Abstract
Hemophilia A (HA) is an X-linked bleeding disease caused by factor VIII (FVIII) deficiency. We previously demonstrated that FVIII is produced specifically in liver sinusoid endothelial cells (LSECs) and to some degree in myeloid cells, and thus, in the present work, we seek to restrict the expression of FVIII transgene to these cells using cell-specific promoters. With this approach, we aim to limit immune response in a mouse model by lentiviral vector (LV)-mediated gene therapy encoding FVIII. To increase the target specificity of FVIII expression, we included miRNA target sequences (miRTs) (i.e., miRT-142.3p, miRT-126, and miRT-122) to silence expression in hematopoietic cells, endothelial cells, and hepatocytes, respectively. Notably, we report, for the first time, therapeutic levels of FVIII transgene expression at its natural site of production, which occurred without the formation of neutralizing antibodies (inhibitors). Moreover, inhibitors were eradicated in FVIII pre-immune mice through a regulatory T cell-dependent mechanism. In conclusion, targeting FVIII expression to LSECs and myeloid cells by using LVs with cell-specific promoter minimized off-target expression and immune responses. Therefore, at least for some transgenes, expression at the physiologic site of synthesis can enhance efficacy and safety, resulting in long-term correction of genetic diseases such as HA.
Keywords: gene therapy, targeted FVIII expression, hemophilia A, inhibitor titers reversion, Tregs
Hemophilia A is an X-linked bleeding disease caused by factor VIII (FVIII) deficiency. Targeting FVIII expression in endothelial and myeloid cells, the natural site of its production, by lentiviral vector gene transfer, Follenzi et al. obtained therapeutic levels of FVIII activity without formation of neutralizing antibodies in hemophilic mice.
Introduction
Hemophilia A (HA) is an X-linked bleeding disorder affecting 1 in 5,000–10,000 live male births and is caused by mutations and/or deletion in the coagulation factor VIII (FVIII) gene.1, 2, 3 Currently, there is no definitive cure for HA, and patients are treated with infusions of FVIII concentrate to treat or prevent bleeding, the latter aimed at sustaining FVIII levels at or above 1% of normal as prophylaxis. Therefore, because HA is a monogenic disease, and even modest amounts of FVIII (≥1%) can ameliorate the bleeding phenotype and improve the quality of life of affected individuals, the potential of gene therapy represents a powerful solution for the permanent treatment of HA.4 Recently, hemophilia B (HB) adult patients were successfully treated with the administration of a single dose of adeno-associated virus (AAV)-derived vectors expressing human factor IX (FIX) in hepatocytes, resulting in long-term FIX activity.5 Moreover, hepatocyte-specific FIX expression using AAV reached therapeutic levels in HB dogs even in the presence of neutralizing antibodies to canine FIX.6 Similarly, hepatocyte-specific FIX expression by lentiviral vectors (LVs)7, 8, 9, 10 with the presence of the hematopoietic-specific microRNA target sequence (mirT) for miR-142-3p11 prevented off-target expression in antigen-presenting cells (APCs) with long-term expression and avoidance of immune response. This approach was successfully applied in dogs and is now under clinical development in humans.12 Thus, circumventing the immune response to FVIII may also enable a gene therapy approach for HA, warranting effort in additional to that focused on improving hepatocyte-targeted LV-induced FVIII expression.13, 14, 15
From the gene therapy perspective, there are two major differences between HA and HB: (1) the normal site of synthesis of mouse or human FVIII is not the hepatocytes, and (2) risk for the formation of long-lasting inhibitor formation to FVIII in HA is 5-fold higher (20%–30% of patients) compared with those receiving replacement therapy for HB. Notably, in AAV-induced hepatocyte expression of human FVIII, FVIII inhibitor develops frequently, whereas using a similar approach, no FIX inhibitor is observed in HB mice injected with AAV-human FIX.16 Similarly, AAV liver gene therapy induced human FVIII expression in non-human primates (NHPs) resulted in inhibition in almost all animals and again in only 20% of those NHPs injected with AAV-human FIX. Whether these discrepancies are due to the use of non-species-specific and/or ectopic expression is unknown.
Recent studies have demonstrated that FVIII is not secreted by hepatocytes but mainly, although not exclusively, by endothelial cells (ECs).17, 18, 19 These cells simultaneously synthesize and release von Willebrand factor (vWF), which stabilizes and protects FVIII against premature degradation.20, 21 The liver is known to induce tolerance, rather than immunity, toward antigens presented locally to T cells22, 23 by specialized resident cells, such as liver sinusoidal ECs (LSECs) and resident liver macrophages, or Kupffer cells (KCs), which express anti-inflammatory mediators (e.g., interleukin 10 [IL-10], transforming growth factor β [TGF-β]),24, 25 thereby directing the immune response toward tolerance.26 Moreover, LSECs and KCs were reported to produce and secrete FVIII when transplanted in a murine model of HA.17, 27 Our goal is to test whether expression of human FVIII in its physiologic site of synthesis is feasible without increased immunogenicity.
To achieve transgene expression only in certain cell types, cell-specific promoters are widely used, such as the endothelial-specific promoter cadherin 5 type 2, also known as vascular endothelial cadherin (VEC),28 the myeloid specific CD11b promoter, or ItgaM.29 VEC is a transmembrane protein expressed mainly in ECs with a particular role in EC biology controlling intercellular cell junctions.28 CD11b is a leukocyte adhesion molecule, expressed mainly in monocytes and macrophages, which mediates myeloid cells binding to ECs and their migration through the vascular wall.30 In addition, specific miRTs can be included in the 3′ UTR of the transgene sequence to improve the stringency of transgene expression regulation.8, 31 In our studies, we use three different miRTs, miRT-122, miRT-126, and miRT-142-3p. miR-122 is a hepatocyte-specific microRNA with an important role in liver development and liver diseases.32 miR-142-3p is a miRNA expressed mainly in hematopoietic cells.11 miR-126 is a microRNA expressed predominantly by ECs controlling angiogenesis33, 34 and plasmacytoid dendritic cells (pDCs), as recently reported.35
Thus, miRTs provide post-transcriptional control to restrict transgene expression in targeted cells. Several efforts were focused on the improvement of LV for FVIII to obtain targeted transgene expression36 and on the production of several bioengineered FVIII compounds.36, 37 However, the development of gene therapy for HA is hampered by the increased risk for developing inhibitory antibodies to FVIII.38 In previous reports, the addition of miRT-142-3p to FVIII-expressing LV under the control of a hepatocyte-specific promoter was unable to prevent the production of neutralizing antibodies in HA mice.13, 31, 39 Consequently, a persistent unmet need exists to develop new strategies to minimize immune response associated with FVIII expression after gene therapy.
In this study, we describe new LV expression cassettes containing GFP and/or the B domain-deleted human FVIII (BDD-hFVIII) under the control of two cell-specific promoters, VEC28, 40 and CD11b.29 Post-transcriptional regulation was combined with different combinations of miRTs to simultaneously obtain long-term transgene expression and functional secretion of therapeutic doses of FVIII and to prevent immune response. Our model is innovative and has two advantages over AAV: (1) neutralizing antibodies do not develop, supporting this approach as viable therapy for a large fraction of HA patients, and (2) upregulation of memory T cells does not occur, preventing vector-capsid immune responses observed in early infection with AAV. This strategy is poised to improve FVIII gene transfer for HA and has additional potential application for inducing tolerance as well as safe and effective expression during gene therapy for other disease conditions. In particular, the present study may open new perspectives for the treatment of HA patients who have or are prone to develop anti-FVIII antibodies.
Results
In Vivo GFP Expression by LV Carrying the VEC Promoter and miRT122–142
Delivery of LV by intravascular injection transduces mainly the liver and spleen. To restrict transgene expression in the endothelial cells (ECs) of these organs, we built an LV construct containing the endothelial-specific murine VEC promoter with miRT122 and miRT142-3p to prevent expression in hepatocytes and hematopoietic cells, respectively (Figure S1A). We administered 5 × 108 TU of LV.VEC-GFP miRT122-miRT142-3p into C57BL/6 mice (n = 9) by the tail vein and analyzed the liver and spleen of injected mice after 1, 4, and 12 weeks. GFP expression in liver was stable for up to 3 months, the longest time tested, largely in the endothelium and mostly in LSECs, without significant immune or inflammatory cell infiltrate accumulation in the parenchyma (not shown). In the spleen of injected mice, GFP expression was restricted to the endothelium inside and surrounding the white pulp. In addition, miRT-122 and miRT-142-3p inclusion efficiently prevents “off-target” transgene expression in hepatocytes and hematopoietic cells, respectively, assuring long-term transgene expression in ECs (Figure 1A). As control, we injected mice with an LV containing only the VEC promoter and GFP (Figure S1A), and we observed off-target GFP expression in a very small fraction of hepatocytes and hematopoietic cells (Figure 1B). The expression cassette (LV.VEC-GFP-142) (Figure S1A) restricted GFP expression to ECs, preventing expression in hematopoietic cells, though a few GFP+ hepatocytes were still present (Figure 1C), confirming that the presence of both miRT and the VEC promoter restricted transgene expression exclusively to ECs (Figure 1A).
Figure 1.
Endothelial- and Myeloid-Specific Transgene Expression in the Liver of LV-Injected Mice
(A–E) GFP expression in the liver and the spleen of mice injected with LV.VEC-GFP-miRT-122–142 (n = 9) (A), LV.VEC-GFP (n = 9) (B), LV.VEC-GFP-miRT-142-3p (n = 9) (C), or LV.CD11b-GFP ± miRT-126 (n = 9) (D and E) 1 week and 3 months after injection. After injection of LV.VEC-GFP, transgene expression was observed in LSECs and splenic endothelial cells, with some off-targets in hepatocytes (white arrowheads) and myeloid cells (white arrows) (B). The addition of the miRT-142-3p avoided transgene expression in hematopoietic cells but not in hepatocytes (C). The addition of the miRT-122–142-3p restricted GFP expression to endothelial cells without off-targets in other cell types, as shown by the co-staining of the endothelial marker Lyve-1(red) with the GFP (green) (A). After injection of LV.CD11b-GFP, transgene expression was observed mainly in hepatic and splenic myeloid cells, with few GFP-expressing endothelial cells (white arrows) (E); by adding miRT-126 to the construct transgene, expression was detected only in myeloid cells, as shown by co-staining for the macrophage marker F4/80 (red) and GFP (green) (D). The scale bars represent 25 μm.
