Graphical abstract
Keywords: Angiogenesis inhibitors, Receptor protein-tyrosine kinase, Tumor microenvironment, Natural products
Abstract
Recent strategies for the treatment of cancer, other than just tumor cell killing have been under intensive development, such as anti-angiogenic therapeutic approach. Angiogenesis inhibition is an important strategy for the treatment of solid tumors, which basically depends on cutting off the blood supply to tumor micro-regions, resulting in pan-hypoxia and pan-necrosis within solid tumor tissues. The differential activation of angiogenesis between normal and tumor tissues makes this process an attractive strategic target for anti-tumor drug discovery. The principles of anti-angiogenic treatment for solid tumors were originally proposed in 1972, and ever since, it has become a putative target for therapies directed against solid tumors. In the early twenty first century, the FDA approved anti-angiogenic drugs, such as bevacizumab and sorafenib for the treatment of several solid tumors. Over the past two decades, researches have continued to improve the performance of anti-angiogenic drugs, describe their drug interaction potential, and uncover possible reasons for potential treatment resistance. Herein, we present an update to the pre-clinical and clinical situations of anti-angiogenic agents and discuss the most recent trends in this field.
Introduction
Cancer is one of the leading causes of death and constitutes a national and international health problem regardless of the development status of the country (developed, developing or undeveloped country) [1]. Yet, no single outstanding anticancer treatment has been discovered. The World Health Organization (WHO) reported ideological failure in changing the mortality attributed to cancer over the past 5 decades (1950–2000), in contrast to other death-causing diseases [2]. Solid tumors constitute more than 94.4% and 96.8% of cancer-caused mortalities in males and females, respectively [3]. Recent strategies, other than just discovering novel anticancer agents, have been under intensive development such as pharmacokinetic utilization of the anti-angiogenic therapeutic approach [4]. Specifically, deregulation of angiogenesis by synthetic and natural products is being accepted as a good target for cancer prevention and treatment [5], [6], [7], [8].
Angiogenesis phenomenon in healthy and diseased tissues
The term “angiogenesis” was introduced in 1787 by the British surgeon John Hunter in order to describe the formation of new vessels in the process of wound healing [9]. Angiogenesis is an essential, temporary physiological process of forming a new vascular tree from an existing one to supply a certain tissue with oxygen and nutrients as well as removing its carbon dioxide and waste products. Apart from embryogenesis, in rare cases, angiogenesis can be a healthy process such as during wound healing and the menstrual cycle [10]. Vasculogenesis is a different process in which blood vessels are formed from angioblast cells (rather than from mature blood vessels) during embryogenesis [11]. Prolonged angiogenesis is usually indicative of a pathological condition such as arthritis, diabetic retinopathy or cancer progression [12]. The differential activation of angiogenesis between normal and tumor cells makes this process an attractive strategic target for anti-tumor drug discovery. The principles of anti-angiogenic treatment were originally proposed by Judah Folkman in 1972, and ever since, the ability of a tumor to form new blood vessels to feed their abnormally high growth rate has become a therapeutic target. Hence, this has become a putative target for therapies directed against solid tumors [12], [13], [14].
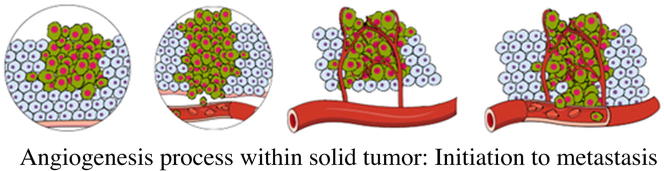
Targeting tumor angiogenesis not only confers relative selectivity to tumor tissue but also enables the targeting of wide-range heterogeneous tumors that only share high angiogenic potential. Within the human body, angiogenesis is orchestrated by two sets of regulatory molecules with opposing functions; pro-angiogenic molecules (such as vascular endothelial growth factor, VEGF) and anti-angiogenic molecules (such as thrombospondin-1) [15]. Under homeostatic conditions, pro/anti angiogenic balance is shifted toward anti-angiogenic factors, resulting in quiescent blood vessels. On the other hand, the angiogenic balance in neoplastic lesions is shifted toward pro-angiogenesis [16]. This pathological transition is known as the angiogenic switch. Tumor hypoxia is believed to be the main pathological deriver behind this switch [17]. The release of pro-angiogenic factors from tumor cells and host cells, such as macrophages, causes disruption of the surrounding vasculature’s basement membrane which is attributed to the activation of a group of proteases, such as plasminogen activator and collagenases [18]. These pro-angiogenic factors also work as chemotactic factors for endothelial cells (ECs), causing migration and proliferation within the tumor tissue and thus forming a vascular lumen structure [10]. In addition, the released angiogenic factors attract circulating bone marrow progenitor cells and stimulate their differentiation into ECs [19]. Then, new basement membrane is formed, and pericytes are attracted to circumvent the neo-vessel [20]. Apart from meeting the metabolic demands of the pre-existing tumor cells, these neo-vessels support further tumor growth and invasion [21]. In addition, intratumoral angiogenesis could serve as potential gateway to spread tumor cells toward distant tissues and facilitate the process of metastasis [22]. Interestingly, pathogenic induction of intratumoral angiogenesis appears to begin as early as during the pre-malignant phase of tumor development [23].
The degree of angiogenesis is not similar in all tumor types. Pancreatic neuroendocrine carcinoma is a highly vascularized tumor, while pancreatic ductal adenocarcinoma possesses low angiogenic potential [24], [25]. In addition, the degree of vascularization varies from one micro-region to another within the same tumor tissue [26]. Sustained activation of angiogenesis within tumor micro-regions ultimately results in hypervascular structure with dysfunctional endothelium. These neo-vessels are characterized by increased permeability and leakiness [27]. In addition to ECs, pericytes are vascular support cells that functionally and structurally support the vascular endothelium. Yet, pericytes tend to be loose around intratumoral vasculature, suggesting a potential reason for the high permeability of tumor vasculature [28]. In 1989, the successful cloning of vascular endothelial growth factor-A (VEGF-A) could be considered the first clue to understanding the molecular bases of angiogenesis in solid tumors [29].
Principles of anti-angiogenic treatment for cancer “tumor under siege strategy”
Normal blood vessels are classified into three major types according to their endothelial lining and their underlying basement membranes. 1 – Continuous capillaries which are characterized by continuous sheets of sub-endothelial basement membrane and tightly packed monolayer of endothelium to prevent uncontrolled transfer of substances such as in blood brain barrier. 2 – Fenestrated capillaries which are characterized by continuous sheets of sub-endothelial basement membrane and loosely packed monolayer of endothelium to allow regular substances transfer (e.g. lung and GIT). 3 – Perforated capillaries which are characterized by perforated sheets of sub-endothelial basement membrane and loosely packed monolayer of endothelium to allow transfer of macromolecules such as hormones and peptides (e.g. endocrine glands). Intratumoral blood vessels are phenotypically similar to perforated capillaries; however, they are premature and possess unique peculiarities. In contrast to normal blood vessels, the intratumoral blood vessels are immature, highly permeable, and chaotic with heterogeneous and interrupted blood flow [30]. Angiogenesis inhibition is a potential novel appealing strategy for the treatment of solid tumors which basically depends on cutting off the blood supply to tumor micro-regions, resulting in pan-hypoxia and pan-necrosis within solid tumor tissues. Selectivity of anti-angiogenic agents toward intratumoral vasculature depends mainly on the phenotypic differences between the premature intratumoral vasculature and normal blood vessels. These phenotypic differences result in relative increased sensitivity of the intratumoral blood vessels to anti-angiogenic agents. The general mechanism of action of angiogenesis inhibitor (AI), nonetheless, vascular disrupting agent (VDA) is through induction of morphologic changes in the intratumoral endothelium; this in turn triggers a cascade of events that ultimately leads to vascular shutdown and tumor necrosis [30]. Initial events can be detected as early as 5–25 min following drug administration in the form of increased vascular permeability, vasoconstriction of tumor-supplying arterioles, reduction of blood flow and tumoral pan-hypoxia [31]. A few hours later (6–24 h), platelet activation, coagulation, vascular occlusion, recruitment of inflammatory cells and vascular remodeling may occur, leading to tumoral pan-necrosis [32]. VDAs are a subclass of AIs that acutely cut off the blood supply with a very early onset of action (a few hours or even minutes). VDA mainly interacts with intratumoral chaotic vasculature; however, a certain degree of ambiguity can occur that might result in adverse pathological changes in normal blood vessels. Anti-angiogenesis is gaining much attention as a unique mechanism for targeting solid tumors [33]. Preventing de novo angiogenesis and leaving a solid tumor mass to die silently without blood supply appears very appealing as an anti-cancer tumor strategy [34].
In the current review, the term AI will be used to represent both subtypes. An important question to understand the clinical effectiveness of using AI should be asked; does it work against large-sized tumors only such as in the primary site or against the small malignant foci of metastasis? Tumor cell proliferation and hence generalized tumor mass growth rate must be accompanied by fast growth of an intratumoral vascular tree. Nutrients and oxygen cannot diffuse from a functioning blood vessel to a tumor cell beyond 100–500 µm [35], which is approximately as small as the size of newly formed metastatic focus. In addition, metastatic tumor cells are originally released to bloodstream from within an intratumoral blood vessel [36]. Yet, bigger and more diverse intratumoral vascular density increases the chance of metastasis. Clinically, high intratumoral vascular density in nearly all types of cancers is associated with increased metastasis and poor survival [37]. Recently, several clinical trials for investigational anti-angiogenic agents against metastatic melanoma, head and neck cancers, malignant melanoma, non-small cell lung cancer and other tumor types have been completed or undergoing [38]. Another conceptual question is whether there is any significance to using AI for hematological malignancies. It is reported that there is excessive angiogenesis and higher microvascular density within bone marrow of patients suffering from hematological neoplasia and is associated with poor survival and prognosis [39], [40]. To the best of our knowledge, there is no approved anti-angiogenic agent for the treatment of hematologic malignancies. However, several clinical trial are under way [40].
Different angiogenic pathways targeted/potentially targeted for anticancer therapeutic purposes
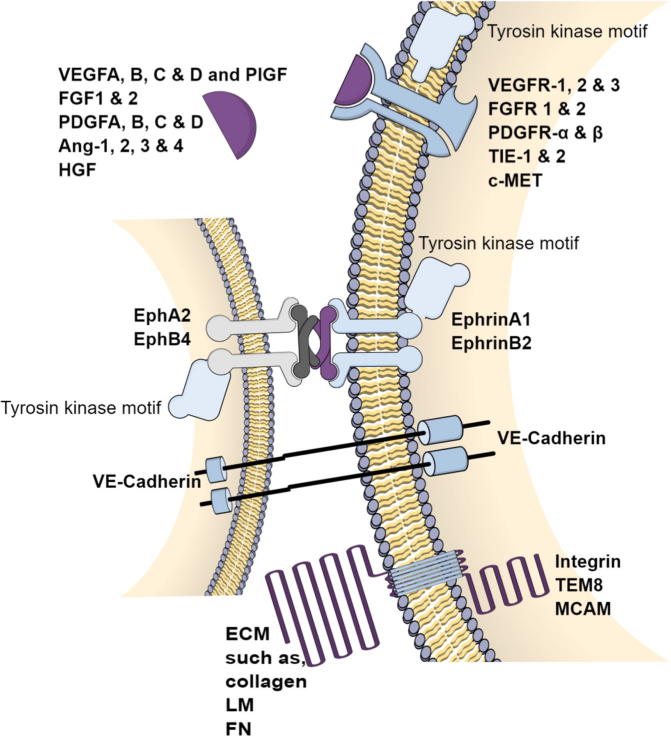
The intratumoral microenvironment is formed of complex soluble, non-soluble and cellular factors that control tumor growth-derived angiogenesis. Formation of an intratumoral neo-vessel takes place when pro-angiogenic factors overweigh anti-angiogenic factors within the intratumoral micro-milieu. Yet, several factors/molecular pathways are known to directly/indirectly influence the process of intratumoral angiogenesis. Targeting one or more of these pathways would result in therapeutic benefits attributed to intratumoral anti-angiogenesis (Fig. 1).
Fig. 1.
Molecular aspects of different angiogenic pathways; brief diagrammatic summary for different molecular pathways involved in angiogenesis. Designed using Mind The Graph™, Zendesk Inc., San Francisco, CA, USA.
VEGF/VEGFR pathway
Vascular endothelial growth factor (VEGF) was appointed by the father of intratumoral angiogenesis, Judah Folkman, as the hallmark symbiotic messenger between tumor cells and ECs [41]. VEGFs are secreted from several cell types (fibroblasts, inflammatory cells and many tumor cell types) to interact with the trans-membrane tyrosine kinase dimeric receptors (VEGFRs) that are abundant on ECs. VEGF/VEGFR interaction within ECs initiates an intracellular cascade of signaling events that ultimately results in ECs’ survival, proliferation, maturation, migration and tube formation [42]. The first FDA-approved anti-angiogenic agent for the treatment of solid tumors was bevacizumab, a humanized monoclonal anti-VEGF antibody [43], [44]. Four different well-identified VEGF ligands (VEGF-A, VEGF-B, VEGF-C and VEGF-D) interact with three VEGF receptors (VEGFR-1, VEGFR-2 and VEGFR-3). Of these interactions, the VEGF-A/VEGFR-2 interaction is the most prominent interaction in promoting intratumoral angiogenesis [45]. VEGF-A and VEGF-B possess the highest affinity to VEGFR-1 and VEGFR-2. Yet, VEGFR-1 is thought to be a decoy receptor involved in negative feedback control of VEGF-A/VEGFR-2 interaction [46]. The other VEGFs (C and D) are responsible for lymphangiogenesis via interacting with VEGFR-3 [47]. Some reports indicated the importance of the VEGF/VEGFR pathway in normal vascular integrity as well [48].
FGF/FGFR pathway
Fibroblast growth factors (FGFs) are heparin-binding growth factors secreted mainly from fibroblasts and stored bound near the basement membrane of EC’s. Two well-identified variants of FGF (FGF-1 and FGF-2) can interact with their corresponding trans-membrane tyrosine kinase receptors, FGFR-1 and FGFR-2, respectively. FGF and particularly, FGF-2/FGFR-2, are involved in EC proliferation, migration and differentiation leading to intratumoral angiogenesis [49], [50]. Light was shed on FGF as an important pro-angiogenic factor due to the involvement of FGF-2 in colorectal cancer resistance to anti-VEGF therapies [50], [51]. FGF-2/FGFR-2 interaction might bypass the role of the VEGF/VEGFR pathway in inducing angiogenesis via activating ECs’ proliferation and inducing differentiation of epiblast cells to ECs. Besides, FGF-2/FGFR-2 interaction is involved in the production of collagenase and urokinase-type plasminogen activator with consequent excessive chemo-attraction and facilitated tissue remodeling for angiogenesis [42], [52]. In 2000, it was the first time to target FGF-2/FGFR-2 interaction as anti-angiogenic approach [53]. Besides, combined anti-VEGF and anti-FGF approaches showed more prominent anti-angiogenesis than either alone [54].
