Abstract
Cassiae semen (Leguminosae), a well-known traditional Chinese medicine, has been used for a number of centuries in areas of Southeast Asia, including Korea, Japan and China. The present review aims to provide updated and comprehensive information, on the botany, phytochemistry and pharmacology of Cassiae semen. The available information on Cassiae semen was collected using several different resources, including classic books on Chinese herbal medicine and a number of scientific databases, including the China Academic Journals full-text database, PubMed, SciFinder, the Web of Science and Science Direct. To date >70 chemical compounds have been isolated from Cassiae semen, and the major components have been determined to be anthraquinones, naphthopyrones and volatile oil. The crude extracts and pure compounds of Cassiae semen have been used as effective agents in preclinical and clinical practice due to their beneficial activities, including antihyperlipidemic, antidiabetic, neuroprotective, hepatoprotective, antibacterial, antioxidant and hypotensive activities. With the body of reported data, it has been suggested that Cassiae semen has convincing medicinal potential. However, the pharmacological mechanisms of the main bioactive compounds and the association between structure and activity require further investigation.
Keywords: Cassiae semen, phytochemistry, pharmacology
1. Introduction
Cassiae semen, also known as ‘Juemingzi’ in Chinese, is the dry and mature seed of Cassia obtusifolia L. or C. tora L., which belong to the Cassia genus of Leguminosae (1). It is cultivated in Korea, Japan and China, and is commonly consumed as a roasted tea (2,3). In traditional Chinese medicine, it has been used in treatments for hyperlipemia, diabetes mellitus, Alzheimer's disease, acute liver injury, inflammation, photophobia, headache, dizziness and hypertension (4–6).
Phytochemical investigations have isolated and identified >70 compounds, including anthraquinones, naphthopyrones, volatile oils and sterols (7,8). Among these, anthraquinones are the primary functional components and possess a wide spectrum of pharmacological properties (9–11), including antihyperlipidemic, neuroprotective, hepatoprotective, antibacterial and antimutagenic activities (12–14). Naphthopyrones, other primary components, exhibit antidiabetic (15,16), antimicrobial (17), antiestrogenic (18), antiallergic (19) and anthelmintic effects (20). At present, the Pharmacopoeia of the People's Republic of China recommends the use of chrysophanol and aurantio-obtusin as the indicator components, and the quality of Cassiae semen is evaluated primarily by assessing the content of these two compounds (1).
The purpose of the present review is to provide comprehensive information on the ethnobotany, phytochemistry and pharmacology of Cassiae semen collated from previous studies, in order to facilitate the further study and application of Cassiae semen, as well as generate a novel basis for the associations between structure and activity, and their molecular mechanisms of action.
2. Ethnobotany
C. obtusifolia is similar to C. tora in terms of botanical morphology. The two are an annual, erect, stout herb, ~1–2 m in length, and their leaves are paripinnate, typically pubescent and are 4–8 cm in length with a conical gland between each of the two lowest pairs of leaflets. Leaflets are formed of 3 leaf pairs and are glaucous, membranous, glabrous or pubescent, and have obcordate or obovate oblong morphology (2–6 cm long × 1.5–2.5 cm wide); the base is somewhat oblique, usually rounded and there are 8–10 pairs of main nerves. The petiolules are 1.5–2 mm in length and their stipules are linear, pilose and caducous. It blossoms from July to September and produces fruit from September to October. Flowers are usually in subsessile pairs in leaf axils, the pedicels are filiform and are 1–1.5 cm in length. Calyces are ovate, glabrous, membranous and comprised of five-parts; there are five petals, which are pale yellow, oblong, obtuse and the upper petal (standard) is two-lobed. The flowers have 10 stamens, while the upper three are reduced to minute staminodes. The pods are slender, puberulous, subtetragonous, obliquely septate and, are ~15 cm in length and 3–4 mm in width.
However, the seeds of C. obtusifolia are a dark brown or green-brown, rhombohedral or short cylindrical, and are 3–7 mm in length and 2–4 mm in width. While C. tora seeds are a light brown, shiny, short cylindrical, and are 3–5 mm in length and 2–3 mm in width (1,21).
C. obtusifolia is cultivated in multiple provinces of China, including Henan, Hubei, Shanxi, Sichuan, Zhejiang and Anhui, and also other countries, including Korea, India and Japan. It is primarily distributed in moist and sunny places, in hillside shrubs and in the sandy soil of river banks (21).
As a widely used traditional Chinese medicine, there are some adulterants of this plant, including the seeds of C. occidentalis (Leguminosae), C. sophera (Leguminosae) and Sesbania aculeata Pers (Papilionaceae) (22–24). To date, a number of methods have been developed to identify and distinguish these, including experiential identification, morphological identification, ultraviolet spectrophotometry, the thin layer chromatography method, high performance liquid chromatography (HPLC), HPLC-coupled with time-of-flight and ion trap mass spectrometry, and SDS-PAGE (25–28). Among these methods, the HPLC method is regarded as the most popular method for evaluating the quality and authenticity of Cassiae semen. Chrysophanol and aurantio-obtusin are used as the indicator compounds to characterize the quality of this plant and the minimum contents are defined as 0.20 and 0.080%, respectively, in the Pharmacopoeia of the People's Republic of China (1).
3. Phytochemistry
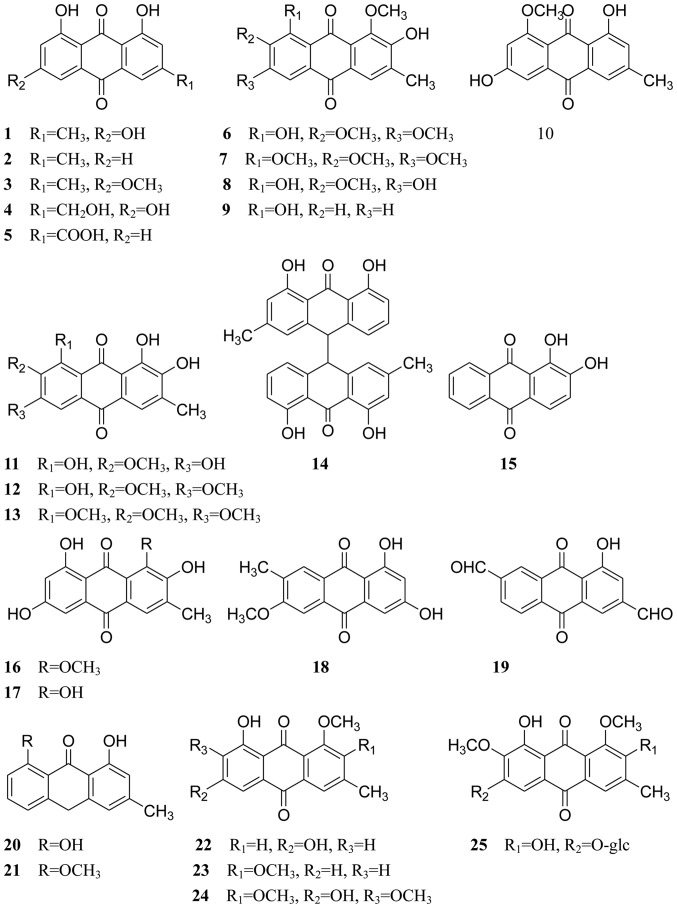
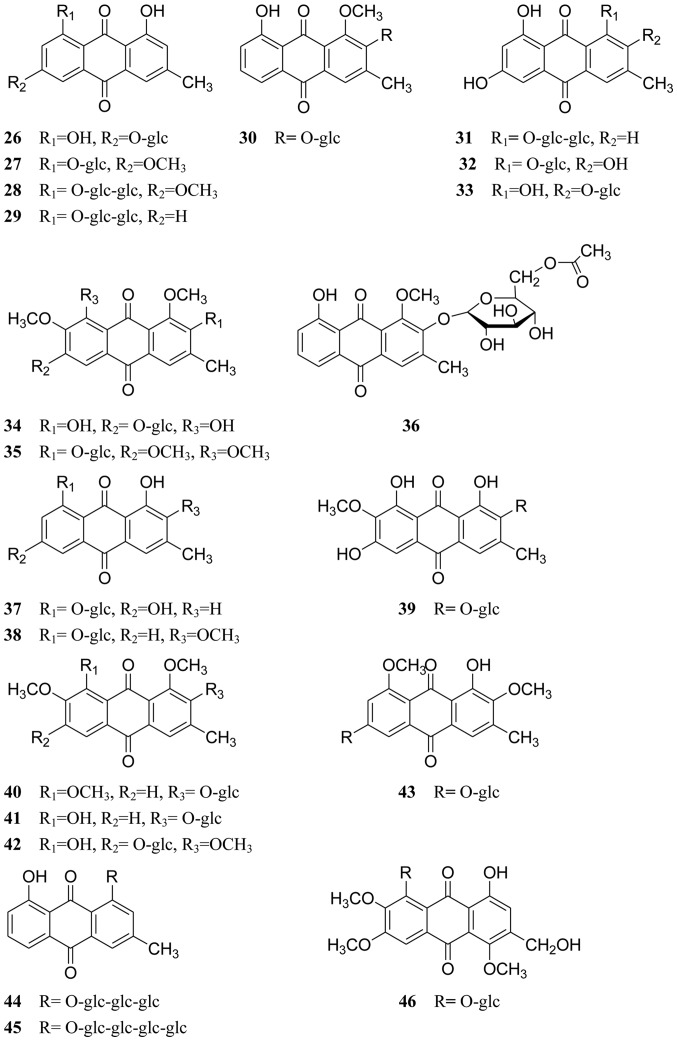
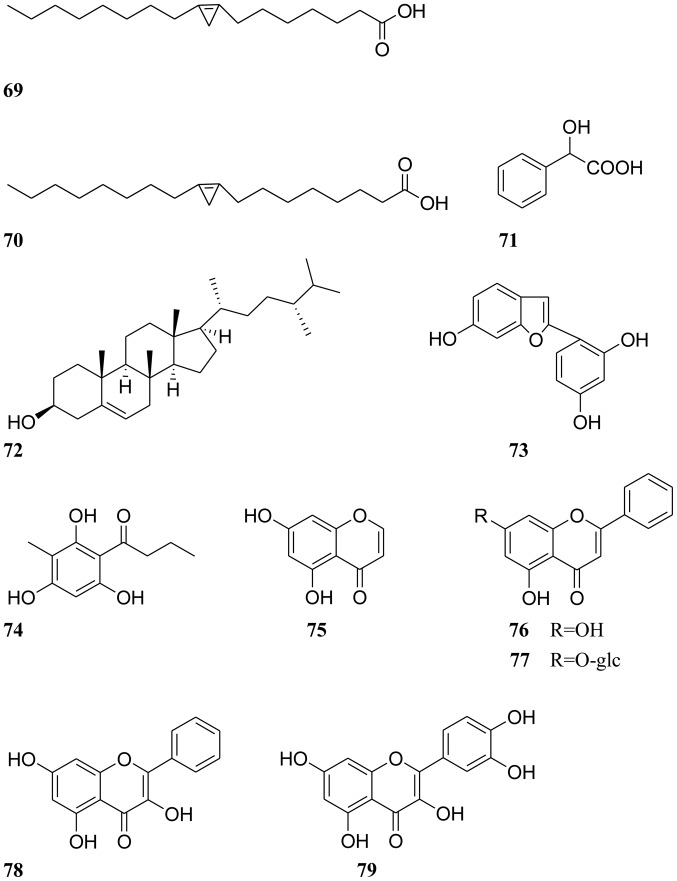
A number of compounds, including anthraquinones, naphthopyrones, volatile oils and sterols, have been isolated from Cassiae semen. Anthraquinones and naphthopyrones, which exhibit multiple pharmacological activities, are considered the primary active ingredients of Cassiae semen. All compounds isolated from Cassiae semen are listed in Table I, and their chemical structures are displayed in Figs. 1–3.
