as per a previous study [27]. The wound-healing index ranged from 0 to 1, with 0 being a circular 1.5 cm wound and 1 being a completely closed wound.
Histological analysis
Collected wound tissues were fixed in 4% phosphate-buffered formaldehyde for 24 h and then prepared according to standard protocols for staining with hematoxylin and eosin (H/E) or Picro Sirius Red. The sections were examined in a blinded fashion using a digital camera LEICA DFC 280 (Leica, Germany) attached to a light microscope LEICA DM 4000B (Leica, Germany). Cell counting and angiogenesis were performed using ImageJ with the Cell Counter plug-in (NIH, USA). Collagen deposition was analyzed using the ImageJ software with the Color Deconvolution plug-in.
Cytokines, MPO, and NAG measurements
A total of 100 mg of tissue was homogenized in 1 ml of PBS, centrifuged (3,000 × g/10 min, 4°C), and supernatants were used to quantify IL-6, TGF-β, TNF-α, IL-1β, and VEGF by Enzyme-Linked Immunosorbent Assay (ELISA; R&D Systems, USA), MPO for neutrophil quantification as described before [27,28], and N-acetylglucosaminidase (NAG) for macrophage measurement [29].
RNA Extraction and qRT-PCR
RNA was extracted using 30 mg of tissue and Ambion PureLink RNA Mini Kit according to the manufacturer’s instructions (Life Technologies, USA). Relative quantification was performed using the ΔΔCt method. For qRT-PCR, the ALX primers used were as follows: forward primer, 5′-TGTTGGGCCCTGGATTTTAGC-3′; and reverse primer, 5′-TGTTACCCCAGGATGCGAAGTT-3′ (antisense: nt 532–553) for LXA4 receptor (Rattus norvegicus). There were no repetitive sequences detected. For MMP8 qRT-PCR analysis, we used commercially available TaqMan primers and probes for the TaqMan Gene Expression Assay (Applied Biosystems, USA).
Statistical analysis
Statistical differences between groups were determined using one-way ANOVA followed by Newman-Keuls post-hoc test or Student’s t-test. Statistical significance was determined as p < 0.05. Data in all figures represent means ± SEM, except for the microparticles diameter and residual charges that are presented as mean ± SD.
Results
Characterization of LXA4-MS and Un-MS
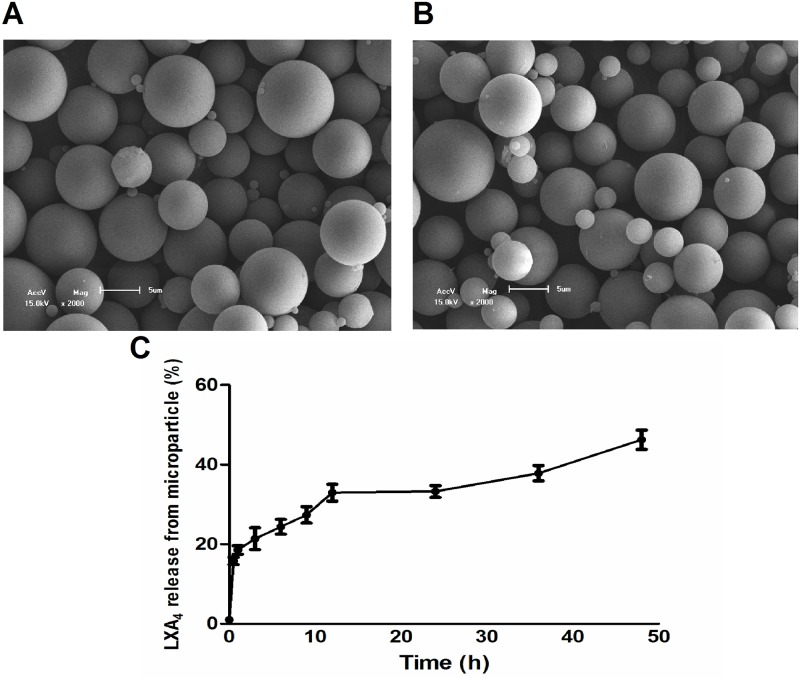
Lyophilized LXA4-MS and Un-MS were dispersed in deionized water, and their size and residual charges were measured. The average diameter was 5.4 μm (± 5.6 μm SD) and 3.9 μm (± 4.4 μm SD) for LXA4-MS and Un-MS, respectively. Analysis of residual charges demonstrated that the molecule, which was encapsulated, did not significantly alter the charges of the polymers. Electrokinetic potential in colloidal dispersions (zeta) was -1.13 mV (± 9.6 mV SD) and -4.56 mV (± 4.8 mV SD) for LXA4-MS and Un-MS, respectively. The high levels of variation in both residual charges and diameter are expected, since during the preparation process, millions of particles are generated with an acceptable range of diameter and electric charges. However, the majority of MS have the appropriated size to penetrate the skin corneum, and negative charge to interact with cell membranes. SEM of Un-MS and LXA4-MS showed the presence of spherical and uniform surfaces (Fig 1A and 1B). Encapsulation efficiency (the amount of LXA4 encapsulated per 10 mg of lyophilized MS) was evaluated using an immunoassay (EIA) kit. We found 800-ng/10 mg or an encapsulation efficiency of 32%. This finding was lower than we observed for leukotriene B4 (LTB4) or prostaglandin E2 (PGE2), which had encapsulation efficiencies of 50% and 75%, respectively [18,19,22]. The in vitro release rates from PLGA MS were evaluated for up to 48 h, and the release profile of LXA4 is shown in Fig 1C. The result demonstrated that LXA4 released from the MS was sustained. Despite the lower encapsulation efficiency, these data showed that PLGA is a suitable strategy for use as a delivery system for lipid mediators.