PDGF/PDGFR pathway
Platelet-derived growth factors (PDGFs) are group of peptides (PDGF-A, B, C and D) which dimerize (homodimers or heterodimers) and interact with trans-membrane tyrosine kinase receptors (PDGFR-α and PDGFR-β) to elicit downstream signaling very similar to VEGFRs, such as MAPK, Raf/Ras, PKC and PI3K [55]. Activation of PDGF signaling (PDGF-B/PDGFR-β) primarily recruits pericytes to neo-vessels with subsequent secretion of a wide range of pro-angiogenic factors leading to EC proliferation, migration and vascular maturation [56].
PlGF/VEGFR pathway
Placental growth factor (PlGF) belongs to the VEGF superfamily and interacts with VEGFR-1. Unlike VEGF, activation of PlGF is merely pathologic in conditions such as inflammation and intratumoral angiogenesis. PlGF knockout mice survive healthy normally with intact vascular system [57]. However, the true potential for anti-PlGF as a therapeutic remedy in inhibiting intratumoral angiogenesis is questionable because it shares the same receptors with the hallmark angiogenic agent, VEGF [58].
ANG/TIE receptors pathway
Angiopoietins (ANG) is a family of growth factors (ANG-1, ANG-2, ANG-3 and ANG-4) which couple tyrosine kinase receptors (TIE-1 and TIE-2) expressed on ECs. Their most prominent intratumoral pro-angiogenic effects are attributed to complicated interaction between ANG-1 and ANG-2 with TIE-2 receptors [59]. ANG-1 is a super agonist that recruits pericytes to premature segments of neo-vessels. On the other hand, ANG-2 is considered as a partial agonist that induces pericytes to reside and exposes ECs to other angiogenic factors [60]. The delicately programmed ANG-1 and ANG-2 interaction with TIE-2 receptors results in intratumoral EC sprouting, vascular remodeling and plasticity. It is worth mentioning that, ANG-1 is merely expressed and secreted from tumor cells [52]. In addition, overexpression of ANG-2 was associated with tumor invasiveness and poor clinical prognosis [52].
HGF/c-MET
The cellular mesenchymal-epithelial transition protein (c-MET) belongs to the trans-membrane tyrosine kinase family which upon activation by the pleiotropic hepatocyte growth factor (HGF) elicits survival, proliferation and motility of normal as well as tumor cells and tumor associated ECs [61]. Interestingly, HGF/c-MET-induced angiogenic response was found to be mediated via excessive release of pro-angiogenic factors such as VEGF. Besides, HGF/c-MET pathway activation is a potential cue in the development of anti-VEGF resistance [62].
RET
The mutated form of the proto-oncogene tyrosine kinase protein, rearranged during transfection (RET), is known for its association with the progression of various tumor types. It is found that RET is involved with intratumoral angiogenesis as well [63]. However, the exact mechanism of RET involvement in intratumoral angiogenesis is not fully understood. It is suggested to be via recruitment of pro-angiogenic cytokine factor [56]. Some clinical studies showed that anti-VEGFR-2 and anti-FGFR treatment can downregulate the expression of RET [64].
Notch signaling pathway
Notch signaling comprises cell-cell interaction mediated by membrane bound Notch receptors (Notch-1, Notch-2, Notch-3 and Notch-4) and membrane bound Notch ligands (Jagged-1, Jagged-2, Dll-1, Dll-3 and Dll-4). All Notch receptors (except Notch-3) and ligands (except Dll-3) are expressed on the outer surface of EC [65]. Notch signaling is upregulated by VEGF activation and mediates tip-to-tip interaction between ECs and vascular stalk sprouting/formation. In other words, Notch signaling mediates the three dimensional awareness of ECs within the newly formed blood vessels. Reciprocally, Notch signaling down-regulates VEGFR-1 and VEGFR-2 [66]. It is worth noting that Dll-4 is highly expressed within different human intratumoral blood vessels [67]. Experimentally, Notch1 antibody and soluble Notch1 receptor (decoy receptor) exert significant anti-angiogenic and antitumor effects against different xenografts models [50]. Notch signaling pathway is physiologically essential during development as Notch-1 homozygous knockout mice results in embryonic fatality [68]. In addition, prolonged exposure to anti-Dll-4 induced abnormal hepatic pathological features and vascular neoplasia in different animal species (mice, rats and monkeys) [69]. Accordingly, it might not be appropriate to depend on the anti-Notch strategy for anti-angiogenic drug development.
Ephrins/Eph receptors pathway
Similar to Notch signaling, ephrins are a family of 9 different membrane bound ligands that mediate cell-cell interaction via coupling with their corresponding Eph tyrosine kinase receptors. Interestingly, Ephrin/Eph signaling is bidirectional in both cell-harboring ligands and cell-harboring receptor [50]. Amongst, ephrinA1/EphA2 and ephrinB2/EphB4 are of special interest in embryonic vasculogenesis, arteriogenesis (arterio-venous anastomoses) and intratumoral angiogenesis [70]. Besides, ephrinA1 was found to be overexpressed in response to elevated VEGF signaling [71]. In addition, several tumor cell types express EphB4, which further activates ECs-expressing ephrinB2. Backward signaling within ECs further promotes the expression of VEGFR-2 and EC tip guidance [72].
Integrins
Integrins are heterodimeric functional extracellular matrix (ECM) signaling peptides composed of several α- and β-subunits. They mediate the cross talk between cells and ECM components, such as fibrinogen, fibronectin and vitronectin, via argenine-glycine-aspartate residue (RGD motif) [73]. Integrins α5β1, αvβ3 and αvβ5 are the most abundant integrins within ECs. They are upregulated during intratumoral angiogenesis [50]. Integrin-αvβ3 mediates FGF-2- and (tumor necrosis factor-α) TNF-α-induced angiogenesis, while integrin-αvβ5 mediates VEGF- and TGF-α-induced angiogenesis [74]. Amongst all integrins, αvβ3 is the most frequently targeted due to high potential as angiogenic factor [50].
MCAM (CD146)
Melanoma cell adhesion molecule (MCAM) is a newly identified VEGFR-2 co-receptor which was found to be overexpressed in wide range of tumor-associated ECs [75]. Although MCAM interacts with several ECM components such as laminin-411, it is believed not to be involved in critical physiological function [76], [77]. Pro-angiogenic effects such as ECs’ proliferation and migration of MCAM were found to be mediated via the interaction with netrin-1 (neuronal guidance protein) [78].
VE-cadherin
VE-cadherin is an endothelial cell-specific-homo-dimeric adhesion molecule that facilitates the formation of cell/cell adherent junctions [79]. VE-cadherin is upregulated and the human VE-cadherin promoter region is highly activated during tumor angiogenesis [80]. Experimentally, several decoy VE-cadherin domains inhibited HUVEC tube formation and possessed considerable anti-tumor activity in xenograft models [81].
TEM8/ANTXR1
Tumor endothelial marker-8 (TEM8) is an anthrax toxin receptor (ANTXR1) expressed on the intratumoral ECs. TEM8 interacts with the α3 subunit of collagen VI and is suspected to elicit angiogenic effects and tumor progression [82]. TEM decoy receptor showed anti-angiogenic and subsequently antitumor effects against xenograft model [50].
Cytokines
Transforming growth factor-β (TGF-β) super-family comprises more than 30 different growth factors. Amongst, three TGF-β isotypes (TGFβ-1, TGFβ-2 and TGFβ-3) which were found to be of special interest for ECM deposition and integrin expression to promote wound healing, ECs proliferation and migration and vascular lumen formation [83]. Besides, the expression of dimeric co-receptor for TGF-β, endoglin (CD105) on adult ECs as well as on the proliferating intratumoral ECs was detected. Exposure to hypoxia or VEGF-blockade induces overexpression of endoglin [50], [84].
The inflammatory cytokine TNF-α is believed to possess angiogenic effects directly via promoting ECs differentiation and indirectly via promoting the secretion of other angiogenic factors [85].
Semaphorins/plexins
Semaphorins (Sema) are secreted proteins that are implicated in neuronal development and immunologic functions. Class 3 semaphorins such as, Sema-3A, Sema-3C and sema-3F, are secreted from the intratumoral ECs and possess autocrine pro-angiogenic and tumor progression effects [86], [87]. In addition, Sema-4D possesses a pathological intratumoral pro-angiogenic effect via Plexin-B1 receptors [88].
Rho-J
Rho-J is an endothelial-expressed Rho-GTPase member of cell division cycle protein-42 (Cdc42). It interacts with cellular cytoskeleton proteins such as actin. Rho-J was found important for ECs focal adhesion, motility, tubulogenesis, and lumen formation during angiogenesis [89].
CLEC14A
CLEC14A is a newly identified specific intratumoral endothelial marker which is overexpressed in a wide range of intratumoral vasculature [90]. CLEC14A interacts with a specific endothelial ECM component, multimerin-2 (MMRN2), and elicits intratumoral angiogenic features which is associated with tumor progression [91].
Anti-angiogenic drug families for cancer treatment
Since the implementation of anti-angiogenesis as a strategy in treating cancer, a long list of chemical synthetic moieties, natural compounds, macromolecules and even treatment modalities have been suggested under this mechanism (Table 1).
Table 1.
List of FDA-approved anti-angiogenic agents.
| Anti-angiogenic agent | Targeted pathway | Clinical indications | Ref. |
|---|---|---|---|
| Bevacizumab | Humanized monoclonal anti-VEGF-A antibody | Several solid tumors such as, non-small cell lung cancer, renal cell cancer, colorectal cancer, ovarian cancer, breast cancer, cervical cancer and glioblastoma | [88] |
| Ziv-aflibercept | Fusion protein directed against VEGF-A, VEGF-B and PlGF | Metastatic colorectal cancer in combination with 5-FU, irinotecan and leucovorin | [92] |
| Sorafenib | Multi-tyrosine kinase inhibitor | Hepatocellular carcinoma, renal cell carcinoma, thyroid carcinoma | [93] |
| Sunitinib | Multi-tyrosine kinase inhibitors | Hepatocellular carcinoma, renal cell carcinoma, thyroid carcinoma | [94] |
| Axitinib | Receptor tyrosine kinase inhibitor | Advanced renal cell carcinoma | [52] |
| Nintedanib | Receptor tyrosine kinase inhibitor | Idiopathic pulmonary fibrosis | [52] |
| Regorafenib | Receptor tyrosine kinase inhibitor | Metastatic colorectal cancer, gastrointestinal stromal tumor and hepatocellular carcinoma | [52] |
| Pazobanib | Receptor tyrosine kinase inhibitor | Advanced renal cell carcinoma, advanced soft tissue sarcoma | [52] |
| Cabozantinib | Receptor tyrosine kinase inhibitor | Metastatic medullary thyroid cancer | [52] |
| Vandetanib | Receptor tyrosine kinase inhibitor | Medullary thyroid cancer | [52] |
| Thalidomide | Inhibitor of Akt phosphorylation | Multiple myeloma in combination with dexamethasone | [95], [96] |
Monoclonal antibodies
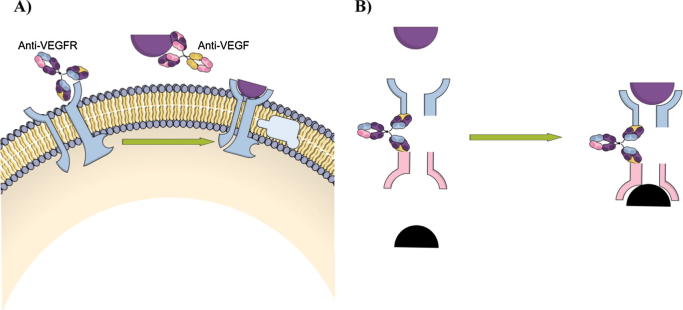
Monoclonal antibodies synthesized to target specific ligands or receptors involved in angiogenesis would be the most straightforward approach to neutralize specific pathogenic pathways. The use of protein-based drugs is technically challenging; however, many monoclonal antibody-based drugs succeeded and got approved by the FDA (Fig. 2A). Another challenge for any highly specific mono-targeting anti-angiogenic agent is the development of molecular bypass utilizing another pro-angiogenic pathway of angiogenesis.
Fig. 2.
Diagrammatic illustration for the interaction between monoclonal antibodies with pro-angiogenic ligand or receptor (A) and the interaction between decoy receptor and soluble pro-angiogenic ligand (B). Designed using Mind The Graph™, Zendesk Inc., San Francisco, CA, USA.
Bevacizumab
Bevacizumab (Avastin®) is the prototype humanized monoclonal anti-VEGF-A antibody used clinically for several solid tumors, such as non-small cell lung cancer, renal cell cancer, colorectal cancer, ovarian cancer, breast cancer, cervical cancer and glioblastoma. Bevacizumab prolonged the progression free survival and overall survival rate in all of these tumor types except breast cancer [56]. It was approved by the FDA in 2004 [97]. Rapid resistance to bevacizumab appeared via the development of non-VEGF pathways for angiogenesis. Bevacizumab failed to provide any survival benefit to EGFR-2 negative breast cancer patients and the FDA withdrew the approval for its use in metastatic breast cancer in 2011 [98].
Ramucirumab and IMC-18F1
Instead of blocking the ligand (as in bevacizumab), ramucirumab is a humanized monoclonal anti-VEGFR-2 antibody which selectively binds the extracellular domain of VEGFR-2. Due to the conflicting results of phase-III trails showing a non-significant clinical improvement in hepatocellular carcinoma patients, it has not yet been approved by the FDA [92]. On the other hand, IMC-18F (recombinant monoclonal anti-VEGFR-2 antibody) has been approved for non-small lung, gastric and metastatic colorectal cancers [99].
Other monoclonal antibodies
Many other humanized monoclonal antibodies targeting particular ligand/receptor involved in angiogenesis are being under consideration for treating malignancies, such as cetuximab (anti-EGFR antibody), volociximab (anti-integrin-αvβ1 monoclonal antibody), etaricizumab or vitaxin (anti-integrin-αvβ3 antibody), MEDI3617 or REGN910 (anti-Ang-2 antibody) and GAL-F2 (anti-FGF-2 antibody) [50]. Many of these were mainly designed to augment or overcome resistance to anti-VEGF pathway antibodies. Another very interesting bi-specific anti-Ang2/anti-VEGF antibody is currently under development; yet, it might be less likely to develop resistance to anti-VEGF therapy [100].