Table I.
Chemical compounds isolated from Cassiae semen.
| Classification | No. | Chemical component | (Refs.) |
|---|---|---|---|
| Anthraquinones | 1 | Emodin | (37,38) |
| 2 | Chrysophanol | (34) | |
| 3 | Physcion | (34) | |
| 4 | Aloe-emodin | (38) | |
| 5 | Rhein | (38) | |
| 6 | Obtusin | (35) | |
| 7 | Chryso-obtusin | (35) | |
| 8 | Aurantio-obtusin | (35) | |
| 9 | Obtusifolin | (35) | |
| 10 | Questin | (36) | |
| 11 | 1-desmethylaurantio-obtusin | (36) | |
| 12 | 1-desmethylobtusin | (36) | |
| 13 | 1-desmethylchryso-obtusin | (36) | |
| 14 | Chrysophanol-10,10′-bianthrone | (36) | |
| 15 | 1,2-dihydroxyanthraquinone | (7) | |
| 16 | 2-hydroxyemodin-1-methylether | (7) | |
| 17 | Alaternin | (29) | |
| 18 | 1,3-dihydroxy-6-methoxy-7-methyl anthraquinone | (30) | |
| 19 | 1-hydroxy-3,7-diformyl anthraquinone | (30) | |
| 20 | Chrysarobin | (30,31) | |
| 21 | 8-O-methylchrysophanol | (32) | |
| 22 | 1-O-methylemodin | (32) | |
| 23 | 1,2-dimethoxy-8-hydroxy-3-methyl-9,10-anthraquinone | (32) | |
| 24 | l,2,7-trimethoxyl-6,8-dihydroxy-3-methylanthraquinone | (33) | |
| 25 | Gluco-aurantioobtusin | (39) | |
| 26 | Emodin-6-glucoside | (30) | |
| 27 | Physcion-8-O-β-D-glucopyranoside | (7,40) | |
| 28 | Physcion-8-O-β-gentiobioside | (41) | |
| 29 | Emodin-1-O-β-gentiobioside | (41) | |
| 30 | Obtusifolin-2-O-β-D-glucoside | (42) | |
| 31 | Chysophanol-1-O-β-gentiobioside | (41) | |
| 32 | Alaternin-1-O-β-D-glucopyranoside | (7) | |
| 33 | Alaternin-2-O-β-D-glucopyranoside | (29) | |
| 34 | Aurantio-obtusin-6-O-β-D-glucopyranoside | (43) | |
| 35 | Chryso-obtusin-2-O-β-D-glucopyranoside | (7) | |
| 36 | Obtusifolin-2-O-β-D-(6′-O-acetyl) glucopyranoside | (44) | |
| 37 | Emodin-8-O-β-D-glucopyranoside | (45) | |
| 38 | 2-methoxyl-chrysophanol-8-O-β-D-glucopyranoside | (46) | |
| 39 | 1-demethylaurantio-obtusin-2-O-β-D-glucopyranoside | (43) | |
| 40 | 1,7,8-trimethoxyl-2-hydroxyl-3-methylanthraquinone-2-O-β-D-glucopyranoside | (33) | |
| 41 | l,7-diinethoxyl-2,8-dihydroxyl-3-methylanthraquinone-2-O-β-D-glucopyranoside | (33) | |
| 42 | l,2,7-trimethoxyl-6,8-dihydroxy-3-methylanthraquinone-6-O-β-D-glucopyranoside | (33) | |
| 43 | 2,8-dimethoxyl-l,6-dihydroxy-3-methylanthraquinone-6-O-β-D-glucopyranoside | (33) | |
| 44 | 1-[(β-D-glucopyranosyl-(1→3)-O-β-glucopyranosyl-(1→6)-O-β-D-glucopyranosyl)oxy]-8-hydroxy-3-methyl-9,10-anthraquinone | (42) | |
| 45 | 1-[(β-D-glucopyranosyl-(1→6)-O-β-glucopyranosyl-(1→3)-O-β-D-glucopyranosyl-(1→6)-O-β-D-glucopyranosyl)oxy]-8-hydroxy-3-methyl-9,10-anthraquinone | (42) | |
| 46 | 4,6,7-trimethoxyl-aloe-emodin-8-O-β-D-glucopyranoside | (46) | |
| Naphthopyrones | 47 | Torachrysone | (48) |
| 48 | Rubrofusarin | (47) | |
| 49 | Rubrofusarin-6-O-β-D-gentiobioside | (47) | |
| 50 | Toralactone | (49) | |
| 51 | Rubrofusarin triglucoside | (17) | |
| 52 | Cassiaside | (50) | |
| 53 | Nor-rubrofusarin gentiobioside | (17) | |
| 54 | Cassiaside B | (50) | |
| 55 | Cassiaside C | (50) | |
| 56 | Torachrysone apiglucoside | (17) | |
| 57 | Torachrysone gentiobioside | (17) | |
| 58 | Torachrysone tetraglucoside | (17) | |
| 59 | Cassiatoroside | (51) | |
| 60 | Demethylflavasperone gentiobioside | (17) | |
| 61 | Cassialactone gentiobioside | (28) | |
| 62 | Torosachrysone | (53) | |
| 63 | Isotoralactone | (53) | |
| 64 | Cassialactone | (53) | |
| 65 | Cassiaside B2 | (19) | |
| 66 | Cassiaside C2 | (19) | |
| 67 | Nor-rubrofusarin-6-O-β-D(6′-o-acetyl) glucopyranoside | (54) | |
| 68 | l-hydroxyl-2-acetyl-3,8-dimethoxy-6-O-[β-D-apiofuranosyl-(l→2)-β-D-glucopyranosyl]-naphthalene | (55) | |
| Other compounds | 69 | Malvalic acid | (5,57) |
| 70 | Sterculic acid | (5) | |
| 71 | Mandelic acid | (5) | |
| 72 | Campesterol | (5) | |
| 73 | Aspidinol | (58) | |
| 74 | 5,7-dihydroxychromone | (58) | |
| 75 | Chrysin | (58) | |
| 76 | Chrysin-7-O-β-D-glucoside | (58) | |
| 77 | Galangin | (58) | |
| 78 | Cyanidenon | (58) |
Figure 1.
Chemical structures of anthraquinones. Chemical structures of anthraquinones.
Figure 3.
Chemical structures of other compounds.
Anthraquinones
Cassiae semen contains structurally diverse and biologically active anthraquinones. Thus far, ~53 anthraquinones have been isolated and identified. The predominant anthraquinones are emodin-type anthraquinones, which include emodin, chrysophanol, physcion, aloe-emodin, rhein, obtusin, chryso-obtusin, aurantio-obtusin, obtusifolin, questin, 1-desmethylaurantio-obtusin, 1-desmethylobtusin, 1-desmethylchryso-obtusin, chrysophanol-10,10′-bianthrone, 1,2-dihydroxyanthraquinone, 2-hydroxyemodin-1-methylether, alaternin, 1,3-dihydroxy-6-methoxy-7-methyl anthraquinone, 1-hydroxy-3,7-diformyl anthraquinone, chrysarobin, 8-O-methylchrysophanol, 1-O-methylemodin, 1,2-dimethoxy-8-hydroxy-3-methyl-9,10-anthraquinone and l,2,7-trimethoxyl-6,8-dihydroxy-3-methylanthraquinone (compounds 1–24, respectively; Fig. 1); these have all been isolated from Cassiae semen (29–38). There are also many combined anthraquinones (compounds 25–46; Fig. 1), which have been isolated from the seeds of C. obtusifolia or C. tora (3,7,30,36,39–46).
