Abstract
Alterations in the ratio of excitatory to inhibitory transmission are emerging as a common component of many nervous system disorders, including autism spectrum disorders (ASDs). Tonic γ-aminobutyric acidergic (GABAergic) transmission provided by peri- and extrasynaptic GABA type A (GABAA) receptors powerfully controls neuronal excitability and plasticity and, therefore, provides a rational therapeutic target for normalizing hyperexcitable networks across a variety of disorders, including ASDs. Our previous studies revealed tonic GABAergic deficits in principal excitatory neurons in the basolateral amygdala (BLA) in the Fmr1−/y knockout (KO) mouse model fragile X syndrome. To correct amygdala deficits in tonic GABAergic neurotransmission in Fmr1−/y KO mice, we developed a novel positive allosteric modulator of GABAA receptors, SGE-872, based on endogenously active neurosteroids. This study shows that SGE-872 is nearly as potent and twice as efficacious for positively modulating GABAA receptors as its parent molecule, allopregnanolone. Furthermore, at submicromolar concentrations (≤1 µM), SGE-872 is selective for tonic, extrasynaptic α4β3δ-containing GABAA receptors over typical synaptic α1β2γ2 receptors. We further find that SGE-872 strikingly rescues the tonic GABAergic transmission deficit in principal excitatory neurons in the Fmr1−/y KO BLA, a structure heavily implicated in the neuropathology of ASDs. Therefore, the potent and selective action of SGE-872 on tonic GABAA receptors containing α4 subunits may represent a novel and highly useful therapeutic avenue for ASDs and related disorders involving hyperexcitability of neuronal networks.
Keywords: fragile X syndrome, amygdala, GABAA receptor, GABA, positive allosteric modulator
Increasing evidence indicates that dysfunction in γ-aminobutyric acidergic (GABAergic) neurotransmission is a primary driver of neuronal network dysfunction in a variety of nervous system disorders. Pathological changes in GABAergic neurotransmission can alter excitatory/inhibitory (E/I) balance in local circuits and are associated with symptoms of a range of neurological and neurodeve-lopmental disorders, such as schizophrenia, epilepsy, and autism spectrum disorders (ASDs), including fragile X syndrome (FXS), Rett syndrome, tuberosclerosis, and idiopathic autism (Zoghbi, 2003; Maguire et al., 2005; Geschwind and Levitt, 2007; Wafford and Ebert, 2008; Maldonado-Avilés et al., 2009; Macdonald et al., 2010; Olmos-Serrano et al., 2010; Paluszkiewicz et al., 2011; Han et al., 2012, 2014; Fatemi et al., 2014). Furthermore, extrasynaptic, tonically active GABA type A (GABAA) receptors that contain the δ subunit in conjunction with α4 or α6 subunits are relatively ubiquitously expressed in key brain regions involved in the neuropathology of these disorders. These areas include the hippocampus (Stell et al., 2003), neocortex (Drasbek, 2005), cerebellum (Brickley et al., 2001), thalamus (Jia, 2005), and amygdala (Olmos-Serrano et al., 2010). Therefore, these receptors constitute a prime target for drug development to augment GABAergic tone, restore E/I balance, and improve cognitive and behavioral symptoms. In support of this concept, recent studies have revealed that pharmacological or genetic rescue of GABAergic neurotransmission can restore molecular and/or behavioral deficits in a variety of animal models of neurodevelopmental disorders, including ASDs (Olmos-Serrano et al., 2010; Han et al., 2012, 2014).
GABA is the main inhibitory neurotransmitter in the adult central nervous system (CNS) acting mainly through chloride-permeable, pentameric GABAA receptors composed of a variety of subunits (α1–6, β1–3, γ1– 3, δ, ε, θ, π, and ρ1–3). These receptors typically take the form of two α subunits and two β subunits, combined with either a γ or a δ subunit (Farrant and Nusser, 2005). The combination of GABAA receptor subunits that forms a functional receptor determines the regional and developmental expression, physiological and pharmacological properties, and the subcellular location (e.g., synaptic or extrasynaptic) of the receptor (Hevers and Lüddens, 1998; Pirker et al., 2000; Mody and Pearce, 2004; Brickley and Mody, 2012). Synaptically located receptors provide fast, phasic conductance by responding to large concentrations of GABA in the synapse (millimolar) and have high desensitization rates. GABA also displays relatively low affinity and high efficacy at these receptors. In contrast, peri- and extrasynaptic receptors, which most often contain δ, α4, α5, or α6 subunits, provide slow, tonic conductance by responding to relatively low concentrations of GABA (in the nanomolar to micromolar range) at lower desensitization rates, with GABA displaying very high affinity and lower efficacy than at their synaptic counterparts (Semyanov et al., 2004; Farrant and Nusser, 2005). These properties allow these GABAA receptors to modulate a potent, dynamic tonic conductance many times larger than that of the collective fast, phasic inhibition (Brickley et al., 1996; Rossi et al., 2003). Thus, tonic conductance crucially affects key cellular and neuronal network functions, such as intrinsic neuronal excitability (Bonin et al., 2007), integration of synaptic inputs (Mitchell and Silver, 2003; Semyanov et al., 2004), and synaptic plasticity (Martin et al., 2010).
Endogenous neurosteroid metabolites of progesterone, such as allopregnanolone, are potent positive allosteric modulators of GABAA receptors (Majewska, 1992) that can have therapeutic efficacy in disorders associated with E/I imbalances, such as epilepsy (Reddy and Rogawski, 2009), and in the BTBR animal model of autism (Han et al., 2014). Although neurosteroids can show particular efficacy at δ subunit-containing GABAA receptors (Wohlfarth et al., 2002), allopregnanolone and its 3β-methylated synthetic analog ganaxolone have potent positive modulation on a wide range of GABAA receptor subtypes, including those that express α1, α2, α3, and γ2L subunits (Carter et al., 1997). This “off-target” efficacy could contribute to unnecessary side effects associated with treatment.
To reduce this possibility and to target neurosteroid modulation specifically to tonic GABAergic transmission, we used allopregnanolone as a molecular platform to develop a novel neurosteroid compound, SGE-872. We show that SGE-872 has exceptional efficacy and selectivity for α4δ subunit-containing GABAA receptors. Furthermore, we tested the ability of this compound to rescue deficient tonic GABAergic currents in the basolateral amygdala (BLA) of the Fmr1−/y knockout (KO) mouse model of FXS, an ASD characterized by social withdrawal and emotional deficits associated with dysfunction of the amygdala (Markram and Markram, 2010). Principal excitatory neurons (PNs) of the BLA that crucially regulate sensory integration and fear modulation (Ehrlich et al., 2009) have deficient tonic GABAergic tone associated with neuronal hyperexcitability and E/I imbalance in Fmr1−/y KO mice (Olmos-Serrano et al., 2010; Martin et al., 2014). This study shows that application of SGE-872 to Fmr1−/y PNs strikingly corrects the tonic GABAergic deficit. Furthermore, at the concentration sufficient to rescue this defect, SGE-872 preferentially affects tonic GABAA receptors and not phasic receptors. Therefore, our data reveal that selective targeting of α4 subunits via a novel positive allosteric modulator can correct amygdala-based GABAergic deficits in an animal model of FXS and may serve to improve E/I balance more broadly across other critical brain regions involved in related syndromic and nonsyndromic etiologies of autism.
MATERIALS AND METHODS
Cell Cultures
The functional potency and efficacy of allopregnanolone and SGE-872 were assessed by whole-cell patch clamp of heterologously expressed GABAA receptors composed of either the α1β2γ2 or the α4β3δ subunits. LTK cells were stably transfected with the human α1β2γ2 subunits of the GABA receptor, and CHO cells were transiently transfected with the human α4β3δ subunits via the lipofectamine method (Dalby et al., 2004). Cells were passaged at a confluence of about 50–80% and then seeded onto 35-mm sterile culture dishes containing 2 ml culture complete medium without antibiotics or antimycotics. Although the two receptor subtypes were expressed in two different host cell systems, allopregnanolone showed similar pharmacology at α1β2γ2 and α4β3δ receptors, consistent with the literature on a variety of cell types (Belelli et al., 2002; Belelli and Lambert, 2005), suggesting that we did not see comparatively different pharmacology in the two populations resulting from differences in the host cell type.