Decoy receptors/fusion peptides
Decoy receptors are soluble form of certain membrane bound receptors that compete with the original membrane-bound receptors with the same affinity to their ligand (Fig. 2B). This competition results in suppressing the downstream signaling of the membrane-bound receptors
Ziv-aflibercept (Zaltrap®)
It is a complicated fusion protein composed of the extracellular domain of both VEGFR-1 and VEGFR-2 linked together via Fc-tag segment. Ziv-aflibercept binds to VEGF-A, VEGF-B and PlGF, prohibiting their interaction with VEGFR and inhibiting angiogenesis [101]. Zaltrap® was recently approved by the FDA for metastatic colorectal cancer in combination with 5-FU, irinotecan and leucovorin [102].
Trebananib
It is biologically-active peptide fused to Fc-tag segment and designed to interrupt the interaction between both Ang-1 and Ang-2 with Tie-2 receptors, leading to anti-angiogenic response [103]. Clinical trials for trebananib against hepatocellular carcinoma have been halted due to lack of efficacy [104]. On the other hand, clinical trials against ovarian cancer are still undergoing despite several disappointing outcomes [105].
Other decoy receptors/fusion peptides
Many anti-angiogenic decoy receptors are being used to suppress intratumoral angiogenesis. FGFR-2 extracellular domain fused to Fc-tag peptide can act as a trap for FGF-2, preventing FGFR-2 downstream signaling [106]. Similarly, Dll4 fused to Fc-tag was used to interrupt the interaction between Notch receptors and Dll4 ligands and to elicit an anti-angiogenic response [107]. In addition, EphA2 extracellular domain fused to Fc-tag interrupted EphA2/ephrinA1 interaction and inhibited angiogenesis experimentally in an in vivo xenograft model [108]. Some peptides, such as Annexin-A2 (Anx-A2), are known for their regulatory effects against VEGF-dependent angiogenesis. Purification of peptides such as Anx-A2, is currently under investigation to inhibit angiogenesis [93], [94].
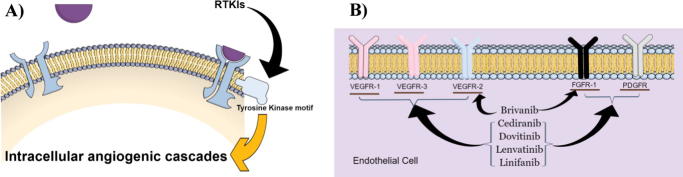
Receptor tyrosine kinase inhibitor small molecules (RTKIs)
These are the most rationalized and common anti-angiogenic agents for cancer therapy. Originally, the first RTKI was designed in 1996 to inhibit VEGFR intracellular tyrosine kinase activity and elicit an anti-angiogenic response [109]. Computational chemistry was used to design such small molecules. Indeed, these RTKI’s inhibit the enzyme activity of the tyrosine kinase motif attached to the intracellular domain of a wide range of receptors involved in angiogenesis, such as VEGFR, FGFR, PDGFR, Tie receptors, RET, c-MET and Eph receptors. RTKI might be specific for one receptor-bound intracellular tyrosine kinase domain or non-specific to cross-react with more than one of the aforementioned receptors (also called dirty tyrosine kinase inhibitor). Non-specific RTKIs can be prescribed as mono-therapy due to its multiple targets [110]. In fact, it is less common to develop resistance to multi-kinase inhibitors as they are covering more than one pathway involved in angiogenesis. However, resistance to more than one RTKI was also reported (Fig. 3A).
Fig. 3.
Diagrammatic sketch for the molecular bases of RTKI’s action (A) and example of the versatile interaction between different investigational new RTKIs and pro-angiogenic receptors (B). Designed using Mind The Graph™, Zendesk Inc., San Francisco, CA, USA.
FDA approved RTKIs
The FDA has approved many RTKIs for the treatment of several solid tumors. Sorafenib and sunitinib are multi-tyrosine kinase inhibitors targeting VEGFR-1, VEGFR-2, VEGFR-3, PDGFR-β and RET receptors. Sorafenib is approved for the treatment of hepatocellular carcinoma, renal cell carcinoma and thyroid carcinoma [111]. Sunitinib is approved for the treatment of gastrointestinal stromal tumor and renal cell carcinoma [112]. Other RTKIs such as axitinib, nintedanib, regorafenib, pazobanib, cabozantinib and vandetanib inhibit various angiogenic mediating receptors. RTKIs have also been approved by the FDA for different types of solid tumors such as, advanced renal cell carcinoma, metastatic medullar thyroid cancer, soft tissue sarcoma, non-small cell lung cancer, metastatic colorectal cancer, gastrointestinal stromal tumor and hepatocellular carcinoma [56].
Investigational RTKIs
Due to the success of many approved RTKIs, intensive research is being carried out on other RTKIs to be approved for other solid tumor types. Similar to other RTKI’s, the new agents are inhibiting more than one angiogenic receptor (Fig. 3B). Amongst, brivanib, cediranib, dovitinib, lenvatinib and linfanib are undergoing or completed phase-III clinical trials against several tumor types such as hepatocellular carcinoma, metastatic colorectal cancer, advanced non-small lung cancer, metastatic renal cell carcinoma and thyroid cancer [113], [114], [115], [116], [117], [118].
Non-RTKI anti-angiogenic small molecules
The vast majority (if not all) of natural products and their derivatives belongs to this group. This group of compounds was discovered via conventional drug screening procedures; later on, these compounds were found to possess anti-angiogenic activity. In many cases, the exact molecular bases for their anti-angiogenic activity are not fully understood. Besides, some small molecules were designed and synthesized to interrupt a particular pathway essential for angiogenesis in addition to inhibiting the tyrosine kinase motif.
Cilengitide
This cyclic RGD complex was designed to interfere with the effect of integrin-αvβ3 and integrin-αvβ5 in promoting intratumoral ECs and angiogenesis [95]. Despite its promising preclinical results, clinical trials against glioblastoma were disappointing and failed to add any significant benefits to patients [96].
Thalidomide and analogues
The old anti-emetic agent, thalidomide, which caused a teratogenic disaster in the middle of the last century was repositioned to be used as anti-angiogenic agent [119]. Thalidomide inhibits the phosphorylation of Akt which is crucial for the downstream signaling of wide range of growth factors such as, VEGF, FGF-2 and hypoxia inducible factor-α (HIF-α) [120]. Due to the severe side effects of thalidomide (teratogenicity, thromboembolism, pancytopenia, neuropathy and tremors), several analogues, such as lenalidomide and pomalidomide are under investigation [120]. In general, thalidomide has not shown any significant anti-angiogenic effect as a monotherapy; however, it has been approved by the FDA to be used in combination with dexamethasone against multiple myeloma [121].
Combretastatin (CA4-P)
Combretastatins are naturally occurring stilbenes isolated from the South African hush pillow, Combretum caffrum [122]. CA4-P is known as a VDA that induces intratumoral ECs’ killing effect as rapidly as within 4 h of administration. It is believed that CA4-P interferes with the microtubular structure of the intratumoral ECs [123], [124]. Clinical trials showed beneficial effects for combining CA4-P with chemotherapeutics in treating platinum-resistant ovarian cancer and anaplastic thyroid tumors [125]. However, CA4-P is not yet clinically approved due to its unwanted adverse effects, which warrant further clinical trials to determine its risk/benefit ratio [126], [127].
Vadimezan (DMXAA, ASA-404)
DMXAA is a synthetic oxygenated xanthone compound with an excellent preclinical profile as an anti-angiogenic agent/VDA. Its mode of action is not fully understood; it is assumed to target the TBK-1/IRF-3 interferon pathway [128]. However, the clinical output of DMXAA was disappointing and failed to provide any significant survival benefit for patients in clinical trials [129].
Vinblastine (VBL) and vincristine (VCR)
Vinca alkaloids are natural compounds derived from Catharanthus roseus. VBL and VCR are well-established anti-cancer agents with a well-known tubulin spindle stabilizing mode of action. Their anti-angiogenic effect is believed to be via inhibiting ECs motility, proliferation and migration attributed to their cellular spindle interfering activity [130], [131].
Paclitaxel
Taxanes are well-recognized naturally occurring anti-cancer drugs; they are originally isolated from the tree Taxus brevifolia and kill tumor cells by interfering with their mitotic tubulin spindles [132]. During the last decade, taxol was found to possess very potent anti-angiogenic activity with much lower doses than its cytotoxic dose. Metronomic therapy (continuous treatment with very much lower dosing) with taxol induced promising anti-angiogenic effects and overall clinical anti-tumor response [131], [133].
Curcumin/ferulic acid
Curcumin is naturally occurring ferulic acid-based polyphenol found in plants such as Curcuma longa. Curcumin possesses a wide range of anticancer pleiotropic effects as well as anti-angiogenic effects [134]. There is no comprehensive understanding of the anti-angiogenic mechanism of curcumin. It might be attributed to the EC-killing effect of the curcumin metabolites, ferulic acid and vanillin [135].
Resveratrol
Resveratrol is a stilbene phytoalexin-based polyphenol with a wide range of pleiotropic health effects; it is found in several edible fruits, berries and nuts. It is the hallmark compound of the French paradox and several studies showed evidence for its chemopreventive, chemotherapeutic and chemomodulatory effects [136], [137]. Resveratrol inhibits ECs proliferation and matrix metalloproteinase-2 (MMP-2) activity required for ECM remodeling and angiogenesis. Besides, resveratrol interrupts VEGF-dependent angiogenesis via inhibiting src kinase and subsequent VE-cadherin phosphorylation [134]. Most interestingly, resveratrol inhibits HIF-1α accumulation, VEGF secretion and VEGFR-2 phosphorylation [52].
Carnosic acid/carnosol
Carnosic acid and carnosol are polyphenolic compounds found in Rosmarinus officinale L. They show an anti-angiogenic response, which is believed to be attributed to direct effect on ECs. Carnosic acid and carnosol inhibit ECM remodeling via inhibiting MMP-2; inhibit proliferation, migration and invasion; and induce apoptosis in ECs [138].
Quercetin
Quercetin belongs to the flavonol family of natural compounds and is found in a wide range of edible plants, such as onions, raspberries, grapes, cherries and leafy plants. Flavonols such as quercetin possess strong anti-angiogenic effects via inhibiting/downregulating the expression of VEGF, MMP’s, NFκB and FGFR signaling [139]. Specifically, quercetin was found to inhibit the VEGFR-2-dependent akt/mTOR pathway in an experimental xenografts model [134], [140].
Genstein
Similar to quercetin, the phytoestrogen genstein is a naturally occurring isoflavone abundant in soy beans (Glycine maxima L.). Genstein down-regulates the expression of several tyrosine kinases which that play essential roles in a wide range of pro-angiogenic pathways. Besides, it down-regulates several ECM remodeling enzymes such as, MMPs [141], [142].
Other natural compounds
Many other natural products showed preliminary experimental and occasional clinical evidence for anti-angiogenic effects, as summarized in Table 2 [52], [134], [143], [144], [145].
Table 2.
Compounds of natural origin with preclinical evidence for anti-angiogenic effects [52], [134], [143], [144], [145].
| Anti-angiogenic agent | Natural source | Anti-angiogenic agent | Natural source |
|---|---|---|---|
| Allin | Allium sativum | Barbaloin, emodin | Aloe vera, Rheum palmatum |
| Apigenin | Apium graveolens | Artemisinin | Artemisia annua |
| Berberine | Berberis vulgaris | Senegin-II, Senegin-III, Senegin-IV, Senegasaponin-a and Senegasaponin-b | Polygala senega |
| Ginkgolide B | Ginkgo biloba | Genistein, daidzein | Glycine maxima |
| Isoliquiritigenin, glabridin | Glycyrrhiza glabra | Protocatechuic acid | Hibiscus sabdariffa |
| Isorhamnetin | Hippophae rhamnoides | Floroglucin | Hypericum perforatum |
| Melatonin | Juglans region | Magnosalin, honokiol, magnolol | Magnoliae spp. |
| Apigenin, fiseti | Matricaria chamomilla | Cortistatins J, K, and L | Marine Sponge Corticium simplex |
| Ponicidin, oridonin | Rabdosia rubescens | Cryptotanshinone | Salvia miltiorrhiza |
| Capsaicin | Capsicum spp. | Baicalein, baicalei | Scutellaria baicalensis |
| Silymarin | Silybum marianum | Parthenolide | Tanacetum parthenium |
| 6-Gingerol | Zingiber officinale | Sanguinarine | Sanguinaria Canadensis |
| Betulinic acid | Prunus dulcis | Pyripyropenes A, B and D | Marine-Derived Fungus of Aspergillus sp. |
| Globostellatic Acid X Methyl Esters | Marine Sponge Rhabdastrella globostellata | Bastadin-6 | Marine Sponge Ianthella basta |
| Aeroplysinin-1 | Marine sponge Phylum Poriphera |
Anti-angiogenic vaccines
Vaccination is the traditional method to eradicate any human disease. Theoretically, vaccines developed against intratumoral ECs are preferred than those developed against tumor cells due to two main reasons. First, the better exposure of ECs to bloodstream and blood born immune cells; in contrast to tumor cells which are most probably deeply hidden and far of reach from immune T-cells. Secondly, ECs possess significantly less genetic instability than tumor cells. Surface epitopes of intratumoral ECs are less frequent to be mutated compared to tumor cells [146]. Yet, some studies showed some kinds of genetic instabilities for intratumoral ECs [147]. Breast cancer ECs and hepatocellular carcinoma ECs showed acquired resistance to chemotherapies such as vincristine and 5-FU/doxorubicin, respectively [148], [149], indicative of genetic instability. On the other hand, it is very challenging to find the antigenic components exclusively expressed on intratumoral ECs when they are not abundant on normal ECs. Cross-reaction of anti-intratumoral EC vaccines with normal blood vessels’ ECs might be devastating and can induce a long list of pathological features from impaired wound healing to autoimmune diseases [150]. However, some vaccines are suggested to target a particular ligand or receptor involved in angiogenesis and showed sort of experimental or even clinical beneficial outcomes.
Despite its importance in hematopoiesis, anti-VEGF vaccine was designed and elicited promising anti-angiogenic anti-tumor effects against several tumor xenograft models. Some anti-VEGF vaccines (CIGB-247: VEGF variant/bacterial adjuvant vaccine) showed reasonable tolerability in phase-I clinical trials with minimal hematopoietic or wound-healing impairments [151]. In addition, DNA and peptide based vaccines against VEGFR-2 showed potent humoral- and cellular-based anti-vascular immunity with subsequent anti-angiogenesis, anti-tumor and anti-metastatic responses [152], [153]. These vaccines showed negligible adverse effects against wound healing and embryogenesis [153]. Interestingly, a specific intratumoral VEGFR-2 peptide (VEGFR2-169 peptide) was identified to elicit a selective T-cell cytotoxic response against intratumoral ECs. In a phase-I clinical study against pancreatic cancer, it was well tolerated in combination with gemcitabine and showed no severe adverse effects [154].
