Abstract
Objectives:
Evidence suggests a possible role of Fusobacterium nucleatum in colorectal carcinogenesis, especially in right-sided proximal colorectum. Considering a change in bowel contents and microbiome from proximal to distal colorectal segments, we hypothesized that the proportion of colorectal carcinoma enriched with F. nucleatum might gradually increase along the bowel subsites from rectum to cecum.
Methods:
A retrospective, cross-sectional analysis was conducted on 1,102 colon and rectal carcinomas in molecular pathological epidemiology databases of the Nurses’ Health Study and the Health Professionals Follow-up Study. We measured the amount of F. nucleatum DNA in colorectal tumor tissue using a quantitative PCR assay and equally dichotomized F. nucleatum-positive cases (high vs. low). We used multivariable logistic regression analysis to examine the relationship of a bowel subsite variable (rectum, rectosigmoid junction, sigmoid colon, descending colon, splenic flexure, transverse colon, hepatic flexure, ascending colon, and cecum) with the amount of F. nucleatum.
Results:
The proportion of F. nucleatum-high colorectal cancers gradually increased from rectal cancers (2.5% 4/157) to cecal cancers (11% 19/178), with a statistically significant linear trend along all subsites (P<0.0001) and little evidence of non-linearity. The proportion of F. nucleatum-low cancers was higher in rectal, ascending colon, and cecal cancers than in cancers of middle segments.
Conclusions:
The proportion of F. nucleatum-high colorectal cancers gradually increases from rectum to cecum. Our data support the colorectal continuum model that reflects pathogenic influences of the gut microbiota on neoplastic and immune cells and challenges the prevailing two-colon (proximal vs. distal) dichotomy paradigm.
Introduction
An increasing body of evidence suggests possible roles of microorganisms in colorectal carcinogenesis.1, 2, 3, 4, 5, 6 Among various microbial species, Fusobacterium nucleatum appears to inhibit antitumor immune response and potentiate colonic neoplasia development in animal models.7, 8, 9, 10 In addition, an enrichment of F. nucleatum can be observed in a subset of human colorectal neoplasms, and a high amount of F. nucleatum in carcinoma tissue has been associated with proximal tumor location, high-level microsatellite instability (MSI-high), and lower density of T cells in tumor tissue.11, 12, 13, 14, 15, 16, 17, 18
As a long organ, the colorectum is typically divided into proximal colon (cecum to transverse colon), distal colon (splenic flexure to sigmoid colon), and rectum in clinical, pathological, and epidemiological studies.19, 20, 21 However, multiple studies have demonstrated that proportions of colorectal cancers with specific molecular features such as MSI-high, CpG island methylator phenotype (CIMP)-high, and BRAF and PIK3CA mutations gradually increase along the bowel subsites from rectum to ascending colon.22, 23, 24, 25 These findings are consistent with the fact that microbiota, bacterial metabolites, and other contents of the large intestine continually (rather than abruptly) change from the proximal to distal segments26, 27, 28, 29 and support the colorectal continuum model rather than the dichotomy or trichotomy model.19, 20, 21 Hence, we hypothesized that the proportion of colorectal cancer enriched with F. nucleatum might gradually change along the bowel subsites from cecum to rectum.
To test this hypothesis, we utilized a database of colorectal carcinoma cases in two US nationwide prospective cohort studies, the Nurses’ Health Study and the Health Professionals Follow-up Study and examined the amount of F. nucleatum in colorectal cancer tissue according to the bowel subsites.
Methods
Study population
We utilized the database of colorectal carcinoma cases in two US nationwide prospective cohort studies, the Nurses’ Health Study (121,701 women enrolled in 1976) and the Health Professionals Follow-up Study (51,529 men enrolled in 1986)30, 31 and conducted a retrospective, cross-sectional analysis to assess the association of the amount of F. nucleatum in colorectal cancer tissue with tumor location. Every 2 years, we sent participants follow-up questionnaires to collect information on lifestyle factors and asked whether they had received diagnoses of major disease, including cancers. Study physicians reviewed medical records for incident colorectal cancer cases and recorded cancer stage (Tumor, Node, Metastasis) and tumor location (cecum, ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, sigmoid colon, rectosigmoid junction, and rectum).22
We collected formalin-fixed paraffin-embedded (FFPE) tissue blocks from hospitals where participants with colorectal carcinoma had undergone tumor resection. A single pathologist (S.O.), who was unaware of other data, conducted a centralized review of hematoxylin and eosin–stained tissue sections of all colorectal carcinoma cases and recorded pathological features. Tumor differentiation was classified into well to moderate or poor (>50% vs. ≤50% glandular area). Written informed consent was obtained from all study participants. The institutional review boards at the Harvard T.H. Chan School of Public Health and the Brigham and Women’s Hospital (Boston, MA) approved the cohort studies.
Quantitative PCR for F. nucleatum
We dissected colorectal cancer tissues from whole-tissue sections of FFPE tissue blocks, and DNA was extracted using the QIAamp DNA FFPE Tissue Kit (Qiagen, Valencia, CA). After the quantitative PCR assay for F. nucleatum was developed and validated as previously described,16 we measured the amount of tissue F. nucleatum DNA in 1,102 colorectal carcinoma cases, while blinded to data on tumor location and other clinical, pathological, and tumor molecular features. Custom TaqMan primer/probe sets (Applied Biosystems, San Diego, CA) for the nusG gene of F. nucleatum and for the reference human gene SLCO2A1 were used as previously described.16 Each reaction contained 80 ng of genomic DNA and was assayed in 20 μl reactions containing 1 × final concentration TaqMan Environmental Master Mix 2.0 (Applied Biosystems) and each TaqMan Gene Expression Assay (Applied Biosystems) in a 96-well optical PCR plate. Amplification and detection of DNA was performed with the StepOnePlus Real-Time PCR Systems (Applied Biosystems) using the following reaction conditions: 10 min at 95 °C and 45 cycles of 15 s at 95 °C and 1 min at 60 °C.
