Abstract
Aeroallergens produced by Alternaria alternata can elicit life-threatening exacerbations of asthma in patients sensitized to this fungus. In this study, the effect of Alternaria on ion transport mechanisms underlying mucociliary clearance and airway epithelial barrier function was investigated in human airway epithelial cells. Apical exposure to Alternaria induced an increase in anion secretion that was inhibited by blockers of CFTR and Ca2+-activated Cl− channels. Stimulation of anion secretion was dependent on Ca2+ uptake from the apical solution. Alternaria exposure also produced an increase in reactive oxygen species (ROS) that was blocked by pretreatment with the oxidant scavenger glutathione (GSH). GSH and the NADPH oxidase inhibitor/complex 1 electron transport inhibitor diphenylene iodonium chloride (DPI) blocked ATP release and the increase in intracellular [Ca2+] evoked by Alternaria. Alternaria also decreased transepithelial resistance, and a portion of this effect was dependent on the increase in ROS. However, the Alternaria-induced increase in unidirectional dextran (molecular mass = 4,000 Da) flux across the epithelium could not be accounted for by increased oxidative stress. These results support the conclusion that oxidative stress induced by Alternaria was responsible for regulating Ca2+-dependent anion secretion and tight junction electrical resistance that would be expected to affect mucociliary clearance.
Keywords: chloride secretion, tight junctions, ATP release, intracellular calcium
alternaria alternata is a saprophytic fungus with worldwide distribution (15, 47). Although considered primarily as an outdoor fungus, it is also found in damp, poorly ventilated buildings (33, 34, 40). Alternaria spore counts are generally highest during warm, dry, and windy weather conditions in late summer and early autumn (17, 41). These spores contain some of the most potent airborne allergens capable of inducing IgE-mediated allergic airway inflammation (16, 18, 29, 42). Moreover, asthma severity appears to be more strongly associated with Alternaria exposure than with sensitization to other aeroallergens, including house dust mite and pollens (18, 29). Alternaria spores are known to induce respiratory distress and airway hyperresponsiveness in asthma patients, and individuals sensitized to Alternaria are more likely to develop asthma than nonsensitized individuals (25, 45). In some populations, sensitization to Alternaria is particularly high, especially in farm workers where greater occupational exposure to Alternaria spores frequently occurs (1, 27, 35, 40, 49). Furthermore, a study investigating the effects of atmospheric CO2 on Alternaria alternata revealed a threefold increase in sporulation and more than twice the level of antigenic protein content/spore in response to elevated CO2 concentrations above 600 μmol/mol (52). These results suggest that continuing increases in atmospheric CO2 associated with global climate change will increase both the level of Alternaria exposure and antigenicity of spores that come in contact with the airways.
Earlier studies of human airway epithelial cells have shown that exposure to Alternaria extracts induces the synthesis and secretion of IL-33, a cytokine that is known to induce Th2-type inflammation in airways (9, 19, 20, 26, 45). IL-33 secretion by the epithelium occurs as a consequence of ATP release following exposure to Alternaria aeroallergens, resulting in activation of P2X7 receptors and sustained elevations in intracellular Ca2+ concentration ([Ca2+]i). (26, 39). This prolonged increase in [Ca2+]i activates processing enzymes that cleave IL-33 into its 19 kDa secreted form and stimulates translocation of the processed cytokine from its storage site within the nucleus into the cytoplasm where it is ultimately secreted into the extracellular fluid. Once released, IL-33 can bind to ST2 receptors expressed by multiple immune cell types. ST2 receptor activation induces the production and secretion of Th2 cytokines necessary for eliciting allergic inflammation within the airway mucosa (9).
Additionally, more recent experiments have demonstrated that Alternaria exposure induces oxidative stress in human airway epithelial cells and that this effect is important in the initiation of IL-33 processing and release (19, 21, 48). This finding is reminiscent of an earlier study where exposure to pollen proteins induces allergic airway inflammation by increasing the production of reactive oxygen species (ROS) by lung epithelial cells (2, 4). The oxidative stress was produced by an endogenous NADPH oxidase(s) associated with pollen grains since removal of pollen NADPH oxidase activity inhibited the inflammatory response. However, exposure of airway epithelial cells to Alternaria extract was shown to activate NADPH dual oxidase 1 (DUOX1) located within the apical membrane of the epithelium following P2Y2 receptor activation. DUOX1-dependent H2O2 production, in turn, stimulated Src and epidermal growth factor receptor signaling that ultimately resulted in calpain-2 activation and subsequent processing and release of IL-33 (19). Interestingly, the mycotoxin altertoxin (ATX) II, a perylene quinone-type compound produced by Alternaria alternata, was found to interact with the nuclear factor erythroid-derived 2-like antioxidant response element (Nrf2/ARE) pathway in HT29 intestinal epithelial cells to induce a concentration-dependent depletion of glutathione (GSH) within the cytoplasm, suggesting that ATX II may constitute at least one component of Alternaria extract that can produce oxidative stress (23).
Exposure of airway epithelial cells to Alternaria allergens has also been shown to significantly reduce barrier function (31). Measurements of transepithelial resistance (TER) and FITC-labeled dextran (molecular mass = 4,000) showed that exposure to Alternaria extracts produced concentration-dependent decreases in TER within 1 h and increased apical-to-basolateral flux of FITC-dextran after 24 h of Alternaria exposure. Heat treatment of the extract was effective at inhibiting the actions of Alternaria on epithelial barrier function, suggesting that the activity of an enzyme within the extract was responsible for barrier disruption. Moreover, experiments with various protease inhibitors including 4-(2-aminoethyl)benzenesulfonyl fluoride hydrochloride (AEBSF) suggested that serine proteases were the predominant class of proteolytic enzymes responsible for the barrier disrupting effects of Alternaria allergens.
In the present study the effects of Alternaria aeroallergen exposure on ion transport function of human airway epithelium was investigated using an immortalized airway epithelial cell line with well-characterized ion transport properties. Our objective was to define the transport mechanisms that are stimulated by these allergens and to identify the underlying signaling pathways responsible for their actions on the epithelium. Our experiments revealed that Alternaria stimulates both Ca2+-dependent and CFTR-dependent anion secretion and that treatment with oxidant scavengers such as GSH and N-acetyl cysteine (NAC) inhibit this response by reducing oxidative stress. In addition, GSH partially blocked the decrease in transepithelial resistance produced by Alternaria exposure. These findings demonstrate that Alternaria allergens are capable of modulating the transport properties of the airway epithelium. However, prolonged exposure leads to disruption of epithelial barrier function that would ultimately reduce mucociliary clearance.
MATERIALS AND METHODS
Materials.
16HBE14o− cells were obtained from Dr. Dieter Gruenert, University of California, San Francisco (5). Eagle's minimum essential medium with Earl's salts (MEM), fetal bovine serum, nonessential amino acids, penicillin-streptomycin, CellROX orange, phosphate-buffered saline (PBS), and Hanks’ balanced salt solution (HBSS) were obtained from ThermoFisher Scientific (Pittsburgh, PA). The acetoxymethyl ester form of fura-2 (fura-2 AM) and 1,2-bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid (BAPTA-AM) were procured from Invitrogen/Life Technologies (Carlsbad, CA). The following compounds were from Sigma-Aldrich Chemical (St. Louis, MO): ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid (EGTA), 4,4′-diisothiocyano-2,2′-stilbenedisulfonic acid (DIDS), 5-[(4-carboxyphenyl)methylene]-2-thioxo-3-[(3-trifluoromethyl)phenyl-4-thiazolidinone (CFTRinh-172), ionomycin, diphenyleneiodonium chloride (DPI), glutathione (GSH), N-acetyl cysteine (NAC), hydrogen peroxide (H2O2), phenylmethylsulfonyl fluoride (PMSF), trans-epoxysuccinyl-l-leucylamido(4-guanidino)butane, l-trans-3-carboxyoxiran-2-carbonyl-l-leucylagmatine, N-(trans-epoxysuccinyl)-l-leucine 4-guanidinobutylamide (E64), 2-[[(4-phenoxyphenyl)sulfonyl]methyl]-thiirane (SB-3CT), 4-(2-aminoethyl)benzenesulfonyl fluoride hydrochloride (AEBSF), and 6-tert-butyl-2-(furan-2-carboxamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylic acid (CaCCinh-Ano1). Trypsin was obtained from Lonza (Allendale, NJ) and GM6001 from EMD Millipore (Billerica, MA). Antibodies for E-cadherin (ab76055), ZO-1 (ab59720), and β-catenin (ab32572) were purchased from Abcam (Cambridge, MA). Antibody validations in human cells for E-cadherin (ab76055), ZO-1 (ab59720), and β-catenin (ab32572) have been previously published (7, 24, 51).
Alternaria alternata extract.
Alternaria extract was purchased from Greer Laboratories (Lenoir, NC). The precise composition of the extract is unknown but earlier studies with human eosinophils indicated that proteinases within the extract play an important role in mediating Alternaria activity (33).
