Abstract
Purpose
The purpose of this investigation was to evaluate human Thiel-embalmed cadavers with the addition of extracorporeal driven ante-grade pulsatile flow in the aorta as a model for simulation training in interventional techniques and endovascular device testing.
Materials and Methods
Three human cadavers embalmed according to the method of Thiel were selected. Extracorporeal pulsatile ante-grade flow of 2.5 L per min was delivered directly into the aorta of the cadavers via a surgically placed connection. During perfusion, aortic pressure and temperature were recorded and optimized for physiologically similar parameters. Pre- and post-procedure CT imaging was conducted to plan and follow up thoracic and abdominal endovascular aortic repair as it would be in a clinical scenario. Thoracic endovascular aortic repair (TEVAR) and endovascular abdominal repair (EVAR) procedures were conducted in simulation of a clinical case, under fluoroscopic guidance with a multidisciplinary team present.
Results
The Thiel cadaveric aortic perfusion model provided pulsatile ante-grade flow, with pressure and temperature, sufficient to conduct a realistic simulation of TEVAR and EVAR procedures. Fluoroscopic imaging provided guidance during the intervention. Pre- and post-procedure CT imaging facilitated planning and follow-up evaluation of the procedure.
Conclusion
The human Thiel-embalmed cadavers with the addition of extracorporeal flow within the aorta offer an anatomically appropriate, physiologically similar robust model to simulate aortic endovascular procedures, with potential applications in interventional radiology training and medical device testing as a pre-clinical model.
Electronic supplementary material
The online version of this article (doi:10.1007/s00270-017-1643-z) contains supplementary material, which is available to authorized users.
Keywords: Cadaveric perfusion, Endovascular training, Endovascular aortic aneurysm repair (EVAR)
Introduction
The need for training of the operator through clinical simulation within the field of interventional radiology is widely recognized [1, 2]. Despite dissimilar anatomy, ethical issues and high cost, animal models continue to provide a high-fidelity training model, with blood flow, percutaneous access and physiological function. Alternatively, human cadaveric models provide more relevant anatomy and anatomical variation in which, for the purposes of aortic endovascular procedures, the human cadaveric aorta in particular has no accurate alternative animal model in terms of vessel geometry, calibre and physiological conditions [3, 4].
Unlike other cadaveric preservation methods, Thiel [5, 6]-embalmed cadavers retain flexibility, colour, tone, have extended durability, negligible infection hazard and, most importantly for an interventional training model, vascular patency. The application of human Thiel-embalmed cadavers in medical research and teaching is increasingly recognized, due to these beneficial properties [7]. Research and development into the introduction of function to fresh human cadaveric models was initiated by Garrett [8], who first described the addition of flow into the blood vessels of fresh frozen cadavers, facilitating fluoroscopic imaging and establishing human cadavers as a model for endovascular training. Garrett’s cadaveric models were not without limitations. Aside from the short shelf life, the biological hazard and increased costs associated with fresh cadavers, the short arterial circuits with low flow rates, prevented peripheral access and therefore constrained their application as a training tool for full clinical simulation and endovascular percutaneous approach. Despite these limitations, the potential benefits of using human cadavers for interventional radiology training were established by this seminal work.
A more recent study introduced the use of Thiel-embalmed cadavers with added function for interventional training [9]. This work demonstrated the successful retrograde filling of the Thiel-embalmed cadaveric aorta to conduct a vascular intervention (TAVI). This study was limited as the cadaveric model was filled but lacked ante-grade pulsatile flow within the arterial system.
This technical note describes a flexible, robust and functional Thiel human cadaveric model with extracorporeal driven ante-grade pulsatile aortic flow, demonstrating the potential of this model in endovascular surgical training, and pre-clinical medical device evaluation.
Materials and Methods
Three human cadavers were preserved using a previously described technique by Thiel. A 2-cm incision was made directly percutaneously into the apex of the heart, and a size 8 endotracheal tube (Portex, Smith Medical, USA) was inserted into the ascending aorta where the balloon was inflated to secure it.
