Abstract
Frizzled receptors are the mediators of the wnt canonical and non-canonical pathways, which play fundamental roles in cell differentiation and organism development. A large body of work indicates that dysregulation of wnt signalling is a feature of oncogenic transformation, but most of the studies published so far focus on the assessment of the consequences of aberrations of the canonical pathway in human cancer. In this review, we discuss the emerging role of the wnt non-canonical pathway regulated by frizzled receptor 6 (Fzd6) in the pathogenesis of different types of human malignancies. The function played by Fzd6 in the physiology of normal and cancer cells has been highlighted in the view that an increased knowledge of the signalling pathways upstream and downstream of this receptor could ultimately result in the identification of new targets for cancer therapy.
Introduction
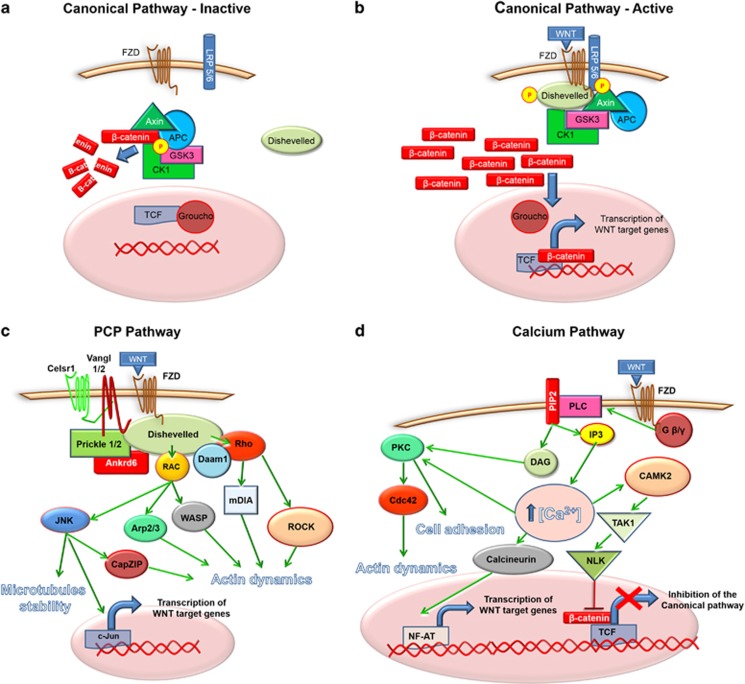
Frizzled receptors (FZDs) are seven-transmembrane-spanning proteins belonging to a sub-class of the G protein-coupled receptor family.1 In humans, there are 10 FZD receptors each encoded by a separate gene. Interaction of wnt ligands with FZDs results in the activation of the canonical or non-canonical wnt signalling pathways regulating embryonal development, cell proliferation, motility, polarity, stem cell maintenance and tissue differentiation.2 The main molecules involved in the canonical and non-canonical pathways are illustrated in Figure 1. Despite recent advances in the understanding of the function of FZDs and wnts, some aspects of the wnt pathway are still poorly understood. This is probably a consequence of the intricate connections between the 19 wnt ligands and the 10 FZDs, making the signalling pathway an intrinsically complicated system to study. Classically, activation of the wnt canonical pathway leads to the stabilization and translocation of β-catenin into the cell nucleus, where it promotes the transcription of wnt-associated genes.3 The term non-canonical pathway refers to a group of wnt-dependent signalling pathways which do not lead to the cytoplasmic stabilization of soluble β-catenin. Two of these pathways have been well characterized: the planar cell polarity (PCP) and the wnt-Calcium pathway. Although, for simplicity, wnt signalling is often dichotomized in two branches, the canonical and the non-canonical pathways often overlap to coordinate complex cellular responses.
Figure 1.
The figure illustrates the wnt canonical (a, b), non-canonical (c, d) pathways and the main molecules involved.
Nusse and Varmus were the first to observe that MMTV (mouse mammary tumour virus) infection induces mammary tumours in mice through activation of the WNT1proto-oncogene.4 Since then, many distinct alterations in the wnt pathway have been related to carcinogenesis and tumour development. Deregulated expression or mutations of FZDs genes have been observed in various human malignancies, indicating the prominent role of these signalling molecules in cancer.5, 6, 7, 8, 9, 10, 11, 12
Physiology of Frizzled receptor 6
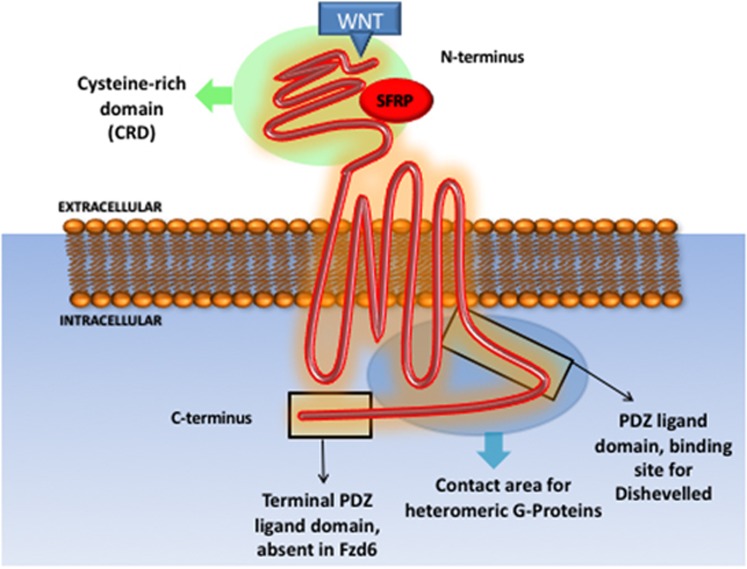
Frizzled receptor 6 (Fzd6) is a 706 amino acid, 7 transmembrane domain receptor, encoded by the FZD6 gene, located in chromosome 8 (8q22.3-q23.1). Similarly, to other FZD receptors, Fzd6 contains an extracellular cysteine-rich domain, the binding site for wnt proteins13 and Soluble Frizzled-related proteins.14 In contrast to other members of the Fzd family, Fzd6 does not contain a second C-terminal PDZ domain-binding motif15 (Figure 2).