Cell culture.
Immortalized human bronchial epithelial cells (16HBE14o− cells) were grown in MEM with 10% fetal bovine serum, 1% nonessential amino acids, and 1% penicillin-streptomycin at 37°C in a humidified CO2 (5%) incubator. Monolayers used in voltage-clamp experiments were grown on Snapwell membrane filters (Fisher Scientific, Pittsburgh, PA) until transepithelial resistance (TER) exceeded 500 Ω·cm2. Cells used for Ca2+ imaging experiments were grown on two-well chamber slides (Laboratory-Tek, VWR International, Chicago, IL).
Electrophysiology.
Transepithelial resistances were measured using an EVOM epithelial voltohmeter connected to Ag/AgCl electrodes (World Precision Instruments, New Haven, CT). Monolayers were mounted in Ussing chambers and bathed on the apical and basolateral sides of the epithelium with symmetric physiologic saline solution (in mM): 130 NaCl, 6 KCl, 1.5 CaCl2, 1 MgCl2, 20 NaHCO3, 0.3 NaH2PO4, and 1.3 Na2HPO4 (pH 7.4), maintained at 37°C and gassed with 95% O2-5% CO2. Cells were apically stimulated with Alternaria extract at concentrations indicated in the figure legends, and all three anion channel blockers were added to the apical solution. Voltage-clamp experiments were performed using DVC1000 epithelial voltage/current clamps (World Precision Instruments), and short-circuit current (Isc) data were acquired using Axoscope 8.1 software (Molecular Devices, Sunnyvale CA).
Calcium imaging.
Ca2+ imaging experiments were performed following a previously published protocol (35). Briefly, 16HBE14o− cells were gown on chamber slides for 48–72 h. Prior to loading cells with fura-2, the media were replaced with Ca2+/Mg2+-free HBSS solution containing 10 mM HEPES buffer. Cells were loaded with fura-2 AM for 1 h, then washed with Ca2+/Mg2+-containing HBSS before mounting onto the stage of a Nikon Diaphot inverted fluorescence microscope. Fluorescence was visualized with a Nikon UV ×20 objective at excitation wavelengths of 340 and 380 nm and a single emission wavelength of 510 nm. Image acquisition and data analysis were performed using Image-1 MetaMorph software. Changes in intracellular [Ca2+] ([Ca2+]i) were determined from the fluorescence ratio (F340/F380) following calibration using the fura-2 AM calcium imaging calibration kit from ThermoFisher Scientific.
Oxidative stress measurements.
Cells were grown for 48–72 h on two-well chamber slides in standard MEM/fetal calf serum-containing media. Oxidative stress induced by Alternaria exposure was measured following incubation with CellROX orange reagent according to the manufacturer’s instructions (ThermoFisher Scientific). After loading for 30 min, cells were exposed to Alternaria (100 μg/ml) or 5 mM GSH + Alternaria (100 μg/ml) or not exposed to either GSH or Alternaria (basal conditions) for 20 min. Images were captured with a Nikon Diaphot fluorescence microscope equipped with a Diagnostics Instruments Model 14.1 monochrome camera and SPOT Advanced imaging software. Relative fluorescence intensity measurements were made using ImageJ software (National Institutes of Health, Bethesda, MD).
ATP release measurements.
ATP measurements in the extracellular media were performed in real time as previously described using a luciferase/luciferin bioluminescence ATP determination kit (ThermoFisher Scientific) (39). ATP-evoked photon emission was measured using a Glomax 20/20 luminometer (Promega, Madison WI). Initially, standard curves were generated over a range of 1–1,000 nM ATP in standard reaction solution (SRS). The ATP concentrations were averaged after subtraction of the background luminescence. Cultured epithelial monolayers grown on 35-mm dishes were then loaded into the luminometer and the apical surface covered with SRS-containing luciferin/luciferase. Monolayers were then stimulated with Alternaria or pretreated with DPI (100 μM) or GSH (5 mM) for 30 min before stimulation with Alternaria. Luminescence was measured over a period of 20 min after the addition of Alternaria, and the signal was converted to [ATP] using the previously generated calibration curve.
Dextran flux measurements.
Monolayers were initially grown on 12-mm Snapwell filters until they reached resistances between ~600 and 1,000 Ω·cm2. Media were removed and the cells were washed twice with serum-free MEM. Fluorescein isothiocyanate (FITC)-labeled dextran (molecular mass 4,000 Da, final concentration; 2.0 mg/ml) was added to the apical solution 1 h before treatment with Alternaria or H2O2 (0.5 mM). Samples of the apical and basolateral solutions were taken at several time points (−1, 0, 1, and 4 h). Fluorescence intensity was measured using a Biotek Synergy 2 fluorescence plate reader equipped with filters for detecting FITC (excitation/emission wavelengths = 485 nm/530 nm). To determine dextran concentration, a standard curve was generated using varying FITC-dextran concentrations (in μM: 0.0625, 0.125, 0.25, 0.5, 1, and 2) and used to calculate the unidirectional fluxes at time 0, 1 and 4 h following Alternaria or H2O2 exposure.
Immunocytochemistry.
Monolayers used for characterizing the effects of Alternaria and H2O2 on TER were fixed with 4% paraformaldehyde for 30 min and incubated with 0.1% Triton X-100 for 20 min before being washed and blocked with 3% bovine serum albumin (BSA) for 2 h. Monolayers were then incubated with primary E-cadherin, ZO-1, or β-catenin antibodies overnight in 3% BSA. The cells were then incubated in secondary Alexa Fluor antibodies diluted in PBS for 45 min then washed five times with PBS. Next, monolayers were treated with 4′,6-diamidino-2-phenylindole (DAPI) diluted in PBS for 10 min and then washed again with PBS. Filters were subsequently mounted on slides, and antibody labeling was examined by confocal microscopy using an Olympus FV1000 confocal microscope with a ×20 fluorescence objective.
Statistics.
Data are presented as means ± SE. Statistical significance was determined by using an unpaired two-tailed t-test (for single comparisons) or an ANOVA followed by Dunnett’s posttest for multiple comparisons with a common control or an ANOVA followed by a Tukey’s posttest for multiple comparisons. A value of P < 0.05 was considered significant. Concentration-response relationships were analyzed using PRISM 6 software (GraphPad Software, La Jolla, CA).
RESULTS
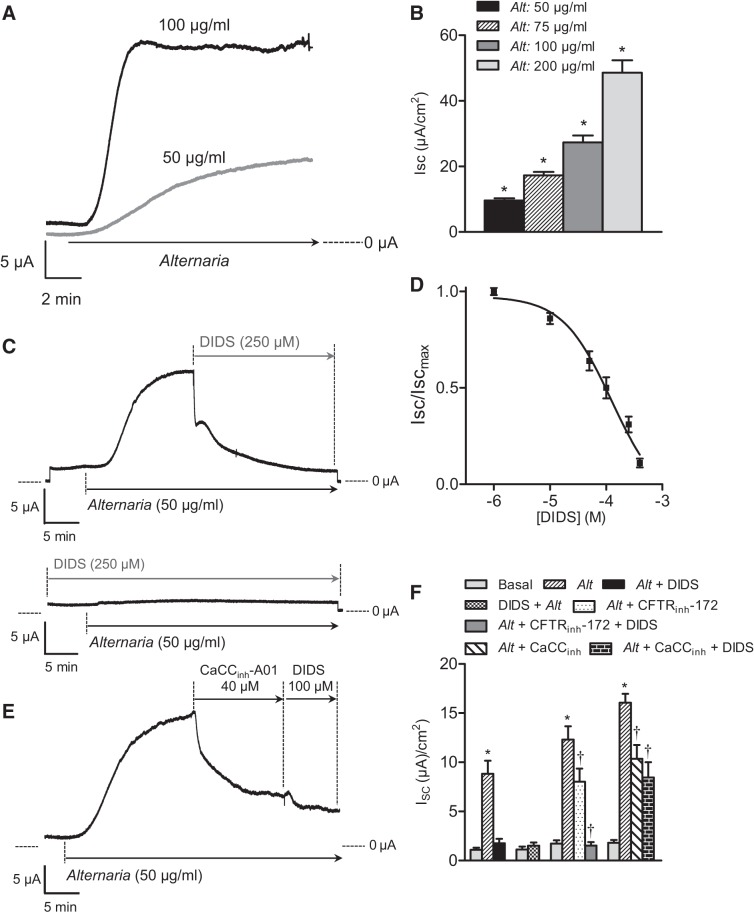
Apical addition of Alternaria extract to monolayers of immortalized human bronchial epithelial cells produced an increase in short-circuit current (Isc) consistent with anion secretion. Figure 1, A and B, shows the kinetics of Alternaria-evoked increases in Isc at concentrations of 50 and 100 μg/ml and a concentration-response relationship for Alternaria ranging from 50 to 200 μg/ml. The rate and magnitude of the Isc increase was found to be dependent on concentration. However, stable measurements of Isc could not be obtained at Alternaria concentrations >200 μg/ml. Treatment of monolayers with the disulfonic stilbene derivative DIDS (250 μM), an inhibitor of calcium-activated Cl− channels (CaCCs), as well as certain anion exchange transporters (27, 42, 43) produced an initial rapid inhibition of Isc, followed by a slower, time-dependent decrease in current to baseline levels (Fig. 1C). Similarly, pretreatment with 250 μM DIDS effectively abolished the increase in Isc typically observed following apical addition of 50 μg/ml Alternaria. A concentration-response relationship for DIDS is shown in Fig. 1D. The IC50 value was calculated to be 100 μM based on a nonlinear least squares fit using a three-parameter logistic function (Prism 6.0). The effect of an inhibitor of the A01 (TMEM16A) calcium-activated Cl− channel (CaCCinh-A01) was also tested and its effect on Isc is shown in Fig. 1E. CaCCinh-A01 inhibited ~50% of the Alternaria-stimulated Isc and blocked the further decrease in current produced by treatment with 100 μM DIDS, indicating that DIDS and CaCCinh-A01 target a common apical transport pathway involved in anion secretion (Fig. 1E). Alternaria also stimulated CFTR-dependent anion secretion as indicated by inhibition of the Alternaria stimulated Isc with CFTRinh-172 shown in Fig. 1F.