Apart from VEGF pathway targeting and its high risk for cross reaction with normal vasculature, several vaccines are investigated for other angiogenic cues. Vaccines against FGF-b, FGFR-2, endolgins, PDGFR-β, TEM-1 and TEM-8 showed considerable anti-angiogenic/anti-tumor effects with minimal adverse effects regarding wound healing and embryogenesis in experimental models [146]. The neonatal lung peptide, HP59, in particular was found overexpressed in the intratumoral vasculature of wide range of solid tumors (lung, colon, ovary and breast) but was not detected in normal vasculature. Vaccines against HP59 suppressed tumor growth of Lewis lung cancer model due to anti-angiogenic effect [155]. Interestingly, clinical evidence showed a significant survival benefit for patients with non-small cell lung cancer who receive anti-EGF vaccine (CIMAvax®) made of recombinant human EGF linked to Neisseria meningitides carrier proteins [156]. CIMAvax® did not induce any significant pathological effects against normal blood vessels nor wound healing process [157].
Drug interaction with anti-angiogenic agents
According to many clinical trials, selective anti-angiogenic remedies (Uni-targeted), such as bevacizumab, are not recommended to be used as monotherapy. One the other hand, multi-kinase inhibitors which target more than one pro-angiogenic pathway could be used as monotherapy [158]. In the current section, the importance, and therefore the inevitability of using anti-angiogenic agents in combination with other classic chemotherapies or with other anti-angiogenic drugs will be discussed. During the early phases of solid tumor exposure to anti-angiogenic treatment, the intratumoral vasculature undergoes what is called vascular normalization. Vascular normalization is accompanied by improved intratumoral perfusion characteristics and better delivery of nutrients, oxygen and chemotherapeutic agents themselves to deeper micro-regions of solid tumors [159]. Further exposure of solid tumors to anti-angiogenic drugs results in vascular shutdown and tumoral tissue necrosis, which is severe but typically restricted to the central part of the tumor, leaving a rim of viable tumor cells [160] that presumably receive their nutritional requirements from nearby normal blood vessels [161]. Tumor cells of the peripheral surviving rim are properly blood-perfused compared to the central compartment of the solid tumor. Yet, it is suggested that the most potent VDA will be unable to eradicate tumor cells based solely on cutting down its blood supply [162]. Besides, time span between anti-angiogenic induced vascular normalization and the ultimate vascular shutdown is called normalization window. The duration of this normalization window differs for each anti-angiogenic agent and represents the intratumoral drug delivery “honeymoon” for optimum intratumoral drug accumulation. This normalization window starts as early as 4 h after delivery of some anti-angiogenic VDA’s such as, combretastatin [163].
Pharmacokinetic drug interaction with anti-angiogenic agents
The two major effects of anti-angiogenic agent on intratumoral vasculature are sequential vascular normalization followed by vascular shutdown. These effects presumably influence intratumoral drug delivery and entrapment, respectively. Pharmacokinetic improvement attributed to anti-angiogenesis is not only due to direct enhancement of vascular perfusion but also due to a decrease in the intratumoral interstitial fluid pressure [164]. Intratumoral drug distribution starts with initial accumulation of drug nearby intratumoral blood vessels due to its enhanced permeation and retention (EPR) effect. Later on, the drug needs to diffuse passively though the crowded avascular tumor parenchyma; this process is challenged by the elevated interstitial fluid pressure within tumor micro-regions [165]. Temporal design of the sequence and time lapse between administering an anti-angiogenic agent and its combined chemotherapy is crucial for optimum anticancer outcomes [166].
Pre-administration of bevacizumab (Avastin®) 1–3 days before topotecan/etoposide administration to neuroblastoma patients, induced vascular normalization (decreased vessel density and improved tumor perfusion) and enhanced tumor uptake of topotecan and etoposide. However, simultaneous co-administration the same drugs or after a 7-days lapse did not improve tumor uptake for topotecan or etoposide within the same tumor type [167]. Similarly, bevacizumab enhanced doxorubicin intratumoral uptake of hepatocellular carcinoma after pre-treatment (3–5 days earlier). However, pre-administration of bevacizumab one day earlier; or 7 days before doxorubicin administration, did not induce any significant improvement of doxorubicin tumoral uptake [168].
Pharmacodynamic drug interaction with anti-angiogenic agents
Combination therapy (or cocktail therapy) is a general principle for chemotherapeutic treatment protocols. This is primarily to avoid potential resistance attributed to the high rate of genetic variability in tumor cells. During the early phase of anti-angiogenic drug discovery, it was believed that ECs are not as genetically unstable as tumor cells. Yet, targeting a specific angiogenesis pathway (such as VEGF) was expected be selective enough to eradicate intratumoral blood vessels with minimal adverse effects [50], [169]. The discovery of non-VEGF angiogenesis pathways and its potential for circumventing VEGF-dependence resulted in treatment failure of mono-therapies of selective mono-targeted monoclonal antibodies [158]. On the other hand, some non-selective (multi-targeted) RTKIs succeeded in some clinical trials as a monotherapy [110].
Therapeutic exposure of solid tumors to anti-angiogenic agent would elicit vascular shutdown and tumoral tissue necrosis, particularly to the central part of tumor micro-regions, leaving a rim of viable tumor cells [160]. Tumor cells in the peripheral rim of tumor micro-regions are properly blood perfused. Yet, this suggests a logical pharmacodynamic-based rationale for the need to combine anti-angiogenic agents with chemotherapy or radiation to achieve full tumor cell eradication [162]. Many reports showed superior antitumor response to combination regimens containing an anti-angiogenic agent with a classic chemotherapeutic drug [170]. Combination between anti-angiogenic agents with radiation seemed very promising. Leftover tumor cells after successful intratumoral shutdown survive mainly due to proper blood perfusion using normal surrounding tissues’ blood vessels. Radiation mainly targets well-perfused and properly oxygenated tumor regions (such as the remaining viable rim) and suffer resistance from the poorly perfused micro-regions [171], [172].
Resistance to anti-angiogenic treatment
Neoplastic cells are known for their high rate of genetic instability and mutations compared to non-malignant cells. Yet, vascular ECs possess much less potential to develop resistance compared to tumor cells. This was the major rationale behind implementing the anti-angiogenic therapeutic strategy. Accordingly, it was assumed that targeting the normal ECs lining tumor blood vessels would be an appropriate way to tackle cancer with minimal risk of developing resistance [173], [174]. However, it was found that resistance to anti-angiogenic therapy can be developed as rapidly as for other conventional therapies. Also, the beneficial effects of anti-angiogenic monotherapy to control tumor progression lack satisfactory sustainability [175], [176], [177]. Unfortunately, it became clear that resistance to angiogenic therapy is major obstacle for cancer treatment options. Resistance to angiogenic therapy is widely believed to be mediated by several mechanisms that fall into two main categories; adaptive and intrinsic resistance [178].
First, adaptive resistance to anti-angiogenic treatment is an indirect method of resistance adopted by malignant cells to cope with blood supply shortage caused by anti-angiogenic therapy. This type of resistance is considered functional compensation of the target inhibited by anti-angiogenic therapy utilizing several molecular mechanisms [178]. Induction of tumor hypoxia attributed to anti-angiogenic therapy seems to upregulates several independent pathways implicated in restoring angiogenesis [179], [180]. In a study conducted in a genetic mouse model of neuroendocrine cancer, it was observed that blocking the VEGF pathway by administering anti-VEGF monoclonal antibody resulted in upregulation of several pro-angiogenic factors, such as FGF-2 [179]. Accordingly, this up-regulation was associated with an increase in intratumoral angiogenesis. Besides, increased plasma levels of FGF-2 were observed in patients treated with the VEGFR inhibitor cediranib [181]. Induction of hypoxia by anti-angiogenic therapy could also elicit a cellular response that might eventually lead to neo-vascularization within tumor micromilieu. Besides, it appears to attract bone marrow-derived endothelial progenitor cells, pericytes and other growth factor-secreting cells to the site of neoplastic lesion in order to further promote tumor angiogenesis [182], [183], [184]. In alignment with experimental studies, clinical evidence suggests that therapy-induced hypoxia seems to promote accumulation of bone marrow-derived progenitor cells around malignant tumors [185]. In addition to the cellular response mediated by bone marrow-derived progenitor cells, pericytes are also implicated in promoting resistance to anti-angiogenic therapy. It was found that tumor blood vessels densely enveloped by pericytes are relatively resistant to anti-angiogenic therapy [186]. Supportive cells are believed to confer resistance to ECs by dual mechanisms. It is believed that pericytes influence negative regulation on endothelial cell proliferation, rendering them quiescent and less responsive to anti-angiogenesis therapy [187]. In addition, these cells offer an alternative signaling pathway to functionally support ECs targeted by anti-VEGF therapy [188], [189]. Therefore, targeting both pericytes and ECs appears to be an effective strategy to guard against the development of anti-angiogenesis resistance. Furthermore, malignant tumor cells could adapt to shortage in blood supply by increasing their invasive potential. Several types of malignant tumors respond to anti-angiogenesis therapy by invading nearby normal tissues to exploit their blood supply [190], [191]. This pathological phenomenon is known in some types of cancer as perivascular tumor invasion, which is believed to be provoked to counteract the inhibition of angiogenesis [178].
The second type of anti-angiogenesis resistance is intrinsic resistance. In this type of resistance, tumors are inherently unresponsive to anti-angiogenic therapy, and patients do not show any sign of clinical or histological response [192]. Intrinsic resistance to anti-angiogenic therapy can be derived from the intrinsic pattern of tumor growth. In pancreatic ductal adenocarcinoma, tumor cells grow and proliferate originally in a hypoxic environment with little or no vascularity [193]. In stage II and III astrocytoma tumors, cells proliferate in an invasive, hypovascularized manner [194]. These malignant tumors with their unique growth patterns are intrinsically unresponsive to anti-angiogenic therapy, as they originally lack the demand for angiogenesis [178]. Other mechanisms could account for this type of resistance, such as compensatory activation of angiogenic receptors by other factors that are not targeted by the anti-angiogenic therapy. Breast cancer tumors only respond to bevacizumab at the early stages of tumor progression [195]. Late-stage tumors usually secrete several growth factors, such as FGF-2 with superior ability to activate VEGF receptors beyond the inhibitory capacity of bevacizumab [196]. Yet, these alternative factors are derived not only from tumor cells but also from the pre-existing immune cells [177].
Finally, there are several well-established and potential molecular mechanisms that could account for both types of resistance. The effects of these mechanisms of resistance on the response to anti-angiogenic therapy are clear, which is reflected on the transient clinical response to angiogenic treatment [197]. Further research is required to investigate the clinical significance of targeting these molecular pathways and also to elucidate additional pathways implicated in this type of resistance.
Conclusions and future perspectives
Targeting intratumoral vasculature as an anticancer treatment strategy is rapidly and successfully emerging due to continuous uncovering of different molecular cues of angiogenesis. Despite reports for some kinds of genetic instability in intratumoral ECs, they are far more genetically stable than tumor cells [146]. Folkman expected that killing one EC would elicit nutritional deprivation capable of killing 100–300 tumor cells [198]. In addition, vascular targeting agents positively influence the anticancer activity of conventional chemo-radiotherapies when properly orchestrated together. Accordingly, several success stories are expected in the near future for anti-angiogenic agents against cancer. Among these, the use of multi-targeted RTKI and decoy receptors are considered the most promising approaches for anti-angiogenic drug discovery. Combination of antiangiogenic agents opens a new horizon for better efficacy and less toxicity of anticancer drugs [199], [200], [201]. On the other hand, anti-angiogenic vaccines seem to have weak potential for the development of marketed drugs due to the excessive risk of debilitating adverse effects, such as autoimmune diseases.
Conflict of interest
The authors have declared no conflict of interest.
Compliance with Ethics Requirements
This article does not contain any studies with human or animal subjects.
Biographies

Ahmed M Al-Abd has graduated (2000) from Faculty of Pharmacy, Ain Shams University; M.Sc. in pharmacology and toxicology (2004-Ain Shams University) from Faculty of Pharmacy; Ph.D. fellow in biomedical sciences (year 2008 – the Catholic University of Republic of Korea); Ph.D. degree in pharmacology and toxicology (year 2011 – Beni-Suif University, Egypt). Since graduation, He has been promoting in the fields of research/academia from research assistant, to a researcher in the National Research Center of Egypt. He is currently an associate professor at King Abdulaziz University and visiting scholar in Bouvé College of Health Sciences, Northeastern University, USA. Ahmed Al-Abd is a co-author of more than 35 peer-reviewed publications and co-inventor of 2 patents. Ahmed Al-Abd has a broad international scientific communication network (collaborator with more than 15 Universities and Research institutes in more than 5 different countries worldwide).

Abdulmohsin J. Alamoudi has graduated in 2013 with a Pharm D degree (Doctor of Pharmacy) from Faculty of Pharmacy, King Abdulaziz University and got his M.Sc. degree (2016) from Imperial College London, London, UK. He is working as a lecturer and a research assistant in Pharmacology and Toxicology Department, Faculty of Pharmacy, King Abdulaziz University, Jeddah, Saudi Arabia. Besides, He was awarded in the scientific forum for the students of King Abdulaziz University and in the nationwide scientific forum of Saudi Arabia for students of higher education.

Ashraf B. Abdel-Naim was graduated (1984) from Faculty of Pharmacy, Cairo University. He obtained a Diploma of Drug Analysis and Bioassay (1987, Al-Azhar University), M.Sc. in Pharmacology (1990, Al-Azhar University), a membership of the Ph.D. channel system “Exchange Visitors” between Al-Azhar University, Egypt and the University of Texas Medical Branch at Galveston, USA (1992–1994) and Ph.D. degree in Pharmacology (1995, Al-Azhar University, Egypt). In 2004, Abdel-Naim had a post-doctoral mission at University of Nebraska, Omaha, USA. In 2000, Abdel-Naim was the head of Pharmacology and Toxicology Department, Faculty of Pharmacy, Ain Shams University. He has a special interest in the role of natural products in protecting against chemically-induced toxicity as well as bioactivation and mechanisms of biotransformation of chemicals and drugs. Abdel-Naim has co-authored over 80 peer-reviewed publications. He obtained several local and regional scientific grants and received many awards in recognition for his efforts in pharmacological and toxicological research.

Thikrayt Neamatallah, is an assistant Professor in Pharmacology (2014). She got her BSc degree from Faculty of Pharmacy, King Abdulaziz University (2008); MSc degree from the University of Glasgow (2010); PhD degree from the University of Srathclyde (2014). Thikryat Neamatallah is interested in cell signaling pathways that are involved in immune responses, cancer, and vascular complications. She has a wide experience in protein analysis (such as, protein purification, SDS-Gels, Western blots, etc.); immunologic methods (such as, ELISA and flow cytometry), cell culture (Maintenance of submerse and adherent cell cultures, isolation of dendritic cells and macrophages from bone marrow); RNA techniques (isolation, purification, RNA integrity analysis, RT-PCR, Real time PCR); DNA preparation for genotyping; and microarray analysis (labeling and data analysis Using GeneSpring Software).

