Abstract
The phytochrome family of sensory photoreceptors directs adaptational changes in gene expression in response to environmental light signals. Using oligonucleotide microarrays to measure expression profiles in wild-type and phytochrome A (phyA) null-mutant Arabidopsis seedlings, we have shown that 10% of the genes represented on the array are regulated by phyA in response to a continuous far-red light signal. Strikingly, 44% of the genes responding to the signal within 1 h are predicted to encode multiple classes of transcriptional regulators. Together with previous data, this observation suggests that phyA may regulate seedling photomorphogenesis by direct targeting of light signals to the promoters of genes encoding a master set of diverse transcriptional regulators, responsible in turn for orchestrating the expression of multiple downstream target genes in various branches of a phyA-regulated transcriptional network.
The phytochrome (phy) family of red- and far-red-light-absorbing photoreceptors regulates multiple plant growth and developmental responses to informational light signals from the environment (1). Although a considerable number of studies have examined expression patterns of a limited number of individual genes underlying these responses (2–4), a global analysis of photoregulated genes has been lacking. The availability of microarray-based expression profiling technology now permits such an analysis (5) and thus the assembly of an integrated picture of these expression patterns. In addition, when coupled to careful early time-course analysis of defined photoresponse mutants, microarray profiling provides a powerful tool for identifying genes in primary transcriptional networks that regulate expression of the array of downstream gene sets, which in turn implement the programmed morphological changes.
In Arabidopsis, the phy family consists of five members, phyA through phyE (6). Mutant analysis has established that the individual phy family members have differential photosensory and/or physiological functions in controlling plant responses (7, 8). phyA is unique within the family in that it is exclusively responsible for seedling responsiveness to continuous far-red light (FRc) (7, 9). Thus, the deetiolation process that is initiated on exposure of seedlings to FRc (7) provides an ideal opportunity to unambiguously define the linkage between a single photoreceptor species and its target gene ensemble. As such, the phyA system represents an excellent example of the “functional module” concept of biological organization and regulation (10, 11), whereby a specific signal (in this case a photon) is perceived by a single molecular species (here a photosensory receptor) and transduced into a discreet biological response (here the global switch from skotomorphogenic to photomorphogenic development) (9). It has been argued that defining and characterizing such modules are key to deciphering the rules that govern complex biological systems (10, 11). Here we examine the expression profiles of genes under phyA control in Arabidopsis using oligonucleotide microarrays.
Materials and Methods
Plant Growth and Irradiation Conditions.
Wild-type Arabidopsis (ecotype RLD) and phyA-101 seeds were sown on growth medium plates (12) containing 0.9% agar. Seeds were stratified for 5 days at 4°C, exposed to white light for 2 h to induce germination and placed in a growth chamber at 21°C in total darkness for 4 days. Seedlings were then irradiated with FRc (740 nm, 2 μmol m⋅2⋅s⋅1), and tissue was harvested and frozen immediately in liquid nitrogen at 1, 3, 6, 12, 18, and 24 h after the start of irradiation. Dark control samples were harvested at 0 and 24 h.
RNA Isolation, cRNA Synthesis, and Microarray Hybridization.
See Note 1, supplemental material at www.pgec.usda.gov/Quail/phyA.html for a complete description of these procedures.
Data Analysis.
See Note 2, supplemental material at www.pgec.usda.gov/Quail/phyA.html for a complete description of the procedures used.
Results and Discussion
phyA Regulates a Major Subset of Arabidopsis Genes.
To identify genes specifically regulated by phyA, we used high-density oligonucleotide arrays (Affymetrix, Santa Clara, CA) to determine the time course of the changes in mRNA levels in etiolated wild-type and phyA-null mutant Arabidopsis seedlings over a 24-h period after transfer from darkness to FRc. These arrays contain probe sets for about 8,200 different Arabidopsis genes (13) representing about one-third of the total number in the genome (14). RNA samples were extracted from seedlings exposed to FRc, as well as from unirradiated dark-control seedlings at both the beginning (time 0 controls) and the end (24-h controls) of the FRc irradiation period. Biotin-labeled cRNAs representing each seedling RNA sample were hybridized individually to separate microarrays and the hybridization signal intensity for each immobilized gene sequence was determined (see Note 1, supplemental material at www.pgec.usda.gov/Quail/phyA.html for details). A high degree of reproducibility between duplicate, independently isolated RNA samples was observed (see Fig. 5, supplemental material at www.pgec.usda.gov/Quail/phyA.html).