Fig. 1.
Alternaria exposure stimulates transepithelial anion secretion. A: kinetics of the short-circuit current (Isc) response following apical exposure to 50 and 100 μg/ml Alternaria extract. B: concentration-response relationship for Alternaria (Alt)-evoked increases in steady-state Isc (n = 4; *significantly different from the basal short-circuit current). C: kinetics of apical DIDS (250 μM) posttreatment and pretreatment on the Alternaria-evoked (50 μg/ml) Isc response (n = 6). D: concentration-response relationship showing the inhibitory effects of apical DIDS on the Alternaria-induced (50 μg/ml) increase in Isc (n = 5). The IC50 = 1.23 × 10−4 M. E: kinetics of apical CaCCinh (40 μM) and DIDS posttreatment on the Alternaria-evoked Isc response (n = 6). F: summary bar graph showing the effects of CFTRinh-172 (20 μM), DIDS (250 or 100 μM), and CaCCinh (40 μM) on steady-state Isc increase evoked by Alternaria (50 μg/ml). *Significant difference between the basal and Alternaria-stimulated Isc. †Significant differences between the Alternaria-stimulated and inhibitor (CFTRinh-172, CaCCinh-Ano1, or DIDS)-treated Isc values.
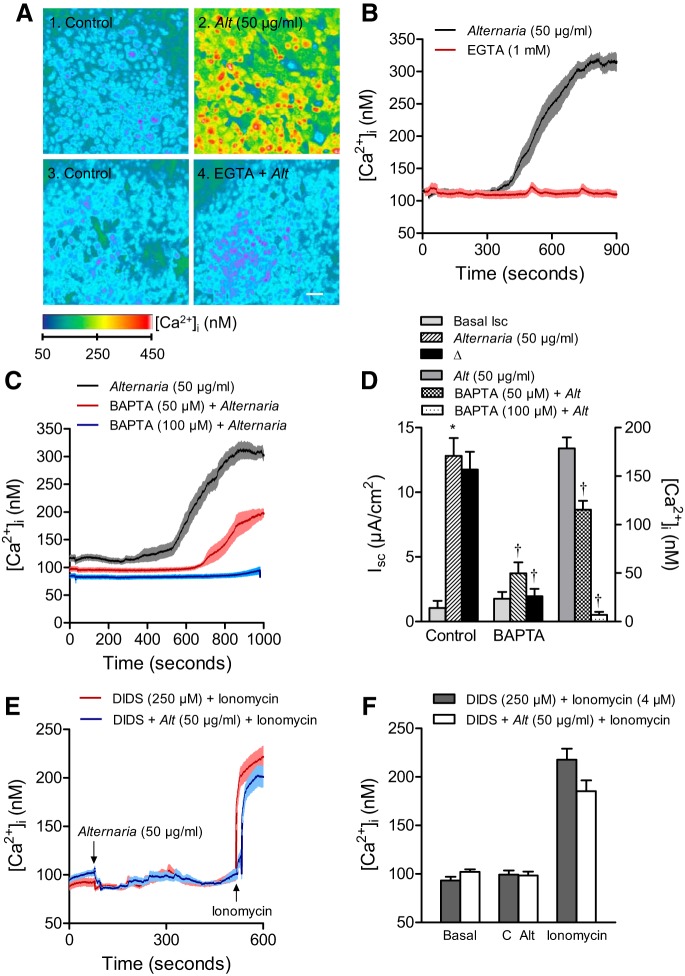
Apical addition of Alternaria (50 μg/ml) also produced a large and sustained increase in [Ca2+]i (Fig. 2, A and B). Moreover, adding 1 mM EGTA to the apical solution, with the purpose of lowering extracellular [Ca2+] before treatment with Alternaria, abolished the increase in [Ca2+]i as shown in panel 4 of Fig. 2A and in the red trace of Fig. 2B. This result indicated that the Alternaria-evoked increase in [Ca2+]i was a consequence of Ca2+ uptake from the extracellular solution. Pretreating cells with a cell-permeable Ca2+-chelating agent (BAPTA-AM) at 50 and 100 μM also reduced the increase in [Ca2+]i associated with apical Alternaria exposure (Fig. 2, C and D). Furthermore, pretreatment with BAPTA-AM also significantly blocked the Isc increase induced by Alternaria (Fig. 2D), suggesting a direct role for [Ca2+]i in regulating Alternaria-evoked anion secretion. In an earlier experiment (Fig. 1C), DIDS was shown to have a biphasic effect on the Alternaria Isc response such that pretreatment with 250 μM DIDS completely blocked the increase in Isc. The slow, time-dependent component of the DIDS response suggested that the compound may be blocking anion secretion by a mechanism independent of inhibiting apical membrane anion channels. To test this hypothesis, we examined the effect of DIDS on the Alternaria-induced increase in [Ca2+]i and found that pretreatment with 250 μM DIDS completely blocked the response, but had no effect on the increase in [Ca2+]i produced by apical addition of the Ca2+ ionophore, ionomycin (4 μM) (Fig. 2, E and F). This result suggested that 250 μM DIDS is capable of blocking the Ca2+ uptake mechanism that is activated by Alternaria in addition to its effect on CaCCs.
Fig. 2.
Alternaria-induced changes in intracellular Ca2+ concentration ([Ca2+]). A: images of 16HBE14o− cell monolayers labeled with fura-2 AM before and after 700 s of stimulation with 50 μg/ml Alternaria extract and after pretreatment with 1 mM EGTA in the extracellular solution. Scale bar = 20 μm. B: kinetics of the Alternaria effect on [Ca2+]i in the absence and presence of 1 mM EGTA in the extracellular solution (n = 25 cells for each condition). C: effects of BAPTA-AM pretreatment (50 and 100 μM) on Alternaria-evoked increases in [Ca2+]i (n = 25 cells for each condition). D: BAPTA pretreatment significantly inhibits the effect of Alternaria on [Ca2+]i (n = 25 cells) and Isc (n = 8 monolayers). *Significantly different from the basal Isc. †Significant differences between basal and Alternaria-treated conditions. E: pretreatment with 250 μM DIDS blocks the Alternaria-evoked increase in [Ca2+]i but does not inhibit ionomycin-induced increases in [Ca2+]i (n = 25 cells for each condition). F: summary of the effects of DIDS on Alternaria and ionomycin-induced increases in [Ca2+]i (n = 25 for each condition).
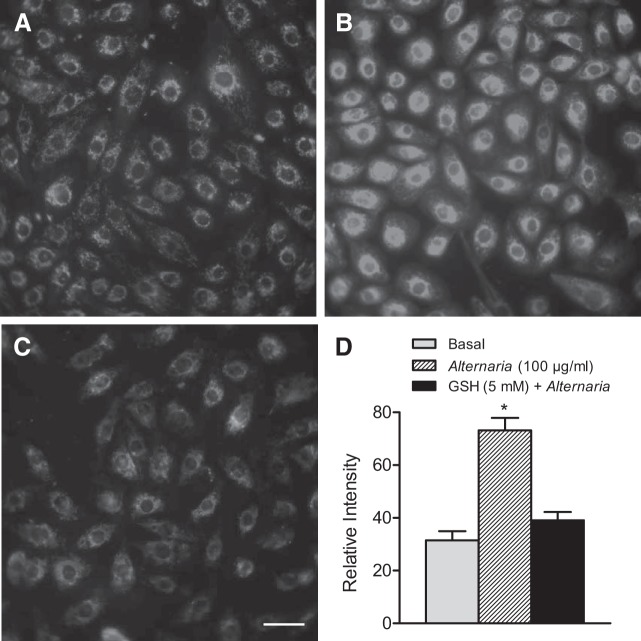
Figure 3 shows the results of experiments designed to determine whether Alternaria exposure produces ROS accumulation leading to oxidative stress. CellROX orange is a cell-permeable fluorescent compound that responds to oxidative stress by increasing fluorescence (18, 26, 49). CellROX orange-loaded cells exhibited a low level of fluorescence under basal, unstimulated conditions (Fig. 3A); however, exposure to 100 μg/ml Alternaria for 15 min produced a significant increase in fluorescence consistent with ROS generation and oxidative stress (Fig. 3B). In contrast, cells that were initially pretreated with 5 mM glutathione (GSH) before Alternaria exposure showed significantly reduced levels of fluorescence compared with Alternaria treatment alone (Fig. 3, C and D). This result demonstrated that oxidant scavenging by GSH is sufficient to block Alternaria-evoked ROS generation.
