Abstract
Novel evidence-based prosthetic designs and biomaterials facilitate the performance of highly successful joint replacement (JR) procedures. To achieve this goal, constructs must be durable, biomechanically sound, and avoid adverse local tissue reactions. Different biomaterials such as metals and their alloys, polymers, ceramics, and composites are currently used for JR implants. This review focuses on (1) the biological response to the different biomaterials used for TJR and (2) the chronic inflammatory and foreign-body response induced by byproducts of these biomaterials. A homeostatic state of bone and surrounding soft tissue with current biomaterials for JR can be achieved with mechanically stable, infection free and intact (as opposed to the release of particulate or ionic byproducts) implants. Adverse local tissue reactions (an acute/chronic inflammatory reaction, periprosthetic osteolysis, loosening and subsequent mechanical failure) may evolve when the latter conditions are not met. This article (Part 2 of 2) summarizes the biological response to the non-metallic materials commonly used for joint replacement including polyethylene, ceramics, and polymethylmethacrylate (PMMA), as well as the foreign body reaction to byproducts of these materials.
Keywords: orthopedic implants, biological response, foreign body response, inflammation, biomaterials
INTRODUCTION
The goal of total joint replacement (TJR) is to treat end-stage arthritis that is painful and disabling for a patient’s activities of daily living and lifestyle. TJRs are mainly performed in large joints (hip, knee, shoulder, and elbow). Different bearing surfaces can be used; the most common are metal-on-polyethylene (MOP), ceramic-on-ceramic (COC), or metal-on-metal (MOM). Besides relieving pain, these biomaterials must be resistant enough to support weight bearing of the lower extremity. Moreover, issues related to biocompatibility are very important, including minimizing adverse effects on the surrounding tissues and the absence of infection. Despite major improvements in these implants, TJRs may not last for a lifetime, especially in young active patients. The production of wear particles and their byproducts interacting with surrounding cells is one biological trigger that may jeopardize the implant’s long-term function.1 It has been shown that each step produces hundreds of thousands to millions of particles for hip and knee replacements.2
TJRs are made with different materials. Metals alloys including titanium alloy, cobalt-chromium alloy, and stainless steel are the most frequent materials. Polymers are the second group of biomaterials and include ultra high molecular weight polyethylene (UHMWPE), highly cross-linked polyethylene (HXLPE), vitamin E enriched polyethylene, and polymethyl-methacrylate (PMMA). Ceramics (alumina and zirconia) and less frequently, composites, represent the last groups. With the current exponential development of new products associated with higher demands of younger patients, a better understanding of the interaction between bone and biomaterials for TJR would prove valuable. The purpose of this review (Part 2 of 2) is to (1) provide an overview of the different polymers and ceramics used in major orthopedic devices for joint replacement and (2) discuss the issue of the chronic inflammatory and foreign body reaction as it relates to byproducts from orthopedic implants.
POLYETHYLENE
Bulk
The literature regarding the effect of bulk polyethylene on bone tissue and cells is scarce. After implantation of bulk polyethylene in the proximal tibia of rabbits for 3 months, a thick fibrous membrane containing fibroblasts and giant cells was observed.3 However, the thickness of the membrane and the number of giant cells and macrophages was significantly lower in the bulk group than in a group in which the same volume of polyethylene (PE) particles was implanted.
Particles
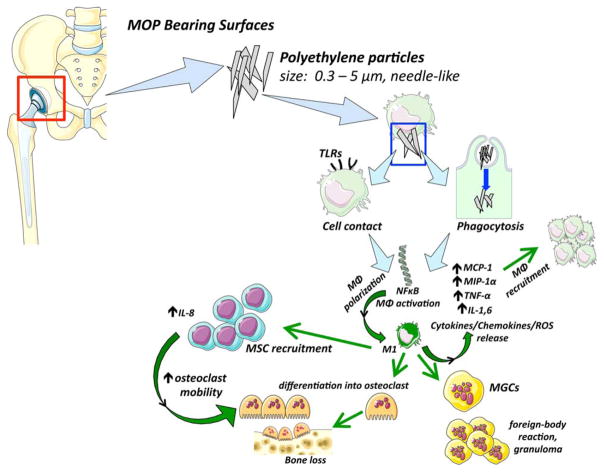
Polyethylene (PE) is one the most widely used bearings for hip replacement in the USA.4 Manufacturers and researchers are constantly working to improve its longevity by inventing more wear-resistant materials. Second-generation HXLPE5 includes the doping of the antioxidant vitamin E within the PE, and repeated treatment with heating and annealing of the polymer.6 PE wear particles migrate within the entire periprosthetic bed,7 known as “the effective joint space.” Interaction with the local cells (resident phagocytic macrophages, osteoblasts, and osteoclasts) triggers a cascade of proinflammatory responses. The macrophage is the key cell8 in this complex innate (nonspecific, nonantigenic) immunological reaction. This response should be distinguished from the specific, antigen-associated, acquired immune response. Using UHMWPE, Xing et al. showed that activation of macrophages occurs either by phagocytosis9 or by cell contact through different membrane receptors1 (Toll-like receptors (TRLs), CD11b, CD14). TLRs are known to function in the innate immune response.10 After particle-induced activation, TLRs act primarily through an adapter protein called myeloid differentiation primary response gene 88 (MyD88) to induce activation NF-kB and other signaling pathways (mitogen-activated protein kinase, IRF3).11 Valladares et al.12 have shown that TLR2 and TLR4 were highly expressed in a calvarial murine model of PE-induced osteolysis. The inflammatory cascade leads to the release of various proinflammatory cytokines (IL-1, IL-6, TNF-α), growth factors (macrophage colony stimulating factor-1) and chemokines (MIP-1α, MCP-1) that would ultimately lead to systemic recruitment of more macrophages to the area. Several in vivo studies have shown that these cytokines contribute to the systemic chemotaxis of macrophages in the presence of UHMWPE particles.2,13–15 As shown by Maitra et al.16 PE particles can also trigger the inflammasome. The CCR1/MIP-1α ligand/receptor axis has been shown to facilitate systemic recruitment of MSCs in the presence of UHMWPE particles.17 Recent studies focused on a new concept of macrophage activation. Depending upon the local environment, macrophages can be polarized to M1 (proinflammatory) and M2 (anti-inflammatory) phenotypes.18,19 M1 macrophages, producers of primarily proinflammatory mediators including TNF-α, IL-1, and IL-6, express inducible nitric oxide synthase (iNOS), whereas M2 macrophages produce primarily anti-inflammatory mediators including IL-4, IL-10, and IL-13, and express mammalian chitinase Ym1, Arginase 1, CD163, and chitotriosidase.20,21 One possible way to decrease local inflammation would be to polarize uncommitted M0 or M1 macrophages toward the M2 phenotype.22 Thus, bone loss was significantly decreased following IL-4 administration to PE treated calvaria.23 Pajarinen et al.24 showed that continuous delivery of IL-4 can modulate macrophage phenotype in vitro from M1 to M2. These results provide promising strategies to mitigate periprosthetic osteolysis by modulating the cytokine microenvironment, and represent avenues for further in vivo studies using clinically relevant models.25–28 Figure 1 summarizes the effect of PE particles.