Fig 1. Scanning electron microscopy (SEM) of microparticles and in vitro release of LXA4 from MS.
Representative images (2,000×) of (A) Unloaded and (B) LXA4-MS morphologies. (C) In vitro cumulative release of LXA4 from LXA4-MS. LXA4 concentration was determined by mass spectrometry over 48 h. Data are representative of two batches.
LXA4-MS reduced neutrophil chemotaxis and accelerated wound closure
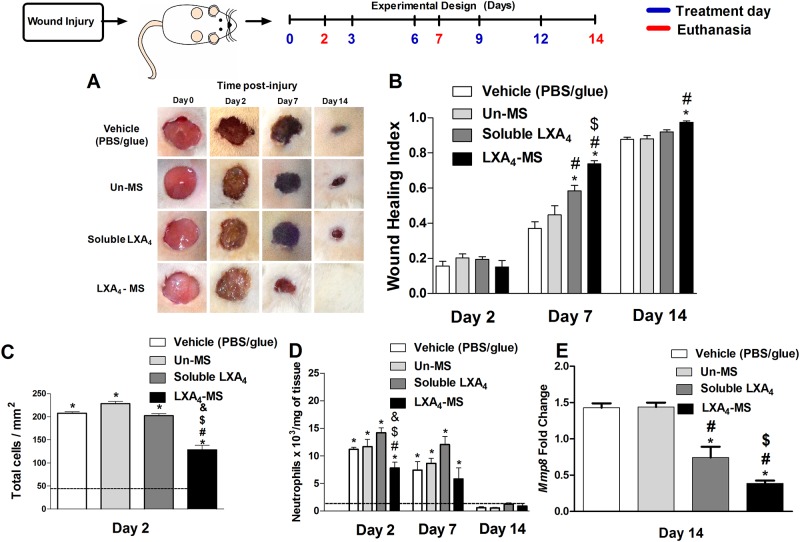
Treatment of skin ulcers with LXA4-MS accelerated wound closure beginning at 7 days post injury (Fig 2A and 2B) as compared to wounds treated with PBS/glue (vehicle), Un-MS, and soluble LXA4. Particularly on day 7, ulcers receiving only fibrin glue and PBS presented only 39% closure of the initial ulcer diameter. Soluble LXA4 and Un-MS improved wound healing by inducing 60% and 45% closure of wounds, respectively. Treatment with LXA4-MS induced closure of 80% of initial ulcers. Interestingly, on day 14, only the induced ulcers treated with LXA4-MS were completely healed. Next, we evaluated leukocyte recruitment to the wound site using two distinct strategies: histological analysis and myeloperoxidase (MPO) measurement. We observed that the number of total cells on wounds treated with LXA4-MS was reduced in comparison to the other groups (Fig 2C and S1A Fig). Assessing tissue MPO and matrix metalloproteinase-8 (MMP8) mRNA abundance (Fig 2D and 2E), we confirmed that neutrophils were lower in LXA4-MS wounds at days 2 and 7 compared to that in the control, Un-MS, and soluble LXA4 groups (although soluble LXA4 also decreased neutrophil recruitment). These data demonstrated that LXA4-MS possessed higher inflammatory resolution activity and was therefore able to expedite wound healing. Moreover, the strategy of encapsulating LXA4 in PLGA efficiently preserved its biological function.
Fig 2. Topical application of LXA4-MS to skin ulcers accelerated wound closure and attenuated neutrophil chemotaxis.
(A) Representative images of 1.5 cm dorsal wounds were collected on days 0, 2, 7, and 14 for the following groups: control (vehicle—PBS/glue), Un-MS, soluble LXA4, and LXA4-MS. (B) Wound healing index values for the groups outlined in (A). Index values range from 0 to 1, where a value of 0 indicates the original wound, and a value of 1 represents a completely closed wound. Values are means ± SEM (n = 10 ulcers/group). One-way ANOVA was done to determine statistical significance (p < 0.05), which is as follows: *, soluble LXA4 or LXA4-MS vs. vehicle (PBS/glue); #, LXA4-MS or soluble LXA4 vs. Un-MS; and $, LXA4-MS vs. soluble LXA4. (C) ImageJ software was used to count inflammatory cells on day 2 in at least 12 random optical 400× fields per group. (D) Neutrophil accumulation (represented as bars) as measured by MPO. Values are means ± SEM (n = 5 wounds/group). One-way ANOVA was done to determine statistical significance (p < 0.05), which is indicated as follows: *, demonstrated significant increase compared to normal tissue (dashed line); #, soluble LXA4 or LXA4-MS vs. Vehicle (PBS/glue); $, LXA4-MS or soluble LXA4 vs. Un-MS; and &, LXA4-MS vs. soluble LXA4. (E) qRT-PCR was performed to assess MMP8 mRNA transcript abundance in skin ulcers collected on days 2, 7, and 14 from the vehicle (PBS/glue), Un-MS, soluble LXA4, and LXA4-MS groups. Data represent means ± SEM (n = 5 ulcers/group). One-way ANOVA was done to determine statistical significance (p < 0.05) and indicated as follows: *, soluble LXA4 or LXA4-MS vs. Vehicle (PBS/glue); #, LXA4-MS or soluble LXA4 vs. Un-MS; and $, LXA4-MS vs. soluble LXA4.