Fig. 3.
Alternaria induces oxidative stress in 16HBE14o− cells. A: basal fluorescence of cells loaded with CellROX orange. B: Alternaria-evoked (50 μg/ml) increase in fluorescence after 15 min of exposure to the extract. C: pretreatment with 5 mM GSH blocks the increase in fluorescence produced by Alternaria exposure. D: summary of the changes in relative fluorescence intensity obtained from images of basal, Alternaria-stimulated, and 5 mM GSH + Alternaria-stimulated conditions (n = 25 cells for each condition). *Significantly different from the basal condition. Scale bar = 20 μm.
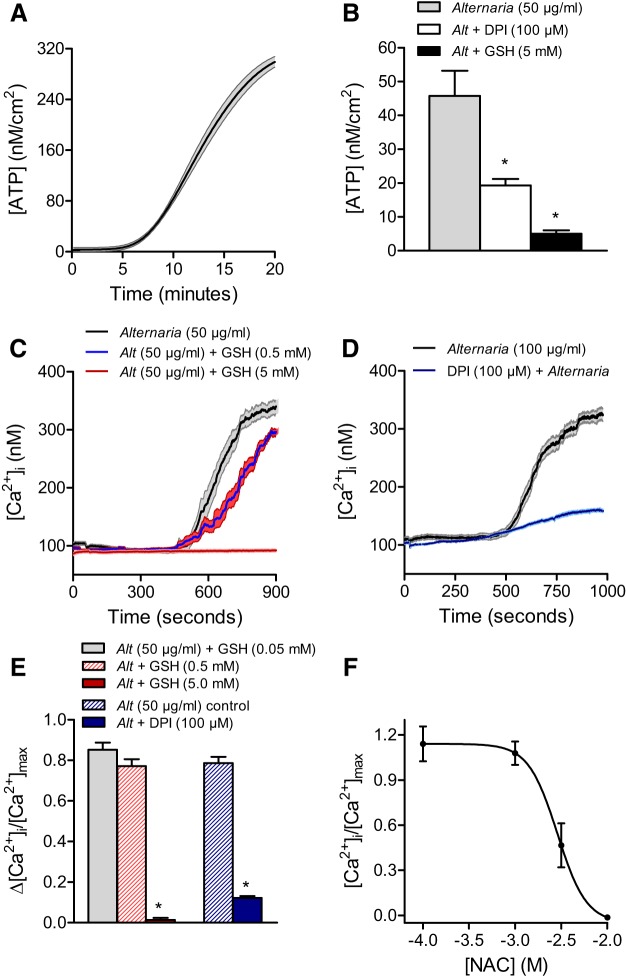
Earlier studies using primary normal human bronchial epithelial (NHBE) cells showed that Alternaria exposure induces ATP release (19, 26, 39). We tested whether a similar response could be detected in 16HBE14o− cells by measuring the release of ATP by photon counting in real time using monolayers where the apical solution contained luciferin and luciferase. Results presented in Fig. 4A show that exposure to Alternaria (50 μg/ml) induces ATP secretion into the apical solution starting at ~5 min and continues to increase up to 20 min in the presence of Alternaria allergens. This increase in ATP release was dramatically blocked by pretreatment with 5 mM GSH, and to a lesser extent following pretreatment with DPI, an inhibitor of NADPH oxidase activity and of complex 1 of the electron transport chain (30) (Fig. 4B). Subsequent Ca2+ imaging experiments revealed that 5 mM GSH and 100 μM DPI were highly effective at inhibiting Alternaria-evoked increases in [Ca2+]i (Fig. 4, C and D) The effects of GSH and DPI on the steady-state levels of [Ca2+]i following Alternaria exposure are shown in Fig. 4E. The dose-response relationship of N-acetyl cysteine (NAC) a clinically used oxidant scavenger, on the steady-state increase in [Ca2+]i induced by Alternaria is reported in Fig. 4F. These data indicate that Alternaria-evoked oxidative stress underlies the increase in both ATP release and Ca2+ uptake that occurs following apical treatment with Alternaria.
Fig. 4.
ROS scavengers and DPI inhibit the effects of Alternaria on ATP release and [Ca2+]i. A: kinetics of ATP release evoked by Alternaria (100 μg/ml). B: effects of DPI (100 μM) and GSH (5 mM) pretreatment (5 min before Alternaria addition) on ATP release evoked by 50 μg/ml Alternaria. *Significantly different from the Alternaria-treated condition. C: GSH inhibits Alternaria-evoked increase in [Ca2+]i (n = 25 cells for each condition). D: DPI inhibits Alternaria-evoked increase in [Ca2+]i (n = 25 cells for each condition). E: summary of the effects of GSH and DPI on [Ca2+]i (n = 25 cells for each condition). *Significantly different from the Alternaria-treated condition. F: concentration-response effects of N-acetyl cysteine (NAC) on [Ca2+]i (n = 25 cells for each condition).
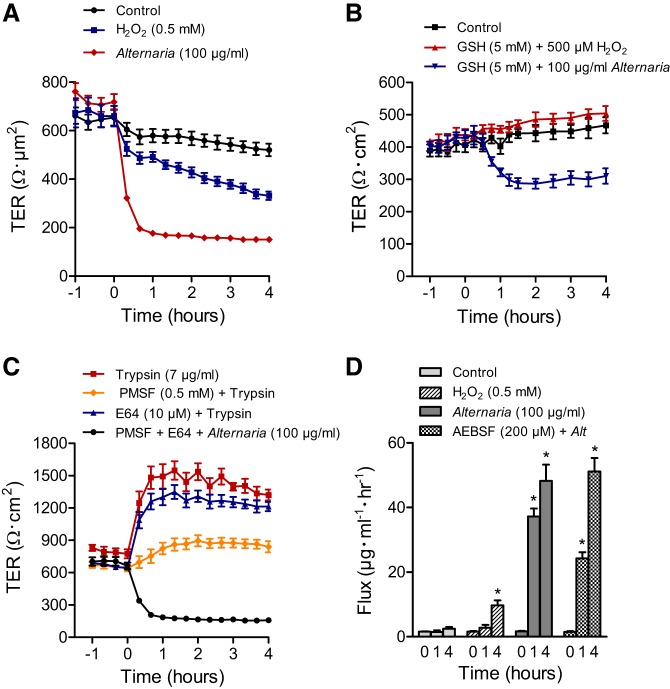
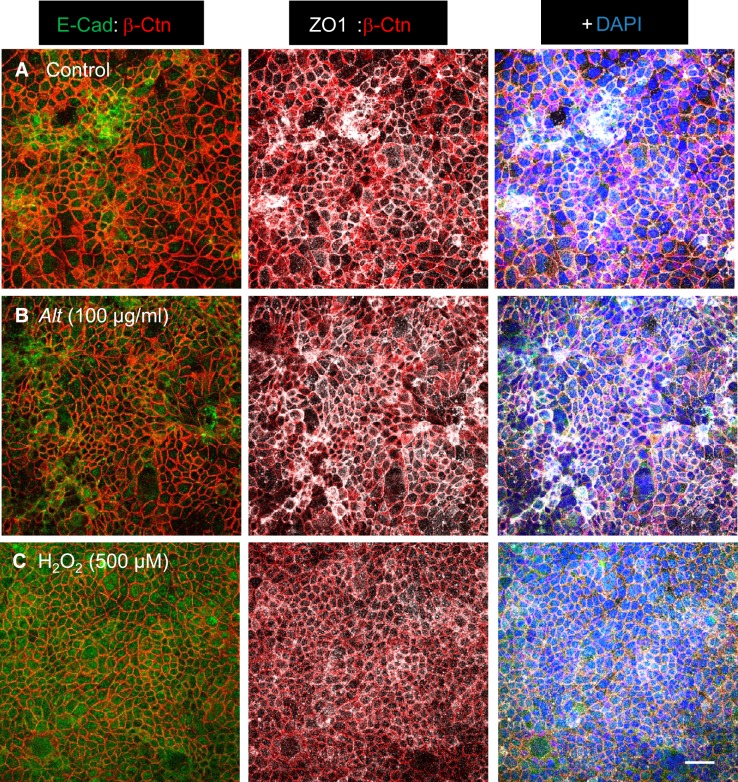
The effects of Alternaria (100 μg/ml) and exogenously added hydrogen peroxide (0.5 mM) on epithelial barrier function was investigated by measuring both transepithelial resistance (TER) and unidirectional (apical-to-basolateral) dextran (molecular mass = 4,000 Da) fluxes (Fig. 5). Exposure to Alternaria produced a decrease in TER from 760 ± 35 to 151 ± 4 Ω·cm2 (n = 6) over a period of 4 h. This change in resistance reflects both the increase in transepithelial Cl− conductance and an increase in tight junction ion permeability. Addition of 0.5 mM H2O2 to the apical solution also reduced the TER (674 ± 60 to 331 ± 18 Ω·cm2; n = 6) but not to the same extent as Alternaria after 4 h (Fig. 5A). Moreover, the time course of the H2O2 response was appreciably slower compared with that of Alternaria, which caused nearly a complete loss of resistance after 1 h of exposure (note: the resistance of the filters alone = ~100 Ω·cm2). When monolayers were initially pretreated with 5 mM GSH and then exposed to either H2O2 or Alternaria, the effect of H2O2 was completely blocked whereas the response to Alternaria was only partially inhibited (Fig. 5B). However, pretreatment of monolayers with various proteinase inhibitors including PMSF (a pan-specific serine proteinase inhibitor), E64 (a cysteine protease inhibitor), GM6001 [a pan-specific matrix metalloproteinase (MMP) inhibitor], SB-3CT (an MMP2 and MMP9 inhibitor), and AEBSF (a pan-specific serine proteinase inhibitor) each failed to inhibit the decrease in TER evoked by Alternaria (Table 1). As a control, we tested the effects of a low concentration of trypsin (7 μg/ml) on TER and discovered that it significantly increased resistance by >2-fold and sustained this increase for 4 h in the continuous presence of the enzyme. Pretreatment with a serine proteinase inhibitor (PMSF) effectively blocked the rise in TER whereas E64, a cysteine proteinase inhibitor, did not (Fig. 5C). To determine whether the effect of Alternaria on TER was due to activation of PAR2 receptors, we tested the effects of the PAR2-activating peptide SLIGKV-NH2 (20 μM) and found that it had no effect on resistance after 4 h of treatment (data not shown). The results of trypsin experiments indicated that low concentrations of the enzyme do not reduce TER and in fact cause an increase that can be blocked by a serine proteinase inhibitor, but is poorly blocked by a cysteine proteinase inhibitor. However, these inhibitors at the same concentrations were ineffective at blocking the Alternaria response. Figure 5D shows that Alternaria increases the permeability of epithelial monolayers to macromolecules as indicated by an increase in the unidirectional flux of dextran (molecular mass = 4,000) after 1 and 4 h of allergen exposure. In contrast, pretreatment with AEBSF was ineffective at blocking the stimulated dextran flux after 