Naphthopyrones
Naphthopyrones are the other characteristic components in Cassiae semen. In 1969, torachrysone, rubrofusarin and rubrofusarin-6-O-β-D-gentiobioside were isolated from C. tora seeds (compounds 47–49, respectively; Fig. 2) (47,48). Subsequently, toralactone, rubrofusarin triglucoside, cassiaside, nor-rubrofusarin gentiobioside, cassiaside B, cassiaside C, torachrysone apiglucoside, torachrysone gentiobioside, torachrysone tetraglucoside, cassiatoroside, demethylflavasperone gentiobioside and cassialactone gentiobioside were isolated from C. tora seeds (compounds 50–61, respectively; Fig. 2) (17,28,49–51). In addition, cassiaside B and cassiaside C were identified in the seeds of C. obtusifolia (52). Other naphthopyrones, including torosachrysone, isotoralactone, cassialactone, cassiaside B2, cassiaside C2, nor-rubrofusarin-6-O-β-D (6′-O-acetyl) glucopyranoside and l-hydroxyl-2-acetyl-3,8-dimethoxy-6-O-[β-D-apiofuranosyl- (l→2)-β-D-glucopyranosyl]-naphthalene were also isolated from C. obtusifolia seeds (compounds 62–68, respectively; Fig. 2) (19,53–55).
Figure 2.
Chemical structures of naphthopyrones.
Volatile oils
Li (56) extracted the volatile oils from Cassiae semen by steam distillation, and subsequently identified 37 components according to the gas chromatography/mass spectrometry analysis. Among these peaks, the major volatile components were 9-octadecenoic acid (E) (22.15%), n-hexadecanoic acid (12.53%), 9,10-anthracenedione, 1,8-dihydroxy-3-methyl (7.66%), octadecanoic acid (4.56%) and 13-octadecenoic acid methyl ester (Z) (3.84%) (56).
Other compounds
A range of other components have been isolated from Cassiae semen, including malvalic acid, sterculic acid, mandelic acid, campesterol, aspidinol and 5,7-dihydroxychromone (compounds 69–74, respectively; Fig. 3) (5,57,58). In addition, the four flavonoid compounds, chrysin, chrysin-7-O-β-D-glucoside, galangin and cyanidenon, were also obtained and identified from Cassiae semen (compounds 75–78, respectively; Fig. 3) (58).
4. Pharmacology
Cassiae semen exerts a great variety of pharmacological activities due to its complex bioactive compounds
An overview of the pharmacological studies on Cassiae semen is presented in detail in the following sections.
Antihyperlipidemic activity
In traditional Chinese herbal medicine, Cassiae semen is used for the prevention and treatment of hyperlipidemia. Several Chinese herbal formulations containing Cassiae semen is available in the Chinese market for preventing the formation of atherosclerotic plaques (59). In certain Asian countries, including China and Korea, it is also commonly drunk as a roasted tea to reduce body weight (60,61). Previous studies using mice have evaluated the reductions in blood lipid contents induced by different Cassiae semen extracts obtained through different methods, including supercritical fluid extraction, systematic solvents (petroleum ether, ethyl acetate, n-butanol, 70% ethanol and water) and ethanol precipitation following water extraction. The results revealed that the n-butanol and ethyl acetate extracts were the most effective (62,63). In addition, the ethanol and aqueous extracts of Cassiae emen significantly decreased the serum levels of total cholesterol (TC), triglyceride (TG) and low-density lipoprotein cholesterol (LDL-C), however, they increased the levels of high-density lipoprotein cholesterol (HDL-C) (13,64,65). Similarly, He et al (66) reported that treatment with the water extract form of C. obtusifolia seeds decreased the blood-lipid level by inhibiting cholesterol synthesis. Cho et al (67) demonstrated that soluble fibers from C. tora seeds markedly decreased liver TC and TG levels in rats fed with a high-cholesterol diet. The underlying mechanism may be mediated by increasing fecal bile acid excretion and downregulating the production of lipogenic enzymes (67). In addition, soluble fibers decreased the serum levels of TC, TG and LDL-C in patients with type II diabetes without serious adverse effects (2). Liu et al (68) revealed that the ethanol extract of Cassiae semen upregulated the expression levels of peroxisome proliferator-activated receptor (PPAR)-γ, sterol regulatory element-binding protein-1c, hormone-sensitive lipase and triacylglycerol hydrolase, however, tumor necrosis factor receptor superfamily member 6 was downregulated in adipose tissue. The anti-hyperlipidemia activity of Cassiae semen is primarily due to its antioxidant components, such as anthraquinones and polysaccharides. There are a variety of bioactive anthraquinone components in Cassiae semen, including chrysophanol, physcion, aurantio-obtusin, obtusifolin and emodin, which have been observed to decrease the levels of TC and TG (69,70). Previous studies have demonstrated that anthraquinones isolated from Cassiae semen were effective substances during hypolipidemic activities (71,72). These results were verified by a previous study, which applied an experimental hyperlipidemic rat model to investigate anthraquinone treatment (80 and 20 mg/kg, per os, for 20 days). The TC, TG and LDL-C levels were significantly reduced in a dose-dependent manner, however, the levels of HDL-C increased. Inhibition of cholesterol synthesis may be one of the underlying mechanisms involved in decreasing blood lipid levels (73). Water-soluble polysaccharides (WSPs) from Cassiae semen markedly inhibited the activities of α-amylase and pancreatic lipase, however, protease activity increased. The results demonstrated that WSPs had the ability to bind to bile acids and reduce the absorption of cholesterol, indicating that WSPs may have potential as an effective herbal ingredient in functional food applications (74).
Antidiabetic activity
A number of studies have demonstrated that Cassiae semen exhibits anti-diabetic activity. A total of three naphthopyrone glucosides (compounds 49, 52 and 55) isolated from the butanol-soluble extract of Cassia semen have been evaluated for their inhibitory activity on advanced glycation end products (AGEs) formation in vitro. The results revealed that these compounds possessed more potent inhibitory activity against AGEs compared with the aminoguanidine positive control (15). In addition, rubrofusarin-6-O-β-d-gentiobioside (compound 49) and cassiaside (compound 52) significantly inhibited the expression of transforming growth factor (TGF)-1 and extracellular matrix protein in glomerular mesangial cells cultured under diabetic conditions, suggesting that the active compounds in Cassiae semen may be effective in the treatment of renal complications associated with diabetes (16). Similarly, Kim et al (75) evaluated the preventive effects of the methanol extract of Cassia semen (200 mg/kg/day, for 12 weeks) on the development of diabetic nephropathy in streptozotocin (STZ)-induced diabetic rats. The results indicated that oral treatment with the Cassia semen methanol extract inhibited the development of diabetic nephropathy by inhibiting AGEs accumulation, receptor for advanced glycosylation end product and cyclooxygenase-2 expression in the renal cortex of STZ-diabetic rats (75,76). In addition, Zhu (77) reported that the water extract of Cassia semen exhibited protective activity against STZ-induced renal fibrosis in diabetic rats. The underlying mechanisms may be associated with its ability to downregulate the expression of TGF-β1, connective tissue growth factor and mothers against decapentaplegic homolog 3 (smad3), as well as upregulating the protein expression of smad6 (77).
Neuroprotective activity
The ethanolic extract from the seeds of C. obtusifolia has been reported to have a neuroprotective effect in brain disease models. Kim et al (6) suggested that C. obtusifolia (25, 50 or 100 mg/kg) significantly attenuated scopolamine or transient bilateral common carotid artery occlusion (2VO)-induced memory impairment. These effects are mediated by the enhancement of the cholinergic nervous system via acetylcholinesterase inhibition in a dose-dependent manner [half maximal inhibitory concentration (IC50)=81.6 µg/ml] (6). In addition, C. obtusifolia (10 or 50 mg/kg/day) exhibited a neuroprotective effect in a mouse transient global ischemia model due to its anti-inflammatory properties and the induced upregulated expression of phosphorylated cyclic AMP response element binding protein and brain-derived neurotrophic factor (78).
Drever et al (79) demonstrated that treatment with C. obtusifolia (0.1–10 µg/ml) significantly attenuated secondary calcium dysregulation and cell death induced by N-methyl-D-aspartate and 3-nitropropionic acid in mouse hippocampal cultures, and no significant effect on cell death was induced by incubation with naturally-secreted oligomers of amyloid (A)β. Yi et al (80) reported for the first time, that C. obtusifolia (10 µg/ml) ameliorated the Aβ-induced synaptic dysfunction model through anti-inflammatory and protein kinase B (Akt)/glycogen synthase kinase-3β pathways. The results suggested that the neuroprotective effect may be attributable to obtusifolin (compound 9) and/or alaternin (compound 17) (80). In a further experiment, C. obtusifolia (0.1–1 µg/ml) inhibited cell damage against oxidopamine (6-OHDA)-induced dopaminergic (DA) neural toxicity in PC12 cells through an anti-oxidant and antimitochondrial-mediated apoptosis mechanism. In a mesencephalic DA culture, C. obtusifolia (0.1–1 µg/ml) protected the DA cells against 6-OHDA- and N-methyl-4-phenylpyridinium iodide-induced toxicities. In addition, C. obtusifolia (50 mg/kg/day for 15 days) significantly protected DA neuronal degeneration in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced mouse Parkinson's disease (PD) model by inhibiting the movement impairment and the loss of DA neurons, indicating that C. obtusifolia may be a useful neuroprotective candidate for PD (81). In addition, protein and anthraquinone glucosides from Cassia semen improved learning and memory capacity, inhibited the malondialdehyde (MDA) and monoamine oxidase levels, and enhanced the level of superoxide dismutase (SOD) in the cerebrum of senile mice (82).
