Abstract
Osthole, a natural coumarin found in traditional Chinese medicinal plants, has shown multiple biological activities. In the present study, we investigated the preventive effects of osthole on inflammatory bowel disease (IBD). Colitis was induced in mice by infusing TNBS into the colonic lumen. Before TNBS treatment, the mice received osthole (100 mg·kg−1·d−1, ip) for 3 d. Pretreatment with osthole significantly ameliorated the clinical scores, colon length shortening, colonic histopathological changes and the expression of inflammatory mediators in TNBS-induced colitis. Pretreatment with osthole elevated serum cAMP levels; but treatment with the PKA inhibitor H89 (10 mg·kg−1·d−1, ip) did not abolish the beneficial effects of osthole on TNBS-induced colitis. In mouse peritoneal macrophages, pretreatment with osthole (50 μmol/L) significantly attenuated the LPS-induced elevation of cytokines at the mRNA level; inhibition of PKA completely reversed the inhibitory effects of osthole on IL-1β, IL-6, COX2, and MCP-1 but not on TNFα. In Raw264.7 cells, the p38 inhibitor SB203580 markedly suppressed LPS-induced upregulation of the cytokines, whereas the PKA inhibitors H89 or KT5720 did not abolish the inhibitory effects of SB203580. Moreover, in LPS-stimulated mouse peritoneal macrophages, SB203580 strongly inhibited the restored expression of IL-1β, IL-6, COX2, and MCP-1, which was achieved by abolishing the suppressive effects of osthole with the PKA inhibitors. Western blot analysis showed that osthole significantly suppressed the phosphorylation of p38, which was induced by TNBS in mice or by LPS in Raw264.7 cells. Inhibition of PKA partially reversed the suppressive effects of osthole on p38 phosphorylation in LPS-stimulated cells. Collectively, our results suggest that osthole is effective in the prevention of TNBS-induced colitis by reducing the expression of inflammatory mediators and attenuating p38 phosphorylation via both cAMP/PKA-dependent and independent pathways, among which the cAMP/PKA-independent pathway plays a major role.
Keywords: inflammatory bowel disease, colitis, mouse peritoneal macrophages, Raw264.7 cells, osthole, MAPK, p38, H89, KT5720, SB203580
Introduction
Inflammatory bowel disease (IBD) is a complex group of chronic, relapsing disorders that affect the gastrointestinal tract and is characterized by inflammation and tissue remodeling1. IBD can occur as two forms, ulcerative colitis and Crohn's disease, which can be distinguished by the portion of GI tract that is affected and the corresponding pathology. IBD is thought to be a consequence of the inappropriate and ongoing activation of the mucosal immune system that is driven by the presence of normal gut flora1. Meanwhile, the impaired barrier functions of the gut epithelium and decreases in blood flow caused by spasms of the colonic muscularis mucosae may also facilitate the development of mucosal inflammation2,3.
The predominant medical therapies for IBD are aminosalicylates and steroids, which are effective but are associated with significant side effects4. In recent years, the tumor necrosis factor α (TNF-α) antibody infliximab was introduced5. It provides significant therapeutic benefits but has disadvantages, including a restriction to non-oral routes of administration, immunogenicity and a high cost of treatment6. During the process of identifying novel approaches for the treatment of IBD, researchers have focused on cAMP-elevating phosphodiesterase (PDE) inhibitors. PDE4 and PDE3 are the major cAMP-hydrolyzing PDEs in most inflammatory cells7. PDE4 inhibitors and the dual selective inhibition of PDE3 and PDE4 have been shown to be effective in preventing and reversing GI inflammation and the disease symptoms of experimental colitis8,9,10, although the clinical use of some investigational PDE4 inhibitors, such as rolipram, was limited due to its adverse side effects11.
Osthole (also known as osthol), 7-methoxy-8-(3-methyl-2-butenyl)-2H-1-benzopyran-2-one, is a natural coumarin first derived from the Cnidium plant. A high content of osthole is found in the mature fruit of Cnidium monnieri (Fructus Cnidii), which is commonly used in the clinical practice of traditional Chinese medicine12,13. Osthole is also widely found in other medicinal plants including Angelica, Archangelica, Citrus, and Clausena. Osthole has been shown to have multiple functions14, among which, immunomodulation and anti-inflammation have been demonstrated by studies on cells, such as macrophages, bronchial epithelial cells, rat peritoneal cells and human peripheral blood mononuclear cells, which were stimulated by lipopolysaccharide (LPS), interleukin-4 (IL-4) or tumor necrosis factor α (TNFα)15,16, and in animal models such as carrageenan-induced hind paw edema and allergy17,18 models. The underlying mechanisms include an elevation of intracellular and tissue levels of both cAMP and cGMP via inhibition of PDE and regulation of the expression of a series of key factors including TNF-α, NF-КB, TGF-β, COX, NO, ERK, and JNK19. So far, the effects of osthole on IBD have not been assessed. In the present study, we induced colitis in mice, investigated the efficacy of osthole in the prevention of colitis and studied the underlying mechanisms.
Materials and methods
Animals and induction of colitis
Eight-week-old male C57BL/6 mice were used in this study. The mice were allowed free access to drinking water and food and were housed under room temperature (25 °C) with an automatic 12 h light and 12 h dark cycle. Osthole (purity >98%) was purchased from MedChem Express (Shanghai, China). Osthole was dissolved in a 1:9 (v/v) mixture of Tween 80 and 0.9% sodium chloride solution and administered intraperitoneally at the indicated doses once daily until the end of the experiments. After pretreatment with osthole (100 mg·kg−1·d−1) for 3 d, the mice were fasted for 14–16 h with free access to water before the induction of colitis. Anesthesia was achieved by the intraperitoneal administration of freshly prepared sodium pentobarbital (60 mg/kg). A 100 μL enema of TNBS (2.8 mg, Sigma, St Louis, USA) in 50% ethanol was infused into the colonic lumen according to a published protocol. In some experiments, H89 (10 mg/kg, Sigma, USA), a specific protein kinase A (PKA) inhibitor, was intraperitoneally administered 1 h prior to the TNBS treatment as indicated. For most studies, the animals were sacrificed and evaluated 2 d after the induction of colitis. For histopathological scoring, the animals were sacrificed 4 d after the induction of colitis. The mice were anesthetized with 60 mg sodium pentobarbital/kg body weight intraperitoneally. An incision was made to access the peritoneal cavity, and the colon was removed, weighed, measured, and used for subsequent analyses as indicated. All animal protocols were pre-approved by the Animal Care and Use Committee of Nanjing Medical University.