4 h, although a significantly lower flux was observed after a 1-h exposure to Alternaria (Fig. 5C). H2O2 had no significant effect on the dextran flux after 1 h; however, a significant increase was detected after 4 h. To determine whether Alternaria or H2O2 exposure for 4 h produced visible signs of monolayer destruction, we performed confocal imaging using antibodies that labeled tight junction proteins including β-catenin, E-cadherin, and ZO-1 (Fig. 6). Our results indicated that epithelial integrity remained intact, with no observable disruption of tight junctions or loss of epithelial cells, although some decrease in the intensity of β-catenin and ZO-1 staining was observed in monolayers treated with H2O2. These findings support the conclusion that Alternaria increases both ionic and macromolecular permeability of the epithelium, but that these changes do not involve observable destruction of monolayer integrity or junctional complexes between the epithelial cells.
Fig. 5.
Alternaria and H2O2 exposure reduces transepithelial resistance (TER) and increases macromolecule permeability of 16HBE14o− cell monolayers. A: time-dependent decreases in TER following exposure to apical Alternaria (100 μg/ml) or H2O2 (0.5 mM) compared with untreated controls (n = 6 for each condition). B: pretreatment with 5 mM GSH completely blocks the inhibitory effect of H2O2 on TER and partially inhibits the effects of Alternaria on TER (n = 6). C: effects of serine protease exposure (trypsin, 7 μg/ml) or Alternaria (100 μl/ml) on TER before and after pretreatment with the serine protease inhibitor PMSF (0.5 mM) or the cysteine protease inhibitor E64 (10 μM) (n = 3 for each condition). D: effects of Alternaria and H2O2 on apical-to-basolateral unidirectional FITC-Dextran (molecular mass = 4,000 Da) fluxes at time 0, 1 h after treatment, and 4 h after treatment. *Significantly different from time 0 (n = 6 for each condition).
Table 1.
Effect of proteinase inhibitors on the Alternaria-induced decrease in TER
| Pretreatment | TER, Ω·cm2 |
|---|---|
| Control | |
| 0 h | 654 ± 34 |
| 4 h | 521 ± 26 |
| Alternaria, 100 μg/ml | |
| 0 h | 718 ± 33 |
| 4 h | 151 ± 4* |
| PMSF, 0.5 mM | |
| 0 h | 554 ± 54 |
| 4 h | 158 ± 3* |
| E64, 10 μM | |
| 0 h | 562 ± 46 |
| 4 h | 155 ± 2* |
| GM6001, 10 μM | |
| 0 h | 1,026 ± 27 |
| 4 h | 150 ± 3* |
| 5B-3CT, 1 μM | |
| 0 h | 1,042 ± 14 |
| 4 h | 154 ± 2* |
| AEBSF, 200 μM | |
| 0 h | 847 ± 104 |
| 4 h | 151 ± 7* |
Values are means ± SE.
P < 0.05.
Fig. 6.
Effects of Alternaria and H2O2 on tight junction complexes in monolayers of 16HBE14o− cells. A: immunocytochemistry showing tight junction expression of E-cadherin, β-catenin, and ZO1 under untreated control conditions. B: exposure to Alternaria for 4 h produced no measurable change in the expression pattern for E-cadherin, β-catenin, or ZO1. C: treatment with 500 μM H2O2 for 4 h diminishes the intensity of β-catenin and ZO1 labeling along the lateral membranes. Scale bar = 20 μm.
DISCUSSION
16HBE14o− cells were originally isolated from a 1-yr-old male heart-lung transplant patient and immortalized following transfection with the SV40 T-antigen (5, 12). These cells have been shown to retain differentiated epithelial morphology including tight junctions, cilia and a transepithelial resistance comparable to primary bronchial epithelial cells. Monolayers of 16HBE14o− cells exhibit β-adrenergic receptor and cAMP-dependent regulation of anion secretion, mediated through activation of apical CFTR channels. Moreover, exposure to bradykinin or the Ca2+ ionophore ionomycin produced increases in intracellular [Ca2+] and stimulated Ca2+-dependent Cl− secretion (12). Using this cell line as an experimental model, we investigated the effects of Alternaria aeroallergens on transepithelial anion transport, a process that underlies mucociliary clearance. Our experiments demonstrated that Alternaria exposure induced a sustained, concentration-dependent increase in Isc, consistent with stimulation of anion secretion. Furthermore, treatment with inhibitors of CFTR and the Ca2+-activated Cl− channel Ano1 significantly inhibited the Isc, suggesting that both channels are activated following apical treatment with Alternaria. However, it is worth noting that CFTRinh-172 has been previously shown to inhibit the ClC-2 Cl− channel as well as CFTR. Earlier whole cell patch-clamp experiments with recombinant ClC-2 and CFTR channels expressed in HEK293 cells revealed that CFTRinh-172 inhibited both forskolin/IBMX-stimulated CFTR currents and lubiprosone-stimulated ClC-2 currents with equal potency (13). Therefore, care must be taken not to conclude that inhibition of anion transport by CFTRinh-172 results from CFTR inhibition alone, especially in cells that are known to express both ClC-2 and CFTR. In a prior study, a molecular approach involving the use of ClC-2 and ClC-3-deficient mice was used to rule out the possible involvement of these ClC channels as contributors to the Cl− conductance present in the basolateral membrane of airway epithelia (22). In the present study, ClC-2/CFTR knockdown experiments were not performed, therefore we are unable to exclude the possibility that ClC-2 channels may be contributing to Alternaria-evoked anion secretion. The disulfonic stilbene compound DIDS, a known Ca2+-dependent Cl− channel inhibitor, produced a biphasic effect on Isc resulting in complete inhibition of the Alternaria-evoked Isc response. The initial decrease in Isc was consistent with rapid inhibition of anion channel activity, whereas the slow, time-dependent decrease in current appeared to involve a separate mechanism necessary for sustained anion secretion. We speculate that the slow component of the DIDS response is related to inhibition of Ca2+ uptake and an associated decrease in [Ca2+]i.