FIGURE 1.
The biological reaction to PE. After phagocytosis or cell contact, PE particles activate nuclear transcription factors and the inflammasome. Subsequent cytokine and chemokine release occurs, leading to systemic recruitment of macrophages. The inflammatory microenvironment polarizes M0 macrophages to proinflammatory M1 macrophages. Macrophages can differentiate into osteoclasts. MSCs increase their secretion of IL-8. Ultimately, the accumulation of osteoclasts leads to osteolysis. The fusion of macrophages leads to MGCs. TLRs = Toll-like receptors; MΦ= macrophage; MCP-1 = monocyte chemoattractant factor 1; MIP-1α= macrophage inflammatory protein 1 alpha; TNF-α= Tumor necrosis factor-alpha; IL = interleukin; MSCs = msenchymal stem cells; ROS = reactive oxygen species; MGCs = multinucleated giant cells; NF-κB = nuclear factor-kappa B.
CERAMICS
Bulk
The response to bulk alumina has been extensively investigated in vivo. Christel29 have shown that when alumina is implanted in muscle, a fibrous membrane containing mostly fibroblasts is induced. The reaction to bulk alumina in bone has been characterized as well. Under nonloaded conditions, a thin fibrous capsule evolves around an alumina implant but osseointegration could be achieved.30 Under loaded conditions, a degree of osseointegration was found with porous alumina.31 More recently, Josset et al.32 showed that bulk alumina and zirconia had no cytostatic or cytotoxic effects when cultured with human osteoblasts.
Particles
Current orthopedic ceramics for joint replacement are actually composites of two of ceramics: alumina (AL2O3) and zirconia (ZrO2) in which alumina is the primary or continuous phase (70–95%) and zirconia is the secondary phase (30% to 5%).33 Zirconia is used to toughen the alumina (and known as ZTA). Typical alumina particles are in the nanometer range and have a bimodal distribution (24 ± 19 nm and 0.05 ± 3.2 μm) as shown by Hatton et al.34 Germain et al.35 showed a cytotoxic effect of clinically relevant alumina particles (size used: 5–20 nm) cultured with human histiocytes. Moreover, the authors also used commercially available alumina particles, with a much larger size (size used: 0.503 ± 0.19 μm) and showed no effect on the viability of the cells. Gutwein et al.36 compared the effect of nanophase versus larger size alumina particles (23 nm vs. 179 nm) on the viability of osteoblasts; the viability was dramatically decreased with larger size particles. Furthermore, the nanophase particles showed no significant effects on cell viability compared to the control group with no particles. Catelas et al.37 using the J774 macrophage cell line showed that ceramic particle phagocytosis increased with increasing concentration for particles up to 2 μm in size. Above that size, the phagocytosis reached a plateau irrespective of the size or the concentration of the particles. The same group also compared alumina, zirconia and PE particles,38 and showed that increasing size and concentration of particles had an increasing effect on cytotoxicity as opposed to the previously mentioned works from Germain et al. and Gutwein et al. However, cytotoxicity still remained very low (10%) for both alumina and zirconia particles. Release of TNF-α followed the same trend and was significantly higher for PE particles. In a related study,39 fluorescence microscopy and DNA laddering showed that macrophage apoptosis was size- and concentration-dependent and reached a plateau above 150 particles per macrophage at 1.3 μm Lucarelli et al.40 analyzed the effect of nano-sized zirconia particles on macrophages with the highest nontoxic dose. Nanoparticles of ZrO2 showed a selective capacity for inducing/increasing expression of TLR3, TLR7, and TLR10, but had a limited stimulatory effect on IL-1β production and no effect on TNF-α production. Interestingly, the authors also showed a proinflammatory effect of ZrO2 nanoparticles, with decreased production of IL-1ra (a marker of M2 macrophages) by M1 polarized macrophages. Kaufman et al.41 found a limited increase in IL-1β and MCP-1 production after challenging human macrophages with alumina particles. Bylski et al.42 challenged human monocytic cells with alumina particles; RANK, TNF-α, and OPG mRNA were only slightly upregulated. Moreover, using clinically relevant alumina size particles, Roualdes et al.43 showed a moderate nonspecific granulomatous response of the synovial membrane in rat knees in vivo. The genotoxicity of alumina particles has been analyzed by Tsaousi et al.44 using primary human fibroblasts. The authors concluded that ceramic particles are only weakly genotoxic to human cells. The authors used fibroblastic cells in their studies; further experiments are needed to evaluate the potential genotoxicity when alumina particles are exposed to osteoprogenitor cells and macrophages. Taken together, alumina particles in their clinically relevant size (nanometer) have limited impact on cell viability and limited influence on cytokines production. This would explain why there is a relatively low incidence of osteolysis in ceramic-on-ceramic implants.45–47 Figure 2 summarizes the effect of ceramic particles.