Figure 2.
Frizzled receptors domains and interacting proteins.
Fzd6 has been shown to regulate both canonical and non-canonical pathways, but most reports indicate a prevalent role in the non-canonical pathway.
Golan and collaborators observed that the ectopic expression of FZD6 in HEK293 cells did not result in the activation of the canonical pathway, even in the presence of canonical ligands. Conversely, the ectopic expression of FZD1 led to a significant activation of the canonical pathway in similar conditions. The group identified Fzd6 as an inhibitor of the canonical pathway through activation of TAK1/NLK kinases, which in turns reduce activation of β-catenin target genes via phosphorylation of TCF/LEF transcription factors.16 These early results indicated that Fzd6 could be upstream of the wnt/Calcium pathway. Accordingly, studies on canine kidney epithelial cells demonstrated that Fzd6 physically binds to the canonical ligand wnt4, but ectopic expression of FZD6, together with WNT4, did not induce any greater wnt4-dependent activation of TCF/LEF reporter, suggesting a non-canonical role for Fzd6 in this system.17 Interaction of Fzd6 with wnt4 was also observed in murine hematopoietic precursor cells; depletion of Fzd6 abrogates the wnt4 dependent expansion of these cells, suggesting that the receptor is critically required for wnt4 function.18 In another study conducted in HelaS3 cells it was reported that downregulation of FZD6 did not change accumulation of nuclear β-catenin, nor the activation of RAC following stimulation with the canonical ligands wnt3a or wnt5a, further suggesting a non-canonical role of the receptor. The same study also revealed that the Fzd6 cysteine-rich domain does not bind to wnt3a and wnt5a.19 A FRAP assay was conducted to identify Fzd6 ligands by measuring the membrane shift of Fzd6 following stimulation with different wnts. The analysis indicated that Fzd6 binds to wnt3a, wnt4, wnt1, wnt5a, wnt9b, wnt10b and wnt16b. However, this technique cannot discern between direct and indirect binding, and there is the possibility that the different ligands interact with other receptors in the proximity of Fzd6.20
In spite these and other studies suggest an involvement of Fzd6 with the non-canonical pathway, other reports indicate otherwise. For example, it was observed that Fzd6 signalling activates β-catenin in a study of patients affected by nail dysplasia caused by germline FZD6 mutations.21 This study reported that wnt3a signalling causes β-catenin accumulation in healthy, but not FZD6-mutant, fibroblasts, indicating a canonical role of Fzd6 in this context. Interestingly, a recent paper revealed that Fzd6 interacts with the heterotrimeric proteins Gαi1/Gαq and that the complex is dissociated following the stimulation with wnt5a.22 However, the G-protein-mediated signalling cascade downstream of Fzd6 and its putative role in disease and cancer remains largely obscure. Fzd6 might also mediate cellular signals through the activation of the c-Jun N-terminal Kinase (JNK), since Fzd6high neuroblastoma stem cells contain higher levels of phosphorylated JNK compared to the Fzd6 negative counterparts.23 JNK is required for convergent extension movements that characterize gastrulation in frogs, controlled by Wnt/PCP signalling, suggesting that Fzd6 could potentially regulate PCP and JNK activation.24
The physiological function of FZD6 is inferred by the analysis of genetic disorders that have been linked to mutations of the locus. For instance, there is a strong correlation between frameshift mutations of FZD6 and neural tube defects, such as failure of neural tube closure along the entire body axis, suggesting the importance of Fzd6 in directing cell migration and cell polarity during brain morphogenesis.25 FZD6 is also important for nail development, since homozygous frameshift mutations result in the onset of a rare form of nail dysplasia. The mutations interfere with the correct localization of the receptor at the cell membrane, resulting in reduced or abrogated signal transduction.21 The disease is characterized by the abnormal pattering and shape of nails, consistent with a role in the control of PCP by Fzd6.