Clinical activity score
Colitis was quantified with a clinical score, as indicated in Table 1, which consisted of assessing weight loss, stool consistency, and bleeding (measured using commercially available kits from Jiancheng, Nanjing, China), as previously described8,9. These scores were added to calculate the total clinical score that ranged from 0 (healthy) to 12 (maximal activity of colitis).
Table 1. Clinical activity scores. Weight loss, stool consistency, and fecal blood were evaluated daily. The clinical score was the average of these parameters.
| Weight loss (%) | Stool | Bloody stool | Index |
|---|---|---|---|
| 0–1 | Well formed pellets | None | 0 |
| 1–5 | Soft and shaped | Between | 1 |
| 5–10 | Pasty and semiformed | Slight | 2 |
| 10–15 | Between | Between | 3 |
| >15 | Diarrhea | Gross bleeding | 4 |
Colon length and histological scoring
During postmortem analyses, the entire colon, from the cecum to the anus, was removed and loosely placed on cellulose. The colon length was measured and observed as an indirect marker of inflammation. The entire mouse colon was then Swiss-rolled, formalin-fixed, and paraffin-embedded. The colon sections were stained with hematoxylin and eosin. The colitis parameters were analyzed and scored according to published histopathological grading standards.
Measurement of cAMP concentrations
The concentration of cAMP in serum samples was determined using commercial enzyme immunoassay kits (Elabscience, China). The frozen serum samples were thawed and centrifuged at 1000 revolutions per minute at 4 °C for 20 min, and the supernatants were analyzed for cAMP levels according to the manufacturer's instructions.
Cell culture and treatment
Raw264.7 cells were obtained from the American Type Culture Collections (VA, USA). The cells were maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% FBS and penicillin/streptomycin. Raw264.7 cells were incubated with the p38 inhibitor SB203580 (MedChem, China) at 10 μmol/L alone or combined with PKA inhibitors H89 at 10 μmol/L or KT5720 (Calbiochem, USA) at 1 μmol/L for 2 h before being exposed to 10 ng/mL LPS (Sigma, USA). After 4 h, the cellular mRNA samples were extracted and quantified using real-time RT-PCR. In the indicated experiments, the Raw264.7 cells were incubated with 50 μmol/L osthole for 24 h. The cells were then treated with 10 μmol/L H89 or 1 μmol/L KT5720 for 2 h before their exposure to 1 μg/mL LPS. Western blot analysis was performed 30 min after the stimulation with LPS.
To isolate peritoneal macrophages, mice were anesthetized with 2%–3% sodium pentobarbital (Sigma, USA), and the abdomen was lavaged with 3 mL of heparinized PBS containing no calcium or magnesium. After abdominal kneading, the peritoneal cells (PCs) were harvested using a syringe. Mouse peritoneal macrophages were obtained by culturing the PCs in RPMI-1640 containing 20% fetal bovine serum for 2 h at 37 °C. After incubation, the adherent cells were collected. The mouse peritoneal macrophages were incubated with 50 μmol/L osthole for 24 h. The cells were then treated with either 10 μmol/L H89 or 1 μmol/L KT5720 alone or combined with 10 μmol/L SB203580 for 2 h before being exposed to 10 ng/mL LPS. After another 4 h, the cellular mRNA samples were extracted and quantified using real-time RT-PCR.
Quantitative real-time PCR
Total cellular RNA was extracted using the Trizol reagent (Takara, Japan) according to the manufacturer's instructions. The total RNA (2 μg) samples were reverse transcribed using the PrimeScript RT reagent kit (Takara, Japan). Real-time PCR was performed with the Power SYBR Green PCR Master Mix (Applied Biosystems), using an Applied Biosystems 7500 Real-Time PCR System. The primer sequences were listed in Table 2. The relative quantities of mRNA were determined using the comparative cycle threshold methods and normalized against GAPDH mRNA.
Table 2. Primers used to detect gene expression at mRNA level.
| Gene name | Primer sequences (5′-3′) |
|---|---|
| IL-6 | F: CTGCAAGAGACTTCCATCCAG |
| R: AGTGGTATAGACAGGTCTGTTGG | |
| IL-1β | F: TTCAGGCAGGCAGTATCACTC |
| R: GAAGGTCCACGGGAAAGACAC | |
| MCP-1 | F: ACTGAAGCCAGCTCTCTCTTCCTC |
| R: TTCCTTCTTGGGGTCAGCACAGAC | |
| CXCL10 | F: CCAAGTGCTGCCGTCATTTTC |
| R: GGCTCGCAGGGATGATTTCAA | |
| COX2 | F: TTCCAATCCATGTCAAAACCGT |
| R: AGTCCGGGTACAGTCACACTT | |
| TNFα | F: CCAGGCGGTGCCTATGTCTC |
| R: CAGCCACTCCAGCTGCTCCT | |
| Claudin1 | F: TGCCCCAGTGGAAGATTTACT |
| R: CTTTGCGAAACGCAGGACAT | |
| ZO1 | F: GCCGCTAAGAGCACAGCAA |
| R: TCCCCACTCTGAAAATGAGGA | |
| GAPDH | F: AGGTCGGTGTGAACGGATTTG |
| R: GGGGTCGTTGATGGCAACA |
Western blot analysis
Gut epithelia were scraped and lysed with RIPA buffer containing protease inhibitors (Roche Diagnostics, Swissland) for 15 min on ice. After centrifugation for 15 min at 20 000×g (4 °C), the total protein content of the samples was determined according to the Bradford method. Proteins (50 μg per lane) were loaded onto SDS-polyacrylamide gels and blotted onto methanol-activated PVDF membranes. Anti-mouse claudin-1 antibodies were obtained from Invitrogen. Phospho-Erk (Thr202/Tyr204), Erk, phospho-p38 (Thr180/Tyr182), p38, phospho-JNK (Thr183/Tyr185), and JNK were obtained from Cell Signaling. GAPDH from Sigma was used as internal control. The bands were detected through enhanced chemiluminescence using the Amersham ECL Western blot detection kit according to the manufacturer's instructions (Amersham Biosciences, USA).
