Abstract
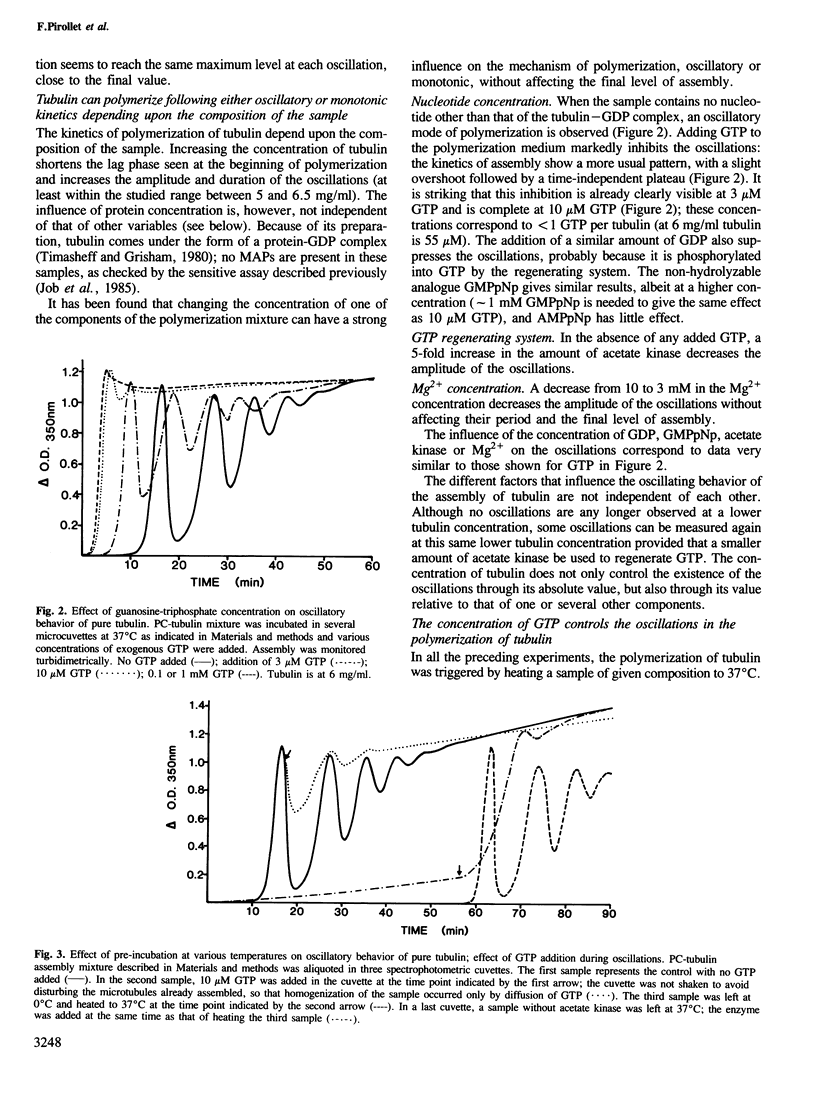
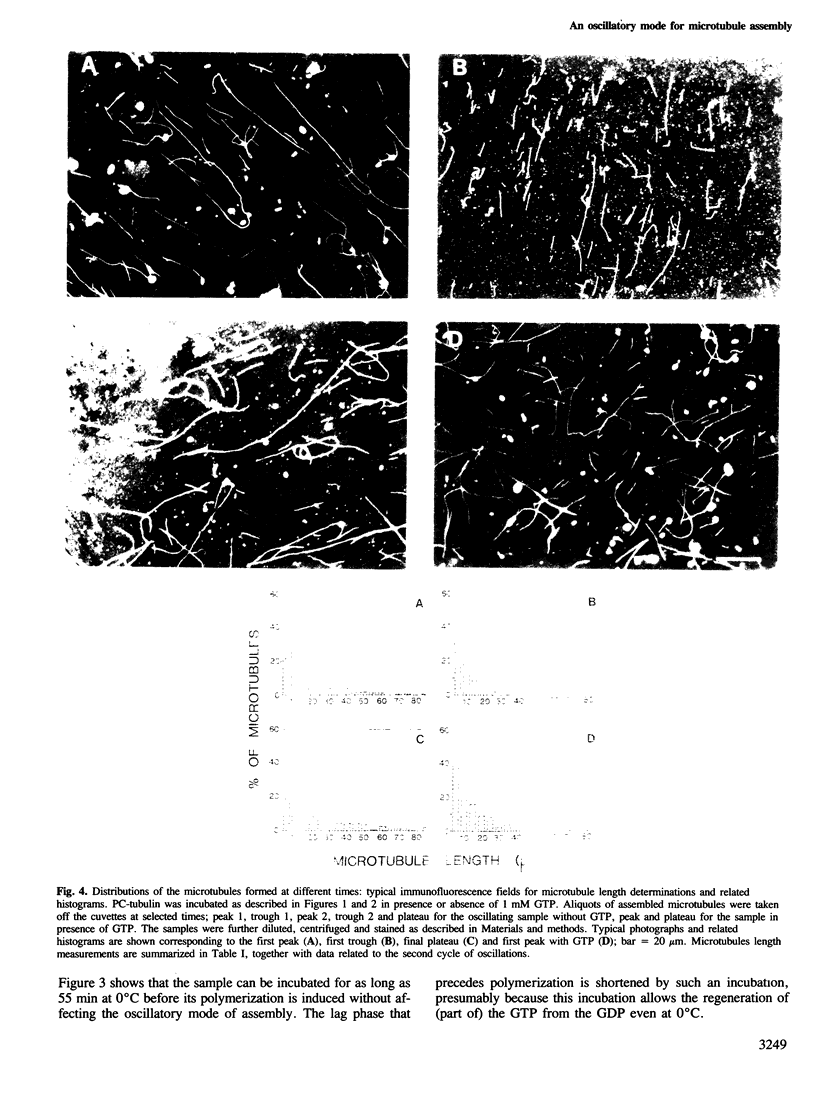
Depending upon the conditions under which polymerization takes place, pure tubulin can assemble into microtubules following either the usual monotonic kinetics or a more complex oscillatory mechanism. When present, these oscillations involve large cyclic changes in the extent of polymer formed before a steady-state is reached. Analysis of the microtubules formed at different times shows that these oscillations involve marked redistribution in both the length and number of microtubules. No significant difference is found between two populations of microtubules corresponding to the same level of assembly, one for which the extent of polymerization will remain stable with time and one for which it will decrease by as much as 90% in the next oscillation. The amplitude of these oscillations is sensitive to changes in the concentrations of protein, nucleotide (GTP, GDP or GMPpNp), magnesium ion or GTP regenerating system. A complete shift from an oscillatory to a monotonic polymerization can be induced by a minor increase in the concentration of free nucleotide, GTP or GDP.
Full text
PDF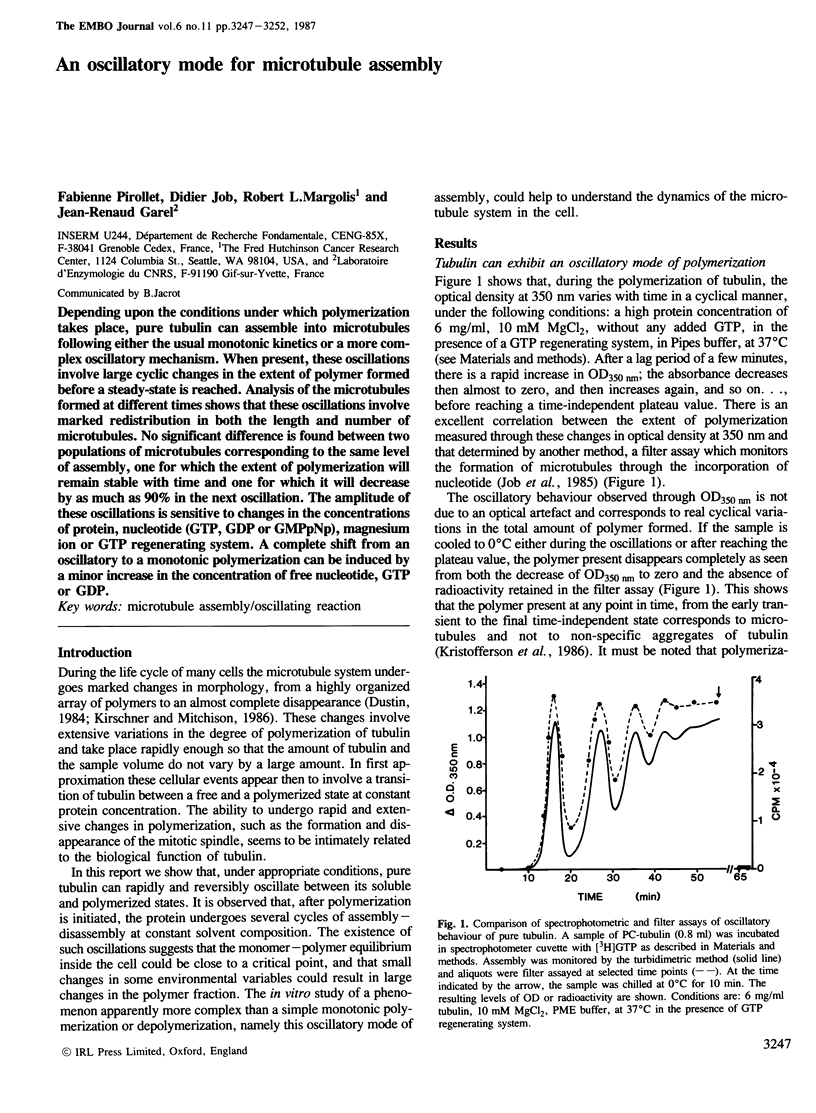



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Asnes C. F., Wilson L. Isolation of bovine brain microtubule protein without glycerol: polymerization kinetics change during purification cycles. Anal Biochem. 1979 Sep 15;98(1):64–73. doi: 10.1016/0003-2697(79)90706-1. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Caplow M., Shanks J., Brylawski B. P. Differentiation between dynamic instability and end-to-end annealing models for length changes of steady-state microtubules. J Biol Chem. 1986 Dec 5;261(34):16233–16240. [PubMed] [Google Scholar]