In agreement with the observations in patients bearing FZD6 mutations, FZD6-null mice manifest a phenotype characterized by defects in cell migration and tissue polarization during organogenesis. FZD6-null mice are viable, but present a disorganized orientation of the hair follicles and defects in the shaping of claws.21, 26 Interestingly, studies conducted on FZD6 and FZD3 double knockout mice revealed a high level of redundancy between the receptors: only abrogation of both FZD6 and FZD3 (but not the knockout of a single receptor) caused severe embryonic defects in neural tube closure and planar orientation of hair bundles on a subset of auditory and vestibular sensory cells.27 Further studies on FZD6-null mice suggested that FZD6 might also be implicated in limiting the activation of platelets.28
In conclusion, the experimental evidence gathered so far suggests that Fzd6 is required to direct the orientation and migration of cells during organogenesis, probably via activation of PCP signalling. One could speculate that reactivation of PCP in adult tissues could contribute to the metastatic dissemination of cancer cells. In the next chapter, we will review recent findings strongly suggesting that Fzd6 does play a role in tumour development and metastasis.
Role of Fzd6 and non-canonical WNT signalling in cancer
It is now well established that the canonical, β-catenin-dependent wnt signalling pathway plays a major role in human cancer.29, 30, 31 Whether the PCP and non-canonical wnt pathways are also major players in oncogenic transformation is still a matter of discussion. There is some evidence though that cancer cells hijack the WNT non-canonical signalling pathway to acquire the ability to migrate and metastasize. For example, overexpression of WNT5a in melanoma and gastric cancer causes increased cancer cell migration and metastasis, but not β-catenin activation.32, 33 Wnt5a effects on cell migration and invasion are dependent on the activation of protein kinase C (PKC) and other effectors of the WNT/calcium pathway.33, 34 Interestingly, Fzd6 has also been shown to activate the WNT/calcium pathway and PKC in human cells and in Xenopus.16, 35 Wnt5a can, among other receptors, bind to Fzd6 and both ligand and receptor are often overexpressed in glioblastoma, suggesting that wnt5a and Fzd6 could potentially cooperate to drive migration and invasion in these tumours.36, 37
Several PCP core components are aberrantly expressed in tumours, contributing to cancer cell proliferation and migration. Overexpression of VANGL-1, a key component of the PCP pathway, is associated with an increased risk of relapse in breast cancer patients and its knockdown reduces breast cancer cell migration.38 VANGL-2 overexpression was also linked to enhanced tumour cell proliferation and poorer prognosis in breast cancer through a mechanism that involves the JNK pathway.39 Notably, mutations of VANGL1, like mutations of FZD6, are associated with neural tube defects, suggesting that these two PCP effectors lie in the same pathway and their aberrations might contribute to cancer.40 Other core components of the PCP such as Celsr1, Prickle1, Fzd3, Fzd7, Dvl2, Dvl3 and casein kinase 1 (CK1)-ε were found upregulated in B lymphocytes of patients with chronic lymphocytic leukaemia. PCP activation in these patients predicts a worse prognosis and is mechanistically implicated in transendothelial cell migration.41
FZD6 is critically required for the malignant transformation of B-cells in Eμ-TCL1 mice that develop chronic lymphocytic leukaemia. FZD6 expression drastically increases during leukaemogenesis, and its key role in B-cells transformation was elegantly demonstrated by crossing Eμ-TCL1 with FZD6-null mice. Mice harbouring FZD6 null alleles develop less leukaemia and have a prolonged survival.42 Overall these studies suggest that core PCP molecules are not only pivotal in regulating the migration of cancer cells and the development of metastasis, but could also be required for tumourigenesis.
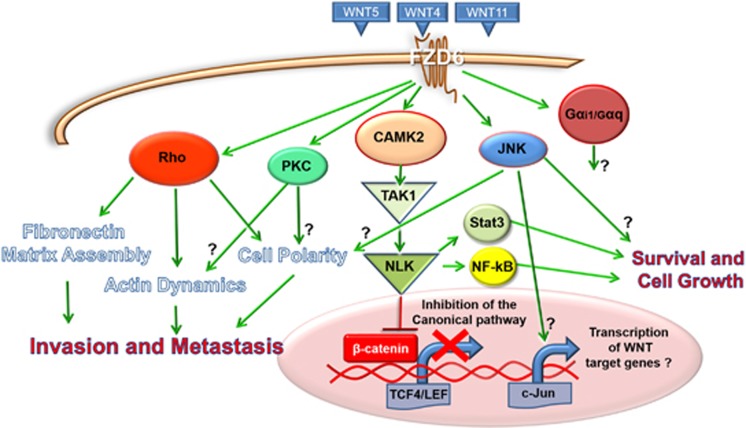
In recent studies, it was investigated whether cues emanating from the cancer associated stroma could regulate PCP. Luga and colleagues demonstrated that exosomes secreted by cancer-associated fibroblasts can instigate breast cancer cells to acquire a motile phenotype by mobilizing PCP core components such as FZD6, DVL1, VANGL1, PK1 and stimulating the autocrine secretion of the non-canonical ligand Wnt11. Knockdown of DVL1, VANGL1, PK1 inhibited cell motility and the migratory phenotype of highly metastatic MDA-MB-231 cells, and lung metastasis were strongly reduced in mice following depletion of Pk-1.43 In agreement with these findings, our group has recently demonstrated that the FZD6 gene is frequently amplified and overexpressed in triple negative breast cancer, regulating the activity of the small GTPase Rho, a key component of the non-canonical wnt pathway.44 Rho and other members of the small GTPases family are key for orchestrating the cytoskeleton rearrangements that occur during cell migration, both in physiological and pathological conditions. Rho signalling is essential to promote cancer metastasis by regulating cell migration, extravasation and angiogenesis.45, 46, 47 Rho also plays a pivotal role in the PCP pathway: non-canonical wnt signalling controls the convergent extension movements through the activation of Rho and other GTPases during morphogenesis.48 During amphibian gastrulation, PCP activated downstream of Fzd7, Rho and wnt11 is implicated in fibronectin matrix assembly, which in turn is fundamental for the correct migration and polarization of embryonic cells during morphogenesis. Interestingly, we found that Fzd6 signalling is important for the regulation of the actin cytoskeleton and extracellular fibronectin matrix in breast cancer cells.44 Fibronectin is a large glycoprotein involved in connecting cells with the extracellular matrix during cell adhesion, growth and migration.49 Fibronectin expression is also a hallmark of Epithelial to Mesenchymal Transition, a process responsible for the metastatic behaviour of cancer cells, modulated by wnt signalling.50 Fibronectin is also implicated in the metastatic dissemination of cancer cells to the bones and lungs of rodents51, 52 and its expression inversely correlates with survival in breast cancer patients.53 Thus, activation of the PCP pathway through gene amplification and overexpression of FZD6 could explain, at least in part, the high metastatic propensity and the organ tropism of triple negative breast cancer cells. Indeed, RNAi knockdown of FZD6 in MDA-MB-231 triple negative breast cancer cells inhibited their motility and caused reduction of bones and liver metastases, suggesting that activation of the PCP pathway is critically important for metastatic dissemination and tumour tropism in vivo.44 A summary of the validated and putative signalling pathways downstream of Fzd6 is shown in Figure 3. FZD6 could also play a role in other forms of human cancer since increased expression has also been detected in liver, prostate, colorectal and squamous cell carcinomas (Table 1)54, 55, 56, 57.