In Vivo Expression of LV Carrying the CD11b Promoter and miRT-126
Similarly, to restrict transgene expression to myeloid cells, such as hepatic macrophages, or Kupffer cells (KCs), splenic monocytes, and macrophages, we prepared an LV containing four copies of the miRT-126 containing GFP under the control of the myeloid-specific CD11b promoter (LV.CD11b-GFP-126) (Figure S1A). In C57BL/6 mice, this vector restricted transgene expression in myeloid cells. GFP expression was maintained up to 3 months after LV injection, the longest time tested, preventing off-target transgene expression in ECs both in liver and spleen (Figure 1D), with no inflammatory infiltrates accumulating in the liver parenchyma (not shown) of injected mice. As control, we injected C57BL/6 mice (n = 9) with 5 × 108 TU of LV containing only the CD11b promoter and GFP as a transgene (LV.CD11b-GFP)29 (Figure S1A). As expected, GFP expression was detected mainly in the monocyte/macrophage compartment of liver and splenic red pulp, with the presence of some off-target expression in ECs, but no GFP+ hepatocytes were detected (Figure 1E).
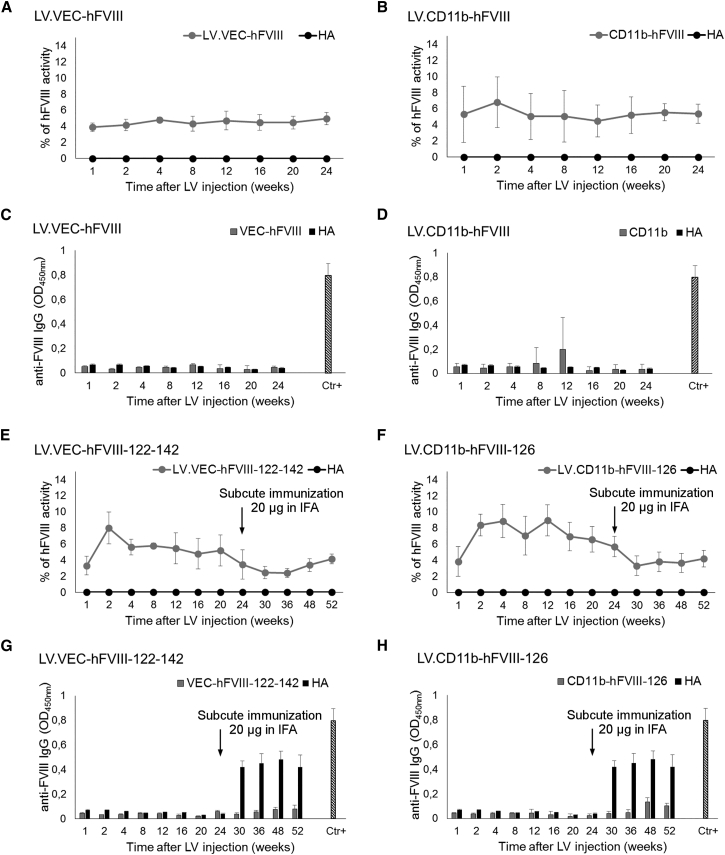
Endothelial and Myeloid Cell-Specific Transgene Expression Facilitates Stable hBDD-FVIII Secretion and Reduces Clotting Times in HA Mice
To evaluate hFVIII expression in ECs, we prepared LV expressing BDD-hFVIII under the control of the VEC promoter (LV.VEC-hFVIII) ± miRT-122 and miRT-142-3p sequences (LV.VEC-hFVIII-122–142) (Figure S1B). Eight- to 10-week-old C57BL/6 HA mice were tail-vein-injected with 109 TU of these constructs (n = 5–9); all injected mice had sustained hFVIII expression for up to 1 year after vector delivery (Figures 2A and 2E). Both groups of mice (i.e., LV.VEC-FVIII and LV.VEC-FVIII-122–142 treated) demonstrate 5%–6% of hFVIII activity. Moreover, ELISA detected no anti-FVIII antibodies, indicating that treatment with LV.VEC-hFVIII ± miRT-122–142-3p did not trigger a humoral immune response in these mice (Figures 2C and 2G).
Figure 2.
Long-Term hFVIII Expression in HA Mice after Injection of LV.VEC-hFVIII ± miRT-122–142-3p and LV.CD11b-hFVIII ± miRT-126
(A–H) HA mice were injected for the expression of hFVIII in endothelial cells with the LV containing the endothelial-specific VEC promoter with (n = 9) or without (n = 5) the addition of miRT-122–142-3p (A, C, E, and G) or in myeloid cells with the LV containing the myeloid-specific CD11b promoter with (n = 5) or without (n = 5) the addition of miRT-126 (B, D, F, and H). (A, B, E, and F) Graphs showing the mean percentages ± SD of hFVIII activity in plasma of HA mice up to 1 year after LV injection. Mice injected with VEC-hFVIII-122–142 (E) or CD11b-hFVIII-126 (F) were immunized with a subcutaneous injection of hFVIII in incomplete Freund’s adjuvant (IFA) (A, B, E, and F: p < 0.001 compared with control HA group). (C, D, G, and H) ELISA showing the presence of anti-FVIII antibodies in plasma of LV injected mice (means ± SD). No anti-FVIII antibodies were detected in plasma of HA mice injected with LV.VEC-hFVIII ± miRT-122–142-3p (C and G) or CD11b-hFVIII-126 (H). Thirty percent of HA mice injected with LV.CD11b-hFVIII developed anti-FVIII antibodies between 8 and 12 weeks after LV injection (D). Plasma dilution was 1:2,000. Ctr+, pooled plasma from LV.PGK-hFVIII-injected mice that developed anti-FVIII antibodies.
On the contrary, intravenous injection of LV containing hFVIII expressed under the control of the ubiquitous PGK promoter with or without the miRT-142-3p sequences (109 TU of LV.PGK-hFVIII ± 142 [n = 5]) in HA mice (Figure S2A) yielded no more than 1.5% FVIII activity 2 weeks after injection, decreasing below 0.5% thereafter (Figure S2B). All mice injected with both LV ± miRT-142-3p sequences developed inhibitors, as assessed by Bethesda assay (Figure S2C), and anti-FVIII IgG1, IgG2a, and IgG2b were detected by ELISA (Figures S2D–S2F). Interestingly, the addition of miRT-142-3p was sufficient to reduce inhibitor titers (30%–40%) (Figure S2C), showing that in HA mice, miRT-142-3p reduces but does not eliminate the immune response against hFVIII expressed under the control of a ubiquitous promoter.
To address the expression of hFVIII in myeloid cells, we tail-vein-injected 109 TU of an LV expressing the BDD-hFVIII transgene under the control of the CD11b promoter (LV.CD11b-hFVIII) ± miRT-126 (LV.CD11b-hFVIII-126) (Figure S1B) in 8- to 10-week-old C57BL/6 HA mice (n = 5–10). As observed for the LV.VEC-injected mice, both mouse groups showed long-term sustained hFVIII expression for up to 1 year, with an average of 5%–6% activity (Figures 2B and 2F). Interestingly, 30% of the mice injected with LV.CD11b-hFVIII (n = 10) developed anti-FVIII antibodies 8–12 weeks after LV injection (Figure 2D), and these mice died mainly from internal or intracranial bleeding 1–2 weeks after developing anti-FVIII antibodies, while none of the mice injected with LV.CD11b-hFVIII-126 (n = 5) developed antibodies (Figure 2H) and continued to express FVIII.
Mice injected with LV.VEC-hFVIII-122–142 and with LV.CD11b-hFVIII-126 were then immunized with 20 μg of a commercial recombinant human BDD-hFVIII (ReFacto, Pfizer) in incomplete Freund’s adjuvant (IFA). This immunization challenge did not result in the development of anti-FVIII antibodies (Figures 2D and 2H), confirming that immune tolerance was achieved consistent with the continuous secretion of FVIII in the circulation.
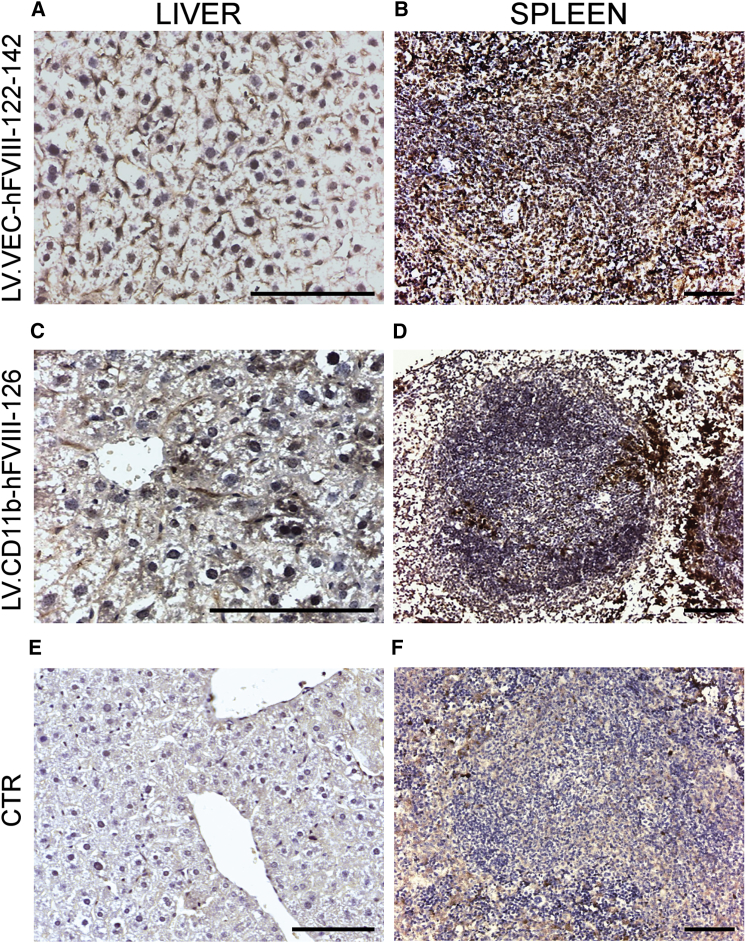
Integrated vector copy number was measured by qPCR in the liver and spleen of injected mice. In KCs and LSECs (Figures S3A and S3B) isolated from liver of both LV.VEC-hFVIII ± miRT-122–142-3p-injected mice and LV.CD11b.hFVIII ± miRT-126-injected mice (Figures S3C and S3D) an average of one or two copies per genome were found. hFVIII expression in LSECs and in macrophages of LV-injected mice was confirmed by immunohistochemistry of liver and spleen sections (Figures 3A–3F).These data indicate that all the LVs were stably integrated and that the transduced cells were not cleared but persisted for a long period of time in the treated mice.