Whole-Cell Patch Clamp of Transfected Cells
Whole-cell currents were measured with HEKA EPC-10 amplifiers in PatchMaster software or by using the high-throughput QPatch platform (Sophion, Stockholm, Sweden). Bath solution for all experiments contained (in mM) NaCl 137, KCl 4, CaCl2 1.8, MgCl2 1, HEPES 10, D-glucose 10, pH (NaOH) 7.4. Intracellular (pipette) solution contained (in mM) KCl 130, MgCl2 1, Mg-ATP 5, HEPES 10, EGTA 5, pH 7.2. During experiments, cells and solutions were maintained at room temperature (19–30°C). For manual patch-clamp recordings, cell culture dishes were placed on the dish holder of the microscope and continuously perfused (1 ml/min) with bath solution, and whole-cell recordings were obtained by manual patch clamp. For experiments with the QPatch system, cells were transferred as a suspension to the QPatch system in the bath solution, and automated whole-cell recordings were performed.
Cells were voltage clamped at a holding potential of −80 mV. For the analysis of test articles, two successive applications of 2 µM GABA were first applied, and then, after wash out, GABA receptors were stimulated again by 2 µM GABA after sequential preincubation of increasing concentrations of the test article. Preincubation duration was 30 sec, and the duration of the GABA stimulus was 4 sec. Test compounds were dissolved in dimethylsulfoxide to form stock solutions (10 mM) and were diluted to 0.01, 0.1, 1.0, and 10 µM in bath solution. All concentrations of test compounds were tested on each cell. The relative percentage of potentiation was defined as the peak amplitude in response to GABA EC20 in the presence of the test article divided by the peak amplitude in response to GABA EC20 alone, multiplied by 100. The ability of SGE-872 to activate GABAA receptors directly was assessed in cells expressing the α4β3δ subunits. Two successive applications of 2 µM GABA were first applied, and then, after wash out, 10 µM SGE-872 was applied. Data are expressed as the peak response to 2 µM GABA alone divided by the peak response to 10 µM SGE-872 alone.
Animal Use
Wild-type (control; strain name FVB.129P2; stock No. 4828) and Fmr1 KO mice (strain name FVB.129P2-Fmr1tm1Cgr/J; stock No. 4624) on the congenic FVB strain background were obtained from The Jackson Laboratory (Genetics Research, Bar Harbor, ME) and maintained as separate congenic stocks at Children’s National Medical Center. Animals were housed and utilized in accordance with protocols approved by the Institutional Animal Care and Use Committee.
Acute Brain Slice Preparation
Acute slices were prepared from postnatal day (P) 21–30 male control and Fmr1−/y mice. Animals were briefly anesthetized with CO2 and decapitated. Brains were removed quickly and placed in cold (4°C) sucrose-based oxygenated (95% O2/5% CO2) cutting solution composed of (in mM) sucrose 234, glucose 11, NaHCO3 26, KCl 2.5, NaH2PO4 · H2O 1.25, MgSO4 · 7H2O 10, and CaCl2 · H2O 0.5. Coronal slices containing the BLA were obtained with a slicing vibratome (VT1200s; Leica, Wetzlar, Germany) by removing the cerebellum with a perpendicular cut to the rostral–caudal plane and gluing the caudal side down on the vibratome stage submerged in cold cutting solution. Slice thickness was 300 µM for all experiments. The slices were immersed in oxygenated (95% O2/5% CO2) artificial cerebral spinal fluid (ACSF) at 34°C for 30–45 min. ACSF was composed of (in mM) NaCl 126, NaHCO3 26, glucose 10, KCl 2.5, NaH2PO4 · H2O 1.25, MgCl2 · 7H2O 2, and CaCl2 · 2H2O2, pH 7.4, with osmolarity maintained at 290–300 mOsm.
Slice Electrophysiology
For all experiments, slices were placed in a submerged slice chamber and continuously perfused with ACSF at 2–4 ml/min maintained at 26–28°C with an inline heater system (Warner Instruments, Hamden, CT). Slices were visualized on a fixed-stage upright microscope (Nikon, Tokyo, Japan) equipped with ×10 and ×60 objectives with differential interference contrast optics, infrared illumination, and an infrared-sensitive camera (Cohu, Poway, CA). Whole-cell patch clamp recordings were performed with glass pipettes with resistance of 2.0–4.0 MΩ when filled with intracellular solution. Access resistance of recordings was <25 MΩ and was monitored throughout the experiment with brief 5-mV steps every 20 sec. Data were discarded if the access resistance changed by >25%. Membrane potentials were adjusted for junction potential (12 mV). Data were acquired with a Multiclamp 700A amplifier and digitized with a Digidata 1322A in pClamp 9.2 (Molecular Devices, Sunnyvale, CA). All recordings were made from PNs initially identified visually as having a large, pyramid-like soma with two to seven primary dendrites and then identified physiologically as having long after hyperpolarizing potentials (AHPs; Sah et al., 2003) induced by prolonged hyperpolarizing current injections (600 msec). Cesium chloride-based intracellular solution contained (in mM) CsCl 135, HEPES 10, EGTA 10, QX-314 5, MgCl2 2, Mg-ATP 4, and Na-GTP 0.3, ECl− = 0 mV. Because cesium interferes with potassium currents involved in AHPs, care was taken to assess the AHP within 4–5 sec of cell membrane rupture before cesium could sufficiently diffuse into the cell. GABAergic tonic currents and phasic spontaneous inhibitory postsynaptic currents (sIPSCs) were recorded in voltage clamp and isolated by blocking ionotropic glutamatergic transmission with 6,7-dinitroquinoxaline-2,3-dione (20 µM final concentration; AMPA/kainate antagonist; Tocris Bioscience, Ellisville, MO) and DL-2-amino-5-phosphonopentanoic acid (50 µM final concentration, NMDA receptor antagonist; Tocris Bioscience) in the ACSF. All experimental compounds were applied locally with a gravity-fed Y-tube following baseline vehicle application. For tonic current recordings, 2 µM exogenous GABA was bath and locally applied during baseline and experimental compound application to stimulate extrasynaptic GABAA receptors. Before application of any experimental compounds or gabazine to measure tonic currents in these experiments, a whole-cell configuration was achieved, followed by exogenous GABA application for at least 10 min to allow the cell to equilibrate and achieve a steady-state tonic current. Spontaneous inhibitory postsynaptic currents (sIPSCs) were recorded in the absence of exogenous GABA.
Statistical Analysis
Tonic currents were acquired and analyzed as reported previously (Krook-Magnuson et al., 2008). Briefly, 10-sec samples were taken from voltage clamp recordings (Vh = –70 mV) under each experimental condition (baseline [IBSLN], compound [SGE-872 or allopregnanolone (Icmpd)] and gabazine [IGBZ, GABAA receptor antagonist SR-95531, 50 µM]). To minimize bias from phasic events, a Gaussian distribution was fit to the right side of an all-points histogram from each sample from a point 1–3 pA left of the peak (Glykys and Mody, 2007). The Gaussian peak determined the mean current for the sample. Total tonic current was calculated from the difference in mean baseline and gabazine currents (IGBZ – IBSLN), and compound-induced current was calculated from the difference in mean baseline and compound currents (Icmpd – IBSLN). To control for differences in cell size/capacitance, calculated currents were expressed as tonic current densities for each cell based on cell capacitance (current density = current [pA]/capacitance [pF]). Capacitance was determined in voltage clamp with brief 10-mV biphasic voltage steps delivered immediately after establishing the whole-cell configuration.
Action-potential-dependent sIPSCs were analyzed for frequency, amplitude, and decay kinetics before and after the application of test compounds in the absence of exogenous GABA. Averaged sIPSCs were fit with the double exponential function f(x) = Afaste−t/tau(fast) + Aslowe−t/tau(slow). Fitted sIPSCs were then used to determine the weighted time constant τd,w = [(Afast τfast) + (Aslow τslow)]/(Afast + Aslow). Phasic synaptic efficacy was estimated by integrating total IPSC charge per 1-sec interval (Huntsman et al., 1999).