Statistical analysis
Data are expressed as the mean±SEM. SPSS version 17.0 software was used to conduct the statistical analyses. Statistical significance was assessed using a two-tailed Student's t-test. In all statistical comparisons, a P value <0.05 was used to indicate a statistically significant difference.
Results
Osthole attenuated TNBS-induced colitis in mice
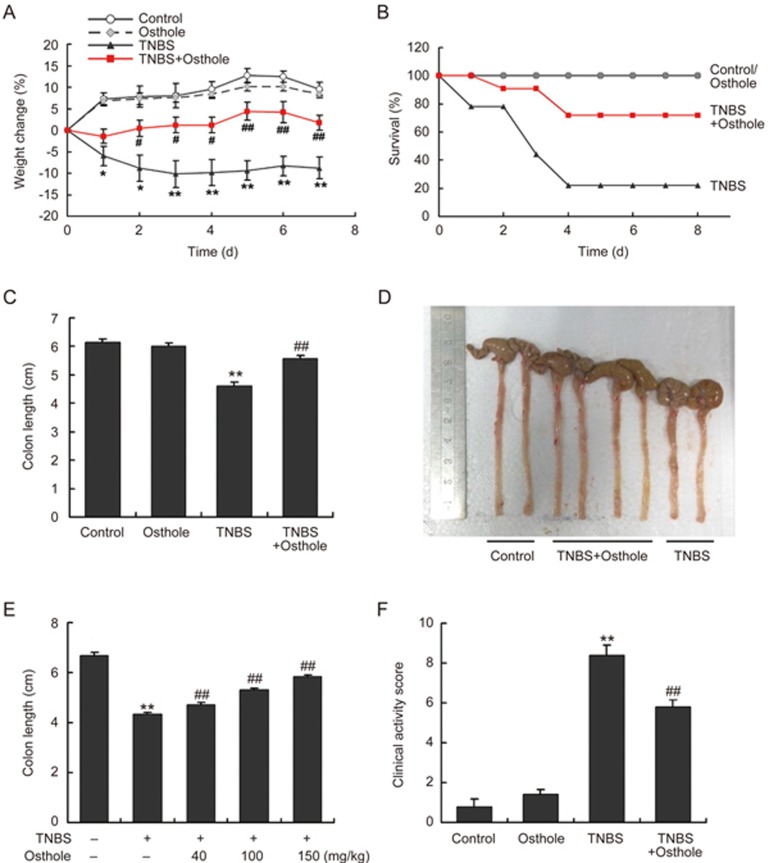
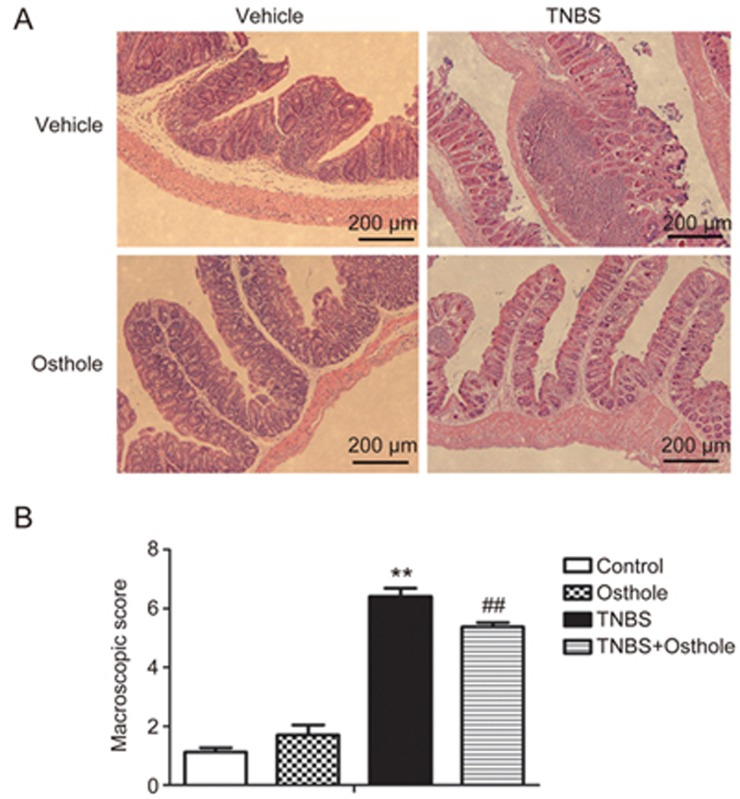
Persistent weight loss was observed in mice starting on d 1 following the TNBS treatments. Pretreatment with osthole (100 mg·kg−1·d−1) significantly attenuated the weight loss (Figure 1A). TNBS treatments led to increased mortality in mice, which was significantly alleviated by osthole (Figure 1B). The colon length is an indirect and reproducible morphological indicator for the severity of colonic inflammation. Significant shortening of the colon was observed in mice on d 2 of post-TNBS treatments. Osthole partially and dose dependently reversed the TNBS-induced reduction in colon length at dosages of 40, 100, and 150 mg·kg−1·d−1 (Figure 1C-1E). Mice exposed to TNBS developed signs of colitis as expressed by clinical scores greater than 8 starting on d 2 (Figure 1F). Treatments with osthole halted the progression of colitis as expressed by lower clinical scores. The histological features of TNBS-treated mice include multiple erosive lesions, transmural inflammation, infiltration of lymphocytes, and the loss of goblet cells (Figure 2A). Osthole significantly decreased the histological scores compared to the TNBS group (Figure 2B). No signs of colitis were observed in mice treated with osthole alone. In the above studies, treatment with osthole began 3 d prior to TNBS and lasted until the end of the experiments. When osthole treatments were started simultaneously with TNBS treatments, no detectable protection was observed (data not shown). Taken together, these results showed the significant beneficial effects of osthole on the prevention of TNBS-induced colitis in mice.
Figure 1.