Figure 3.
hFVIII Expression in Hepatic and Splenic Endothelial Cells and Myeloid Cells of HA Mice 1 Year after LV Injection
(A–F) Immunohistochemistry showing hFVIII expression in hepatic and splenic endothelial cells after injection of LV.VEC-hFVIII-122–142 (A and B) and in hepatic and splenic myeloid cells after injection of LV.CD11b-hFVIII-126 (C and D). Control liver and spleen from HA mice showed no hFVIII positive staining (E and F). The scale bars represent 100 μm.
Endothelial-Specific Expression Allows hFVIII Secretion and Results in the Reversion of Anti-FVIII Antibody Titer in Previously Immunized Mice
One of the major obstacles for HA gene therapy is the treatment of inhibitor-positive patients.41 Hepatocyte-specific FIX expression results in the reduction of anti-FIX antibody titers in previously FIX-immunized HB mice.7 However, reduction of anti-FVIII antibody titers using gene therapy in previously FVIII-immunized HA mice requires the additional treatment with anti-CD3 or anti-CD20 antibodies.42, 43 We therefore induced anti-FVIII antibodies in C57BL/6 HA mice by subcutaneous injection of human recombinant FVIII (ReFacto) in IFA7 (Figure S4). All mice developed 15–24 Bethesda unit (BU) inhibitor titers 4 weeks following immunization (Figure 4C). These immunized mice were injected with 109 TU of LV.VEC-hFVIII ± miRT-122–142-3p (n = 3). One week following LV injection, mice expressed an average of 1% hFVIII with a stable increase to 5%–6% in hFVIII activity for 1 year following injection (Figures 4A and 4B). Concurrently, plasma BU decreased progressively starting from 4 weeks after LV injection (Figure 4C). We anticipate that because plasma anti-FVIII IgG1 decreased in parallel with BU in injected mice, anti-FVIII inhibitors are IgG1 isotype antibodies (Figure 4D), in contrast to anti-FVIII IgG2a and IgG2b antibodies, which remained detectable in injected mice (Figures 4E and 4F). To evaluate the potential of this LV delivery approach to induce immune tolerance to FVIII, we injected these LV-treated mice with 4 U of human recombinant FVIII (ReFacto)44 24 weeks after LV delivery and demonstrate that antibody titers remained undetectable with continued FVIII activity, indicating tolerance to FVIII (Figures 4A–4E).
Figure 4.
hFVIII Activity after LV.VEC-hFVIII ± miRT-122–142-3p Injection in HA Mice Previously Immunized with hFVIII
HA mice were immunized with FVIII, and 4 weeks later injected with LV.VEC-hFVIII ± 122–142. (A and B) hFVIII activity in plasma of mice injected with LV.VEC-hFVIII (A) and LV.VEC-hFVIII-122–142 (B) increased over time, reaching 5%–6% FVIII activity, and remained stable up to 1 year. Rechallenge with hFVIII had no effects on hFVIII activity in plasma of both groups of mice (A and B: p < 0.001 compared with control group). (C) BU present in plasma of immunized mice decreased over time after injection of LV.VEC-hFVIII ± 122–142. (D–F) Line-graphs showing anti-FVIII IgG1 (D), IgG2a (E), and IgG2b (F) antibody titers in plasma of FVIII-immunized HA mice injected with LV.VEC-hFVIII and LV.VEC-hFVIII-122–142 (C–F: p < 0.001 compared with control group). Indicated are means ± SD.
Our present data demonstrate that we were even able to reverse the pre-existing anti-FVIII immunity in HA mice. Indeed, inhibitors levels in the plasma of these mice decreased, with a concomitant correlated increase of FVIII activity. Similar results were reported in the case of the hepatocyte-directed FIX expression mediated by LVs containing mirT-142-3p, which was able to reverse anti-FIX pre-existing immunity in HB mice.7
miRT-126 Prevents Transgene Expression in pDCs
As previously described, pDCs express miR-126.35 We therefore hypothesized that the addition of miRT-126 would also reduce transgene expression in pDCs, blunting the immune response to FVIII. Initially, C57BL/6 mice were injected with 5 × 108 TU of LV.CD11b-GFP ± miRT-126 (n = 6–7) or LV.PGK-GFP (n = 6), and GFP expression was analyzed in hepatic and splenic pDCs 2 weeks following injection. Fluorescence-activated cell sorting (FACS) analysis demonstrates GFP expression in pDCs of mice injected with either LV.PGK-GFP or LV.CD11b-GFP. Transgene expression, instead, was drastically reduced only in pDCs but not in other cell types after injection of LV.CD11b-GFP-126 (% of GFP+ pDCs: CD11b-GFP 12 ± 3 versus CD11b-GFP-126 1.8 ± 0.5 in liver and CD11b-GFP 15 ± 3 versus CD11b-GFP-126 1.3 ± 0.6 in spleen; p < 0.01), with no effects on the total number of splenic and hepatic pDCs. Moreover, we observed comparable GFP expression in DCs in mice treated with and without miRT-126 (Figures 5A and 5B). We hypothesize that these data may explain the development of anti-FVIII antibodies in 30% of LV.CD11b-hFVIII-injected mice. Perhaps presentation of FVIII antigen by pDCs is able to trigger an immune response in injected mice; the addition of miRT-126 abrogated this response, allowing long-term FVIII expression in LV-treated HA mice.
Figure 5.
GFP Expression in Hepatic and Splenic Plasmacytoid Dendritic Cells after Injection of LV.CD11b-GFP ± miRT-126
(A and B) FACS analysis showing GFP expression in pDCs (CD11c+, B220+, pDCA-1+) and in DCs (CD11c+, pDCA-1−) from liver (A) and spleen (B) of mice injected with LV.CD11b-GFP ± miRT-126. Mice injected with the LV.PGK-GFP were used as controls. The addition of miRT-126 to the LV.CD11b-GFP drastically reduced the GFP expression in hepatic and splenic pDCs. Indicated are mean percentages ± SD (n = 6 or 7 per group).
Endothelial-Specific Expression of FVIII-RH and Codon-Optimized FVIII Results in Higher FVIII Activity without Antibody Formation
We recently described a codon-optimized (co)-hFVIII version that augments gene expression in vivo.45 To study the efficiency of an endothelial-specific expression system and evaluate the possibility of improving transgene expression, we compared the non-codon-optimized BDD-hFVIII version (LV.VEC-hFVIII ± 122–142) with its co-hFVIII counterpart (LV.VEC-co-hFVIII-122–142) (n = 3–5 for each LV). We also compared these forms with the RH-FVIII form, a recently described FVIII variant with enhanced pro-coagulant activity in HA mice. This variant (RH-FVIII) contain an amino acid change at the furin cleavage site within the B domain (position R1645H) that mimics the canine sequence (HHQR versus human RHQR).46 Two weeks after LV.VEC-co-hFVIII-122–142 injection, HA mice showed higher FVIII activity; the FVIII activity in mice injected with co-hFVIII increased over time up to 12%, remaining stable up to 36 weeks following injection. In comparison, LV injection with LV.VEC-hFVIII ± 122–142 exhibited the typical FVIII activity (up to 5%), remaining stable 52 weeks following vector delivery (Figure 6A), while LV.VEC-RH reached 8% FVIII activity (higher than LV.VEC-hFVIII ± 122–142 but lower than co-hFVIII) and remained stable for 1 year following LV injection. As observed for the BDD-hFVIII form, no anti-FVIII antibodies or inhibitors were detectable in the plasma of mice injected with LV.VEC-co-hFVIII-122–142 or LV.VEC-RH (Figure 6B). Moreover, FVIII immunization did not result in anti-FVIII antibody production, and FVIII activity remained stable in LV-injected mice (Figures 6A and 6B). Blood loss and bleeding time analyzed 24 weeks after LV injection confirmed correction in all injected mice, with higher efficiency in mice injected with LV.VEC-co-hFVIII-122–142 (Figures 6C and 6D). Taken together, these data demonstrate that endothelial-specific FVIII expression is improved by using a codon-usage-optimized hFVIII or a different FVIII form with greater pro-coagulant activity.
Figure 6.
Comparison of Different Forms of BDD-hFVIII
HA mice (n = 3–5) were injected with LV containing the VEC promoter driving expression of BDD-hFVIII ± miRT-122–142, RH, and co-hFVIII-miRT-122–142.