Hepatoprotective activity
It was recorded in the Compendium of Materia Medica that Cassiae semen exhibited the functions of nourishing the liver and improving vision (83). In Korea, the aqueous extract of C. tora L. seeds has been used for the protection of the liver. A weak anti-hepatotoxic activity in CCl4-induced mice was observed when the drug was administered orally at a dose of 670 mg/kg (84). The methanol extract of C. tora L. seeds exhibited significantly protective effects in primary cultured hepatocytes against CCl4 and toxicity. A total of two anthraquinone glycosides (compounds 44 and 45) and three naphthoypyrone glycosides (compounds 49, 51 and 53) from the methanol extract of C. tora seeds were the primary chemical constituents (42,50). The preventative effects of Cassiae semen on acute liver injury in mice induced by CCl4 have also been investigated (85,86). When compared with the control group, varying concentrations of the aqueous extract of Cassiae semen significantly increased the serum levels of SOD, and decreased the serum levels of aspartate transaminase (AST), alanine transaminase (ALT) and MDA. These results indicate that Cassiae semen is potentially beneficial in the treatment or prevention of hepatic damage (85,86). In addition, the ethanol extract of Cassiae semen has been observed to increase the serum levels of SOD and to decrease the serum levels of TG, TC, MDA, AST and ALT (86). Total anthraquinones from Cassiae semen exhibited a protective effect on alcohol-induced acute liver injury in mice by regulating fat metabolism, improving liver function and increasing the mRNA and protein expression levels of PPAR-γ (10,87).
Antibacterial activity
Antibacterial activity, an important effect of Cassiae semen, has been comprehensively investigated. Naphthalenes (compounds 47 and 50) and anthraquinones (compounds 1, 4 and 5) isolated from C. tora seeds exhibited significant antibacterial effects on four strains of methicillin-resistant Staphylococcus aureus [minimal inhibitory concentration (MIC) was 2–64 µg/ml] and a strain of methicillin-sensitive S. aureus. In addition, rhein (compound 5) and torachrysone (compound 47) from the seeds of C. tora exhibited antibacterial activity against Escherichia coli K12 with MIC values of 512 and 128 µg/ml, respectively (17). Kim et al (88) were the first to demonstrate that emodin (compound 1) from C. tora seeds has a median lethal dose (LC50) value of 0.102, 0.163, 0.385 and 0.046 g/l against Rhizoctonia solani, Botrytis cinerea, Phytophthora infestans and Erysiphe graminis, respectively, and physcion (compound 3) has an LC50 value of 0.248, 0.263, 0.518, and 0.073 g/l against R. solani, B. cinerea, P. infestans and E. graminis, respectively. In addition, the LC50 value of rhein (compound 5) is 0.375, 0.478, and 0.047 g/l against R. solani, B. cinerea and P. infestans, respectively (88).
It has been reported that ethanol and aqueous extracts of C. obtusifolia seeds were inhibitory against Helicobacter pylori strains (MIC were 100 and 60 µg/ml, respectively) (89). In addition, 1,2-dihydroxyanthraquinone (compound 15) isolated from C. obtusifolia seeds was revealed to inhibit the growth of Clostridium perfringens and E. coli., indicating that this drug exhibited potent growth-inhibiting activities towards human intestinal bacteria (90). Li et al (91) demonstrated that the chloroform extract of the seeds of C. obtusifolia also exhibited different inhibitory activities against Fusarium oxysporum and B. cinerea (IC50 values were 0.57 mg/ml and 0.97 mg/ml).
Antioxidant activity
The water extract of C. tora seeds accelerated the oxidation of deoxyribose induced by Fe3+-EDTA/H2O2 and exhibited 94% inhibition of linoleic acid peroxidation at a concentration of 0.2 mg/ml. The underlying mechanisms of this may be mediated by reducing metal ions, scavenging hydroxyl radical and chelating ferrousion (92,93). Xv and Hu (94) demonstrated that the water extract of Cassiae semen exhibited a potent ability to scavenge free oxygen radicals [IC50 values were 2 mg/ml and 2 µg/ml for hydroxyl radicals (OH-) and hydrogen peroxide (H2O2), respectively]. WSP from Cassiae semen (0.022 mg/ml) effectively inhibited superoxide radicals (O2-) induced by pyrogallol autoxidation (95). The inhibitory effects of WSP on serum levels of MDA were used to evaluate its antioxidation capabilities. The results demonstrated that WSP decreased MDA serum levels with an IC50 value of 15.80% (96). In another study, Liu et al (97) optimized the extraction conditions for WSP of Cassiae semen (temperature 80°C, extraction time 3.5 h, solid-liquid ratio 1:30) and observed that WSP (94.03 µg/ml) had the ability to scavenge hydroxyl and superoxide radicals with scavenging rates of 43.32 and 64.97%, respectively.
In addition, ethyl acetate fraction and n-butanol fraction of Cassiae semen were evaluated by DPPH radical scavenging activity. The results revealed that the ethyl acetate fraction had a lower IC50 value of 56.4 g/ml, when compared with the value of 80.6 g/ml for n-butanol fraction. 1-Desmethylaurantio-obtusin (compound 11) exhibited good scavenging activity on DPPH with an IC50 value of 4.5±0.7 g/ml, while aurantio-obtusin-6-O-β-D-glucopyranoside (compound 34) and questin (compound 10) exhibited moderate antioxidant activity, and their IC50 values were 103.2±1.5 g/ml and 185.2±1.8 g/ml, respectively. When compared with these results, chryso-obtusin (compound 7) and aurantio-obtusin (compound 8) demonstrated weaker antioxidant activity (IC50 >200 µg/ml) (98). The methanolic extract of C. tora seeds exhibited a high antioxidant activity on lipid peroxidation (99). Similarly, in another study, Yen et al (12) demonstrated that the methanolic extract of C. tora seeds exerted a greater antioxidant activity than the other organic solvents (n-hexane and ethyl acetate). Emodin was also revealed to be an antioxidative component (12). In addition, alaternin (compound 17), cassiaside (compound 52) and rubrofusarin-6-O-β-D-gentiobioside (compound 49) isolated from C. tora seeds exhibited good scavenging activity against DPPH radicals with IC50 values of 17.59, 32.52 and 18.04 µg/ml, respectively (100).
Hypotensive activity
Aqueous and ethanol extracts of Cassiae semen have been reported to possess hypotensive effects (101). Koo et al (101) reported that the water extract of C. tora seeds (3.75, 7.5, 15, 30, 60 and 250 mg/kg) consistently reduced arterial blood pressure in anesthetized rats. A potential reflex mechanism of this hypotensive action may involve a vagal reflex, which reciprocally inhibits the peripheral vasomotor tone via a reflex reduction in the sympathetic neural outflow to blood vessels (102). In addition, the media portion of the medullary reticular formation has been revealed to be directly involved in the hypotensive effect of C. tora seeds (103). Furthermore, the ethanol extract of Cassiae semen significantly decreased blood pressure in hypertensive rats by inhibiting receptor-controlled calcium channels on vessels and regulating the secretion of nitric oxide and inducible nitric oxide synthase (104).
Other activities
In addition to the pharmacological effects described above, Cassiae semen and its ingredients have other pharmacological effects, including estrogenic, anti-allergic, antigenotoxic, anti-aggregatory, antimutagenic and cardioprotective effects. Some of these effects are discussed briefly below.
The estrogenic activity of C. obtusifolia seeds was evaluated by a recombinant yeast screening assay. The results revealed that 70% EtOH extracts of this drug exhibited estrogenic relative potency [half maximal effective concentration (EC50) was 60.2 µg/ml) (105). Cassiaside C2 (compound 66) isolated from C. obtusifolia seeds exhibited a potent anti-allergic activity by inhibiting the histamine release from mast cells induced by antigen-antibody reaction (19). Furthermore, gluco-aurantioobtusin (compound 25) from C. obtusifolia seeds possessed potent inhibitory activities against arachidonic-acid-, ADP- and collagen-induced platelet aggregations (39). Wu and Yen (106) demonstrated that the water extract of C. tora seeds exhibited potential antigenotoxic activities against the dietary mutagens Glu-P-1 and TrpP-1 in the Ames test and the Comet assay. The potential mechanisms may be associated with neutralization of the reactive intermediate of Trp-P-1 and an antioxidant effect of the tested compounds (106). Anthraquinone aglycones (compounds 2, 7 and 8) and naphthopyrone glycosides (compounds 49 and 52) from C. tora seeds exhibited significant antimutagenic activity in vitro. The mechanism associated with these compounds may be mediated via interactions with a microsomal activating system (14). Fu et al (107) reported that the water extract of Cassiae semen (10 mg/kg/day, for one week) effectively improved myocardial function, and attenuated myocardial ischemia and reperfusion-induced injury and apoptosis in diabetic animals, which is potentially attributable to the reduced plasma lipid levels and the triggered cell survival Akt and extracellular signal-regulated kinases 1/2 signaling.
5. Conclusions
In traditional Chinese medicine, Cassiae semen has long been used to clean the liver, brighten the eye, loosen the bowel to relieve constipation, and for the treatment of inflammation, photophobia, headaches, dizziness, hyperlipemia and Alzheimer's disease. In addition, Cassiae semen is commonly used in the composition of other herbs. Although modern experiments have confirmed that this drug alone exhibits multiple pharmacological activities, it is important to investigate the molecular mechanisms of Cassiae semen combined with other herbs based on traditional uses.
A number of studies have investigated the effective constituents of Cassiae semen from different batches and geographical areas. HPLC-fingerprint chromatography is a common method to compare the differences (108–111). Zhang et al (3) developed a sensitive and reliable ultra-high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry (UHPLC-ESI-MS/MS) method to evaluate the quality of Cassiae semen through simultaneous determination of 13 components, providing a novel basis for the overall assessment of the quality of this plant. In addition, a novel nonaqueous capillary electrophoresis method was used for the analysis of aurantio-obtusin, emodin and rhein in Cassiae semen with satisfactory results (112). Yang et al (113) was the first to simultaneously determine 7 anthraquinones in rat plasma by UHPLC-MS/MS following oral administration of Cassiae semen extract. These results may support investigations into the bioactivity mechanism and clinical application of this drug (113). Anthraquinones and naphthopyrones are considered to be the major constituents. Therefore, characteristic compounds or a biological index should be established to evaluate the quality and ensure their clinical application is suitable. In the Pharmacopoeia of the People's Republic of China, chrysophanol and aurantio-obtusin are used as the indicator compounds to characterize the quality of Cassiae semen with the minimum contents of 0.20 and 0.080%, respectively (1).
