Key Clinical Message
We evaluate the clinical findings and the treatment response of a late‐diagnosed case with a novel homozygous insertion c.13_14insG (p.P6Afs*202) result in a frameshift mutation in MLYCD gene. Both cardiac and neurologic involvements were mild when compared to previously reported cases, and see low‐fat/high‐carbohydrate diet treatment is highly effective.
Keywords: Diet therapy, malonyl‐CoA decarboxylase deficiency, MLYCD gene, noncompaction cardiomyopathy
Introduction
Malonic aciduria (MIM 248360) is a rare autosomal recessive disorder caused by deficiency of malonyl‐CoA decarboxylase (MLYCD, EC 4.1.1.9), encoded by the MLYCD gene on chromosome 16q24 (MIM 606761) 1, 2. MLYCD catalyzes the decarboxylation of malonyl‐CoA to acetyl‐CoA and has an important role in fatty acid metabolism both in mitochondria and peroxisomes 3. Increased malonyl‐CoA levels resulting from the MLYCD deficiency inhibit long‐chain carnitine acyltransferases and Krebs cycle 3, 4 (Fig. 1).
Figure 1.
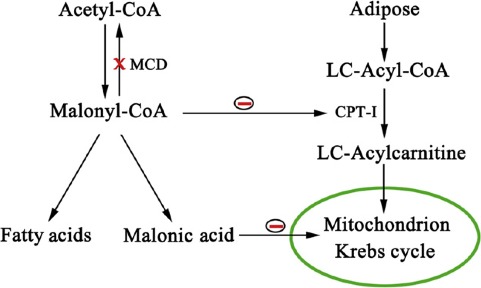
Pathways of malonyl‐CoA and effects on fatty acid metabolism and Krebs cycle.
Malonic acid is most commonly present in heart muscle more than skeletal muscle, brain, intestine, liver, and kidney 5. Thus, MLYCD is characterized by varying severity of cardiomyopathy, muscle weakness, developmental delay, seizures, short structure, hypoglycemia, and metabolic acidosis 6, 7.
The diagnosis of malonic aciduria is based on high urinary excretion of malonic, methylmalonic, methylcitric acids in addition to mild increases in dicarboxylic acids and elevated levels of malonylcarnitine (C3DC) in tandem mass spectrometry. Early diagnosis is important to prevent and treat cardiomyopathy which is the main cause of morbidity and mortality. Similar to fatty acid oxidation disorders, a high‐carbohydrate and low‐fat diet is recommended for treatment 8, 9.
The diet should consist of total energy to support normal weight gain for infants and children and maintain appropriate weight for height in adults: 10–12% of total energy from protein and 30% of total energy from fat (30% long‐chain triglycerides – LCTs; 70% medium‐chain triglycerides – MCTs). To avoid the lack of essential fatty acid (EFA) deficiency, the diet should include linoleic and α‐linolenic acid. The rest of total energy (~ 60–65%) should be carbohydrate.
The MLYCD gene is located on chromosome 16 and has five coding exons. Several different mutations have been described including missense, nonsense, small insertions, and deletions, as well as large genomic deletions.
Here, we report a one‐year‐old boy's clinical and biochemical findings with a novel homozygous MLYCD mutation and discuss the treatment and follow‐up under the highlight of the literature.
Case Report
A one‐year‐old boy was referred to the hospital with failure to thrive and neuromotor development delay. He was the first child of consanguineous parents with an unremarkable family history. He was born at term following an uneventful pregnancy and delivery. Birth weight was 3000 g. He had phototherapy for physiological jaundice at postnatal fourth day and experienced no additional problem. He had no hypoglycemia or acidosis in his medical history.
On physical examination, his weight was under 3rd percentile, height was between 3rd and 10th percentile, and head circumference was 50th percentile. He was mildly hypotonic and exhibited a mild delay in language and motor development with normal social development. He was sitting without support for 15 days.
Biochemical investigations were not diagnostic. On his metabolic screening, free carnitine was 12.3 μmol/L (normal range: 8.6–90 μmol/L) and C3DC was 3.78 μmol/L (normal range: 0–0.6 μmol/L). Urine organic acids showed marked elevation of malonic acid (824 mg/g creatinine; normal range: <30 mg/g creatinine) and methylmalonic acid (124 mg/g creatinine; normal range: <50 mg/g creatinine) and mild elevation of methylcitric, adipic, suberic, sebacic, and ethylmalonic acids (41, 65, 58, 22, 15 mg/g creatinine; normal range: <22, <15, <20, <22, <10 mg/g creatinine, respectively). Homocysteine, folate and vitamin B12 levels were all within normal ranges.
