Abstract
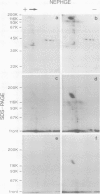
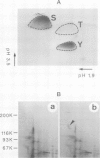
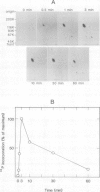
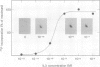
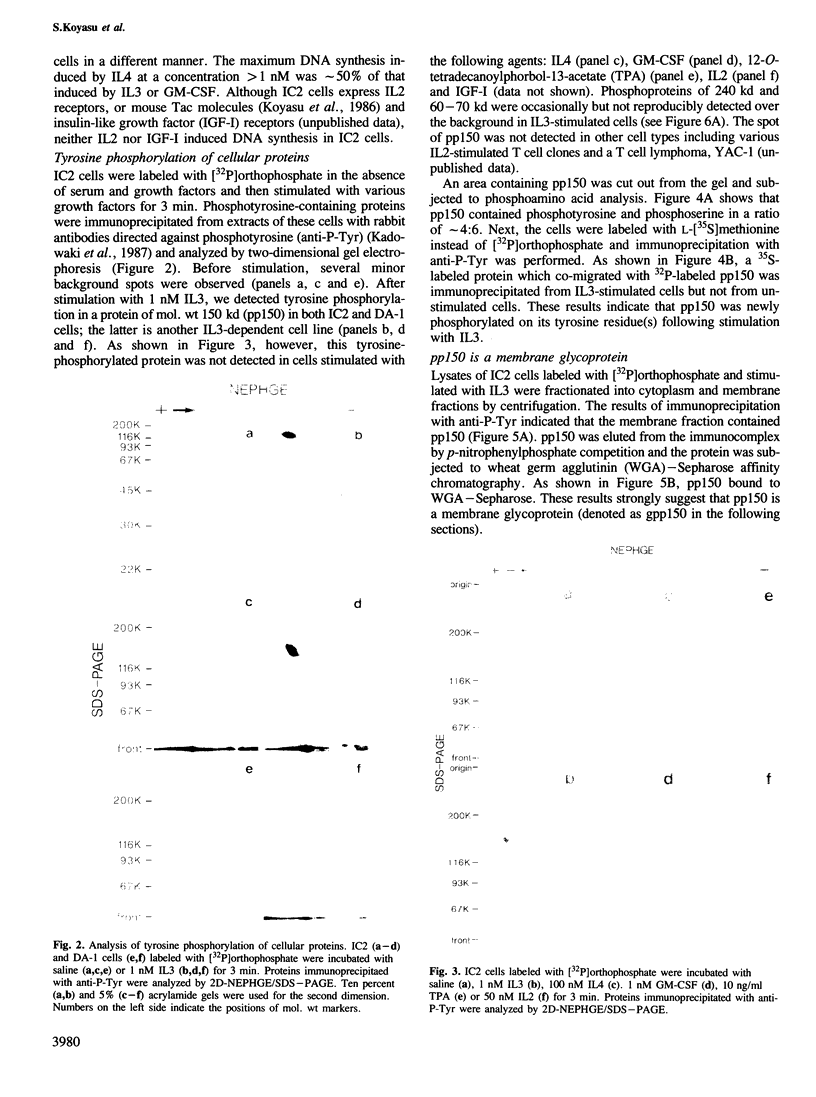
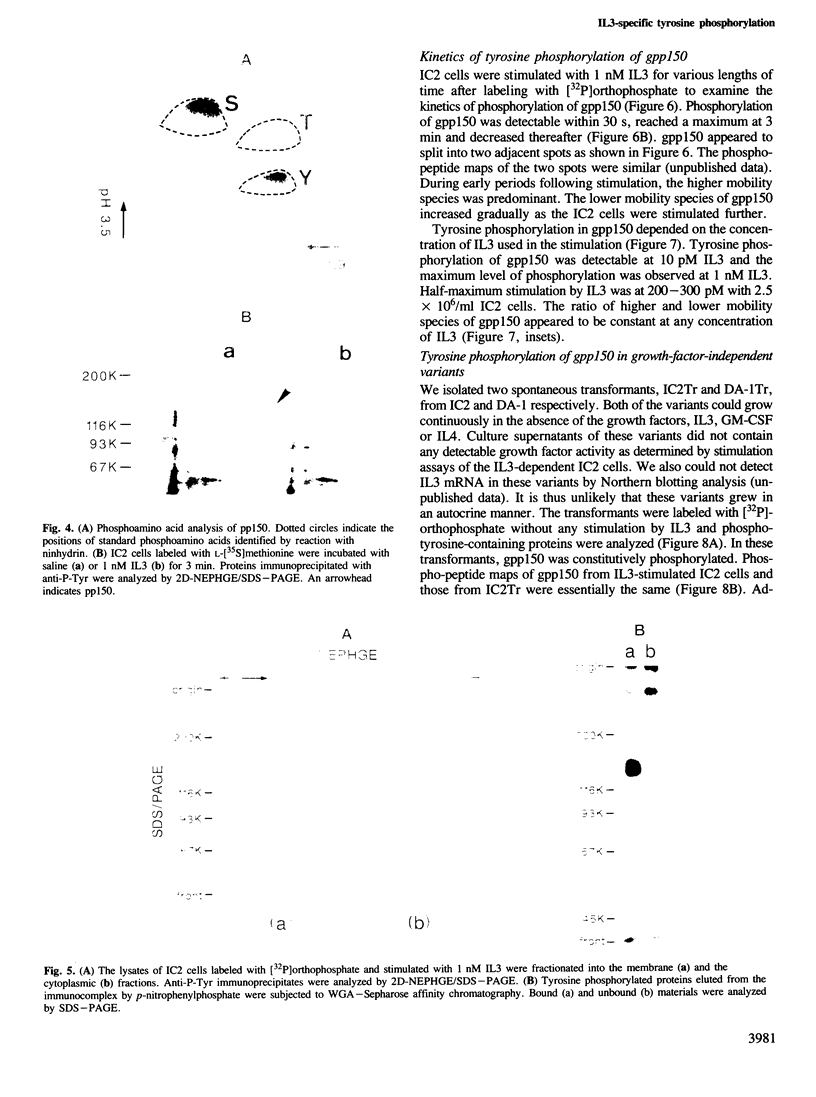
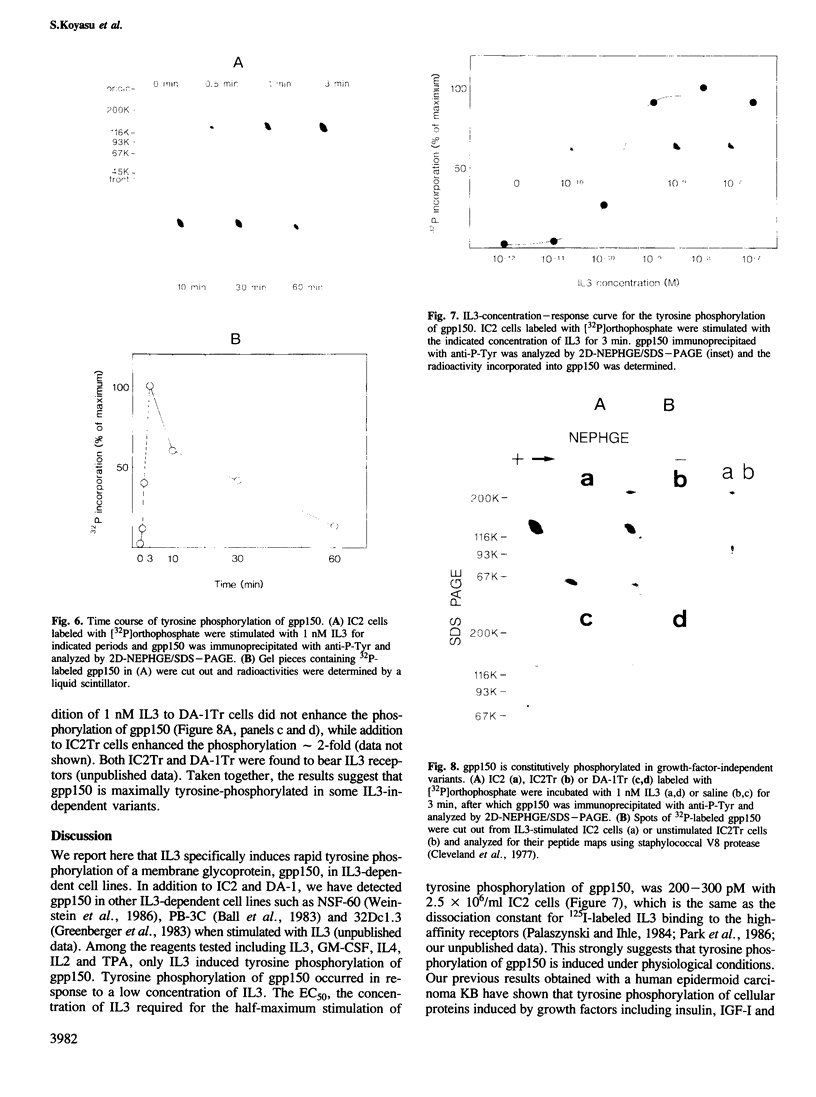
Tyrosine phosphorylation of cellular proteins induced by various hematopoietic growth factors such as interleukin 3 (IL3), granulocyte-macrophage colony-stimulating factor (GM-CSF) and interleukin 4 (IL4) was studied in several multi-factor-dependent myeloid cell lines. Among the growth factors, IL3 specifically induced rapid tyrosine phosphorylation of a membrane glycoprotein of mol. wt 150 kd (gpp150) in the IL3-dependent cell lines, IC2 and DA-1. The IL3-induced tyrosine phosphorylation of gpp150 was detected within 30 s, reached a maximum at 3 min and decreased thereafter. The concentration of IL3 required for half-maximum stimulation of gpp150 tyrosine phosphorylation with 2.5 x 10(6)/ml cells was approximately 200 pM, which is the same as the dissociation constant for 125I-labeled IL3 binding. gpp150 was constitutively phosphorylated on tyrosine residue(s) in growth factor independent variants, IC2Tr and DA-1Tr, derived from IC2 and DA-1 respectively. Neither variant synthesized IL3. The present findings suggest that tyrosine phosphorylation of gpp150 is a critical event involved in both IL3-dependent and -independent growth.
Full text
PDF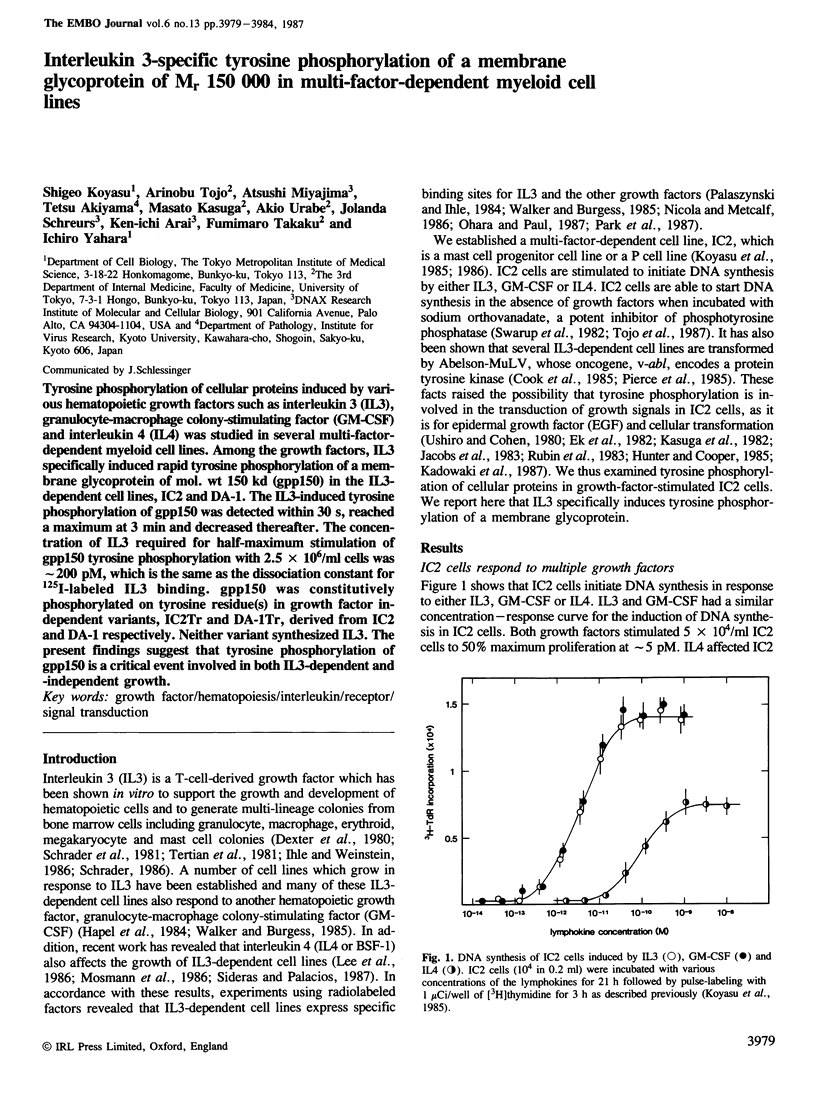


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akiyama T., Sudo C., Ogawara H., Toyoshima K., Yamamoto T. The product of the human c-erbB-2 gene: a 185-kilodalton glycoprotein with tyrosine kinase activity. Science. 1986 Jun 27;232(4758):1644–1646. doi: 10.1126/science.3012781. [DOI] [PubMed] [Google Scholar]