A total of 79 compounds including anthraquinones, naphthopyrones and volatile oil have been isolated and identified from Cassiae semen (Table I; Figs. 1–3). It has also been suggested that certain efforts should be made to isolate and identify novel compounds from Cassiae semen, in order to strengthen its pharmacological profile to develop it further as a candidate for novel drug developments in the future.
Pharmacological studies have revealed that Cassiae semen possesses a variety of biological effects, including anti-hyperlipidemic, anti-diabetic, neuroprotective, hepatoprotective, antimicrobial, anti-oxidant and hypotensive activities (5,90,93,114,115). Extracts and compounds responsible for the pharmacological properties have also been determined, as presented in Table II. Although the pharmacological properties of certain traditional uses of Cassiae semen have been validated, these studies were primarily conducted in vitro (16,116,117). Therefore, the effects of these compounds require verification in vivo. In addition, the association between structure and activity, and the potential synergistic action exerted by the bioactive compounds requires further elucidation. It is anticipated that the comprehensive and current research on the pharmacological activities of extracts, as well as on active molecules isolated from Cassiae semen, provided in this review will inspire novel strategies in therapeutics for curing a number of different ailments.
Table II.
Pharmacological activities of Cassiae semen.
| Pharmacological activities | Actions | Extracts/compounds | Application | (Refs.) |
|---|---|---|---|---|
| Anti-hyperlipidemia activity | Reduces blood lipid levels | SFE, systematic solvents (petroleum ether, ethyl acetate, n.butanol, 70% ethanol and water) | In vivo | (62,63) |
| Decreases the levels of TC, TG and LDL-C; Increases the level of HDL-C | Ethanol and aqueous extracts | In vivo | (13,64,65) | |
| Inhibits the synthesis of cholesterol | Water extract | In vitro | (66) | |
| Increases fecal bile acid excretion and downregulates the production of lipogenic enzymes | Soluble fibers | In vivo | (67) | |
| Anti-hyperlipidemia activity | Upregulates the expression levels of PPARγ, SREBP-1c, HSL and TGH; Downregulates the levels of FAS | Ethanol extract | In vivo | (68) |
| Decreases TC and TG levels | Chrysophanol, physcion, aurantio-obtusin, obtusifolin and emodin | In vitro | (69,70) | |
| Decreases TC, TG and LDL-C levels; Increases HDL-C levels | Anthraquinones | In vivo | (73) | |
| Binds bile acids and reduces the absorption of cholesterol | Water-soluble polysaccharides | In vitro | (74) | |
| Anti-diabetic activity | Inhibits AGEs activity | Cassiaside, cassiaside C, rubrofusarin-6-O-β-D-gentiobioside | In vitro | (15) |
| Inhibits the expression of TGF-1 and ECM proteins | Rubrofusarin-6-O-β-D-gentiobioside, cassiaside | In vitro | (16) | |
| Inhibits AGEs accumulation and, RAGE and COX-2 expression | Methanol extract | In vitro/vivo | (75,76) | |
| Downregulates the expression of TGF-β1, CTGF and smad3, and upregulates the protein expression of smad6 | Water extract | In vivo | (77) | |
| Neuroprotective activity | Inhibits AChE activity | Ethanol extract | In vivo | (6) |
| Upregulates the expression of pCREB and BDNF | Ethanol extract | In vivo | (78) | |
| Attenuates secondary calcium dysregulation and cell death | Ethanol extract | In vivo | (79) | |
| Ameliorates the Aβ-induced synaptic dysfunction model | Obtusifolin, alaternin | In vivo | (80) | |
| Inhibits cell damage and protects DA neuronal degeneration | Ethanol extract | In vitro/vivo | (81) | |
| Improves learning and memory capacity; Inhibits MDA and MAO levels; Enhances the level of SOD | Protein and anthraquinone glucosides | In vivo | (82) | |
| Hepatoprotective activity | Hepatoprotective effects | Methanol extract | In vitro | (42,50) |
| Increases the serum levels of SOD and decreases the serum levels of AST, ALT and MDA | Aqueous extract | In vivo | (85,86) | |
| Increases the serum levels of SOD and decreases the serum levels of TG, TC, MDA, AST and ALT | Ethanol extract | In vivo | (10,87) | |
| Antibacterial activity | Anti-Staphylococcus aureus; Anti-Escherichia coli K12 | Naphthalenes, anthraquinones | In vitro | (17) |
| Exhibits fungicidal activity against Botrytis cinerea, Erysiphe graminis, Phytophthora infestans, Puccinia recondita, Phacelia grisea, and Rhizoctonia solani. | Anthraquinones | In vitro | (88) | |
| Anti-Helicobacter pylori | Ethanol and aqueous extracts | In vitro | (89) | |
| Anti-Clostridium perfringens; Anti-E. coli | 1,2-dihydroxyanthraquinone | In vitro | (90) | |
| Anti-Fusarium oxysporum; Anti-B. cinerea | Chloroform extract | In vitro | (91) | |
| Antioxidant activity | Accelerates the oxidation of deoxyribose; inhibits linoleic acid peroxidation | Water extract | In vitro | (92,93) |
| Scavenges free oxygen radicals | Water extract | In vitro | (94) | |
| Inhibits superoxide radicals | Water-soluble polysaccharides | In vitro | (95) | |
| Decreases MDA serum levels | Water-soluble polysaccharides | In vitro | (96) | |
| Scavenges hydroxyl and superoxide radicals | Water-soluble polysaccharides | In vitro | (97) | |
| Scavenges DPPH radicals | Ethyl acetate fraction | In vitro | (98) | |
| Antioxidant effect | Methanol extract | In vitro | (12) | |
| Scavenges DPPH radicals | Alaternin, cassiaside and rubrofusarin-6-O-β-D-gentiobioside | In vitro | (100) | |
| Hypotensive activity | Reduces arterial blood pressure | Water extract | In vivo | (101) |
| Hypotensive activity | Water extract | In vivo | (103) | |
| Decreases the blood pressure | Ethanol extract | In vitro | (104) | |
| Other activities | Estrogenic activity | 70% EtOH extract | In vitro | (105) |
| Inhibits histamine release from mast cells | Cassiaside C2 | In vitro | (19) | |
| Anti-platelet aggregation | Gluco-aurantioobtusin | In vitro | (39) | |
| Antigenotoxic activity | Water extract | In vitro | (106) | |
| Antimutagenic activity | Anthraquinone aglycones and naphthopyrone glycosides | In vitro | (14) | |
| Improves myocardial function and attenuates MI/R-induced injury | Water extract | In vivo | (107) |
TC, total cholesterol; TG, triglyceride; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; PPARγ, peroxisome proliferator-activated receptor; SREBP-1c, sterol regulatory element-binding protein-1c; HSL, hormone-sensitive lipase; TGH, triacylglycerol hydrolase; FAS, tumor necrosis factor receptor superfamily member 6; AGEs, advanced glycation end products; TGF-1, transforming growth factor-1; ECM, extracellular matrix; RAGE, receptor for advanced glycosylation end product; COX-2, cyclooxygenase-2; CTGF, connective tissue growth factor; Smad3/6, mothers against decapentaplegic homolog 3 and 6; AChE, acetylcholinesterase; pCREB, phosphorylated cyclic AMP response element binding protein; BDNF, brain-derived neurotrophic factor; DA, dopaminergic; MDA, malondialdehyde; MAO, monoamine oxidase; SOD, superoxide dismutase; AST, aspartate transaminase; ALT, alanine transaminase; DPPH, 1-diphenyl-2-picrylhydrazyl; MI/R, myocardial ischemia and reperfusion.
Acknowledgements
The present review was financially supported by the Collaborative Innovation Construction Plan of Beijing University of Chinese Medicine (grant no. 2013-XTCX-03).