To objectively identify sequences exhibiting significant phyA-mediated changes in abundance in response to FRc, we developed a set of quantitative criteria that were applied individually to the data set at each time point in the time course. Genes were classified as phyA-regulated if at the time point in question they fulfilled both of the following criteria: (i) The expression level in FRc-irradiated wild-type seedlings deviated (positively or negatively) 2-fold or more from that of the wild-type time-zero dark control. (ii) The expression level in the FRc-irradiated wild type deviated statistically (for triplicate seedling samples at the 1-h time point), or 2-fold or more (for the other time points), from that of the FRc-irradiated phyA mutant at that time point. In addition, the full time-course profile for each gene selected by these criteria was inspected visually for internal consistency and continuity of pattern. Genes exhibiting erratic patterns, such as large repetitive oscillations over multiple sequential points on the time-course curve, were eliminated (see Note 2, supplemental material at www.pgec.usda.gov/Quail/phyA.html for details of data analysis). This combination of strictly objective selection criteria and more subjective deselection criteria might be considered somewhat analogous to the hybrid supervised/unsupervised cluster analysis of expression profiles used by Eisen et al. (15).
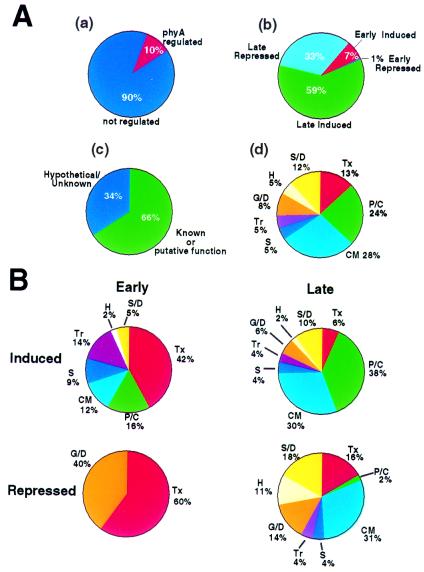
Rigorous application of these criteria resulted in identification of genes that were induced or repressed 2-fold or more in the wild type in response to the FRc light signal, but not in the phyA-minus mutant. On this basis, 10% (812) of the different genes on the array were identified as being regulated by phyA (Table 1, Fig. 1A). These genes are listed in Tables 2–7, supplemental material at www.pgec.usda.gov/Quail/phyA.html. Of these, 67% were induced and 33% were repressed, with 8% exhibiting changes in mRNA abundance within 1 h of transfer to FRc (“early-response” genes) and the remaining 92% first exhibiting changes at a later point during the remainder of the 24-h irradiation period (“late-response” genes) (Table 1, Fig. 1A; see Note 2, supplemental material at www.pgec.usda.gov/Quail/phyA.html for details of analysis). Thirty-four percent of these genes displayed a 2- to 3-fold light-induced change in mRNA abundance, 50% a 3- to 10-fold change, and 16% a 10-fold or greater change. Overall, 66% of the phyA-responsive genes are currently annotated as encoding proteins of known or putative function and the remainder as being of unknown function (Fig. 1A). Of the known category, over half are functionally classified as being involved in either photosynthesis/chloroplast biogenesis (24%) or general cellular metabolism (28%), with genes putatively involved in transcriptional regulation (13%) or stress and defense (12%) also significantly represented (Fig. 1A). The remaining 23% are comprised about equally of genes putatively involved in signaling, transport, hormone pathways, or growth and development (Fig. 1A). A list of all identified genes, assigned to each functional category, can be found in Tables 2–7, supplemental material at www.pgec.usda.gov/Quail/phyA.html.
Table 1.
Number of phyA-regulated genes categorized by functional class and temporal expression pattern
| Functional classification | Early-response genes
(1 h)
|
Late-response genes (3–24 h)
|
Total | ||
|---|---|---|---|---|---|
| Induced | Repressed | Induced | Repressed | ||
| Transcription (Tx) | 18 | 3 | 20 | 29 | 70 |
| Photosynthesis/Chloroplast (P/C) | 7 | 0 | 115 | 4 | 126 |
| Cellular Metabolism (CM) | 5 | 0 | 93 | 54 | 152 |
| Signaling (S) | 4 | 0 | 13 | 8 | 25 |
| Transporters (Tr) | 6 | 0 | 11 | 8 | 25 |
| Growth and Development (G/D) | 0 | 2 | 18 | 25 | 45 |
| Hormone-pathway related (H) | 1 | 0 | 6 | 20 | 27 |
| Stress/Defense (S/D) | 2 | 0 | 32 | 30 | 64 |
| Hypothetical/Unknown (H/U) | 13 | 1 | 178 | 86 | 278 |
| Totals | 56 | 6 | 486 | 264 | 812 |
Genes represented on the microarray whose mRNA abundance was either increased (induced) or decreased (repressed) 2-fold or more under phyA control within 1 h (early) or between 3 and 24 h (late) of the start of FRc irradiation were scored and classified into the broad functional categories shown, according to established or putative function in the plant.
Figure 1.