FIGURE 2.
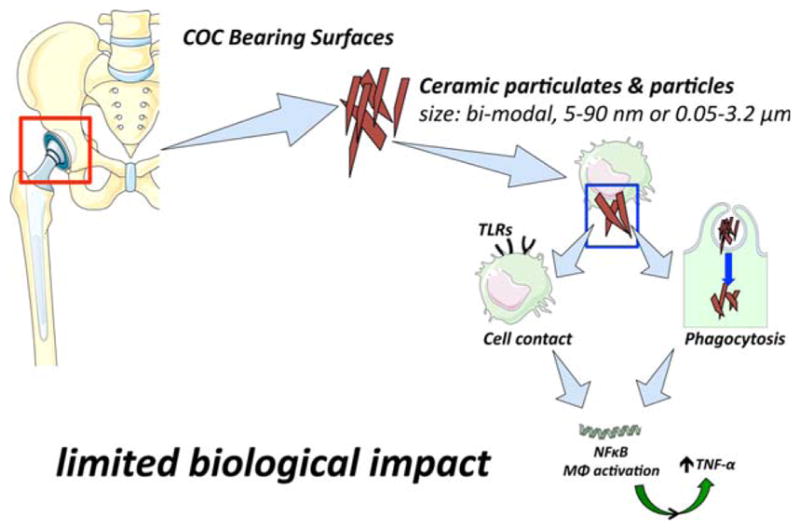
The biological reaction to ceramic. Ceramics are responsible for a mild inflammatory response. The cytotoxicity is very low and ceramic particles induce a slight increase in the production of inflammatory cytokines. TLRs = Toll-like receptors; MΦ= macrophage; NF-κB = nuclear factor-kappa B.
POLYMETHYLMETHACRYLATE (PMMA)
Bulk
Retrieval studies allow us to better understand the effect of bulk PMMA on the surrounding tissues. For instance, Charnley48 showed that the fibrous tissue between bone and cement underwent a metaplasia into fibrous cartilage with ossification as a result of mechanical pressure. Maloney et al.49 found excellent osseointegration and no intervening fibrous tissue around cemented components. Localized areas of osteolysis were due to microfractures within the bulk PMMA, that release cement particles, resulting in a localized foreign-body response.50 Willert et al.51 analyzed the bone-cement interface at the level of localized osteolysis. They found large foreign-body granulomas and giant cells with PMMA debris inside the cells.
In vivo studies52 with bulk PMMA (unmodified) have shown a limited biological response. After implantation of bulk PMMA, the response mainly consisted in the production of a thin fibrous membrane with occasional giant cells, lymphocytes, and histiocytes. The thickness of the membrane and the amount of cells was much lower than after implantation of PMMA particles. Consistently, the organ culture of the fibrous membrane harvested around both bulk PMMA and PMMA particles showed different profiles PGE2 production: lower levels of PGE2 were produced after implantation of bulk PMMA compared to PMMA particles (size: 10–100μm).53 During polymerization, PMMA releases free radicals that have been shown to be cytotoxic for osteoblastic cells.54
Particles
The influence of PMMA particles has been widely studied, and was originally called “cement disease” because of the chronic inflammatory reaction to bone cement breakdown products.55 Quinn et al.56 co-cultured osteoblasts with PMMA-challenged macrophages. After 14 days of culture they found an increased number of multinucleated tartrate-resistant acid phosphatase (TRAP)-positive cells (osteoclasts) and lacunar osteolysis. Huang et al.57 challenged macrophages with PMMA articles and showed an increase in MCP-1 production. Interestingly, the conditioned media (CM) led to chemotaxis of both macrophages and MSCs. The osteolytic potential of PMMA particles has been found in other studies,58–61 and has been associated with increased production of proinflammatory cytokines. Yaszay et al.62 challenged human fibroblasts with PMMA particles and found increased release of MCP-1 and IL-6. Chiu et al.61 challenged bone marrow osteoprogenitor cells and demonstrated that PMMA particles (size: 1–10μm) inhibited osteoblastic differentiation. Ramachandran et al.63 showed that PMMA particles (size: 4–10μm) did not promote the death of human osteoblasts after 21 days and there were no significant effects on alkaline phosphatase or osteocalcin levels. Antonios et al.64 investigated the time course of murine macrophage polarization and cytokine release in response to challenge with combinations of PMMA particles, lipopolysaccharide (LPS) and IL-4 in vitro. As expected, PMMA particles increased the levels of IL-1β and TNF-α. The most effective protocol to mitigate this response was to add IL-4 before PMMA particle challenge to M1 proinflammatory macrophages.64 Thus, the polarization of M1 to an M2 macrophage phenotype could represent a strategy to mitigate particle-associated inflammation and peri-prosthetic bone loss. Figure 3 summarizes the effect of PMMA particles.
FIGURE 3.
The biological reaction to PMMA. After phagocytosis of PMMA particles, macrophages become activated and secrete proinflammatory cytokines. The response is both local through an autocrine mechanism perpetuating macrophags activation, and systemic with recruitment of macrophages to the site of inflammation. Macrophages can differentiate into osteoclasts leading to osteolysis. MΦ= macrophage; MCP-1 = monocyte chemoattractant factor 1; TNF-α= tumor necrosis factor alpha; IL = interleukin; MSCs = mesenchymal stem cells; TRAP = tartrate resistant acid phosphatase.