All recordings were analyzed off-line (pClamp, RRI-D:rid_000085; Mini Analysis Program, RRID:SciRes_000143; Patchmaster, RRID:SciRes_000168). Values are mean ± SE. Two-tailed Student’s t-test was used for measures of evoked and tonic currents, sIPSC amplitude, frequency, weighted decay, and synaptic efficacy (Microcal Origin, RRID:rid_000069; Graphpad Prism, RRID:rid_000081). Paired Student’s t-test was used to determine significance for within-group comparisons between conditions (i.e., baseline and drug), and ANOVA with Bonferroni correction for multiple comparisons was used to test differences in baseline and drug conditions across genotypes (control and Fmr1−/y) or between compounds (allopregnanolone and SGE-872).
RESULTS
SGE-872 Is a Potent and Selective Modulator of GABAA Receptors
We first wanted to assess the pharmacology of SGE-872 compared with allopregnanolone. To accomplish this we used patch-clamp electrophysiology in mammalian cells expressing recombinant human GABAA receptors. GABAA receptors composed of the α1β2γ2 subunits were used as surrogates for synaptic receptors, and GABAA receptors composed of the α4β3δ subunits were used as surrogates for extrasynaptic receptors (Belelli and Lambert, 2005).
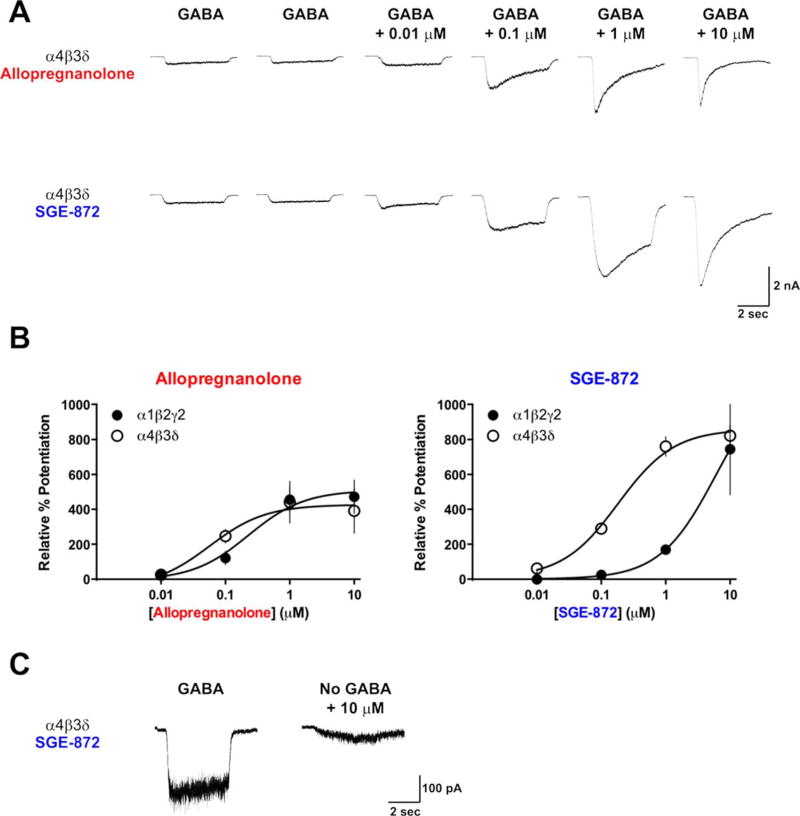
Allopregnanolone potentiated GABA-evoked currents in a concentration-dependent manner with an EC50 of 185 nM and Emax of 476% in cells expressing α1β2γ2 and an EC50 of 80 nM and Emax of 418% in cells expressing α4β3δ (Fig. 1A, top, Fig. 1B, left). In contrast, SGE-872 displayed greater selectivity for GABAA receptors composed of α4β3δ subunits compared with receptors composed of α1β2γ2 subunits. In cells expressing the α4β3δ subunits, SGE-872 potentiated GABA-evoked currents with an EC50 of 160 nM and Emax of 838% (Fig. 1A, bottom, Fig. 1B, right). However, in cells expressing α1β2γ2 subunits, SGE-872 was substantially less potent, and the concentration–response curve did not reach an asymptote by the highest concentration tested, so potency and efficacy values could not be estimated. At 10 µM, SGE-872 potentiated GABA-evoked currents by 744% ± 261% (mean ± SD, n = 3).
Fig. 1.
SGE-872 is a more potent and selective positive allosteric modulator than allopregnanolone. A: Representative current responses from LTK cells transfected with α4β3δ GABAA receptors in response to 2 sec of stimulus with either GABA (2 µM) or GABA plus each modulator at increasing concentrations. B: Dose–response curves for allopregnanolone (at left) and SGE-872 (right) indicate that SGE-872 potentiates α4β3δ-mediated GABAergic currents more than allopregnanolone at concentrations greater than 0.1 µM. In addition, SGE-872 is more selective for α4β3δ-containing receptors vs. α1β2γ2 receptors compared with allopregnanolone. C: The highest concentration tested (10 µM) minimally directly activates α4β3δ GABAA receptors in the absence of GABA.
The ability of SGE-872 to activate GABAA receptors directly was also investigated in cells that expressed α4β3δ subunits. 10 µM SGE-872 had minimal direct activation, evoking a current that was 22.9% ± 3.5% of the current evoked by an EC20 concentration of GABA (2 µM, n = 3; Fig. 1C).
SGE-872 Enhances Tonic GABAA Receptor-Mediated Currents in BLA PNs
GABAergic tone crucially regulates network function in the BLA (Ehrlich et al., 2009), and dysfunction in the amygdala has been associated with symptoms of ASDs (Baron-Cohen et al., 2000; Amaral et al., 2003; Markram and Markram, 2010). In animal models of ASDs, GABAergic alterations exist in the BLA in particular (Olmos-Serrano et al., 2010; Paluszkiewicz et al., 2011; Vislay et al., 2013) Furthermore, in the Fmr1−/y KO mouse model of FXS, PNs of the BLA display deficits in tonic GABAergic transmission in association with increases in E/I balance that can be rescued by tonic GABAergic augmentation (Olmos-Serrano et al., 2010). Specifically, Fmr1−/y PNs show a decreased physiological response to the GABAA receptor superagonist 4,5, 6,7-tetrahydroisoxa-zolo[5,4-c]pyridin-3-ol (THIP), also known as gaboxadol, at a concentration shown to affect α4δ-containing receptors preferentially (Brown et al., 2002; Stórustovu and Ebert, 2006; Martin et al., 2014), consistent with decreased function of these receptors in these cells. With these findings, we next tested the ability of allopregnanolone and SGE-872 to enhance tonic GABAA receptor-mediated currents in BLA principal neurons in acute brain slices from both control and Fmr1−/y mice (Fig. 2).
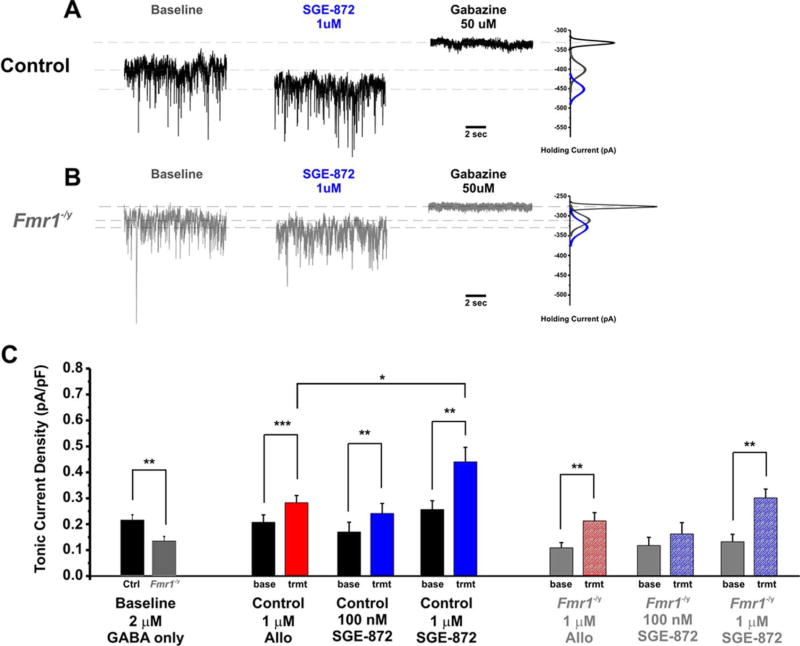
Fig. 2.