Percentage distribution of genes in various categories. (A) (a) Percentage of total genes represented on the microarray that are regulated or not regulated by phyA in response to FRc. (b) Percentage of phyA-regulated genes that are induced or repressed, early (within 1 h) or late (between 3 and 24 h), in response to FRc. (c) Percentage of phyA-regulated genes currently annotated as having a known or putative function and those being of unknown, hypothetical, or undesignated function. (d) Overall percentage of phyA-regulated genes in the functional categories listed in Table 1. (B) Percentage of phyA-regulated genes in each functional category that are induced or repressed, early or late, in response to FRc irradiation. See Table 1 for abbreviations.
Global Analysis of Temporal Expression Patterns.
To begin to identify gene classes involved in the primary phase of phyA-induced deetiolation, as opposed to later elaboration of the induced changes, we examined the temporal patterns of expression exhibited by the genes in each functional category, with particular focus on whether changes in abundance were first detectable within 1 h of FRc exposure or later. Striking differences between some categories are apparent. In particular, whereas genes involved in photosynthesis and general metabolism are together clearly the predominant group in the late-response category (56% of the known genes in this category), genes encoding transcription-related proteins are the major class among early-response genes (44% of the known genes in this category) (Table 1, Fig. 1B).
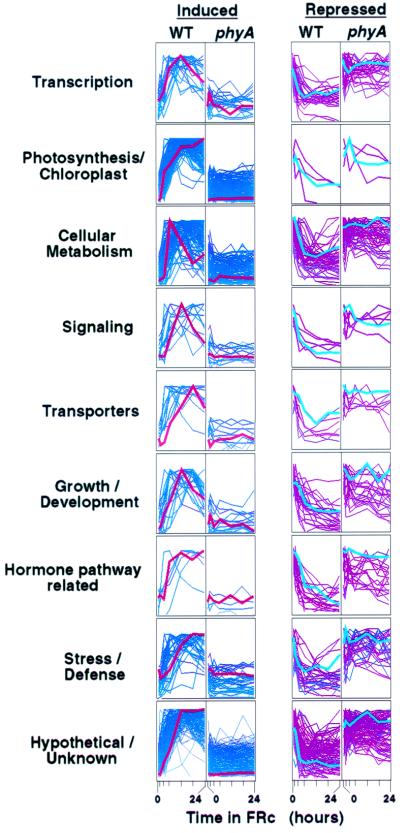
The assembled expression profiles for each functional category are presented for the “late” genes in Fig. 2 and for the “early” genes in Fig. 3. In Fig. 2, the normalized profiles of all late genes within a category have been generated and superimposed using genespring software (Silicon Genetics, Redwood City, CA) and are presented with the wild-type and mutant profiles for induced and repressed genes in separate adjacent panels. In Fig. 3, the expression profile of each “early” gene is presented separately in the miniaturized panels shown, with the full 24-h time course of both wild type and mutant together on the right and the means and standard errors for the triplicate 0- and 1-h values in the histograms on the left. The separate profiles for each of the individual genes presented in Figs. 2 and 3 can be viewed as links to the gene list in Tables 2–7, supplemental material at www.pgec.usda.gov/Quail/phyA.html.
Figure 2.
Late-response genes in various functional categories respond coordinately to FRc irradiation in wild-type but not phyA-null mutant seedlings. The 24-h time-course expression profiles for all late-response genes in each category have been superimposed for the wild-type and phyA-null mutants separately by using genespring software. (Left) Genes induced by FRc. (Right) Genes repressed by FRc. All profiles are normalized to the maximum value in the time course for that gene. A single example profile has been highlighted for each category of induced (red) and repressed (blue) genes. The complete list of late-response genes is presented in Tables 6 and 7, supplemental material, with links to the separate time-course curves for each individual gene, at www.pgec.usda.gov/Quail/phyA.html.
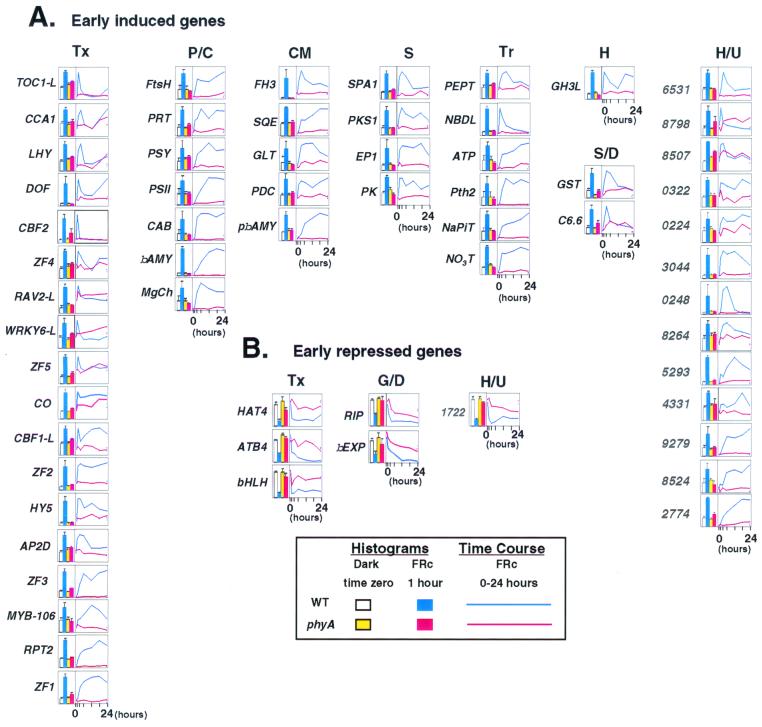
Figure 3.