Echocardiogram revealed left ventricular noncompaction cardiomyopathy characterized by deep trabeculation of the left ventricular posterior wall and apex. The heart muscle lost its normal shape and was mildly dilated. Cardiac function was normal with ejection fraction of 57% (Fig. 2A and B).
Figure 2.
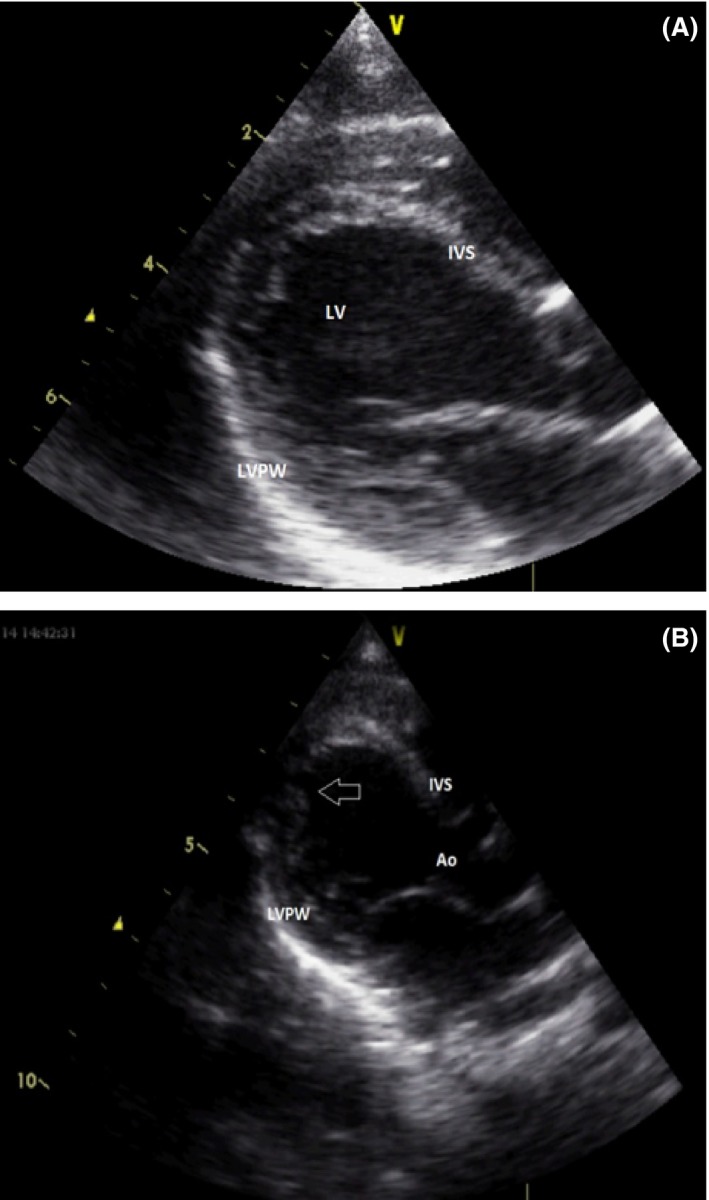
Echocardiography: (A) The glob shape of heart. (B) The trabeculation at left ventricle mainly at apex. Ao, aorta; IVS, interventricular septum; LVPW, left ventricular posterior wall; LV, left ventricle.
Magnetic resonance imaging of the brain showed hyperintensity at bilateral dentate nucleus and global cerebral atrophy and periventricular hyperintensity and delayed myelination (Fig. 3A and B).
Figure 3.