- Gaskin F., Cantor C. R., Shelanski M. L. Turbidimetric studies of the in vitro assembly and disassembly of porcine neurotubules. J Mol Biol. 1974 Nov 15;89(4):737–755. doi: 10.1016/0022-2836(74)90048-5. [DOI] [PubMed] [Google Scholar]
- Horio T., Hotani H. Visualization of the dynamic instability of individual microtubules by dark-field microscopy. Nature. 1986 Jun 5;321(6070):605–607. doi: 10.1038/321605a0. [DOI] [PubMed] [Google Scholar]
- Job D., Margolis R. L. Isolation from bovine brain of a superstable microtubule subpopulation with microtubule seeding activity. Biochemistry. 1984 Jun 19;23(13):3025–3031. doi: 10.1021/bi00308a028. [DOI] [PubMed] [Google Scholar]
- Job D., Pabion M., Margolis R. L. Generation of microtubule stability subclasses by microtubule-associated proteins: implications for the microtubule "dynamic instability" model. J Cell Biol. 1985 Nov;101(5 Pt 1):1680–1689. doi: 10.1083/jcb.101.5.1680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirschner M., Mitchison T. Beyond self-assembly: from microtubules to morphogenesis. Cell. 1986 May 9;45(3):329–342. doi: 10.1016/0092-8674(86)90318-1. [DOI] [PubMed] [Google Scholar]
- Kristofferson D., Mitchison T., Kirschner M. Direct observation of steady-state microtubule dynamics. J Cell Biol. 1986 Mar;102(3):1007–1019. doi: 10.1083/jcb.102.3.1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchison T., Kirschner M. Dynamic instability of microtubule growth. Nature. 1984 Nov 15;312(5991):237–242. doi: 10.1038/312237a0. [DOI] [PubMed] [Google Scholar]