(A) Two weeks after LV injection, FVIII activity in plasma of injected mice showed an average of 4% for BDD-hFVIII ± miRT-122–142 and RH, and 7% for co-hFVIII-miRT-122–142. Although hFVIII activity remained stable to 5% in plasma of mice injected with LV.VEC-hFVIII ± miRT-122–142, LV.VEC-RH-, and LV.VEC-co-hFVIII-miRT-122–142-injected HA mice showed an average of 8% and 11%–12% of hFVIII activity, respectively, and remained stable up to 52 and 36 weeks after injection, and immunization with FVIII did not affect the FVIII activity in plasma of these mice (p < 0.001 for LV.VEC-cohFVIII-122–142 versus LV.VEC-hFVIII ± 122–142). (B) ELISA showing the absence of anti-FVIII antibodies in plasma of mice injected with the different LVs. Immunization with FVIII did not result in anti-FVIII antibody production in LV-injected mice. Plasma dilution was 1:2,000. Ctr+, pooled plasma from LV.PGK-hFVIII-injected mice that developed anti-FVIII antibodies. (A and B) Indicated are means ± SD. (C) Blood loss (mean ± SD) was evaluated 24 weeks after LV injection by measuring the absorbance at 597 nm (A597nm) of blood content in the collection tubes by lysis of red blood cells. (D) Tail bleeding times (mean ± SD) were assessed by monitoring the blood flow into collection tubes containing saline solution, and times to stop bleeding were recorded. **p < 0.01; ***p < 0.001.
LSEC-Induced Regulatory T Cells Allow Tolerance to FVIII
To study whether long term FVIII expression in LSECs was regulated by regulatory T cells (Tregs), we depleted Tregs in vivo using an anti-CD25 antibody.47 We have explored the ability of LSECs to induce Tregs using two different protocols: (1) LV-VEC-hFVIII ± 122–142 injection in HA mice followed by Treg depletion (Figures S5A and S5B) and (2) an initial Treg depletion followed by injection of LV-VEC-hFVIII ± 122–142 (Figures S5C and S5D).48 For the first group of mice, we injected LV.VEC-hFVIII ± 122–142 (n = 5), assessing plasma hFVIII activity and anti-hFVIII antibodies starting 1 week after LV delivery. After 10 weeks, hFVIII activity was stable (Figure 7A), and no anti-hFVIII antibodies were detected (Figures 7B–7E). Eleven weeks after LV injection, we depleted Tregs using anti-CD25 and analyzed plasma samples from the subsequent week. After 1 week, Tregs were almost totally depleted (Figure S5B), with no effect on hFVIII activity or anti-hFVIII antibody production. Three weeks after Treg depletion, hFVIII activity in the plasma of all mice was halved (Figure 7A), and 5 weeks after Treg depletion, anti-hFVIII antibodies and inhibitors were detectable (Figures 7B–7E) even though Tregs were almost back to normal (Figure S5B). Anti-FVIII antibodies and plasma BU peaked at 12 weeks following Treg depletion in injected mice (Figures 7B–7E) with undetectable plasma hFVIII activity (Figure 7A). Interestingly, 16 weeks after Treg depletion, plasma hFVIII activity rebounds, returning to the original levels observed before Treg depletion (Figure 7A) 3 weeks later, with a concurrent decrease in BU levels (Figure 7B). Moreover, blood loss and bleeding time after tail clip challenge confirms the physiological function of restored FVIII activity to the initial levels before Treg depletion (Figures 7F and 7G). Again, BU kinetics correlate mainly with anti-FVIII IgG1 while anti-FVIII IgG2a and IgG2b remained stable after the immunization (Figures 7B–7E).
Figure 7.
Effects of Treg Depletion after Gene Therapy with LV.VEC-hFVIII ± 122–142
(A) hFVIII activity in plasma of LV-injected mice reached a level of 4% with LV.VEC-hFVIII ± 122–142 (n = 4 or 5). FVIII activity remained stable up to 10 weeks after LV injection. hFVIII activity started to decrease 3 weeks after Treg depletion, reached almost undetectable levels, and increased again 16 weeks after Treg depletion, returning to levels observed before Treg depletion 4 weeks later (p < 0.001, treated mice compared with control HA group). (B) Bethesda units (BU) started to be detected 3 weeks after Treg depletion and reached the peak 12 weeks after Treg depletion, and then BU decreased again 8 weeks later. (C–E) ELISA showed no anti-FVIII antibodies in plasma of LV-injected mice up to 14 weeks after LV injection. Three weeks after Treg depletion, the presence of anti-FVIII IgG1 (C), IgG2A (D), and IgG2b (E) was detected in the plasma of treated mice. Although anti-FVIII IgG2a (D) and IgG2b (E) titers remained high, anti-FVIII IgG1 (C) titers decreased concomitantly with BU 12 weeks after Treg depletion. (A–E) Indicated are means ± SD. (F) Blood loss (mean ± SD) was evaluated at different times after LV injection by measuring the absorbance at 597 nm (A597nm) of blood content in the collection tubes by lysis of red blood cells. (G) Tail bleeding times (mean ± SD) were assessed by monitoring the blood flow into collection tubes containing saline solution, and times to stop bleeding were recorded. n = 3 or 4 per group. **p < 0.01; ***p < 0.001.
In the second group of animals, we depleted Tregs by injection of anti-CD25 antibodies (n = 6) with complete Treg depletion in 5 days; mice were then injected with LV-VEC-hFVIII ± 122–142 (n = 3 for each group) (Figures S5C and S5D). We analyzed plasma samples of injected mice for the presence of hFVIII activity, BU, and anti-hFVIII antibodies starting 2 weeks after LV injection. All of these mice showed only very low hFVIII activity in plasma (<1%) up to 52 weeks after LV injection (Figure S6A) and developed anti-hFVIII antibodies and inhibitors starting 2 weeks after LV-injection (Figures S6B–S6E). These data demonstrate a clear role for Tregs in establishing tolerance to FVIII during LV-induced gene expression in ECs under the VEC promoter. Similar results were reported for mice immunized with FIX and injected with an AAV containing the FIX transgene under a hepatocyte-specific promoter.49 Overall, LSECs appear to be the ideal target cell for FVIII transgene expression.
Endothelial-Specific Expression of FVIII in BALB/c and B6/129 HA mice
To this point, we analyzed HA mice on a C57BL/6 background. To study whether endothelial-specific FVIII production would support long-term transgene expression in a different immunocompetent mouse strain,50 we injected BALB/c (n = 4) and B6/129 (n = 5) HA mice with LV.VEC-hFVIII. Two weeks after LV injection, plasma FVIII activity is detected in treated BALB/c and B6/129 mice up to 28 and 24 weeks after injection, respectively, with comparable activity (5%–6%) with that observed with immunocompetent C57BL/6 HA mouse (Figures 8A and 8C). Moreover, no plasma anti-FVIII antibodies were detected in either LV injected HA mouse strain (Figures 8B and 8D) with diminished blood loss and shortened bleeding time at 28 and 20 weeks after injection in BALB/c and B6/129 HA mice, respectively, confirming that correction was achieved in all injected mice (Figures 8E and 8F).
Figure 8.
Endothelial-Specific FVIII Expression in Different HA Mouse Strains
(A–D) BALB/c HA mice (n = 4) and B6/129 HA mice (n = 5) were injected with LV.VEC-hFVIII and hFVIII activity (A and C), and eventual presence of anti-FVIII antibodies (B and D) was evaluated starting from 2 weeks after injection. hFVIII activity in the plasma of LV-injected mice reached 5%–6% up to 28 weeks after injection in BALB/c HA mice (A), and the same activity was detected in plasma of treated B6/129 HA mice up to 24 weeks after injection. (B and D) ELISA showing no anti-FVIII antibodies in the plasma of injected BALB/c and B6/129 HA mice up to 28 and 24 weeks, respectively, after injection. Plasma dilutions were 1:200 and 1:2,000. Ctr+, pooled plasma from LV.PGK-hFVIII-injected mice that developed anti-FVIII antibodies. (A–D) Indicated are means ± SD. (E) Blood loss (mean ± SD) was evaluated 28 weeks and 20 weeks after LV injection in BALB/c-HA and B6/129-HA mice, respectively, by measuring the absorbance at 597 nm (A597nm) of blood content in the collection tubes after lysis of red blood cells. (F) Tail bleeding times (mean ± SD) were assessed by monitoring the blood flow into collection tubes containing saline solution, and times to stop bleeding were recorded. n = 3–5 per group. **p < 0.01; and ***p < 0.001.
Finally, we performed a direct comparison to verify if there is any discrepancy between the FVIII activity measured by activated partial thromboplastin time (aPTT) or by chromogenic assays. We tested in parallel some of our samples, and we found that the chromogenic assay showed up to 20% higher level than the aPTT assay we normally perform in the laboratory (Figure S7). However, ELISA for FVIII-Ag detected higher amount of hFVIII antigen (up to 50%–60%) compared with both aPTT and chromogenic assays. This last result was expected because the ELISA evaluates only the presence of the antigen but not its functionality. We can conclude that our aPTT measurements underestimated FVIII activity of about 20%, as previously reported by Potgieter et al.51 Taken together, our data support our hypothesis that endothelial-specific expression, even without the miRT-specific sequences, supports FVIII expression with FVIII activity without formation of anti-FVIII antibodies in all three different immunoreactive HA mouse strains studied.
Discussion
HA, like HB, is a good candidate for gene therapy because modest amounts of FVIII or FIX can ameliorate bleeding and improve quality of life in affected individuals.52 For both diseases, long-term expression has been successfully achieved in animal models3, 13, 53, 54, 55, 56 and several therapeutic gene transfer strategies have been attempted in recent years. However, in light of three failed clinical trials for HA, additional strategies and modifications of prior approaches are necessary for long-term therapeutic correction in patients.57 Our present data demonstrate the potential for using LVs containing non-hepatocyte liver-specific promoters in association with post-transcriptional control sequences (miRTs). As a consequence, transgene expression was selectively restricted to ECs, mainly LSECs for VEC promoter, or to splenic/hepatic macrophages for the CD11b promoter, resulting in long-term GFP expression in the absence of an immune response.