Alternaria exposure also produced a sustained increase in intracellular [Ca2+] with characteristics similar to results from earlier studies using primary human bronchial epithelial (NHBE) cells (25, 38) and 16HBE14o− cells (3). Chelation of extracellular Ca2+ with EGTA before addition of Alternaria abolished the [Ca2+]i increase indicating that Alternaria exposure had activated a Ca2+ uptake mechanism located in the apical membrane. Similarly, pretreatment with the cell permeable Ca2+ chelating agent BAPTA-AM also blocked the Alternaria-induced increase in [Ca2+]i and significantly reduced the Isc response, demonstrating that stimulation of anion secretion was dependent on the increase in [Ca2+]i. Comparison of the kinetics of the Alternaria effect on both Isc and [Ca2+]i shows a greater delay in the onset of ATP release and the Ca2+ response. This apparent discrepancy in the temporal cause and effect relationship between the initiation of ATP secretion, Ca2+ uptake and stimulation of Cl− secretion is most likely due to the fact that the ATP and Ca2+ measurements were performed at room temperature (~22°C), whereas the Isc measurements were conducted at 37°C. This 15 degree difference in temperature would be expected to slow the effect of Alternaria on signaling mechanisms involved in regulation of Cl− secretion.
Pretreatment of cells with DIDS also inhibited the Alternaria-evoked increase in [Ca2+]i; however, the ionomycin response was not affected. This result established that the concentration of DIDS used to completely block the Alternaria-induced Isc response inhibited Ca2+ uptake across the plasma membrane, which accounts for the slow, time-dependent decrease in Isc that occurs following inhibition of anion channel activity. Previous experiments with NHBE cells showed that P2X7 receptors were responsible for the increase in [Ca2+]i produced by Alternaria (23) and activation of P2X7 receptors by 2,3-O-(4-benzoylbenzoyl) ATP (BzATP) in pancreatic duct epithelial cells was shown to be blocked by high concentrations (>100 μM) of DIDS (8). P2X receptor activation in 16HBE14o− cells is likely to occur since the present study showed that Alternaria stimulated a robust and sustained increase in ATP release from the epithelium. Moreover, previous studies have shown that P2X7 receptor activation with BzATP increases [Ca2+]i in submandibular gland acinar and duct epithelial cells and stimulates Cl− secretion by activation of CFTR and Ca2+-activated Cl− channels (32, 37, 38).
Recently, Alternaria exposure was shown to induce oxidative stress in NHBE cells and this response could be blocked by pretreatment with oxidant scavengers such as GSH or by Nrf2 modulators such as CDDO-Me (bardoxolone) or resveratrol (19, 21, 48). Oxidative stress following treatment with Alternaria was directly observed in the current study by an increase in fluorescence associated with a cell-permeable ROS indicator. Addition of exogenous GSH completely blocked this change in fluorescence, consistent with ROS scavenging. Pretreatment with ROS scavengers and DPI, an NADPH oxidase/complex 1 electron transport inhibitor (30), also inhibited ATP release and the increase in [Ca2+]i induced by Alternaria, signifying that oxidative stress was critical for triggering signaling pathways that activate ATP secretion and Ca2+-uptake across the apical membrane, ultimately stimulating Cl− secretion. In an earlier study using Calu-3 cells, a human submucosal gland serous cell line, ROS exposure was shown to stimulate CFTR-dependent anion secretion (11). Although measurements of [Ca2+]i were not performed, pretreatment with the adenylyl cyclase inhibitor SQ22536 completely blocked CFTR activation, indicating that ROS exposure produced an increase in cAMP, which is known to regulate both apical CFTR channels and basolateral KCNQ1/KCNE3 K channels. ROS exposure is also known to stimulate the production of prostaglandin-like compounds known as isoprostanes (10). Exposure of Calu-3 cells to the isoprostane 8-iso-prostaglandin E2 was shown to stimulate CFTR-dependent anion secretion in part by activation of the thromboxane A2 (TPα) receptor. Previous studies have shown that ligand-dependent activation of TPα receptors increases adenylyl cyclase activity, leading to an increase in [cAMP] and activation of ion channels involved in anion secretion. A similar mechanism could potentially contribute to the Alternaria-induced CFTR activation observed in the present study. Oxidative stress was also shown to induce increases in [Ca2+]i and Cl− secretion in human nasal epithelial cells and 16HBE14o− cells following treatment with exogenous H2O2 (14). The increase in [Ca2+]i was abolished when EDTA was added to the extracellular media, indicating that H2O2 exposure stimulates a Ca2+ uptake mechanism in the plasma membrane. In addition, oxidative stress also produced inhibition of Na-K-ATPase activity, which resulted in a time-dependent increase in intracellular [Na+]. The results of this study are consistent with the conclusion that oxidative stress induced by Alternaria is responsible for increasing both [Ca2+]i and anion secretion in airway epithelial cells.
Earlier studies of the house dust mite allergen Der p 1, a protein with cysteine protease activity, on epithelial barrier function showed time-dependent disruption of tight junctions and loss of ZO-1 content in 16HBE14o− cell monolayers (50). Morphologic changes in tight junction structure were associated with increased junctional permeability as determined by measurements of [14C]mannitol fluxes. Tight junction disruption was linked to cleavage of ZO-1 and occludin, suggesting that sensitization and allergic inflammatory responses to house dust mite allergens may depend in part on loss of airway epithelial tight junction integrity. Similarly, alkaline serine proteinase activity associated with allergens Pen ch 13 and Asp f 13 derived from two species of fungi, Pencillium and Aspergillus, have been shown to induce the release of PGE2, IL-8, and TGF-β1 from airway epithelial cells and to cleave occludin within the tight junctions of 16HBE14o− cells (44, 46). Although measurements of tight junction permeability were not performed, the authors speculated that occludin degradation could facilitate allergen transport across the epithelial barrier to promote an inflammatory response. More recently, the effects of Pen ch 13 were investigated in a human lung adenocarcinoma cell line (NCI-H441 cells) where it was shown to induce degradation of occludin, ZO-1, and E-cadherin after 2 h of exposure and to reduce TER (6). The results of the present study showed that Alternaria induces a significant decrease in TER within 1 h after exposure. In contrast, treatment with 0.5 mM H2O2 was less complete after 4 h of exposure. Interestingly, pretreatment with several proteinase inhibitors including PMSF, E64, AEBSF, and two MMP inhibitors failed to block the effects of Alternaria on TER. Moreover, in control experiments using a low concentration of trypsin added to the apical surface, TER increased by twofold and this effect was blocked by pretreatment with the serine proteinase inhibitor PMSF, but only slightly reduced by the cysteine proteinase inhibitor E64, a result confirming the serine proteinase selectivity of PMSF. In contrast to the lack of effect of proteinase inhibitors on the decrease in TER evoked by Alternaria, pretreatment with GSH produced a partial inhibition of the response, suggesting that oxidative stress contributed in part to the decrease in electrical resistance of the monolayer. Furthermore, fluorescent dextran flux experiments demonstrated that Alternaria produced a significant increase in tight junction macromolecule permeability within 1 h of exposure, whereas oxidative stress induced by H2O2 required 4 h of treatment to produce a significant increase in permeability that was significantly less than observed after treatment with Alternaria. Pretreatment with the serine proteinase inhibitor AEBSF affected the magnitude of the unidirectional dextran flux after 1 h, but at 4 h the flux was not significantly different than Alternaria treatment alone. These findings indicated that acute oxidative stress predominately affects the electrical resistance of the epithelium, which would increase paracellular ion movement and reduce the shunt resistance of the monolayer. This would ultimately diminish net transepithelial electrolyte transport and reduce overall mucociliary clearance. However, the increase in macromolecule permeability induced by Alternaria appears to be less affected by oxidative stress, but the exact mechanism is not clear from the results of the present study. It is possible that a proteinase(s) that are insensitive to the inhibitors used in this study may be involved, but additional experiments will be required to reveal their identity.
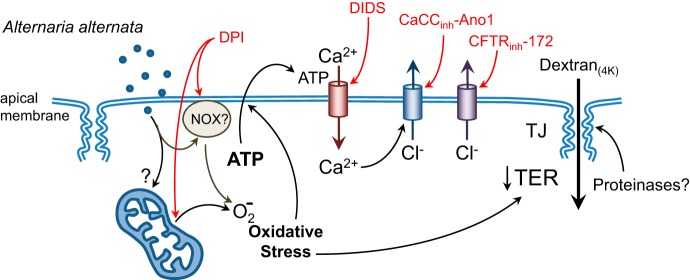
A model that summarizes the key findings of this study is presented in Fig. 7. The results suggest that apical exposure to Alternaria allergens induces oxidative stress, possibly by activation of NADPH oxidase activity or by uncoupling mitochondrial respiration. Consequently, ATP release from the epithelium occurs, which activates Ca2+ uptake through a DIDS-sensitive conductive pathway in the apical membrane. The increase in [Ca2+]i stimulates apical Cl− efflux and transepithelial Cl− secretion, which is completely blocked by CaCCinh-Ano1, a known inhibitor of Ca2+-activated Cl− channels, and CFTRinh-172, an inhibitor of CFTR as well as ClC-2. The increase in oxidative stress also contributes to a time-dependent decrease in transepithelial resistance; however, the increase in dextran permeability appears to be independent of the increase in oxidative stress and may involve proteinase activation.