Allopregnanolone and SGE-872 significantly potentiate tonic currents in principal neurons of the BLA. A,B: Representative whole-cell voltage clamp traces recorded from control (A) and Fmr1−/y (B) principal neurons show 10-sec samples before (baseline) and after (SGE-872) direct application of SGE-872 1 µM in the presence of 2 µM exogenous GABA, followed by application of gabazine 50 µM (Vhold = –70 mV). Gaussian distributions (right) for each sample indicate the differences in mean holding current at each condition. C: Baseline tonic density is decreased in Fmr1−/y cells in the presence of 2 µM GABA (left). Pairwise comparisons show that allopregnanolone and SGE-872 significantly potentiate baseline tonic current density in control and Fmr1−/y cells. However, Fmr1−/y cells require a higher concentration of SGE-872 (1 µM vs. 100 nM) to have a significant effect. Allopregnanolone also significantly potentiates tonic current density in both genotypes. However, in wild-type cells, SGE-872 (1 µM) has a stronger potentiation than allopregnanolone (ANOVA). *P < 0.05, **P < 0.01, ***P < 0.001.
First, we observed that baseline tonic GABAA receptor-mediated current densities across all experimental groups in the presence of exogenous GABA (2 µM) were significantly decreased in Fmr1−/y cells compared with control neurons (total control baseline, 0.21 ± 0.02 pA/pF, n = 22; Fmr1−/y baseline, 0.12 ± 0.02, n = 23; ANOVA with Bonferroni correction, F[1,43] = 0.290, n = 45, P = 0.003; Fig. 2C). Current densities were determined by the change in holding current following the application of the GABAA receptor antagonist gabazine (50 µM). This result is consistent with reduced tonic GABAergic tone in the Fmr1−/y BLA, as we have previously characterized (Olmos-Serrano et al., 2010). Application of SGE-872 at a low concentration (100 nM) significantly enhanced tonic current density in control cells (control baseline, 0.17 ± 0.04 pA/pF; control SGE-872 100 nM, 0.24 ± 0.04 pA/pF; n = 8, paired t-test, t[7] = −5.18, P = 0.001) but not in Fmr1−/y cells (Fmr1−/y baseline, 0.12 ± 0.03 pA/pF; Fmr1−/y SGE-872 100 nM, 0.16 ± 0.04 pA/pF; n = 7, paired t-test, t[6] = −1.75, P = 0.13). In contrast, application of a higher concentration (1 µM) of SGE-872 significantly enhanced tonic current density in both control and Fmr1−/y BLA PNs (Fig. 2A,B; control baseline, 0.26 ± 0.03 pA/pF; control 1 µM SGE-872, 0.44 ± 0.06 pA/pF; n = 8, paired t-test, t[7] = −4.62, P = 0.002; Fmr1−/y baseline, 0.13 ± 0.03 pA/pF; Fmr1−/y 1 µM SGE-872, 0.30 ± 0.03 pA/pF; n = 8, paired t-test, t[7] = −4.55, P = 0.003; Fig. 2C). Furthermore, treatment with SGE-872 at 1 µM restored tonic current density in Fmr1−/y PNs to control baseline levels (Fmr1−/y 0.30 pA/pF vs. control 0.26 pA/pF, ANOVA with Bonferroni correction, F[1,14] = 0.899, n = 15, P = 0.360).
SGE-872 Is More Effective Than Allopregnanolone for Augmenting Tonic Current in BLA PNs
Allopregnanolone (1 µM) also significantly enhanced tonic GABAA receptor-mediated current density in both control and Fmr1−/y PNs (control baseline, 0.20 ± 0.03 pA/pF; control allopregnanolone 1 µM, 0.28 ± 0.03; n = 9, paired t-test, t[8] = −5.30, P = 0.0007; Fmr1−/y baseline, 0.10 ± 0.02 pA/pF; Fmr1−/y allopregnanolone 1 µM, 0.21 ± 0.03 pA/pF; n = 7, paired t-test, t[6] = –4.87, P = 0.003). However, in control cells but not in Fmr1−/y cells, SGE-872 demonstrated a stronger potentiation of these current densities than allopregnanolone, consistent with higher efficacy of SGE-872 on recombinant GABAA receptors shown in Figure 1 (control allopregnanolone 1 µM, 0.28 ± 0.03 pA/pF; control SGE-872 1 µM, 0.44 ± 0.06 pA/pF; ANOVA with Bonferroni correction, F[1,15] = 7.02, P = 0.018; Fmr1−/y allopregnanolone 1 µM, 0.21 ± 0.03 pA/pF; Fmr1−/y SGE-82 1 µM, 0.30 ± 0.03 pA/pF; ANOVA with Bonferroni correction, F[1,13] = 4.02, P = 0.07; Fig. 2C).
SGE-872 Demonstrates Specificity for Tonic Over Phasic GABAA Receptors in BLA PNs
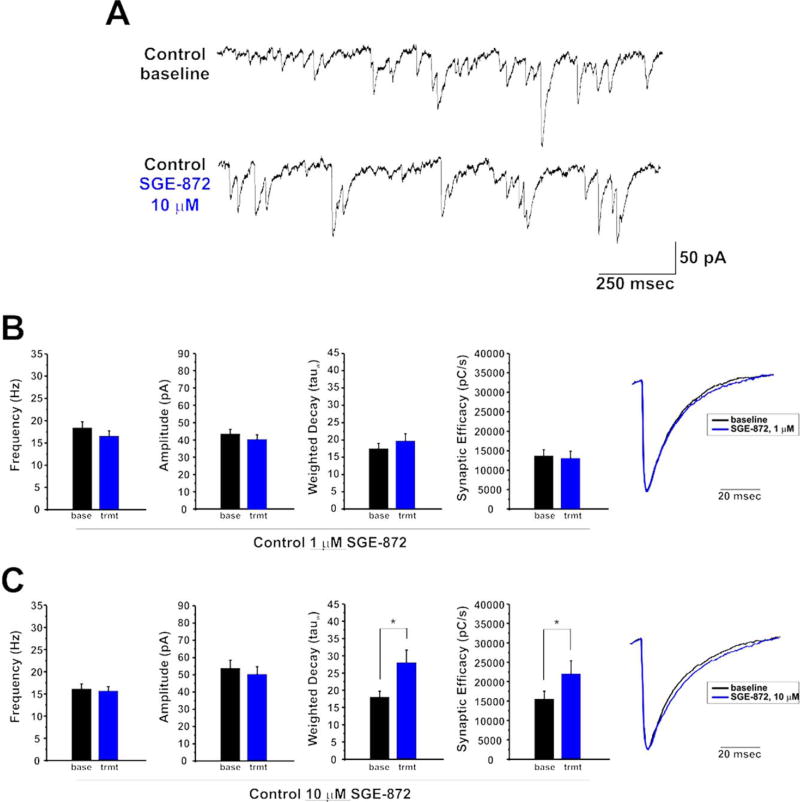
Characterization of GABA currents in a recombinant system demonstrated that SGE-872 is more active at putative extrasynaptic receptors (α4β3δ) than at putative synaptic receptors (α1β2γ2) at low concentrations (≤1 µM; Fig. 1B). Therefore, we wanted to test whether this preference also existed in BLA PNs. To accomplish this, we recorded sIPSCs in the absence of exogenous GABA and compared baseline sIPSC properties with those at low and high concentrations of SGE-872 (Fig. 3). Upon application of 1 µM SGE-872 to control BLA PNs, a concentration that significantly augmented tonic current density (Fig. 2A,C), we observed no significant change in sIPSC frequency, amplitude, weighted decay constant, or estimated phasic synaptic efficacy (frequency baseline, 18.3 ± 1.36 Hz; frequency SGE-872 1 µM, 16.4 ± 1.45 Hz; t[8] = 2.11, P = 0.070; amplitude baseline, 43.8 ± 2.56 pA; amplitude SGE-872 1 µM, 40.6 ± 2.66 pA; t[8] = 2.10, P = 0.072; tauω baseline, 17.4 ± 1.53; tauω SGE-872 1 µM, 19.6 ± 2.05; t[8] = –1.27, P = 0.241; efficacy baseline, 13,571.3 ± 1,608.1 pC/sec; efficacy SGE-872, 1 µM 12,966.4 ± 1,820.3 pC/sec, t[8] = 0.292, P = 0.778; n = 9, paired t-test; Fig. 3B). However application of 10 µM SGE-872 caused significant changes in sIPSC kinetics and an associated increase in phasic GABAergic synaptic efficacy, indicating significant enhancement of phasic synaptic receptors in addition to tonically active receptors (frequency baseline, 16.1 ± 1.07 Hz; frequency SGE-872 1 µM, 15.7 ± 1.01 Hz; t[6] = 0.556, P = 0.598; amplitude baseline, 53.0 ± 4.56 pA; amplitude SGE872 1 µM, 50.4 ± 4.28 pA; t[6] = 1.47, P = 0.192; tauω baseline, 18.0 ± 1.68; tauω SGE-872 1 µM, 28.1 ± 3.63; t[6] = –2.91, P = 0.027; efficacy baseline, 15,514.0 ± 2,004.3 pC/sec; efficacy SGE-872 1 µM, 22,057.3 ± 3,352.8 pC/sec; t[6] = –2.38, P = 0.027; n = 7, paired t-test; Fig. 3A,C). Therefore, SGE-872 preferentially modulates tonically active GABAA receptors at low concentrations (≤ 1 µM) compared with high concentrations (10 µM) in mouse BLA PNs.