- Ball P. E., Conroy M. C., Heusser C. H., Davis J. M., Conscience J. F. Spontaneous, in vitro, malignant transformation of a basophil/mast cell line. Differentiation. 1983;24(1):74–78. doi: 10.1111/j.1432-0436.1983.tb01305.x. [DOI] [PubMed] [Google Scholar]
- Cleveland D. W., Fischer S. G., Kirschner M. W., Laemmli U. K. Peptide mapping by limited proteolysis in sodium dodecyl sulfate and analysis by gel electrophoresis. J Biol Chem. 1977 Feb 10;252(3):1102–1106. [PubMed] [Google Scholar]
- Cook W. D., Metcalf D., Nicola N. A., Burgess A. W., Walker F. Malignant transformation of a growth factor-dependent myeloid cell line by Abelson virus without evidence of an autocrine mechanism. Cell. 1985 Jul;41(3):677–683. doi: 10.1016/s0092-8674(85)80048-9. [DOI] [PubMed] [Google Scholar]
- Dexter T. M., Garland J., Scott D., Scolnick E., Metcalf D. Growth of factor-dependent hemopoietic precursor cell lines. J Exp Med. 1980 Oct 1;152(4):1036–1047. doi: 10.1084/jem.152.4.1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ek B., Heldin C. H. Use of an antiserum against phosphotyrosine for the identification of phosphorylated components in human fibroblasts stimulated by platelet-derived growth factor. J Biol Chem. 1984 Sep 10;259(17):11145–11152. [PubMed] [Google Scholar]
- Ek B., Westermark B., Wasteson A., Heldin C. H. Stimulation of tyrosine-specific phosphorylation by platelet-derived growth factor. Nature. 1982 Feb 4;295(5848):419–420. doi: 10.1038/295419a0. [DOI] [PubMed] [Google Scholar]
- Evans S. W., Rennick D., Farrar W. L. Multilineage hematopoietic growth factor interleukin 3 and direct activators of protein kinase C stimulate phosphorylation of common substrates. Blood. 1986 Oct;68(4):906–913. [PubMed] [Google Scholar]