Our validation study has previously shown that, in colorectal carcinoma cases with detectable F. nucleatum DNA, the cycle threshold (Ct) values in the quantitative PCR for F. nucleatum and SLCO2A1 decreased linearly with the log-transformed amount of input DNA from the same specimen (r2>0.99), and that the interassay coefficient of variation of Ct values from the same specimen in five different batches was ≤1% for all targets.16 Each specimen was analyzed in duplicate for each target in a single batch, and we used the mean of the two Ct values for each target. Spearman’s rank-correlation coefficients between the two Ct values (in duplicated runs) in each of cases with detectable target amplification in the quantitative PCR assays for F. nucleatum and SLCO2A1 were 0.95 and 0.92, respectively.16 The amount of tissue F. nucleatum DNA in each specimen was calculated as a relative unitless value normalized with SLCO2A1 using the 2−ΔCt method (where ΔCt=“the mean Ct value of F. nucleatum”−“the mean Ct value of SLCO2A1”).16
Cases with detectable F. nucleatum DNA were categorized as low or high based on the median cutpoint while cases without detectable F. nucleatum DNA were categorized as negative, to keep consistent classification system with our previous study.32
Analyses of MSI, DNA methylation, and KRAS, BRAF, and PIK3CA mutations
Using DNA extracted from FFPE colorectal carcinoma tissue, MSI status was analyzed with the use of 10 microsatellite markers (D2S123, D5S346, D17S250, BAT25, BAT26, BAT40, D18S55, D18S56, D18S67, and D18S487) as previously described.33 We defined MSI-high as the presence of instability in ≥30% of the markers, and MSI-low/microsatellite stable (MSS) as instability in <30% of the markers. Methylation analyses of long interspersed nucleotide element-1 (LINE-1)34 and eight promoter CpG islands specific for CIMP (CACNA1G, CDKN2A, CRABP1, IGF2, MLH1, NEUROG1, RUNX3, and SOCS1) were performed as previously described.35, 36 PCR reaction and pyrosequencing were performed for KRAS (codons 12, 13, 61, and 146),37, 38 BRAF (codon 600),33 and PIK3CA (exons 9 and 20).39, 40
Statistical analysis
All statistical analyses were conducted using SAS (version 9.3, SAS Institute, Cary, NC) and all P values were two-sided. For our primary hypothesis testing, we examined the relationship of the tumor location variable (the nine subsites) with the amount of F. nucleatum DNA in colorectal cancer tissue (as an outcome variable). There was an initial flexibility of the outcome variable, which could be raw continuous, log-transformed (continuous), ordinal three-tiered (high vs. low vs. negative), binary (high vs. low/negative), or binary (high/low vs. negative). Considering these five possibilities, we used adjusted two-sided α level of 0.01 (=0.05/5). Neither the amount of F. nucleatum DNA nor its log-transformed value fitted a normal distribution with the use of the Kolmogorov–Smirnov test for normality (P<0.01). Thus we used multivariable logistic regression analysis to examine the relationship of the nine subsites (as a predictor variable; tested for a linear trend with one degree of freedom) with categorical outcome variables of F. nucleatum. For the ordinal outcome variable, the proportionality of odds assumption was not satisfied in ordinal logistic regression models (P=0.019). According to the distribution of colorectal cancer cases by the subsites and the amount of F. nucleatum (Figure 1), we used a binary outcome variable (F. nucleatum-high vs. -low/negative) in the logistic regression model. For the subsite variable, we assigned population average distance from anal verge to each bowel subsite (either the midpoint or junction/flexure), which was calculated based on published data using computed tomographic colonography,22, 41 as follows: rectum (the midpoint), 9.8 cm; rectosigmoid junction, 20 cm; sigmoid colon (the midpoint), 44 cm; descending colon (the midpoint), 85 cm; splenic flexure, 102 cm; transverse colon (the midpoint), 131 cm; hepatic flexure, 160 cm; ascending colon (the midpoint), 171 cm; and cecum (the midpoint), 186 cm. A significant P value by the Wald’s test on the bowel subsite variable indicated a linear relationship of the bowel subsite with tissue F. nucleatum, but a curvilinear relationship might exist. Thus we assessed the non-linearity by a likelihood ratio test comparing the model with squared and/or cubic subsite variables with the model without squared or cubic subsite variable; a significant likelihood ratio test result would indicate the presence of non-linearity (curvilinearity).
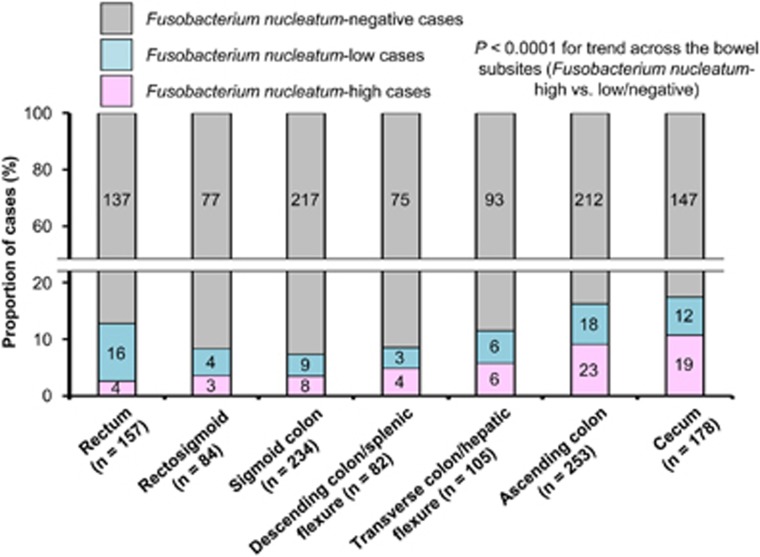
Figure 1.