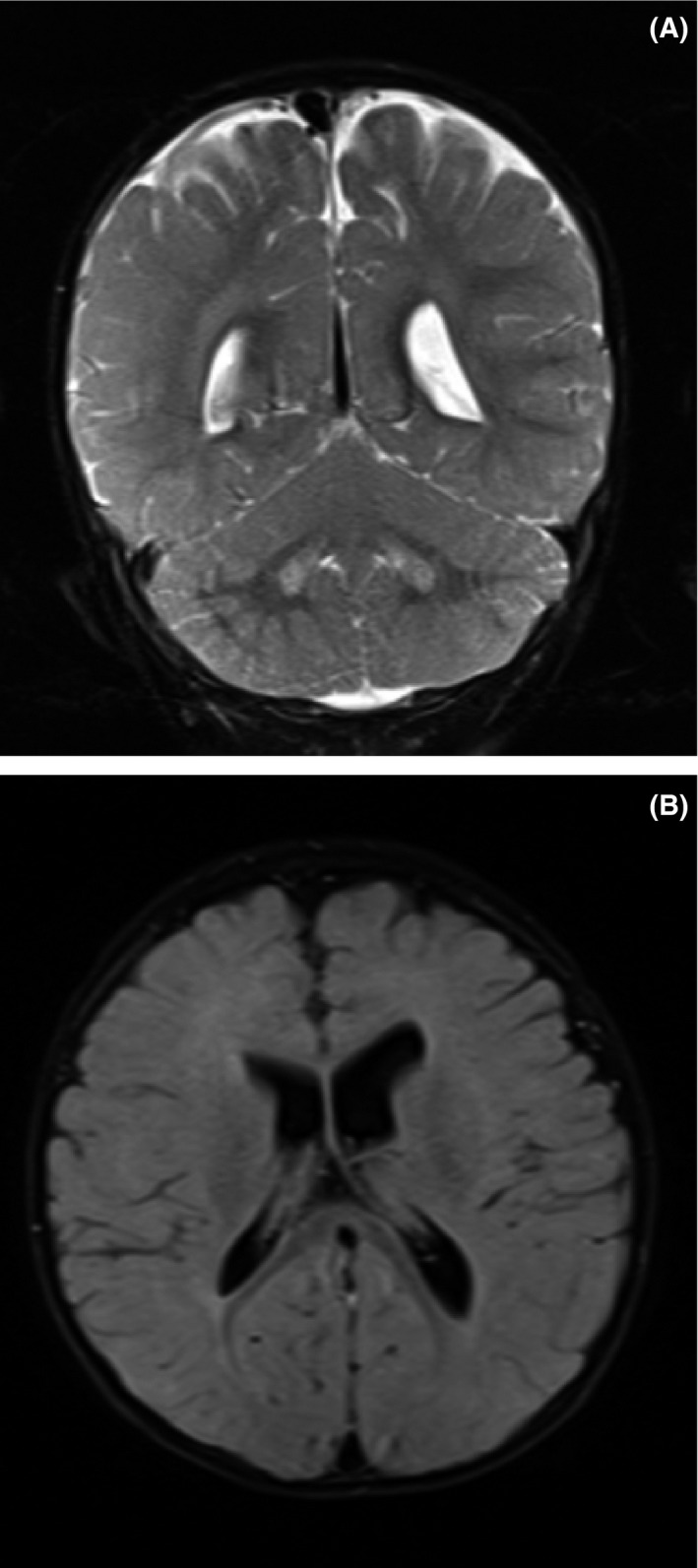
Magnetic resonance imaging: (A) Hyperintensity at bilateral dentate nucleus. (B) Global cerebral atrophy; periventricular hyperintensity; delayed myelination.
MLYCD gene sequence analysis was performed with MiSeq next‐generation sequencing (NGS) platform (Illumina, San Diego, CA). Genomic DNA isolation was performed according to the manufacturer's standard procedure with Anatolia magnetic bead kit (Anatolia, Turkey). Coding exons and flanking regions were amplified with in‐house designed PCR primers, designed with PRIMER© – Primer Designer v.2.0 (Scientific & Educational Software programme: Denver, CO, USA) software. Library preparation was carried out with the Nextera XT kit (Illumina Inc.: San Diego, CA, USA). Sequences were aligned to the hg19 genome within MiSeq Reporter software (Illumina Inc.: San Diego, CA, USA) and visualized with IGV 2.3 (Broad Institute: Cambridge, MA, USA) software.
This study showed a novel homozygous insertion NM_012213: c.13_14insG (p. P6Afs*202) result in a frameshift mutation.
We could not found this variant in the literature. In silico analysis with MutationTaster software predicted this variant as a disease‐causing mutation (prob: 1). As this insertion was happened at very first part of the gene, this variant most probably causes a severe damage on protein and its function. This variant was not present in 1250 clinical exome sequencing data of our own patients. It was reported as a likely pathogenic variant. Functional studies are needed for exact decision.