Osthole attenuated the TNBS-induced colitis in mice. Mice received osthole treatments intraperitoneally at 100 mg/kg once daily starting at three days before the TNBS treatments and until the end of the experiments. Colitis was induced by infusing the mouse colonic lumen with 100 μL enema of TNBS (2.8 mg) in 50% ethanol. (A) The change in body weights of the mice. (B) Survival of the mice. (C and D) The colon lengths and representative macroscopic changes were determined 2 d after the initial rectal TNBS administration. (E) Treatment with osthole dose dependently reduced colon length shortening. (F) The degree of colitis was quantified by the clinical score 2 d after the initial rectal TNBS administration, as described in materials and methods. Data are expressed as the mean±SEM (numerical data are shown in the Supplementary file), n=5-9 mice per group. *P<0.05, **P<0.01 compared to control. #P<0.05, ##P<0.01 compared to TNBS.
Figure 2.
Effects of osthole on the histological manifestations of TNBS-induced colitis. (A) H&E staining of colonic morphology at 4 d after the initial rectal TNBS administration. The magnification factor of the images is 50-fold. (B) Scores are reported as the mean±SEM. **P<0.01 compared to control. ##P<0.01 compared to TNBS.
Osthole reduced the expression of inflammatory cytokines
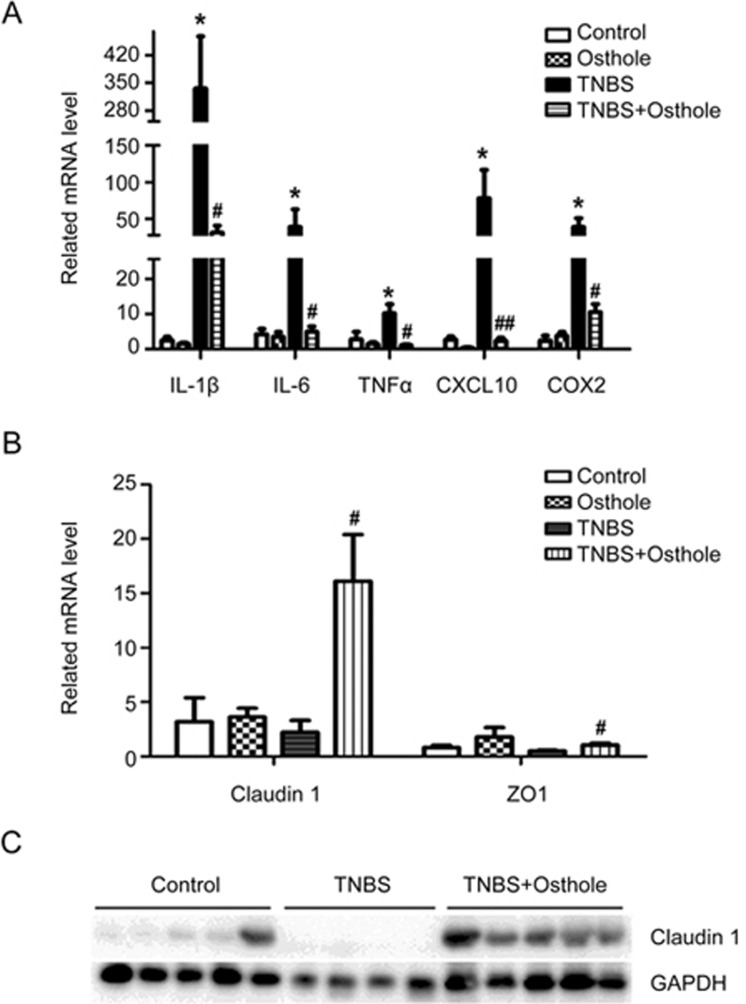
To investigate the mechanisms underlying the preventive effects of osthole, we first examined the expression levels of several inflammatory factors that play crucial roles during the pathogenesis of IBD. TNBS significantly increased the mRNA levels of IL-1β, IL-6, TNFα, CXCL10, and COX2 in colonic tissues on d 2 post-TNBS treatment, whereas pretreatment with osthole reduced them significantly, indicating the possible role of osthole in the attenuation of inflammation (Figure 3A). Because impaired barrier function of the gut epithelium is likely to facilitate the development of mucosal inflammation, we examined the expression of the tight junction proteins claudin-1 and ZO-1 in colonic mucosa. In mice treated with TNBS or osthole alone, no detectable changes in either of these genes at the mRNA level were observed. However, when mice were treated with osthole prior to TNBS, a significant upregulation of claudin-1 and ZO-1 mRNA in colonic mucosa was detected compared to the TNBS group (Figure 3B). Western blot analysis of colonic mucosal lysates showed a significant reduction in claudin-1 in TNBS-treated mice when compared to the control group. Pretreatment with osthole strongly enhanced the expression of claudin-1 to higher than baseline levels (Figure 3C). These results suggested that the efficacy of osthole in the prevention of colitis was related to its anti-inflammation properties.
Figure 3.
Osthole reduced the expression of inflammatory cytokines. Mice received osthole intraperitoneally at 100 mg/kg once daily starting at three days before being exposed to TNBS and until the end of the experiments. Colitis was induced by infusing a 100 μL enema of TNBS (2.8 mg) in 50% ethanol into the colonic lumen. (A and B) Mice were sacrificed on d 2 post-TNBS treatments and the mRNA levels of inflammatory cytokines in the colonic mucosa samples were quantified by real-time RT-PCR. (C) Western blot analysis of colonic mucosa homogenates was performed 2 d after the initial rectal TNBS administration. Data are expressed as the mean±SEM (numerical data are shown in the Supplementary file), n=4–6 mice per group. *P<0.05 compared to control. #P<0.05, ##P<0.01 compared to TNBS.