Proportions of Fusobacterium nucleatum-negative, F. nucleatum-low, and F. nucleatum-high colorectal carcinoma cases along the bowel subsites. P-value was calculated by the linear trend test across the bowel subsite variable (population average distance from anal verge to each subsite (cm)) as a continuous variable in the univariable logistic regression model to predict the amount of tissue F. nucleatum (as a binary outcome variable (high vs. low/negative)).
The multivariable logistic regression models were adjusted for clinical features, including age (continuous), sex, year of diagnosis (continuous), and family history of colorectal carcinoma in any first-degree relative (present vs. absent). Studies have shown an enrichment of F. nucleatum in colorectal adenomas (before progression to carcinomas),9, 13, 15 suggesting that F. nucleatum may be involved in early colorectal carcinogenesis. Hence, pathological and tumor molecular features of colorectal carcinoma may be present downstream in the causal sequence of events after the Fusobacterium variable, and adjusting for the tumor pathological and molecular features might cause biased results. Thus we did not include the pathological and tumor molecular variables in the multivariable logistic regression models. For cases with missing information on family history of colorectal carcinoma in a first-degree relative (1.3%), we included those cases in a majority category of a given covariate to minimize the number of variables in multivariable logistic regression models. We confirmed that excluding the cases with missing information on family history of colorectal carcinoma in a first-degree relative did not substantially alter the results (data not shown).
To assess the associations between the amount of tissue F. nucleatum and other categorical variables, chi-square test was performed. To compare mean age and mean LINE-1 methylation levels, an analysis of variance was performed. These comparisons represented secondary analyses, and we used adjusted α level of 0.003 (=0.05/14) by simple Bonferroni correction for multiple hypothesis testing.
Results
F. nucleatum in colorectal carcinoma tissue
We measured the amount of tissue F. nucleatum DNA in 1,102 colorectal carcinoma cases within the two prospective cohort studies using the quantitative PCR assay that was previously validated.16 F. nucleatum DNA was detected (positive) in colorectal carcinoma tissue in 138 (13%) of the 1,102 cases and undetectable (negative) in the remaining 964 cases (87%). We equally dichotomized the 138 cases with detectable F. nucleatum DNA levels into two groups to keep consistency with our previous study.32 Clinical, pathological, and tumor molecular features according to the amount (high vs. low. vs. negative) of tissue F. nucleatum are summarized in Table 1. High-level F. nucleatum in colorectal cancer tissue was associated with proximal tumor location, poor tumor differentiation, MSI-high, MLH1 hypermethylation, CIMP-high, and BRAF mutation (P≤0.0004 with the adjusted α level of 0.003 for multiple hypothesis testing).
Table 1. Characteristics of colorectal cancer cases according to the amount of tissue Fusobacterium nucleatum.
| Characteristicsa | All patients (n=1,102) |
The amount of tissue
Fusobacterium nucleatum |
P valueb | ||
|---|---|---|---|---|---|
| Negative (n=964) | Low (n=69) | High (n=69) | |||
| Mean age±s.d. (years) | 69.5±8.9 | 69.4±8.9 | 70.9±9.0 | 69.2±8.5 | 0.39 |
| Sex | 0.35 | ||||
| Men | 466 (42%) | 415 (43%) | 27 (39%) | 24 (35%) | |
| Women | 636 (58%) | 549 (57%) | 42 (61%) | 45 (65%) | |
| Year of diagnosis | 0.026 | ||||
| Prior to 1995 | 352 (32%) | 323 (34%) | 12 (17%) | 17 (25%) | |
| 1996–2000 | 301 (27%) | 262 (27%) | 19 (28%) | 20 (29%) | |
| 2001–2008 | 449 (41%) | 379 (39%) | 38 (55%) | 32 (46%) | |
| Family history of colorectal carcinoma in a first-degree relative | 0.26 | ||||
| Absent | 877 (81%) | 762 (80%) | 60 (88%) | 55 (81%) | |
| Present | 211 (19%) | 190 (20%) | 8 (12%) | 13 (19%) | |
| Tumor location | 0.0004 | ||||
| Proximal colon | 536 (49%) | 452 (47%) | 36 (53%) | 48 (72%) | |
| Distal colon | 316 (29%) | 292 (31%) | 12 (18%) | 12 (18%) | |
| Rectum | 241 (22%) | 214 (22%) | 20 (29%) | 7 (10%) | |
| Nine bowel subsites c | <0.0001d | ||||
| Cecum | 178 | 147 (83%) | 12 (6.7%) | 19 (11%) | |
| Ascending colon | 253 | 212 (84%) | 18 (7.1%) | 23 (9.1%) | |
| Hepatic flexure | 32 | 28 (88%) | 2 (6.3%) | 2 (6.3%) | |
| Transverse colon | 73 | 65 (89%) | 4 (5.5%) | 4 (5.5%) | |
| Splenic flexure | 29 | 27 (93%) | 1 (3.5%) | 1 (3.5%) | |
| Descending colon | 53 | 48 (91%) | 2 (3.8%) | 3 (5.7%) | |
| Sigmoid colon | 234 | 217 (93%) | 9 (3.8%) | 8 (3.4%) | |
| Rectosigmoid junction | 84 | 77 (92%) | 4 (4.8%) | 3 (3.6%) | |
| Rectum | 157 | 137 (87%) | 16 (10%) | 4 (2.5%) | |
| Disease stage | 0.006 | ||||
| I | 247 (25%) | 230 (26%) | 10 (16%) | 7 (11%) | |
| II | 331 (33%) | 279 (32%) | 23 (37%) | 29 (45%) | |
| III | 286 (29%) | 246 (28%) | 25 (40%) | 15 (24%) | |
| IV | 135 (13%) | 117 (14%) | 5 (7.9%) | 13 (20%) | |
| Tumor differentiation | <0.0001 | ||||
| Well to moderate | 994 (90%) | 887 (92%) | 57 (84%) | 50 (72%) | |
| Poor | 106 (9.6%) | 76 (7.9%) | 11 (16%) | 19 (28%) | |
