Abstract
Central nervous system immune reconstitution inflammatory syndrome (CNS-IRIS), which occurs most often in HIV-infected patients, is an exacerbation of inflammatory reactions related to opportunistic infections as well as primary CNS malignancies both of which mostly occur in HIV-infected patients. However, differential diagnoses are challenging both clinically and radiologically. We describe a patient with CNS-IRIS due to toxoplasmosis whose 11C-methionine uptake suggested lymphoma but whose arterial spin-labelling MRI led to the correct diagnosis.
Keywords: Radiology, Neuroimaging
Background
Patients with HIV after initiation of antiretroviral therapy (ART) can present complications related to ART-induced immune reconstitution inflammatory syndrome (IRIS). The incidence of IRIS in treated patients with HIV as been reported to be as high as 38%.1 Central nervous system (CNS)-IRIS can be induced by opportunistic infections such as Cryptococcus, John Cunningham virus, Cytomegalovirus, tuberculosis or cerebral toxoplasmosis1 and by malignancies such as primary CNS lymphoma.2 The most common causes of focal brain lesions in HIV-infected patients are toxoplasmosis and lymphoma.2 3 Moreover, differentiation of the two is often difficult clinically and radiologically.2 3
11C-methionine (MET)-PET is reported to be useful for differential diagnoses of intra-axial malignant tumours from other lesions such as low-grade gliomas or non-neoplastic inflammatory diseases, but its results are not always conclusive.4 5
Arterial spin-labelling MRI imaging (ASL-MRI) can estimate brain function non-invasively by measuring cerebral blood flow (CBF).6 Here, we describe a patient with CNS-IRIS due to toxoplasmosis whose high MET uptake suggested lymphoma but whose ASL-MRI led to the correct diagnosis.
Case presentation
Our institutional review board approved this report. A 46-year-old man presented progressively worsening back pain for a few days and was diagnosed with pyogenic spondylitis at a nearby hospital. In the process of intensive examination, he was found to be HIV-antibody-positive, and he was transferred to our hospital. Laboratory studies on admission revealed a white blood cell count of 3970/µL, lactate dehydrogenase level of 154 IU/L, C reactive protein level of 1.79 mg/dL, cluster of differentiation 4 (CD4) cell count of 136/µL and CD4/CD8 of 0.3. Tests for hepatitis B core antibody, HIV antibody, rapid plasma regain and Treponema pallidum haemagglutination were positive. His HIV RNA level was 71 000 copies/mL. His toxoplasma IgG antibody level was 108 IU/mL (normal, <7 IU/mL). AIDS was diagnosed, and ART was started immediately. One month later, however, he had a sudden onset of convulsions on the left side of the body. Laboratory studies revealed that the CD4 cell count had increased slightly, to 193/µL, and HIV RNA was not detected. Cerebrospinal fluid (CSF) was not cloudy and had a cell count of 7.4/µL. CSF levels of sugar and protein were 50 and 93 mg/dL, respectively. Although CNS-IRIS due to toxoplasmosis or lymphoma was suspected, a definitive diagnosis was difficult. Unenhanced CT showed a 20 mm isoattenuation nodular lesion with surrounding oedema at the right frontoparietal lobe and a 15 mm low-attenuation lesion at the right occipital lobe. No acute intracranial haemorrhage was observed. On MRI, T2-weighted image (T2WI) showed a hyperintense nodular lesion surrounded by mild oedema at the right frontoparietal lobe (figure 1a) and the right occipital lobe. Postcontrast T1-weighted image (T1WI) showed 20 mm and 7 mm solid enhanced nodules at the right frontoparietal (figure 1b) and occipital lobe, respectively. The lesions showed increased signal intensity on diffusion-weighted image (DWI) (figure 1c). The apparent diffusion coefficient value was 0.82×10−3 mm2/s for the right frontoparietal lesion (figure 1d), which was fairly suggestive of lymphoma rather than toxoplasmosis. However, no increased regional CBF was detected on ASL-MRI (figure 1e), which made lymphoma considerably less likely.
Figure 1.
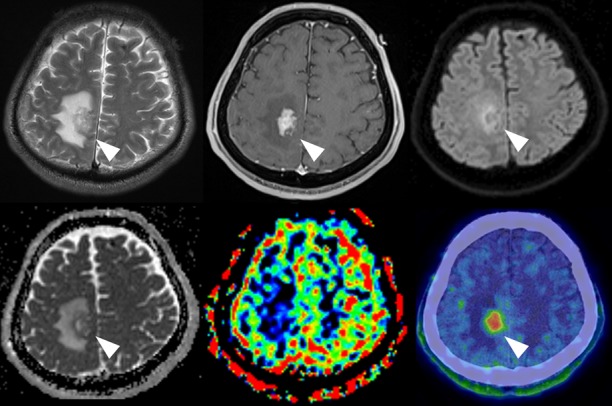
T2WI shows a hyperintense nodular lesion with mild surrounding oedema at the right frontoparietal lobe (a, arrowhead). Postcontrast T1WI shows a solid enhanced area (b, arrowhead) with an increased signal intensity on DWI (c, arrowhead) in conjunction with an ADC decrease (d, arrowhead). A low-CBF lesion is observed on ASL-MRI (e, arrowhead), but MET-PET 60 min after the injection shows a high uptake (f, arrowhead). ADC, apparent diffusion coefficient; ASL, arterial spin-labelling; CBF, cerebral blood flow; DWI, diffusion-weighted image; MET, 11C-methionine; T1WI, T1-weighted image; T2WI, T2-weighted image.