Perfusion
Extracorporeal flow was introduced directly into the cadaver vasculature via the connecting tube linking the aorta and the heart lung bypass machine (HL30 Maquet, Germany) with a modified master pump in order to recreate a pulsatile-type flow at 60 beats per minute. The master pump was prepared with 6 m of 13-mm-diameter silicone–platinum-coated tubing (Silex, UK), delivering a flow rate of approximately 2.5 L per min as the standard setting.
Temperature Assessment
Endovascular devices with thermal memory require the temperature within the aortic lumen to be within 35–39 degrees centigrade. To achieve this temperature at the deployment site, the reservoir was heated to 60 degrees centigrade prior to perfusion and temperatures were continuously measured and recorded (TS2, Optocon, Germany). Fibre-optic thermocouple (Fotemp-4, Optocon, Germany) was located within the circuit and temperatures during perfusion recorded.
Pressure Measurement
A continuous invasive aortic pressure measurement was taken post-intervention in two of the cadavers at three separate sites: extracorporeal circuit descending thoracic aorta and abdominal aorta. An arterial catheter (Careflow, BD, USA) was inserted into each site and attached to a pressure transducer (Standard, Baxter, UK) using a standard giving set (Protect-A-Line 2, Vygon, France) with a high-pressure bag of saline prepared to retain line patency (0.9% Saline, Baxter, UK). The pressure transducer was connected to a haemodynamic monitoring system (M3046A, HP, Germany) to provide a continuous pressure measurement (Table 3).
Table 3.
Pressure assessment during standard flow conditions (mmHg)
| MAP cadaver 1 | MAP cadaver 2 | |
|---|---|---|
| Arch of aorta | 38.8 | 52 |
| Distal aorta | 37.5 | 54 |
| Extracorporeal | 78.1 | 110 |
Multi-Modal Imaging
The following standard parameters were used throughout interventions.
Pre-intervention Planning
Arterial land markings for abdominal aortic stent grafting and sizing of the aorta to select appropriate device were conducted by an interventional radiologist, using the CT data, allowing the appropriate device to be selected. Secondly, nearby vessels were assessed for arterial disease to determine risks, e.g. great vessel or renal artery disease. Finally, the access vessels were assessed for lumen tortuosity and diameter to allow “safe” device delivery, deployment and delivery system retrieval.
Intervention
The simulation was designed as a multidisciplinary clinical scenario with consultant radiologist, nurse and radiographer present.
All devices were manufactured by Captivia Delivery Systems, Medtronic Inc., USA. One cadaver had two procedures, a thoracic stent graft followed by an abdominal stent graft, bifurcating to the left and right iliac arteries of the cadaver.