The liver has been implicated as the major FVIII-producing organ, because liver transplantation is able to cure HA in canine models and humans.58, 59 Moreover, hepatocyte-specific transgene expression may induce tolerance, reducing the risk for immune reaction against the transgene; such an approach addresses the current limitations in reaching therapeutic efficacy with FVIII for HA.52 Previous work demonstrated the importance of hepatocytes as the main physiologic source of FVIII. More recently, we and others have demonstrated that FVIII is largely secreted by ECs, particularly by LSECs, and to a lesser extent by hematopoietic cells in both humans and mice.17, 19, 60, 61, 62 In fact, cell therapy approaches demonstrated a role for LSECs and myeloid cells in FVIII secretion, decreasing bleeding in HA mice.17, 19, 27
We thus targeted FVIII expression to endothelial or myeloid cells using LVs containing endothelial-specific (LV.VEC)28, 40 or myeloid-specific (LV.CD11b)29 promoters. Furthermore, the addition of miRTs in the 3′-UTR of the LVs allowed us to reach full cell-specific transgene expression. The addition of miRTs-122 or 142-3p prevented transgene expression in hepatocytes or hematopoietic cells,48 respectively, in LV.VEC (LV.VEC-122–142). The addition of miRT-126 prevented FVIII expression in endothelial and pDCs63 in LV.CD11b (LV.CD11b-126). By this strategy, specific targeting of ECs, mainly LSECs,17, 19 resulted in long-term therapeutic levels of FVIII expression in HA mice and induced Tregs by a TGF-β-mediated mechanism,26 thus inhibiting the immune response. This effect was observed also in the absence of miRTs, strongly suggesting that the presence of the endothelial promoter alone is enough to induce long-term FVIII expression. Additionally, it has been reported that FVIII expression in hepatocytes could induce a cellular stress, supporting our hypothesis that FVIII expression is optimally induced in cells that are physiologically involved in FVIII expression and secretion.64
In contrast to the limited immune response observed in EC-specific FVIII expression, targeting FVIII expression in myeloid cells results in the formation of anti-FVIII antibodies in 30% of the injected mice. The addition of miRT-126 to myeloid-specific FVIII expression prevents anti-FVIII antibody generation. miR-126 has been reported to be expressed in pDCs,35 which could be involved, possibly as APC, in the anti-FVIII adaptive immune response triggered by transgene expression. Indeed, when we used GFP in LV.CD11b-126 construct, we observed a strong reduction of GFP expression in pDCs, with no effect on transgene expression in conventional dendritic cells (cDCs) and macrophages. Thus, eliminating transgene expression in pDCs during LV transduction prevents the immune response to the transgene, enabling long-term FVIII secretion and activity. On the other hand, it was reported that LV delivery activates pDCs to secrete type I IFN immediately after vector administration, and specifically within the liver, contributing to immune-mediated clearance of transduced cells, playing a major role in preventing stable gene transfer.39 In future studies it will be interesting to elucidate the exact mechanism triggering the innate host response in LV-injected animals with pDC activation and whether IFN production limits LV transduction of LSEC and myeloid cells as reported for hepatocytes.
Using more active forms of FVIII, such as RH-FVIII and co-hFVIII,14, 45, 46, 65 we can further increase the therapeutic index of gene transfer by LV. Similar results were reported by Siner et al.,46 who used this transgene under the control of a hepatocyte-specific promoter in AAV-8 to obtain long-term expression without inducing an immune response. In this context, Nathwani’s group14 reported phenotypic correction using codon-optimized hFVIII variants (BDD, N6, and V3) under the control of a hepatocyte-specific promoter and, by further restricting hFVIII expression in the liver using AAV-8, obtained a 10-fold increase in transgene expression compared with the levels obtained using AAV-5.
Finally, we demonstrate stable FVIII activity in the plasma of HA mice also on BALB/c and B6/129 strains (in addition to C57BL/6) after injection with EC-specific LV (LV.VEC-hFVIII). We investigated whether FVIII expression and antibody formation against the transgene after LV injection would be influenced by the genetic background of the HA mice, as previously reported for injection of recombinant FVIII in HA mice in both C57BL/6 and BALB/c background.50 The lack of difference in the immune response among different strains in our data can be explained by the fact that expressing FVIII in its natural site of production results not only in long-term FVIII expression but also in the inhibition of a secondary immune response to FVIII through the direct interaction of effector T cells with ECs, mainly LSECs,66 thus endorsing a tolerance response to FVIII.
In conclusion, our study demonstrates successful long-term expression of FVIII using LVs specifically targeting non-hepatocyte cells in the liver. This approach is complementary to the promising early clinical trial using AAV to target hepatocyte-specific FVIII expression (BMN 270, BioMarin). Because these initial studies select HA patients without inhibitors, the final results may not be applicable to all HA patients (e.g., those with anti-FVIII inhibitors). As a consequence, additional strategies to prevent or reverse immune responsiveness to FVIII may be necessary. We propose that approaches such as those presented here serve as important pre-clinical models of circumventing or reversing immune response using non-hepatocyte-specific FVIII expression in HA.
Materials and Methods
LV Production
We inserted the B-domain-deleted hFVIII (BDD-hFVIII) sequence, and a de novo synthesized co-hFVIII45 or the RH-hFVIII46 in LV transfer constructs containing the ubiquitous PGK promoter, the endothelial-specific VEC promoter, or the myeloid-specific CD11b promoter. LV.PGK-GFP-126, LV.PGK-GFP-142-3p, and LV.PGK-GFP-122–142-3p constructs were kindly provided by Professor L. Naldini (HSR-TIGET). We subcloned the miRT sequences from these constructs into LV.PGK-hFVIII, LV.VEC-GFP, LV.VEC-hFVIII, LV.CD11b-GFP, and LV.CD11b-hFVIII plasmids.
LVs were produced by transient co-transfection of 293T cells with third-generation packaging plasmids using the calcium phosphate method, according to published protocols.67 LV titers were determined on 293T by qPCR, for hFVIII-containing LV, as previously described.68
Mice
For in vivo delivery of LVs we used C57BL/6 mice for GFP studies or HA mice in C57BL/6,27 B6/129, or BALC/c background, obtained by breeding HA mice in BALB/c for 10 generations. B6/129 HA (B6;129S-F8tm1Kaz/J, The Jackson Laboratory) mice were kindly provided by Professor L. Naldini (HSR-TIGET). To induce anti-FVIII antibodies, mice were injected subcutaneously with 20 μg of ReFacto in IFA (Sigma-Aldrich), and plasma samples were analyzed from 2 weeks after immunization. Animal studies were performed according to an approved protocol by the Animal Care and Use Committee of Università del Piemonte Orientale.
FVIII Activity Assays
FVIII activity was assessed by aPTT assay, using the Coagulation Analyzer Coatron M4 (TECO Medical Instruments), and by chromogenic assay, using the COATEST SP4 FVIII (Chromogenix, Werfen - Instrumentation Laboratory). Standard curves were generated using serial dilutions of pooled human plasma or purified BDD-hFVIII (ReFacto) in hemophilic mouse plasma. Results were expressed as percentage of hFVIII activity.
The presence of FVIII inhibitors in plasma of treated mice was assessed using the Nijmegen modification of the Bethesda assay, as previously described.69
ELISA for Anti-FVIII Antibodies and FVIII-Ag
Plasma samples were diluted 1:200 and 1:2,000 and tested in a solid-phase ELISA, in which ReFacto (0.2 μg/well) or BSA, used as a specificity control, was adsorbed on PVC microwells and saturated with 0.2% BSA. Serum reactivity was detected with horseradish peroxidase-conjugated goat anti-mouse total IgG (Thermo Fisher Scientific), IgG1, IgG2a, and IgG2b (Southern Biotec), and the addition of the chromogen 3,3′,5,5′-tetramethylbenzidine (TMB; Sigma-Aldrich). For IgG isotype quantification, standard curves of IgG1, IgG2a, and IgG2b (Southern Biotec) were tested in parallel. Samples were diluted as described above and pooled plasma from LV.PGK-hFVIII-injected mice that developed anti-FVIII antibodies or a commercial mouse anti-FVIII monoclonal antibody (Clone GMA-8015, Green Mountain Antibodies) were used as positive controls. ELISA for FVIII-Ag detection was performed as reported by McIntosh et al.14 Absorbance was read at 450 nm on a Victor X (PerkinElmer) spectrophotometer.
In Vivo Treg Depletion
For Treg depletion, HA mice were injected via the tail vein with 250–500 μg of anti-CD25 (clone PC61, Bio X Cell) monoclonal antibody (mAb) kindly provided by Dr. A. Annoni (HSR-TIGET).
To break the tolerance, HA mice were injected with 109 TU of LV.VEC-hFVIII (n = 5) or LV.VEC-hFVIII-122–142 (n = 4) and, 11 weeks later, injected with anti-CD25 antibodies. Peripheral blood samples were collected starting 5 days after Treg depletion for Treg quantification by FACS analysis. Plasma was analyzed for hFVIII activity and for anti-FVIII antibodies.
