ABSTRACT
Introduction:
The frequency of gastric neuroendocrine tumors is increasing. Reasons are the popularization of endoscopy and its technical refinements. Despite this, they are still poorly understood and have complex management.
Aim:
Update the knowledge on gastric neuroendocrine tumor and expose the future perspectives on the diagnosis and treatment of this disease.
Method:
Literature review using the following databases: Medline/PubMed, Cochrane Library and SciELO. Search terms were: gastric carcinoid, gastric neuroendocrine tumor, treatment. From the selected articles, 38 were included in this review.
Results:
Gastric neuroendocrine tumors are classified in four clinical types. Correct identification of the clinical type and histological grade is fundamental, since treatment varies accordingly and defines survival.
Conclusion:
Gastric neuroendocrine tumors comprise different subtypes with distinct management and prognosis. Correct identification allows for a tailored therapy. Further studies will clarify the diseases biology and improve its treatment.
HEADINGS: Gastric carcinoid; Gastric neuroendocrine tumor; Treatment, review
RESUMO
Introdução:
A frequência de tumores neuroendócrinos gástricos está aumentando. As razões são a popularização da endoscopia e seus refinamentos técnicos. Apesar disso, os gástricos ainda são pouco compreendidos e têm manejo complexo.
Objetivo:
Atualizar os conhecimentos nos tumores neuroendócrinos gástricos e expor as perspectivas futuras no diagnóstico e tratamento.
Método:
Revisão da literatura utilizando as seguintes bases de dados: Medline/PubMed, Cochrane Library e SciELO. Os descritores da busca foram: carcinóide gástrico, tumor neuroendócrino gástrico, tratamento. Dos artigos selecionados, 38 foram incluídos nesta revisão.
Resultados:
Tumores neuroendócrinos gástricos são classificados em quatro tipos clínicos. A identificação correta do tipo clínico e grau histológico é fundamental, pois a conduta é variável e define a sobrevida.
Conclusão:
Tumor neuroendócrino gástrico possui diferentes subtipos com tratamento e prognóstico distintos. A identificação correta destes e seu entendimento permite o tratamento individualizado. Estudos futuros ajudarão a esclarecer a biologia desta doença e melhorar o tratamento.
INTRODUCTION
Gastric neuroendocrine tumors (gNETs) are neoplasms derived from the enterochromaffin-like cells (ECL cells) of the gastric mucosa. They are rare lesions with an indolent behavior and neuroendocrine differentiation. Although uncommon, their diagnosis is increasing, due to the widespread use of upper digestive endoscopy and the technical refinement of endoscopists 4 .
The ECL cells are avid for silver salts and play a fundamental role in the regulation of acid secretion. Following food intake, the G cells of the antrum secrete gastrin that stimulates the ECL cells and the histamine-producing parietal cells to secrete hydrochloric acid (HCL). Negative feed-back comes from the D cells, which are stimulated by the HCL and secrete somatostatin who acts reducing the secretion of gastrin 4 .
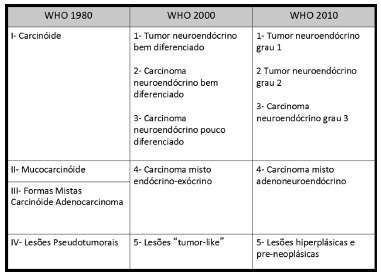
It is essential to understand these mechanisms to classify gastric gNETs in four clinical subtypes, with distinct treatment management and prognosis 4 , 12 , 27 . It is also important to emphasize that this classification is different from the three histological grades proposed by the World Health Organization (WHO). Furthermore, the WHO terminology for gNETs underwent changes in recent years, which amplified the difficulty to understand this complex disease (Figure 1).
FIGURE 1. Evolution in gNETs terminology.
METHOD
Literature review using the following databases: Medline/PubMed, Cochrane Library and SciELO. Search terms were: gastric carcinoid, gastric neuroendocrine tumor, treatment. Articles in English and Portuguese were considered.