Putative transcription factor genes are the predominant functional class of early-response genes. The expression profile for each of the early-response genes identified here is presented in a separate panel and grouped vertically under functional category (see Table 1 for abbreviations). (A) Induced genes; (B) repressed genes. The 24-h time-course profiles for each gene are shown on the right of each panel for wild-type (blue) and phyA-null mutant (red). All profiles are normalized to the maximum value in the time course for that gene. The histograms to the left of each panel show the expression level for each gene at 1 h of FRc in wild-type (blue) and phyA mutant (red) compared with the time-zero dark control wild-type (white) and phyA mutant (yellow). The histogram data represent the means ± standard errors (bars) of triplicate microarray measurements performed on three RNA samples independently prepared from separate tissue samples. All data have been normalized to the maximum value for that gene in the histogram dataset. Encoded-protein names and accession nos.: (A) Early induced genes: TOC1-L, timing of CAB1-like (AAC33497); CCA1, circadian clock-associated protein1 (AAC33507); LHY, late elongated hypocotyl (CAB42406); DOF, H-protein promoter-binding factor-2a (AAC28390); CBF2, CRT/DRE-binding factor 2 (AAD15976); ZF4, putative zinc-finger protein (CAB38816); RAV2-L, related to ABI3/VP1–2-like (AAD26965); WRKY6-L, WRKY transcription factor-like (AAB60774); ZF5, zinc-finger protein (CAA67232); CO, constans (CAA71588); CBF1-L, CRT/DRE-binding factor 1-like (CAA18178); ZF2, constans-like zinc finger protein (CAA64819); HY5, (BAA21327); AP2D, putative AP2 domain transcription factor (AAD21489); ZF3, putative CONSTANS-like B-box zinc-finger protein (AAD26481); MYB106, putative Myb-related transcription factor (AAF26160); RPT2, root phototropism 2 (AAB63085); ZF1, zinc-finger protein 1 (AAD33769); FtsH, chloroplast FtsH protease (CAA68141); PRT, choloroplast prenyltransferase (AAD30584); PSY, phytoene synthase (AAA32836); PSII, putative protein 1 photosystem II oxygen-evolving complex (CAB42911); CAB, Lhcb2 protein (AAD28769); βAMY, β-amylase (CAB58423); MgCh, magnesium chelatase subunit (CAA92802); FH3, flavanone 3-hydroxylase (CAB62646); SQE, squalene epoxidase-like protein (CAB38924); GLT, putative glucosyl transferase (AAD20156); PDC, pyruvate decarboxylase (AAB16855); pβAMY, putative β-amylase (CAB58423); SPA1, phyA suppressor (AAD30124); PKS1, phy kinase substrate 1 (AAD38033); EP1, strong similarity to glycoprotein EP1 (AAC83044); PK, putative protein kinase (AAC31848); PEPT, putative peptide transporter (CAB41143); NBDL, NBD-like protein (AAD20643); ATP, ATPase (CAA73318); Pth2, phosphate transporter 2 (CAA66116); NaPiT, putative Na+-dependent inorganic phosphate cotransporter (AAC35230); NO3T, nitrate transporter (CAB38706); GH3L, GH3-like protein (AAD14468); GST, glutathione S-transferase (AAD32887); C6.6, cold regulated (CAA38894). Hypothetical and unknown proteins are identified by the last four digits of their protein accession numbers. Full accession numbers are: CAB36531, AAC18798, AAC28507, CAB10322, AAD30224, AAD23044, CAB10248, CAB38264, AAB95293, AAB64331, AAD39279, AAC78524, and AAD32774. (B) Early repressed genes: HAT4 (CAA79670); ATB4, homeodomain transcription factor (ATHB-4) (AAC31833); bHLH, (AAC34226); RIP, ripening-like protein (CAA23067); βEXP, β-expansin (AAD20920). Hypothetical/unknown: (AAD21722). See Tables 2–5, supplemental material, for links to the separate time-course curves for each individual early-response gene, at www.pgec.usda.gov/Quail/phyA.html.