LXA4-MS affected pro and anti-inflammatory cytokine production
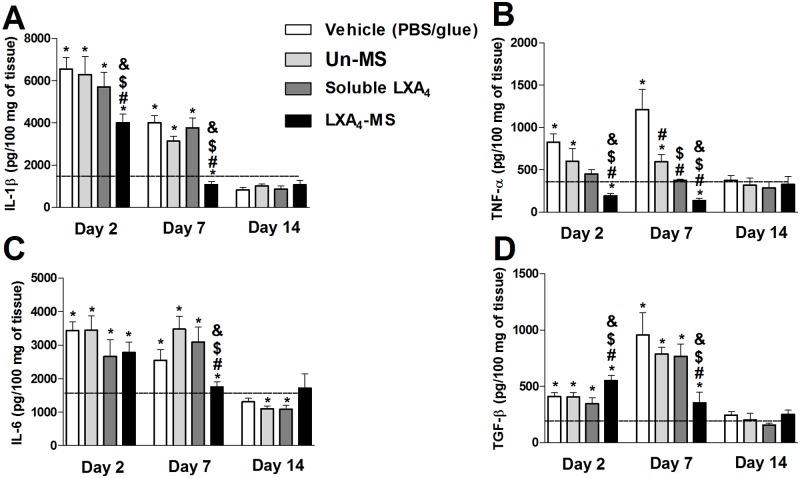
We investigated the effects of all treatments on cytokine production in skin ulcers (Fig 3). We observed that LXA4-MS treatment reduced the production of IL-1β, TNF-α, and IL-6 at days 2 and 7, although with variable intensity. No differences were observed after 14 days post skin injury (Fig 3A, 3B and 3C). Interestingly, LXA4-MS treatment first induced an increase in TGF-β production at day 2 and a decrease in TGF-β production at day 7 (Fig 3D). In ulcers from the other groups, we observed little or no effect on the production of cytokines. At day 14, cytokine concentrations resembled levels found in non-injured skin (day 0) and were thus used to obtain a basal level of cytokines, serving as internal controls.
Fig 3. LXA4-MS modulated cytokines generation in the skin.
Skin ulcer tissues collected on days 0, 2, 7, and 14 from the control (vehicle—PBS/glue), Un-MS, soluble LXA4, and LXA4-MS groups were homogenized to assess (A) IL-1β, (B) TNF-α, (C) IL-6, and (D) TGF-β production by ELISAs. Data (represented as bars) represent means ± SEM (n = 5 ulcers/group). One-way ANOVA was done to determine statistical significance (p < 0.05) and indicated as follows: *, demonstrated significant differences compared to normal tissues (dashed line); #, soluble LXA4 or LXA4-MS vs. Vehicle (PBS/glue); $, LXA4-MS or soluble LXA4 vs. Un-MS; and &, LXA4-MS vs. soluble LXA4.
LXA4-MS promoted and increased deposition of collagen, angiogenesis, and vascular endothelial growth factor (VEGF) production
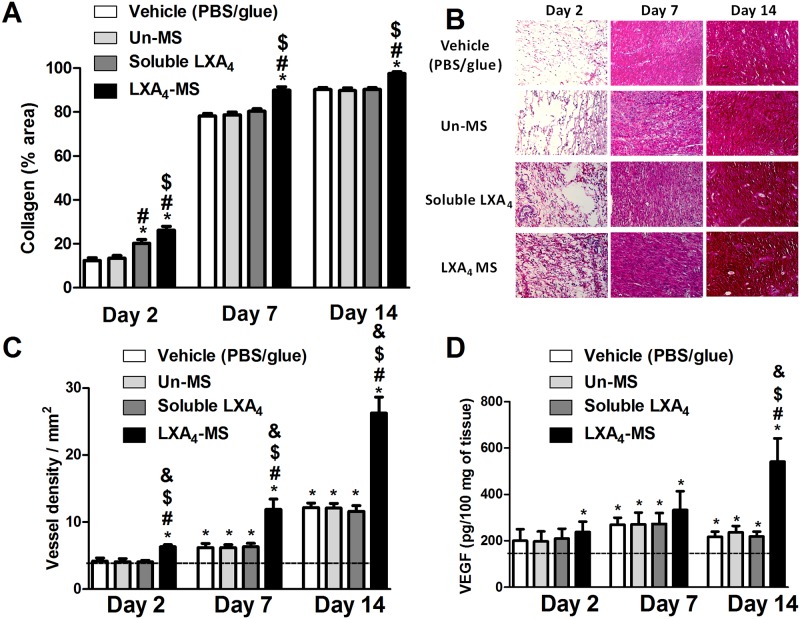
Since we observed that LXA4-MS increased wound healing, we investigated its impact on the elements important for skin restoration, such as collagen deposition and angiogenesis. We observed that LXA4-MS enhanced collagen deposition beginning at day 2 and peaking at day 14 (Fig 4A). The enhancement of collagen deposition was confirmed by histological analysis. LXA4-MS induced massive deposition of collagen on skin ulcers; although with less intensity, soluble LXA4 also induced collagen deposition (Fig 4B). The number of blood vessels in histological sections stained by hematoxylin/eosin (H/E) and VEGF quantification in skin ulcers were additionally evaluated. As expected, LXA4-MS stimulated massive neovascularization beginning on day 2 (Fig 4C and S1B Fig) and peaking at 14 days. However, an increase in VEGF was observed only after 14 days, the last day of observation (Fig 4D). Soluble LXA4 and Un-MS did not induce blood vessel formation nor increase in VEGF levels.
Fig 4. LXA4-MS increased collagen deposition and angiogenesis and affected VEGF production.