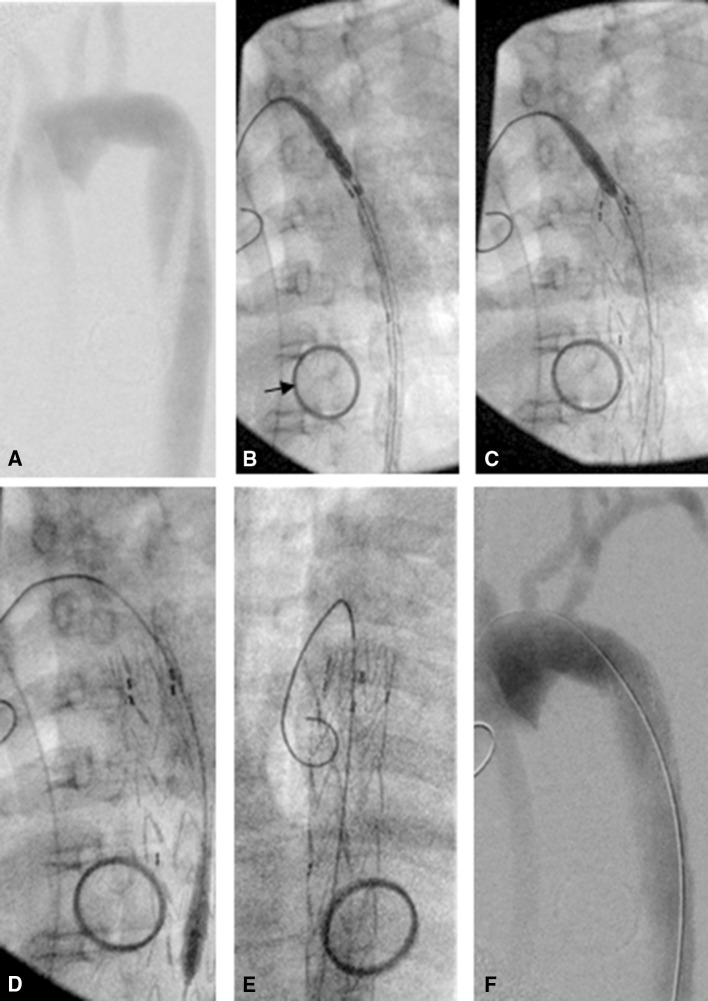
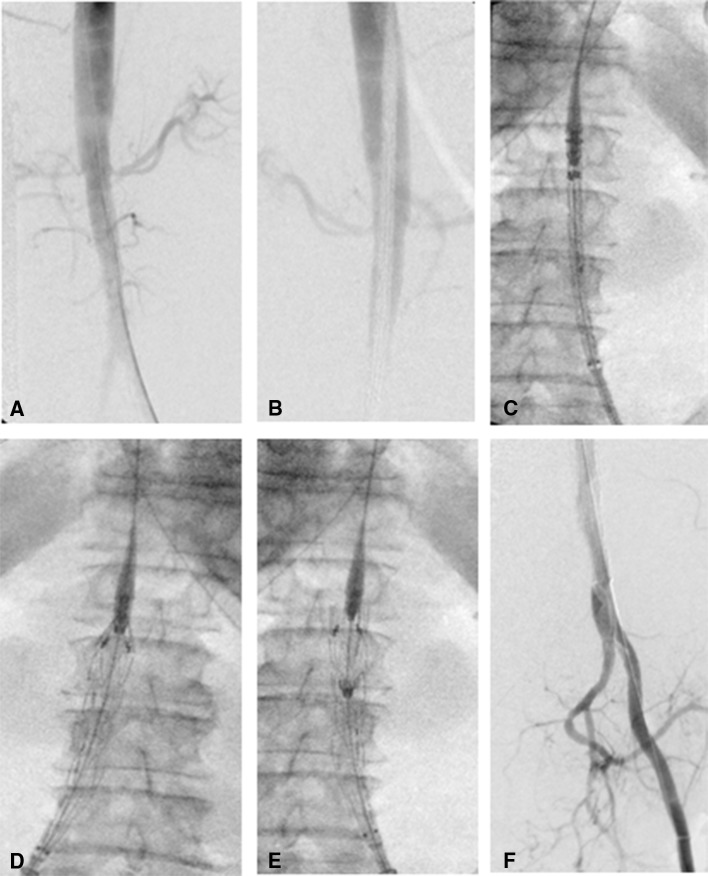
A range of different aortic stent graft types were deployed (Table 1): thoracic tube stent graft (Fig. 1), an aorto-uniiliac (Fig. 2) and bifurcated stent graft (Fig. 3). For all interventions, aortic access was secured via the left or right common femoral artery with a 22Fr Sheath (DrySeal sheath, Gore, USA) in situ. A straight stiff guide wire, 0.035 inch diameter × 260 mm long (Amplatz Super Stiff, Boston Scientific), was advanced to the previously planned target location under fluoroscopic guidance. The devices were delivered and deployed according to the manufacturer’s instructions.
Table 1.