Collectively, these expression profiles provide support for a number of generalizations. First, the data confirm that the genes in all categories exhibit significant responses to FRc in the wild type relative to the starting point at time 0 in the dark control but not in the phyA null mutant. In addition, the majority of these genes showed little or no change in expression level in control seedlings retained in darkness over the 24-h FRc-irradiation period for either wild-type or mutant, or where such changes did occur, they mostly occurred in both wild type and mutant in parallel (data omitted in Fig. 2 for clarity; individual data points at 24 h in Fig. 3). Thus, these data provide robust verification that the genes identified here are regulated by phyA in response to the FRc light signal. Second, the expression profiles collectively indicate that the majority of genes begin to respond to the light signal within 3 h and reach or approach their maximum response within 12 h. This pattern suggests that the major part of the redirection in cellular function underlying the deetiolation process is well established within 12 h of the onset of the light signal. This generalized pattern is most striking for the most abundant classes of late genes, the photosynthesis/chloroplast, cellular metabolism, and unknown classes (Fig. 2), and is indicative of the highly coordinated regulation of expression levels anticipated for this complex transition from heterotrophic to autotrophic development. Third, the overall kinetic behavior of the genes in the various functional categories is generally consistent with their established or proposed functional roles. Again, this is most obvious for the genes involved in chloroplast biogenesis and photosynthesis, where the vast majority are strongly induced but is also apparent for those involved in general cellular metabolism and ion or metabolite transport, where subsets of each are induced or repressed by the light signal (Fig. 2). This pattern presumably reflects the selective induction, enhancement, or phasing out of various metabolic pathways as the transition toward a new homeostatic state is effected. Similarly, subsets of genes with regulatory functions, such as those involved in hormone action, signaling, transcription, and growth and development, display a variety of enhanced or reduced expression profiles in response to FRc (Figs. 2 and 3), indicative of the need for coordinate regulation of the diverse array of activities associated with deetiolation (16). Detailed analysis of specific pathways or individual genes in the late category is beyond the scope of this initial report. However, the data assembled here should serve as a foundational framework providing a rich resource for such detailed analysis in the future (see Tables 2–7, supplemental material at www.pgec.usda.gov/Quail/phyA.html).
Early-Response Genes.
Detailed analysis of the small number of genes induced or repressed within 1 h of FRc treatment has provided intriguing insight into the nature of the early events triggered by the light signal (Fig. 3). The most striking observation is the preponderance of transcription-factor-related genes represented in this category. The rapid induction or repression of these genes suggests that they are integral members of an early-response transcriptional network under phyA control. Moreover, because these genes encode multiple classes of factors, the data suggest rapid amplification and broadening of the categories of genes that are downstream targets of these factors in the phyA-induced transcriptional cascade. It is noteworthy that zinc-finger proteins constitute the single largest class in this group, but that bZIP, homeodomain, MYB, AP2-domain, WRKY, and bHLH factors are also represented (Fig. 3). Together, these include representatives from the majority of the main transcription-factor classes present in Arabidopsis (17).
Some of these genes have already been implicated in phy-regulated responses. The bZIP protein HY5, originally identified genetically, is well documented to have a central role in light-regulated deetiolation, with evidence that this involves recognition of G-box DNA-binding sites in target genes (18). The MYB-related factor, CCA1, was isolated by its capacity to bind to a functionally defined cis element in an Arabidopsis CAB gene promoter and has been shown to be involved in regulating the light-induced expression of that gene (19). In addition, CCA1, the related MYB-like factor, LHY, and TOC1-L have all been implicated in circadian clock regulation (19–23), which is known to be regulated by light signals through the phy system (24). Intriguingly, all three, together with the zinc-finger protein, DOF, identified as binding to the promoter of a light-regulated glycine decarboxylase subunit involved in photorespiration (ref. 25; H. K. R. Abbaraju and D. J. Oliver, GenBank accession no. AF079503), display strong very rapid transient increases in mRNA levels, followed by a second slower rise toward the end of the 24-h period (Fig. 3). This pattern may reflect circadian activity, and if so, it will be interesting to determine whether DOF has a clock-related function. On the other hand, RPT2, identified as a signaling component in blue-light-induced phototropism (26), but which is also clock-regulated (24), shows a more gradual monotonic increase in mRNA abundance over the 24-h period. Other factors previously shown to be light-related include the zinc-finger factor, CO, involved in photoperiodic control of flowering (27), and the two down-regulated homeodomain factors, HAT4 and ATB4 (28). None of the remaining transcription-related factors appear to have been shown to be involved in photoresponses before but are now clearly of interest in this regard.