(A) Collagen deposition was measured using the ImageJ software with the Color Deconvolution plug-in, which measured densitometry in at least 12 random 400× fields in all groups, at days 2, 7 and 14. Percentages of collagen deposition are represented as means ± SEM (n = 6 ulcers/group). One-way ANOVA was used to determine statistical significance (p < 0.05) and is indicated as follows: *, soluble LXA4 or LXA4-MS vs. Vehicle (PBS/glue); #, LXA4-MS or soluble LXA4 vs. Un-MS; and $, LXA4-MS vs. soluble LXA4. (B) Photomicrographs of wounds stained with Picro Sirius Red (200×) show collagen deposition at days 2, 7, and 14. (C) Histogram showing quantitative analysis of vascular density using the ImageJ software with the Cell Counter plug-in on 200× images. One-way ANOVA was performed to determine statistical significance (p < 0.05), which is indicated as follows: *, significant VEGF increase as compared to normal tissue (dashed line); #, soluble LXA4 or LXA4-MS vs. Vehicle (PBS/glue); $, LXA4-MS or soluble LXA4 vs. Un-MS; and &, LXA4-MS vs. soluble LXA4. (D) VEGF was quantified in all groups at days 2, 7 and 14 (represented as bars) in wounds via ELISAs as proxy to blood vessel density. Values are means ± SEM (n = 6 wounds/group). One-way ANOVA was performed to determine statistical significance (p< 0.05), which is indicated as follows: *, significant VEGF increase as compared to normal tissue (dashed line); #, soluble LXA4 or LXA4-MS vs. Vehicle (PBS/glue); $, LXA4-MS or soluble LXA4 vs. Un-MS; and &, LXA4-MS vs. soluble LXA4.
LXA4-MS induced increase of macrophages and IL-4 in the skin
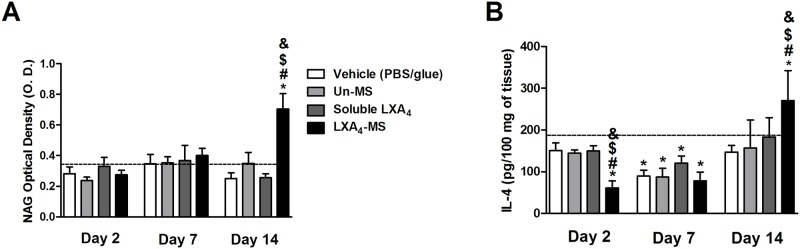
Type II macrophages are known to release IL-4 [30], which regulates scar formation. Thus, we estimated macrophage infiltration by NAG quantification, a marker of these leukocytes [31]. We observed that LXA4-MS treated lesions exhibited a significant increase on macrophage infiltration at day 14 (Fig 5A). We also observed enhanced IL-4 production, which was probably released by infiltrating macrophages (Fig 5B), as described before [30]. Un-MS and soluble LXA4 did not increase NAG and IL-4 production.
Fig 5. LXA4-MS increased macrophages and IL-4 production.
(A) NAG (OD values) was quantified in skin ulcers collected on days 2, 7, and 14 from the vehicle (PBS/glue), Un-MS, soluble LXA4, and LXA4-MS groups (n = 5 ulcers/ group). (B) ELISAs were used to measure IL-4 production in skin ulcers collected on day 14 from the vehicle (PBS/glue), Un-MS, soluble LXA4, and LXA4-MS groups. Basal levels were also assessed. Data represent means ± SEM (n = 5 ulcers/ group). One-way ANOVA was done to determine statistical significance (p < 0.05) and indicated as follows: *, demonstrated significant differences compared to normal tissues (dashed line); #, LXA4-MS vs. Vehicle (PBS/glue); $, LXA4-MS vs. Un-MS; and &, LXA4-MS vs. soluble LXA4.
LXA4-MS increased ALX receptor expression and treatment with a specific ALX receptor antagonist reversed LXA4-MS wound healing capabilities
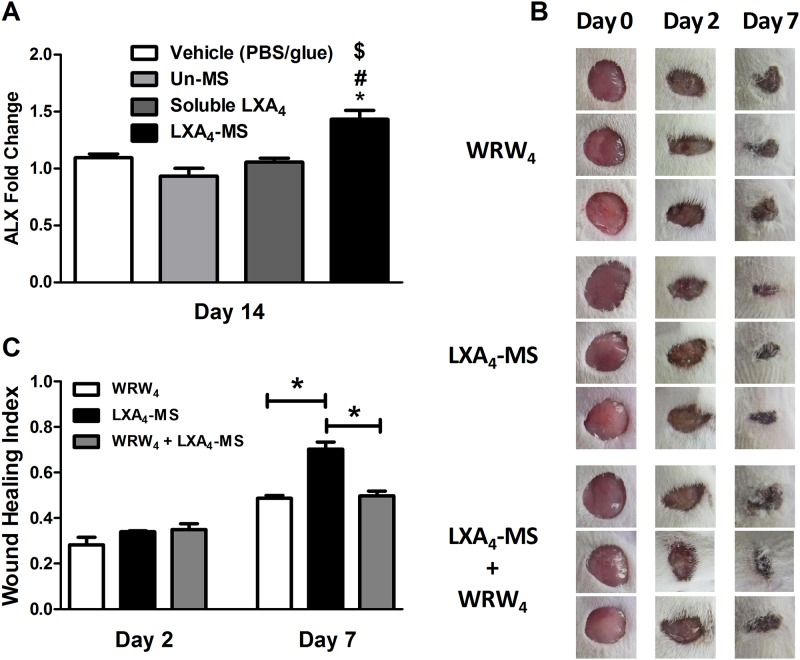
To investigate whether LXA4 released from the microparticles induced wound healing via its own receptor ALX, we assessed ALX expression via qRT-PCR on day 14; the healed skin appeared to be similar to the intact skin. Our results demonstrated that at 14 days post lesion, remarkable up regulation occurred in the mRNA ALX receptor (Fig 6A). As seen in Fig 2A, 7 days after injury, LXA4-MS promoted 80% wound closure compared to the initial wound. Therefore, day 7 was chosen for the analysis of the role of LXA4 receptor. We treated skin lesions with LXA4-MS in the presence or absence of WRW4, a specific LXA4 receptor antagonist. WRW4 treatment reversed LXA4-MS ability to efficiently heal skin ulcers as assessed microscopically and via the wound healing index values (Fig 6B and 6C). Taken together, these results demonstrated that LXA4 released from the microparticles improved wound healing expressly through its ALX receptor.