References
- 1.Chinese Pharmacopoeia. 2015. Medical Science and Technology Press; Beijing, China: 2015. Editorial Committee of Chinese Pharmacopoeia; p. 145. [Google Scholar]
- 2.Cho SH, Kim TH, Lee NH, Son HS, Cho IJ, Ha TY. Effects of Cassia tora fiber supplement on serum lipids in Korean diabetic patients. J Med Food. 2005;8:311–318. doi: 10.1089/jmf.2005.8.311. [DOI] [PubMed] [Google Scholar]
- 3.Zhang W, Wang Y, Wang Q, Yang WJ, Gu Y, Wang R, Song XM, Wang XJ. Quality evaluation of Semen Cassiae (Cassia obtusifolia L.) by using ultra-high performance liquid chromatography coupled with mass spectrometry. J Sep Sci. 2012;35:2054–2062. doi: 10.1002/jssc.201200009. [DOI] [PubMed] [Google Scholar]
- 4.Chen JK, Chen TT. Chinese Medical Herbology and Pharmacology. CA: Art of Medicine Press; 2001. p. 803. [Google Scholar]
- 5.Hao YJ, Sang YL, Zhao YQ. The advancement of the studies on the seeds of Cassia obtusifolia. Chinese Tradit Herb Drugs. 2001;32:858–859. [Google Scholar]
- 6.Kim DH, Yoon BH, Kim YW, Lee S, Shin BY, Jung JW, Kim HJ, Lee YS, Choi JS, Kim SY, et al. The seed extract of Cassia obtusifolia ameliorates learning and memory impairments induced by scopolamine or transient cerebral hypoperfusion in mice. J Pharmacol Sci. 2007;105:82–93. doi: 10.1254/jphs.FP0061565. [DOI] [PubMed] [Google Scholar]
- 7.Kitanaka S, Kimura F, Takido M. Studies on the Constituents of seeds of Cassia obusifolia LINN. The structures of two new anthraquinone glycosides. Chem Pharm Bull. 1985;33:1274–1276. doi: 10.1248/cpb.33.1274. [DOI] [Google Scholar]
- 8.Zhang C, Li GL, Xiao YQ, Pang LZ. Two new glycosides from the seeds of Cassia obtusifolia. Chinese Chem Lett. 2009;20:1097–1099. doi: 10.1016/j.cclet.2009.04.006. [DOI] [Google Scholar]
- 9.Deng ZY, Zhang JW, Li J, Fan YW, Cao SW, Huang RL, Yin YL, Zhong HY, Li TJ. Effect of polysaccharides of cassiae seeds on the intestinal microflora of piglets. Asia Pac J Clin Nutr. 2007;16:143–147. (Suppl 1) [PubMed] [Google Scholar]
- 10.Luo X, Xu X, Huang C, Wu X, Liu J, Lan B, Xu J. Experiment study of total anthraquinone in Cassiae Semen on lipid peroxidation and PPAR-gamma expression in liver tissues of rats with alcoholic fatty liver. Zhongguo Zhong Yao Za Zhi. 2011;36:1654–1658. (In Chinese) [PubMed] [Google Scholar]
- 11.Su H, Wang Z, Tang L. Simultaneous determination of 4 major components in Cassiae Semen obtusifoline by HPLC. Zhongguo Zhong Yao Za Zhi. 2011;36:1327–1329. (In Chinese) [PubMed] [Google Scholar]
- 12.Yen GC, Chen HW, Duh PD. Extraction and identification of an antioxidative component from Jue Ming Zi (Cassia tora L.) J Agr Food Chem. 1998;46:820–824. doi: 10.1021/jf970690z. [DOI] [Google Scholar]
- 13.Patil UK, Saraf S, Dixit VK. Hypolipidemic activity of seeds of Cassia tora Linn. J Ethnopharmacol. 2004;90:249–252. doi: 10.1016/j.jep.2003.10.007. [DOI] [PubMed] [Google Scholar]
- 14.Choi JS, Lee HJ, Park KY, Ha JO, Kang SS. In vitro antimutagenic effects of anthraquinone aglycones and naphthopyrone glycosides from Cassia tora. Planta Med. 1997;63:11–14. doi: 10.1055/s-2006-957593. [DOI] [PubMed] [Google Scholar]
- 15.Lee GY, Jang DS, Lee YM, Kim JM, Kim JS. Naphthopyrone glucosides from the seeds of Cassia tora with inhibitory activity on advanced glycation end products (AGEs) formation. Arch Pharm Res. 2006;29:587–590. doi: 10.1007/BF02969270. [DOI] [PubMed] [Google Scholar]
- 16.Jung DH, Kim YS, Kim NH, Lee J, Jang DS, Kim JS. Extract of Cassiae semen and its major compound inhibit S100b-induced TGF-beta1 and fibronectin expression in mouse glomerular mesangial cells. Eur J Pharmacol. 2010;641:7–14. doi: 10.1016/j.ejphar.2010.04.061. [DOI] [PubMed] [Google Scholar]
- 17.Hatano T, Uebayashi H, Ito H, Shiota S, Tsuchiya T, Yoshida T. Phenolic constituents of Cassia seeds and antibacterial effect of some naphthalenes and anthraquinones on methicillin-resistant Staphylococcus aureus. Chem Pharm Bull (Tokyo) 1999;47:1121–1127. doi: 10.1248/cpb.47.1121. [DOI] [PubMed] [Google Scholar]
- 18.Das C, Dash S, Sahoo DC, Mohanty A, Rout D. Cassia tora: A phyto-pharmacological overview. Int J Res Ayurveda Pharm. 2011;2:1162–1174. [Google Scholar]
- 19.Kitanaka S, Nakayama T, Shibano T, Ohkoshi E, Takido M. Antiallergic agent from natural sources. Structures and inhibitory effect of histamine release of naphthopyrone glycosides from seeds of Cassia obtusifolia L. Chem Pharm Bull (Tokyo) 1998;46:1650–1652. doi: 10.1248/cpb.46.1650. [DOI] [PubMed] [Google Scholar]
- 20.Deore SL, Khadabadi SS, Kamdi KS, Ingle VP, Kawalkar NG, Sawarkar PS, Patil UA, Vyas AJ. In vitro Anthelmintic activity of Cassia tora. Int J Chem Tech Res. 2009;1:177–179. [Google Scholar]
- 21.Editorial Board of Flora of China. Science Publishing House Press; 1988. p. 126. [Google Scholar]
- 22.Du ZX. Comparative identification of Cassiae Semen and Sesbania aculeata Pers. Chinese Pharm J. 1995;30:204–205. [Google Scholar]
- 23.Zhu SY, Xv HJ, Xv MG. Comparative identification of Semen Cassiae and Cassia sophera. Chin Tradit Herb Drugs. 2003;34:379–380. [Google Scholar]
- 24.Liu J, Zou CC, Sun ZF. Comparative identification of Cassiae Semen and Cassia occidentalis L. Heilongjiang Med Pharm. 2006;29:63–64. [Google Scholar]
- 25.Hu YJ, Wan L, Zhang JX, et al. Identification of Cassia obtusifolia L. by TLC. Lishizhen Med Mater Med Res. 2006;17:2129. [Google Scholar]
- 26.Sun GF. Identification of Cassia obtusifolia L., Cassia tora. and Cassia occidentalis L. by SDS-Polyacrylamide gelelectrophoresis. Tianjin Pharm. 1996;8:69–71. [Google Scholar]
- 27.Wang JB, Zhou X, Hu ZF. Quality evaluation of Cassiae Semen by both indicated component determination and HPLC fingerprint. Chin Tradit Herb Drugs. 2008;39:917–919. [Google Scholar]
- 28.Luo Y, Zhang L, Wang WH, Li B. Components identification in Cassiae Semen by HPLC-IT-TOF MS. Chin J Pharm Anal. 2015;35:1408–1516. [Google Scholar]
- 29.Lee HJ, Choi JS, Jung JH, Kang SS. Alaternin glucoside isomer from Cassia tora. Phytochemistry. 1998;49:1403–1404. doi: 10.1016/S0031-9422(98)00151-4. [DOI] [Google Scholar]
- 30.Chen QD, Xv R, Xv ZN, et al. Progress in studies of active coustituents of anthraquinones and their biological activities from Cassiae Semen. Chins J Mod Appl Pharm. 2003;20:120–123. [Google Scholar]
- 31.DiGiovanni J, Boutwell RK. Tumor promoting activity of 1, 8-dihydroxy-3-methyl-9-anthrone (chrysarobin) in female SENCAR mice. Carcinogenesis. 1983;4:281–284. doi: 10.1093/carcin/4.3.281. [DOI] [PubMed] [Google Scholar]
- 32.Jia ZB, Chen WW, Jiang JX, DING XL. Study on anthraquinone constituents in the seed of Cassia tora L. Chem Ind Forest Prod. 2009;29:100–102. [Google Scholar]
- 33.Xv YL. Studies on the chemical constituents from Semen Cassiae and the influence of processing. Beijing: Chinese Academy of Medical Sciences. 2014;1:15. [Google Scholar]
- 34.Takido M. Studies on the constituents of the seeds of Cassia obtusifolia L. I. The Structure of Obtusifolin. Chem Pharm Bull. 1958;6:397–400. doi: 10.1248/cpb.6.397. [DOI] [PubMed] [Google Scholar]
- 35.Takido M. Studies on the constituents of seeds of Cassia obusifolia L. II. The structure of Obtusin, Chryso-obutsin and Aurantio-obyusin. Chem Pharm Bull. 1960;8:246. doi: 10.1248/cpb.8.246. [DOI] [Google Scholar]
- 36.Kitanaka S, Takido M. Studies on the constituents of the seeds of Cassia obusifolia: The structures of three new anthraquinones. Chem Pharm Bull. 1984;32:860–864. doi: 10.1248/cpb.32.860. [DOI] [Google Scholar]
- 37.Tang LY, Wang ZJ, Fu HM, et al. Study on anthraquinones constituents from Semen Cassiae. J Chinese Med Mater. 2009a;32:717–719. [Google Scholar]
- 38.Zhang ZX, Liang YF. Isolation and identification of chemical constituents from seeds of Cassia obtusifolia. China Pharm. 2012;23:1782–1783. [Google Scholar]
- 39.Yun-Choi HS, Kim JH, Takido M. Potential inhibitors of platelet aggregation from plant sources, V. Anthraquinones from seeds of Cassia obtusifolia and related compounds. J Nat Prod. 1990;53:630–633. doi: 10.1021/np50069a014. [DOI] [PubMed] [Google Scholar]
- 40.Hao YJ, Sang YL, Zhao YQ. Study on anthraquinone constituents in Cassiae Semen. Chinese Tradit Herb Drugs. 2003;34:18–19. [Google Scholar]
- 41.Li CH, Wei XY, Li XE. A new anthraquinone glycoside from the seed of Cassia obtusifolia. Chinese Chem Lett. 2004;15:1448–1450. [Google Scholar]