Imaging parameters
| Modality | Equipment | Settings | Contrast |
|---|---|---|---|
| X-ray fluoroscopy | OEC Elite 9900 C Arm (GE, USA) | Fluoroscopic imaging 8–12 f.p.s and digital subtraction angiography 25 f.p.s | 20-ml Bolus contrast (Omnipaque 300, GE Healthcare, Auckland) was introduced manually |
| CT | Siemens Biograph mCT scanner (Siemens, Erlangen DE) | Slice thickness 0.6 mm, pitch 0.8, kV 120. Effective mat rotation time, 0.5 s. | Bolus-tracked contrast was initiated manually (Omnipaque 300, GE Healthcare, New Zealand). |
Fig. 1.
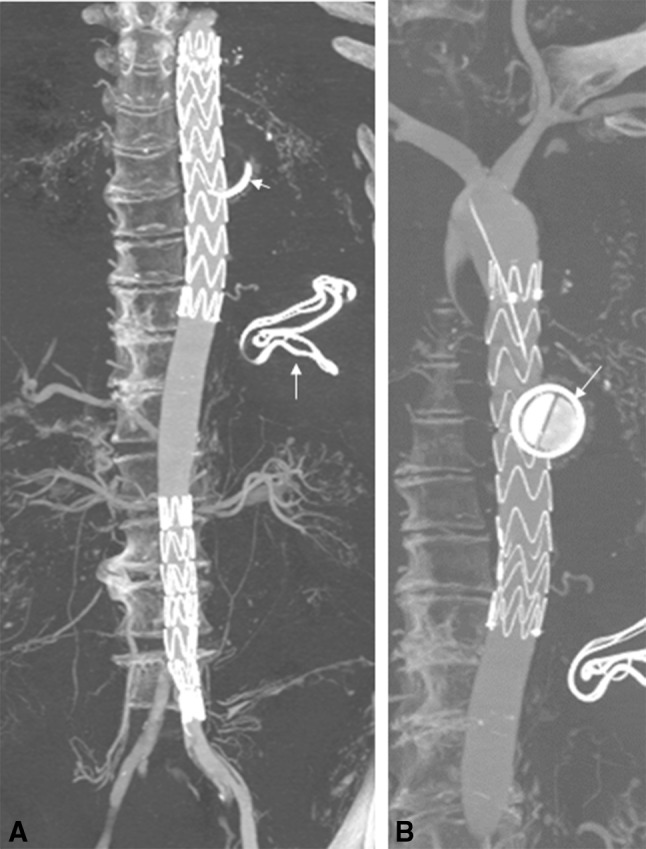
Post-procedure CT scan. CT angiogram (Table 1), MIP 3D reconstruction, allowing vessel and device evaluation (arrows point at post-mortem devices unrelated to this work). A Thoracic stent and abdominal stent conducted under fluoroscopic guidance, B thoracic stent in situ
Fig. 2.
Valiant thoracic stent graft Captivia Delivery System intervention to the thoracic aorta of a Thiel-embalmed human cadaver. A Digital subtraction angiography cadaveric aorta with Captivia catheter located in the descending aorta. B Fluoroscopic-guided catheter advanced to position in cadaveric aortic arch. Arrows points to ring and swab placed during post-mortem training which was not related to this procedure. C Stent graft deployed in the thoracic aorta. D Catheter undocked and withdrawn. E Guide wire remains in device to allow angiographic catheter to be placed in aorta. F Post-procedure digital subtraction angiography demonstrating patent aorta
Fig. 3.