Figure 3.
Summary of validated and putative signalling pathways downstream of Fzd6. The question marks indicate molecular functions that are only hypothetical and not fully demonstrated in previous studies.
Table 1. Summary of the studies in which increased expression of Frizzled 6 was detected in different cancers.
| Tumour type | Activation mechanism | Phenotype | Reference |
|---|---|---|---|
| Breast cancer | Gene amplification, overexpression | Increased invasion and metastasis, predicts worse prognosis in patients | 44 |
| Mesenchymal glioblastoma | Overexpression | Increased tumour growth in mice xenografts, predicts worse prognosis in patients | 62 |
| Neuroblastoma | Expression in cancer stem cells | Drug resistance, increased tumour growth in mice xenografts, worse prognosis in patients | 23 |
| Chronic lymphocytic leukaemia | Progressive upregulation during CD5(+) B cells leukaemogenesis | Reduced survival in mice | 42 |
| Hepatocellular carcinoma | Overexpression | High expression associated with lower tumour differentiation | 56 |
| Prostate cancer | Overexpression | Not determined | 54 |
| Squamous cell carcinoma | Overexpression | Not determined | 57 |
| Colorectal cancer | Overexpression | Not determined | 55 |
Fzd6 and cancer stem cells
Cancer stem cells are thought to derive from normal counterparts harbouring oncogenic mutations or transformed epithelial cells that underwent epithelial to mesenchymal transition and acquired a stem-like phenotype.58 In a previous study, our group has demonstrated that the Fzd6 receptor is expressed on the surface of stem-like cells in neuroblastomas. Fzd6high neuroblastoma cells expressed high levels of mesenchymal markers such as Notch1and Twist1, formed neurospheres with high efficiency in liquid culture, were drug resistant and highly metastatic when injected orthotopically into immunodeficient mice.23 These biological features could be dependent on JNK activity since it is known that this kinase regulates stem cell functions through activation of the transcription factor AP-1 and the PCP pathway.24, 59 A further indication of the critical role played by Fzd6 in regulating cell stemness is provided by a recent study in which Fzd6 was shown to control the balance between self-renewal and survival of hematopoietic stem cells. Ablation of FZD6 strongly inhibited the repopulation of the hematopoietic compartment in the bone marrow after irradiation and transplant of donor cells in mice.60 It is intriguing to speculate that deregulation of Fzd6 signalling in cancer stem cells might promote uncontrolled self-renewal, homing and survival of metastatic cells in secondary organs. In agreement with this hypothesis, FZD6 is highly expressed in brain tumours containing mesenchymal, stem-like glioblastoma cells. Patients with FZD6high mesenchymal glioblastomas have a higher risk of recurrence and worse prognosis compared with other glioblastoma molecular and cellular subtypes.61, 62 Ectopic expression of FZD6 in proneural glioblastoma cells induced the expression of stem cell markers and resulted in increased cell proliferation and sphere formation in vitro and in vivo. Conversely, ablation of FZD6 expression decreased proliferation of mesenchymal glioblastoma cells.62 Fzd6 controls glioblastoma cell survival and proliferation through activation of the wnt/calcium pathway, Nf-kB and Stat3, which are known to promote a mesenchymal, stem-like state and aggressive behaviour of glioblastoma cells.62, 63, 64
FZD6 as a potential prognostic marker in cancer
Biomarkers are important as diagnostic tools, to predict disease outcome and ultimately direct clinical decisions and therapeutic strategies.65 An emblematic biomarker in oncology is HER2, whose expression is critical in determining therapeutic strategies in breast cancer.66 It would be important to identify new biomarkers that predicted tumour recurrence. This would benefit patients by allowing to discriminate those requiring aggressive treatments from patients who can be spared chemo- or radio-therapy.67 The identification of reliable markers to calculate the risk of metastatic recurrence would be particularly desirable, since metastasis is the primary cause of death in cancer patients. Tumour relapse occurs when dormant cancer cells are reactivated after months or years after chemotherapy and cancer stem cells markers have been proposed to be good prognostic indicators for tumour recurrence and metastasis.68, 69, 70 Cancer stem cells express membrane transporters that promote the active excretion of cytotoxic drugs and are resistant to drug killing, therefore are likely to constitute the bulk of the dormant tumour cell population.71, 72 In agreement with the hypothesis that FZD6 could be a cancer stem cell marker, high levels of FZD6 mRNA in neuroblastoma tumours indicate poor patient prognosis and in lymph node negative breast cancer high FZD6 expression predicts local and distant tumour relapse.23, 44 Positive Fzd6 immunostaining is correlated with lower distant relapse-free survival of triple negative breast cancer patients.23, 44 FZD6 expression is also of prognostic value in glioblastoma. Low expression of the transcription factor TCF4 coupled to high levels of FZD6 predict low survival of glioblastoma patients.62 These findings suggest that expression of FZD6, like other stem cell markers, could be potentially used as a prognostic biomarker in different types of human cancer.