THE FOREIGN BODY REACTION
The foreign body reaction is an adverse innate host reaction to an implanted medical device.65 The innate immune reaction is nonantigen specific where as the acquired (also known as adaptive) immune response is antigen specific. The innate immune system is complex and requires interaction among cytokines, chemokines and different types of cells, with the macrophage as the key cell.8 B and T lymphocytes and plasma cells, cells associated with the adaptive immune system, are scarce in tissues with ceramic, PMMA and PE debris.1 Local macrophages are activated by wear particles either by phagocytosis or cell contact. Retrieval studies have shown that macrophages are widely represented among the cell population.66–68 After activation, macrophages secrete potent proinflammatory cytokines and chemokines to systematically recruit more macrophages and inflammatory cells. Nakashima et al.69 analyzed tissues from failed arthroplasties undergoing revision surgery. The most numerous cells were macrophages and multi-nucleated giant cells within the granulomatous tissue. MCP-1 and MIP-1α were detected in macrophage-rich areas. Those two chemokines play a major role recruiting macrophages and mesenchymal stem cells (MSCs) to the periprosthetic tissues.15,17 Table I summarizes the most prominent chemokines and cytokines that are involved in the innate reaction to particulate debris. Shanbhag et al.70 in a retrieval study using tissues harvested from hip revision surgeries performed for peri-prosthetic osteolysis or aseptic loosening found high levels of IL-8, MCP-1, TGF-β1 and adhesion molecules such as sICAM-1. Interestingly, Wang et al.71 compared cytokine profiles of synovial fluid from primary versus revision hip arthroplasties (metal-on-polyethylene). Fluids from revised patients had higher RANKL expression on osteoblastic cells, interleukin (IL)–6, IL-8, IL-10, interferon-γ-inducible protein (IP)–10, MCP-1, monokine induced by interferon-γ (MIG), and lower OPG/RANKL ratios in their synovial fluid compared to primary THAs.
TABLE I.
Cytokines and Chemokines Involved in the Biological Response to Orthopedic Implants for Joint Replacement (Polyethylene, Ceramics, PMMA)
| Cytokines/Chemokines | Function |
|---|---|
| MCP-1 | Immediate early stress-response factor. Important in systemic migration of MΦto local site. Produced by monocytes and activated NK cells, fibroblasts and bone-marrow derived primary osteoblasts. |
| MIP-1α | MIP-1α enhances the release of IL-1 and IL-6 affecting neighboring cells in a paracrine manner. Produced by activated MΦ and T lymphocytes. |
| IL-1α and IL-1β | IL-1 activates MΦ, neutrophils and endothelial cells; stimulates fibroblasts and osteoclasts, and induces prostaglandin E2 and collagenase synthesis. IL-1α and IL-1β are produced by two distinct genes. Secreted by many cell types including macrophages. |
| IL-6 | Activates T and B cells and induces B cells to differentiate and secrete immunoglobulins. Secreted by macrophages, T cells, fibroblasts and other cell types. |
| TNF-α | Stimulates fibroblasts and granulocytes; some of the effects are similar to IL-1. Secreted by activated lymphocytes, monocytes, MΦand other cells. |
| PDGF-α | Increases class-II antigen expression in macrophages, stimulates osteoclasts to resorb bone, induces collagenase and prostaglandin production, and is chemotactic for fibroblasts, monocytes and neutrophils. Secreted by MΦ, platelets, endothelial cells and fibroblasts. |
| TGF-β | Stimulates fibroblast growth, extracellular matrix formation and suppresses T- and B-cell proliferation. TGF-β also stimulates osteoblast and inhibits osteoclast function. Secreted by T cells, activated MΦ, and other cell types. |
MΦ= macrophage; TNF-α= tumor necrosis factor alpha; MCP-1 = monocyte chemoattractant factor 1; IL = interleukin.
Thus, the interface surrounding failed loose implants with periprosthetic osteolysis demonstrates fibrohistiocytic tissue with numerous foreign body giant cells and osteoclasts, which produce increased amounts of proinflammatory cytokines, chemokines, and other substances.
Rao et al.22 showed that retrieved periprosthetic tissues demonstrated increased M1/M2 macrophage ratios compared to non-operated osteoarthritic synovial tissues. This increased number of M1 macrophages maintains a local inflammatory state that ultimately leads to a chronic inflammatory and foreign body reaction. Foreign body giant cells, also called polykaryons, have been widely found within the bone-implant interface.72 MGCs come from fusion of multiple macrophages in response to hematopoietic growth factors73 (GM-CSF) and interleukins (IL-3, IL-4).74,75 Adhesion molecules are also involved in the MGCs development, including intercellular adhesion molecule-1(ICAM-1/CD54); the receptor CR3 (CD11b/CD18) is also expressed by multinucleated giant cells.76 Locally, the presence of MGCs increases both osteoclastic bone resorption and osteoclast-like cell growth and differentiation by their ability to release TGF-β and other factors.
CONCLUSION
Orthopedic implants used for joint replacement are effective in relieving pain and restoring function. These implants have limited life expectancy due wear and the host reaction to wear byproducts. Most modern materials for TJR are well tolerated by the body as long as they remain in bulk form, achieve mechanical stability within bone, and are not colonized by microorganisms to produce chronic infection. If there is excessive wear of the materials and generation of wear particles or ionic complexes, the prosthesis will be associated with an acute and chronic inflammation, which may induce periprosthetic osteolysis, loss of bony support subsequent loosening, and failure of the implant. A more comprehensive understanding of the local and systemic biological pathways associated with implants for joint replacement will optimize the selection of appropriate materials and design parameters for future arthroplasties.