- Greenberger J. S., Eckner R. J., Sakakeeny M., Marks P., Reid D., Nabel G., Hapel A., Ihle J. N., Humphries K. C. Interleukin 3-dependent hematopoietic progenitor cell lines. Fed Proc. 1983 Jul;42(10):2762–2771. [PubMed] [Google Scholar]
- Hapel A. J., Warren H. S., Hume D. A. Different colony-stimulating factors are detected by the "interleukin-3"-dependent cell lines FDC-Pl and 32D cl-23. Blood. 1984 Oct;64(4):786–790. [PubMed] [Google Scholar]
- Haring H. U., Kasuga M., White M. F., Crettaz M., Kahn C. R. Phosphorylation and dephosphorylation of the insulin receptor: evidence against an intrinsic phosphatase activity. Biochemistry. 1984 Jul 3;23(14):3298–3306. doi: 10.1021/bi00309a028. [DOI] [PubMed] [Google Scholar]
- Hunter T., Cooper J. A. Protein-tyrosine kinases. Annu Rev Biochem. 1985;54:897–930. doi: 10.1146/annurev.bi.54.070185.004341. [DOI] [PubMed] [Google Scholar]
- Ihle J. N., Weinstein Y. Immunological regulation of hematopoietic/lymphoid stem cell differentiation by interleukin 3. Adv Immunol. 1986;39:1–50. doi: 10.1016/s0065-2776(08)60347-8. [DOI] [PubMed] [Google Scholar]
- Ishii T., Sugamura K., Nakamura M., Hinuma Y. Interleukin 2 (IL-2) rapidly induces phosphorylation of a cellular protein, pp67, in an IL-2 dependent murine cell line. Biochem Biophys Res Commun. 1986 Mar 13;135(2):487–494. doi: 10.1016/0006-291x(86)90020-3. [DOI] [PubMed] [Google Scholar]
- Jacobs S., Kull F. C., Jr, Earp H. S., Svoboda M. E., Van Wyk J. J., Cuatrecasas P. Somatomedin-C stimulates the phosphorylation of the beta-subunit of its own receptor. J Biol Chem. 1983 Aug 25;258(16):9581–9584. [PubMed] [Google Scholar]
- Kadowaki T., Koyasu S., Nishida E., Tobe K., Izumi T., Takaku F., Sakai H., Yahara I., Kasuga M. Tyrosine phosphorylation of common and specific sets of cellular proteins rapidly induced by insulin, insulin-like growth factor I, and epidermal growth factor in an intact cell. J Biol Chem. 1987 May 25;262(15):7342–7350. [PubMed] [Google Scholar]