Fig 6. WRW4, a selective LXA4 receptor antagonist, reversed wound healing properties of LXA4-MS.
(A) qRT-PCR analysis was performed to assess the LXA4 receptor ALX mRNA’s abundance in skin ulcers collected on days 2, 7, and 14 in the control (vehicle—PBS/glue), Un-MS, soluble LXA4, and LXA4-MS groups. Data represent means ± SEM (n = 5 ulcers/group). One-way ANOVA was done to determine statistical significance (p < 0.05), which is indicated as follows: *, LXA4-MS vs. Vehicle (PBS/glue); #, LXA4-MS vs. Un-MS; and $, LXA4-MS vs. soluble LXA4. (B) Representative images of 1.5 cm dorsal wounds at days 2 and 7, with day 0 images serving as pre-injury images, are presented for the following groups: WRW4 (25 μl per animal—from a main peptide solution of 1 mg/ml), WRW4 + LXA4-MS (WRW4 applied 10 minutes before MS application– 10 mg of LXA4-MS), and LXA4-MS (10 mg). (C) Wound healing index values for the groups outlined in (B). Index values range from 0 to 1, where a value of 0 indicates the original wound, and a value of 1 represents a completely closed wound. Data represent means ± SEM (n = 9 ulcers/group); One-way ANOVA was done to determine statistical significance (*p < 0.05).
Discussion
After injury, wound healing elicits a coordinated cascade of intracellular and intercellular events to restore homeostasis and tissue integrity [1]. It is a dynamic process divided into distinct phases [1]. Crucial to healing and cell recruitment are cytokines and lipid mediators production, collagen deposition, and blood vessel formation [1,2]. Among the soluble mediators involved in the healing process, LXA4 is a key factor, especially for cornea healing [15]. This mediator is derived from AA metabolism [8,32] and a potent inhibitor of neutrophil recruitment to the site of inflammation and regulator of leukocyte chemotaxis, and activation [9–13]. Macrophages are then activated to transmigrate and to promote phagocytosis of apoptotic PMN, creating an environment conducive for wound-healing [15]. However, the role of LXA4 in skin regeneration after injury remains unknown.
LXA4 is an unstable mediator [17], a feature that impairs its use for treating inflammatory diseases or tissue lesions. We took advantage of our expertise in lipid mediator encapsulation in PLGA microparticles in order to preserve its wound healing activities and provide controlled release [18,19,22]. Using the single emulsion oil/water (o/w) method [18], we prepared and characterized the Un-MS and LXA4-MS to ensure appropriate size 5.4 μm (± 5.6 μm SD) and 3.9 μm (± 4.4 μm SD) for LXA4-MS and Un-MS] respectively, and charge (-1.13 mV and -4.56 mV for LXA4-MS and Un-MS respectively) needed to interact with cells (Fig 1). According to Jalon’s study, microparticles with a size of around 5 μm are appropriate to penetrate into the stratum corneum, reaching the epidermis and promoting sustained drug release into the skin [33]. The amount of PLGA microparticles loaded with LXA4 (10 mg of MS containing 800 ng of LXA4) used to treat the wound in our study was selected based on literature data, demonstrating that 1 μg of soluble LXA4 accelerated epithelial wound healing in the cornea [15]. To pursue this aim, we used a well-characterized model to study skin healing in rats [34,35]. After inducing skin lesions, ulcers were treated with each PBS, Un-MS, soluble LXA4, or LXA4-MS and then covered with fibrin glue. The healing process was assessed 2, 7, and 14 days later. We showed that using PLGA microparticles to encapsulate LXA4 preserved its biological activities, as evidenced by our calculated wounding healing index values (Fig 2A and 2B). Remarkably, lesions treated with LXA4-MS were completely closed after 14 days. Soluble LXA4 has been shown to reduce neutrophil infiltration in the cornea [15], and our data confirmed that LXA4-MS also reduced neutrophil infiltration in the wound site at 2 and 7 days as per the decrease in MPO (Fig 2D) and MMP8 mRNA (Fig 2E), which are both markers of neutrophil infiltration [36,37]. Decrease in neutrophil infiltration has been described as beneficial for wound healing. A recent study demonstrated that the ‘neutrophil extracellular trap’ (NET) facilitated matrix collagen degradation, impairing wound-healing [38]. Our data suggested that LXA4-MS attenuated neutrophil chemotaxis (Fig 4A and 4B); this effect allows collagen deposition, as previously demonstrated by others [38,39]. As expected, among the other groups, only soluble LXA4 increased the wound-healing index at 7 days and reduced MMP8 mRNA at 14 days (Fig 2B and 2E). However, the soluble mediator minimally affected lesion closure, probably due to its intrinsic instability and thus loss in its biological properties. It’s important to note that MMP8 was not measured days 2 and 7 owing to low tissue recovery and intense inflammatory cell infiltration, which may lead to false-positive results.