The patient was started on levocarnitine 50 mg/kg/day twice daily, and high doses of carnitine were avoided similar to fatty acid oxidation disorders. Carnitine dose was adjusted according to the carnitine profile. A high‐carbohydrate/low‐fat diet (30% long‐chain triglycerides – LCTs; 70% MCTs) was started. Fish oil was supplemented at a dose of 100 mg/kg/day to provide essential fatty acid. Anticongestive therapy was not required. In the early days of the treatment, due to the unpleasant taste he refused to consume the MCTs. He soon began to thrive. At the end of the 6 month of the treatment, he had gained weight and arrived at the 3rd‐10th percentile. He began pulling to stand and walked for several seconds. There was no deterioration in the trabeculation of left ventricle and myocardial function. Ejection fraction remained at 58%.
Malonic acid level decreased to 396 mg/g creatinine (normal range: <30 mg/g creatinine), and methyl malonic acid and methylcitrate were 56 and 27 mg/g creatinine, respectively. The metabolites of the Krebs cycle disappeared. CD3DC decreased to 1.06 μmol/L (normal range: 0–0.6 μmol/L). He continued his treatment on high‐MCT diet and levocarnitine.
Discussion
The clinical findings of malonic aciduria due to malonyl‐CoA decarboxylase deficiency involve mainly heart and skeletal muscles and the central nervous system. This is described by accumulation of malonyl‐CoA, which is a potent inhibitor of carnitine palmitoyltransferase‐I (CPT‐I), causing impaired beta‐oxidation of fatty acids in both mitochondria and peroxisomes 3, 4. CPT‐I enzymes that are expressed in the heart, skeletal muscle and brain have the highest affinity to malonyl‐CoA 5.
Cardiomyopathy has been reported in up to 40% of patients with MLYD, similar to fatty acid oxidation disorders 7, 10. Together with dilated and hypertrophic types, noncompaction cardiomyopathy is common in MLYCD. Left ventricular noncompaction (LVNC) is a clinical heterogeneous disorder characterized anatomically by deep trabeculations in the ventricular wall, and it is associated with ventricular dysfunction, arrhythmias, and embolic events 11, 12. Although it has been defined as an arrest of myocardial maturation during embryogenesis, LVNC can occur in the course of metabolic disease with an unexplained mechanism 8. MLYCD is necessary for normal sarcomere function and embryological compaction of the left ventricular myocardium 13. MCD plays different pleiotropic roles in pathways in mitochondria and peroxisome 14, 15. Echocardiograms of several patients identified by newborn screening were normal 16. Interestingly, trabeculation was at the apex and limited part of the left ventricular posterior wall of the heart in this case. The structure of the other ventricular wall was normal. It is difficult to say whether the LVNC was congenital or acquired in our patient. Serial cardiac evaluations in patients identified by newborn screening can highlight the pathogenesis and the course of these complications in MLYCD. The factors that determine the severity of the affected heart muscle tissues are unclear. Heart muscle of our case was affected slightly.
Treatment with carnitine supplementation together with a low‐fat, high‐MCT, and high‐carbohydrate diet halts of cardiomyopathy 8, 9, 17. Medium‐chain triglycerides (MCTs) should be at least 70% of the total fat content of the diet. Reducing the dose of MCTs or dissonance of the treatment can cause clinical worsening. The patient's diet must be absolute, and they should be screened for complications such as cardiomyopathy, including patients identified by NBS 18.
During 6 months under the therapy, we observed that the structure of the myocardium did not exacerbate.
Hypotony depends on both central nervous system involvement and muscle weakness. It can be said that the diet therapy also has a positive effect on skeletal muscles. There is insufficient data concerning the effects of diet therapy on neuromotor development in published literature.
Both white and gray matter involvement was demonstrated together or alone in malonic aciduria 6, 7, 8. Subcortical white matter intensity alterations, delayed myelination, cortical pachygyria, polymicrogyria, and periventricular heterotopia were reported in several cases 19, 20. Elevated malonyl‐CoA level can disturb neuronal migration and cause cortical dysplasia. Generalized cerebral and cerebellar atrophy is one of the prominent imaging findings 6, 21. High intensities in the basal ganglions were reported in some patients 6. To the best of our knowledge, dentate nucleus hyperintensity has not been reported previously in MLYCD. Dentate nucleus involvement is one of the neuroimaging findings of mitochondrial impairment, secondary disturbance of mitochondrial dysfunction as seen in organic acidemias and cerebrotendinous xanthomatosis 22, 23, 24. There is additional evidence indicating MLYCD may cause mitochondrial dysfunction 25.