To differentiate between toxoplasmosis and lymphoma, MET-PET was performed. The lesions to contralateral frontal cortex ratio of MET uptake were calculated. The ratio of the right frontoparietal lesion 20 min after MET injection was 2.82 and that at 60 min was 2.71; for the right occipital lesion, the values were 2.24 after 20 min and 1.76 after 60 min (figure 1f). Both lesions showed high MET uptake.
Medical treatment with pyrimethamine, calcium folinate and sulfadiazine for toxoplasmosis had been initiated immediately after onset of convulsions; after MET-PET findings complicated the diagnosis, a partial resection of the right frontoparietal lesion was performed to confirm the diagnosis. Numerous inflammatory cells, predominantly neutrophils and some lymphocytes, had infiltrated in the brain tissue. Inflammation cells were accumulated mainly around the blood vessels. Grocott, Periodic acid-Schiff and Gram stains showed no obvious source of infection (eg, Toxoplasma gondii). However, no atypical lymphocytes were observed, and lymphoma was ruled out. Later, a T. gondii gene was detected by PCR of a CSF sample; therefore, CNS-IRIS due to toxoplasmosis was clinically confirmed. It was considered that T. gondii was not detected in the biopsy specimen because of the start of antitoxoplasmosis therapy prior to the biopsy. Treatment for toxoplasmosis was continued, and postcontrast T1WI showed both enhanced lesions markedly decreased at 8 months from onset.
Discussion
IRIS is often caused by exaggerated activation of the immune system following the initiation of ART.1 Several risk factors for the development of IRIS have been proposed: a rapid decline HIV viral load especially in the first 3 months after ART, a low baseline CD4 cell count (below 50/µL) and a rapid increase of CD4 cell count after initiation of ART.1 The present case made CNS-IRIS the most likely clinical diagnosis because of the HIV viral load suppression, the slightly increased CD4 cell count 1 month after starting ART and the T. gondii gene found in the CSF—even though there was no pronounced decrease of the CD4 cell count before treatment and no infectious pathogen was found in the biopsy specimen.
The most common causes of focal brain lesion in patients with HIV are toxoplasmosis and lymphoma.3 7 However, the treatment of toxoplasmosis and lymphoma are completely different. Particularly, lymphoma usually progresses rapidly in untreated cases, so that early diagnosis by brain biopsy and an early start to targeted lymphoma therapy is critical.7 By contrast, toxoplasmosis responds well to antitoxoplasmosis treatment, and more than 90% of patients with toxoplasmosis improve significantly by 14 days.7 The presenting clinical features of the two diseases are non-specific and demonstrate significant overlapping.3
The typical CT and MRI findings of AIDS-related lymphoma are reported to be solitary, isointensity to hyperintensity on T2WI with peritumoural oedema, hyperintensity on DWI and increased CBV on dynamic susceptibility contrast (DSC)-MRI.4 7 8 More than 50% of AIDS-related lymphoma cases show ring enhancement.4 However, the CT and MRI features of toxoplasmosis have been reported to be variable depending on pathologic evolution: namely, necrotising, organising and chronic abscesses based on the host’s immune response to T. gondii.3 The imaging features of toxoplasmosis are multiple, with various intensities on T2WI with perilesional oedema, hypointensity on DWI and decreased CBV on DSC-MRI.3 7 8 Moreover, lesions >10 mm in diameter often show ring enhancement while those that are smaller show various enhancement patterns.3 In practice, however, differentiating is often challenging because there is some overlap between toxoplasmosis and AIDS-related lymphoma.7 8
In the present case, no CBF increase on ASL-MRI gave negative evidence for lymphoma, although conventional MRI findings were non-specific, or the restricted diffusion within the lesion might suggest lymphoma. ASL-MRI typically shows an increased CBF in CNS lymphoma. Yamashita et al reported the relative CBF normalised by grey matter as 1.24±0.37 (mean±SD), which indicated that CNS lymphoma had CBF equal to or more than that in grey matter.6 In our case, however, the CBF in the lesion was apparently lower than that in the circumferential grey matter, which made lymphoma considerably less likely. This is the first report describing a patient with CNS-IRIS caused by toxoplasmosis showing a low CBF on ASL-MRI.