LXA4 mediates the delicate balance among inflammatory, anti-inflammatory and regulatory cytokines, which modulate tissue regeneration [15]. Particularly, LXA4 inhibits NF-κB and AP-1 and consequently decreases pro-inflammatory cytokines production [7]. Simultaneously, LXA4 activates the transcriptional factor PPAR-γ, generating a resolutive profile [40]. Our results demonstrated that the acceleration of wound closure and the reduction in neutrophil recruitment induced by LXA4-MS treatment correlated with altered cytokine profiles. We also observed that LXA4-MS application reduced the concentration of the inflammatory cytokines IL-1β, TNF-α, and IL-6. Previous works have shown that such reduction is accompanied by increased TGF-β, an anti-inflammatory cytokine, and collagen deposition [41]. LXA4-MS increased collagen deposition and blood vessel density in coordination with VEGF release (Fig 4C and 4D; S1B Fig). Moreover, at the site of the lesion, LXA4-MS increased macrophages and IL-4. These findings suggested that the accumulation of type II macrophages, sources of IL-4 a regulatory cytokine, is mediates LXA4 resolution properties [30]. Furthermore, wound-healing may function to mediate recruitment of macrophages to infection sites in order to promote phagocytosis of apoptotic PMN and clearance and remodeling of the tissue [14].
Finally, using a selective antagonist of LXA4 receptor, WRW4 [42], we demonstrated that the beneficial effects of LXA4-MS treatment were indeed mediated by the interaction of LXA4 specifically to its receptor ALX (Fig 6) and not due to unspecific effects of the PLGA microparticles. The participation of ALX receptor in an in vitro model of regeneration was described recently in a study using soluble LXA4 [43]. We observed that 14 days after injury (the skin regeneration period), an increase in ALX mRNA occurred. This finding suggested that, at least within this timeframe, endogenous LXA4 is not produced in the necessary quantity to close the lesion. However, we could not rule out the possibility of LXA4 being released beyond 14 days, the same period of spontaneous lesion closure. Also, the relevance of LXA4 to eye tissue regeneration has been demonstrated [15]. Our results suggested that LXA4 released from LXA4-MS accelerated skin wound-healing via its ALX receptors, findings that corroborated a previous study [7].
Conclusions
In summary, we show the retention of LXA4 as a resolution, wound healer, and reparative mediator via the use of PLGA microparticles. Our study demonstrated LXA4-MS as an effective strategy for the treatment of skin ulcers, and it may be a viable treatment option for healing and repair.
Supporting information
(A) Animals were topically treated with PBS/glue, Un-MS, soluble LXA4 and LXA4-MS. At day 2, animals were euthanized, wounds were removed and paraffin-wound sections were stained with HE to evaluate inflammatory infiltrate by image analysis. The sections were photographed at 400×. The cell counting was performed using the software ImageJ, plug-in Cell Counting in at least 12 random optic fields per group. (B) Animals were topically treated with vehicle (PBS/glue) or LXA4-MS. Paraffin wound sections were stained with HE and photographed at 400×. The blood vessels were counted using the software ImageJ, plug-in Cell Counting in at least 12 random optic fields per group.
(TIF)
Acknowledgments
This work was supported by grants from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP; grant #2014/07125-6) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). We would like to thank Dr. Celio Lopes Silva for providing PLGA and Ana Paula Masson, Izaira Tincani Brandão, Wendy Martin Rios and Guilherme Gelfuso for technical support.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This work was supported by grants from Sao Paulo Research Foundation (FAPESP; grant #2014/07125-6) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Singer A, Clark R. Cutaneous Wound Healing. N Engl J Med. 1999;341: 738–746. doi: 10.1056/NEJM199909023411006 [DOI] [PubMed] [Google Scholar]
- 2.Sivamani RK. Eicosanoids and Keratinocytes in Wound Healing. Adv Wound Care. 2014;3: 476–481. doi: 10.1089/wound.2014.0523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bosanquet DC, Harding KG. Wound duration and healing rates: cause or effect? Wound Repair Regen. 2014;22: 143–150. doi: 10.1111/wrr.12149 [DOI] [PubMed] [Google Scholar]
- 4.Wilgus T a., Vodovotz Y, Vittadini E, Clubbs E a., Oberyszyn TM. Reduction of scar formation in full-thickness wounds with topical celecoxib treatment. Wound Repair Regen. 2003;11: 25–34. doi: 10.1046/j.1524-475X.2003.11106.x [DOI] [PubMed] [Google Scholar]
- 5.Sen CK, Gordillo GM, Roy S, Kirsner R, Lambert L, Hunt TK, et al. Health and the Economy. Wound Repair Regen 2009. 2009;17: 763–771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Serhan CN, Sheppard KA. Lipoxin formation during human neutrophil-platelet interactions: Evidence for the transformation of leukotriene A4 by platelet 12-lipoxygenase in vitro. J Clin Invest. 1990;85: 772–780. doi: 10.1172/JCI114503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chiang N, Serhan CN, Dahlén S-E, Drazen JM, Hay DWP, Rovati GE, et al. The lipoxin receptor ALX: potent ligand-specific and stereoselective actions in vivo. Pharmacol Rev. 2006;58: 463–487. doi: 10.1124/pr.58.3.4 [DOI] [PubMed] [Google Scholar]