Stent graft intervention to abdominal aorta of Thiel-embalmed human cadaver (thoracic Valiant device and Captivia Delivery System used to demonstrate viability of technique). A ipsilateral catheter placement—DSA aortogram [Table 1). Renal arteries evident to plan location of device deployment, B DSA Captivia catheter evident, located in aorta to deliver device to infra-renal position, C fluoroscopic image of catheter in situ, D Valiant stent graft being deployed under fluoroscopic guidance, E guide wire remains in device to allow angiographic catheter to be placed in aorta, F post-procedure DSA of distal abdominal aorta and left iliac artery which branches distally into internal and external iliac artery. Right iliac occluded by stent graft and therefore not visible on image
Post-intervention Evaluation
Stent graft placement was evaluated post-procedure by fluoroscopic and DSA angiography. Post-procedure CT was conducted in one cadaver to evaluate device placement, migration and endoleak (Fig. 1).
Results
The simulation followed the pathway similar to live recipients of these devices, with pre-procedure CT providing images to plan the procedure, procedural angiography and post-procedure CT to evaluate the deployed devices (Fig. 4). The arterial landmarks were identified, and the presence and severity of any iliac arterial stenotic disease or tortuosity were noted in all cases. The pre-procedure CT allowed planning of the stent graft procedure and patient-specific co-morbidities into account in the planning simulation, while post-procedure CT allowed assessment of complications such as endovascular leak.
Fig. 4.
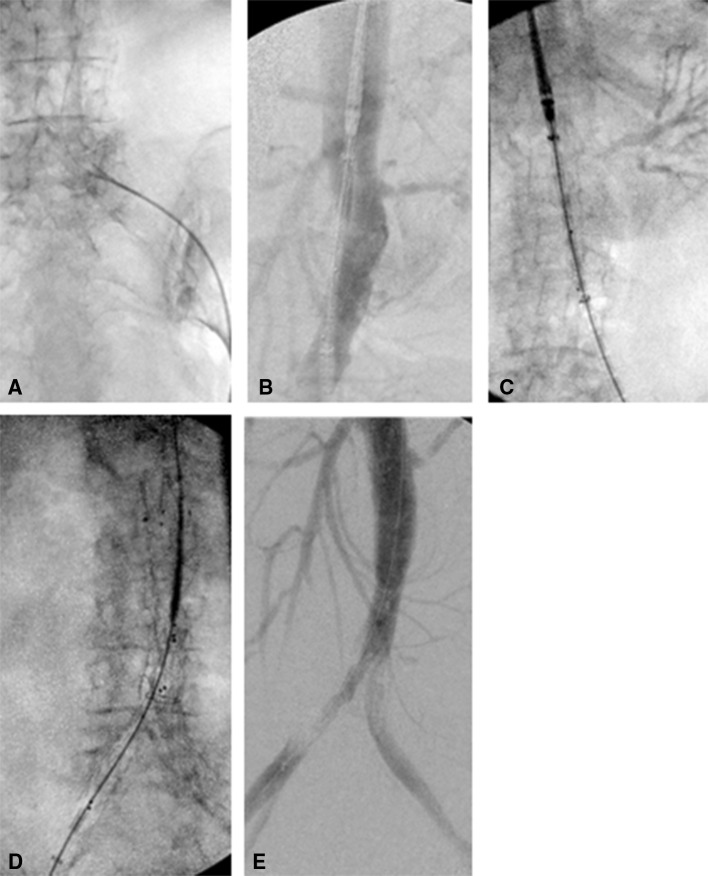
Bifurcating Endurant stent graft intervention to Thiel-embalmed human cadaver. A Fluoroscopic image of pigtail angiographic catheter advancing over guide wire from common femoral artery access paint. B Pre-deployment digital subtraction angiography showing renal arteries and superior mesenteric artery. C Catheter advanced to desired location to locate device in an infra-renal position. D Contralateral leg deployment, catheter advanced to dock into device. Leg deployed, E post-procedure DSA demonstrating patent device
The intravascular temperatures in two cadaveric models were evaluated, with an initial average intraluminal temperature of 16 degrees centigrade which increased to 39.1 degrees centigrade after 4-min perfusion at 2.5 L per min perfusion. Pressures within the aorta were measured during standard perfusion conditions detailed in Table 2.
Table 2.