Concluding remarks
Despite huge research efforts, no drugs targeting the wnt signalling pathway have been yet approved for clinical use, but clinical trials are ongoing in which new molecules targeting the wnt pathway are being tested.73 For example, the monoclonal antibody vantictumab is currently in phase 1 trials. The antibody binds to Fzds 1, 2, 5, 7 and 8, and has been shown to reduce activation of canonical wnt signalling in preclinical cancer models.74 One could envisage that a naked or toxin conjugated monoclonal antibody targeting Fzd6 could be developed to treat cancers bearing high Fzd6 expression levels. In principle, such antibody could be used to deplete rare subpopulations of drug resistant, metastatic cancer stem cells within the tumour. The phenotype of mice and humans carrying genetic inactivation and/or mutation of Fzd6 is mild, mainly manifesting in the form of defects in nail and hair development.21, 26 Thus, it could be predicted that antibodies targeting Fzd6 should have a good therapeutic potential and be well tolerated, without affecting the function of normal cells and tissues. Time will tell whether Fzd6 targeting has a future in cancer therapeutics.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors declare no conflict of interest.
References
- Schulte G. International Union of Basic and Clinical Pharmacology. LXXX. The class Frizzled receptors. Pharmacol Rev 2010; 62: 632–667. [DOI] [PubMed] [Google Scholar]
- van Amerongen R, Nusse R. Towards an integrated view of Wnt signaling in development. Development 2009; 136: 3205–3214. [DOI] [PubMed] [Google Scholar]
- Daniels D L, Weis WI. β-catenin directly displaces Groucho/TLE repressors from Tcf/Lef in Wnt-mediated transcription activation. Nat struct mol biol 2005; 12: 364–371. [DOI] [PubMed] [Google Scholar]
- Nusse R, van Ooyen A, Cox D, Fung YKT, Varmus H. Mode of proviral activation of a putative mammary oncogene (int-1) on mouse chromosome 15. Nature 1984; 307: 131–136. [DOI] [PubMed] [Google Scholar]
- Gupta S, Iljin K, Sara H, Mpindi JP, Mirtti T, Vainio P et al. FZD4 as a mediator of ERG oncogene-induced WNT signaling and epithelial-to-mesenchymal transition in human prostate cancer cells. Cancer Res 2010; 70: 6735–6745. [DOI] [PubMed] [Google Scholar]
- Lu D, Zhao Y, Tawatao R, Cottam HB, Sen M, Leoni LM et al. Activation of the Wnt signaling pathway in chronic lymphocytic leukemia. Proc Natl Acad Sci USA 2004; 101: 3118–3123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milovanovic T, Planutis K, Nguyen A, Marsh JL, Lin F, Hope C et al. Expression of Wnt genes and frizzled 1 and 2 receptors in normal breast epithelium and infiltrating breast carcinoma. Int J Oncol 2004; 25: 1337–1342. [PubMed] [Google Scholar]
- Nagayama S, Fukukawa C, Katagiri T, Okamoto T, Aoyama T, Oyaizu N et al. Therapeutic potential of antibodies against FZD10, a cell-surface protein, for synovial sarcomas. Oncogene 2005; 24: 6201–6212. [DOI] [PubMed] [Google Scholar]
- Reddy KB, Yin S, Daniel RB, Banerjee S, Sarkar F, Sethi S. Tumor initiating cells and FZD8 play a major role in drugresistance and tumor progression in triple negative breast cancer. Cancer Res 2013; 73(8 Supplement): 241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinhart Z, Hart T, Chandrashekhar M, Pavlovic Z, Robitaille M, Wang X et al. A CRISPR screen reveals a WNT7B-FZD5 signaling circuit as a therapeutic opportunity in pancreatic cancer. bioRxiv 2016; 23: 041996. [Google Scholar]
- Ueno K, Hazama S, Mitomori S, Nishioka M, Suehiro Y, Hirata H et al. Down-regulation of frizzled-7 expression decreases survival, invasion and metastatic capabilities of colon cancer cells. Br J Cancer 2009; 101: 1374–1381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winn RA, Marek L, Han SY, Rodriguez K, Rodriguez N, Hammond M et al. Restoration of Wnt-7a expression reverses non-small cell lung cancer cellular transformation through frizzled-9-mediated growth inhibition and promotion of cell differentiation. J Biol Chem 2005; 280: 19625–19634. [DOI] [PubMed] [Google Scholar]
- Xu YK, Nusse R. The Frizzled CRD domain is conserved in diverse proteins including several receptor tyrosine kinases. Curr Biol 1998; 8: R405–R406. [DOI] [PubMed] [Google Scholar]