References
- 1.Tuan RS, Lee FY, YTK, Wilkinson JM, Smith RL. What are the local and systemic biologic reactions and mediators to wear debris, and what host factors determine or modulate the biologic response to wear particles? J Am Acad Orthop Surg. 2008;16(Suppl 1):S42–S48. doi: 10.5435/00124635-200800001-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Goodman SB, Ma T. Cellular chemotaxis induced by wear particles from joint replacements. Biomaterials. 2010;31:5045–5050. doi: 10.1016/j.biomaterials.2010.03.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Goodman SB, Fornasier VL, Kei J. The effects of bulk versus particulate ultra-high-molecular-weight polyethylene on bone. J Arthroplasty. 1988;3(Suppl):S41–S46. doi: 10.1016/s0883-5403(88)80007-x. [DOI] [PubMed] [Google Scholar]
- 4.Bozic KJ, Kurtz S, Lau E, Ong K, Chiu V, Vail TP, Rubash HE, Berry DJ. The epidemiology of bearing surface usage in total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91:1614–1620. doi: 10.2106/JBJS.H.01220. [DOI] [PubMed] [Google Scholar]
- 5.Goodman SB, Gomez Barrena E, Takagi M, Konttinen YT. Biocompatibility of total joint replacements: A review. J Biomed Mater Res A. 2009;90:603–618. doi: 10.1002/jbm.a.32063. [DOI] [PubMed] [Google Scholar]
- 6.Essner A, Herrera L, Yau S-S, Wang A, Dumbleton J, Manley M. Sequentially crosslinked and annealed UHMWPE CR knee wear. Transactions of the 51st Annual Meeting of the Orthopedic Research Society; Washington, DC: Orthopedic Research Society; 2005. [Google Scholar]
- 7.Schmalzried TP, Jasty M, Harris WH. Periprosthetic bone loss in total hip arthroplasty. Polyethylene wear debris and the concept of the effective joint space. J Bone Joint Surg Am. 1992;74:849–863. [PubMed] [Google Scholar]
- 8.Nich C, Goodman SB. Role of macrophages in the biological reaction to wear debris from joint replacements. J Long Term Eff Med Implants. 2014;24:259–265. doi: 10.1615/jlongtermeffmedimplants.2014010562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xing S, Waddell JE, Boynton EL. Changes in macrophage morphology and prolonged cell viability following exposure to polyethylene particulate in vitro. Microsc Res Technol. 2002;57:523–529. doi: 10.1002/jemt.10106. [DOI] [PubMed] [Google Scholar]
- 10.Pajarinen J, Jamsen E, Konttinen YT, Goodman SB. Innate immune reactions in septic and aseptic osteolysis around hip implants. J Long Term Eff Med Implants. 2014;24:283–296. doi: 10.1615/jlongtermeffmedimplants.2014010564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Akira S, Takeda K, Kaisho T. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol. 2001;2:675–680. doi: 10.1038/90609. [DOI] [PubMed] [Google Scholar]
- 12.Valladares RD, Nich C, Zwingenberger S, Li C, Swank KR, Gibon E, Rao AJ, Yao Z, Goodman SB. Toll-like receptors-2 and 4 are overexpressed in an experimental model of particle-induced osteolysis. J Biomed Mater Res A. 2014;102:3004–3011. doi: 10.1002/jbm.a.34972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ren PG, Huang Z, Ma T, Biswal S, Smith RL, Goodman SB. Surveillance of systemic trafficking of macrophages induced by UHMWPE particles in nude mice by noninvasive imaging. J Biomed Mater Res A. 2010;94:706–711. doi: 10.1002/jbm.a.32744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ren PG, Irani A, Huang Z, Ma T, Biswal S, Goodman SB. Continuous infusion of UHMWPE particles induces increased bone macrophages and osteolysis. Clin Orthop Relat Res. 2011;469:113–122. doi: 10.1007/s11999-010-1645-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gibon E, Ma T, Ren P-G, Fritton K, Biswal S, Yao Z, Smith L, Goodman SB. Selective inhibition of the MCP-1-CCR2 ligand-receptor axis decreases systemic trafficking of macrophages in the presence of UHMWPE particles. J Orthop Res. 2012;30:547–553. doi: 10.1002/jor.21548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maitra R, Clement CC, Scharf B, Crisi GM, Chitta S, Paget D, Purdue PE, Cobelli N, Santambrogio L. Endosomal damage and TLR2 mediated inflammasome activation by alkane particles in the generation of aseptic osteolysis. Mol Immunol. 2009;47:175–184. doi: 10.1016/j.molimm.2009.09.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gibon E, Yao Z, Rao AJ, Zwingenberger S, Batke B, Valladares R, Smith RL, Biswal S, Gambhir SS, Goodman SB. Effect of a CCR1 receptor antagonist on systemic trafficking of MSCs and polyethylene particle-associated bone loss. Biomaterials. 2012;33:3632–3638. doi: 10.1016/j.biomaterials.2012.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brown BN, Ratner BD, Goodman SB, Amar S, Badylak SF. Macrophage polarization: an opportunity for improved outcomes in biomaterials and regenerative medicine. Biomaterials. 2012;33:3792–3802. doi: 10.1016/j.biomaterials.2012.02.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Konttinen YT, Pajarinen J, Takakubo Y, Gallo J, Nich C, Takagi M, Goodman SB. Macrophage polarization and activation in response to implant debris: Influence by “particle disease” and “ion disease”. J Long Term Eff Med Implants. 2014;24:267–281. doi: 10.1615/jlongtermeffmedimplants.2014011355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ho VW, Sly LM. Derivation and characterization of murine alternatively activated (M2) macrophages. Methods Mol Biol (Clifton, NJ) 2009;531:173–185. doi: 10.1007/978-1-59745-396-7_12. [DOI] [PubMed] [Google Scholar]