Osama M. Ashour has graduated (1981) from Faculty of Medicine, Cairo University; M.Sc. in Pediatrics (1987-Faculty of Medicine, Cairo University); M.Sc. in Pharmacology (1989-Faculty of Medicine, Minia University); a member of the Ph.D. channel system “Exchange Visitors” between Minia University, Egypt and the University of Alabama at Birmingham, USA (1991–1996); Ph.D. in Pharmacology (1998, Faculty of Medicine, Minia University, Egypt). Currently, Osama Ashour is a Professor at Faculty of Medicine, Minia University, Egypt and visiting professor at Faculty of Pharmacy, King Abdulaziz University, Saudi Arabia. Osama Ashour has co-authored over 25 peer-reviewed publications by the year 2016.
Footnotes
Peer review under responsibility of Cairo University.
References
- 1.Siegel R.L., Miller K.D., Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66(1):7–30. doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- 2.1950 mortality data – CDC/NCHS, NVSS, mortality Revised. 2002 mortality Data-NVSR-Death final Data 2002-Volume 53, No. 5. Cost data from American Cancer Society Cancer & Figures 2005; 2002.
- 3.Torre L.A., Bray F., Siegel R.L., Ferlay J., Lortet-Tieulent J., Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 4.Al-Abd A.M., Aljehani Z.K., Gazzaz R.W., Fakhri S.H., Jabbad A.H., Alahdal A.M. Pharmacokinetic strategies to improve drug penetration and entrapment within solid tumors. J Control Release. 2015;219:269–277. doi: 10.1016/j.jconrel.2015.08.055. [DOI] [PubMed] [Google Scholar]
- 5.Albini A., Tosetti F., Li V.W., Noonan D.M., Li W.W. Cancer prevention by targeting angiogenesis. Nat Rev Clin Oncol. 2012;9(9):498–509. doi: 10.1038/nrclinonc.2012.120. [DOI] [PubMed] [Google Scholar]
- 6.Zhao Y., Adjei A.A. Targeting angiogenesis in cancer therapy: moving beyond vascular endothelial growth factor. Oncologist. 2015;20(6):660–673. doi: 10.1634/theoncologist.2014-0465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang Z., Dabrosin C., Yin X., Fuster M.M., Arreola A., Rathmell W.K. Broad targeting of angiogenesis for cancer prevention and therapy. Semin Cancer Biol. 2015;35(Suppl.):S224–S243. doi: 10.1016/j.semcancer.2015.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Khalid E.B., Ayman E.E., Rahman H., Abdelkarim G., Najda A. Natural products against cancer angiogenesis. Tumour Biol. 2016;37(11):14513–14536. doi: 10.1007/s13277-016-5364-8. [DOI] [PubMed] [Google Scholar]
- 9.Lenzi P., Bocci G., Natale G. John Hunter and the origin of the term “angiogenesis”. Angiogenesis. 2016;19(2):255–256. doi: 10.1007/s10456-016-9496-7. [DOI] [PubMed] [Google Scholar]
- 10.Folkman J., Shing Y. Control of angiogenesis by heparin and other sulfated polysaccharides. Heparin and related polysaccharides. Adv Exp Med Biol. 1992;313:355–364. doi: 10.1007/978-1-4899-2444-5_34. [DOI] [PubMed] [Google Scholar]
- 11.Swift M.R., Weinstein B.M. Arterial–venous specification during development. Circ Res. 2009;104(5):576–588. doi: 10.1161/CIRCRESAHA.108.188805. [DOI] [PubMed] [Google Scholar]
- 12.Hanahan D., Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996;86(3):353–364. doi: 10.1016/s0092-8674(00)80108-7. [DOI] [PubMed] [Google Scholar]
- 13.Folkman J. Anti-angiogenesis: new concept for therapy of solid tumors. Ann Surg. 1972;175(3):409–416. doi: 10.1097/00000658-197203000-00014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maj E., Papiernik D., Wietrzyk J. Antiangiogenic cancer treatment: the great discovery and greater complexity (Review) Int J Oncol. 2016;49(5):1773–1784. doi: 10.3892/ijo.2016.3709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Baeriswyl V, Christofori G, editors. The angiogenic switch in carcinogenesis. Semin Cancer Biol 2009;19(5):329–37. [DOI] [PubMed]
- 16.Payen V.L., Brisson L., Dewhirst M.W., Sonveaux P. Common responses of tumors and wounds to hypoxia. Cancer J. 2015;21(2):75–87. doi: 10.1097/PPO.0000000000000098. [DOI] [PubMed] [Google Scholar]
- 17.Hashimoto T., Shibasaki F. Hypoxia-inducible factor as an angiogenic master switch. Front Pediatr. 2015;3:33. doi: 10.3389/fped.2015.00033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ribatti D., Nico B., Crivellato E., Roccaro A., Vacca A. The history of the angiogenic switch concept. Leukemia. 2007;21(1):44–52. doi: 10.1038/sj.leu.2404402. [DOI] [PubMed] [Google Scholar]
- 19.Laurenzana A., Fibbi G., Margheri F., Biagioni A., Luciani C., Del Rosso M. Endothelial progenitor cells in sprouting angiogenesis: proteases pave the way. Curr Mol Med. 2015;15(7):606–620. doi: 10.2174/1566524015666150831131214. [DOI] [PubMed] [Google Scholar]
- 20.Kirsch M., Schackert G., Black P.M. Angiogenesis, metastasis, and endogenous inhibition. J Neurooncol. 2000;50(1–2):173–180. doi: 10.1023/a:1006453428013. [DOI] [PubMed] [Google Scholar]
- 21.Nicosia R.F., Tchao R., Leighton J. Interactions between newly formed endothelial channels and carcinoma cells in plasma clot culture. Clin Exp Metas. 1986;4(2):91–104. doi: 10.1007/BF00119076. [DOI] [PubMed] [Google Scholar]
- 22.Bielenberg D.R., Zetter B.R. The contribution of angiogenesis to the process of metastasis. Cancer J. 2015;21(4):267–273. doi: 10.1097/PPO.0000000000000138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Raica M., Cimpean A.M., Ribatti D. Angiogenesis in pre-malignant conditions. Eur J Cancer. 2009;45(11):1924–1934. doi: 10.1016/j.ejca.2009.04.007. [DOI] [PubMed] [Google Scholar]
- 24.Turner H.E., Harris A.L., Melmed S., Wass J.A. Angiogenesis in endocrine tumors. Endocr Rev. 2003;24(5):600–632. doi: 10.1210/er.2002-0008. [DOI] [PubMed] [Google Scholar]
- 25.Olive K.P., Jacobetz M.A., Davidson C.J., Gopinathan A., McIntyre D., Honess D. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324(5933):1457–1461. doi: 10.1126/science.1171362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kirsch M., Schackert G., Black P.M. Metastasis and angiogenesis. Cancer Treat Res. 2004;117:285–304. doi: 10.1007/978-1-4419-8871-3_17. [DOI] [PubMed] [Google Scholar]
- 27.Nagy JA, Chang S-H, Shih S-C, Dvorak AM, Dvorak HF, editors. Heterogeneity of the tumor vasculature. Semin Thromb Hemost 2010;36(3):321–31. [DOI] [PMC free article] [PubMed]
- 28.Raza A., Franklin M.J., Dudek A.Z. Pericytes and vessel maturation during tumor angiogenesis and metastasis. Am J Hematol. 2010;85(8):593–598. doi: 10.1002/ajh.21745. [DOI] [PubMed] [Google Scholar]
- 29.Kech P., Hauser S., Krivi G., Sanzo K., Warren T., Feder J. Vascular permeability factor, an endothelial cell mitogen related to platelet derived growth factor. Science. 1989;246(4935):1309–1312. doi: 10.1126/science.2479987. [DOI] [PubMed] [Google Scholar]
- 30.Tozer G.M., Kanthou C., Baguley B.C. Disrupting tumour blood vessels. Nat Rev Cancer. 2005;5(6):423–435. doi: 10.1038/nrc1628. [DOI] [PubMed] [Google Scholar]
- 31.Skliarenko J.V., Lunt S.J., Gordon M.L., Vitkin A., Milosevic M., Hill R.P. Effects of the vascular disrupting agent ZD6126 on interstitial fluid pressure and cell survival in tumors. Can Res. 2006;66(4):2074–2080. doi: 10.1158/0008-5472.CAN-05-2046. [DOI] [PubMed] [Google Scholar]
- 32.Robinson S.P., McIntyre D.J., Checkley D., Tessier J.J., Howe F.A., Griffiths J.R. Tumour dose response to the antivascular agent ZD6126 assessed by magnetic resonance imaging. Br J Cancer. 2003;88(10):1592–1597. doi: 10.1038/sj.bjc.6600926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Seidi K., Jahanban-Esfahlan R., Zarghami N. Tumor rim cells: from resistance to vascular targeting agents to complete tumor ablation. Tumour Biol. 2017;39(3) doi: 10.1177/1010428317691001. 1010428317691001. [DOI] [PubMed] [Google Scholar]
- 34.Wong P.P., Bodrug N., Hodivala-Dilke K.M. Exploring novel methods for modulating tumor blood vessels in cancer treatment. Curr Biol. 2016;26(21):R1161–R1166. doi: 10.1016/j.cub.2016.09.043. [DOI] [PubMed] [Google Scholar]
- 35.Bielenberg D.R., Zetter B.R. The contribution of angiogenesis to the process of metastasis. Cancer J. 2015;21(4):267–273. doi: 10.1097/PPO.0000000000000138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Paduch R. The role of lymphangiogenesis and angiogenesis in tumor metastasis. Cell Oncol (Dordr) 2016;39(5):397–410. doi: 10.1007/s13402-016-0281-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.S-h Li., Tian H., W-m Yue., Li L., W-j Li., Z-t Chen. Overexpression of metastasis-associated protein 1 is significantly correlated with tumor angiogenesis and poor survival in patients with early-stage non-small cell lung cancer. Ann Surg Oncol. 2011;18(7):2048–2056. doi: 10.1245/s10434-010-1510-5. [DOI] [PubMed] [Google Scholar]
- 38.Antiangiogenesis – list results – Clinicaltrials.Gov. Clinicaltrials.gov. N.p., 2017. Web. 23 Apr. 2017; 2017.
- 39.Ran S., Volk L., Hall K., Flister M.J. Lymphangiogenesis and lymphatic metastasis in breast cancer. Pathophysiology. 2010;17(4):229–251. doi: 10.1016/j.pathophys.2009.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Medinger M., Passweg J. Role of tumour angiogenesis in haematological malignancies. Swiss Med Wkly. 2014;144:w14050. doi: 10.4414/smw.2014.14050. [DOI] [PubMed] [Google Scholar]
- 41.Shing Y., Folkman J., Sullivan R., Butterfield C., Murray J., Klagsbrun M. Heparin affinity: purification of a tumor-derived capillary endothelial cell growth factor. Science. 1984;223(4642):1296–1299. doi: 10.1126/science.6199844. [DOI] [PubMed] [Google Scholar]
- 42.Carmeliet P., Jain R.K. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473(7347):298–307. doi: 10.1038/nature10144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kim K.J., Li B., Houck K., Winer J., Ferrara N. The vascular endothelial growth factor proteins: identification of biologically relevant regions by neutralizing monoclonal antibodies. Growth Factors. 1992;7(1):53–64. doi: 10.3109/08977199209023937. [DOI] [PubMed] [Google Scholar]
- 44.Vasudev N.S., Reynolds A.R. Anti-angiogenic therapy for cancer: current progress, unresolved questions and future directions. Angiogenesis. 2014;17(3):471–494. doi: 10.1007/s10456-014-9420-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dvorak H.F. Vascular permeability factor/vascular endothelial growth factor: a critical cytokine in tumor angiogenesis and a potential target for diagnosis and therapy. J Clin Oncol. 2002;20(21):4368–4380. doi: 10.1200/JCO.2002.10.088. [DOI] [PubMed] [Google Scholar]
- 46.Fischer C., Mazzone M., Jonckx B., Carmeliet P. FLT1 and its ligands VEGFB and PlGF: drug targets for anti-angiogenic therapy? Nat Rev Cancer. 2008;8(12):942–956. doi: 10.1038/nrc2524. [DOI] [PubMed] [Google Scholar]
- 47.Tammela T., Alitalo K. Lymphangiogenesis: molecular mechanisms and future promise. Cell. 2010;140(4):460–476. doi: 10.1016/j.cell.2010.01.045. [DOI] [PubMed] [Google Scholar]
- 48.Lee S., Chen T.T., Barber C.L., Jordan M.C., Murdock J., Desai S. Autocrine VEGF signaling is required for vascular homeostasis. Cell. 2007;130(4):691–703. doi: 10.1016/j.cell.2007.06.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zhang J, Li Y, editors. Therapeutic uses of FGFs. Semin Cell Dev Biol 2016;53:144–54. [DOI] [PubMed]
- 50.Khan K.A., Bicknell R. Anti-angiogenic alternatives to VEGF blockade. Clin Exp Metas. 2016;33(2):197–210. doi: 10.1007/s10585-015-9769-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kopetz S., Hoff P.M., Morris J.S., Wolff R.A., Eng C., Glover K.Y. Phase II trial of infusional fluorouracil, irinotecan, and bevacizumab for metastatic colorectal cancer: efficacy and circulating angiogenic biomarkers associated with therapeutic resistance. J Clin Oncol. 2010;28(3):453–459. doi: 10.1200/JCO.2009.24.8252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Khalid E.B., Ayman E.E., Rahman H., Abdelkarim G., Najda A. Natural products against cancer angiogenesis. Tumor Biol. 2016;37(11):14513–14536. doi: 10.1007/s13277-016-5364-8. [DOI] [PubMed] [Google Scholar]
- 53.Compagni A., Wilgenbus P., Impagnatiello M.-A., Cotten M., Christofori G. Fibroblast growth factors are required for efficient tumor angiogenesis. Can Res. 2000;60(24):7163–7169. [PubMed] [Google Scholar]
- 54.Wang L., Park H., Chhim S., Ding Y., Jiang W., Queen C. A novel monoclonal antibody to fibroblast growth factor 2 effectively inhibits growth of hepatocellular carcinoma xenografts. Mol Cancer Ther. 2012;11(4):864–872. doi: 10.1158/1535-7163.MCT-11-0813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Heldin C.-H., Eriksson U., Östman A. New members of the platelet-derived growth factor family of mitogens. Arch Biochem Biophys. 2002;398(2):284–290. doi: 10.1006/abbi.2001.2707. [DOI] [PubMed] [Google Scholar]