Fig. 7.
A model summarizing the key findings of this study. See discussion for details.
GRANTS
This work was supported by the National Institute of Biomedical Imaging and Bioengineering (Grant F31 EB-018707 to N. A. Zaidman) and the National Heart, Lung, and Blood Institute (Grant R01 HL-110539 to H. Kita and S. M. O’Grady).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
AUTHOR CONTRIBUTIONS
N.A.Z., H.K., and S.M.O. conceived and designed research; N.A.Z., K.E.O., N.P., F.M., and P.J.M. performed experiments; N.A.Z., K.E.O., N.P., F.M., P.J.M., and S.M.O. analyzed data; N.A.Z., K.E.O., F.M., and P.J.M. prepared figures; N.A.Z. drafted manuscript; N.A.Z., K.E.O., H.K., and S.M.O. edited and revised manuscript; N.A.Z., K.E.O., N.P., F.M., P.J.M., H.K., and S.M.O. approved final version of manuscript; K.E.O., N.P., H.K., and S.M.O. interpreted results of experiments.
REFERENCES
- 1.Adhikari A, Reponen T, Lee SA, Grinshpun SA. Assessment of human exposure to airborne fungi in agricultural confinements: personal inhalable sampling versus stationary sampling. Ann Agric Environ Med 11: 269–277, 2004. [PubMed] [Google Scholar]
- 2.Bacsi A, Dharajiya N, Choudhury BK, Sur S, Boldogh I. Effect of pollen-mediated oxidative stress on immediate hypersensitivity reactions and late-phase inflammation in allergic conjunctivitis. J Allergy Clin Immunol 116: 836–843, 2005. doi: 10.1016/j.jaci.2005.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Boitano S, Flynn AN, Sherwood CL, Schulz SM, Hoffman J, Gruzinova I, Daines MO. Alternaria alternata serine proteases induce lung inflammation and airway epithelial cell activation via PAR2. Am J Physiol Lung Cell Mol Physiol 300: L605–L614, 2011. doi: 10.1152/ajplung.00359.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Boldogh I, Bacsi A, Choudhury BK, Dharajiya N, Alam R, Hazra TK, Mitra S, Goldblum RM, Sur S. ROS generated by pollen NADPH oxidase provide a signal that augments antigen-induced allergic airway inflammation. J Clin Invest 115: 2169–2179, 2005. doi: 10.1172/JCI24422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Boncoeur E, Criq VS, Bonvin E, Roque T, Henrion-Caude A, Gruenert DC, Clement A, Jacquot J, Tabary O. Oxidative stress induces extracellular signal-regulated kinase 1/2 mitogen-activated protein kinase in cystic fibrosis lung epithelial cells: potential mechanism for excessive IL-8 expression. Int J Biochem Cell Biol 40: 432–446, 2008. doi: 10.1016/j.biocel.2007.08.013. [DOI] [PubMed] [Google Scholar]
- 6.Chen JC, Chuang JG, Su YY, Chiang BL, Lin YS, Chow LP. The protease allergen Pen c 13 induces allergic airway inflammation and changes in epithelial barrier integrity and function in a murine model. J Biol Chem 286: 26667–26679, 2011. doi: 10.1074/jbc.M110.193987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chen YC, Statt S, Wu R, Chang HT, Liao JW, Wang CN, Shyu WC, Lee CC. High mobility group box 1-induced epithelial mesenchymal transition in human airway epithelial cells. Sci Rep 6: 18815–18828, 2016. doi: 10.1038/srep18815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Christoffersen BC, Hug MJ, Novak I. Different purinergic receptors lead to intracellular calcium increases in pancreatic ducts. Pflugers Arch 436: 33–39, 1998. doi: 10.1007/s004240050601. [DOI] [PubMed] [Google Scholar]
- 9.Cohen ES, Scott IC, Majithiya JB, Rapley L, Kemp BP, England E, Rees DG, Overed-Sayer CL, Woods J, Bond NJ, Veyssier CS, Embrey KJ, Sims DA, Snaith MR, Vousden KA, Strain MD, Chan DT, Carmen S, Huntington CE, Flavell L, Xu J, Popovic B, Brightling CE, Vaughan TJ, Butler R, Lowe DC, Higazi DR, Corkill DJ, May RD, Sleeman MA, Mustelin T. Oxidation of the alarmin IL-33 regulates ST2-dependent inflammation. Nat Commun 6: 8327, 2015. doi: 10.1038/ncomms9327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cowley EA. Isoprostane-mediated secretion from human airway epithelial cells. Mol Pharmacol 64: 298–307, 2003. doi: 10.1124/mol.64.2.298. [DOI] [PubMed] [Google Scholar]
- 11.Cowley EA, Linsdell P. Oxidant stress stimulates anion secretion from the human airway epithelial cell line Calu-3: implications for cystic fibrosis lung disease. J Physiol 543: 201–209, 2002. doi: 10.1113/jphysiol.2002.022400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cozens AL, Yezzi MJ, Kunzelmann K, Ohrui T, Chin L, Eng K, Finkbeiner WE, Widdicombe JH, Gruenert DC. CFTR expression and chloride secretion in polarized immortal human bronchial epithelial cells. Am J Respir Cell Mol Biol 10: 38–47, 1994. doi: 10.1165/ajrcmb.10.1.7507342. [DOI] [PubMed] [Google Scholar]
- 13.Cuppoletti J, Chakrabarti J, Tewari KP, Malinowska DH. Differentiation between human ClC-2 and CFTR Cl− channels with pharmacological agents. Am J Physiol Cell Physiol 307: C479–C492, 2014. doi: 10.1152/ajpcell.00077.2014. [DOI] [PubMed] [Google Scholar]
- 14.Dazy AC, Auger F, Bailbé D, Blouquit S, Lombet A, Marano F. The toxicity of H2O2 on the ionic homeostasis of airway epithelial cells in vitro. Toxicol In Vitro 17: 575–580, 2003. doi: 10.1016/S0887-2333(03)00092-4. [DOI] [PubMed] [Google Scholar]
- 15.De Lucca AJ. Harmful fungi in both agriculture and medicine. Rev Iberoam Micol 24: 3–13, 2007. doi: 10.1016/S1130-1406(07)70002-5. [DOI] [PubMed] [Google Scholar]
- 16.Denning DW, O’Driscoll BR, Hogaboam CM, Bowyer P, Niven RM. The link between fungi and severe asthma: a summary of the evidence. Eur Respir J 27: 615–626, 2006. doi: 10.1183/09031936.06.00074705. [DOI] [PubMed] [Google Scholar]
- 17.Filali Ben Sidel F, Bouziane H, Del Mar Trigo M, El Haskouri F, Bardei F, Redouane A, Kadiri M, Riadi H, Kazzaz M. Airborne fungal spores of Alternaria, meteorological parameters and predicting variables. Int J Biometeorol 59: 339–346, 2015. doi: 10.1007/s00484-014-0845-1. [DOI] [PubMed] [Google Scholar]
- 18.Gabriel MF, Postigo I, Tomaz CT, Martínez J. Alternaria alternata allergens: markers of exposure, phylogeny and risk of fungi-induced respiratory allergy. Environ Int 89-90: 71–80, 2016. doi: 10.1016/j.envint.2016.01.003. [DOI] [PubMed] [Google Scholar]
- 19.Hristova M, Habibovic A, Veith C, Janssen-Heininger YM, Dixon AE, Geiszt M, van der Vliet A. Airway epithelial dual oxidase 1 mediates allergen-induced IL-33 secretion and activation of type 2 immune responses. J Allergy Clin Immunol 137: 1545–1556.e11, 2016. doi: 10.1016/j.jaci.2015.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Iijima K, Kobayashi T, Hara K, Kephart GM, Ziegler SF, McKenzie AN, Kita H. IL-33 and thymic stromal lymphopoietin mediate immune pathology in response to chronic airborne allergen exposure. J Immunol 193: 1549–1559, 2014. doi: 10.4049/jimmunol.1302984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Iijima K, Kobayashi T, Uchida M, Anderson EL, Squillace D, Kephart GM, O’Grady SM, Kita H. Oxidative stress responses to the fungal allergen Alternaria mediate IL-33 secretion by airway epithelial cells and type 2 immunity in the airways. J Allergy Clin Immunol 137: AB199, 2016. doi: 10.1016/j.jaci.2015.12.779. [DOI] [Google Scholar]
- 22.Itani OA, Lamb FS, Melvin JE, Welsh MJ. Basolateral chloride current in human airway epithelia Am J Physiol Lung Cell Mol Physiol 293: L991–L999, 2007. doi: 10.1152/ajplung.00077.2007. [DOI] [PubMed] [Google Scholar]