- 42.Wong SM, Wong MM, Seligmann O, Wagner H. Anthraquinone glycosides from the seeds of Cassia tora. Phytochemistry. 1989;28:211–214. doi: 10.1016/0031-9422(89)85040-X. [DOI] [PubMed] [Google Scholar]
- 43.Tang LY, Wang ZJ, Fu MH, He Y, Wu HW, Huang LQ. A new anthraquinone glycoside from seeds of Cassia obtusifolia. Chinese Chem Lett. 2008;19:1083–1085. doi: 10.1016/j.cclet.2008.06.006. [DOI] [Google Scholar]
- 44.Li G, Xiao Y, Li L, Zhang C, Pang Z. Studies on chemical constituents of roasted seeds of Cassia obtusifolia. Zhongguo Zhong Yao Za Zhi. 2009;34:54–56. (In Chinese) [PubMed] [Google Scholar]
- 45.Tang LY, Wang ZJ, He Y, et al. Glycosides from seeds of Cassia obtusifolia. Chinese J Exp Tradit Med Formulae. 2009b;15:35–37. [Google Scholar]
- 46.Jia ZB, Ding XL. Anthraquinone constituents from seeds of Cassia tora L. J Chinese Med Mater. 2006;29:28–29. [Google Scholar]
- 47.Miyuki K, Eisaku M, Shoji S. Chemical Studies on the Oriental Plant Drugs. XXI. The Constituents of Cassia tora L. (2). A Glycoside of Rubrofusarin. Chem Pharm Bull. 1969;17:458–461. doi: 10.1248/cpb.17.458. [DOI] [PubMed] [Google Scholar]
- 48.Shibata S, Morishita E, Kaneda M. Chemical Studies on the Oriental Plant Drugs. XX. The Constituents of Cassia tora L. (1). The Structure of Torachrysone. Chem Pharm Bull. 1969;17:454–457. doi: 10.1248/cpb.17.454. [DOI] [PubMed] [Google Scholar]
- 49.Takahashi S, Takido M. Studies on the constituents of the seeds of Cassia tora L. II. (On the purgative crude drugs. VII). The structure of the new naphthopyrone derivative, toralactone. Yakugaku Zasshi. 1973;93:261. doi: 10.1248/yakushi1947.93.3_261. [DOI] [PubMed] [Google Scholar]
- 50.Wong SM, Wong MM, Seligmann O, Wagner H. New antihepatotoxic naphthopyrone glycosides from the seeds of Cassia tora. Planta Med. 1989;55:276–280. doi: 10.1055/s-2006-962003. [DOI] [PubMed] [Google Scholar]
- 51.Choi JS, Jung JH, Lee HJ, Lee HJ, Lee JH, Kang SS. A naphthalene glycoside from Cassia tora. Phytochemistry. 1995;40:997–999. doi: 10.1016/0031-9422(95)00318-2. [DOI] [Google Scholar]
- 52.Tianaka S, Takido M. Studies on the constituents of the seeds of Cassia obtusifolia L. The structures of two naphthopyrone glycosides. Chem Pharm Bull. 1988;36:3980–3984. doi: 10.1248/cpb.36.3980. [DOI] [Google Scholar]
- 53.Kitanaka S, Miehio T. Studies on the constituents of the seeds of Cassia obtusifolia: The structures of two new lactones, isotoralactone and cassialactone. Phytochemistry. 1981;20:1951–1953. doi: 10.1016/0031-9422(81)84042-3. [DOI] [Google Scholar]
- 54.Li L, Zhang C, Xiao YQ, Li W, Yin X, Chen D, Tian G, Wang Y. Glycosides of roasted seeds of Cassia obtusifolia. Zhongguo Zhong Yao Za Zhi. 2010;35:1566–1568. doi: 10.4268/cjcmm20101213. [DOI] [PubMed] [Google Scholar]
- 55.Wang ZJ, Wu QP, Tang LY, Fu MH, He Y, Gong QF, Hung LQ. Two new glycosides from the genus of Cassia. Chinese Chem Lett. 2007;18:1218–1220. doi: 10.1016/j.cclet.2007.08.004. [DOI] [Google Scholar]
- 56.Li YM. Comparison of volatile components of Cassiae Semen and semen seeds tea. J Med Plants Res. 2012;6:3865–3869. [Google Scholar]
- 57.Jiao SF, Han HD. Studies on chemical constituents of Cassiae Semen. Chinese J Clin Ration Drug Use. 2010;3:81–82. [Google Scholar]
- 58.Wu XH. Study on the chemical constituents, quality control and metabolism of Cassia obtusifolia. Wuhan: Huazhong University of Science and Technology. 2010;1:11–12. [Google Scholar]
- 59.Guan Y, Zhao S. Yishou jiangzhi (de-blood-lipid) tablets in the treatment of hyperlipidemia. J Tradit Chin Med. 1995;15:178–179. [PubMed] [Google Scholar]
- 60.Yang Y, Liu J, Lai XH. Observation on cassia seed tea combined with walking exercise on weight loss in the elderly. Modern Prev Med. 2013;40:2468–2474. [Google Scholar]
- 61.Lai XH. Double intervention of cassia seed tea and sports on older women weight loss. Chinese J Gerontol. 2011;31:2402–2404. [Google Scholar]
- 62.Zhang R, Feng ML, Wu YP. Experimental study on the active situs of fetid cassia seed to reduce blood lipid and their dose-effect relation. China Remedies Clin. 2005b;5:183–185. [Google Scholar]
- 63.Zhang JX, Wan L, Hu YJ, Qu OL, Shi JY. Study on the effective part of reducing blood lipid in Semen Cassiae. Lishizhen Med Mater Med Res. 2006;17:904–905. [Google Scholar]
- 64.Li CH, Li XE, Guo BJ. The effects of Cassia obtusifolia seeds extracts on reducing blood lipid. J South China Normal Univ. 2002;98:29–32. [Google Scholar]
- 65.Wang YH, Gao L, Zhou WJ, Ma WJ. Effects of ethanol extraction from Cassiae Semen on Serum IL-6 and TNF-α in hyperlipidemia rats. Chinese J Exp Tradit Med Formulae. 2014;20:178–181. [Google Scholar]
- 66.He JY, Liu SQ, Peng YF, et al. Study of the Mechanism of Cassia Obtusifolia L in decreasing blood -lipid. China Pharm. 2003;14:202–203. [Google Scholar]
- 67.Cho IJ, Lee C, Ha TY. Hypolipidemic effect of soluble fiber isolated from seeds of Cassia tora Linn. In rats fed a high-cholesterol diet. J Agric Food Chem. 2007;55:1592–1596. doi: 10.1021/jf0622127. [DOI] [PubMed] [Google Scholar]
- 68.Liu SM, Sun C, Xie WH. Effect of Semen Cassiae extracts on expression of lipogenesis genes in hyperlipidemia model mice. Chinese Tradit Herb Drugs. 2009;40:583–587. [Google Scholar]
- 69.Wei N, Lv HR, Liu MF. Study on the chemical constitutions of reducing blood lipid in Cassiae Semen. Guangdong Chem Ind. 2012;39:99–100. [Google Scholar]
- 70.Li BL. The active ingredients of reducing blood lipid in Semen Cassia. China Prac Med. 2012;7:172–173. [Google Scholar]
- 71.Guo CY, Horn W, Pin DD. Extraction and identification of an antioxidative component from Jue Ming Zi (Cassia tora L.) J Agric Food Chem. 1998;46:820–824. doi: 10.1021/jf970690z. [DOI] [Google Scholar]
- 72.Li XE, Guo BJ. Effects of protein and anthraquinone glucosides from Cassia seed on serum lipid of hyperlipidemia rats. Zhongguo Zhong Yao Za Zhi. 2002;27:374–376. (In Chinese) [PubMed] [Google Scholar]
- 73.Li CH, Li XE, Fang KY, Guo BJ. Effects of anthraquinones from Cassia obtusifolia L. on cholesterol biosynthesis in cells. J Clin Rehabilit Tissue Eng Res. 2008;12:6593–6596. [Google Scholar]
- 74.Huang YL, Chow CJ, Tsai YH. Composition, characteristics, and in-vitro physiological effects of the water-soluble polysaccharides from Cassia seed. Food Chem. 2012;134:1967–1972. doi: 10.1016/j.foodchem.2012.03.127. [DOI] [PubMed] [Google Scholar]
- 75.Kim YS, Jung DH, Sohn E, Lee YM, Kim CS, Kim JS. Extract of Cassiae semen attenuates diabetic nephropathy via inhibition of advanced glycation end products accumulation in streptozotocin-induced diabetic rats. Phytomedicine. 2014;21:734–739. doi: 10.1016/j.phymed.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 76.Jung DH, Kim YS, Sohn EJ, et al. Effects of CS, a BuOH-Soluble Fraction of Cassiae Semen Methanolic Extract, on COX-2 Expression in Renal Cortex of STZ-Induced Diabetic Rats and Cultured Glomerular Mesangial Cells. Diabetes. 2007;56:pA155–pA155. [Google Scholar]
- 77.Zhu TC. Inhibitory Effects of Cassia Seed on the Renal Fibrosis in Diabetic Rats. Chin J Exp Tradit Med Formulae. 2012;18:315–319. [Google Scholar]
- 78.Kim DH, Kim S, Jung WY, Park SJ, Park DH, Kim JM, Cheong JH, Ryu JH. The neuroprotective effects of the seeds of Cassia obtusifolia on transient cerebral global ischemia in mice. Food Chem Toxicol. 2009;47:1473–1479. doi: 10.1016/j.fct.2009.03.028. [DOI] [PubMed] [Google Scholar]
- 79.Drever BD, Anderson WG, Riedel G, Kim DH, Ryu JH, Choi DY, Platt B. The seed extract of Cassia obtusifolia offers neuroprotection to mouse hippocampal cultures. J Pharmacol Sci. 2008;107:380–392. doi: 10.1254/jphs.08034FP. [DOI] [PubMed] [Google Scholar]
- 80.Yi JH, Park HJ, Lee S, Jung JW, Kim BC, Lee YC, Ryu JH, Kim DH. Cassia obtusifolia seed ameliorates amyloid β-induced synaptic dysfunction through anti-inflammatory and Akt/GSK-3β pathways. J Ethnopharmacol. 2016;178:50–57. doi: 10.1016/j.jep.2015.12.007. [DOI] [PubMed] [Google Scholar]