RESULTS
Classification and diagnosis
Type I
Type I lesions correspond to the majority of gNETs found in the stomach (70-80%) and they are associated with autoimmune chronic atrophic gastritis. The patient has anti-parietal cell or anti-intrinsic factor antibodies, leading to the destruction of the gastric parietal cell, reducing the level of HCL (achlorhydria), consequently increasing the gastrin production by G cells (hypergastrinemia) 12 , 21 . This hormone excess promotes ECL cells hyperplasia, favoring the appearance of multiple small lesions, usually with little aggressive behavior and good prognosis 21 .
A decrease in the intrinsic factor with reduction on vitamin B12 absorption also occurs leading to macrocytic anemia (pernicious or megaloblastic) 15 .
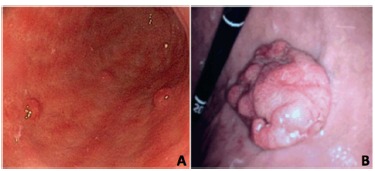
Diagnosis of type I is made by upper gastrointestinal (GI) endoscopy with biopsy. Endoscopic findings consist of pale, yellowish and transparent blood vessels of the antral mucosa, contrasting with the smooth and reddish mucosa of normal areas. Neuroendocrine tumors are visualized as small, reddish polyps and often being multiple (Figure 2A).
FIGURE 2. Endoscopic images: A) showing chronic atrophic pangastritis with three low grade gNETs; B) with solitary gNET type III.
Histological examination shows atrophy of the mucosa cells, absence of parietal cells and neuroendocrine cell hyperplasia. It also confirms the diagnosis of NET. Increased serum gastrin and low serum vitamin B12 are usually observed. Anti-parietal cell and anti-intrinsic factor antibodies may also be present 12 , 15 , 18 . Finally, the gastric acidity dosage reveals high pH (pH≥7) 22 , 32 .
Type II
Type II lesions are caused by gastrinomas (gastrin-producing tumors), also known as Zollinger-Ellison syndrome. In most cases, the patient has multiple endocrine neoplasia type I (MEN-1) and should be investigated with serum sequencing for MEN1 gene 16 .
The frequency of type II gNETs is around 7% and the lesions are usually small and multiple. The metastatic potential is also low, although higher than in type I 4 , 12 .
For diagnostic confirmation, upper GI endoscopy with gastric biopsy reveals normal or hypertrophic gastric mucosa 29 . Hypergastrinemia and gastric pH<2 (hyperchlorhydria) are observed. Serial measurement of gastrin levels following intravenous administration of secretin can also be performed revealing an increase in gastrin levels for patients with gastrinoma, whereas they decrease in healthy individuals 3 .
After confirming the diagnosis, research should continue aiming to localize the gastrinoma and, if possible, remove it surgically. Most of these lesions are located in the triangle of gastrinomas determined by the junction of the cystic duct with the common hepatic duct, the transition from second to third duodenal portions and the pancreatic cervix 33 . Computed tomography (CT) scan, magnetic resonance imaging (MRI), endoscopic ultrasound, scintigraphy with octreotide, selective angiography, positron emission tomography and intraoperative ultrasonography are usefull methods that help finding the lesion.
Type III
Type III gNETs consist of a sporadic lesion and has the greatest potential to generate metastasis. The survival of these patients is also worse (75-80% at five years compared to 90-95% for type I) 4 , 12 . Generally, the lesion is unique and greater than 1 cm, with normal gastrinemia.
Diagnosis is made by upper GI endoscopy with biopsy, observing a single lesion in normal gastric mucosa (Figure 2B). Although rare, carcinoid syndrome (due to the presence of liver metastasis) can be the initial manifestation.
Type IV
It is worth mentioning that three recent reports suggest a fourth type of gNET. It consists of multiple small lesions and the histological examination reveals hypertrophy and hyperplasia of parietal cells with vacuolated cytoplasm. A structural abnormality prevents the HCL, produced by these cells, from being secreted. Consequently, achlorhydria, hypergastrinemia and hyperplasia of neuroendocrine cells occur 1 , 23 , 27 .