- 8.Serhan C, Hamberg M, Samuelsson B. Trihydroxytetraenes: a novel series of compounds formed from arachidonic acid in human leukocytes. Biochem Biophys Res Commun. 1984;118: 943–949. [DOI] [PubMed] [Google Scholar]
- 9.Serhan CN, Chiang N, Dalli J. The resolution code of acute inflammation: Novel pro-resolving lipid mediators in resolution. Semin Immunol. 2015;27: 200–215. doi: 10.1016/j.smim.2015.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Serhan CN, Chiang N, Dalli J, Levy BD. Lipid Mediators in the Resolution of Inflammation. Cold Spring Harb Perspect Biol. 2015;7: a016311 doi: 10.1101/cshperspect.a016311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chiang N, Serhan CN. Structural elucidation and physiologic functions of specialized pro-resolving mediators and their receptors. Mol Aspects Med. 2017; doi: 10.1016/j.mam.2017.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Serhan CN. Treating inflammation and infection in the 21st century: new hints from decoding resolution mediators and mechanisms. FASEB J. 2017;31: 1273–1288. doi: 10.1096/fj.201601222R [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Serhan CN. Discovery of specialized pro-resolving mediators marks the dawn of resolution physiology and pharmacology. Mol Aspects Med. 2017; doi: 10.1016/j.mam.2017.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Godson C, Mitchell S, Harvey K, Petasis NA, Hogg N, Brady HR. Cutting edge: lipoxins rapidly stimulate nonphlogistic phagocytosis of apoptotic neutrophils by monocyte-derived macrophages. J Immunol. 2000;164: 1663–7. [DOI] [PubMed] [Google Scholar]
- 15.Gronert K. Lipoxins in the eye and their role in wound healing. Prostaglandins Leukot Essent Fat Acids. 2005;73: 221–229. doi: 10.1016/j.plefa.2005.05.009 [DOI] [PubMed] [Google Scholar]
- 16.Hu F, Liu X-X, Wang X, Alashkar M, Zhang S, Xu J-T, et al. Lipoxin A4 inhibits proliferation and inflammatory cytokine/chemokine production of human epidermal keratinocytes associated with the ERK1/2 and NF-κB pathways. J Dermatol Sci. Japanese Society for Investigative Dermatology; 2015;78: 181–188. doi: 10.1016/j.jdermsci.2015.03.009 [DOI] [PubMed] [Google Scholar]
- 17.Clish CB, Levy BD, Chiang N, Tai HH, Serhan CN. Oxidoreductases in lipoxin A4 metabolic inactivation: A novel role for 15-oxoprostaglandin 13-reductase/leukotriene B4 12-hydroxydehydrogenase in inflammation. J Biol Chem. 2000;275: 25372–25380. doi: 10.1074/jbc.M002863200 [DOI] [PubMed] [Google Scholar]
- 18.Nicolete R, Lima K de M, Júnior JMR, Baruffi MD, de Medeiros AI, Bentley MVLB, et al. In vitro and in vivo activities of leukotriene B4-loaded biodegradable microspheres. Prostaglandins Other Lipid Mediat. 2007;83: 121–9. doi: 10.1016/j.prostaglandins.2006.10.007 [DOI] [PubMed] [Google Scholar]
- 19.Nicolete R, Lima K de M, Júnior JMR, Jose PJ, Sanz M-J, Faccioli LH. Prostaglandin E(2)-loaded microspheres as strategy to inhibit phagocytosis and modulate inflammatory mediators release. Eur J Pharm Biopharm Off J Arbeitsgemeinschaft für Pharm Verfahrenstechnik eV. 2008;70: 784–90. doi: 10.1016/j.ejpb.2008.06.019 [DOI] [PubMed] [Google Scholar]
- 20.dos Santos DF, Nicolete R, de Souza PRM, Bitencourt C da S, dos Santos RR, Bonato VLD, et al. Characterization and in vitro activities of cell-free antigens from Histoplasma capsulatum-loaded biodegradable microspheres. Eur J Pharm Sci. 2009;38: 548–55. doi: 10.1016/j.ejps.2009.10.003 [DOI] [PubMed] [Google Scholar]
- 21.Nicolete R, Secatto A, Pereira PAT, Soares EG, Faccioli LH. Leukotriene B4-loaded microspheres as a new approach to enhance antimicrobial responses in Histoplasma capsulatum-infected mice. Int J Antimicrob Agents. 2009;34: 365–9. doi: 10.1016/j.ijantimicag.2009.06.009 [DOI] [PubMed] [Google Scholar]
- 22.Dos Santos DF, Bitencourt CS, Gelfuso GM, Pereira PAT, de Souza PRM, Sorgi CA, et al. Biodegradable microspheres containing leukotriene B(4) and cell-free antigens from Histoplasma capsulatum activate murine bone marrow-derived macrophages. Eur J Pharm Sci. 2011;44: 580–8. doi: 10.1016/j.ejps.2011.10.005 [DOI] [PubMed] [Google Scholar]
- 23.Nicolete R, dos Santos DF, Faccioli LH. The uptake of PLGA micro or nanoparticles by macrophages provokes distinct in vitro inflammatory response. Int Immunopharmacol. 2011;11: 1557–63. doi: 10.1016/j.intimp.2011.05.014 [DOI] [PubMed] [Google Scholar]
- 24.Ronfard V, Broly H, Mitchell V, Galizia JP, Hochart D, Chambon E, et al. Use of human keratinocytes cultured on fibrin glue in the treatment of burn wounds. Burns. 1991;17: 181–4. [DOI] [PubMed] [Google Scholar]
- 25.Horch RE, Bannasch H, Kopp J, Andree C, Stark GB. Single-cell suspensions of cultured human keratinocytes in fibrin-glue reconstitute the epidermis. Cell Transplant. 1998;7: 309–17. [DOI] [PubMed] [Google Scholar]