The protective effects of osthole against TNBS-induced colitis was mainly mediated by the cAMP/PKA-independent pathway
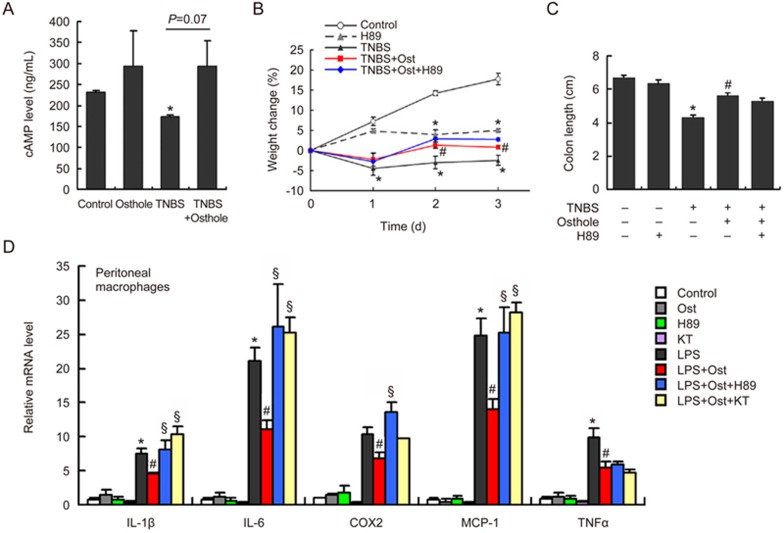
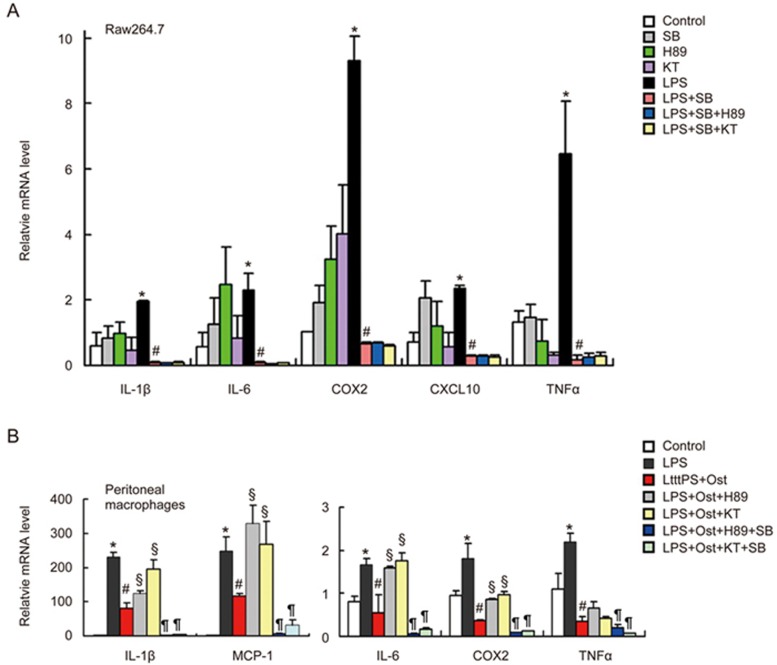
Osthole was found to elevate intracellular and tissue cAMP levels by inhibiting PDE4, the main enzyme that metabolizes cAMP in inflammatory cells. In general, intracellular elevations in cAMP are associated with broad anti-inflammatory effects20. The anti-inflammatory and immunomodulatory effects of PDE4 inhibitors in human leukocytes, endothelium and epithelium have been well documented21. To test the role of cAMP in the protective effects of osthole against TNBS-induced colitis, we determined the serum cAMP levels. A significant reduction in serum cAMP levels was observed on d 2 after TNBS treatment. Osthole led to elevated cAMP levels (P=0.07) compared to the TNBS group (Figure 4A), which is consistent with previous reports. It is known that cAMP mainly works by activating PKA20. To test whether the protective effects of osthole are mediated by the cAMP/PKA pathway, H89, a potent selective inhibitor of PKA, was administered intraperitoneally 1 h prior to TNBS treatment. Unexpectedly, we found that H89 pretreatment failed to abolish osthole-induced alleviation of weight loss and colon length shortening (Figure 4B, 4C). We cultured mouse peritoneal macrophages and stimulated them with LPS. We observed that osthole decreased the elevation of IL-1β, IL-6, COX2, MCP-1, and TNFα at the mRNA level in response to LPS (Figure 4D). Inhibition of PKA with H89 or KT5720 completely reversed the effects of osthole on IL-1β, IL-6, COX2, and MCP-1 but not on TNFα (Figure 4D). Therefore, the suppression of inflammatory cytokines by osthole may be mediated by two distinct mechanisms through the cAMP/PKA pathways. Because the inhibition of PKA had no effect on the protective effects of osthole against colitis, we can infer that the cAMP/PKA-independent mechanism plays a major role in this process.
Figure 4.
The protective effects of osthole against TNBS-induced colitis were mainly mediated by the cAMP/PKA-independent pathway. (A) Serum cAMP levels were determined 2 d after the initial rectal TNBS administration. (B) H89 was intraperitoneally administered at 10 mg/kg 1 h prior to TNBS. The changes in the body weights of mice were determined. (C) H89 was intraperitoneally administered at 10 mg/kg 1 h prior to TNBS treatments. The animals were sacrificed on d 2 post-TNBS treatments, and the colon lengths were determined. (D) Mouse peritoneal macrophages were isolated and incubated with 50 μmol/L osthole for 24 h. Cells were then treated with 10 μmol/L H89 or 1 μmol/L KT5720 for 2 h before being exposed to 10 ng/mL LPS. After another 4 h, the cellular mRNA samples were extracted and quantified by real-time RT-PCR. Data are expressed as the mean±SEM (numerical data are shown in the Supplementary file), n=5–9 mice or n=3 wells per group. *P<0.05 compared to control. #P<0.05 compared to TNBS or LPS. §P<0.05 compared to TNBS plus osthole or LPS plus osthole.