Elevated malonyl‐CoA level can cause growth retardation in utero and also during infancy 26. Besides short structure, failure to thrive appears to be one of the clinical outcomes of MLYCD (unpublished data). Although the birthweight of our case was normal, his weight gain halted by the time. He began to gain weight after adjusting to the taste of MCT oil.
Conclusion
Although malonic aciduria is a rare inherited metabolic disease, diet therapy can prevent complications in cases identified by newborn screening and facilitate improvement in late‐diagnosed cases. In this report, we evaluate the clinical findings and the treatment response of a late‐diagnosed case with a novel mutation. Both cardiac and neurologic involvements were mild when compared to previously reported cases. The treatment is highly effective.
The definition of natural course, genotype–phenotype relationship, and the effect of treatment should be the subject of further studies.
Authorship
ME: contributed to conception, design, and interpretation, and drafted and clinically revised the manuscript. MBA: contributed to conception, design, and interpretation. SC: contributed to conception, design, and analysis. NCB: contributed to conception, design, and interpretation, and drafted and clinically revised the manuscript.
Conflict of Interest
None declared.
References
- 1. FitzPatrick, D. R. , Hill A., Tolmie J. L., Thorburn D. R., and Christodoulou J.. 1999. The molecular basis of malonyl‐CoA decarboxylase deficiency. Am. J. Hum. Genet. 65:318–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Wightman, P. J. , Santer R., Ribes A., Dougherty F., McGill N., Thorburn D. R., et al. 2003. MLYCD mutation analysis: evidence for protein mistargeting as a cause of MLYCD deficiency. Hum. Mutat. 22:288–300. [DOI] [PubMed] [Google Scholar]
- 3. Bennett, M. J. , Harthcock P. A., Boriack R. L., and Cohen J. C.. 2001. Impaired mitochondrial fatty acid oxidative flux in fibroblasts from a patient with malonyl‐CoA decarboxylase deficiency. Mol. Genet. Metab. 73:276–279. [DOI] [PubMed] [Google Scholar]
- 4. McGarry, J. D. , and Brown N. F.. 1997. The mitochondrial carnitine palmitoyltransferase system. From concept to molecular analysis. Eur. J. Biochem. 244:1–14. [DOI] [PubMed] [Google Scholar]
- 5. Price, N. , van der Leij F., Jackson V., Corstorphine C., Thomson R., Sorensen A., et al. 2002. A novel brain‐expressed protein related to carnitine palmitoyltransferase I. Genomics 80:433. [DOI] [PubMed] [Google Scholar]
- 6. Ozand, P. T. , Nyhan W. L., al Aqeel A., and Christodoulou J.. 1994. Malonic aciduria. Brain Dev. 16(S):7–11. [DOI] [PubMed] [Google Scholar]
- 7. Salomons, G. S. , Jakobs C., Pope L. L., Errami A., Potter M., Nowaczyk M., et al. 2007. Clinical, enzymatic and molecular characterization of nine new patients with malonyl‐coenzyme A decarboxylase deficiency. J. Inherit. Metab. Dis. 30:23–28. [DOI] [PubMed] [Google Scholar]
- 8. Ficicioglu, C. , Chrisant M. R., Payan I., and Chace D. H.. 2005. Cardiomyopathy and hypotonia in a 5‐month‐old infant with malonyl‐Coa decarboxylase deficiency: potential for preclinical diagnosis with expanded newborn screening. Pediatr. Cardiol. 26:881–883. [DOI] [PubMed] [Google Scholar]
- 9. Footitt, E. J. , Stafford J., Dixon M., Burch M., Jakobs C., Salomons G. S., et al. 2010. Use of a long‐chain triglyceride‐restricted/medium‐chain triglyceride‐supplemented diet in a case of malonyl‐CoA decarboxylase deficiency with cardiomyopathy. J. Inherit. Metab. Dis. 33 Suppl 3:S253–256. [DOI] [PubMed] [Google Scholar]
- 10. Yano, S. , Sweetman L., Thorburn D. R., Mofidi S., and Williams J. C.. 1997. A new case of malonyl coenzyme A decarboxylase deficiency presenting with cardiomyopathy. Eur. J. Pediatr. 156:382–383. [DOI] [PubMed] [Google Scholar]