- Kasuga M., Zick Y., Blithe D. L., Crettaz M., Kahn C. R. Insulin stimulates tyrosine phosphorylation of the insulin receptor in a cell-free system. Nature. 1982 Aug 12;298(5875):667–669. doi: 10.1038/298667a0. [DOI] [PubMed] [Google Scholar]
- Koyasu S., Nakauchi H., Kitamura K., Yonehara S., Okumura K., Tada T., Yahara I. Production of interleukin 3 and gamma-interferon by an antigen-specific mouse suppressor T cell clone. J Immunol. 1985 May;134(5):3130–3136. [PubMed] [Google Scholar]
- Koyasu S., Yodoi J., Nikaido T., Tagaya Y., Taniguchi Y., Honjo T., Yahara I. Expression of interleukin 2 receptors on interleukin 3-dependent cell lines. J Immunol. 1986 Feb 1;136(3):984–987. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lee F., Yokota T., Otsuka T., Meyerson P., Villaret D., Coffman R., Mosmann T., Rennick D., Roehm N., Smith C. Isolation and characterization of a mouse interleukin cDNA clone that expresses B-cell stimulatory factor 1 activities and T-cell- and mast-cell-stimulating activities. Proc Natl Acad Sci U S A. 1986 Apr;83(7):2061–2065. doi: 10.1073/pnas.83.7.2061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May W. S., Ihle J. N. Affinity isolation of the interleukin-3 surface receptor. Biochem Biophys Res Commun. 1986 Mar 28;135(3):870–879. doi: 10.1016/0006-291x(86)91009-0. [DOI] [PubMed] [Google Scholar]
- Miyajima A., Otsu K., Schreurs J., Bond M. W., Abrams J. S., Arai K. Expression of murine and human granulocyte-macrophage colony-stimulating factors in S. cerevisiae: mutagenesis of the potential glycosylation sites. EMBO J. 1986 Jun;5(6):1193–1197. doi: 10.1002/j.1460-2075.1986.tb04346.