Immunohistochemistry
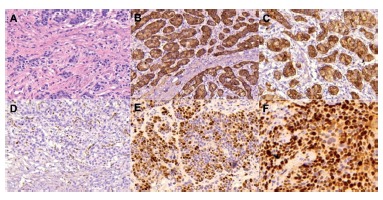
Immunohistochemical analysis is essential in NETs. It allows diagnostic confirmation and permits classifying the lesion according to the histological grades defined by the WHO (Figure 3) 8 , 30 . For diagnostic confirmation chromogranin A and synaptophysin are necessary, while for prognostic definition the proliferative index Ki-67 and the number of mitoses per high magnification field are required (Figure 4) 8 , 30 . Other markers, such as p53, have being studied, and also relates to prognosis and risk of metastasis 31 .
FIGURE 3. Classification according to histological grades.
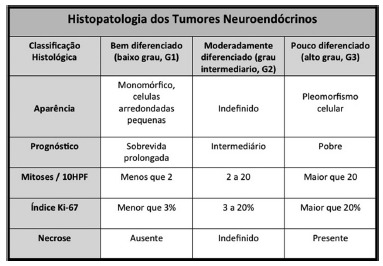
FIGURE 4. Histological sections of well-differentiated gNET (x 200): A) hematoxylin-eosin; B) positivity for chromogranin A; C) positivity for synaptophysin; D) Ki-67 nuclear proliferative index <2%; E) Ki-67 of 50-60%; F) Ki-67 of 70-80%.
Staging
CT scan of the abdomen is recommended for type I and II gNETs larger than 2 cm and for all type III lesions. MRI of the abdomen, octreotide scintigraphy and PET-CT may be usefull in specific cases 34 .
Treatment
The treatment of gNETs depends on the clinical type, disease extent, the differentiation of the lesion and the presence or absence of poor prognostic factors (Figure 5). Accordingly to the WHO, these neoplasms are classified into three histologic degrees with distinct prognosis (Figure 3) 30 .
FIGURE 5. Treatment algorithm.
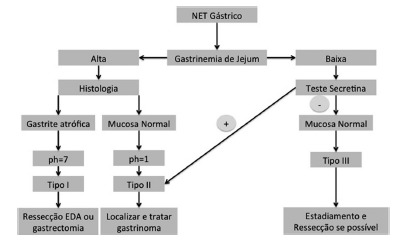
Poor prognosis factors are: lesion ≥2 cm; deep submucosa invasion or beyond (at least 24% are metastatic); Ki-67 ≥3%; vascular invasion; low degree of structural differentiation; presence of atypia and/or necrosis.
Carcinoid crisis should also be prevented before and after any tumor manipulation or anesthesia. This may be accomplished by the administration of intravenous or subcutaneous octreotide 35 9 .
Type I
As most of these lesions are small, well-differentiated and with excellent prognosis, treatment usually consists in the serial endoscopic resection of these lesions 12 . Supplementation of vitamin B12 is also recommended.
Surgical treatment is necessary only when endoscopic resection is not feasible or when poor prognostic factors are present. While the surgical indication is obvious when deep invasion is observed, for those cases with lymph node metastasis or for lesions not suitable for endoscopic resection, there is no evidence that supports or not surgery when there is only necrosis, vascular invasion or an elevated Ki-67. A recent article validated the WHO classification, demonstrating the presence of a lymph node metastasis in a patient with a small and superficial type I gNET whose only poor prognostic factor was a Ki-67 of 7% (G2 according to the WHO grading) 19 . This also highlights the need for diligent analysis of all resected lymph node. Our group recommends Carnoy`s solution as specimen fixative to improve lymph node detection 9 , 28 .