SPA1 and PKS1 have both previously been implicated as negative regulators of phyA signaling (29, 30). The transient phyA-induced expression of SPA1 observed here closely matches that previously determined by blot analysis (29). PKS1 was identified as a phyA-binding protein, which can be phosphorylated by phyA-associated kinase activity. The phyA-induced increase in transcript levels reported here may be consistent with the proposed function of PKS1 as a cytoplasmic phyA retention factor (30). EP1 and PK have apparently not previously been implicated in phyA signaling.
The gene GH3L is closely related to FIN219, a locus recently identified as being involved in phyA signaling (31). Both genes are related to GH3, a soybean gene long known to respond rapidly to the hormone auxin, and indeed FIN219 is also rapidly induced by auxin (31). The strong rapid induction of GH3L here (Fig. 3) suggests a close link between phyA and auxin activity. The down-regulated β-expansin, β-EXP, and ripening-related, RIP, genes (Fig. 3) are potentially involved in cell-wall metabolism (32). These genes could, therefore, potentially be involved in the changes in cell expansion rates responsible for the well known phyA-induced changes in growth rates accompanying deetiolation (7).
The detection of the initial phases of induction at 1 h of several strongly induced photosynthesis-related genes (Fig. 3) is indicative that the necessary transcriptional activators are already operative at this time. The relative temporal patterns of the expression profiles of CAB and the MYB-factor gene, CCA1, which encodes an established regulator of CAB (19), provides correlative evidence to this effect and is suggestive of a transcriptional cascade with CAB as the terminal output gene (Fig. 6, supplemental material, www.pgec.usda.gov/Quail/phyA.html). Similar temporal relationships are observed for HY5 and CHS and for DOF and GDCH (Figs. 7 and 8, supplemental material, www.pgec.usda.gov/Quail/phyA.html). The expression profiles of the remaining functionally annotated genes in the early-response category are indicative of the need for rapid deployment of enhanced levels of a variety of membrane transporters, such as those involved in nitrate (NO3T) and phosphate (NaPiT) transport, and for altered sugar metabolism, as indicated by the pronounced induction of two β-amylase genes (βAMY and pβAMY) and the glucosyl transferase gene, GLT (Fig. 3). Elucidation of the functional roles of the substantial group of currently “unknown” genes in this category (Fig. 3) can be expected to yield further insight into early phyA-regulated transcriptional events.
A phyA-Regulated Transcriptional Network.
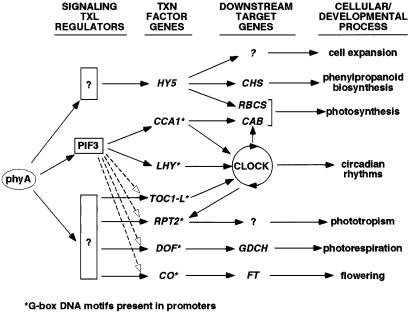
The expression profiles presented here not only identify and catalog, to our knowledge for the first time, the more than 800 Arabidopsis genes on the chip that are phyA regulated but more importantly have begun to provide insight into the structure of the primary transcriptional network that initiates and coordinates the genome-wide response to phyA signals (Fig. 4). To what extent the genes in the early-response category might be direct targets of the primary phyA signaling pathway remains to be investigated. However, there is evidence suggesting that at least two genes, CCA1 and LHY, in the transcription factor category, are such targets. Each of these genes carries a G-box promoter element capable of binding the basic helix–loop–helix factor, PIF3, and each requires PIF3 for rapid light-induced transcript accumulation mediated by phyB (33). PIF3 in turn binds either phyA or phyB specifically on light-induced conversion of the photoreceptor to its biologically active Pfr form (33, 35), suggesting a direct signaling pathway from these phys to target genes via physical interaction of the light-activated photoreceptor molecules with a promoter-bound transcriptional regulator. Consistent with this suggestion, there is compelling evidence that the phys are induced to translocate into the nucleus on photoconversion to the Pfr form (36, 37). Apart from CCA1 and LHY, a number of the other early-response genes in the transcription factor category also carry G-box motifs in their promoters (Fig. 4; data not shown). Whether PIF3 is involved in the phyA-regulated expression of these transcription-factor genes remains to be determined, as the relationship between G-box elements and their cognate-binding proteins is complex (38).
Figure 4.