Fig. 3.
SGE-872 is selective for tonic vs. phasic GABAA receptors at low concentrations. A: Representative voltage clamp traces of sIPSCs recorded in a principal neuron in the BLA (all measurements taken from control brain slices) before (top) and after (bottom) application of SGE-872 (10 µM) in the absence of exogenous GABA (Vhold = –70 mV). B,C: Quantification of changes in sIPSC properties of cells exposed to SGE-872 at 1 µM (B) and 10 µM (C) indicates average sIPSC frequency, amplitude, weighted decay, and overall GABAergic synaptic efficacy before and after SGE-872 treatment. Representative average sIPSC waveforms (far right, scaled for amplitude) demonstrate changes in weighted decay constant (tauω) after SGE-872 application. At 1 µM, SGE-872 had no significant effect on any sIPSC properties (B). In contrast, at 10 µM, SGE-872 significantly increased both the weighted decay constant (tauω) of sIPSCs and the total GABAergic synaptic efficacy (C). *P < 0.05, paired t-test.
DISCUSSION
Tonic GABAergic Transmission in Neurodevelopmental Disorders
Alterations in GABAergic transmission are pervasive in ASDs and other related neurodevelopmental disorders such as schizophrenia and tuberosclerosis (Chao et al., 2010; Markram and Markram, 2010; Pizzarelli and Cherubini, 2011; Rudolph and Mohler, 2014). These disruptions affect local and regional network synchronization, synaptic plasticity, and neuronal excitability, all of which rely heavily on tonic GABAergic conductance (Pizzarelli and Cherubini, 2011; Brickley and Mody, 2012). The power of dysfunctional GABAergic neurotransmission has recently been exemplified by findings showing that rescue of GABAergic signaling alone can ameliorate dysfunction in animal models of autism (Han et al., 2012, 2014).
In FXS specifically, multiple studies have revealed hyperexcitability and/or hypersynchronization of neuronal networks in key brain regions such as sensory cortex (Gibson et al., 2008; Gonçalves et al., 2013), prefrontal cortex (Testa-Silva et al., 2012), and amygdala (Olmos-Serrano et al., 2010; Martin et al., 2014), brain regions that contribute to core symptoms of ASDs such as hypersensitivity, anxiety, social withdrawal, and cognitive impairment (Markram and Markram, 2010). Tonic GABAergic conductance mediated by δ subunit-containing GABAA receptors can modulate excitability (Semyanov et al., 2004) and regulate γ-frequency band oscillations (Mann and Mody, 2010) that are crucial for regional synchronization and cognitive processing (Fries, 2009). Therefore, given the widespread disruption in GABAergic tone in the FXS brain, potent and specific modulation of tonic inhibitory tone by SGE-872 has the potential to normalize both local and regional neuronal communication and improve overall brain function. Additional testing in behavioral paradigms is required to determine whether the enhancement of tonic GABAergic tone by SGE-872 can also improve behavioral symptoms in FXS and other animal models of ASDs.
Expression of the δ subunit (mRNA and protein) is broadly decreased in the FXS brain in association with the functional loss of fragile X mental retardation protein (D’Hulst et al., 2006; Gantois et al., 2006), the transcriptional regulator that underlies dysfunction in the syndrome (Bear et al., 2004). Furthermore, tonic GABAergic transmission is compromised in key brain regions, such as the subiculum (Curia et al., 2009) and the BLA (Olmos-Serrano et al., 2010; Martin et al., 2014). Here we confirm previous data revealing significantly lower tonic GABAergic currents in BLA PNs in Fmr1−/y mice compared with controls and extend this work to reveal that this deficit is rescued by application of SGE-872. Previous molecular and electrophysiological analyses have indicated that BLA PNs likely express at least three distinct tonically active, extrasynaptic GABAA receptor subtypes, the δ, α5, and α3 subunit-containing receptors (Olmos-Serrano et al., 2010; Marowsky et al., 2012; Martin et al., 2014). From the determined specificity of SGE-872, δ subunit-containing receptors expressed with α4 likely mediate the increased tonic currents that we observe. However, because δ subunit expression is probably reduced in the Fmr1−/y BLA (Gantois et al., 2006; Martin et al., 2014), we cannot discount the possible participation of uncharacteristically expressed GABAA receptors that contain α4 in the absence of δ in the augmented currents. Regardless of the mechanism, our results reveal that selective α4 targeting is highly efficacious for correcting underlying GABAergic alterations in the Fmr1−/y KO BLA.
SGE-872 Potency and Specificity for Extrasynaptic GABAA Receptors
Our characterization of SGE-872 introduces a novel neurosteroid-based compound that acts as a potent, selective positive allosteric modulator at tonically active GABAA receptors. In transfected cells, SGE-872 clearly showed a preference for putative extrasynaptic (α4β3δ) GABAA receptors over putative synaptic receptors (α1β2γ2) with an ability to gate α4β3δ receptors directly at the highest concentration tested (10 µM). The compound also exhibited greater efficacy at lower concentrations than the endogenous neurosteroid allopregnanolone, at least at α4β3δ-containing receptors. In addition, we observed some desensitization of the GABA response in the presence of SGE-872 in these transfected cells, especially at higher concentrations (1 and 10 µM; Fig. 1). To assess these effects, we attempted an additional experiment to assess steady-state GABA current in α4β3δ-containing receptors in the presence of SGE-872 (data not shown) in which we first applied GABA for 90 sec to allow it to reach steady state, followed by application of SGE-872 for 90 sec to observe a second, higher potentiated steady state. Unfortunately, we did not obtain a sufficient number of stable recordings in this experiment to attain an accurate measurement of steady-state potentiation in α4β3δ-containing receptors.
We did, however, observe sustained augmentation of the tonic GABAergic current by SGE-872 in PNs in the mouse BLA in both controls and the Fmr1−/y KO mouse model of FXS. In the Fmr1−/y slices, SGE-872 enhanced tonic GABAergic currents to control levels at a concentration that preferentially modulated extrasynaptic rather than synaptic GABAA receptors.