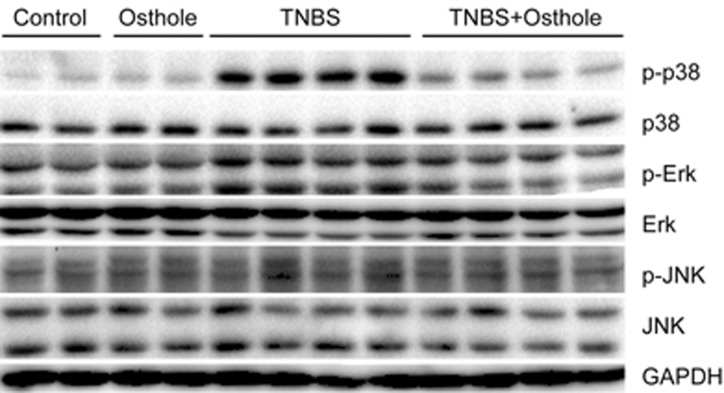
Osthole attenuated p38 phosphorylation in TNBS-induced colitis and LPS-activated macrophages
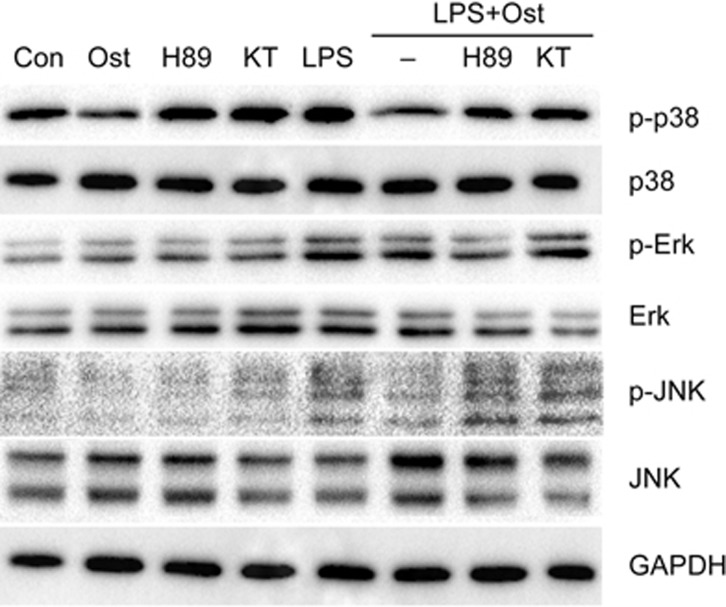
Because the MAPK pathway affects the production of inflammatory cytokines22, we examined the phosphorylation levels of MAPK in colonic tissues. Compared to the control group, significantly higher phosphorylation of p38 was observed in mice exposed to TNBS on d 2. Osthole significantly inhibited the TNBS-induced p38 activation. No detectable changes were observed in the phosphorylation levels of Erk and JNK (Figure 5). To explore the effects of p38 inhibition on inflammation, Raw264.7 cells were treated with the p38 inhibitor SB203580 before exposure to LPS. We found that LPS-induced elevation of IL-1β, IL-6, COX2, CXCL10, and TNFα levels was strongly suppressed by SB203580 (Figure 6A), indicating that p38 phosphorylation was required for the upregulation of inflammatory cytokines caused by LPS. Inhibition of PKA with H89 or KT5720 failed to abolish the effects of SB203580 on these cytokines, suggesting that the suppression of inflammation induced by p38 inhibition is not via the cAMP/PKA pathway. The role of p38 inhibition in the anti-inflammatory actions of osthole was tested in mouse peritoneal macrophages activated by LPS. The results showed that the elevated expression levels of IL-1β, IL-6, COX2, and MCP-1 induced by PKA inhibitors in osthole-treated cells was completely abolished by SB203580 (Figure 6B), suggesting that p38 inhibition acts downstream of cAMP/PKA in the osthole-regulated expression of cytokines. In fact, SB203580 suppressed the expression of these cytokines to levels much lower than that of osthole alone in LPS-activated cells. The expression of TNFα was also strongly inhibited by SB203580, suggesting that the inhibition of p38 might be a common intersection in the multiple pathways mediating the anti-inflammation actions of osthole. The phosphorylation levels of MAPK were also examined in Raw264.7 cell by western blot analysis. Consistent with the findings in vivo, LPS stimulated an increased phosphorylation of p38 (Figure 7). Pretreatment with osthole significantly reduced the phosphorylation levels of p38. Inhibition of PKA with H89 or KT5720 partially reversed the suppressive effects of osthole on p38 phosphorylation, further supporting the view that osthole inhibited p38 activation partly through the cAMP/PKA pathway. No detectable changes were observed in the phosphorylation levels of Erk due to pretreatment with osthole. For JNK, only a very low level of phosphorylation was detected in response to LPS, suggesting that JNK might play a minor role in this process.
Figure 5.
Osthole attenuated the phosphorylation of p38 in TNBS-induced colitis. Mice received osthole intraperitoneally at 100 mg/kg once daily starting at three days before being exposed to TNBS treatments and until the end of the experiments. Colitis was induced by infusing a 100 μL enema of TNBS (2.8 mg) in 50% ethanol into the colonic lumen. Western blot analysis of the colonic mucosa homogenates was performed 2 d after the initial rectal TNBS administration.
Figure 6.
Effect of p38 or PKA inhibition in osthole-induced suppression of inflammation evoked by LPS. (A) Raw264.7 cells were incubated with 10 μmol/L SB203580 alone or combined with either 10 μmol/L H89 or 1 μmol/L KT5720 for 2 h before being exposed to 10 ng/mL LPS. After 4 h, the mRNA samples from the cells were extracted and quantified by real-time RT-PCR. (B) Mouse peritoneal macrophages were isolated and incubated with 50 μmol/L osthole for 24 h. Cells were then treated with either 10 μmol/L H89 or 1 μmol/L KT5720 alone or combined with 10 μmol/L SB203580 for 2 h before being exposed to 10 ng/mL LPS. After another 4 h, the mRNA in cells was extracted and quantified by real-time RT-PCR. Data are expressed as the mean±SEM (numerical data are shown in the Supplementary file), n=3 wells per group. *P<0.05 compared to control. #P<0.05 compared to LPS. §P<0.05 compared to LPS plus osthole. ¶P<0.05 compared to LPS plus osthole and H89 or KT5720.
Figure 7.
Effects of PKA inhibition on the osthole-induced suppression of p38 phosphorylation in LPS-stimulated cells. Raw264.7 cells were incubated with 50 μmol/L osthole for 24 h. Cells were then treated with 10 μmol/L H89 or 1 μmol/L KT5720 for 2 h before exposure to 1 μg/mL LPS. Western blot analysis was performed 30 min after stimulation with LPS.
Discussion
Osthole, a natural product found widely in medicinal plants, especially in Fructus Cnidii, has been demonstrated to display multiple pharmacological activities, including neuroprotective, osteogenic, immunomodulatory, anticancer, hepatoprotective, cardiovascular protective, and antimicrobial activities19. In the present study, we investigated the effects of osthole in the prevention of TNBS-induced colitis. Treatment with osthole ameliorated the clinical score, dose dependently reduced colon length shortening, improved the colonic histopathological changes, and decreased the expression of inflammatory mediators in colonic tissue. These findings clearly indicated the beneficial effects of osthole in the prevention of TNBS-induced colitis and enriched the current knowledge of the pharmacological activities of this coumarin.