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyatake S., Otsuka T., Yokota T., Lee F., Arai K. Structure of the chromosomal gene for granulocyte-macrophage colony stimulating factor: comparison of the mouse and human genes. EMBO J. 1985 Oct;4(10):2561–2568. doi: 10.1002/j.1460-2075.1985.tb03971.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosmann T. R., Bond M. W., Coffman R. L., Ohara J., Paul W. E. T-cell and mast cell lines respond to B-cell stimulatory factor 1. Proc Natl Acad Sci U S A. 1986 Aug;83(15):5654–5658. doi: 10.1073/pnas.83.15.5654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicola N. A., Metcalf D. Binding of iodinated multipotential colony-stimulating factor (interleukin-3) to murine bone marrow cells. J Cell Physiol. 1986 Aug;128(2):180–188. doi: 10.1002/jcp.1041280207. [DOI] [PubMed] [Google Scholar]
- Nicola N. A., Peterson L. Identification of distinct receptors for two hemopoietic growth factors (granulocyte colony-stimulating factor and multipotential colony-stimulating factor) by chemical cross-linking. J Biol Chem. 1986 Sep 15;261(26):12384–12389. [PubMed] [Google Scholar]
- O'Farrell P. Z., Goodman H. M., O'Farrell P. H. High resolution two-dimensional electrophoresis of basic as well as acidic proteins. Cell. 1977 Dec;12(4):1133–1141. doi: 10.1016/0092-8674(77)90176-3. [DOI] [PubMed] [Google Scholar]
- Ohara J., Paul W. E. Receptors for B-cell stimulatory factor-1 expressed on cells of haematopoietic lineage. Nature. 1987 Feb 5;325(6104):537–540. doi: 10.1038/325537a0. [DOI] [PubMed] [Google Scholar]
- Palaszynski E. W., Ihle J. N. Evidence for specific receptors for interleukin 3 on lymphokine-dependent cell lines established from long-term bone marrow cultures. J Immunol. 1984 Apr;132(4):1872–1878. [PubMed] [Google Scholar]
- Park L. S., Friend D., Gillis S., Urdal D. L. Characterization of the cell surface receptor for a multi-lineage colony-stimulating factor (CSF-2 alpha). J Biol Chem. 1986 Jan 5;261(1):205–210. [PubMed] [Google Scholar]