Aortic stent grafts used during the procedures
| Fig. No. | Device | Dimensions (mm) | Size | Implant location |
|---|---|---|---|---|
| Figure 1 | Valiant thoracic | 204 × 30 | 22Fr | Thoracic aorta |
| Valiant thoracic | 164 × 30 | 22Fr | Thoracic aorta | |
| Figure 2 | Valiant thoracic | 124 × 30 | 22Fr | Distal abdominal aorta |
| Endurant II system | 181 × 28 | Abdominal aorta—bifurcating to L& R iliac arteries | ||
| Figure 3 | Endurant II system | 9319 to 13 | Abdominal aorta—iliac artery |
Four aortic stent grafts were successfully deployed according to the procedure plan, in all three cadavers (Figs. 2, 3 and 4). During the procedures, it was noted by the radiologists that the haptic and geometric sensations during the procedure were very similar to that experienced in a live human procedure and superior to that of an animal simulation (85 kg pigs). In particular, the proximal stent positioning, stent deployment, placement of extension pieces, contralateral limb selection and deployment, device retrieval, post-implantation balloon dilatation and angiography check for endoleaks, were found to closely simulate the clinical procedure. Ancillary angioplasty of a coincidental renal artery stenosis and iliac stenosis was also performed with similar favourable results. Post-procedure CT confirmed the precise placement of the TEVAR and EVARS with no evidence of an endoleak (Fig. 1).
Discussion
Our model is the first to fully perfuse the aorta of a Thiel-embalmed human cadaver by the addition of extracorporeal ante-grade pulsatile flow, originating from the ascending aorta to the entire arterial tree along with temperature and pressure measurement. This approach uniquely provides arterial access from the common femoral arteries and potentially other peripheral vessels to conduct interventions. A great advantage of this model is pulsatile flow within the vessels which allows palpation or ultrasound identification of target vessels followed by standard vascular access technique, providing a closer simulation of the human experience in comparison with animal models, or synthetic and virtual models [10]. Simple virtual training simulators such as Mentice (VIST LAB, Mentice AB, Gothenburg, Sweden) and anatomical simulators with fluoroscopic imaging, such as silicon vascular phantoms (Elstrat, Switzerland), are beneficial tools for gaining basic interventional skills and procedural knowledge but are limited to individual learning and do not provide the complexity of human anatomy, the haptic properties and realism of clinical simulation that the cadaveric model can provide (Table 3).
The use of Thiel cadavers means a longer “shelf life” with negligible infection hazard compared to fresh frozen cadavers. The application of this Thiel cadaveric perfusion model for endovascular techniques of aortic stent graft placement in the thoracic and abdominal aorta (both aorto-uniiliac and bifurcated grafts) has been shown to be suitable both for device testing and also as an aid to operator training. Particular cadaveric advantages are the range of human anatomy, the presence of complicating co-morbidity such as vessel tortuosity, arterial stenosis and the use of aortic perfusion. In addition, for the purposes of both device testing and operator training, the Thiel cadaveric procedures use “off-the-shelf” standard medical devices (guide wires, catheters, balloons, stent grafts) and standard imaging systems, both fluoroscopic and CT units, imaging contrast media and image acquisition protocols.
In comparison with other published cadaveric models, the Thiel cadaveric model we describe has the capacity to introduce higher volumes of flow at greater velocities due to size of the extracorporeal connection to the aorta. The increased flow rates allow perfused organs such as the kidneys to be fluoroscopically imaged, offering further potential training in interventional techniques as yet to be explored.
Intravascular pressures measured within the cadaveric aorta were lower than normal human intra-aortic pressures. This was expected as peripheral resistance is not present within the cadaver model. Despite the pressure not matching “normal” human in vivo aortic pressure, the flow within the aorta was sufficient to conduct the simulated intervention.
Using Thiel-embalmed cadavers to conduct this type of training limits its application to facilities where these resources and expertise are available. This limitation is balanced by the lack of good In Vivo alternatives and therefore offers a specialized solution to the unmet need in this field.