- 56.Zhao Y., Adjei A.A. Targeting angiogenesis in cancer therapy: moving beyond vascular endothelial growth factor. Oncologist. 2015;20(6):660–673. doi: 10.1634/theoncologist.2014-0465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Dewerchin M., Carmeliet P. PlGF: a multitasking cytokine with disease-restricted activity. Cold Spring Harb Perspect Med. 2012;2:a011056. doi: 10.1101/cshperspect.a011056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Nielsen D.L., Sengeløv L. Inhibition of placenta growth factor with TB-403: a novel antiangiogenic cancer therapy. Expert Opin Biol Ther. 2012;12(6):795–804. doi: 10.1517/14712598.2012.679655. [DOI] [PubMed] [Google Scholar]
- 59.Thomas M., Augustin H.G. The role of the angiopoietins in vascular morphogenesis. Angiogenesis. 2009;12(2):125–137. doi: 10.1007/s10456-009-9147-3. [DOI] [PubMed] [Google Scholar]
- 60.Yuan H.T., Khankin E.V., Karumanchi S.A., Parikh S.M. Angiopoietin 2 is a partial agonist/antagonist of Tie2 signaling in the endothelium. Mol Cell Biol. 2009;29(8):2011–2022. doi: 10.1128/MCB.01472-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Graveel C.R., Tolbert D., Woude G.F.V. MET: a critical player in tumorigenesis and therapeutic target. Cold Spring Harb Perspect Biol. 2013;5(7):a009209. doi: 10.1101/cshperspect.a009209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jahangiri A., De Lay M., Miller L.M., Carbonell W.S., Hu Y.-L., Lu K. Gene expression profile identifies tyrosine kinase c-Met as a targetable mediator of antiangiogenic therapy resistance. Clin Cancer Res. 2013;19(7):1773–1783. doi: 10.1158/1078-0432.CCR-12-1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Verrienti A., Tallini G., Colato C., Boichard A., Checquolo S., Pecce V. RET mutation and increased angiogenesis in medullary thyroid carcinomas. Endocr Relat Cancer. 2016;23(8):665–676. doi: 10.1530/ERC-16-0132. [DOI] [PubMed] [Google Scholar]
- 64.Elisei R., Schlumberger M.J., Müller S.P., Schöffski P., Brose M.S., Shah M.H. Cabozantinib in progressive medullary thyroid cancer. J Clin Oncol. 2013;31(29):3639–3646. doi: 10.1200/JCO.2012.48.4659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kuhnert F., Kirshner J.R., Thurston G. Dll4-Notch signaling as a therapeutic target in tumor angiogenesis. Vasc Cell. 2011;3(1):1. doi: 10.1186/2045-824X-3-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Hellström M., Phng L.-K., Hofmann J.J., Wallgard E., Coultas L., Lindblom P. Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature. 2007;445(7129):776–780. doi: 10.1038/nature05571. [DOI] [PubMed] [Google Scholar]
- 67.Patel N.S., Li J.-L., Generali D., Poulsom R., Cranston D.W., Harris A.L. Up-regulation of delta-like 4 ligand in human tumor vasculature and the role of basal expression in endothelial cell function. Can Res. 2005;65(19):8690–8697. doi: 10.1158/0008-5472.CAN-05-1208. [DOI] [PubMed] [Google Scholar]
- 68.Limbourg F.P., Takeshita K., Radtke F., Bronson R.T., Chin M.T., Liao J.K. Essential role of endothelial Notch1 in angiogenesis. Circulation. 2005;111(14):1826–1832. doi: 10.1161/01.CIR.0000160870.93058.DD. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Yan M., Callahan C.A., Beyer J.C., Allamneni K.P., Zhang G., Ridgway J.B. Chronic DLL4 blockade induces vascular neoplasms. Nature. 2010;463(7282):E6–E7. doi: 10.1038/nature08751. [DOI] [PubMed] [Google Scholar]
- 70.Mosch B, Reissenweber B, Neuber C, Pietzsch J. Eph receptors and ephrin ligands: important players in angiogenesis and tumor angiogenesis. J Oncol 2010;2010. [DOI] [PMC free article] [PubMed]
- 71.Cheng N., Brantley D.M., Liu H., Lin Q., Enriquez M., Gale N. Blockade of EphA receptor tyrosine kinase activation inhibits vascular endothelial cell growth factor-induced angiogenesis. Mol Cancer Res. 2002;1(1):2–11. [PubMed] [Google Scholar]
- 72.Sawamiphak S., Seidel S., Essmann C.L., Wilkinson G.A., Pitulescu M.E., Acker T. Ephrin-B2 regulates VEGFR2 function in developmental and tumour angiogenesis. Nature. 2010;465(7297):487–491. doi: 10.1038/nature08995. [DOI] [PubMed] [Google Scholar]
- 73.Weis S, Cheresh D. alphaV integrins in angiogenesis and cancer. Cold Spring Harb Perspect Med 2011;1:a006478. [DOI] [PMC free article] [PubMed]
- 74.Friedlander M., Brooks P.C., Shaffer R.W., Kincaid C.M. Definition of two angiogenic pathways by distinct alphav integrins. Science. 1995;270(5241):1500–1502. doi: 10.1126/science.270.5241.1500. [DOI] [PubMed] [Google Scholar]
- 75.Wang Z., Yan X. CD146, a multi-functional molecule beyond adhesion. Cancer Lett. 2013;330(2):150–162. doi: 10.1016/j.canlet.2012.11.049. [DOI] [PubMed] [Google Scholar]
- 76.Zeng Q., Wu Z., Duan H., Jiang X., Tu T., Lu D. Impaired tumor angiogenesis and VEGF-induced pathway in endothelial CD146 knockout mice. Protein Cell. 2014;5(6):445–456. doi: 10.1007/s13238-014-0047-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Flanagan K., Fitzgerald K., Baker J., Regnstrom K., Gardai S., Bard F. Laminin-411 is a vascular ligand for MCAM and facilitates TH17 cell entry into the CNS. PLoS ONE. 2012;7(7):e40443. doi: 10.1371/journal.pone.0040443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Tu T., Zhang C., Yan H., Luo Y., Kong R., Wen P. CD146 acts as a novel receptor for netrin-1 in promoting angiogenesis and vascular development. Cell Res. 2015;25(3):275–287. doi: 10.1038/cr.2015.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Dejana E., Giampietro C. Vascular endothelial-cadherin and vascular stability. Curr Opin Hematol. 2012;19(3):218–223. doi: 10.1097/MOH.0b013e3283523e1c. [DOI] [PubMed] [Google Scholar]
- 80.Prandini M.-H., Dreher I., Bouillot S., Benkerri S., Moll T., Huber P. The human VE-cadherin promoter is subjected to organ-specific regulation and is activated in tumour angiogenesis. Oncogene. 2005;24(18):2992–3001. doi: 10.1038/sj.onc.1208483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Li H., Shi X., Liu J., Hu C., Zhang X., Liu H. The soluble fragment of VE-cadherin inhibits angiogenesis by reducing endothelial cell proliferation and tube capillary formation. Cancer Gene Ther. 2010;17(10):700–707. doi: 10.1038/cgt.2010.26. [DOI] [PubMed] [Google Scholar]
- 82.Chaudhary A., Hilton M.B., Seaman S., Haines D.C., Stevenson S., Lemotte P.K. TEM8/ANTXR1 blockade inhibits pathological angiogenesis and potentiates tumoricidal responses against multiple cancer types. Cancer Cell. 2012;21(2):212–226. doi: 10.1016/j.ccr.2012.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ucuzian A.A., Gassman A.A., East A.T., Greisler H.P. Molecular mediators of angiogenesis. J Burn Care Res. 2010;31(1):158–175. doi: 10.1097/BCR.0b013e3181c7ed82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Nolan-Stevaux O., Zhong W., Culp S., Shaffer K., Hoover J., Wickramasinghe D. Endoglin requirement for BMP9 signaling in endothelial cells reveals new mechanism of action for selective anti-endoglin antibodies. PLoS ONE. 2012;7(12):e50920. doi: 10.1371/journal.pone.0050920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Umikawa M., Umikawa A., Asato T., Takei K., Matsuzaki G., K-i Kariya. Angiopoietin-like protein 2 induces proinflammatory responses in peritoneal cells. Biochem Biophys Res Commun. 2015;467(2):235–241. doi: 10.1016/j.bbrc.2015.09.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Gaur P., Bielenberg D.R., Samuel S., Bose D., Zhou Y., Gray M.J. Role of class 3 semaphorins and their receptors in tumor growth and angiogenesis. Clin Cancer Res. 2009;15(22):6763–6770. doi: 10.1158/1078-0432.CCR-09-1810. [DOI] [PubMed] [Google Scholar]
- 87.Neufeld G., Sabag A.D., Rabinovicz N., Kessler O. Semaphorins in angiogenesis and tumor progression. Cold Spring Harb Perspect Med. 2012;2(1):a006718. doi: 10.1101/cshperspect.a006718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhou H., Binmadi N.O., Yang Y.-H., Proia P., Basile J.R. Semaphorin 4D cooperates with VEGF to promote angiogenesis and tumor progression. Angiogenesis. 2012;15(3):391–407. doi: 10.1007/s10456-012-9268-y. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 89.Leszczynska K., Kaur S., Wilson E., Bicknell R., Heath V.L. The role of RhoJ in endothelial cell biology and angiogenesis. Biochem Soc Trans. 2011 Dec;39(6):1606–1611. doi: 10.1042/BST20110702. [DOI] [PubMed] [Google Scholar]
- 90.Mura M., Swain R., Zhuang X., Vorschmitt H., Reynolds G., Durant S. Identification and angiogenic role of the novel tumor endothelial marker CLEC14A. Oncogene. 2012;31(3):293–305. doi: 10.1038/onc.2011.233. [DOI] [PubMed] [Google Scholar]
- 91.Noy P., Lodhia P., Khan K., Zhuang X., Ward D., Verissimo A. Blocking CLEC14A-MMRN2 binding inhibits sprouting angiogenesis and tumour growth. Oncogene. 2015 doi: 10.1038/onc.2015.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Berretta M., Rinaldi L., Di Benedetto F., Lleshi A., De Re V., Facchini G. Angiogenesis inhibitors for the treatment of hepatocellular carcinoma. Front Pharmacol. 2016;7:428. doi: 10.3389/fphar.2016.00428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Raddum A.M., Evensen L., Hollås H., Grindheim A.K., Lorens J.B., Vedeler A. Domains I and IV of annexin A2 affect the formation and integrity of in vitro capillary-like networks. PLoS ONE. 2013;8(3):e60281. doi: 10.1371/journal.pone.0060281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Raddum A.M., Hollås H., Shumilin I.A., Henklein P., Kretsinger R., Fossen T. The native structure of annexin A2 peptides in hydrophilic environment determines their anti-angiogenic effects. Biochem Pharmacol. 2015;95(1):1–15. doi: 10.1016/j.bcp.2015.02.013. [DOI] [PubMed] [Google Scholar]
- 95.Shimizu T., Kurozumi K., Ishida J., Ichikawa T., Date I. Adhesion molecules and the extracellular matrix as drug targets for glioma. Brain Tumor Pathol. 2016;33(2):97–106. doi: 10.1007/s10014-016-0261-9. [DOI] [PubMed] [Google Scholar]
- 96.Lombardi G., Pambuku A., Bellu L., Farina M., Della Puppa A., Denaro L. Effectiveness of antiangiogenic drugs in glioblastoma patients: a systematic review and meta-analysis of randomized clinical trials. Crit Rev Oncol Hematol. 2017;111:94–102. doi: 10.1016/j.critrevonc.2017.01.018. [DOI] [PubMed] [Google Scholar]
- 97.Tewari K.S., Sill M.W., Long H.J., III, Penson R.T., Huang H., Ramondetta L.M. Improved survival with bevacizumab in advanced cervical cancer. N Engl J Med. 2014;370(8):734–743. doi: 10.1056/NEJMoa1309748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Lin Z., Zhang Q., Luo W. Angiogenesis inhibitors as therapeutic agents in cancer: challenges and future directions. Eur J Pharmacol. 2016 doi: 10.1016/j.ejphar.2016.10.039. [DOI] [PubMed] [Google Scholar]
- 99.Grabowski J., Glode A. Ramucirumab: a vascular endothelial growth factor receptor-2 inhibitor with activity in several malignancies. Am J Health Syst Pharm. 2016;73(13):957–968. doi: 10.2146/ajhp150188. [DOI] [PubMed] [Google Scholar]
- 100.Kienast Y., Klein C., Scheuer W., Raemsch R., Lorenzon E., Bernicke D. Ang-2-VEGF-A CrossMab, a novel bispecific human IgG1 antibody blocking VEGF-A and Ang-2 functions simultaneously, mediates potent antitumor, antiangiogenic, and antimetastatic efficacy. Clin Cancer Res. 2013;19(24):6730–6740. doi: 10.1158/1078-0432.CCR-13-0081. [DOI] [PubMed] [Google Scholar]
- 101.Ricci V., Ronzoni M., Fabozzi T. Aflibercept a new target therapy in cancer treatment: a review. Crit Rev Oncol Hematol. 2015;96(3):569–576. doi: 10.1016/j.critrevonc.2015.07.001. [DOI] [PubMed] [Google Scholar]
- 102.Scartozzi M., Vincent L., Chiron M., Cascinu S. Aflibercept, a new way to target angiogenesis in the second line treatment of metastatic colorectal cancer (mCRC) Target Oncol. 2016;11(4):489–500. doi: 10.1007/s11523-016-0447-4. [DOI] [PubMed] [Google Scholar]
- 103.Liontos M., Lykka M., Bamias A. Profile of trebananib (AMG386) and its potential in the treatment of ovarian cancer. Onco Targets Ther. 2014;7:1837–1845. doi: 10.2147/OTT.S65522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Abou-Alfa GK, Blanc J-F, Miles S, Ganten TM, Trojan J, Cebon JS, et al., editors. Phase II study of first-line trebananib plus sorafenib in patients with advanced hepatocellular carcinoma (HCC). In: ASCO annual meeting proceedings; 2014. [DOI] [PMC free article] [PubMed]
- 105.Al Wadi K., Ghatage P. Efficacy of trebananib (AMG 386) in treating epithelial ovarian cancer. Expert Opin Pharmacother. 2016;17(6):853–860. doi: 10.1517/14656566.2016.1161027. [DOI] [PubMed] [Google Scholar]