When tested in the Fmr1−/y KO model of FXS, SGE-872 was less effective at lower concentrations (100 nM) in Fmr1−/y KO neurons than in control PNs. This result was expected given the reduced expression of GABAA receptor subunits, including the δ and the α4 subunits in the FXS brain (D’Hulst et al., 2006). Despite the decreased effectiveness of the compound to enhance tonic GABAergic current in Fmr1−/y PNs, application at 1 µM showed significant efficacy, indicating that SGE-872 can effectively rescue the tonic current deficit in the Fmr1−/y BLA. In addition, further evaluation showed that, despite potentiation of α1β2γ2 receptors by ~200% in transfected cells, response of phasic events (sIPSCs) to 1 µM application in the acute control brain slice was not significantly affected by SGE-872 in control PNs. Several factors could account for the lack of enhancement of sIPSCs at this concentration (1 µM), including a differential GABAA receptor pool in the BLA PNs (i.e., more α2/α3 subunit-containing receptors than α1; Vislay et al., 2013), lack of penetration of the compound into the slice, strong activity of GABA transporters in the synaptic cleft resulting in reduced effect, and/or desensitization of the receptor to the presence of the modulator. However, at 10 µM, application of SGE-872, phasic receptors showed increased efficacy, accounted for by increased weighted decay of synaptic events but no significant increase in sIPSC amplitude, perhaps because neurosteroids likely primarily potentiate GABAA receptors by increasing receptor open time (Barker et al., 1987).
Potential Clinical Implications of Findings
Given the crucial role of tonic GABAergic transmission in regulating neuronal excitability and synaptic plasticity (Brickley and Mody, 2012), extrasynaptic receptors have become targets of drug development for a wide range of CNS disorders, such as insomnia, anxiety, depression, schizophrenia, and autism (Wafford and Ebert, 2006; Rudolph and Mohler, 2014). Accordingly, attempts to synthesize α4δ-selective molecules have produced δ subunit-selective agonists such as THIP and gaboxadol (Wafford and Ebert, 2008) and positive allosteric modulators with specificity for α4δ-GABAA receptors such as DS2 (Wafford et al., 2009) and AA29504 (Hoestgaard-Jensen et al., 2010). Although deficits in tonic GABAergic conductance represent only one of the many defects in GABAergic signaling in Fmr1−/y mice, including GABA production, inhibitory synapse number, receptor expression, and GABA production and release (Olmos-Serrano et al., 2010), augmentation of the tonic current acts as a powerful regulator of increased cellular excitability and a potential therapeutic tool. For instance, gaboxadol alone can physiologically rescue the hyperexcitable phenotype of Fmr1−/y PNs in the acute amygdala brain slice (Olmos-Serrano et al., 2010) and rescue at least one behavioral phenotype in the Fmr1−/y mouse, hyperactivity (Olmos-Serrano et al., 2011). However, determination of the full potential therapeutic value of tonic GABAergic modulators for FXS requires additional behavioral studies with SGE-872 and related compounds.
Here we show that SGE-872 consistently displays stronger positive allosteric modulation of tonic GABAA receptors than the principle endogenous neurosteroid allopregnanolone in both human transfected cells and in BLA PNs in acute mouse brain slices. In fact, SGE-872 displays efficacy at α4β3δ GABAA receptors equal to or greater than that of DS2, AA29504, allopregnanolone, or its 3β-methylated homologue ganaxolone (Carter et al., 1997) at concentrations selective for α4δ subunit-containing receptors. This superior efficacy and selectivity signify a possible therapeutic advantage of SGE-872 for treatment of disorders that have significantly reduced expression of the α4δ GABAA receptors, such as schizophrenia (Maldonado-Avilés et al., 2009), FXS (D’Hulst et al., 2006; Gantois et al., 2006), idiopathic ASDs (Fatemi et al., 2010, 2014), and those disorders in which allopregnanolone has shown possible efficacy for improving symptoms, including generalized anxiety (Schüle et al., 2014), depression (Bäckström et al., 2014), posttraumatic stress disorder (Pinna, 2014), and epilepsy (Carver and Reddy, 2013).
In summary, this study shows that SGE-872 is a potent and selective positive allosteric modulator of α4 subunit-containing GABAA receptors that augments deficient tonic GABAergic tone in the BLA of the Fmr1−/y KO mouse model of FXS. Therefore, augmentation of tonic GABAergic neurotransmission may prove effective for improving amygdala-based symptoms in FXS and related ASDs.
SIGNIFICANCE.
This study shows the potential of targeting a particular type of neurotransmission, tonic γ-aminobutyric acidergic transmission, with a novel, selective compound, SGE-872, to treat neurophysiological symptoms such as social anxiety in individuals with fragile X syndrome. SGE-872 and related molecules may especially improve those symptoms related to the amygdala, a primary brain region responsible for the regulation of anxiety.
Acknowledgments
Contract grant sponsor: SAGE Therapeutics, Inc.
The authors thank the members of the Corbin and Huntsman labioratories for helpful discussions.
ROLE OF AUTHORS
BSM, MAA, and JJD performed the experiments and analyzed the data. The work was overseen and directed by JGC, MMH and JJD with intellectual contribution/data analysis input from GM-B, CML, FGS, AJR, MAA. All authors collaborated on editing and writing the article.
Footnotes
CONFLICT OF INTEREST STATEMENT
GM-B, CML, FGS, AJR, MAA, and JJD are currently or were formerly employed by Sage Therapeutics, Inc. Joshua G Corbin and Molly M Huntsman served as paid consultants on the Sage Therapeutics Scientific Advisory Board.
References
- Amaral DG, Bauman MD, Schumann CM. The amygdala and autism: implications from nonhuman primate studies. Genes Brain Behav. 2003;2:295–302. doi: 10.1034/j.1601-183x.2003.00043.x. [DOI] [PubMed] [Google Scholar]
- Bäckström T, Bixo M, Johansson M, Nyberg S, Ossewaarde L, Ragagnin G, Savic I, Strömberg J, Timby E, van Broekhoven F, van Wingen G. Allopregnanolone and mood disorders. Prog Neurobiol. 2014;113:88–94. doi: 10.1016/j.pneurobio.2013.07.005. [DOI] [PubMed] [Google Scholar]
- Barker JL, Harrison NL, Lange GD, Owen DG. Potentiation of gamma-aminobutyric-acid-activated chloride conductance by a steroid anaesthetic in cultured rat spinal neurones. J Physiol. 1987;386:485–501. doi: 10.1113/jphysiol.1987.sp016547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baron-Cohen S, Ring HA, Bullmore ET, Wheelwright S, Ashwin C, Williams SC. The amygdala theory of autism. Neurosci Biobehav Rev. 2000;24:355–364. doi: 10.1016/s0149-7634(00)00011-7. [DOI] [PubMed] [Google Scholar]
- Bear MF, Huber KM, Warren ST. The mGluR theory of fragile X mental retardation. Trends Neurosci. 2004;27:370–377. doi: 10.1016/j.tins.2004.04.009. [DOI] [PubMed] [Google Scholar]
- Belelli D, Lambert JJ. Neurosteroids: endogenous regulators of the GABAA receptor. Nat Rev Neurosci. 2005;6:565–575. doi: 10.1038/nrn1703. [DOI] [PubMed] [Google Scholar]
- Belelli D, Casula A, Ling A, Lambert JJ. The influence of subunit composition on the interaction of neurosteroids with GABAA receptors. Neuropharmacology. 2002;43:651–661. doi: 10.1016/s0028-3908(02)00172-7. [DOI] [PubMed] [Google Scholar]
- Bonin RP, Martin LJ, MacDonald JF, Orser BA. Alpha5 GABAA receptors regulate the intrinsic excitability of mouse hippocampal pyramidal neurons. J Neurophysiol. 2007;98:2244–2254. doi: 10.1152/jn.00482.2007. [DOI] [PubMed] [Google Scholar]