Because TNBS-induced colitis is characterized by transmural inflammation1, we investigated the mechanisms underlying the anti-inflammatory actions of osthole in this model. Previous reports identified osthole as a natural PDE4 inhibitor23. Consistently, elevated serum cAMP levels were observed in mice pretreated with osthole before being exposed to TNBS in our study. Intracellular elevation of cAMP levels demonstrated its anti-inflammatory activity in numerous in vitro and in vivo studies20. The efficacy of PDE4 inhibitors, such as mesopram, rolipram, and roflumilast, in the treatment of experimental colitis has been reported8,9,10. However, in the present study, the inhibition of PKA, a major target of cAMP, failed to abolish the protective effects of osthole on TNBS-induced colitis. This discrepancy may be explained by the finding that osthole regulated TNFα, a crucial factor in the pathogenesis of IBD24,25, in a way that is independent of the cAMP/PKA pathway. It is interesting to find that osthole regulates TNFα differently than cytokines such as IL-6 and IL-1β. We speculate that it might be due to differences in promoter structures and regulatory proteins. It should be noted that in contrast to PDE4 inhibitors that merely raise cellular cAMP levels, osthole has many other bioactivities, such as raising cGMP levels via the inhibition of other PDEs, regulating iron channels, such as calcium channels, and inhibiting 5-lipoxygenase, as well as antioxidative and anti-apoptosis properties19. Consequently, the regulation of genes by osthole may be due to the complicated and integrated effects of its multiple actions.
Osthole has been shown to affect MAPK activation by several studies. In prostate cancer cells and HepG2 cells, osthole was shown to attenuate their metastatic ability and inflammatory responses through inhibiting the phosphorylation of Erk, JNK, and p3826,27. However, osthole was also shown to alleviate hepatic injury after trauma-hemorrhage by enhancing p38 activation28. Therefore, the effects of osthole on the activation of MAPK are related to the cellular context and the type of stimulus. Consistent with a previous study on J774A.1 macrophages that showed that the phosphorylation of p38 and JNK was inhibited by osthole29, we observed that osthole attenuated the phosphorylation of p38 in mouse peritoneal macrophages and Raw264.7 cells. Moreover, we showed that the inhibitory effects of osthole on p38 phosphorylation was mediated via both cAMP/PKA-dependent and -independent pathways. Evidence supporting the involvement of the cAMP/PKA/MAPK pathway in the effects of osthole was also reported by a previous study on neutrophils, which showed that the anti-inflammatory effects of osthole were mediated by cAMP/PKA-dependent inhibition of Akt and ERK activation23. Further studies are needed to reveal other molecules that mediate the effects of osthole on MAPK.
In this study, the mechanisms underlying the protective effects of osthole against TNBS-induced colitis were focused on the reduction of inflammatory cytokines. Because osthole also demonstrated immunomodulatory activities19, changes in the mucosal immune system may be involved in its effects on colitis. In addition, the role of vasculature and disturbances in blood flow are thought to be important pathogenic mechanisms in IBD2. It was reported that osthole showed vasorelaxant properties due to its Ca2+-channel antagonistic effects and the upregulation of cAMP and cGMP levels in vascular smooth muscle cells30,31,32. Therefore, it is probable that the anti-inflammatory action of osthole is not the only mechanism that explains its ability to inhibit colitis.
In conclusion, we demonstrated that osthole is effective in the prevention of TNBS-induced colitis. Osthole reduced the expression of inflammatory mediators, probably through the inhibition of p38 phosphorylation via both cAMP/PKA-dependent and independent pathways, among which the cAMP/PKA-independent pathway plays a major role. Osthole can be considered a potential natural treatment to prevent IBD.
Author contribution
Wu SUN and Hao LI designed the study; Wu SUN, Yun CAI, and Xin-xin ZHANG performed the in vivo experiments; Wu SUN and Yun CAI were responsible for the cell experiments; Xin-xin ZHANG and Hao CHEN assisted in caring for the animals; Wu SUN, Yan-die LIN, and Hao CHEN performed the data analyses; Hao LI and Wu SUN wrote the manuscript.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No 81270944 and 81070678), and the Qinglan Project (2014) awarded to Hao LI. Dr Hao LI is an Associate Fellow at the Collaborative Innovation Center for Cardiovascular Disease Translational Medicine.
Footnotes
Supplementary information is available on the website of Acta Pharmacologica Sinica.
Supplementary Information
References
- Keighley MR, Stockbrugger RW. Inflammatory bowel disease. Aliment Pharmacol Ther 2003; 18: 66–70. [DOI] [PubMed] [Google Scholar]
- Tsujii M, Kawano S, Tsuji S, Kobayashi I, Takei Y, Nagano K, et al. Colonic mucosal hemodynamics and tissue oxygenation in patients with ulcerative colitis: investigation by organ reflectance spectrophotometry. J Gastroenterol 1995; 30: 183–8. [DOI] [PubMed] [Google Scholar]
- Ma TY, Iwamoto GK, Hoa NT, Akotia V, Pedram A, Boivin MA, et al. TNF-alpha-induced increase in intestinal epithelial tight junction permeability requires NF-kappa B activation. Am J Physiol Gastrointest Liver Physiol 2004; 286: G367–76. [DOI] [PubMed] [Google Scholar]
- Farrell RJ, Kelleher D. Glucocorticoid resistance in inflammatory bowel disease. J Endocrinol 2003; 178: 339–46. [DOI] [PubMed] [Google Scholar]
- Rutgeerts P, Sandborn WJ, Feagan BG, Reinisch W, Olson A, Johanns J, et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N Engl J Med 2005; 353: 2462–76. [DOI] [PubMed] [Google Scholar]