| MSI status | <0.0001 | ||||
| MSI-low/MSS | 885 (84%) | 805 (87%) | 44 (67%) | 36 (54%) | |
| MSI-high | 171 (16%) | 118 (13%) | 22 (33%) | 31 (46%) | |
| MLH1 hypermethylation | <0.0001 | ||||
| Absent | 869 (86%) | 782 (89%) | 50 (79%) | 37 (58%) | |
| Present | 140 (14%) | 100 (11%) | 13 (21%) | 27 (42%) | |
| CIMP status | <0.0001 | ||||
| Low/negative | 823 (82%) | 737 (84%) | 50 (79%) | 36 (56%) | |
| High | 186 (18%) | 145 (16%) | 13 (21%) | 28 (44%) | |
| BRAF mutation | <0.0001 | ||||
| Wild type | 892 (84%) | 795 (85%) | 52 (79%) | 45 (66%) | |
| Mutant | 172 (16%) | 135 (15%) | 14 (21%) | 23 (34%) | |
| KRAS mutation | 0.51 | ||||
| Wild type | 569 (57%) | 501 (57%) | 30 (51%) | 38 (61%) | |
| Mutant | 435 (43%) | 382 (43%) | 29 (49%) | 24 (39%) | |
| PIK3CA mutation | 0.88 | ||||
| Wild type | 841 (84%) | 738 (84%) | 49 (82%) | 54 (83%) | |
| Mutant | 162 (16%) | 140 (16%) | 11 (18%) | 11 (17%) | |
| Mean LINE-1 methylation level, %±s.d. | 63.5±10.2 | 63.3±10.2 | 65.0±10.6 | 65.4±8.9 | 0.14 |
CIMP, CpG island methylator phenotype; LINE-1, long interspersed nucleotide element-1; MSI, microsatellite instability; MSS, microsatellite stable.
Percentage (%) indicates the proportion of cases with a specific clinical, pathological, or tumor molecular feature according to the amount of tissue Fusobacterium nucleatum. There were cases which had missing values for any of the characteristics except for age, sex, and year of diagnosis.
To assess associations between the ordinal categories (negative, low, and high) of the amount of tissue F. nucleatum and categorical variables, the chi-square test was performed. To compare mean age and mean LINE-1 methylation levels, an analysis of variance was performed. We adjusted two-sided α level to 0.003 (=0.05/14) by simple Bonferroni correction for multiple hypothesis testing.
Percentage indicates the proportion of F. nucleatum-negative, F. nucleatum-low, or F. nucleatum-high cases among all tumors in a given bowel subsite.
P value was calculated by the linear trend test across the bowel subsite variable (population average distance from anal verge to each subsite (cm)) as a continuous variable in the univariable logistic regression model to predict the amount of tissue F. nucleatum (as a binary outcome variable (high vs. low/negative)).
The relationship of the bowel subsites with the amount of F. nucleatum in colorectal cancer tissue
The amount of F. nucleatum in colorectal cancer tissue according to the bowel subsites from rectum to cecum is shown in Figure 1. The proportion of F. nucleatum-high cancers gradually increased from rectal cancers (2.5%=4/157) to cecal cancers (11%=19/178). In contrast, the proportion of F. nucleatum-low cancers was higher in rectal cancers (10%=16/157), ascending colon cancers (7.1%=18/253), and cecal cancers (6.7%=12/178) than in cancers of middle segments.
We assessed the relationship of the bowel subsite (as a predictor variable) with the amount of F. nucleatum in colorectal cancer tissue (as a binary outcome variable (high vs. low/negative)) by multivariable logistic regression analysis that adjusted for potential confounders (Table 2). The bowel subsite variable was significantly associated with high-level F. nucleatum in colorectal cancer tissue (Ptrend<0.0001 for trend across the bowel subsites from rectum to cecum, with the adjusted α level of 0.01). We demonstrated little evidence of non-linearity (P≥0.61) using likelihood ratio test, which compared the model with squared and/or cubic subsite variables to the model without squared or cubic subsite variable.
Table 2. Assessment of the linearity and non-linearity on the relationship of the bowel subsites with the amount of Fusobacterium nucleatum in colorectal cancer tissue by multivariable logistic regression analyses.
| Bowel subsite variable (distance from anal verge to each subsite (cm)) |
Squared subsite variable |
Cubic subsite variable |
Likelihood ratio test |
|||
|---|---|---|---|---|---|---|
| P value (Wald’s test)a | Included | P value (Wald’s test) | Included | P value (Wald’s test) | Degree of freedom | P valueb |
| Model for the amount of tissue Fusobacterium nucleatum (as an outcome variable (high vs. low/negative)) | ||||||
| All cases | ||||||
| <0.0001 | No | — | No | — | — | Referent |
| 0.87 | Yes | 0.61 | No | — | 1 | 0.61 |
| 0.65 | Yes | 0.74 | Yes | 0.67 | 2 | 0.80 |
| Cases from sigmoid colon to cecum (excluding rectal and rectosigmoid cancers) | ||||||
| 0.002 | No | — | No | — | — | Referent |
| 0.82 | Yes | 0.51 | No | — | 1 | 0.50 |
| 0.84 | Yes | 0.85 | Yes | 0.80 | 2 | 0.77 |
Multivariable logistic regression model included age, sex, year of diagnosis, family history of colorectal cancer in parent or sibling, and the bowel subsite variable with or without the squared and cubic subsite variable, as indicated in the Table. We adjusted two-sided α level to 0.01 (=0.05/5) for multiple hypothesis testing.
P value was calculated by the Wald’s test on the bowel subsite variable (population average distance from anal verge to each subsite (cm)) as a continuous variable in the multivariable logistic regression model to predict the amount of tissue Fusobacterium nucleatum (as a binary outcome variable (high vs. low/negative)).