- 21.Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004;25:677–686. doi: 10.1016/j.it.2004.09.015. [DOI] [PubMed] [Google Scholar]
- 22.Rao AJ, Gibon E, Ma T, Yao Z, Smith RL, Goodman SB. Revision joint replacement, wear particles, and macrophage polarization. Acta Biomater. 2012;8:2815–2823. doi: 10.1016/j.actbio.2012.03.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rao AJ, Nich C, Dhulipala LS, Gibon E, Valladares R, Zwingenberger S, Smith RL, Goodman SB. Local effect of IL-4 delivery on polyethylene particle induced osteolysis in the murine calvarium. J Biomed Mater Res A. 2013;101:1926–1934. doi: 10.1002/jbm.a.34486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pajarinen J, Tamaki Y, Antonios JK, Lin T-H, Sato T, Yao Z, Takagi M, Konttinen YT, Goodman SB. Modulation of mouse macrophage polarization in vitro using IL-4 delivery by osmotic pumps. J Biomed Mater Res A. 2015;103:1339–1345. doi: 10.1002/jbm.a.35278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ma T, Huang Z, Ren PG, McCally R, Lindsey D, Smith RL, Goodman SB. An in vivo murine model of continuous intramedullary infusion of polyethylene particles. Biomaterials. 2008;29:3738–3742. doi: 10.1016/j.biomaterials.2008.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ortiz SG, Ma T, Epstein NJ, Smith RL, Goodman SB. Validation and quantification of an in vitro model of continuous infusion of submicron-sized particles. J Biomed Mater Res B Appl Biomater. 2008;84:328–333. doi: 10.1002/jbm.b.30875. [DOI] [PubMed] [Google Scholar]
- 27.Ortiz SG, Ma T, Regula D, Smith RL, Goodman SB. Continuous intramedullary polymer particle infusion using a murine femoral explant model. J Biomed Mater Res B Appl Biomater. 2008;87:440–446. doi: 10.1002/jbm.b.31122. [DOI] [PubMed] [Google Scholar]
- 28.Ma T, Ortiz SG, Huang Z, Ren P, Smith RL, Goodman SB. In vivo murine model of continuous intramedullary infusion of particles–a preliminary study. J Biomed Mater Res B Appl Biomater. 2009;88:250–253. doi: 10.1002/jbm.b.31175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Christel PS. Biocompatibility of surgical-grade dense polycrystalline alumina. Clin Orthop Relat Res. 1992:10–18. [PubMed] [Google Scholar]
- 30.Plenk H, Salzer M, Locke H, Stark N, Punzet G, Zweymuller K. Extracortical attachment of bioceramic endoprostheses to long bones without bone cement. Clin Orthop Relat Res. 1978;132:252–265. [PubMed] [Google Scholar]
- 31.Hulbert SF, Matthews JR, Klawitter JJ, Sauer BW, Leonard RB. Effect of stress on tissue ingrowth into porous aluminum oxide. J Biomed Mater Res. 1974;3:85–97. doi: 10.1002/jbm.820080310. [DOI] [PubMed] [Google Scholar]
- 32.Josset Y, Oum’Hamed Z, Zarrinpour A, Lorenzato M, Adnet JJ, Laurent-Maquin D. In vitro reactions of human osteoblasts in culture with zirconia and alumina ceramics. J Biomed Mater Res. 1999;47:481–493. doi: 10.1002/(sici)1097-4636(19991215)47:4<481::aid-jbm4>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 33.Kurtz SM, Kocagoz S, Arnholt C, Huet R, Ueno M, Walter WL. Advances in zirconia toughened alumina biomaterials for total joint replacement. J Mech Behav of Biomed Mater. 2014;31:107–116. doi: 10.1016/j.jmbbm.2013.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hatton A, Nevelos JE, Nevelos AA, Banks RE, Fisher J, Ingham E. Alumina-alumina artificial hip joints. Part I: A histological analysis and characterisation of wear debris by laser capture microdissection of tissues retrieved at revision. Biomaterials. 2002;23:3429–3440. doi: 10.1016/s0142-9612(02)00047-9. [DOI] [PubMed] [Google Scholar]
- 35.Germain MA, Hatton A, Williams S, Matthews JB, Stone MH, Fisher J, Ingham E. Comparison of the cytotoxicity of clinically relevant cobalt-chromium and alumina ceramic wear particles in vitro. Biomaterials. 2003;24:469–479. doi: 10.1016/s0142-9612(02)00360-5. [DOI] [PubMed] [Google Scholar]
- 36.Gutwein LG, Webster TJ. Increased viable osteoblast density in the presence of nanophase compared to conventional alumina and titania particles. Biomaterials. 2004;25:4175–4183. doi: 10.1016/j.biomaterials.2003.10.090. [DOI] [PubMed] [Google Scholar]
- 37.Catelas I, Huk OL, Petit A, Zukor DJ, Marchand R, Yahia L. Flow cytometric analysis of macrophage response to ceramic and polyethylene particles: Effects of size, concentration, and composition. J Biomed Mater Res. 1998;41:600–607. doi: 10.1002/(sici)1097-4636(19980915)41:4<600::aid-jbm12>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- 38.Catelas I, Petit A, Marchand R, Zukor DJ, Yahia L, Huk OL. Cytotoxicity and macrophage cytokine release induced by ceramic and polyethylene particles in vitro. J Bone Joint Surg Br. 1999;81:516–521. doi: 10.1302/0301-620x.81b3.8737. [DOI] [PubMed] [Google Scholar]