- Rattner A, Hsieh JC, Smallwood PM, Gilbert DJ, Copeland NG, Jenkins NA et al. A family of secreted proteins contains homology to the cysteine-rich ligand-binding domain of frizzled receptors. Proc Natl Acad Sci USA 1997; 94: 2859–2863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tokuhara M, Hirai M, Atomi Y, Terada M, Katoh M. Molecular cloning of HumanFrizzled-6. Biochem Biophys Res Commun 1998; 243: 622–627. [DOI] [PubMed] [Google Scholar]
- Golan T, Yaniv A, Bafico A, Liu G, Gazit A. The human Frizzled 6 (HFz6) acts as a negative regulator of the canonical Wnt. beta-catenin signaling cascade. J Biol Chem 2004; 279: 14879–14888. [DOI] [PubMed] [Google Scholar]
- Lyons JP, Mueller UW, Ji H, Everett C, Fang X, Hsieh J et al. Wnt-4 activates the canonical β-catenin-mediated Wnt pathway and binds Frizzled-6 CRD: functional implications of Wnt/β-catenin activity in kidney epithelial cells. Exp Cell Res 2004; 298: 369–387. [DOI] [PubMed] [Google Scholar]
- Heinonen KM, Vanegas JR, Lew D, Krosl J, Perreault C. Wnt4 enhances murine hematopoietic progenitor cell expansion through a planar cell polarity-like pathway. PLoS ONE 2011; 6: e19279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato A, Yamamoto H, Sakane H, Koyama H, Kikuchi A. Wnt5a regulates distinct signalling pathways by binding to Frizzled2. EMBO J 2010; 29: 41–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilander MB, Dahlström J, Schulte G. Assessment of Frizzled 6 membrane mobility by FRAP supports G protein coupling and reveals WNT-Frizzled selectivity. Cell Signal 2014; 26: 1943–1949. [DOI] [PubMed] [Google Scholar]
- Fröjmark A, Schuster J, Sobol M, Entesarian M, Kilander MB, Gabrikova D et al. Mutations in Frizzled 6 cause isolated autosomal-recessive nail dysplasia. Am J Hum Genet 2011; 88: 852–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilander MB, Petersen J, Andressen KW, Ganji RS, Levy FO, Schuster J et al. Disheveled regulates precoupling of heterotrimeric G proteins to Frizzled 6. FASEB J 2014; 28: 2293–2305. [DOI] [PubMed] [Google Scholar]
- Cantilena S, Pastorino F, Chayka O, Pezzolo A, Pistoia V, Ponzoni M et al. Frizzled receptor 6 marks rare, highly tumourigenic stem-like cells in mouse and human neuroblastomas. Oncotarget 2011; 2: 976–983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamanaka H, Moriguchi T, Masuyama N, Kusakabe M, Hanafusa H, Takada R et al. JNK functions in the non-canonical Wnt pathway to regulate convergent extension movements in vertebrates. EMBO Rep 2002; 3: 69–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Marco P, Merello E, Rossi A, Piatelli G, Cama A, Kibar Z et al. FZD6 is a novel gene for human neural tube defects. Hum Mutat 2012; 33: 384–390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo N, Hawkins C, Nathans J. Frizzled6 controls hair patterning in mice. Proc Natl Acad Sci USA 2004; 101: 9277–9281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Guo N, Nathans J. The role of Frizzled3 and Frizzled6 in neural tube closure and in the planar polarity of inner-ear sensory hair cells. J Neurosci 2006; 26: 2147–2156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steele BM, Harper MT, Macaulay IC, Morrell CN, Perez-Tamayo A, Foy M et al. Canonical Wnt signaling negatively regulates platelet function. Proc Natl Acad Sci USA 2009; 106: 19836–19841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klaus A, Birchmeier W. Wnt signalling and its impact on development and cancer. Nat Rev Cancer 2008; 8: 387–398. [DOI] [PubMed] [Google Scholar]
- Polakis P. The many ways of Wnt in cancer. Curr Opin Genet Dev 2007; 17: 45–51. [DOI] [PubMed] [Google Scholar]
- Reya T, Clevers H. Wnt signalling in stem cells and cancer. Nature 2005; 434: 843–850. [DOI] [PubMed] [Google Scholar]
- Kurayoshi M, Oue N, Yamamoto H, Kishida M, Inoue A, Asahara T et al. Expression of Wnt-5a is correlated with aggressiveness of gastric cancer by stimulating cell migration and invasion. Cancer Res 2006; 66: 10439–10448. [DOI] [PubMed] [Google Scholar]
- Weeraratna AT, Jiang Y, Hostetter G, Rosenblatt K, Duray P, Bittner M et al. Wnt5a signaling directly affects cell motility and invasion of metastatic melanoma. Cancer cell 2002; 1: 279–288. [DOI] [PubMed] [Google Scholar]
- Anastas JN, Moon RT. WNT signalling pathways as therapeutic targets in cancer. Nat Rev Cancer 2013; 13: 11–26. [DOI] [PubMed] [Google Scholar]