- Park L. S., Friend D., Grabstein K., Urdal D. L. Characterization of the high-affinity cell-surface receptor for murine B-cell-stimulating factor 1. Proc Natl Acad Sci U S A. 1987 Mar;84(6):1669–1673. doi: 10.1073/pnas.84.6.1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce J. H., Di Fiore P. P., Aaronson S. A., Potter M., Pumphrey J., Scott A., Ihle J. N. Neoplastic transformation of mast cells by Abelson-MuLV: abrogation of IL-3 dependence by a nonautocrine mechanism. Cell. 1985 Jul;41(3):685–693. doi: 10.1016/s0092-8674(85)80049-0. [DOI] [PubMed] [Google Scholar]
- Rubin J. B., Shia M. A., Pilch P. F. Stimulation of tyrosine-specific phosphorylation in vitro by insulin-like growth factor I. 1983 Sep 29-Oct 5Nature. 305(5933):438–440. doi: 10.1038/305438a0. [DOI] [PubMed] [Google Scholar]
- Schrader J. W., Lewis S. J., Clark-Lewis I., Culvenor J. G. The persisting (P) cell: histamine content, regulation by a T cell-derived factor, origin from a bone marrow precursor, and relationship to mast cells. Proc Natl Acad Sci U S A. 1981 Jan;78(1):323–327. doi: 10.1073/pnas.78.1.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schrader J. W. The panspecific hemopoietin of activated T lymphocytes (interleukin-3). Annu Rev Immunol. 1986;4:205–230. doi: 10.1146/annurev.iy.04.040186.001225. [DOI] [PubMed] [Google Scholar]
- Sideras P., Palacios R. Bone marrow pro-T and pro-B lymphocyte clones express functional receptors for interleukin (IL) 3 and IL 4/BSF-1 and nonfunctional receptors for IL 2. Eur J Immunol. 1987 Feb;17(2):217–221. doi: 10.1002/eji.1830170211. [DOI] [PubMed] [Google Scholar]
- Sorensen P., Farber N. M., Krystal G. Identification of the interleukin-3 receptor using an iodinatable, cleavable, photoreactive cross-linking agent. J Biol Chem. 1986 Jul 15;261(20):9094–9097. [PubMed] [Google Scholar]