- 39.Catelas I, Petit A, Zukor DJ, Marchand R, Yahia L, Huk OL. Induction of macrophage apoptosis by ceramic and polyethylene particles in vitro. Biomaterials. 1999;20:625–630. doi: 10.1016/s0142-9612(98)00214-2. [DOI] [PubMed] [Google Scholar]
- 40.Lucarelli M, Gatti AM, Savarino G, Quattroni P, Martinelli L, Monari E, Boraschi D. Innate defence functions of macrophages can be biased by nano-sized ceramic and metallic particles. Eur Cytokine Netw. 2004;15:339–346. [PubMed] [Google Scholar]
- 41.Kaufman AM, Alabre CI, Rubash HE, Shanbhag AS. Human macrophage response to UHMWPE, TiAlV, CoCr, and alumina particles: Analysis of multiple cytokines using protein arrays. J Biomed Mater Res A. 2008;84:464–474. doi: 10.1002/jbm.a.31467. [DOI] [PubMed] [Google Scholar]
- 42.Bylski D, Wedemeyer C, Xu J, Sterner T, Loer F, von Knoch M. Alumina ceramic particles, in comparison with titanium particles, hardly affect the expression of RANK-, TNF-alpha-, and OPG-mRNA in the THP-1 human monocytic cell line. J Biomed Mater Res A. 2009;89:707–716. doi: 10.1002/jbm.a.31956. [DOI] [PubMed] [Google Scholar]
- 43.Roualdes O, Duclos M-E, Gutknecht D, Frappart L, Chevalier Jrm, Hartmann DJ. In vitro and in vivo evaluation of an aluminazirconia composite for arthroplasty applications. Biomaterials. 2010;31:2043–2054. doi: 10.1016/j.biomaterials.2009.11.107. [DOI] [PubMed] [Google Scholar]
- 44.Tsaousi A, Jones E, Case CP. The in vitro genotoxicity of orthopedic ceramic (Al2O3) and metal (CoCr alloy) particles. Mutat Res. 2010;697:1–9. doi: 10.1016/j.mrgentox.2010.01.012. [DOI] [PubMed] [Google Scholar]
- 45.Hannouche D, Hamadouche M, Nizard R, Bizot P, Meunier A, Sedel L. Ceramics in total hip replacement. Clin Orthop Relat Res. 2005;430:62–71. doi: 10.1097/01.blo.0000149996.91974.83. [DOI] [PubMed] [Google Scholar]
- 46.Nich C, Sariali E-H, Sari Ali E-H, Hannouche D, Nizard R, Witvoet J, Sedel L, Bizot P. Long-term results of alumina-on-alumina hip arthroplasty for osteonecrosis. Clin Orthop Relat Res. 2003:102–111. doi: 10.1097/01.blo.0000096820.67494.bf. [DOI] [PubMed] [Google Scholar]
- 47.Hamadouche M, Boutin P, Daussange J, Bolander ME, Sedel L. Alumina-on-alumina total hip arthroplasty: a minimum 18. 5-year follow-up study. J Bone Joint Surg Am. 2002;84:69–77. [PubMed] [Google Scholar]
- 48.Charnley J. The reaction of bone to self-curing acrylic cement. A long-term histological study in man. J Bone Joint Surg Br. 1970;52:340–353. [PubMed] [Google Scholar]
- 49.Maloney WJ, Jasty M, Burke DW, O’Connor DO, Zalenski EB, Bragdon C, Harris WH. Biomechanical and histologic investigation of cemented total hip arthroplasties. A study of autopsy-retrieved femurs after in vivo cycling. Clin Orthop Relat Res. 1989;6:129–140. [PubMed] [Google Scholar]
- 50.Maloney WJ, Jasty M, Rosenberg A, Harris WH. Bone lysis in well-fixed cemented femoral components. J Bone Joint Surg Br. 1990;72:966–970. doi: 10.1302/0301-620X.72B6.2246299. [DOI] [PubMed] [Google Scholar]
- 51.Willert HG, Bertram H, Buchhorn GH. Osteolysis in alloarthroplasty of the hip. The role of bone cement fragmentation. Clin Orthop Relat Res. 1990;258:108–121. [PubMed] [Google Scholar]
- 52.Goodman SB, Fornasier VL, Kei J. The effects of bulk versus particulate polymethylmethacrylate on bone. Clin Orthop Relat Res. 1988;232:255–262. [PubMed] [Google Scholar]
- 53.Goodman SB, Chin RC. Prostaglandin E2 levels in the membrane surrounding bulk and particulate polymethylmethacrylate in the rabbit tibia. A preliminary study. Clin Orthop Relat Res. 1990;257:305–309. [PubMed] [Google Scholar]
- 54.Moreau MF, Chappard D, Lesourd M, Monthéard JP, Baslé MF. Free radicals and side products released during methylmethacrylate polymerization are cytotoxic for osteoblastic cells. J Biomed Mater Res. 1998;40:124–131. doi: 10.1002/(sici)1097-4636(199804)40:1<124::aid-jbm14>3.0.co;2-o. [DOI] [PubMed] [Google Scholar]
- 55.Jones LC, Hungerford DS. Cement disease. Clin Orthop Relat Res. 1987;225:192–206. [PubMed] [Google Scholar]
- 56.Quinn J, Joyner C, Triffitt JT, Athanasou NA. Polymethylmethacrylate-induced inflammatory macrophages resorb bone. J Bone Joint Surg Br. 1992;74:652–658. doi: 10.1302/0301-620X.74B5.1527108. [DOI] [PubMed] [Google Scholar]
- 57.Huang Z, Ma T, Ren P-G, Smith RL, Goodman SB. Effects of orthopedic polymer particles on chemotaxis of macrophages and mesenchymal stem cells. J Biomed Mater Res A. 2010;94:1264–1269. doi: 10.1002/jbm.a.32803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Glant TT, Jacobs JJ, Molnar G, Shanbhag AS, Valyon M, Galante JO. Bone resorption activity of particulate-stimulated macrophages. J Bone Miner Res. 1993;8:1071–1079. doi: 10.1002/jbmr.5650080907. [DOI] [PubMed] [Google Scholar]