A significant P value by the likelihood ratio test indicates a non-linear (curvilinear) relationship, and a combination of insignificant P values by the likelihood ratio test and a significant P value by the Wald test on the bowel subsite variable in the model without the squared or cubic subsite variable indicates a linear relationship.
To exclude a potential influence of preoperative chemotherapy and/or radiation therapy for rectal cancers as a secondary analysis, we excluded cancers in the rectum and rectosigmoid and performed a linearity test. The bowel subsite variable (from sigmoid colon to cecum) was significantly associated with high-level tissue F. nucleatum (Ptrend=0.002 for trend across the bowel subsites, with the adjusted α level of 0.01), and there was no evidence for non-linearity (P≥0.50; Table 2).
Discussion
We found that the proportion of F. nucleatum-high colorectal cancers increased linearly along the large intestine from rectum to cecum. Although differences in clinical, pathological, and epidemiological features between proximal and distal colon cancers and between colon and rectal cancers have been known for decades,19, 20, 21 emerging evidence indicates a gradual change in the proportions of key molecular features of colorectal cancer along the bowel subsites.22, 23, 24, 25 Our data challenge the common dichotomy model and support the colorectal continuum model that recently emerged.42, 43
Analyses of molecular pathology are increasing importance in cancer research.44, 45, 46, 47, 48, 49, 50, 51 Accumulating evidence suggests that colorectal tumors arise with sets of genomic and epigenomic alterations through interactions between neoplastic cells, immune cells, and microbiota that vary along the proximal to distal axis of colorectum.42 In the current study, high-level tissue F. nucleatum DNA was associated with the molecular features of colorectal cancer, including MSI-high, CIMP-high, and BRAF mutation. Some studies reported that butyrate might suppress colonic inflammation and tumor development.52, 53 In contrast, emerging evidence suggests that butyrate may promote the growth of colonic tumors that exhibit DNA mismatch repair deficiency.5, 54 F. nucleatum is known to be one of the microbes that produce butyrate.1, 5 Thus F. nucleatum might promote the development and progression of colorectal tumors through the production of butyrate, although additional studies are needed to elucidate the exact mechanisms underlying the association between F. nucleatum and colorectal carcinogenesis.
Cecal carcinomas represent an interesting subgroup of colorectal carcinomas characterized by high prevalence of KRAS mutations.22, 55 In addition, along with rectum, cecum shows the highest incidence of carcinoma occurrence per surface area of mucosa.56 Our current study has shown that F. nucleatum-enriched carcinomas are most prevalent in cecum compared with other subsites. Future studies should investigate the role of microbiota in cecal carcinogenesis.
Epidemiological evidence indicates a recent increase in the proportion of proximal colon cancers in Western countries57, 58 and the association between postcolonoscopy cancer and proximal tumor location.59, 60 Because any experimental system cannot perfectly recapitulate the complex nature of human tumor or microorganisms, analyses of human cancer tissue in a large population are useful in elucidating the relationship between microorganisms and cancer. However, no previous study has examined the amount of specific microbial species in human colorectal cancers according to detailed subsites (using an enough sample size), as we did in this study. Our population-based human data would guide future mechanistic investigations. Considering that diet, lifestyle, pharmacological factors (including antibiotics), and probiotics and prebiotics can influence the composition of intestinal microbiota,61, 62, 63 future investigations may be warranted to examine potential influences of those modifiable factors on the intestinal microflora and tumorigenic processes.
Strengths of this study include the use of our molecular pathological epidemiology64, 65 database (of 1,102 colorectal carcinoma cases in the two US nationwide, prospective cohort studies), which integrates epidemiological exposures, clinical characteristics, and tissue F. nucleatum in colorectal carcinoma. The sample size and the comprehensiveness of the colorectal cancer database enabled us to examine the amounts of F. nucleatum in colorectal cancer tissue in each of the bowel subsites and test the linearity of the relationship of the bowel subsites with the amount of F. nucleatum, while adjusting for clinical features. Importantly, our data set of colorectal cancer cases represented a population-based sample derived from a large number of hospitals in diverse settings across the United States that increases the generalizability of our findings.
We recognize limitations of our study. First, routine histopathology processing might have influenced the performance of the quantitative PCR assay to detect microorganisms in FFPE tissue specimens. Although measurement errors in FFPE tissue specimens would have likely driven our results toward the null hypothesis, we cannot exclude unmeasured confounding factors. However, our validation study has demonstrated a high linearity (r2>0.99) and a high reproducibility (interassay coefficient of variation ≤1%) of the quantitative PCR assay for F. nucleatum with the use of FFPE tissue specimens.16 In addition, our data on the relationships of F. nucleatum with clinicopathological characteristics and tumor molecular features, including MSI and CIMP status, are consistent with the study using a quantitative PCR assay for frozen tissue specimens.14 Second, rectal cancers are commonly treated by preoperative chemotherapy and/or radiation, which might have changed the gut and tumor microbiota. Therefore, we excluded preoperatively treated rectal cancers in which adequate pretreatment biopsy specimens were unavailable. In addition, as a secondary analysis, we excluded rectal and rectosigmoid cancers and observed similar findings on the association of bowel subsites (from the sigmoid colon to cecum) with the amount of tissue F. nucleatum. In the current study, rectal cancers showed a high proportion of F. nucleatum-low cases (10%) compared with rectosigmoid (4.8%) and sigmoid colon cancers (3.8%). These findings need to be validated by additional studies. Third, we did not examine other microbes (including Escherichia coli and Bacteroides fragilis66, 67) in colorectal cancer tissue or data on stool microbiota. Certainly, future comprehensive metagenomic analyses on tissue and stool microbiota may provide further insights on roles of gut microorganisms in the development and progression of colorectal tumors. Fourth, as an observational study, we could not conclude on a potential causal effect of F. nucleatum. Nonetheless, given complex roles of interactions between microbial and host factors in human carcinogenesis, we believe that our novel data on F. nucleatum (which appears to have a role in carcinogenesis in experimental studies8, 9, 10) in >1,000 colorectal cancer cases along the bowel subsites represent valuable information.