It is also unclear when surgery should be indicated for patients with frequent recurrences or when there is a high number of lesions. At this time there is no evidence in the literature that allows a strong recommendation, since there is no consensus of what is a “frequent recurrence” or a “high number” of lesions. Thus, management of these cases should be tailored and discussed with the patient.
The best operation for type I gNETs is also controversial 37 . Antrectomy has been proposed to remove the gastrin-producing G cells; however, failure may occur due to improper removal of these cells or because the ECL cells became autonomous. For these reasons subtotal or total gastrectomy are more suitable options. Subtotal gastrectomy allows adequate removal of G cells, while total gastrectomy is reserved for those cases with substantial disease in the gastric fundus 3 , 7 , 12 . Concerning lymphadenectomy, it should be performed when there is any evidence of extra gastric disease or when poor prognosis factors are present. Its extension is not stablished in the literature, option for D1, D1+ or D2 should be performed on an individual basis. Minimally invasive procedures are adequate for these patients 2 .
It is worth mentioning the clinical treatment of type I gNETs, although this will hardly be an effective option in the long term. Some authors used somatostatin analogues (octreotide) to decrease gastrinemia in small groups of patients. However, after discontinuation of the treatment serum gastrin rose again in a one year follow-up, although no new lesions were observed in the short term 13 , 17 . Therefore, this treatment should be reserved for those patients unfit for surgical resection.
Type II
Treatment of type II gNETs consists in localizing and resecting the gastrinoma. As for gastric lesions, unless there is some factor of poor prognosis, endoscopic resection is enough.
Type III
These lesions should be managed aggressively with total or subtotal gastrectomy (depending on location) associated with lymphadenectomy.
If there is resectable metastatic disease, it should also be treated. For unresectable liver disease, local therapies such as arterial embolization or radioablation have a success rate of 50% 14 , 20 . If there is extrahepatic metastasis or recurrent symptomatic disease, systemic therapy with cytotoxic chemotherapy (streptozocin combined with 5-fluorouracil or cyclophosphamide, doxorubicin mono drug or with 5-fluorouracil, dacarbazine or temozolamide, oxaliplatin with capecitabine or 5-flurouracil with leucovorin) or molecular targeted agents (bevacizumab, sorafenib, sunitinib, pazopanib and everolimus) can be introduced. The goal is to keep the disease stable, with a small gain in progression-free survival, since the response rate is very low and there is no evidence of gain in overall survival. Preferably, these patients should be included in randomized clinical studies 24 .
Carcinoid syndrome
This is a rare event in gNETs and its clinical manifestation is atypical consisting exclusively of redness due to histamine production 26 . Symptoms control is achieved with somatostatin analogs (octreotide or lanreotide) and interferon alfa in low doses for refractory cases 10 , 26 .
Follow up
Disease evolution is quite heterogeneous, with a median survival ranging from 13 months to more than 10 years 36 .
The recommendation of the National Comprehensive Cancer Network (NCCN) for follow-up consists of anamnesis, physical examination, upper GI endoscopy, abdominal CT scan or MRI and serum chromogranin A, every six months for 1-2 years, annually for four more years and then every two years until 10 years after surgery 24 . Dosage of urinary 5-hydroxyindoleacetic acid is not recommended since gNETs do not produce serotonin 26 . Types I and II lesions <2 cm and without poor prognostic factors may be followed only with anamnesis, physical examination and upper GI endoscopy every 6-12 months 24 .
Serum chromogranin A is used as a prognostic factor and response marker for chemotherapy 3 . However, it should be analyzed with caution due to its low specificity, for instance: somatostatin analogs and proton pump inhibitors may alter it 6 .
Trends
The presence of somatostatin receptors in NETs have been used as starting point for the development of new diagnostic tests and therapeutic methods. A cheaper and faster octreotide scintigraphy has been obtained after labeling it with technetium instead of indium 25 . Another recent innovation is the use of gallium marked octreotide, which can be captured by positron emission tomography scans (PET and PET-CT). Recent data showed undoubted superiority of this method over the octreotide scintigraphy and probably it will become the new standard in detecting NETs 11 , 26 .