Immunostaining
Mouse liver and spleen were fixed in 4% paraformaldehyde (PFA) in PBS for 2 hr at 4°C, equilibrated in sucrose, embedded in Killik cryostat embedding medium (Bio-Optica), and stored at –80°C; part of the liver samples were fixed in formalin and enclosed in paraffin. Cryostat sections (5–6 μm) were incubated in blocking buffer, while 5- to 6-μm-thick paraffin-embedded mouse liver sections were treated in boiling 50 mM EDTA (pH 8) for antigen retrieval using a microwave oven and then blocked in a buffer containing 5% goat serum, 1% BSA, and 0.1% Triton X-100 in PBS. For FVIII detection, samples were stained with rabbit anti-FVIII (1:1,000) for 2 hr at room temperature (RT).19 For GFP detection, samples were stained with rabbit anti-GFP (1:1,000; Molecular Probes, Thermo Fisher Scientific) for 1 hr at RT. In order to characterize GFP- or FVIII-expressing cells, co-staining was performed with rat anti-mouse F4/80 (1:400; AbD serotec, Bio-Rad), rat anti-mouse Lyve-1 (1:200; eBioscience, Affymetrix), or rat anti-mouse CD31 (1:75; BD PharMingen, BD Biosciences). After washing in PBS containing 0.1% Triton X-100, Alexa Fluor 488-conjugated goat anti-rabbit IgG and Alexa Fluor 546-conjugated goat anti-rat IgG (1:500, Molecular Probes, Thermo Fisher Scientific) were added for 1 hr. Nuclei were stained with DAPI (Sigma-Aldrich). Immunohistochemical reactions were performed using a standard procedure, as previously reported.19, 70 Immunostaining was performed with a Dako Cytomation Envision plus system (DAKO Cytomation), using diaminobenzidine as a chromogen. Sections were counterstained with hemalume (Merck).
Liver Cell Isolation
Hepatic cells were isolated by in situ liver perfusion with collagenase digestion, as previously reported.17, 71 Briefly, liver was perfused at 5.5–6 mL/min via the portal vein at 37°C for 5 min with buffer containing 1.9 mg/ml EGTA, for 2 min with buffer lacking EGTA, and for 12–15 min with buffer containing 0.025% (w/v) collagenase type IV and 5 mM CaCl2. The perfusion buffer contained 10 mmol/L HEPES, 3 mmol/L KCl, 130 mmol/L NaCl, 1 mmol/L NaH2PO4·H2O, and 10 mmol/l d-glucose (pH 7.4). The liver was dissociated, and cells were filtered through Dacron fabric with 80 μm pores and centrifuged at 50 × g for 5 min to isolate hepatocytes. Non-parenchymal cells (NPCs) in the supernatant were pelleted at 350 × g for 10 min, and after red blood cell lysis for 6 min on ice, LSECs or KCs were immunomagnetically selected using anti-CD146 or anti-CD11b + anti-F4/80 (Miltenyi Biotec), respectively. Chemicals and collagenase were from Sigma-Aldrich.
Genomic DNA Isolation and qPCR
Genomic DNA (gDNA) was isolated from cells, liver, or spleen samples using the ReliaPrep gDNA Tissue Miniprep System (Promega). gDNA (50 ng) was used for the qPCR using the GoTaq qPCR Master Mix (Promega). The PCR protocol was as follows: initial denaturation at 95°C for 10 min followed by 35 cycles of denaturation at 95°C for 30 s, annealing, and extension at 60°C for 45 s. Primers used were GAPDH (sense: atcactgccacccagaagact; antisense: atcgaaggtggaagagtggga) and Wpre-dNEF (sense: tggattctgcgcgggacgtc; antisense: ggctaagatctacagctgccttg). Copy number was assessed for each sample by comparison with GAPDH and LV standard curves.
Flow Cytometric Analysis
For hepatic and splenic pDC analysis, livers and spleens were harvested and processed as previously described.35 Samples were stained with PE-conjugated anti-mouse CD11c (Miltenyi Biotec) or PE-conjugated anti-mouse B220 (eBioscience, Affymetrix) and APC-conjugated anti-mouse PDCA-1 (Miltenyi Biotec). For Treg analysis, peripheral blood was collected and analyzed using FACSCalibur for CD4, CD25, and Foxp3 expression starting 5 days after anti-CD25 injection using the Mouse Regulatory T Cell Staining Kit #2 (eBioscience, Affymetrix). For Treg FACS analysis, we used another clone of anti-mouse CD25, clone 7D4 (Miltenyi Biotec), to avoid FACS staining problems caused by using the same clone as was used for Treg depletion. For each sample, 1–2 × 105 events were acquired by FACSCalibur. Data were analyzed using FlowJo software (Tree Star).
Tail Clip Challenge
Tail clip assay was performed as previously described.72 Briefly, mice were anesthetized, and tail tips (2.5–3 mm in diameter) were cut and immersed in saline at 37°C. Bleeding was carried on for a maximum of 10 min; tails were then removed from saline solution and cauterized. Times to stop bleeding were recorded, and the amount of blood loss was evaluated by centrifuging and resuspending samples in red blood lysis buffer. Absorbance was read at 597 nm on a Victor X (PerkinElmer).
Statistical Analysis
Data are shown as mean ± SD. Significance was analyzed using t tests and one-way or two-way ANOVA with Bonferroni post hoc tests in GraphPad Prism version 5 (GraphPad Software); p values < 0.05 were considered to indicate statistical significance.
Author Contributions
S.M. and E.S.C. planned and performed research and analyzed data. E.B. and G.V. performed research and analyzed data. V.B. prepared LVs. V.R.A. and P.S. provided reagents and advice on coagulation assays. T.V., M.K.C., and W.T. generated and characterized the codon-optimized BDD-FVIII. M.P. helped design the FVIII immunization experiments in mice and analyzed data. A.F. conceived the study, generated funding, designed the experiments, and analyzed data. A.F. and S.M. wrote the paper, which was revised by all authors.
Conflicts of Interest
The authors declare no conflict of interest.
Acknowledgments
We would like to thank M.L. Attinà for technical assistance, Professor L. Naldini (HSR-TIGET) for the miRTs, Dr. A. Annoni (HSR-TIGET) for helpful discussion on Treg experiments, and Professor Y. Ginzburg and Dr. C. Borsotti for English revision and critical reading of the manuscript. A.F. was supported in part by the Telethon Foundation (grant GGP09280); European Research Council startup grant 261178; E-Rare HEMO-iPS 2011, AIRC 2012 (project 13166), and CSP 2012. T.V., M.C., and W.T. were supported by VUB GEAR IOF (GENECURE), VUB SRP “Grower” (GENEFIX), and FWO grants.
Footnotes
Supplemental Information includes seven figures and can be found with this article online at http://dx.doi.org/10.1016/j.ymthe.2017.04.029.
Supplemental Information
References
- 1.Bolton-Maggs P.H., Pasi K.J. Haemophilias A and B. Lancet. 2003;361:1801–1809. doi: 10.1016/S0140-6736(03)13405-8. [DOI] [PubMed] [Google Scholar]
- 2.Graw J., Brackmann H.H., Oldenburg J., Schneppenheim R., Spannagl M., Schwaab R. Haemophilia A: from mutation analysis to new therapies. Nat. Rev. Genet. 2005;6:488–501. doi: 10.1038/nrg1617. [DOI] [PubMed] [Google Scholar]
- 3.Roth D.A., Tawa N.E., Jr., O’Brien J.M., Treco D.A., Selden R.F., Factor V.T.T.S.G., Factor VIII Transkaryotic Therapy Study Group Nonviral transfer of the gene encoding coagulation factor VIII in patients with severe hemophilia A. N. Engl. J. Med. 2001;344:1735–1742. doi: 10.1056/NEJM200106073442301. [DOI] [PubMed] [Google Scholar]
- 4.Ishiwata A., Mimuro J., Mizukami H., Kashiwakura Y., Takano K., Ohmori T., Madoiwa S., Ozawa K., Sakata Y. Liver-restricted expression of the canine factor VIII gene facilitates prevention of inhibitor formation in factor VIII-deficient mice. J. Gene Med. 2009;11:1020–1029. doi: 10.1002/jgm.1391. [DOI] [PubMed] [Google Scholar]
- 5.Nathwani A.C., Reiss U.M., Tuddenham E.G., Rosales C., Chowdary P., McIntosh J., Della Peruta M., Lheriteau E., Patel N., Raj D. Long-term safety and efficacy of factor IX gene therapy in hemophilia B. N. Engl. J. Med. 2014;371:1994–2004. doi: 10.1056/NEJMoa1407309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Crudele J.M., Finn J.D., Siner J.I., Martin N.B., Niemeyer G.P., Zhou S., Mingozzi F., Lothrop C.D., Jr., Arruda V.R. AAV liver expression of FIX-Padua prevents and eradicates FIX inhibitor without increasing thrombogenicity in hemophilia B dogs and mice. Blood. 2015;125:1553–1561. doi: 10.1182/blood-2014-07-588194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Annoni A., Cantore A., Della Valle P., Goudy K., Akbarpour M., Russo F., Bartolaccini S., D’Angelo A., Roncarolo M.G., Naldini L. Liver gene therapy by lentiviral vectors reverses anti-factor IX pre-existing immunity in haemophilic mice. EMBO Mol. Med. 2013;5:1684–1697. doi: 10.1002/emmm.201302857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brown B.D., Cantore A., Annoni A., Sergi L.S., Lombardo A., Della Valle P., D’Angelo A., Naldini L. A microRNA-regulated lentiviral vector mediates stable correction of hemophilia B mice. Blood. 2007;110:4144–4152. doi: 10.1182/blood-2007-03-078493. [DOI] [PubMed] [Google Scholar]
- 9.Cantore A., Nair N., Della Valle P., Di Matteo M., Màtrai J., Sanvito F., Brombin C., Di Serio C., D’Angelo A., Chuah M. Hyperfunctional coagulation factor IX improves the efficacy of gene therapy in hemophilic mice. Blood. 2012;120:4517–4520. doi: 10.1182/blood-2012-05-432591. [DOI] [PubMed] [Google Scholar]