Simplified schematic of postulated phyA-regulated transcriptional network. It is proposed that the phyA-regulated transcription (TXN)-factor genes identified here are primary targets of phyA signaling via “signaling transcriptional (TXL) regulators” constitutively present before light signal perception, and that these TXN-factor genes encode a master set of regulators, each of which regulates one or more major branches of cellular or developmental activity by controlling the expression of specific downstream target genes. PIF3, a bHLH factor, is proposed to function as one such “signaling TXL regulator” on the basis of its capacity to bind to G-box sequence elements in the promoters of CCA1 and LHY (35) and to bind specifically to the active Pfr form of phyA (37), thereby targeting light signals directly to these genes. The promoters of several other of the transcription-factor genes also carry G-box motifs (asterisks) making them potential PIF3 targets (dashed arrows). For genes lacking functionally relevant PIF3-binding sites, such as HY5, which appears to lack a G-box, we postulate that other yet to be identified “signaling TXL regulators” (question marks in boxes) may fulfill this role. Some of the key downstream genes in the different pathways, known or proposed to be targets of the TXN-factor gene products listed, are indicated. CHS, chalcone synthase; RBCS, ribulose bisphosphate carboxylase small subunit; CAB, chlorophyll a/b-binding protein; GDCH, H-protein subunit of glycine decarboxylase; FT, flowering locus T.
Taken together, the data presented here suggest the possibility that phyA may regulate the process of deetiolation, at least in part, by direct targeting of light signals to the promoters of a master set of multiple classes of transcription factor genes (Fig. 4). The observation that phyA localizes to multiple nuclear speckles after translocation from the cytoplasm (36, 37) is consistent with the proposed direct regulation of multiple genes at different loci in the genome. The diversity of factors in this set (Fig. 3) suggests extensive and immediate branching in the phyA signaling network via amplification and diversification of the spectrum of downstream genes that are potentially the targets of these factors. Fig. 4 depicts the major downstream cellular processes known or proposed to be under the control of these factors. The coupling of microarray analysis, such as that presented here, with mutants carrying lesions in specific members of the early-response gene category, or in upstream factors like PIF3, will greatly assist dissection of this network. Available evidence already indicates that this network is likely to represent an intricate and complex web of intersecting branches rather than simple linear pathways.
Acknowledgments
We thank Stacey Harmer and Steve Kay for providing data before publication and helpful discussions, the members of our laboratory for useful discussions and comments on the manuscript, Julie Mathern for help with the web pages, and Ron Wells for manuscript preparation. This work was supported by the Torrey Mesa Research Institute, National Institutes of Health Grant GM47475, and Agricultural Research Service Grant 5335–21000-010–00D.
Abbreviations
- phy
phytochrome
- FRc
continuous far-red light
References
- 1.Smith H. Nature (London) 2000;407:585–591. doi: 10.1038/35036500. [DOI] [PubMed] [Google Scholar]
- 2.Tobin E, Kehoe D M. Semin Cell Biol. 1994;5:335–346. doi: 10.1006/scel.1994.1040. [DOI] [PubMed] [Google Scholar]
- 3.Terzaghi W B, Cashmore A R. Annu Rev Plant Physiol Plant Mol Biol. 1995;46:445–474. [Google Scholar]
- 4.Kuno N, Muramatsu T, Hamazato F, Furuya M. Plant Physiol. 2000;122:15–24. doi: 10.1104/pp.122.1.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lipshutz R J, Fodor S P A, Gingeras T R, Lockhart D J. Nat Genet Suppl. 1999;21:20–24. doi: 10.1038/4447. [DOI] [PubMed] [Google Scholar]
- 6.Mathews S, Sharrock R A. Plant Cell Environ. 1997;20:666–671. [Google Scholar]
- 7.Whitelam G C, Devlin P F. Plant Cell Environ. 1997;20:752–758. [Google Scholar]
- 8.Devlin P F, Patel S R, Whitelam G C. Plant Cell. 1998;10:1479–1487. doi: 10.1105/tpc.10.9.1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Deng X-W, Quail P H. Semin Cell Dev Biol. 1999;10:121–129. doi: 10.1006/scdb.1999.0287. [DOI] [PubMed] [Google Scholar]