In conclusion, utilizing the database of the 1,102 colorectal carcinoma cases in the US nationwide prospective cohort studies, we have found that the proportion of colorectal cancer enriched with F. nucleatum increases linearly along the bowel subsites from rectum to cecum. Our human population-based data suggest a continuum model of pathogenic influences of F. nucleatum on colorectal carcinogenesis, which may be targeted for colorectal cancer prevention and treatment in the future.
Study Highlights

Acknowledgments
We thank the participants and staff of the Nurses’ Health Study and the Health Professionals Follow-up Study for their valuable contributions as well as the following state cancer registries for their help: AL, AZ, AR, CA, CO, CT, DE, FL, GA, ID, IL, IN, IA, KY, LA, ME, MD, MA, MI, NE, NH, NJ, NY, NC, ND, OH, OK, OR, PA, RI, SC, TN, TX, VA, WA, and WY. We assume full responsibility for analyses and interpretation of these data. We have used HUGO (Human Genome Organisation)-approved official symbols for genes and gene products, including BRAF, CACNA1G, CDKN2A, CRABP1, IGF2, KRAS, MLH1, NEUROG1, PIK3CA, RUNX3, SLCO2A1, and SOCS1, all of which are described at www.genenames.org.
Footnotes
Supplementary Information accompanies this paper on the Clinical and Translational Gastroenterology website (http://www.nature.com/ctg)
Guarantor of the article: Shuji Ogino, MD, PhD, MS.
Specific author contributions: All authors contributed to review and revision. Kosuke Mima, Caitlin A. Brennan, Danny A. Milner, Levi A. Garraway, Jeffrey A. Meyerhardt, Wendy S. Garrett, Curtis Huttenhower, Matthew Meyerson, Edward L. Giovannucci, Andrew T. Chan, Charles S. Fuchs, and Shuji Ogino developed the main concept and designed the study. Andrew T. Chan, Charles S. Fuchs, and Shuji Ogino wrote grant applications. Kosuke Mima, Yin Cao, Reiko Nishihara, Zhi Rong Qian, Jonathan A. Nowak, Yohei Masugi, Yan Shi, Annacarolina da Silva, Mancang Gu, Wanwan Li, Tsuyoshi Hamada, Keisuke Kosumi, Akiko Hanyuda, Li Liu, Mingyang Song, Jeffrey A. Meyerhardt, Edward L. Giovannucci, Andrew T. Chan, Charles S. Fuchs, and Shuji Ogino were responsible for collection of tumor tissue and acquisition of epidemiological, clinical and tumor tissue data, including histopathological and immunohistochemical characteristics. Kosuke Mima, Aleksandar D. Kostic, Susan Bullman, Caitlin A. Brennan, Wendy S. Garrett, Curtis Huttenhower, Matthew Meyerson, Charles S. Fuchs, and Shuji Ogino performed data analysis and interpretation. Kosuke Mima, Yin Cao, Reiko Nishihara, and Shuji Ogino drafted the manuscript. Yin Cao, Andrew T. Chan, Mingyang Song, Marios Giannakis, Caitlin A. Brennan, Hideo Baba, Wendy S. Garrett, Matthew Meyerson, Jeffrey A. Meyerhardt, Edward L. Giovannucci, Charles S. Fuchs, Reiko Nishihara, and Shuji Ogino contributed to editing and critical revision for important intellectual contents. All authors approved the final draft submitted.
Financial support: This work was supported by US National Institutes of Health (NIH) grants (P01 CA87969 to M.J. Stampfer; UM1 CA186107 to M.J. Stampfer; P01 CA55075 to W.C. Willett; UM1 CA167552 to W.C. Willett; P50 CA127003 to C.S.F.; R01 CA137178 to A.T.C.; R01 CA151993 to S.O.; R35 CA197735 to S.O.; and K07 CA190673 to R.N.); and by grants from the Project P Fund, The Friends of the Dana-Farber Cancer Institute, Bennett Family Fund, and the Entertainment Industry Foundation through National Colorectal Cancer Research Alliance. K.M. is supported by a grant from Program for Advancing Strategic International Networks to Accelerate the Circulation of Talented Researchers from Japan Society for the Promotion of Science. The content is solely the responsibility of the authors and does not necessarily represent the official views of NIH. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Potential competing interests: Chan previously served as a consultant for Bayer Healthcare, Millennium Pharmaceuticals, Pozen, and Pfizer. This study was not funded by Bayer Healthcare, Millennium Pharmaceuticals, Pozen, or Pfizer. This is redundant, and we will keep the last sentence. Meyerson has applied for a patent on Fusobacterium in colorectal cancer diagnosis and had ownership interest in and was a consultant and advisory board member for Foundation Medicine. He also receives research support from Bayer. The other authors declare no competing financial interest.