- 10.Mátrai J., Cantore A., Bartholomae C.C., Annoni A., Wang W., Acosta-Sanchez A., Samara-Kuko E., De Waele L., Ma L., Genovese P. Hepatocyte-targeted expression by integrase-defective lentiviral vectors induces antigen-specific tolerance in mice with low genotoxic risk. Hepatology. 2011;53:1696–1707. doi: 10.1002/hep.24230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen C.Z., Li L., Lodish H.F., Bartel D.P. MicroRNAs modulate hematopoietic lineage differentiation. Science. 2004;303:83–86. doi: 10.1126/science.1091903. [DOI] [PubMed] [Google Scholar]
- 12.Cantore A., Ranzani M., Bartholomae C.C., Volpin M., Valle P.D., Sanvito F., Sergi L.S., Gallina P., Benedicenti F., Bellinger D. Liver-directed lentiviral gene therapy in a dog model of hemophilia B. Sci. Transl. Med. 2015;7:277ra28. doi: 10.1126/scitranslmed.aaa1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Matsui H., Hegadorn C., Ozelo M., Burnett E., Tuttle A., Labelle A., McCray P.B., Jr., Naldini L., Brown B., Hough C., Lillicrap D. A microRNA-regulated and GP64-pseudotyped lentiviral vector mediates stable expression of FVIII in a murine model of Hemophilia A. Mol. Ther. 2011;19:723–730. doi: 10.1038/mt.2010.290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McIntosh J., Lenting P.J., Rosales C., Lee D., Rabbanian S., Raj D., Patel N., Tuddenham E.G., Christophe O.D., McVey J.H. Therapeutic levels of FVIII following a single peripheral vein administration of rAAV vector encoding a novel human factor VIII variant. Blood. 2013;121:3335–3344. doi: 10.1182/blood-2012-10-462200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Park F., Ohashi K., Kay M.A. Therapeutic levels of human factor VIII and IX using HIV-1-based lentiviral vectors in mouse liver. Blood. 2000;96:1173–1176. [PubMed] [Google Scholar]
- 16.Sabatino D.E., Mackenzie T.C., Peranteau W., Edmonson S., Campagnoli C., Liu Y.L., Flake A.W., High K.A. Persistent expression of hF.IX After tolerance induction by in utero or neonatal administration of AAV-1-F.IX in hemophilia B mice. Mol. Ther. 2007;15:1677–1685. doi: 10.1038/sj.mt.6300219. [DOI] [PubMed] [Google Scholar]
- 17.Follenzi A., Benten D., Novikoff P., Faulkner L., Raut S., Gupta S. Transplanted endothelial cells repopulate the liver endothelium and correct the phenotype of hemophilia A mice. J. Clin. Invest. 2008;118:935–945. doi: 10.1172/JCI32748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shahani T., Covens K., Lavend’homme R., Jazouli N., Sokal E., Peerlinck K., Jacquemin M. Human liver sinusoidal endothelial cells but not hepatocytes contain factor VIII. J. Thromb. Haemost. 2014;12:36–42. doi: 10.1111/jth.12412. [DOI] [PubMed] [Google Scholar]
- 19.Zanolini D., Merlin S., Feola M., Ranaldo G., Amoruso A., Gaidano G., Zaffaroni M., Ferrero A., Brunelleschi S., Valente G. Extrahepatic sources of factor VIII potentially contribute to the coagulation cascade correcting the bleeding phenotype of mice with hemophilia A. Haematologica. 2015;100:881–892. doi: 10.3324/haematol.2014.123117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Terraube V., O’Donnell J.S., Jenkins P.V. Factor VIII and von Willebrand factor interaction: biological, clinical and therapeutic importance. Haemophilia. 2010;16:3–13. doi: 10.1111/j.1365-2516.2009.02005.x. [DOI] [PubMed] [Google Scholar]
- 21.Weiss H.J., Sussman I.I., Hoyer L.W. Stabilization of factor VIII in plasma by the von Willebrand factor. Studies on posttransfusion and dissociated factor VIII and in patients with von Willebrand’s disease. J. Clin. Invest. 1977;60:390–404. doi: 10.1172/JCI108788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Crispe I.N. Hepatic T cells and liver tolerance. Nat. Rev. Immunol. 2003;3:51–62. doi: 10.1038/nri981. [DOI] [PubMed] [Google Scholar]
- 23.Thomson A.W., Knolle P.A. Antigen-presenting cell function in the tolerogenic liver environment. Nat. Rev. Immunol. 2010;10:753–766. doi: 10.1038/nri2858. [DOI] [PubMed] [Google Scholar]
- 24.Breous E., Somanathan S., Vandenberghe L.H., Wilson J.M. Hepatic regulatory T cells and Kupffer cells are crucial mediators of systemic T cell tolerance to antigens targeting murine liver. Hepatology. 2009;50:612–621. doi: 10.1002/hep.23043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tiegs G., Lohse A.W. Immune tolerance: what is unique about the liver. J. Autoimmun. 2010;34:1–6. doi: 10.1016/j.jaut.2009.08.008. [DOI] [PubMed] [Google Scholar]
- 26.Carambia A., Freund B., Schwinge D., Heine M., Laschtowitz A., Huber S., Wraith D.C., Korn T., Schramm C., Lohse A.W. TGF-β-dependent induction of CD4+CD25+Foxp3+ Tregs by liver sinusoidal endothelial cells. J. Hepatol. 2014;61:594–599. doi: 10.1016/j.jhep.2014.04.027. [DOI] [PubMed] [Google Scholar]
- 27.Follenzi A., Raut S., Merlin S., Sarkar R., Gupta S. Role of bone marrow transplantation for correcting hemophilia A in mice. Blood. 2012;119:5532–5542. doi: 10.1182/blood-2011-07-367680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gory S., Vernet M., Laurent M., Dejana E., Dalmon J., Huber P. The vascular endothelial-cadherin promoter directs endothelial-specific expression in transgenic mice. Blood. 1999;93:184–192. [PubMed] [Google Scholar]
- 29.Simard A.R., Soulet D., Gowing G., Julien J.P., Rivest S. Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer’s disease. Neuron. 2006;49:489–502. doi: 10.1016/j.neuron.2006.01.022. [DOI] [PubMed] [Google Scholar]
- 30.Cifarelli V., Libman I.M., Deluca A., Becker D., Trucco M., Luppi P. Increased expression of monocyte CD11b (Mac-1) in overweight recent-onset type 1 diabetic children. Rev. Diabet. Stud. 2007;4:112–117. doi: 10.1900/RDS.2007.4.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Brown B.D., Venneri M.A., Zingale A., Sergi Sergi L., Naldini L. Endogenous microRNA regulation suppresses transgene expression in hematopoietic lineages and enables stable gene transfer. Nat. Med. 2006;12:585–591. doi: 10.1038/nm1398. [DOI] [PubMed] [Google Scholar]
- 32.Davoodian N., Lotfi A.S., Soleimani M., Mola J. MicroRNA-122 overexpression promotes hepatic differentiation of human adipose tissue-derived stem cells. J. Cell. Biochem. 2014;115:1582–1593. doi: 10.1002/jcb.24822. [DOI] [PubMed] [Google Scholar]
- 33.Fish J.E., Santoro M.M., Morton S.U., Yu S., Yeh R.F., Wythe J.D., Ivey K.N., Bruneau B.G., Stainier D.Y., Srivastava D. miR-126 regulates angiogenic signaling and vascular integrity. Dev. Cell. 2008;15:272–284. doi: 10.1016/j.devcel.2008.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang S., Aurora A.B., Johnson B.A., Qi X., McAnally J., Hill J.A., Richardson J.A., Bassel-Duby R., Olson E.N. The endothelial-specific microRNA miR-126 governs vascular integrity and angiogenesis. Dev. Cell. 2008;15:261–271. doi: 10.1016/j.devcel.2008.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Agudo J., Ruzo A., Tung N., Salmon H., Leboeuf M., Hashimoto D., Becker C., Garrett-Sinha L.A., Baccarini A., Merad M., Brown B.D. The miR-126-VEGFR2 axis controls the innate response to pathogen-associated nucleic acids. Nat. Immunol. 2014;15:54–62. doi: 10.1038/ni.2767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Doering C.B., Spencer H.T. Advancements in gene transfer-based therapy for hemophilia A. Expert Rev. Hematol. 2009;2:673–683. doi: 10.1586/EHM.09.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Selvaraj S.R., Scheller A.N., Miao H.Z., Kaufman R.J., Pipe S.W. Bioengineering of coagulation factor VIII for efficient expression through elimination of a dispensable disulfide loop. J. Thromb. Haemost. 2012;10:107–115. doi: 10.1111/j.1538-7836.2011.04545.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lillicrap D., VandenDriessche T., High K. Cellular and genetic therapies for haemophilia. Haemophilia. 2006;12(Suppl 3):36–41. doi: 10.1111/j.1365-2516.2006.01259.x. [DOI] [PubMed] [Google Scholar]
- 39.Brown B.D., Sitia G., Annoni A., Hauben E., Sergi L.S., Zingale A., Roncarolo M.G., Guidotti L.G., Naldini L. In vivo administration of lentiviral vectors triggers a type I interferon response that restricts hepatocyte gene transfer and promotes vector clearance. Blood. 2007;109:2797–2805. doi: 10.1182/blood-2006-10-049312. [DOI] [PubMed] [Google Scholar]
- 40.De Palma M., Venneri M.A., Naldini L. In vivo targeting of tumor endothelial cells by systemic delivery of lentiviral vectors. Hum. Gene Ther. 2003;14:1193–1206. doi: 10.1089/104303403322168028. [DOI] [PubMed] [Google Scholar]