- 10.Young R A. Cell. 2000;102:9–15. doi: 10.1016/s0092-8674(00)00005-2. [DOI] [PubMed] [Google Scholar]
- 11.Hartwell L H, Hopfield J J, Leibler S, Murray A W. Nature Suppl. 1999;402:C47–C52. doi: 10.1038/35011540. [DOI] [PubMed] [Google Scholar]
- 12.Valvekens D, Van Montagu M, van Lijsebettens M. Proc Natl Acad Sci USA. 1988;85:5536–5540. doi: 10.1073/pnas.85.15.5536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhu T, Wang X. Plant Physiol. 2000;124:1472–1476. doi: 10.1104/pp.124.4.1472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Arabidopsis Genome Initiative. Nature (London) 2000;408:796–815. doi: 10.1038/35048692. [DOI] [PubMed] [Google Scholar]
- 15.Eisen M B, Spellman P T, Brown P O, Botstein D. Proc Natl Acad Sci USA. 1998;95:14863–14868. doi: 10.1073/pnas.95.25.14863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Alon U, Barkai N, Notterman D A, Gish K, Ybarra S, Mack D, Levine A J. Proc Natl Acad Sci USA. 1999;96:6745–6750. doi: 10.1073/pnas.96.12.6745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Riechmann J L, Heard J, Martin G, Reuber L, Jiang C Z, Keddie J, Adam L, Pineda O, Ratcliffe O J, Samaha R R, et al. Science. 2000;290:2105–2110. doi: 10.1126/science.290.5499.2105. [DOI] [PubMed] [Google Scholar]
- 18.Chattopadhyay S, Ang L-H, Puente P, Deng X-W, Wei N. Plant Cell. 1998;10:673–683. doi: 10.1105/tpc.10.5.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang Z-Y, Tobin E M. Cell. 1998;93:1207–1217. doi: 10.1016/s0092-8674(00)81464-6. [DOI] [PubMed] [Google Scholar]
- 20.Schaffer R, Ramsay N, Samach A, Corden S, Putterill F, Carrè I A, Coupland G. Cell. 1998;93:1219–1229. doi: 10.1016/s0092-8674(00)81465-8. [DOI] [PubMed] [Google Scholar]
- 21.Green R M, Tobin E M. Proc Natl Acad Sci USA. 1999;96:4176–4179. doi: 10.1073/pnas.96.7.4176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Strayer C, Oyama T, Schultz T F, Raman R, Somers D E, Mas P, Panda S, Kreps J A, Kay S A. Science. 2000;289:768–771. doi: 10.1126/science.289.5480.768. [DOI] [PubMed] [Google Scholar]
- 23.Matsushika A, Makino S, Kojima M, Mizuno T. Plant Cell Physiol. 2000;41:1002–1012. doi: 10.1093/pcp/pcd043. [DOI] [PubMed] [Google Scholar]
- 24.Harmer S L, Hogenesch J B, Straume M, Chang H-S, Han B, Zhu T, Wang X, Kreps J A, Kay S A. Science. 2000;290:2110–2113. doi: 10.1126/science.290.5499.2110. [DOI] [PubMed] [Google Scholar]
- 25.Srinivasan R, Oliver D J. Plant Physiol. 1995;109:161–168. doi: 10.1104/pp.109.1.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sakai T, Wada T, Ishiguro S, Okada K. Plant Cell. 2000;12:225–236. doi: 10.1105/tpc.12.2.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Samach A, Onouchi H, Gold S E, Ditta G S, Schwarz-Sommer Z, Yanofsky M F, Coupland G. Science. 2000;288:1613–1616. doi: 10.1126/science.288.5471.1613. [DOI] [PubMed] [Google Scholar]
- 28.Morelli G, Ruberti I. Plant Physiol. 2000;122:621–626. doi: 10.1104/pp.122.3.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hoecker U, Tepperman J M, Quail P H. Science. 1999;284:496–499. doi: 10.1126/science.284.5413.496. [DOI] [PubMed] [Google Scholar]
- 30.Fankhauser C, Yeh K C, Lagarias J C, Zhang H, Elich T D, Chory J. Science. 1999;284:1539–1541. doi: 10.1126/science.284.5419.1539. [DOI] [PubMed] [Google Scholar]
- 31.Hsieh H-L, Okamoto H, Wang M, Ang L-H, Matsui M, Goodman H, Deng X W. Genes Dev. 2000;14:1958–1970. [PMC free article] [PubMed] [Google Scholar]
- 32.Cosgrove D J. Annu Rev Plant Physiol Plant Mol Biol. 1999;50:391–417. doi: 10.1146/annurev.arplant.50.1.391. [DOI] [PubMed] [Google Scholar]
- 33.Martínez-García J F, Huq E, Quail P H. Science. 2000;288:859–863. doi: 10.1126/science.288.5467.859. [DOI] [PubMed] [Google Scholar]
- 34.Ni M, Tepperman J M, Quail P H. Nature (London) 1999;400:781–784. doi: 10.1038/23500. [DOI] [PubMed] [Google Scholar]
- 35.Zhu Y, Tepperman J M, Fairchild C D, Quail P. Proc Natl Acad Sci USA. 2000;97:13419–13424. doi: 10.1073/pnas.230433797. . (First Published November 7, 2000; 10.1073/pnas.230433797) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kim L, Kircher S, Toth R, Adam E, Schäfer E, Nagy F. Plant J. 2000;22:125–133. doi: 10.1046/j.1365-313x.2000.00729.x. [DOI] [PubMed] [Google Scholar]
- 37.Nagy F, Schäfer E. EMBO J. 2000;19:157–163. doi: 10.1093/emboj/19.2.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Quail P H. Semin Cell Dev Biol. 2000;11:457–466. doi: 10.1006/scdb.2000.0199. [DOI] [PubMed] [Google Scholar]