- 23.Jarolim K, Del Favero G, Pahlke G, Dostal V, Zimmermann K, Heiss E, Ellmer D, Stark TD, Hofmann T, Marko D. Activation of the Nrf2-ARE pathway by the Alternaria alternata mycotoxins altertoxin I and II. Arch Toxicol 91: 203–216, 2017. doi: 10.1007/s00204-016-1726-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jones WM, Chao AT, Zavortink M, Saint R, Bejsovec A. Cytokinesis proteins Tum and Pav have a nuclear role in Wnt regulation. J Cell Sci 123: 2179–2189, 2010. doi: 10.1242/jcs.067868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kauffman HF, van der Heide S. Exposure, sensitization, and mechanisms of fungus-induced asthma. Curr Allergy Asthma Rep 3: 430–437, 2003. doi: 10.1007/s11882-003-0080-z. [DOI] [PubMed] [Google Scholar]
- 26.Kouzaki H, Iijima K, Kobayashi T, O’Grady SM, Kita H. The danger signal, extracellular ATP, is a sensor for an airborne allergen and triggers IL-33 release and innate Th2-type responses. J Immunol 186: 4375–4387, 2011. doi: 10.4049/jimmunol.1003020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Krysińska-Traczyk E. [Microflora of the farming work environment as an occupational risk factor]. Med Pr 51: 351–355, 2000. [PubMed] [Google Scholar]
- 29.Kustrzeba-Wójcicka I, Siwak E, Terlecki G, Wolańczyk-Mędrala A, Mędrala W. Alternaria alternata and its allergens: a comprehensive review. Clin Rev Allergy Immunol 47: 354–365, 2014. doi: 10.1007/s12016-014-8447-6. [DOI] [PubMed] [Google Scholar]
- 30.Lambert AJ, Buckingham JA, Boysen HM, Brand MD. Diphenyleneiodonium acutely inhibits reactive oxygen species production by mitochondrial complex I during reverse, but not forward electron transport. Biochim Biophys Acta 1777: 397–403, 2008. doi: 10.1016/j.bbabio.2008.03.005. [DOI] [PubMed] [Google Scholar]
- 31.Leino MS, Loxham M, Blume C, Swindle EJ, Jayasekera NP, Dennison PW, Shamji BW, Edwards MJ, Holgate ST, Howarth PH, Davies DE. Barrier disrupting effects of alternaria alternata extract on bronchial epithelium from asthmatic donors. PLoS One 8: e71278, 2013. doi: 10.1371/journal.pone.0071278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li Q, Luo X, Zeng W, Muallem S. Cell-specific behavior of P2X7 receptors in mouse parotid acinar and duct cells. J Biol Chem 278: 47554–47561, 2003. doi: 10.1074/jbc.M308306200. [DOI] [PubMed] [Google Scholar]
- 33.Matsuwaki Y, Wada K, White TA, Benson LM, Charlesworth MC, Checkel JL, Inoue Y, Hotta K, Ponikau JU, Lawrence CB, Kita H. Recognition of fungal protease activities induces cellular activation and eosinophil-derived neurotoxin release in human eosinophils. J Immunol 183: 6708–6716, 2009. doi: 10.4049/jimmunol.0901220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.McGrath JJ, Wong WC, Cooley JD, Straus DC. Continually measured fungal profiles in sick building syndrome. Curr Microbiol 38: 33–36, 1999. doi: 10.1007/PL00006768. [DOI] [PubMed] [Google Scholar]
- 35.Monsó E. Occupational asthma in greenhouse workers. Curr Opin Pulm Med 10: 147–150, 2004. doi: 10.1097/00063198-200403000-00010. [DOI] [PubMed] [Google Scholar]
- 37.Novak I. Purinergic signalling in epithelial ion transport: regulation of secretion and absorption. Acta Physiol (Oxf) 202: 501–522, 2011. doi: 10.1111/j.1748-1716.2010.02225.x. [DOI] [PubMed] [Google Scholar]
- 38.Novak I, Jans IM, Wohlfahrt L. Effect of P2X(7) receptor knockout on exocrine secretion of pancreas, salivary glands and lacrimal glands. J Physiol 588: 3615–3627, 2010. doi: 10.1113/jphysiol.2010.190017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.O’Grady SM, Patil N, Melkamu T, Maniak PJ, Lancto C, Kita H. ATP release and Ca2+ signalling by human bronchial epithelial cells following Alternaria aeroallergen exposure. J Physiol 591: 4595–4609, 2013. doi: 10.1113/jphysiol.2013.254649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Prester L, Macan J. Determination of Alt a 1 (Alternaria alternata) in poultry farms and a sawmill using ELISA. Med Mycol 48: 298–302, 2010. doi: 10.3109/13693780903115402. [DOI] [PubMed] [Google Scholar]
- 41.Rodríguez-Rajo FJ, Iglesias I, Jato V. Variation assessment of airborne Alternaria and Cladosporium spores at different bioclimatical conditions. Mycol Res 109: 497–507, 2005. doi: 10.1017/S0953756204001777. [DOI] [PubMed] [Google Scholar]
- 42.Sanchez H, Bush RK. A review of Alternaria alternata sensitivity. Rev Iberoam Micol 18: 56–59, 2001. [PubMed] [Google Scholar]
- 43.Scudieri P, Sondo E, Ferrera L, Galietta LJ. The anoctamin family: TMEM16A and TMEM16B as calcium-activated chloride channels. Exp Physiol 97: 177–183, 2012. doi: 10.1113/expphysiol.2011.058198. [DOI] [PubMed] [Google Scholar]
- 44.Shen HD, Tam MF, Tang RB, Chou H. Aspergillus and Penicillium allergens: focus on proteases. Curr Allergy Asthma Rep 7: 351–356, 2007. doi: 10.1007/s11882-007-0053-8. [DOI] [PubMed] [Google Scholar]
- 45.Snelgrove RJ, Gregory LG, Peiró T, Akthar S, Campbell GA, Walker SA, Lloyd CM. Alternaria-derived serine protease activity drives IL-33-mediated asthma exacerbations. J Allergy Clin Immunol 134: 583–592.e6, 2014. doi: 10.1016/j.jaci.2014.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tai HY, Tam MF, Chou H, Peng HJ, Su SN, Perng DW, Shen HD. Pen ch 13 allergen induces secretion of mediators and degradation of occludin protein of human lung epithelial cells. Allergy 61: 382–388, 2006. doi: 10.1111/j.1398-9995.2005.00958.x. [DOI] [PubMed] [Google Scholar]
- 47.Thomma BP. Alternaria spp.: from general saprophyte to specific parasite. Mol Plant Pathol 4: 225–236, 2003. doi: 10.1046/j.1364-3703.2003.00173.x. [DOI] [PubMed] [Google Scholar]
- 48.Uchida M, Anderson EL, Squillace DL, Patil N, Maniak PJ, Iijima K, Kita H, O’Grady SM. Oxidative stress serves as a key checkpoint for IL-33 release by airway epithelium. Allergy. [Epub ahead of print]. doi: 10.1111/all.13158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Viegas C, Carolino E, Malta-Vacas J, Sabino R, Viegas S, Veríssimo C. Fungal contamination of poultry litter: a public health problem. J Toxicol Environ Health A 75: 1341–1350, 2012. doi: 10.1080/15287394.2012.721165. [DOI] [PubMed] [Google Scholar]
- 50.Wan H, Winton HL, Soeller C, Gruenert DC, Thompson PJ, Cannell MB, Stewart GA, Garrod DR, Robinson C. Quantitative structural and biochemical analyses of tight junction dynamics following exposure of epithelial cells to house dust mite allergen Der p 1. Clin Exp Allergy 30: 685–698, 2000. doi: 10.1046/j.1365-2222.2000.00820.x. [DOI] [PubMed] [Google Scholar]
- 51.Weyemi U, Redon CE, Choudhuri R, Aziz T, Maeda D, Boufraqech M, Parekh PR, Sethi TK, Kasoji M, Abrams N, Merchant A, Rajapakse VN, Bonner WM. The histone variant H2A.X is a regulator of the epithelial-mesenchymal transition. Nat Commun 7: 10711–10723, 2016. doi: 10.1038/ncomms10711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wolf J, O’Neill NR, Rogers CA, Muilenberg ML, Ziska LH. Elevated atmospheric carbon dioxide concentrations amplify Alternaria alternata sporulation and total antigen production. Environ Health Perspect 118: 1223–1228, 2010. doi: 10.1289/ehp.0901867. [DOI] [PMC free article] [PubMed] [Google Scholar]