- 106.Li D., Wei X., Xie K., Chen K., Li J., Fang J. A novel decoy receptor fusion protein for FGF-2 potently inhibits tumour growth. Br J Cancer. 2014;111(1):68–77. doi: 10.1038/bjc.2014.282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Noguera-Troise I., Daly C., Papadopoulos N.J., Coetzee S., Boland P., Gale N.W. Blockade of Dll4 inhibits tumour growth by promoting non-productive angiogenesis. Nature. 2006;444(7122):1032–1037. doi: 10.1038/nature05355. [DOI] [PubMed] [Google Scholar]
- 108.Brantley D.M., Cheng N., Thompson E.J., Lin Q., Brekken R.A., Thorpe P.E. Soluble Eph A receptors inhibit tumor angiogenesis and progression in vivo. Oncogene. 2002;21(46):7011–7026. doi: 10.1038/sj.onc.1205679. [DOI] [PubMed] [Google Scholar]
- 109.Strawn L.M., McMahon G., App H., Schreck R., Kuchler W.R., Longhi M.P. Flk-1 as a target for tumor growth inhibition. Can Res. 1996;56(15):3540–3545. [PubMed] [Google Scholar]
- 110.Baumann K., Du Bois A., Meier W., Rau J., Wimberger P., Sehouli J. A phase II trial (AGO 2.11) in platinum-resistant ovarian cancer: a randomized multicenter trial with sunitinib (SU11248) to evaluate dosage, schedule, tolerability, toxicity and effectiveness of a multitargeted receptor tyrosine kinase inhibitor monotherapy. Ann Oncol. 2012;23(9):2265–2271. doi: 10.1093/annonc/mds003. [DOI] [PubMed] [Google Scholar]
- 111.Iyer R., Fetterly G., Lugade A., Thanavala Y. Sorafenib: a clinical and pharmacologic review. Expert Opin Pharmacother. 2010;11(11):1943–1955. doi: 10.1517/14656566.2010.496453. [DOI] [PubMed] [Google Scholar]
- 112.Carlisle B, Demko N, Freeman G, Hakala A, MacKinnon N, Ramsay T, et al. Benefit, risk, and outcomes in drug development: a systematic review of sunitinib. J Natl Cancer Inst 2016;108(1):djv292. [DOI] [PMC free article] [PubMed]
- 113.Johnson P.J., Qin S., Park J.-W., Poon R.T., Raoul J.-L., Philip P.A. Brivanib versus sorafenib as first-line therapy in patients with unresectable, advanced hepatocellular carcinoma: results from the randomized phase III BRISK-FL study. J Clin Oncol. 2013;31(28):3517–3524. doi: 10.1200/JCO.2012.48.4410. [DOI] [PubMed] [Google Scholar]
- 114.Wedge S.R., Kendrew J., Hennequin L.F., Valentine P.J., Barry S.T., Brave S.R. AZD2171: a highly potent, orally bioavailable, vascular endothelial growth factor receptor-2 tyrosine kinase inhibitor for the treatment of cancer. Can Res. 2005;65(10):4389–4400. doi: 10.1158/0008-5472.CAN-04-4409. [DOI] [PubMed] [Google Scholar]
- 115.Angevin E., Lopez-Martin J.A., Lin C.-C., Gschwend J.E., Harzstark A., Castellano D. Phase I study of dovitinib (TKI258), an oral FGFR, VEGFR, and PDGFR inhibitor, in advanced or metastatic renal cell carcinoma. Clin Cancer Res. 2013;19(5):1257–1268. doi: 10.1158/1078-0432.CCR-12-2885. [DOI] [PubMed] [Google Scholar]
- 116.Matsui J., Funahashi Y., Uenaka T., Watanabe T., Tsuruoka A., Asada M. Multi-kinase inhibitor E7080 suppresses lymph node and lung metastases of human mammary breast tumor MDA-MB-231 via inhibition of vascular endothelial growth factor-receptor (VEGF-R) 2 and VEGF-R3 kinase. Clin Cancer Res. 2008;14(17):5459–5465. doi: 10.1158/1078-0432.CCR-07-5270. [DOI] [PubMed] [Google Scholar]
- 117.Cainap C., Qin S., Huang W.-T., Chung I.J., Pan H., Cheng Y. Linifanib versus Sorafenib in patients with advanced hepatocellular carcinoma: results of a randomized phase III trial. J Clin Oncol. 2015;33(2):172–179. doi: 10.1200/JCO.2013.54.3298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Zhou J., Goh B.C., Albert D.H., Chen C.S. ABT-869, a promising multi-targeted tyrosine kinase inhibitor: from bench to bedside. J Hematol Oncol. 2009;2:33. doi: 10.1186/1756-8722-2-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Kumar V., Chhibber S. Thalidomide: an old drug with new action. J Chemother. 2011;23(6):326–334. doi: 10.1179/joc.2011.23.6.326. [DOI] [PubMed] [Google Scholar]
- 120.Sherbet G.V. Therapeutic potential of thalidomide and its analogues in the treatment of cancer. Anticancer Res. 2015;35(11):5767–5772. [PubMed] [Google Scholar]
- 121.Kumar S.K., Vij R., Noga S.J., Berg D., Brent L., Dollar L. Treating multiple myeloma patients with oral therapies. Clin Lymphoma Myeloma Leuk. 2017;17(5):243–251. doi: 10.1016/j.clml.2017.02.024. [DOI] [PubMed] [Google Scholar]
- 122.Cragg G.M., Newman D.J. Plants as a source of anti-cancer agents. J Ethnopharmacol. 2005;100(1–2):72–79. doi: 10.1016/j.jep.2005.05.011. [DOI] [PubMed] [Google Scholar]
- 123.Anderson H.L., Yap J.T., Miller M.P., Robbins A., Jones T., Price P.M. Assessment of pharmacodynamic vascular response in a phase I trial of combretastatin A4 phosphate. J Clin Oncol. 2003;21(15):2823–2830. doi: 10.1200/JCO.2003.05.186. [DOI] [PubMed] [Google Scholar]
- 124.Jaroch K., Karolak M., Gorski P., Jaroch A., Krajewski A., Ilnicka A. Combretastatins: in vitro structure-activity relationship, mode of action and current clinical status. Pharmacol Rep. 2016;68(6):1266–1275. doi: 10.1016/j.pharep.2016.08.007. [DOI] [PubMed] [Google Scholar]
- 125.Zweifel M., Jayson G.C., Reed N., Osborne R., Hassan B., Ledermann J. Phase II trial of combretastatin A4 phosphate, carboplatin, and paclitaxel in patients with platinum-resistant ovarian cancer. Ann Oncol. 2011;22(9):2036–2041. doi: 10.1093/annonc/mdq708. [DOI] [PubMed] [Google Scholar]
- 126.Ibrahim M.A., Do D.V., Sepah Y.J., Shah S.M., Van Anden E., Hafiz G. Vascular disrupting agent for neovascular age related macular degeneration: a pilot study of the safety and efficacy of intravenous combretastatin a-4 phosphate. BMC Pharmacol Toxicol. 2013;14:7. doi: 10.1186/2050-6511-14-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Liu P., Qin Y., Wu L., Yang S., Li N., Wang H. A phase I clinical trial assessing the safety and tolerability of combretastatin A4 phosphate injections. Anticancer Drugs. 2014;25(4):462–471. doi: 10.1097/CAD.0000000000000070. [DOI] [PubMed] [Google Scholar]
- 128.Na Y. Recent cancer drug development with xanthone structures. J Pharm Pharmacol. 2009;61(6):707–712. doi: 10.1211/jpp/61.06.0002. [DOI] [PubMed] [Google Scholar]
- 129.LoRusso P.M., Boerner S.A., Hunsberger S. Clinical development of vascular disrupting agents: what lessons can we learn from ASA404? J Clin Oncol. 2011;29(22):2952–2955. doi: 10.1200/JCO.2011.36.1311. [DOI] [PubMed] [Google Scholar]
- 130.Kruczynski A., Poli M., Dossi R., Chazottes E., Berrichon G., Ricome C. Anti-angiogenic, vascular-disrupting and anti-metastatic activities of vinflunine, the latest vinca alkaloid in clinical development. Eur J Cancer. 2006;42(16):2821–2832. doi: 10.1016/j.ejca.2006.06.019. [DOI] [PubMed] [Google Scholar]
- 131.Banerjee S, Hwang DJ, Li W, Miller DD. Current advances of tubulin inhibitors in nanoparticle drug delivery and vascular disruption/angiogenesis. Molecules 2016;21(11). [DOI] [PMC free article] [PubMed]
- 132.Wani M.C., Taylor H.L., Wall M.E., Coggon P., McPhail A.T. Plant antitumor agents. VI. Isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc. 1971;93(9):2325–2327. doi: 10.1021/ja00738a045. [DOI] [PubMed] [Google Scholar]
- 133.Slaughter K.N., Moore K.N., Mannel R.S. Anti-angiogenic therapy versus dose-dense paclitaxel therapy for frontline treatment of epithelial ovarian cancer: review of phase III randomized clinical trials. Curr Oncol Rep. 2014;16(11):412. doi: 10.1007/s11912-014-0412-2. [DOI] [PubMed] [Google Scholar]
- 134.Kumar M., Dhatwalia S.K., Dhawan D.K. Role of angiogenic factors of herbal origin in regulation of molecular pathways that control tumor angiogenesis. Tumour Biol. 2016;37(11):14341–14354. doi: 10.1007/s13277-016-5330-5. [DOI] [PubMed] [Google Scholar]
- 135.Kunnumakkara A.B., Anand P., Aggarwal B.B. Curcumin inhibits proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signaling proteins. Cancer Lett. 2008;269(2):199–225. doi: 10.1016/j.canlet.2008.03.009. [DOI] [PubMed] [Google Scholar]
- 136.Abdel-Latif G.A., Al-Abd A.M., Tadros M.G., Al-Abbasi F.A., Khalifa A.E., Abdel-Naim A.B. The chemomodulatory effects of resveratrol and didox on herceptin cytotoxicity in breast cancer cell lines. Sci Rep. 2015;5:12054. doi: 10.1038/srep12054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Khaleel S.A., Al-Abd A.M., Ali A.A., Abdel-Naim A.B. Didox and resveratrol sensitize colorectal cancer cells to doxorubicin via activating apoptosis and ameliorating P-glycoprotein activity. Sci Rep. 2016;6:36855. doi: 10.1038/srep36855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.López-Jiménez A., García-Caballero M., Medina M.Á., Quesada A.R. Anti-angiogenic properties of carnosol and carnosic acid, two major dietary compounds from rosemary. Eur J Nutr. 2013;52(1):85–95. doi: 10.1007/s00394-011-0289-x. [DOI] [PubMed] [Google Scholar]
- 139.Igura K., Ohta T., Kuroda Y., Kaji K. Resveratrol and quercetin inhibit angiogenesis in vitro. Cancer Lett. 2001;171(1):11–16. doi: 10.1016/s0304-3835(01)00443-8. [DOI] [PubMed] [Google Scholar]
- 140.Pratheeshkumar P., Budhraja A., Son Y.-O., Wang X., Zhang Z., Ding S. Quercetin inhibits angiogenesis mediated human prostate tumor growth by targeting VEGFR-2 regulated AKT/mTOR/P70S6K signaling pathways. PLoS ONE. 2012;7(10):e47516. doi: 10.1371/journal.pone.0047516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Su S.J., Yeh T.M., Chuang W.J., Ho C.L., Chang K.L., Cheng H.L. The novel targets for anti-angiogenesis of genistein on human cancer cells. Biochem Pharmacol. 2005;69(2):307–318. doi: 10.1016/j.bcp.2004.09.025. [DOI] [PubMed] [Google Scholar]
- 142.Hosseini A., Ghorbani A. Cancer therapy with phytochemicals: evidence from clinical studies. Avicenna J Phytomed. 2015;5(2):84–97. [PMC free article] [PubMed] [Google Scholar]
- 143.Garcia-Vilas J.A., Martinez-Poveda B., Quesada A.R., Medina M.A. Aeroplysinin-1, a sponge-derived multi-targeted bioactive marine drug. Mar Drugs. 2015;14(1):1. doi: 10.3390/md14010001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Wahl O., Oswald M., Tretzel L., Herres E., Arend J., Efferth T. Inhibition of tumor angiogenesis by antibodies, synthetic small molecules and natural products. Curr Med Chem. 2011;18(21):3136–3155. doi: 10.2174/092986711796391570. [DOI] [PubMed] [Google Scholar]
- 145.Niemann H., Marmann A., Lin W., Proksch P. Sponge derived bromotyrosines: structural diversity through natural combinatorial chemistry. Nat Prod Commun. 2015;10(1):219–231. [PubMed] [Google Scholar]
- 146.Wagner S.C., Ichim T.E., Ma H., Szymanski J., Perez J.A., Lopez J. Cancer anti-angiogenesis vaccines: is the tumor vasculature antigenically unique? J Transl Med. 2015;13:340. doi: 10.1186/s12967-015-0688-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Bussolati B., Deambrosis I., Russo S., Deregibus M.C., Camussi G. Altered angiogenesis and survival in human tumor-derived endothelial cells. FASEB J. 2003;17(9):1159–1161. doi: 10.1096/fj.02-0557fje. [DOI] [PubMed] [Google Scholar]
- 148.Grange C., Bussolati B., Bruno S., Fonsato V., Sapino A., Camussi G. Isolation and characterization of human breast tumor-derived endothelial cells. Oncol Rep. 2006;15(2):381–386. [PubMed] [Google Scholar]
- 149.Xiong Y.Q., Sun H.C., Zhang W., Zhu X.D., Zhuang P.Y., Zhang J.B. Human hepatocellular carcinoma tumor–derived endothelial cells manifest increased angiogenesis capability and drug resistance compared with normal endothelial cells. Clin Cancer Res. 2009;15(15):4838–4846. doi: 10.1158/1078-0432.CCR-08-2780. [DOI] [PubMed] [Google Scholar]
- 150.Li Y., Bohlen P., Hicklin D.J. Vaccination against angiogenesis-associated antigens: a novel cancer immunotherapy strategy. Curr Mol Med. 2003;3(8):773–779. doi: 10.2174/1566524033479438. [DOI] [PubMed] [Google Scholar]
- 151.Gavilondo J., Hernández-Bernal F., Ayala-Ávila M., de la Torre A., de la Torre J., Morera-Díaz Y. Specific active immunotherapy with a VEGF vaccine in patients with advanced solid tumors. Results of the CENTAURO antigen dose escalation phase I clinical trial. Vaccine. 2014;32(19):2241–2250. doi: 10.1016/j.vaccine.2013.11.102. [DOI] [PubMed] [Google Scholar]
- 152.Chen R., Wang S., Yao Y., Zhou Y., Zhang C., Fang J. Anti-metastatic effects of DNA vaccine encoding single-chain trimer composed of MHC I and vascular endothelial growth factor receptor 2 peptide. Oncol Rep. 2015;33(5):2269–2276. doi: 10.3892/or.2015.3820. [DOI] [PubMed] [Google Scholar]