- Mitchison T., Kirschner M. Microtubule assembly nucleated by isolated centrosomes. Nature. 1984 Nov 15;312(5991):232–237. doi: 10.1038/312232a0. [DOI] [PubMed] [Google Scholar]
- Penefsky H. S. Reversible binding of Pi by beef heart mitochondrial adenosine triphosphatase. J Biol Chem. 1977 May 10;252(9):2891–2899. [PubMed] [Google Scholar]
- Rothwell S. W., Grasser W. A., Murphy D. B. End-to-end annealing of microtubules in vitro. J Cell Biol. 1986 Feb;102(2):619–627. doi: 10.1083/jcb.102.2.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandoval I. V., MacDonald E., Jameson J. L., Cuatrecasas P. Role of nucleotides in tubulin polymerization: effect of guanylyl 5'-methylenediphosphonate. Proc Natl Acad Sci U S A. 1977 Nov;74(11):4881–4885. doi: 10.1073/pnas.74.11.4881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timasheff S. N., Grisham L. M. In vitro assembly of cytoplasmic microtubules. Annu Rev Biochem. 1980;49:565–591. doi: 10.1146/annurev.bi.49.070180.003025. [DOI] [PubMed] [Google Scholar]
- Weingarten M. D., Lockwood A. H., Hwo S. Y., Kirschner M. W. A protein factor essential for microtubule assembly. Proc Natl Acad Sci U S A. 1975 May;72(5):1858–1862. doi: 10.1073/pnas.72.5.1858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson L., Synder K. B., Thompson W. C., Margolis R. L. A rapid filtration assay for analysis of microtubule assembly, disassembly, and steady-state tubulin flux. Methods Cell Biol. 1982;24:159–169. doi: 10.1016/s0091-679x(08)60653-6. [DOI] [PubMed] [Google Scholar]