- 11. Lorsheyd, A. , Cramer M. J., Velthuis B. K., Vonken E. J., van der Smagt J., van Tintelen P., et al. 2006. Familial occurrence of isolated non‐compaction cardiomyopathy. Eur. J. Heart Fail. 8:826–831. [DOI] [PubMed] [Google Scholar]
- 12. Towbin, J. A. 2010. Left ventricular noncompaction: a new form of heart failure. Heart Fail. Clin. 6:453–469. [DOI] [PubMed] [Google Scholar]
- 13. Richardson, P. , McKenna W., Bristow M., Maisch B., Mautner B., O'Connell J., et al. 1996. Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the Definition and Classification of cardiomyopathies. Circulation 93:841–842. [DOI] [PubMed] [Google Scholar]
- 14. Dyck, J. R. , Hopkins T. A., Bonnet S., Michelakis E. D., Young M. E., Watanabe M., et al. 2006. Absence of malonyl coenzyme A decarboxylase in mice increases cardiac glucose oxidation and protects the heart from ischemic injury. Circulation 114:1721–1728. [DOI] [PubMed] [Google Scholar]
- 15. Joly, E. , Bendayan M., Roduit R., Saha A. K., Ruderman N. B., and Prentki M., 2005. Malonyl‐CoA decarboxylase is present in the cytosolic, mitochondrial and peroxisomal compartments of rat hepatocytes. FEB Lett. 579:6581–6586. [DOI] [PubMed] [Google Scholar]
- 16. Prada, C. E. , Jefferies J. L., Grenier M. A., Huth C. M., Page K. I., Spicer R. L., et al. 2012. Malonyl coenzyme A decarboxylase deficiency: early dietary restriction and time course of cardiomyopathy. Pediatrics 130:456–460. [DOI] [PubMed] [Google Scholar]
- 17. Celato, A. , Mitola C., Tolve M., Giannini M. T., De Leo S., Carducci C., et al. 2013. A new case of malonic aciduria with a presymptomatic diagnosis and an early treatment. Brain Dev. 35:675–680. [DOI] [PubMed] [Google Scholar]
- 18. Baertling, F. , Mayatepek E., Thimm E., Schlune A., Kovacevic A., Distelmaier F., et al. 2014. Malonic aciduria: long‐term follow‐up of new patients detected by newborn screening. Eur. J. Pediatr. 173:1719–1722. [DOI] [PubMed] [Google Scholar]
- 19. Malvagia, S. , Papi L., Morrone A., Donati M. A., Ciani F., Pasquini E., et al. 2007. Fatal malonyl Co‐A decarboxylase deficiency due to maternal uniparental isodisomy of the telomeric end of chromosome 16. Ann. Hum. Genet. 71:705–712. [DOI] [PubMed] [Google Scholar]
- 20. Xue, J. , Peng J., Zhou M., Zhong L., Yin F., Liang D., et al. 2012. Novel compound heterozygous mutation of MLYCD in a Chinese patient with malonic aciduria. Mol. Genet. Metab. 105:79–83. [DOI] [PubMed] [Google Scholar]
- 21. Krawinkel, M. B. , Oldigs H. D., Santer R., Lehnert W., Wendel U., and Schaub J., 1994. Association of malonyl‐CoA decarboxylase deficiency and heterozygote state for hemoglobin C disease. J. Inherit. Metab. Dis. 17:288–300. [DOI] [PubMed] [Google Scholar]
- 22. Barth, P. G. , Hoffmann G. F., Jaeken J., Lehnert W., Hanefeld F., van Gennip A. H., et al. 1992. L‐2‐hydroxyglutaric acidemia: a novel inherited neurometabolic disease. Ann. Neurol. 32:66–71. [DOI] [PubMed] [Google Scholar]
- 23. Kinghorn, K. J. , Kaliakatsos M., Blakely E. L., Taylor R. W., Rich P., Clarke A., et al. 2013. Hypertrophic olivary degeneration on magnetic resonance imaging in mitochondrial syndromes associated with POLG and SURF1 mutations. J. Neurol. 260:3–9. [DOI] [PubMed] [Google Scholar]
- 24. Nie, S. , Chen G., Cao X., and Zhang Y.. 2014. Cerebrotendinous xanthomatosis: a comprehensive review of pathogenesis, clinical manifestations, diagnosis, and management. Orphanet. J. Rare. Dis. 9:179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Cavanagh, J. B. , and Harding B. N.. 1994. Pathogenic factors underlying the lesions in Leigh's disease. Tissue responses to cellular energy deprivation and their clinico‐pathological consequences. Brain 117:1357–1376. [DOI] [PubMed] [Google Scholar]
- 26. Benn, P. 1998. Trisomy 16 and trisomy 16 mosaicism: a review. Am. J. Med. Genet. 79:121–123. [PubMed] [Google Scholar]