- 153.Liang P.H., Zhang K.Q., Xu G.L., Li Y.F., Wang L.F., Nie Z.L. Construction of a DNA vaccine encoding Flk-1 extracellular domain and C3d fusion gene and investigation of its suppressing effect on tumor growth. Cancer Immunol Immunother. 2010;59(1):93–101. doi: 10.1007/s00262-009-0727-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Miyazawa M., Ohsawa R., Tsunoda T., Hirono S., Kawai M., Tani M. Phase I clinical trial using peptide vaccine for human vascular endothelial growth factor receptor 2 in combination with gemcitabine for patients with advanced pancreatic cancer. Cancer Sci. 2010;101(2):433–439. doi: 10.1111/j.1349-7006.2009.01416.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Fu C., Bardhan S., Cetateanu N.D., Wamil B.D., Wang Y., Yan H.-P. Identification of a novel membrane protein, HP59, with therapeutic potential as a target of tumor angiogenesis. Clin Cancer Res. 2001;7(12):4182–4194. [PubMed] [Google Scholar]
- 156.García B., Neninger E., de la Torre A., Leonard I., Martínez R., Viada C. Effective inhibition of the epidermal growth factor/epidermal growth factor receptor binding by anti-epidermal growth factor antibodies is related to better survival in advanced non-small-cell lung cancer patients treated with the epidermal growth factor cancer vaccine. Clin Cancer Res. 2008;14(3):840–846. doi: 10.1158/1078-0432.CCR-07-1050. [DOI] [PubMed] [Google Scholar]
- 157.Lorente A.F., Brooks S.A., Vinageras E.N., Alvarez Md.C.B., Brito B.W., Concepción M.T. Effect of blockade of the EGF system on wound healing in patients vaccinated with CIMAvax® EGF. World J Surg Oncol. 2013;11:275. doi: 10.1186/1477-7819-11-275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Nerini I.F., Cesca M., Bizzaro F., Giavazzi R. Combination therapy in cancer: effects of angiogenesis inhibitors on drug pharmacokinetics and pharmacodynamics. Chin J Cancer. 2016;35(1):61. doi: 10.1186/s40880-016-0123-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Jain R.K. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307(5706):58–62. doi: 10.1126/science.1104819. [DOI] [PubMed] [Google Scholar]
- 160.Siemann D.W., Rojiani A.M. Enhancement of radiation therapy by the novel vascular targeting agent ZD6126. Int J Radiat Oncol Biol Phys. 2002;53(1):164–171. doi: 10.1016/s0360-3016(02)02742-6. [DOI] [PubMed] [Google Scholar]
- 161.Liang W., Ni Y., Chen F. Tumor resistance to vascular disrupting agents: mechanisms, imaging, and solutions. Oncotarget. 2016;7(13):15444–15459. doi: 10.18632/oncotarget.6999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Horsman M.R., Siemann D.W. Pathophysiologic effects of vascular-targeting agents and the implications for combination with conventional therapies. Can Res. 2006;66(24):11520–11539. doi: 10.1158/0008-5472.CAN-06-2848. [DOI] [PubMed] [Google Scholar]
- 163.Nagaiah G., Remick S.C. Combretastatin A4 phosphate: a novel vascular disrupting agent. Future Oncol. 2010;6(8):1219–1228. doi: 10.2217/fon.10.90. [DOI] [PubMed] [Google Scholar]
- 164.Kłosowska-Wardęga A., Hasumi Y., Burmakin M., Åhgren A., Stuhr L., Moen I. Combined anti-angiogenic therapy targeting PDGF and VEGF receptors lowers the interstitial fluid pressure in a murine experimental carcinoma. PLoS ONE. 2009;4(12):e8149. doi: 10.1371/journal.pone.0008149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Al-Abd A.M., Al-Abbasi F.A., Torchilin V.P. Intratumoral pharmacokinetics: challenges to nanobiomaterials. Curr Pharm Des. 2015;21(22):3208–3214. doi: 10.2174/1381612821666150531170621. [DOI] [PubMed] [Google Scholar]
- 166.Martinelli M., Bonezzi K., Riccardi E., Kuhn E., Frapolli R., Zucchetti M. Sequence dependent antitumour efficacy of the vascular disrupting agent ZD6126 in combination with paclitaxel. Br J Cancer. 2007;97(7):888–894. doi: 10.1038/sj.bjc.6603969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Dickson P.V., Hamner J.B., Sims T.L., Fraga C.H., Ng C.Y., Rajasekeran S. Bevacizumab-induced transient remodeling of the vasculature in neuroblastoma xenografts results in improved delivery and efficacy of systemically administered chemotherapy. Clin Cancer Res. 2007;13(13):3942–3950. doi: 10.1158/1078-0432.CCR-07-0278. [DOI] [PubMed] [Google Scholar]
- 168.Zhou F., Hu J., Shao J.-H., Zou S.-B., Shen S.-L., Luo Z.-Q. Metronomic chemotherapy in combination with antiangiogenic treatment induces mosaic vascular reduction and tumor growth inhibition in hepatocellular carcinoma xenografts. J Cancer Res Clin Oncol. 2012;138(11):1879–1890. doi: 10.1007/s00432-012-1270-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Carbone C., Tamburrino A., Piro G., Boschi F., Cataldo I., Zanotto M. Combined inhibition of IL1, CXCR1/2, and TGFβ signaling pathways modulates in-vivo resistance to anti-VEGF treatment. Anticancer Drugs. 2016;27(1):29–40. doi: 10.1097/CAD.0000000000000301. [DOI] [PubMed] [Google Scholar]
- 170.Ma J., Waxman D.J. Combination of antiangiogenesis with chemotherapy for more effective cancer treatment. Mol Cancer Ther. 2008;7(12):3670–3684. doi: 10.1158/1535-7163.MCT-08-0715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Hsu H.-W., Wall N.R., Hsueh C.-T., Kim S., Ferris R.L., Chen C.-S. Combination antiangiogenic therapy and radiation in head and neck cancers. Oral Oncol. 2014;50(1):19–26. doi: 10.1016/j.oraloncology.2013.10.003. [DOI] [PubMed] [Google Scholar]
- 172.Kamrava M., Bernstein M.B., Camphausen K., Hodge J.W. Combining radiation, immunotherapy, and antiangiogenesis agents in the management of cancer: the three musketeers or just another quixotic combination? Mol BioSyst. 2009;5(11):1262–1270. doi: 10.1039/b911313b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Kerbel R.S. A cancer therapy resistant to resistance. Nature. 1997;390(6658):335–336. doi: 10.1038/36978. [DOI] [PubMed] [Google Scholar]
- 174.Kerbel R.S. Inhibition of tumor angiogenesis as a strategy to circumvent acquired resistance to anti-cancer therapeutic agents. BioEssays. 1991;13(1):31–36. doi: 10.1002/bies.950130106. [DOI] [PubMed] [Google Scholar]
- 175.Kindler H.L., Niedzwiecki D., Hollis D., Sutherland S., Schrag D., Hurwitz H. Gemcitabine plus bevacizumab compared with gemcitabine plus placebo in patients with advanced pancreatic cancer: phase III trial of the Cancer and Leukemia Group B (CALGB 80303) J Clin Oncol. 2010;28(22):3617–3622. doi: 10.1200/JCO.2010.28.1386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Saltz L.B., Lenz H.-J., Kindler H.L., Hochster H.S., Wadler S., Hoff P.M. Randomized phase II trial of cetuximab, bevacizumab, and irinotecan compared with cetuximab and bevacizumab alone in irinotecan-refractory colorectal cancer: the BOND-2 study. J Clin Oncol. 2007;25(29):4557–4561. doi: 10.1200/JCO.2007.12.0949. [DOI] [PubMed] [Google Scholar]
- 177.Shojaei F., Wu X., Malik A.K., Zhong C., Baldwin M.E., Schanz S. Tumor refractoriness to anti-VEGF treatment is mediated by CD11b+ Gr1+ myeloid cells. Nat Biotechnol. 2007;25(8):911–920. doi: 10.1038/nbt1323. [DOI] [PubMed] [Google Scholar]
- 178.Bergers G., Hanahan D. Modes of resistance to anti-angiogenic therapy. Nat Rev Cancer. 2008;8(8):592–603. doi: 10.1038/nrc2442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Fernando N.T., Koch M., Rothrock C., Gollogly L.K., D'Amore P.A., Ryeom S. Tumor escape from endogenous, extracellular matrix–associated angiogenesis inhibitors by up-regulation of multiple proangiogenic factors. Clin Cancer Res. 2008;14(5):1529–1539. doi: 10.1158/1078-0432.CCR-07-4126. [DOI] [PubMed] [Google Scholar]
- 180.Mizukami Y., Jo W.-S., Duerr E.-M., Gala M., Li J., Zhang X. Induction of interleukin-8 preserves the angiogenic response in HIF-1α–deficient colon cancer cells. Nat Med. 2005;11(9):992–997. doi: 10.1038/nm1294. [DOI] [PubMed] [Google Scholar]
- 181.Batchelor T.T., Sorensen A.G., di Tomaso E., Zhang W.-T., Duda D.G., Cohen K.S. AZD2171, a pan-VEGF receptor tyrosine kinase inhibitor, normalizes tumor vasculature and alleviates edema in glioblastoma patients. Cancer Cell. 2007;11(1):83–95. doi: 10.1016/j.ccr.2006.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Asahara T., Murohara T., Sullivan A., Silver M., van der Zee R., Li T. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275(5302):964–966. doi: 10.1126/science.275.5302.964. [DOI] [PubMed] [Google Scholar]
- 183.Song S., Ewald A.J., Stallcup W., Werb Z., Bergers G. PDGFRβ+ perivascular progenitor cells in tumours regulate pericyte differentiation and vascular survival. Nat Cell Biol. 2005;7(9):870–879. doi: 10.1038/ncb1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Yang L., DeBusk L.M., Fukuda K., Fingleton B., Green-Jarvis B., Shyr Y. Expansion of myeloid immune suppressor Gr+ CD11b+ cells in tumor-bearing host directly promotes tumor angiogenesis. Cancer Cell. 2004;6(4):409–421. doi: 10.1016/j.ccr.2004.08.031. [DOI] [PubMed] [Google Scholar]
- 185.Shaked Y., Ciarrocchi A., Franco M., Lee C.R., Man S., Cheung A.M. Therapy-induced acute recruitment of circulating endothelial progenitor cells to tumors. Science. 2006;313(5794):1785–1787. doi: 10.1126/science.1127592. [DOI] [PubMed] [Google Scholar]
- 186.Mancuso M.R., Davis R., Norberg S.M., O’Brien S., Sennino B., Nakahara T. Rapid vascular regrowth in tumors after reversal of VEGF inhibition. J Clin Invest. 2006;116(10):2610–2621. doi: 10.1172/JCI24612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Bussard K.M., Mutkus L., Stumpf K., Gomez-Manzano C., Marini F.C. Tumor-associated stromal cells as key contributors to the tumor microenvironment. Breast Cancer Res: BCR. 2016;18(1):84. doi: 10.1186/s13058-016-0740-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Benjamin L.E., Hemo I., Keshet E. A plasticity window for blood vessel remodelling is defined by pericyte coverage of the preformed endothelial network and is regulated by PDGF-B and VEGF. Development. 1998;125(9):1591–1598. doi: 10.1242/dev.125.9.1591. [DOI] [PubMed] [Google Scholar]
- 189.Darland D., Massingham L., Smith S., Piek E., Saint-Geniez M., D'amore P. Pericyte production of cell-associated VEGF is differentiation-dependent and is associated with endothelial survival. Dev Biol. 2003;264(1):275–288. doi: 10.1016/j.ydbio.2003.08.015. [DOI] [PubMed] [Google Scholar]
- 190.Narayana A., Kelly P., Golfinos J., Parker E., Johnson G., Knopp E. Antiangiogenic therapy using bevacizumab in recurrent high-grade glioma: impact on local control and patient survival: clinical article. J Neurosurg. 2009;110(1):173–180. doi: 10.3171/2008.4.17492. [DOI] [PubMed] [Google Scholar]
- 191.Norden A., Young G., Setayesh K., Muzikansky A., Klufas R., Ross G. Bevacizumab for recurrent malignant gliomas Efficacy, toxicity, and patterns of recurrence. Neurology. 2008;70(10):779–787. doi: 10.1212/01.wnl.0000304121.57857.38. [DOI] [PubMed] [Google Scholar]
- 192.Jimenez-Valerio G., Martinez-Lozano M., Bassani N., Vidal A., Ochoa-de-Olza M., Suarez C. Resistance to antiangiogenic therapies by metabolic symbiosis in renal cell carcinoma PDX models and patients. Cell Rep. 2016;15(6):1134–1143. doi: 10.1016/j.celrep.2016.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Sofuni A., Iijima H., Moriyasu F., Nakayama D., Shimizu M., Nakamura K. Differential diagnosis of pancreatic tumors using ultrasound contrast imaging. J Gastroenterol. 2005;40(5):518–525. doi: 10.1007/s00535-005-1578-z. [DOI] [PubMed] [Google Scholar]
- 194.Kaur B., Tan C., Brat D.J. Genetic and hypoxic regulation of angiogenesis in gliomas. J Neurooncol. 2004;70(2):229–243. doi: 10.1007/s11060-004-2752-5. [DOI] [PubMed] [Google Scholar]
- 195.Marty M., Pivot X. The potential of anti-vascular endothelial growth factor therapy in metastatic breast cancer: clinical experience with anti-angiogenic agents, focusing on bevacizumab. Eur J Cancer. 2008;44(7):912–920. doi: 10.1016/j.ejca.2008.01.005. [DOI] [PubMed] [Google Scholar]
- 196.Griffioen A.W. Therapeutic approaches of angiogenesis inhibition: are we tackling the problem at the right level? Trends Cardiovasc Med. 2007;17(5):171–176. doi: 10.1016/j.tcm.2007.03.007. [DOI] [PubMed] [Google Scholar]
- 197.Miller K.D., Sweeney C.J., Sledge G.W., Jr Can tumor angiogenesis be inhibited without resistance? EXS. 2005;94:95–112. doi: 10.1007/3-7643-7311-3_7. [DOI] [PubMed] [Google Scholar]
- 198.Folkman J. Fighting cancer by attacking its blood supply. Sci Am. 1996;275(3):150–154. doi: 10.1038/scientificamerican0996-150. [DOI] [PubMed] [Google Scholar]
- 199.Kuusk T., Albiges L., Escudier B., Grivas N., Haanen J., Powles T. Antiangiogenic therapy combined with immune checkpoint blockade in renal cancer. Angiogenesis. 2017;20(2):205–215. doi: 10.1007/s10456-017-9550-0. [DOI] [PubMed] [Google Scholar]
- 200.Eisermann K., Fraizer G. The androgen receptor and VEGF: mechanisms of androgen-regulated angiogenesis in prostate cancer. Cancers (Basel) 2017;9(4) doi: 10.3390/cancers9040032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Cao Y. Future options of anti-angiogenic cancer therapy. Chin J Cancer. 2016;35:21. doi: 10.1186/s40880-016-0084-4. [DOI] [PMC free article] [PubMed] [Google Scholar]