- van Deventer SJ. Small therapeutic molecules for the treatment of inflammatory bowel disease. Gut 2002; 50: III47–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torphy TJ. Phosphodiesterase isozymes: molecular targets for novel antiasthma agents. Am J Respir Crit Care Med 1998; 157: 351–70. [DOI] [PubMed] [Google Scholar]
- Hartmann G, Bidlingmaier C, Siegmund B, Albrich S, Schulze J, Tschoep K, et al. Specific type IV phosphodiesterase inhibitor rolipram mitigates experimental colitis in mice. J Pharmacol Exp Ther 2000; 292: 22–30. [PubMed] [Google Scholar]
- Loher F, Schmall K, Freytag P, Landauer N, Hallwachs R, Bauer C, et al. The specific type-4 phosphodiesterase inhibitor mesopram alleviates experimental colitis in mice. J Pharmacol Exp Ther 2003; 305: 549–56. [DOI] [PubMed] [Google Scholar]
- Rieder F, Siegmund B, Bundschuh DS, Lehr HA, Endres S, Eigler A. The selective phosphodiesterase 4 inhibitor roflumilast and phosphodiesterase 3/4 inhibitor pumafentrine reduce clinical score and TNF expression in experimental colitis in mice. PLoS One 2013; 8: e56867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sturgess I, Searle GF. The acute toxic effect of the phosphodiesterase inhibitor rolipram on plasma osmolality. Br J Clin Pharmacol 1990; 29: 369–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- You L, Feng S, An R, Wang X. Osthole: a promising lead compound for drug discovery from a traditional Chinese medicine (TCM). Nat Prod Commun 2009; 4: 297–302. [PubMed] [Google Scholar]
- Wang R, Kong J, Wang D, Lien LL, Lien EJ. A survey of Chinese herbal ingredients with liver protection activities. Chin Med 2007; 2: 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoult JR, Paya M. Pharmacological and biochemical actions of simple coumarins: natural products with therapeutic potential. Gen Pharmacol 1996; 27: 713–22. [DOI] [PubMed] [Google Scholar]
- Chiu PR, Lee WT, Chu YT, Lee MS, Jong YJ, Hung CH. Effect of the Chinese herb extract osthol on IL-4-induced eotaxin expression in BEAS-2B cells. Pediatr Neonatol 2008; 49: 135–40. [DOI] [PubMed] [Google Scholar]
- Zimecki M, Artym J, Cisowski W, Mazol I, Wlodarczyk M, Glensk M. Immunomodulatory and anti-inflammatory activity of selected osthole derivatives. Z Naturforsch C 2009; 64: 361–8. [DOI] [PubMed] [Google Scholar]
- Liu J, Zhang W, Zhou L, Wang X, Lian Q. Anti-inflammatory effect and mechanism of osthole in rats. Zhong Yao Cai 2005; 28: 1002–6. [PubMed] [Google Scholar]
- Matsuda H, Tomohiro N, Ido Y, Kubo M. Anti-allergic effects of cnidii monnieri fructus (dried fruits of Cnidium monnieri) and its major component, osthol. Biol Pharm Bull 2002; 25: 809–12. [DOI] [PubMed] [Google Scholar]
- Zhang ZR, Leung WN, Cheung HY, Chan CW. Osthole: A review on its bioactivities, pharmacological properties, and potential as alternative medicine. Evid Based Complement Alternat Med 2015; 2015: 919616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serezani CH, Ballinger MN, Aronoff DM, Peters-Golden M. Cyclic AMP: master regulator of innate immune cell function. Am J Respir Cell Mol Biol 2008; 39: 127–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banner KH, Trevethick MA. PDE4 inhibition: a novel approach for the treatment of inflammatory bowel disease. Trends Pharmacol Sci 2004; 25: 430–6. [DOI] [PubMed] [Google Scholar]
- Dang PM, Morel F, Gougerot-Pocidalo MA, El Benna J. Phosphorylation of the NADPH oxidase component p67(PHOX) by ERK2 and P38MAPK: selectivity of phosphorylated sites and existence of an intramolecular regulatory domain in the tetratricopeptide-rich region. Biochemistry 2003; 42: 4520–6. [DOI] [PubMed] [Google Scholar]
- Tsai YF, Yu HP, Chung PJ, Leu YL, Kuo LM, Chen CY, et al. Osthol attenuates neutrophilic oxidative stress and hemorrhagic shock-induced lung injury via inhibition of phosphodiesterase 4. Free Radic Biol Med 2015; 89: 387–400. [DOI] [PubMed] [Google Scholar]
- Horie Y, Chiba M, Suzuki T, Kudo T, Kamata A, Iizuka M, et al. Induction of major histocompatibility complex class II antigens on human colonic epithelium by interferon-gamma, tumor necrosis factor-alpha, and interleukin-2. J Gastroenterol 1998; 33: 39–47. [PubMed] [Google Scholar]
- Pender SL, Fell JM, Chamow SM, Ashkenazi A, MacDonald TT. A p55 TNF receptor immunoadhesin prevents T cell-mediated intestinal injury by inhibiting matrix metalloproteinase production. J Immunol 1998; 160: 4098–103. [PubMed] [Google Scholar]
- Wen YC, Lee WJ, Tan P, Yang SF, Hsiao M, Lee LM, et al. By inhibiting snail signaling and miR-23a-3p, osthole suppresses the EMT-mediated metastatic ability in prostate cancer. Oncotarget 2015; 6: 21120–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu SJ. Osthole attenuates inflammatory responses and regulates the expression of inflammatory mediators in HepG2 cells grown in differentiated medium from 3T3-L1 preadipocytes. J Med Food 2015; 18: 972–9. [DOI] [PubMed] [Google Scholar]
- Yu HP, Liu FC, Tsai YF, Hwang TL. Osthole attenuates hepatic injury in a rodent model of trauma-hemorrhage. PLoS One 2013; 8: e65916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao PC, Chien SC, Ho CL, Wang EI, Lee SC, Kuo YH, et al. Osthole regulates inflammatory mediator expression through modulating NF-kappaB, mitogen-activated protein kinases, protein kinase C, and reactive oxygen species. J Agric Food Chem 2010; 58: 10445–51. [DOI] [PubMed] [Google Scholar]
- Fusi F, Sgaragli G, Ha le M, Cuong NM, Saponara S. Mechanism of osthole inhibition of vascular Cav1.2 current. Eur J Pharmacol 2012; 680: 22–7. [DOI] [PubMed] [Google Scholar]
- Ko FN, Wu TS, Liou MJ, Huang TF, Teng CM. Vasorelaxation of rat thoracic aorta caused by osthole isolated from Angelica pubescens. Eur J Pharmacol 1992; 219: 29–34. [DOI] [PubMed] [Google Scholar]
- Guh JH, Yu SM, Ko FN, Wu TS, Teng CM. Antiproliferative effect in rat vascular smooth muscle cells by osthole, isolated from Angelica pubescens. Eur J Pharmacol 1996; 298: 191–7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.