- Brickley SG, Mody I. Extrasynaptic GABAA receptors: their function in the CNS and implications for disease. Neuron. 2012;73:23–34. doi: 10.1016/j.neuron.2011.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brickley SG, Cull-Candy SG, Farrant M. Development of a tonic form of synaptic inhibition in rat cerebellar granule cells resulting from persistent activation of GABAA receptors. J Physiol. 1996;497:753–759. doi: 10.1113/jphysiol.1996.sp021806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brickley SG, Revilla V, Cull-Candy SG, Wisden W, Farrant M. Adaptive regulation of neuronal excitability by a voltage-independent potassium conductance. Nature. 2001;409:88–92. doi: 10.1038/35051086. [DOI] [PubMed] [Google Scholar]
- Brown N, Kerby J, Bonnert TP, Whiting PJ, Wafford KA. Pharmacological characterization of a novel cell line expressing human alpha(4)beta(3)delta GABAA receptors. Br J Pharmacol. 2002;136:965–974. doi: 10.1038/sj.bjp.0704795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter RB, Wood PL, Wieland S, Hawkinson JE, Belelli D, Lambert JJ, White HS, Wolf HH, Mirsadeghi S, Tahir SH, Bolger MB, Lan NC, Gee KW. Characterization of the anticonvulsant properties of ganaxolone (CCD 1042; 3alpha-hydroxy-3beta-methyl-5alpha-pregnan-20-one), a selective, high-affinity, steroid modulator of the gamma-aminobutyric acid(A) receptor. J Pharmacol Exp Ther. 1997;280:1284–1295. [PubMed] [Google Scholar]
- Carver C, Reddy D. Neurosteroid interactions with synaptic and extrasynaptic GABAA receptors: regulation of subunit plasticity, phasic and tonic inhibition, and neuronal network excitability. Psychopharmacology. 2013;230:151–188. doi: 10.1007/s00213-013-3276-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chao H-T, Chen H, Samaco RC, Xue M, Chahrour M, Yoo J, Neul JL, Gong S, Lu H-C, Heintz N, Ekker M, Rubenstein JLR, Noebels JL, Rosenmund C, Zoghbi HY. Dysfunction in GABA signalling mediates autism-like stereotypes and Rett syndrome phenotypes. Nature. 2010;468:263–269. doi: 10.1038/nature09582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curia G, Papouin T, Séguéla P, Avoli M. Downregulation of tonic GABAergic inhibition in a mouse model of fragile X syndrome. Cereb Cortex. 2009;19:1515–1520. doi: 10.1093/cercor/bhn159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalby B, Cates S, Harris A, Ohki EC, Tilkins ML, Price PJ, Ciccarone VC. Advanced transfection with Lipofectamine 2000 reagent: primary neurons, siRNA, and high-throughput applications. Methods. 2004;33:95–103. doi: 10.1016/j.ymeth.2003.11.023. [DOI] [PubMed] [Google Scholar]
- D’Hulst C, de Geest N, Reeve SP, van Dam D, de Deyn PP, Hassan BA, Kooy RF. Decreased expression of the GABAA receptor in fragile X syndrome. Brain Res. 2006;1121:238–245. doi: 10.1016/j.brainres.2006.08.115. [DOI] [PubMed] [Google Scholar]
- Drasbek KR. THIP, a hypnotic and antinociceptive drug, enhances an extrasynaptic GABAA receptor-mediated conductance in mouse neocortex. Cereb Cortex. 2005;16:1134–1141. doi: 10.1093/cercor/bhj055. [DOI] [PubMed] [Google Scholar]
- Ehrlich I, Humeau Y, Grenier F, Ciocchi S, Herry C, Lüthi A. Amygdala inhibitory circuits and the control of fear memory. Neuron. 2009;62:757–771. doi: 10.1016/j.neuron.2009.05.026. [DOI] [PubMed] [Google Scholar]
- Farrant M, Nusser Z. Variations on an inhibitory theme: phasic and tonic activation of GABAA receptors. Nat Rev Neurosci. 2005;6:215–229. doi: 10.1038/nrn1625. [DOI] [PubMed] [Google Scholar]
- Fatemi SH, Reutiman TJ, Folsom TD, Rooney RJ, Patel DH, Thuras PD. mRNA and protein levels for GABAAalpha4, alpha5, beta1 and GABABR1 receptors are altered in brains from subjects with autism. J Autism Dev Disord. 2010;40:743–750. doi: 10.1007/s10803-009-0924-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fatemi SH, Reutiman TJ, Folsom TD, Rustan OG, Rooney RJ, Thuras PD. Downregulation of GABAA receptor protein subunits α6, β2, δ, ε, γ2, θ, and ρ2 in superior frontal cortex of subjects with autism. J Autism Dev Disord. 2014;44:1833–1845. doi: 10.1007/s10803-014-2078-x. [DOI] [PubMed] [Google Scholar]
- Fries P. Neuronal gamma-band synchronization as a fundamental process in cortical computation. Annu Rev Neurosci. 2009;32:209–224. doi: 10.1146/annurev.neuro.051508.135603. [DOI] [PubMed] [Google Scholar]
- Gantois I, Vandesompele J, Speleman F, Reyniers E, D’Hooge R, Severijnen L-A, Willemsen R, Tassone F, Kooy RF. Expression profiling suggests underexpression of the GABAA receptor subunit delta in the fragile X knockout mouse model. Neurobiol Dis. 2006;21:346–357. doi: 10.1016/j.nbd.2005.07.017. [DOI] [PubMed] [Google Scholar]
- Geschwind DH, Levitt P. Autism spectrum disorders: developmental disconnection syndromes. Curr Opin Neurobiol. 2007;17:103–111. doi: 10.1016/j.conb.2007.01.009. [DOI] [PubMed] [Google Scholar]
- Gibson JR, Bartley AF, Hays SA, Huber KM. Imbalance of neo-cortical excitation and inhibition and altered UP states reflect network hyperexcitability in the mouse model of fragile X syndrome. J Neurophysiol. 2008;100:2615–2626. doi: 10.1152/jn.90752.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glykys J, Mody I. The main source of ambient GABA responsible for tonic inhibition in the mouse hippocampus. J Physiol. 2007;582:1163–1178. doi: 10.1113/jphysiol.2007.134460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonçalves JT, Anstey JE, Golshani P, Portera-Cailliau C. Circuit level defects in the developing neocortex of fragile X mice. Nat Neurosci. 2013;16:903–909. doi: 10.1038/nn.3415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han S, Tai C, Westenbroek RE, Yu FH, Cheah CS, Potter GB, Rubenstein JL, Scheuer T, de la Iglesia HO, Catterall WA. Autistic-like behaviour in Scn1a+/− mice and rescue by enhanced GABA-mediated neurotransmission. Nature. 2012;489:385–390. doi: 10.1038/nature11356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han S, Tai C, Jones CJ, Scheuer T, Catterall WA. Enhancement of inhibitory neurotransmission by GABA. Neuron. 2014;81:1282–1289. doi: 10.1016/j.neuron.2014.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hevers W, Lüddens H. The diversity of GABAA receptors. Pharmacological and electrophysiological properties of GABAA channel subtypes. Mol Neurobiol. 1998;18:35–86. doi: 10.1007/BF02741459. [DOI] [PubMed] [Google Scholar]
- Hoestgaard-Jensen K, Dalby NO, Wolinsky TD, Murphey C, Jones KA, Rottländer M, Frederiksen K, Watson WP, Jensen K, Ebert B. Pharmacological characterization of a novel positive modulator at alpha 4 beta 3 delta-containing extrasynaptic GABAA receptors. Neuropharmacology. 2010;58:702–711. doi: 10.1016/j.neuropharm.2009.12.023. [DOI] [PubMed] [Google Scholar]
- Huntsman MM, Porcello DM, Homanics GE, DeLorey TM, Huguenard JR. Reciprocal inhibitory connections and network synchrony in the mammalian thalamus. Science. 1999;283:541–543. doi: 10.1126/science.283.5401.541. [DOI] [PubMed] [Google Scholar]
- Jia F. An extrasynaptic GABAA receptor mediates tonic inhibition in thalamic VB neurons. J Neurophysiol. 2005;94:4491–4501. doi: 10.1152/jn.00421.2005. [DOI] [PubMed] [Google Scholar]