Supplementary Material
References
- Louis P, Hold GL, Flint HJ. The gut microbiota, bacterial metabolites and colorectal cancer. Nat Rev Microbiol 2014; 12: 661–672. [DOI] [PubMed] [Google Scholar]
- Keku TO, Dulal S, Deveaux A et al. The gastrointestinal microbiota and colorectal cancer. Am J Physiol Gastrointest Liver Physiol 2015; 308: G351–G363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ericsson AC, Akter S, Hanson MM et al. Differential susceptibility to colorectal cancer due to naturally occurring gut microbiota. Oncotarget 2015; 6: 33689–33704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dejea CM, Wick EC, Hechenbleikner EM et al. Microbiota organization is a distinct feature of proximal colorectal cancers. Proc Natl Acad Sci USA 2014; 111: 18321–18326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belcheva A, Irrazabal T, Robertson SJ et al. Gut microbial metabolism drives transformation of MSH2-deficient colon epithelial cells. Cell 2014; 158: 288–299. [DOI] [PubMed] [Google Scholar]
- Dulal S, Keku TO. Gut microbiome and colorectal adenomas. Cancer J 2014; 20: 225–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sears CL, Garrett WS. Microbes, microbiota, and colon cancer. Cell Host Microbe 2014; 15: 317–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gur C, Ibrahim Y, Isaacson B et al. Binding of the Fap2 protein of Fusobacterium nucleatum to human inhibitory receptor TIGIT protects tumors from immune cell attack. Immunity 2015; 42: 344–355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kostic AD, Chun E, Robertson L et al. Fusobacterium nucleatum potentiates intestinal tumorigenesis and modulates the tumor-immune microenvironment. Cell Host Microbe 2013; 14: 207–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubinstein MR, Wang X, Liu W et al. Fusobacterium nucleatum promotes colorectal carcinogenesis by modulating E-cadherin/beta-catenin signaling via its FadA adhesin. Cell Host Microbe 2013; 14: 195–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kostic AD, Gevers D, Pedamallu CS et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res 2012; 22: 292–298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castellarin M, Warren RL, Freeman JD et al. Fusobacterium nucleatum infection is prevalent in human colorectal carcinoma. Genome Res 2012; 22: 299–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCoy AN, Araujo-Perez F, Azcarate-Peril A et al. Fusobacterium is associated with colorectal adenomas. PLoS One 2013; 8: e53653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tahara T, Yamamoto E, Suzuki H et al. Fusobacterium in colonic flora and molecular features of colorectal carcinoma. Cancer Res 2014; 74: 1311–1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito M, Kanno S, Nosho K et al. Association of Fusobacterium nucleatum with clinical and molecular features in colorectal serrated pathway. Int J Cancer 2015; 137: 1258–1268. [DOI] [PubMed] [Google Scholar]
- Mima K, Sukawa Y, Nishihara R et al. Fusobacterium nucleatum and T cells in colorectal carcinoma. JAMA Oncol 2015; 1: 653–661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosho K, Sukawa Y, Adachi Y et al. Association of Fusobacterium nucleatum with immunity and molecular alterations in colorectal cancer. World J Gastroenterol 2016; 22: 557–566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li YY, Ge QX, Cao J et al. Association of Fusobacterium nucleatum infection with colorectal cancer in Chinese patients. World J Gastroenterol 2016; 22: 3227–3233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iacopetta B. Are there two sides to colorectal cancer? Int J Cancer 2002; 101: 403–408. [DOI] [PubMed] [Google Scholar]
- Gervaz P, Bucher P, Morel P. Two colons-two cancers: paradigm shift and clinical implications. J Surg Oncol 2004; 88: 261–266. [DOI] [PubMed] [Google Scholar]
- Carethers JM. One colon lumen but two organs. Gastroenterology 2011; 141: 411–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamauchi M, Morikawa T, Kuchiba A et al. Assessment of colorectal cancer molecular features along bowel subsites challenges the conception of distinct dichotomy of proximal versus distal colorectum. Gut 2012; 61: 847–854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phipps AI, Buchanan DD, Makar KW et al. BRAF mutation status and survival after colorectal cancer diagnosis according to patient and tumor characteristics. Cancer Epidemiol Biomarkers Prev 2012; 21: 1792–1798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosty C, Young JP, Walsh MD et al. PIK3CA activating mutation in colorectal carcinoma: associations with molecular features and survival. PLoS One 2013; 8: e65479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phipps AI, Limburg PJ, Baron JA et al. Association between molecular subtypes of colorectal cancer and patient survival. Gastroenterology 2015; 148: 77–87 e72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cummings JH, Pomare EW, Branch WJ et al. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 1987; 28: 1221–1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi H, Takahashi R, Nishi T et al. Molecular analysis of jejunal, ileal, caecal and recto-sigmoidal human colonic microbiota using 16S rRNA gene libraries and terminal restriction fragment length polymorphism. J Med Microbiol 2005; 54: 1093–1101. [DOI] [PubMed] [Google Scholar]
- Li X, LeBlanc J, Truong A et al. A metaproteomic approach to study human-microbial ecosystems at the mucosal luminal interface. PLoS One 2011; 6: e26542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mowat AM, Agace WW. Regional specialization within the intestinal immune system. Nat Rev Immunol 2014; 14: 667–685. [DOI] [PubMed] [Google Scholar]
- Liao X, Lochhead P, Nishihara R et al. Aspirin use, tumor PIK3CA mutation, and colorectal-cancer survival. N Engl J Med 2012; 367: 1596–1606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishihara R, Wu K, Lochhead P et al. Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med 2013; 369: 1095–1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mima K, Nishihara R, Qian ZR et al. Fusobacterium nucleatum in colorectal carcinoma tissue and patient prognosis. Gut 2016. in press. [DOI] [PMC free article] [PubMed]