- 26.Horch RE, Bannasch H, Stark GB. Transplantation of cultured autologous keratinocytes in fibrin sealant biomatrix to resurface chronic wounds. Transplant Proc. 2001;33: 642–4. [DOI] [PubMed] [Google Scholar]
- 27.Fronza M, Caetano GF, Leite MN, Bitencourt CS, Paula-Silva FWG, Andrade T a. M, et al. Hyaluronidase Modulates Inflammatory Response and Accelerates the Cutaneous Wound Healing. PLoS One. 2014;9: e112297 doi: 10.1371/journal.pone.0112297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Andrade TAM, Iyer A, Das PK, Foss NT, Garcia SB, Coutinho-Netto J, et al. The inflammatory stimulus of a natural latex biomembrane improves healing in mice. Braz J Med Biol Res. 2011;44: 1036–47. [DOI] [PubMed] [Google Scholar]
- 29.Shibata H, Yagi T. Rate assay of with 4-nitrophenyl as an artificial substrate. Clin Chim Acta. 1996;251: 53–64. doi: 10.1016/0009-8981(96)06292-4 [DOI] [PubMed] [Google Scholar]
- 30.La Flamme AC, Kharkrang M, Stone S, Mirmoeini S, Chuluundorj D, Kyle R. Type II-activated murine macrophages produce IL-4. PLoS One. 2012;7: e46989 doi: 10.1371/journal.pone.0046989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dean RT, Hylton W, Allison AC. Lysosomal enzyme secretion by macrophages during intracellular storage of particles. BBA—Gen Subj. 1979;584: 57–65. doi: 10.1016/0304-4165(79)90235-6 [DOI] [PubMed] [Google Scholar]
- 32.Serhan CN, Hamberg M, Samuelsson B. Lipoxins: novel series of biologically active compounds formed from arachidonic acid in human leukocytes. Proc Natl Acad Sci U S A. 1984;81: 5335–5339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.de Jalón EG, Blanco-Príeto MJ, Ygartua P, Santoyo S. PLGA microparticles: possible vehicles for topical drug delivery. Int J Pharm. 2001;226: 181–4. [DOI] [PubMed] [Google Scholar]
- 34.Caetano GF, Fronza M, Leite MN, Gomes A, Frade MAC. Comparison of collagen content in skin wounds evaluated by biochemical assay and by computer-aided histomorphometric analysis. Pharm Biol. 2016;209: 1–5. doi: 10.3109/13880209.2016.1170861 [DOI] [PubMed] [Google Scholar]
- 35.Caetano GF, Frade MAC, Andrade TAM, Leite MN, Bueno CZ, Moraes ÂM, et al. Chitosan-alginate membranes accelerate wound healing. J Biomed Mater Res B Appl Biomater. 2015;103: 1013–22. doi: 10.1002/jbm.b.33277 [DOI] [PubMed] [Google Scholar]
- 36.Bradley PP, Priebat D a, Christensen RD, Rothstein G. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J Invest Dermatol. Elsevier Masson SAS; 1982;78: 206–209. doi: 10.1111/1523-1747.ep12506462 [DOI] [PubMed] [Google Scholar]
- 37.Hasty A, Goldberg GI, Thompson P, Spinella G, Stevens M, Mainardi L, et al. Human Neutrophil Collagenase. J Biol Chem. 1990;265: 11421–11424. [PubMed] [Google Scholar]
- 38.Wong SL, Demers M, Martinod K, Gallant M, Wang Y, Goldfine AB, et al. Diabetes primes neutrophils to undergo NETosis, which impairs wound healing. Nat Med. 2015;21 doi: 10.1038/nm.3887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dovi J V, He L, Dipietro L a. Accelerated wound closure in neutrophil-depleted mice Abstract: The infiltration of neutrophils into in- jured tissue is known to protect wounds from in- repair process. To investigate the role of neutro-. J Leukoc Biol. 2003;73: 448–455. doi: 10.1189/jlb.0802406 [DOI] [PubMed] [Google Scholar]
- 40.Weinberger B, Quizon C, Vetrano AM, Archer F, Laskin JD, Laskin DL. Mechanisms mediating reduced responsiveness of neonatal neutrophils to lipoxin A4. Pediatr Res. 2008;64: 393–398. doi: 10.1203/PDR.0b013e318180e4af [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ignotz R a, Massagué J. Transforming growth factor-beta stimulates the expression of fibronectin and collagen and their incorporation into the extracellular matrix. J Biol Chem. 1986;261: 4337–45. [PubMed] [Google Scholar]
- 42.Karlsson J, Fu H, Boulay F, Bylund J, Dahlgren C. The peptide Trp-Lys-Tyr-Met-Val-D-Met activates neutrophils through the formyl peptide receptor only when signaling through the formylpeptde receptor like 1 is blocked. A receptor switch with implications for signal transduction studies with inhibitors an. Biochem Pharmacol. 2006;71: 1488–1496. doi: 10.1016/j.bcp.2006.02.010 [DOI] [PubMed] [Google Scholar]
- 43.Herrera BS, Kantarci A, Zarrough A, Hasturk H, Leung KP, Van Dyke TE. LXA4 actions direct fibroblast function and wound closure. Biochem Biophys Res Commun. Elsevier Ltd; 2015;464: 1072–1077. doi: 10.1016/j.bbrc.2015.07.076 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A) Animals were topically treated with PBS/glue, Un-MS, soluble LXA4 and LXA4-MS. At day 2, animals were euthanized, wounds were removed and paraffin-wound sections were stained with HE to evaluate inflammatory infiltrate by image analysis. The sections were photographed at 400×. The cell counting was performed using the software ImageJ, plug-in Cell Counting in at least 12 random optic fields per group. (B) Animals were topically treated with vehicle (PBS/glue) or LXA4-MS. Paraffin wound sections were stained with HE and photographed at 400×. The blood vessels were counted using the software ImageJ, plug-in Cell Counting in at least 12 random optic fields per group.
(TIF)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.