Somatostatin analogues have also been utilized as carriers of radioactive molecules. This allows specific treatment of NET cells expressing somatostatin receptors, minimizing side effects and maximizing results. Currently, this therapy is only available in few specialized centers 5 .
New serum markers that help the diagnosis are also being studied. Serum enolase which is most often released by aggressive undifferentiated tumors is an example. From this knowledge more specific follow-up tests for each histological tumor type may be achieved 24 .
CONCLUSIONS
Gastric NETs consist of a complex disease that includes different subtypes with distinct management and prognosis. Correct identification of the clinical type and histological grade allows for a tailored management. Further studies will clarify the diseases biology and improve its treatment.
Footnotes
Financial source: none
REFERENCES
- 1.Abraham SC, Carney JA, Ooi A, Choti MA, Argani P. Achlorhydria, parietal cell hyperplasia, and multiple gastric carcinoids: a new disorder. Am J Surg Pathol. 2005;29:969–975. doi: 10.1097/01.pas.0000163363.86099.9f. [DOI] [PubMed] [Google Scholar]
- 2.Barchi LC, Jacob CE, Bresciani CJ, et al. Minimally invasive surgery for gastric cancer: time to change the paradigm. Arq Bras Cir Dig. 2016;29(2):117–120. doi: 10.1590/0102-6720201600020013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Borch K, Ahrén B, Ahlman H, Falkmer S, Granérus G, Grimelius L. Gastric carcinoids biologic behavior and prognosis after differentiated treatment in relation to type. Ann Surg. 2005;242(1):64–73. doi: 10.1097/01.sla.0000167862.52309.7d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Burkitt MD, Pritchard DM. Review article: pathogenesis and management of gastric carcinoid tumours. Aliment Pharmacol Ther. 24:1305–1320. doi: 10.1111/j.1365-2036.2006.03130.x. [DOI] [PubMed] [Google Scholar]
- 5.Bushnell DL, Jr, O'Dorisio TM, O'Dorisio MS. 90Y-edotreotide for metastatic carcinoid refractory to octreotide. J Clin Oncol. 2010;28(10):1652–1652. doi: 10.1200/JCO.2009.22.8585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Campana D, Nori F, Piscitelli L. Chromogranin A is it a useful marker of neuroendocrine tumors? J Clin Oncol. 2007;25(15):1967–1967. doi: 10.1200/JCO.2006.10.1535. [DOI] [PubMed] [Google Scholar]
- 7.Dakin GF, Warner RR, Pomp A, Salky B, Inabnet WB. Presentation, treatment, and outcome of type 1 gastric carcinoid tumors. J Surg Oncol. 2006;93(5):368–372. doi: 10.1002/jso.20468. [DOI] [PubMed] [Google Scholar]
- 8.Delle Fave G, Kwekkeboom DJ, Van Cutsem E. ENETS Consensus Guidelines for the management of patients with gastroduodenal neoplasms. Neuroendocrinology. 2012;95(2):74–87. doi: 10.1159/000335595. [DOI] [PubMed] [Google Scholar]
- 9.Dias AR, Pereira MA, Mello ES, Zilberstein B, Cecconello I, Ribeiro U., Junior Carnoy's solution increases the number of examined lymph nodes following gastrectomy for adenocarcinoma a randomized trial. Gastric Cancer. 2016;19(1):136–142. doi: 10.1007/s10120-014-0443-2. [DOI] [PubMed] [Google Scholar]