MET-PET evaluates the protein synthesis rate and the activity of amino acid transport of the pathologies, and tumourous lesions with increased metabolism and active amino acids transport show high MET uptake. In contrast to 18F-fluorodeoxyglucose (FDG), the background uptake of MET in normal tissue is low, providing good contrast with tumour uptake. Therefore, MET-PET has been reported to provide useful information to evaluate brain tumour and differentiate benign from malignant lesions in the brain. Ogawa et al reported that MET-PET showed high uptake in all lymphoma lesions and was useful for delineating lymphoma.9 However, Kawai et al reported that MET uptake in the tumour was obscure in lymphoma patients with atypical MRI findings, such as disseminated lesions, no lesion and ring-like enhanced lesions, in contrast to lymphoma patients with typical MRI findings.4 Furthermore, a high MET uptake has been reported in non-tumourous lesions, such as abscesses, haematomas and cerebral infarctions, and this mechanism may be related to disruption of the blood–brain barrier and increased metabolism or active amino acid transport.5 In terms of discrimination between toxoplasmosis and lymphoma, the utility of MET-PET is limited. In the present case, right frontoparietal and occipital lesions showed high uptake on MET-PET, suggesting lymphoma. However, the real cause was toxoplasmosis.
Learning points.
Central nervous system (CNS)-immune reconstitution inflammatory syndrome (IRIS) is an exacerbation of inflammatory reactions related to opportunistic infections as well as primary central nervous system malignancies after starting antiretroviral therapy for HIV-infected patients.
11C-methionine-PET or diffusion-weighted imaging made it difficult to differentiate toxoplasmosis from lymphoma in our case with CNS-IRIS.
Arterial spin-labelling MRI showed a low cerebral blood flow and proved helpful in identifying the true cause as toxoplasmosis.
Footnotes
Contributors: NW wrote the manuscript. TN analysed MRI, ASL-MRI and MET-PET of this case and contributed to writing the manuscript. TA was an attending doctor of the patient and contributed to writing the manuscript. TT contributed to writing the manuscript. All the authors read, reviewed and approved the final version of this case report.
Competing interests: None declared.
Patient consent: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Müller M, Wandel S, Colebunders R, et al. Immune reconstitution inflammatory syndrome in patients starting antiretroviral therapy for HIV infection: a systematic review and meta-analysis. Lancet Infect Dis 2010;10:251–61. 10.1016/S1473-3099(10)70026-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gopal S, Patel MR, Achenbach CJ, et al. Lymphoma immune reconstitution inflammatory syndrome in the center for AIDS research network of integrated clinical systems cohort. Clin Infect Dis 2014;59:279–86. 10.1093/cid/ciu270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sakamoto N, Maeda T, Mikita K, et al. Clinical presentation and diagnosis of toxoplasmic encephalitis in Japan. Parasitol Int 2014;63:701–4. 10.1016/j.parint.2014.05.007 [DOI] [PubMed] [Google Scholar]
- 4.Kawai N, Okubo S, Miyake K, et al. Use of PET in the diagnosis of primary CNS lymphoma in patients with atypical MR findings. Ann Nucl Med 2010;24:335–43. 10.1007/s12149-010-0356-z [DOI] [PubMed] [Google Scholar]
- 5.Dethy S, Goldman S, Blecic S, et al. Carbon-11-methionine and fluorine-18-FDG PET study in brain hematoma. J Nucl Med 1994;35:1162–6. [PubMed] [Google Scholar]
- 6.Yamashita K, Yoshiura T, Hiwatashi A, et al. Differentiating primary CNS lymphoma from glioblastoma multiforme: assessment using arterial spin labeling, diffusion-weighted imaging, and (18)F-fluorodeoxyglucose positron emission tomography. Neuroradiology 2013;55:135–43. 10.1007/s00234-012-1089-6 [DOI] [PubMed] [Google Scholar]
- 7.Utsuki S, Oka H, Abe K, et al. Primary central nervous system lymphoma in acquired immune deficiency syndrome mimicking toxoplasmosis. Brain Tumor Pathol 2011;28:83–7. 10.1007/s10014-010-0001-5 [DOI] [PubMed] [Google Scholar]
- 8.Ernst T, Chang L, Arnold S. Increased glial metabolites predict increased working memory network activation in HIV brain injury. Neuroimage 2003;19:1686–93. 10.1016/S1053-8119(03)00232-5 [DOI] [PubMed] [Google Scholar]
- 9.Ogawa T, Kanno I, Hatazawa J, et al. Methionine PET for follow-up of radiation therapy of primary lymphoma of the brain. Radiographics 1994;14:101–10. 10.1148/radiographics.14.1.8128041 [DOI] [PubMed] [Google Scholar]