- Ogino S, Nosho K, Kirkner GJ et al. CpG island methylator phenotype, microsatellite instability, BRAF mutation and clinical outcome in colon cancer. Gut 2009; 58: 90–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irahara N, Nosho K, Baba Y et al. Precision of pyrosequencing assay to measure LINE-1 methylation in colon cancer, normal colonic mucosa, and peripheral blood cells. J Mol Diagn 2010; 12: 177–183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogino S, Kawasaki T, Brahmandam M et al. Precision and performance characteristics of bisulfite conversion and real-time PCR (MethyLight) for quantitative DNA methylation analysis. J Mol Diagn 2006; 8: 209–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogino S, Kawasaki T, Kirkner GJ et al. Evaluation of markers for CpG island methylator phenotype (CIMP) in colorectal cancer by a large population-based sample. J Mol Diagn 2007; 9: 305–314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogino S, Kawasaki T, Brahmandam M et al. Sensitive sequencing method for KRAS mutation detection by Pyrosequencing. J Mol Diagn 2005; 7: 413–421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imamura Y, Lochhead P, Yamauchi M et al. Analyses of clinicopathological, molecular, and prognostic associations of KRAS codon 61 and codon 146 mutations in colorectal cancer: cohort study and literature review. Mol Cancer 2014; 13: 135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosho K, Kawasaki T, Ohnishi M et al. PIK3CA mutation in colorectal cancer: relationship with genetic and epigenetic alterations. Neoplasia 2008; 10: 534–541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao X, Morikawa T, Lochhead P et al. Prognostic role of PIK3CA mutation in colorectal cancer: cohort study and literature review. Clin Cancer Res 2012; 18: 2257–2268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khashab MA, Pickhardt PJ, Kim DH et al. Colorectal anatomy in adults at computed tomography colonography: normal distribution and the effect of age, sex, and body mass index. Endoscopy 2009; 41: 674–678. [DOI] [PubMed] [Google Scholar]
- Yamauchi M, Lochhead P, Morikawa T et al. Colorectal cancer: a tale of two sides or a continuum? Gut 2012; 61: 794–797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jess P, Hansen IO, Gamborg M et al. A nationwide Danish cohort study challenging the categorisation into right-sided and left-sided colon cancer. BMJ Open 2013; 3: e002608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kudryavtseva AV, Lipatova AV, Zaretsky AR et al. Important molecular genetic markers of colorectal cancer. Oncotarget 2016. in press. [DOI] [PMC free article] [PubMed]
- Colussi D, Brandi G, Bazzoli F et al. Molecular pathways involved in colorectal cancer: implications for disease behavior and prevention. Int J Mol Sci 2013; 14: 16365–16385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin JH, Giovannucci E. Environmental exposure and tumor heterogeneity in colorectal cancer risk and outcomes. Curr Colorectal Cancer Rep 2014; 10: 94–104. [Google Scholar]
- Kocarnik JM, Shiovitz S, Phipps AI. Molecular phenotypes of colorectal cancer and potential clinical applications. Gastroenterol Rep (Oxf) 2015; 3: 269–276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuipers EJ, Grady WM, Lieberman D et al. Colorectal cancer. Nat Rev Dis Primers 2015; 1: 15065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zong L, Abe M, Ji J et al. Tracking the correlation between CpG island methylator phenotype and other molecular features and clinicopathological features in human colorectal cancers: a systematic review and meta-analysis. Clin Transl Gastroenterol 2016; 7: e151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellatt DF, Stevens JR, Wolff RK et al. Expression profiles of miRNA subsets distinguish human colorectal carcinoma and normal colonic mucosa. Clin Transl Gastroenterol 2016; 7: e152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kedrin D, Gala MK. Genetics of the serrated pathway to colorectal cancer. Clin Transl Gastroenterol 2015; 6: e84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lupton JR. Microbial degradation products influence colon cancer risk: the butyrate controversy. J Nutr 2004; 134: 479–482. [DOI] [PubMed] [Google Scholar]
- Singh N, Gurav A, Sivaprakasam S et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity 2014; 40: 128–139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donohoe DR, Collins LB, Wali A et al. The Warburg effect dictates the mechanism of butyrate-mediated histone acetylation and cell proliferation. Mol Cell 2012; 48: 612–626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosty C, Young JP, Walsh MD et al. Colorectal carcinomas with KRAS mutation are associated with distinctive morphological and molecular features. Mod Pathol 2013; 26: 825–834. [DOI] [PubMed] [Google Scholar]
- Phipps AI, Chan AT, Ogino S. Anatomic subsite of primary colorectal cancer and subsequent risk and distribution of second cancers. Cancer 2013; 119: 3140–3147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong RJ. Marked variations in proximal colon cancer survival by race/ethnicity within the United States. J Clin Gastroenterol 2010; 44: 625–630. [DOI] [PubMed] [Google Scholar]
- Cheng L, Eng C, Nieman LZ et al. Trends in colorectal cancer incidence by anatomic site and disease stage in the United States from 1976 to 2005. Am J Clin Oncol 2011; 34: 573–580. [DOI] [PubMed] [Google Scholar]
- Samadder NJ, Curtin K, Tuohy TM et al. Characteristics of missed or interval colorectal cancer and patient survival: a population-based study. Gastroenterology 2014; 146: 950–960. [DOI] [PubMed] [Google Scholar]
- Robertson DJ, Lieberman DA, Winawer SJ et al. Colorectal cancers soon after colonoscopy: a pooled multicohort analysis. Gut 2014; 63: 949–956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biedermann L, Zeitz J, Mwinyi J et al. Smoking cessation induces profound changes in the composition of the intestinal microbiota in humans. PLoS One 2013; 8: e59260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Keefe SJ, Li JV, Lahti L et al. Fat, fibre and cancer risk in African Americans and rural Africans. Nat Commun 2015; 6: 6342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zitvogel L, Galluzzi L, Viaud S et al. Cancer and the gut microbiota: an unexpected link. Sci Transl Med 2015; 7: 271ps271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogino S, Chan AT, Fuchs CS et al. Molecular pathological epidemiology of colorectal neoplasia: an emerging transdisciplinary and interdisciplinary field. Gut 2011; 60: 397–411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogino S, Nishihara R, VanderWeele TJ et al. Review article: The role of molecular pathological epidemiology in the study of neoplastic and non-neoplastic diseases in the era of precision medicine. Epidemiology 2016; 27: 602–611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arthur JC, Perez-Chanona E, Muhlbauer M et al. Intestinal inflammation targets cancer-inducing activity of the microbiota. Science 2012; 338: 120–123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu S, Rhee KJ, Albesiano E et al. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17T cell responses. Nat Med 2009; 15: 1016–1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.