- 59.Pandey R, Quinn J, Joyner C, Murray DW, Triffitt JT, Athanasou NA. Arthroplasty implant biomaterial particle associated macrophages differentiate into lacunar bone resorbing cells. Ann Rheum Dis. 1996;55:388–395. doi: 10.1136/ard.55.6.388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sabokbar A, Pandey R, Quinn JM, Athanasou NA. Osteoclastic differentiation by mononuclear phagocytes containing biomaterial particles. Arch Orthop Trauma Surg. 1998;117:136–140. doi: 10.1007/s004020050213. [DOI] [PubMed] [Google Scholar]
- 61.Chiu R, Ma T, Smith RL, Goodman SB. Polymethylmethacrylate particles inhibit osteoblastic differentiation of bone marrow osteoprogenitor cells. J Biomed Mater Res A. 2006;77:850–856. doi: 10.1002/jbm.a.30697. [DOI] [PubMed] [Google Scholar]
- 62.Yaszay B, Trindade MC, Lind M, Goodman SB, Smith RL. Fibroblast expression of C-C chemokines in response to orthopedic biomaterial particle challenge in vitro. J Orthop Res. 2001;19:970–976. doi: 10.1016/S0736-0266(01)00003-1. [DOI] [PubMed] [Google Scholar]
- 63.Ramachandran R, Goodman SB, Smith RL. The effects of titanium and polymethylmethacrylate particles on osteoblast phenotypic stability. J Biomed Mater Res A. 2006;77:512–517. doi: 10.1002/jbm.a.30649. [DOI] [PubMed] [Google Scholar]
- 64.Antonios JK, Yao Z, Li C, Rao AJ, Goodman SB. Macrophage polarization in response to wear particles in vitro. Cell Mol Immunol. 2013;10:471–482. doi: 10.1038/cmi.2013.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Major MR, Wong VW, Nelson ER, Longaker MT, Gurtner GC. The foreign body response: At the interface of surgery and bioengineering. Plast Reconstr Surg. 2015;135:1489–1498. doi: 10.1097/PRS.0000000000001193. [DOI] [PubMed] [Google Scholar]
- 66.Goodman SB, Huie P, Song Y, Schurman D, Maloney W, Woolson S, Sibley R. Cellular profile and cytokine production at prosthetic interfaces. Study of tissues retrieved from revised hip and knee replacements. J Bone Joint Surg Br. 1998;80:531–539. doi: 10.1302/0301-620x.80b3.8158. [DOI] [PubMed] [Google Scholar]
- 67.Bostrom M, O’Keefe R. What experimental approaches (e.g., in vivo, in vitro, tissue retrieval) are effective in investigating the biologic effects of particles? J Am Acad Orthop Surg. 2008;16(Suppl 1):S63–S67. doi: 10.5435/00124635-200800001-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kadoya Y, Revell PA, al-Saffar N, Kobayashi A, Scott G, Freeman MA. Bone formation and bone resorption in failed total joint arthroplasties: Histomorphometric analysis with histochemical and immunohistochemical technique. J Orthop Res. 1996;14:473–482. doi: 10.1002/jor.1100140318. [DOI] [PubMed] [Google Scholar]
- 69.Nakashima Y, Sun DH, Trindade MC, Chun LE, Song Y, Goodman SB, Schurman DJ, Maloney WJ, Smith RL. Induction of macrophage C-C chemokine expression by titanium alloy and bone cement particles. J Bone Joint Surg Br. 1999;81:155–162. doi: 10.1302/0301-620x.81b1.8884. [DOI] [PubMed] [Google Scholar]
- 70.Shanbhag AS, Kaufman AM, Hayata K, Rubash HE. Assessing osteolysis with use of high-throughput protein chips. J Bone Joint Surg Am. 2007;89:1081–1089. doi: 10.2106/JBJS.F.00330. [DOI] [PubMed] [Google Scholar]
- 71.Wang C-T, Lin Y-T, Chiang B-L, Lee S-S, Hou S-M. Over-expression of receptor activator of nuclear factor-kappaB ligand (RANKL), inflammatory cytokines, and chemokines in periprosthetic osteolysis of loosened total hip arthroplasty. Biomaterials. 2010;31:77–82. doi: 10.1016/j.biomaterials.2009.09.017. [DOI] [PubMed] [Google Scholar]
- 72.Goldring SR, Jasty M, Roelke MS, Rourke CM, Bringhurst FR, Harris WH. Formation of a synovial-like membrane at the bone-cement interface. Its role in bone resorption and implant loosening after total hip replacement. Arthritis Rheum. 1986;29:836–842. doi: 10.1002/art.1780290704. [DOI] [PubMed] [Google Scholar]
- 73.Elliott MJ, Vadas MA, Cleland LG, Gamble JR, Lopez AF. IL-3 and granulocyte-macrophage colony-stimulating factor stimulate two distinct phases of adhesion in human monocytes. J Immunol. 1990;145:167–176. [PubMed] [Google Scholar]
- 74.McInnes A, Rennick DM. Interleukin 4 induces cultured monocytes/macrophages to form giant multinucleated cells. J Exp Med. 1988;167:598–611. doi: 10.1084/jem.167.2.598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.McNally AK, Anderson JM. Interleukin-4 induces foreign body giant cells from human monocytes/macrophages. Differential lymphokine regulation of macrophage fusion leads to morphological variants of multinucleated giant cells. Am J Pathol. 1995;147:1487–1499. [PMC free article] [PubMed] [Google Scholar]
- 76.al-Saffar N, Mah JT, Kadoya Y, Revell PA. Neovascularisation and the induction of cell adhesion molecules in response to degradation products from orthopedic implants. Ann Rheum Dis. 1995;54:201–208. doi: 10.1136/ard.54.3.201. [DOI] [PMC free article] [PubMed] [Google Scholar]