- Sheldahl LC, Park M, Malbon CC, Moon RT. Protein kinase C is differentially stimulated by Wnt and Frizzled homologs in aG-protein-dependent manner. Curr Biol 1999; 9: 695–698. [DOI] [PubMed] [Google Scholar]
- Hirano H, Yonezawa H, Yunoue S, Habu M, Uchida H, Yoshioka T et al. Immunoreactivity of Wnt5a, Fzd2, Fzd6, and Ryk in glioblastoma: evaluative methodology for DAB chromogenic immunostaining. Brain Tumor Pathol 2014; 31: 85–93. [DOI] [PubMed] [Google Scholar]
- Kamino M, Kishida M, Kibe T, Ikoma K, Iijima M, Hirano H et al. Wnt‐5a signaling is correlated with infiltrative activity in human glioma by inducing cellular migration and MMP‐2. Cancer sci 2011; 102: 540–548. [DOI] [PubMed] [Google Scholar]
- Anastas J, Biechele T, Robitaille M, Muster J, Allison K, Angers S et al. A protein complex of SCRIB, NOS1AP and VANGL1 regulates cell polarity and migration, and is associated with breast cancer progression. Oncogene 2012; 31: 3696–3708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puvirajesinghe TM, Bertucci F, Jain A, Scerbo P, Belotti E, Audebert S et al. Identification of p62/SQSTM1 as a component of non-canonical Wnt VANGL2-JNK signalling in breast cancer. Nat commun 2016; 7: 10318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kibar Z, Bosoi CM, Kooistra M, Salem S, Finnell RH, De Marco P et al. Novel mutations in VANGL1 in neural tube defects. Hum Mutat 2009; 30: E706–E715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaucka M, Plevova K, Pavlova S, Janovska P, Mishra A, Verner J et al. The planar cell polarity pathway drives pathogenesis of chronic lymphocytic leukemia by the regulation of B-lymphocyte migration. Cancer Res 2013; 73: 1491–1501. [DOI] [PubMed] [Google Scholar]
- Wu Q, Claudia Z, Ranheim EA. Dysregulation of frizzled 6 is a critical component of B cell leukemogenesis in a mouse model of chronic lymphocytic leukemia. Blood 2007; 110: 347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luga V, Zhang L, Viloria-Petit AM, Ogunjimi AA, Inanlou MR, Chiu E et al. Exosomes mediate stromal mobilization of autocrine Wnt-PCP signaling in breast cancer cell migration. Cell 2012; 151:1542–1556. [DOI] [PubMed] [Google Scholar]
- Corda G, Sala G, Lattanzio R, Iezzi M, Sallese M, Fragassi G et al. Functional and prognostic significance of the genomic amplification of frizzled 6 (FZD6) in breast cancer. J Pathol 2016; 241: 350–361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yagi H, Tan W, Dillenburg-Pilla P, Armando S, Amornphimoltham P, Simaan M et al. A synthetic biology approach reveals a CXCR4-G13-Rho signaling axis driving transendothelial migration of metastatic breast cancer cells. Sci Signal 2011; 4:ra60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itoh K, Yoshioka K, Akedo H, Uehata M, Ishizaki T, Narumiya S. An essential part for Rho–associated kinase in the transcellular invasion of tumor cells. Nat Med 1999; 5: 221–225. [DOI] [PubMed] [Google Scholar]
- Croft DR, Sahai E, Mavria G, Li S, Tsai J, Lee WM et al. Conditional ROCK activation in vivo induces tumor cell dissemination and angiogenesis. Cancer Res 2004; 64: 8994–9001. [DOI] [PubMed] [Google Scholar]
- Habas R, Dawid IB, He X. Coactivation of Rac and Rho by Wnt/Frizzled signaling is required for vertebrate gastrulation. Genes Dev 2003; 17: 295–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearlstein E, Gold LI, Garcia-Pardo A. Fibronectin: a review of its structure and biological activity. Mol Cell Biochem 1980; 29: 103–128. [DOI] [PubMed] [Google Scholar]
- Brabletz T, Jung A, Spaderna S, Hlubek F, Kirchner T. Migrating cancer stem cells—an integrated concept of malignant tumour progression. Nat Rev Cancer 2005; 5: 744–749. [DOI] [PubMed] [Google Scholar]
- Saad S, Gottlieb DJ, Bradstock KF, Overall CM, Bendall LJ. Cancer cell-associated fibronectin induces release of matrix metalloproteinase-2 from normal fibroblasts. Cancer Res 2002; 62: 283–289. [PubMed] [Google Scholar]
- Huang L, Cheng H, Isom R, Chen C, Levine RA, Pauli BU. Protein kinase Cϵ mediates polymeric fibronectin assembly on the surface of blood-borne rat breast cancer cells to promote pulmonary metastasis. J Biol Chem 2008; 283: 7616–7627. [DOI] [PubMed] [Google Scholar]
- Ioachim E, Charchanti A, Briasoulis E, Karavasilis V, Tsanou H, Arvanitis D et al. Immunohistochemical expression of extracellular matrix components tenascin, fibronectin, collagen type IV and laminin in breast cancer: their prognostic value and role in tumour invasion and progression. Eur J Cancer 2002; 38: 2362–2370. [DOI] [PubMed] [Google Scholar]