- 41.Kuether E.L., Schroeder J.A., Fahs S.A., Cooley B.C., Chen Y., Montgomery R.R., Wilcox D.A., Shi Q. Lentivirus-mediated platelet gene therapy of murine hemophilia A with pre-existing anti-factor VIII immunity. J. Thromb. Haemost. 2012;10:1570–1580. doi: 10.1111/j.1538-7836.2012.04791.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liu C.L., Ye P., Lin J., Butts C.L., Miao C.H. Anti-CD20 as the B-cell targeting agent in a combined therapy to modulate anti-factor VIII immune responses in hemophilia A inhibitor mice. Front. Immunol. 2014;4:502. doi: 10.3389/fimmu.2013.00502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Peng B., Ye P., Rawlings D.J., Ochs H.D., Miao C.H. Anti-CD3 antibodies modulate anti-factor VIII immune responses in hemophilia A mice after factor VIII plasmid-mediated gene therapy. Blood. 2009;114:4373–4382. doi: 10.1182/blood-2009-05-217315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gaitonde P., Ramakrishnan R., Chin J., Kelleher R.J., Jr., Bankert R.B., Balu-Iyer S.V. Exposure to factor VIII protein in the presence of phosphatidylserine induces hypo-responsiveness toward factor VIII challenge in hemophilia A mice. J. Biol. Chem. 2013;288:17051–17056. doi: 10.1074/jbc.C112.396325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ward N.J., Buckley S.M., Waddington S.N., Vandendriessche T., Chuah M.K., Nathwani A.C., McIntosh J., Tuddenham E.G., Kinnon C., Thrasher A.J., McVey J.H. Codon optimization of human factor VIII cDNAs leads to high-level expression. Blood. 2011;117:798–807. doi: 10.1182/blood-2010-05-282707. [DOI] [PubMed] [Google Scholar]
- 46.Siner J.I., Iacobelli N.P., Sabatino D.E., Ivanciu L., Zhou S., Poncz M., Camire R.M., Arruda V.R. Minimal modification in the factor VIII B-domain sequence ameliorates the murine hemophilia A phenotype. Blood. 2013;121:4396–4403. doi: 10.1182/blood-2012-10-464164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Setiady Y.Y., Coccia J.A., Park P.U. In vivo depletion of CD4+FOXP3+ Treg cells by the PC61 anti-CD25 monoclonal antibody is mediated by FcgammaRIII+ phagocytes. Eur. J. Immunol. 2010;40:780–786. doi: 10.1002/eji.200939613. [DOI] [PubMed] [Google Scholar]
- 48.Annoni A., Brown B.D., Cantore A., Sergi L.S., Naldini L., Roncarolo M.G. In vivo delivery of a microRNA-regulated transgene induces antigen-specific regulatory T cells and promotes immunologic tolerance. Blood. 2009;114:5152–5161. doi: 10.1182/blood-2009-04-214569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Markusic D.M., Hoffman B.E., Perrin G.Q., Nayak S., Wang X., LoDuca P.A., High K.A., Herzog R.W. Effective gene therapy for haemophilic mice with pathogenic factor IX antibodies. EMBO Mol. Med. 2013;5:1698–1709. doi: 10.1002/emmm.201302859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Qadura M., Waters B., Burnett E., Chegeni R., Hough C., Othman M., Lillicrap D. Immunoglobulin isotypes and functional anti-FVIII antibodies in response to FVIII treatment in Balb/c and C57BL/6 haemophilia A mice. Haemophilia. 2011;17:288–295. doi: 10.1111/j.1365-2516.2010.02397.x. [DOI] [PubMed] [Google Scholar]
- 51.Potgieter J.J., Damgaard M., Hillarp A. One-stage vs. chromogenic assays in haemophilia A. Eur. J. Haematol. 2015;94(Suppl 77):38–44. doi: 10.1111/ejh.12500. [DOI] [PubMed] [Google Scholar]
- 52.High K.H., Nathwani A., Spencer T., Lillicrap D. Current status of haemophilia gene therapy. Haemophilia. 2014;20(Suppl 4):43–49. doi: 10.1111/hae.12411. [DOI] [PubMed] [Google Scholar]
- 53.Brown B.D., Shi C.X., Powell S., Hurlbut D., Graham F.L., Lillicrap D. Helper-dependent adenoviral vectors mediate therapeutic factor VIII expression for several months with minimal accompanying toxicity in a canine model of severe hemophilia A. Blood. 2004;103:804–810. doi: 10.1182/blood-2003-05-1426. [DOI] [PubMed] [Google Scholar]
- 54.Brown B.D., Shi C.X., Rawle F.E., Tinlin S., McKinven A., Hough C., Graham F.L., Lillicrap D. Factors influencing therapeutic efficacy and the host immune response to helper-dependent adenoviral gene therapy in hemophilia A mice. J. Thromb. Haemost. 2004;2:111–118. doi: 10.1111/j.1538-7836.2004.00552.x. [DOI] [PubMed] [Google Scholar]
- 55.Hu C., Cela R.G., Suzuki M., Lee B., Lipshutz G.S. Neonatal helper-dependent adenoviral vector gene therapy mediates correction of hemophilia A and tolerance to human factor VIII. Proc. Natl. Acad. Sci. U S A. 2011;108:2082–2087. doi: 10.1073/pnas.1015571108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Powell J.S., Ragni M.V., White G.C., 2nd, Lusher J.M., Hillman-Wiseman C., Moon T.E., Cole V., Ramanathan-Girish S., Roehl H., Sajjadi N. Phase 1 trial of FVIII gene transfer for severe hemophilia A using a retroviral construct administered by peripheral intravenous infusion. Blood. 2003;102:2038–2045. doi: 10.1182/blood-2003-01-0167. [DOI] [PubMed] [Google Scholar]
- 57.Lheriteau E., Davidoff A.M., Nathwani A.C. Haemophilia gene therapy: Progress and challenges. Blood Rev. 2015;29:321–328. doi: 10.1016/j.blre.2015.03.002. [DOI] [PubMed] [Google Scholar]
- 58.Lewis J.H., Bontempo F.A., Spero J.A., Ragni M.V., Starzl T.E. Liver transplantation in a hemophiliac. N. Engl. J. Med. 1985;312:1189–1190. doi: 10.1056/NEJM198505023121812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Webster W.P., Zukoski C.F., Hutchin P., Reddick R.L., Mandel S.R., Penick G.D. Plasma factor VIII synthesis and control as revealed by canine organ transplantation. Am. J. Physiol. 1971;220:1147–1154. doi: 10.1152/ajplegacy.1971.220.5.1147. [DOI] [PubMed] [Google Scholar]
- 60.Do H., Healey J.F., Waller E.K., Lollar P. Expression of factor VIII by murine liver sinusoidal endothelial cells. J. Biol. Chem. 1999;274:19587–19592. doi: 10.1074/jbc.274.28.19587. [DOI] [PubMed] [Google Scholar]
- 61.Hellman L., Smedsröd B., Sandberg H., Pettersson U. Secretion of coagulant factor VIII activity and antigen by in vitro cultivated rat liver sinusoidal endothelial cells. Br. J. Haematol. 1989;73:348–355. doi: 10.1111/j.1365-2141.1989.tb07751.x. [DOI] [PubMed] [Google Scholar]
- 62.Kumaran V., Benten D., Follenzi A., Joseph B., Sarkar R., Gupta S. Transplantation of endothelial cells corrects the phenotype in hemophilia A mice. J. Thromb. Haemost. 2005;3:2022–2031. doi: 10.1111/j.1538-7836.2005.01508.x. [DOI] [PubMed] [Google Scholar]
- 63.van Solingen C., Seghers L., Bijkerk R., Duijs J.M., Roeten M.K., van Oeveren-Rietdijk A.M., Baelde H.J., Monge M., Vos J.B., de Boer H.C. Antagomir-mediated silencing of endothelial cell specific microRNA-126 impairs ischemia-induced angiogenesis. J. Cell. Mol. Med. 2009;13(8A):1577–1585. doi: 10.1111/j.1582-4934.2008.00613.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lange A.M., Altynova E.S., Nguyen G.N., Sabatino D.E. Overexpression of factor VIII after AAV delivery is transiently associated with cellular stress in hemophilia A mice. Mol. Ther. Methods Clin. Dev. 2016;3:16064. doi: 10.1038/mtm.2016.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Pipe S.W., Kaufman R.J. Characterization of a genetically engineered inactivation-resistant coagulation factor VIIIa. Proc. Natl. Acad. Sci. U S A. 1997;94:11851–11856. doi: 10.1073/pnas.94.22.11851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Carambia A., Frenzel C., Bruns O.T., Schwinge D., Reimer R., Hohenberg H., Huber S., Tiegs G., Schramm C., Lohse A.W., Herkel J. Inhibition of inflammatory CD4 T cell activity by murine liver sinusoidal endothelial cells. J. Hepatol. 2013;58:112–118. doi: 10.1016/j.jhep.2012.09.008. [DOI] [PubMed] [Google Scholar]
- 67.Follenzi A., Naldini L. Generation of HIV-1 derived lentiviral vectors. Methods Enzymol. 2002;346:454–465. doi: 10.1016/s0076-6879(02)46071-5. [DOI] [PubMed] [Google Scholar]
- 68.Geraerts M., Willems S., Baekelandt V., Debyser Z., Gijsbers R. Comparison of lentiviral vector titration methods. BMC Biotechnol. 2006;6:34. doi: 10.1186/1472-6750-6-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Verbruggen B., Novakova I., Wessels H., Boezeman J., van den Berg M., Mauser-Bunschoten E. The Nijmegen modification of the Bethesda assay for factor VIII:C inhibitors: improved specificity and reliability. Thromb. Haemost. 1995;73:247–251. [PubMed] [Google Scholar]
- 70.Valente G., Mamo C., Bena A., Prudente E., Cavaliere C., Kerim S., Nicotra G., Comino A., Palestro G., Isidoro C., Beatrice F. Prognostic significance of microvessel density and vascular endothelial growth factor expression in sinonasal carcinomas. Hum. Pathol. 2006;37:391–400. doi: 10.1016/j.humpath.2005.11.021. [DOI] [PubMed] [Google Scholar]
- 71.Merlin S., Bhargava K.K., Ranaldo G., Zanolini D., Palestro C.J., Santambrogio L., Prat M., Follenzi A., Gupta S. Kupffer cell transplantation in mice for elucidating monocyte/macrophage biology and for potential in cell or gene therapy. Am. J. Pathol. 2016;186:539–551. doi: 10.1016/j.ajpath.2015.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Schuettrumpf J., Herzog R.W., Schlachterman A., Kaufhold A., Stafford D.W., Arruda V.R. Factor IX variants improve gene therapy efficacy for hemophilia B. Blood. 2005;105:2316–2323. doi: 10.1182/blood-2004-08-2990. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.