- Swarup G., Cohen S., Garbers D. L. Inhibition of membrane phosphotyrosyl-protein phosphatase activity by vanadate. Biochem Biophys Res Commun. 1982 Aug;107(3):1104–1109. doi: 10.1016/0006-291x(82)90635-0. [DOI] [PubMed] [Google Scholar]
- Tertian G., Yung Y. P., Guy-Grand D., Moore M. A. Long-term in vitro culture of murine mast cells. I. Description of a growth factor-dependent culture technique. J Immunol. 1981 Aug;127(2):788–794. [PubMed] [Google Scholar]
- Tojo A., Kasuga M., Urabe A., Takaku F. Vanadate can replace interleukin 3 for transient growth of factor-dependent cells. Exp Cell Res. 1987 Jul;171(1):16–23. doi: 10.1016/0014-4827(87)90247-3. [DOI] [PubMed] [Google Scholar]
- Ushiro H., Cohen S. Identification of phosphotyrosine as a product of epidermal growth factor-activated protein kinase in A-431 cell membranes. J Biol Chem. 1980 Sep 25;255(18):8363–8365. [PubMed] [Google Scholar]
- Walker F., Burgess A. W. Specific binding of radioiodinated granulocyte-macrophage colony-stimulating factor to hemopoietic cells. EMBO J. 1985 Apr;4(4):933–939. doi: 10.1002/j.1460-2075.1985.tb03721.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinstein Y., Ihle J. N., Lavu S., Reddy E. P. Truncation of the c-myb gene by a retroviral integration in an interleukin 3-dependent myeloid leukemia cell line. Proc Natl Acad Sci U S A. 1986 Jul;83(14):5010–5014. doi: 10.1073/pnas.83.14.5010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokota T., Lee F., Rennick D., Hall C., Arai N., Mosmann T., Nabel G., Cantor H., Arai K. Isolation and characterization of a mouse cDNA clone that expresses mast-cell growth-factor activity in monkey cells. Proc Natl Acad Sci U S A. 1984 Feb;81(4):1070–1074. doi: 10.1073/pnas.81.4.1070. [DOI] [PMC free article] [PubMed] [Google Scholar]