- Krook-Magnuson EI, Li P, Paluszkiewicz SM, Huntsman MM. Tonically active inhibition selectively controls feed-forward circuits in mouse barrel cortex. J Neurophysiol. 2008;100:932–944. doi: 10.1152/jn.01360.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macdonald RL, Kang J-Q, Gallagher MJ. Mutations in GABAA receptor subunits associated with genetic epilepsies. J Physiol. 2010;588:1861–1869. doi: 10.1113/jphysiol.2010.186999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maguire JL, Stell BM, Rafizadeh M, Mody I. Ovarian cycle-linked changes in GABAA receptors mediating tonic inhibition alter seizure susceptibility and anxiety. Nat Neurosci. 2005;8:797–804. doi: 10.1038/nn1469. [DOI] [PubMed] [Google Scholar]
- Majewska MD. Neurosteroids: endogenous bimodal modulators of the GABAA receptor. Mechanism of action and physiological significance. Prog Neurobiol. 1992;38:379–395. doi: 10.1016/0301-0082(92)90025-a. [DOI] [PubMed] [Google Scholar]
- Maldonado-Avilés JG, Curley AA, Hashimoto T, Morrow AL, Ramsey AJ, O’Donnell P, Volk DW, Lewis DA. Altered markers of tonic inhibition in the dorsolateral prefrontal cortex of subjects with schizophrenia. Am J Psychiatry. 2009;166:450–459. doi: 10.1176/appi.ajp.2008.08101484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann EO, Mody I. Control of hippocampal gamma oscillation frequency by tonic inhibition and excitation of interneurons. Nat Neurosci. 2010;13:205–212. doi: 10.1038/nn.2464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markram K, Markram H. The intense world theory: a unifying theory of the neurobiology of autism. Front Hum Neurosci. 2010;4:224. doi: 10.3389/fnhum.2010.00224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marowsky A, Rudolph U, Fritschy J-M, Arand M. Tonic inhibition in principal cells of the amygdala: a central role for 3 subunit-containing GABAA receptors. J Neurosci. 2012;32:8611–8619. doi: 10.1523/JNEUROSCI.4404-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin BS, Corbin JG, Huntsman MM. Deficient tonic GABAergic conductance and synaptic balance in the fragile-X syndrome amygdala. J Neurophysiol. 2014;112:890–902. doi: 10.1152/jn.00597.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin LJ, Zurek AA, MacDonald JF, Roder JC, Jackson MF, Orser BA. Alpha5GABAA receptor activity sets the threshold for long-term potentiation and constrains hippocampus-dependent memory. J Neurosci. 2010;30:5269–5282. doi: 10.1523/JNEUROSCI.4209-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell SJ, Silver RA. Shunting inhibition modulates neuronal gain during synaptic excitation. Neuron. 2003;38:433–445. doi: 10.1016/s0896-6273(03)00200-9. [DOI] [PubMed] [Google Scholar]
- Mody I, Pearce RA. Diversity of inhibitory neurotransmission through GABAA receptors. Trends Neurosci. 2004;27:569–575. doi: 10.1016/j.tins.2004.07.002. [DOI] [PubMed] [Google Scholar]
- Olmos-Serrano JL, Paluszkiewicz SM, Martin BS, Kaufmann WE, Corbin JG, Huntsman MM. Defective GABAergic neurotransmission and pharmacological rescue of neuronal hyperexcitability in the amygdala in a mouse model of fragile X syndrome. J Neurosci. 2010;30:9929–9938. doi: 10.1523/JNEUROSCI.1714-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olmos-Serrano JL, Corbin JG, Burns MP. The GABAA receptor agonist THIP ameliorates specific behavioral deficits in the mouse model of fragile X syndrome. Dev Neurosci. 2011;33:395–403. doi: 10.1159/000332884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paluszkiewicz SM, Martin BS, Huntsman MM. Fragile X syndrome: the GABAergic system and circuit dysfunction. Dev Neurosci. 2011;33:349–364. doi: 10.1159/000329420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinna G. Targeting neurosteroidogenesis as therapy for PTSD. Front Pharmacol. 2014;4:166. doi: 10.3389/fphar.2013.00166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pirker S, Schwarzer C, Wieselthaler A, Sieghart W, Sperk G. GABAA receptors: immunocytochemical distribution of 13 subunits in the adult rat brain. Neuroscience. 2000;101:815–850. doi: 10.1016/s0306-4522(00)00442-5. [DOI] [PubMed] [Google Scholar]
- Pizzarelli R, Cherubini E. Alterations of GABAergic signaling in autism spectrum disorders. Neural Plasticity. 2011;2011:297153. doi: 10.1155/2011/297153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy DS, Rogawski MA. Neurosteroid replacement therapy for catamenial epilepsy. Neurotherapeutics. 2009;6:392–401. doi: 10.1016/j.nurt.2009.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossi DJ, Hamann M, Attwell D. Multiple modes of GABAergic inhibition of rat cerebellar granule cells. J Physiol. 2003;548:97–110. doi: 10.1113/jphysiol.2002.036459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudolph U, Mohler H. GABAA receptor subtypes: therapeutic potential in Down syndrome, affective disorders, schizophrenia, and autism. Annu Rev Pharmacol Toxicol. 2014;54:483–507. doi: 10.1146/annurev-pharmtox-011613-135947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sah P, Faber ESL, Lopez De Armentia M, Power J. The amygdaloid complex: anatomy and physiology. Physiol Rev. 2003;83:803–834. doi: 10.1152/physrev.00002.2003. [DOI] [PubMed] [Google Scholar]
- Schüle C, Nothdurfter C, Rupprecht R. The role of allopregnanolone in depression and anxiety. Prog Neurobiol. 2014;113:79–87. doi: 10.1016/j.pneurobio.2013.09.003. [DOI] [PubMed] [Google Scholar]
- Semyanov A, Walker MC, Kullmann DM, Silver RA. Tonically active GABA A receptors: modulating gain and maintaining the tone. Trends Neurosci. 2004;27:262–269. doi: 10.1016/j.tins.2004.03.005. [DOI] [PubMed] [Google Scholar]
- Stell BM, Brickley SG, Tang CY, Farrant M, Mody I. Neuroactive steroids reduce neuronal excitability by selectively enhancing tonic inhibition mediated by delta subunit-containing GABAA receptors. Proc Natl Acad Sci U S A. 2003;100:14439–14444. doi: 10.1073/pnas.2435457100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stórustovu SÍ, Ebert B. Pharmacological characterization of agonists at delta-containing GABAA receptors: functional selectivity for extrasynaptic receptors is dependent on the absence of gamma2. J Pharmacol Exp Ther. 2006;316:1351–1359. doi: 10.1124/jpet.105.092403. [DOI] [PubMed] [Google Scholar]
- Testa-Silva G, Loebel A, Giugliano M, de Kock CPJ, Mansvelder HD, Meredith RM. Hyperconnectivity and slow synapses during early development of medial prefrontal cortex in a mouse model for mental retardation and autism. Cereb Cortex. 2012;22:1333–1342. doi: 10.1093/cercor/bhr224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vislay RL, Martin BS, Olmos-Serrano JL, Kratovac S, Nelson DL, Corbin JG, Huntsman MM. Homeostatic responses fail to correct defective amygdala inhibitory circuit maturation in fragile X syndrome. J Neurosci. 2013;33:7548–7558. doi: 10.1523/JNEUROSCI.2764-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wafford KA, Ebert B. Gaboxadol—a new awakening in sleep. Curr Opin Pharmacol. 2006;6:30–36. doi: 10.1016/j.coph.2005.10.004. [DOI] [PubMed] [Google Scholar]
- Wafford KA, Ebert B. Emerging anti-insomnia drugs: tackling sleeplessness and the quality of wake time. Nat Rev Drug Discov. 2008;7:530–540. doi: 10.1038/nrd2464. [DOI] [PubMed] [Google Scholar]
- Wafford KA, van Niel MB, Ma QP, Horridge E, Herd MB, Peden DR, Belelli D, Lambert JJ. Neuropharmacology. Neuropharmacology. 2009;56:182–189. doi: 10.1016/j.neuropharm.2008.08.004. [DOI] [PubMed] [Google Scholar]
- Wohlfarth KM, Bianchi MT, Macdonald RL. Enhanced neurosteroid potentiation of ternary GABAA receptors containing the delta sub-unit. J Neurosci. 2002;22:1541–1549. doi: 10.1523/JNEUROSCI.22-05-01541.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zoghbi HY. Postnatal neurodevelopmental disorders: meeting at the synapse? Science. 2003;302:826–830. doi: 10.1126/science.1089071. [DOI] [PubMed] [Google Scholar]