A further limitation of human cadaveric models includes the absence of pathology in predictable and desired locations; in this model, aneurysmal or dissection pathology was absent. However, the cadaveric model did offer vascular pathologies with iliac and renal stenosis being identified, which added to the complexity and relevance of the simulation.
This study demonstrates the Thiel human cadaveric model as a robust, reproducible, high-quality model that is ethically sound, to train multidisciplinary teams in complex endovascular interventions.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
This work was funded by Dr Hadwen Trust. The Dr Hadwen Trust (DHT) is the UK’s leading non-animal biomedical research charity that exclusively funds and promotes human-relevant research that replaces the use of animal’s while supporting the progress of medicine. This project has received funding from the European Union’s Seventh Framework Programme for research, technological development and demonstration under Grant agreement no 324487. This work followed on from EU 7th Framework-funded IIIOS (FP7/2007-2013, under Grant agreement no 238802). The researchers are very grateful to Professor Sir Alfred Cuschieri for the use of equipment during this research. We would like to thank the people who made this work possible by the generous donation of their bodies to medical research and science.
Compliance with Ethical Standards
Conflict of interest
On behalf of all the authors, the corresponding author states that there is no conflict of interest.
Ethical Approval
Study procedures involving human cadaveric participants were in accordance with the institutional research ethics committee and the Anatomy Act (Scotland) 1984 as amended in 2006.
Informed Consent
It does not apply to this study.
Footnotes
Electronic supplementary material
The online version of this article (doi:10.1007/s00270-017-1643-z) contains supplementary material, which is available to authorized users.
References
- 1.Belli AM, Reekers JA, Lee M. The importance of curriculum-based training and assessment in interventional radiology. Cardiovasc Interv Radiol. 2014;37:8–10. doi: 10.1007/s00270-013-0750-8. [DOI] [PubMed] [Google Scholar]
- 2.Diethrich EB. Will our training programs meet the challenges of increasingly complex endovascular aortic surgery? Semin Vasc Surg. 2012;25(3):177–179. doi: 10.1053/j.semvascsurg.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 3.Stefanidis D, Yonce TC, Green JM, Coker AP. Cadavers versus pigs: which are better for procedural training of surgery residents outside the OR? Surgery. 2013;154(1):34–37. doi: 10.1016/j.surg.2013.05.001. [DOI] [PubMed] [Google Scholar]
- 4.Gilbody J, Prasthofer AW, Ho K, Costa ML. The use and effectiveness of cadaveric workshops in higher surgical training: a systematic review. Ann R Coll Surg Engl. 2011;93:347–352. doi: 10.1308/147870811X582954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thiel W. The preservation of the whole corpse with natural color. Ann Anat. 1992;174:185–195. doi: 10.1016/S0940-9602(11)80346-8. [DOI] [PubMed] [Google Scholar]
- 6.Thiel W. Supplement to the conservation of an entire cadaver according to W. Thiel. Ann Anat. 2002;184:267–269. doi: 10.1016/S0940-9602(02)80121-2. [DOI] [PubMed] [Google Scholar]
- 7.Eisma R, Wilkinson T. From, “silent teachers” to models. PLoS Biol. 2014;12(10):e1001971. doi: 10.1371/journal.pbio.1001971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Garrett HE. A human cadaveric circulation model. J Vasc Surg. 2001;33(5):1128–1130. doi: 10.1067/mva.2001.114214. [DOI] [PubMed] [Google Scholar]
- 9.Chevallier C, Willaert W, Kawa E, et al. Postmortem circulation: a new model for testing endovascular devices and training clinicians in their use. Clin Anat. 2014;27(4):556–562. doi: 10.1002/ca.22357. [DOI] [PubMed] [Google Scholar]
- 10.Gould D. Using simulation for interventional radiology training. Br J Radiol. 2010;83(991):546–553. doi: 10.1259/bjr/33259594. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.