- Wissmann C, Wild PJ, Kaiser S, Roepcke S, Stoehr R, Woenckhaus M et al. WIF1, a component of the Wnt pathway, is down‐regulated in prostate, breast, lung, and bladder cancer. J Pathol 2003; 201: 204–212. [DOI] [PubMed] [Google Scholar]
- Kim BK, Yoo HI, Kim I, Park J, Kim Yoon S. FZD6 expression is negatively regulated by miR-199a-5p in human colorectal cancer. BMB Rep 2015; 48: 360–366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bengochea A, De Souza M, Lefrancois L, Le Roux E, Galy O, Chemin I et al. Common dysregulation of Wnt/Frizzled receptor elements in human hepatocellular carcinoma. Br J Cancer 2008; 99: 143–150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haider AS, Peters SB, Kaporis H, Cardinale I, Fei J, Ott J et al. Genomic analysis defines a cancer-specific gene expression signature for human squamous cell carcinoma and distinguishes malignant hyperproliferation from benign hyperplasia. J Invest Dermatol 2006; 126: 869–881. [DOI] [PubMed] [Google Scholar]
- Singh A, Settleman J. EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene 2010; 29: 4741–4751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leppä S, Bohmann D. Diverse functions of JNK signaling and c-Jun in stress response and apoptosis. Oncogene 1999; 18: 6158–6162. [DOI] [PubMed] [Google Scholar]
- Abidin BM, Owusu Kwarteng E, Heinonen KM. Frizzled-6 regulates hematopoietic stem/progenitor cell survival and self-renewal. J Immunol 2015; 195: 2168–2176. [DOI] [PubMed] [Google Scholar]
- Iwadate Y. Epithelial-mesenchymal transition in glioblastoma progression (Review). Oncol lett 2016; 11: 1615–1620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang T, Alvarez AA, Pangeni RP, Horbinski M C, Lu S, Kim SH et al. A regulatory circuit of miR-125b/miR-20b and Wnt signalling controls glioblastoma phenotypes through FZD6-modulated pathways. Nat Commun 2016; 7: 12885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carro MS, Lim WK, Alvarez MJ, Bollo RJ, Zhao X, Snyder EY et al. The transcriptional network for mesenchymal transformation of brain tumours. Nature 2010; 463: 318–325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhat KP, Balasubramaniyan V, Vaillant B, Ezhilarasan R, Hummelink K, Hollingsworth F et al. Mesenchymal differentiation mediated by NF-κB promotes radiation resistance in glioblastoma. Cancer cell 2013; 24: 331–346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry NL, Hayes DF. Cancer biomarkers. Mol oncol 2012; 6: 140–146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baselga J, Swain SM. Novel anticancer targets: revisiting ERBB2 and discovering ERBB3. Nat Rev Cancer 2009; 9: 463–475. [DOI] [PubMed] [Google Scholar]
- Sestak I, Cuzick J. Markers for the identification of late breast cancer recurrence. Breast Cancer Res 2015; 17: 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collina F, Di Bonito M, Li Bergolis V, De Laurentiis M, Vitagliano C, Cerrone M et al. Prognostic value of cancer stem cells markers in triple-negative breast cancer. BioMed res int 2015; 2015: 158682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng B, Yang G, Jiang R, Cheng Y, Yang H, Pei L et al. Cancer stem cell markers predict a poor prognosis in renal cell carcinoma: a meta-analysis. Oncotarget 2016; 7: 65862–65875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aguirre-Ghiso JA. Models, mechanisms and clinical evidence for cancer dormancy. Nat Rev Cancer 2007; 7: 834–846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean M, Fojo T, Bates S. Tumour stem cells and drug resistance. Nat Rev Cancer 2005; 5: 275–284. [DOI] [PubMed] [Google Scholar]
- Fulda S. Regulation of apoptosis pathways in cancer stem cells. Cancer Lett 2013; 338: 168–173. [DOI] [PubMed] [Google Scholar]
- Lu B, Green BA, Farr JM, Lopes F, Van Raay TJ. Wnt drug discovery: weaving through the screens, patents and clinical trials. Cancers 2016; 8: 82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messersmith W, Cohen S, Shahda S, Lenz H, Weekes C, Dotan E et al. Phase 1b study of WNT inhibitor vantictumab (VAN, human monoclonal antibody) with nab-paclitaxel (Nab-P) and gemcitabine (G) in patients (pts) with previously untreated stage IV pancreatic cancer (PC). Ann Oncol 2016; 27(Suppl 6): 677P. [Google Scholar]