- 81.Ju MS, Kim HG, Choi JG, Ryu JH, Hur J, Kim YJ, Oh MS. Cassiae semen, a seed of Cassia obtusifolia, has neuroprotective effects in Parkinson's disease models. Food Chem Toxicol. 2010;48:2037–2044. doi: 10.1016/j.fct.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 82.Liu JZ, Lin X, Li XE, Guo BJ. Effect of protein and anthraquinone glucosides from Semen Cassiae on learning and memory capacity and related substances of senile mice induced by D-galactone. Zhongguo Zhong Yao Za Zhi. 2007;32:516–519. (In Chinese) [PubMed] [Google Scholar]
- 83.Jiangsu New Medical College: Encyclopedia of Chinese MateriaMedica, I. Shanghai Science and Technology Press; Shanghai, China: 1975. p. 94984. [Google Scholar]
- 84.Yun HS, Chang IM. Plants with liver protective activities (I) Korean J Pharmacogn. 1977;8:125–129. [Google Scholar]
- 85.Gao Q, Xu H, Chen J. Liver-protective and bowel-lubricating and defecation-promoting effects of crude and processed Semen Cassiae. Tradit Chinese Drug Res Clin Pharmacol. 2007;18:194–196. [Google Scholar]
- 86.Lin DJ, Jin Z. Experimental Study on Protective Effect of Semen Cassiae Extract Against Acute Liver Injury. Lishizhen Med Mater Med Res. 2006;17:214–215. [Google Scholar]
- 87.Niu YF, Zhao T, Zeng T, et al. Study on the protective effect of Cassiae Semen extract against alcohol-induced acute liver injury in mice. J Toxicol. 2010;24:58–61. [Google Scholar]
- 88.Kim YM, Lee CH, Kim HG, Lee HS. Anthraquinones isolated from Cassia tora (Leguminosae) seed show an antifungal property against phytopathogenic fungi. J Agr Food Chem. 2004;52:6096–6100. doi: 10.1021/jf049379p. [DOI] [PubMed] [Google Scholar]
- 89.Li Y, Xu C, Zhang Q, Liu JY, Tan RX. In vitro anti-Helicobacter pylori action of 30 Chinese herbal medicines used to treat ulcer diseases. J Ethnopharmacol. 2005;98:329–333. doi: 10.1016/j.jep.2005.01.020. [DOI] [PubMed] [Google Scholar]
- 90.Sung BK, Kim MK, Lee WH, Lee DH, Lee HS. Growth responses of Cassia obtusifolia toward human intestinal bacteria. Fitoterapia. 2004;75:505–509. doi: 10.1016/j.fitote.2004.03.012. [DOI] [PubMed] [Google Scholar]
- 91.Li XH, Gong CR, Cao H, et al. Primary study on inhabiting of the extracts from Cassia obtusifolia seeds against Fusarium oxysporum and Botrytis cinerea. J Shanxi Agric Univ. 2006;26:348–350. [Google Scholar]
- 92.Yen GC, Chung DY. Antioxidant Effects of Extracts from Cassia tora L. Prepared under different degrees of roasting on the oxidative damage to biomolecules. J Agr Food Chem. 1999;47:1326–1332. doi: 10.1021/jf9810618. [DOI] [PubMed] [Google Scholar]
- 93.Yen GC, Chuang DY. Antioxidant properties of water extracts from Cassia tora L. In relation to the degree of roasting. J Agr Food Chem. 2000;48:2760–2765. doi: 10.1021/jf991010q. [DOI] [PubMed] [Google Scholar]
- 94.Xv JG, Hu QP. Study on Free Radical Scavenging Capacity by Cassia Seed Water Extract in vitro. Food Sci. 2006;27:73–75. [Google Scholar]
- 95.Guo CQ, Yan J, Wu XY, Xu GY, Fan CH, Gou XJ. Study on purification and antioxidation of water-soluble polysaccharide isolated from semen cassia. Food Sci. 2007;28:205–208. [Google Scholar]
- 96.Liu J, Deng ZY, Yu HH. Antioxidation Study on Water-soluble Polysaccharide Isolated from Semen Cassiae. Food Sci. 2006;27:61–63. [Google Scholar]
- 97.Liu C, Liu Q, Sun J, Jiang Bb, Yan J. Extraction of water-soluble polysaccharide and the antioxidant activity from Cassiae Semen. J Food Drug Anal. 2014;22:492–499. doi: 10.1016/j.jfda.2014.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Zeng H, Liu Q, Wang M, Jiang S, Zhang L, He X, Wang J, Chen X. Target-guided separation of antioxidants from Semen cassia via off-line two-dimensional high-speed counter-current chromatography combined with complexation and extrusion elution mode. J Chromatogr B. 2015;1001:58–65. doi: 10.1016/j.jchromb.2015.07.046. [DOI] [PubMed] [Google Scholar]
- 99.Kim SY, Kim JH, Kim SK, Oh MJ, Jung MY. Antioxidant activities of selected oriental herb extracts. J Am Oil Chem Soc. 1994;71:633–640. doi: 10.1007/BF02540592. [DOI] [Google Scholar]
- 100.Choi JS, Lee HJ, Kang SS. Alatemin, cassiaside and rubrofusarin gentiobioside, radical scavenging principles from the seeds of Cassia tora on 1, 1-diphenyl-2-picrylhydrazyl (DPPH) radical. Arch Pharm Res. 1994;17:462–466. doi: 10.1007/BF02979126. [DOI] [PubMed] [Google Scholar]
- 101.Koo A, Wang JC, Li KM. Extraction of hypotensive principles from seeds of Cassia tora. Am J Chin Med (Gard City N Y) 1976;4:245–248. doi: 10.1142/S0192415X76000330. [DOI] [PubMed] [Google Scholar]
- 102.Koo A, Chan WS, Li KM. A possible reflex mechanism of hypotensive action of extract from Cassia tora seeds. Am J Chin Med (Gard City N Y) 1976;4:249–255. doi: 10.1142/S0192415X76000342. [DOI] [PubMed] [Google Scholar]
- 103.Chan SH, Koo A, Li KM. The involvement of medullary reticular formation in the hypotensive effect of extracts from seeds of Cassia tora. Am J Chin Med (Gard City N Y) 1976;4:383–389. doi: 10.1142/S0192415X76000482. [DOI] [PubMed] [Google Scholar]
- 104.Mao WH, Shang QH, Liu AD, et al. Effects of Cassiae Semen Extracts on Vasorelaxation and Its Mechanisms in Rat Aorta. Chin J Hypertens. 2010;18:60–63. [Google Scholar]
- 105.Zhang CZ, Wang SX, Zhang Y, Chen JP, Liang XM. In vitro estrogenic activities of Chinese medicinal plants traditionally used for the management of menopausal symptoms. J Ethnopharmacol. 2005;98:295–300. doi: 10.1016/j.jep.2005.01.033. [DOI] [PubMed] [Google Scholar]
- 106.Wu CH, Yen GC. Antigenotoxic properties of Cassia tea (Cassia tora L.): Mechanism of action and the influence of roasting process. Life Sci. 2004;76:85–101. doi: 10.1016/j.lfs.2004.07.011. [DOI] [PubMed] [Google Scholar]
- 107.Fu F, Tian F, Zhou H, Lv W, Tie R, Ji L, Li R, Shi Z, Yu L, Liang X, et al. Semen cassiae attenuates myocardial ischemia and reperfusion injury in high-fat diet streptozotocin-induced type 2 diabetic rats. Am J Chinese Med. 2014;42:95–108. doi: 10.1142/S0192415X14500062. [DOI] [PubMed] [Google Scholar]
- 108.Zhang P, Chen JW. HPLC Fingerprint of Cassiae Semen. Chinese Tradit Herb Drugs. 2007;38:372–375. [Google Scholar]
- 109.Luo W, Liu B, Wang W, et al. HPLC fingerprint chromatogram of Cassiae Semen. J Beijing Univ Tradit Chin Med. 2009;32:115–117. [Google Scholar]
- 110.Wang WY, Zhao Q, Zhang TJ, et al. HPLC Fingerprint and chemical pattern recognition of Cassiae Semen. Chinese Tradit Herb Drugs. 2009;40:1638–1641. [Google Scholar]
- 111.Tang YL, Liang TZ, Zhang HX, Xu JY. Study on fingerprint of Cassiae Semen, ultramicro powder and powder particle. Zhong Yao Cai. 2011;34:1861–1866. (In Chinese) [PubMed] [Google Scholar]
- 112.Wang N, Wu Y, Wu X, Liang S, Sun H. A novel nonaqueous capillary electrophoresis method for effective separation and simultaneous determination of aurantio-obtusin, emodin and rhein in semen cassiae and cassia seed tea. Anal Methods. 2014;6:5133–5139. doi: 10.1039/C4AY00588K. [DOI] [Google Scholar]
- 113.Yang C, Wang S, Guo X, Sun J, Liu L, Wu L. Simultaneous determination of seven anthraquinones in rat plasma by Ultra High Performance Liquid Chromatography-tandem Mass Spectrometry and pharmacokinetic study after oral administration of Semen Cassiae extract. J Ethnopharmacol. 2015;169:305–313. doi: 10.1016/j.jep.2015.03.024. [DOI] [PubMed] [Google Scholar]
- 114.Hyun SK, Lee H, Kang SS, Chung HY, Choi JS. Inhibitory activities of Cassia tora and its anthraquinone constituents on angiotensin-converting enzyme. Phytother Res. 2009;23:178–184. doi: 10.1002/ptr.2579. [DOI] [PubMed] [Google Scholar]
- 115.Dhanasekaran M, Ignacimuthu S, Agastian P. Potential hepatoprotective activity of ononitol monohydrate isolated from Cassia tora L. on carbon tetrachloride induced hepatotoxicity in Wistar rats. Phytomedicine. 2009;16:891–895. doi: 10.1016/j.phymed.2009.02.006. [DOI] [PubMed] [Google Scholar]
- 116.Jung HA, Ali MY, Jung HJ, Jeong HO, Chung HY, Choi JS. Inhibitory activities of major anthraquinones and other constituents from Cassia obtusifolia against β-secretase and cholinesterases. J Ethnopharmacol. 2016;191:152–160. doi: 10.1016/j.jep.2016.06.037. [DOI] [PubMed] [Google Scholar]
- 117.Wen CC, Shyur LF, Jan JT, Liang PH, Kuo CJ, Arulselvan P, Wu JB, Kuo SC, Yang NS. Traditional Chinese medicine herbal extracts of Cibotium barometz, Gentiana scabra, Dioscorea batatas, Cassia tora and Taxillus chinensis inhibit SARS-CoV replication. J Tradit Complement Med. 2011;1:41–50. doi: 10.1016/S2225-4110(16)30055-4. [DOI] [PMC free article] [PubMed] [Google Scholar]