- 10.Frank M, Klose KJ, Wied M, Ishaque N, Schade-Brittinger C, Arnold R. Combination therapy with octreotide and alpha-interferon effect on tumor growth in metastatic endocrine gastroenteropancreatic tumors. Am J Gastroenterol. 1999;94(5):1381–1381. doi: 10.1111/j.1572-0241.1999.01090.x. [DOI] [PubMed] [Google Scholar]
- 11.Frilling A, Akerström G, Falconi M. Neuroendocrine tumor disease an evolving landscape. Endocr Relat Cancer. 2012;19(5):R163–R185. doi: 10.1530/ERC-12-0024. [DOI] [PubMed] [Google Scholar]
- 12.Gladdy RA, Strong VE, Coit D. Defining surgical indications for type I gastric carcinoid tumor. Ann Surg Oncol. 2009;16(11):3154–3160. doi: 10.1245/s10434-009-0687-y. [DOI] [PubMed] [Google Scholar]
- 13.Grozinsky-Glasberg S, Kaltsas G, Gur C. Long-acting somatostatin analogues are an effective treatment for type 1 gastric carcinoid tumours. Eur J Endocrinol. 2008;159(4):475–482. doi: 10.1530/EJE-08-0420. [DOI] [PubMed] [Google Scholar]
- 14.Gupta S, Johnson MM, Murthy R. Hepatic arterial embolization and chemoembolization for the treatment of patients with metastatic neuroendocrine tumors variables affecting response rates and survival. Cancer. 2005;104(8):1590–1602. doi: 10.1002/cncr.21389. [DOI] [PubMed] [Google Scholar]
- 15.Hung OY, Maithel SK, Willingham FF, Farris 3rd AB, Kauh JS. Hypergastrinemia, type 1 gastric carcinoid tumors diagnosis and management., J Clin. Oncol. 2011;29(25):e713–e715. doi: 10.1200/JCO.2011.35.3235. [DOI] [PubMed] [Google Scholar]
- 16.Jordan PH, Jr, Barroso A, Sweeney J. Gastric carcinoids in patients with hypergastrinemia. J Am Coll Surg. 2004;199(4):552–555. doi: 10.1016/j.jamcollsurg.2004.06.019. [DOI] [PubMed] [Google Scholar]
- 17.Khuroo MS, Khuroo MS, Khuroo NS. Treatment of type I gastric neuroendocrine tumors with somatostatin analogs. J Gastroenterol Hepatol. 2010;25(3):548–554. doi: 10.1111/j.1440-1746.2009.06131.x. [DOI] [PubMed] [Google Scholar]
- 18.Liu Y, Uemura N, Xiao SD, Tytgat GN, Kate FJ. Agreement between endoscopic and histological gastric atrophy scores. J Gastroenterol. 2005;40(2):123–127. doi: 10.1007/s00535-004-1511-x. [DOI] [PubMed] [Google Scholar]
- 19.Lupinacci RM, Dias AR, Mello ES, Kondo A. Minute type I gastric carcinoid with regional lymph node metastasis. Int J Surg Pathol. 2013;21(2):169–172. doi: 10.1177/1066896912457201. [DOI] [PubMed] [Google Scholar]
- 20.Mazzaglia PJ, Berber E, Siperstein AE. Radiofrequency thermal ablation of metastatic neuroendocrine tumors in the liver. Curr Treat Options Oncol. 2007;8:322–322. doi: 10.1007/s11864-007-0038-9. [DOI] [PubMed] [Google Scholar]
- 21.Modlin IM, Kidd M, Latich I, Zikusoka MN, Shapiro MD. Current status of gastrointestinal carcinoids. Gastroenterology. 2005;128(6):1717–1751. doi: 10.1053/j.gastro.2005.03.038. [DOI] [PubMed] [Google Scholar]
- 22.Modlin IM, Oberg K, Chung DC. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008;9(1):61–72. doi: 10.1016/S1470-2045(07)70410-2. [DOI] [PubMed] [Google Scholar]
- 23.Nakata K, Aishima S, Ichimiya H. Unusual multiple gastric carcinoids with hypergastrinemia report of a case. Surg Today. 2010;40(3):267–271. doi: 10.1007/s00595-009-4032-7. [DOI] [PubMed] [Google Scholar]
- 24.National Comprehensive Cancer Network . Neuroendocrine tumors NCCN guidelines. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp [Google Scholar]
- 25.Nock B, Maina T. Tetraamine-coupled peptides and resulting (99m)Tc-radioligands an effective route for receptor-targeted diagnostic imaging of human tumors. Curr Top Med Chem. 2012;12(23):2655–2667. doi: 10.2174/1568026611212230003. [DOI] [PubMed] [Google Scholar]
- 26.Öberg K, Knigge U, Kwekkeboom D, Perren A. ESMO Guidelines Working Group Neuroendocrine gastro-entero-pancreatic tumors ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23(7):vii124–vii130. doi: 10.1093/annonc/mds295. [DOI] [PubMed] [Google Scholar]
- 27.Ooi A, Ota M, Katsuda S, Nakanishi I, Sugawara H, Takahashi I. An unusual case of multiple gastric carcinoids associated with diffuse endocrine cell hyperplasia and parietal cell hypertrophy. Endocr Pathol. 1995;6:229–237. doi: 10.1007/BF02739887. [DOI] [PubMed] [Google Scholar]
- 28.Pereira MA, Dias AR, Faraj SF. Carnoy's solution is an adequate tissue fixative for routine surgical pathology, preserving cell morphology and molecular integrity. Histopathology. 2015;66(3):388–397. doi: 10.1111/his.12532. [DOI] [PubMed] [Google Scholar]
- 29.Pritchard DM. Zollinger-Ellison syndrome still a diagnostic challenge in the 21st century? Gastroenterology. 2011;140(5):1380–1383. doi: 10.1053/j.gastro.2011.03.026. [DOI] [PubMed] [Google Scholar]
- 30.Rindi G, Arnold R, Bosman FT, et al. Bosman FT, Carneiro F, Hruban RH, Theise ND, et al. WHO classification of tumors of the digestive system. Lyon: 2010. Nomenclature and classification of neuroendocrine neoplasms of the digestive system; pp. 13–14. [Google Scholar]
- 31.Safatle-Ribeiro AV, Ribeiro U, Jr, Corbett CE. Prognostic value of immunohistochemistry in gastric neuroendocrine (carcinoid) tumors. Eur J Gastroenterol Hepatol. 2007;19(1):21–28. doi: 10.1097/01.meg.0000250582.30737.bd. [DOI] [PubMed] [Google Scholar]
- 32.Soga J. Early-stage carcinoids of the gastrointestinal tract an analysis of 1914 reported cases. Cancer. 2005;103(8):1587–1595. doi: 10.1002/cncr.20939. [DOI] [PubMed] [Google Scholar]
- 33.Stabile BE, Morrow DJ, Passaro E., Jr The gastrinoma triangle operative implications. Am J Surg. 1984;147(1):25–31. doi: 10.1016/0002-9610(84)90029-1. [DOI] [PubMed] [Google Scholar]
- 34.Sundin A, Vullierme MP, Kaltsas G, Plöckinger U. Mallorca Consensus Conference participants.European Neuroendocrine Tumor Society ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Tumors radiological examinations. Neuroendocrinology. 2009;90(2):167–183. doi: 10.1159/000184855. [DOI] [PubMed] [Google Scholar]
- 35.Woodside KJ, Townsend CM, Jr, Mark Evers B. Current management of gastrointestinal carcinoid tumors. J Gastrointest Surg. 2004;8(6):742–742. doi: 10.1016/j.gassur.2004.04.010. [DOI] [PubMed] [Google Scholar]
- 36.Yao JC, Hassan M, Phan A. One hundred years after "carcinoid" epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26(18):3063–3063. doi: 10.1200/JCO.2007.15.4377. [DOI] [PubMed] [Google Scholar]
- 37.Zilberstein B, Malheiros C, Lourenço LG, et al. Brazilian consensus in gastric cancer: guidelines for gastric cancer in Brazil. Arq Bras Cir Dig. 2013;26(1):2–6. doi: 10.1590/s0102-67202013000100002. [DOI] [PubMed] [Google Scholar]


