Abstract
Background
Despite men being less prone to develop autoimmune diseases, male sex has been associated with a more severe disease course in several systemic autoimmune diseases. In the present study, we aimed to investigate differences in the clinical presentation of primary Sjögren’s syndrome (pSS) between the sexes and establish whether male sex is associated with a more severe form of long-term pSS.
Methods
Our study population included 967 patients with pSS (899 females and 68 males) from Scandinavian clinical centers. The mean follow-up time (years) was 8.8 ± 7.6 for women and 8.5 ± 6.2 for men (ns). Clinical data including serological and hematological parameters and glandular and extraglandular manifestations were compared between men and women.
Results
Male patient serology was characterized by more frequent positivity for anti-Ro/SSA and anti-La/SSB (p = 0.02), and ANA (p = 0.02). Further, men with pSS were more frequently diagnosed with interstitial lung disease (p = 0.008), lymphadenopathy (p = 0.04) and lymphoma (p = 0.007). Conversely, concomitant hypothyroidism was more common among female patients (p = 0.009).
Conclusions
We observe enhanced serological responses and higher frequencies of lymphoma-related extraglandular manifestations in men with pSS. Notably, lymphoma itself was also significantly more common in men. These observations may reflect an aggravated immune activation and a more severe pathophysiological state in male patients with pSS and indicate a personalized managing of the disease due to the influence of the sex of patients with pSS.
Keywords: Sjögren’s syndrome, Autoimmunity, Sex difference, Disease severity, Extraglandular manifestations
Background
It has been widely established that women are more prone to develop autoimmune diseases [1]. Primary Sjögren’s syndrome (pSS) is a systemic autoimmune disease characterized by inflammation of the salivary and lacrimal glands, causing a reduction in exocrine secretion that ultimately leads to the clinical presentation of sicca symptoms. The reported population-based female to male ratio is 14:1 [2]. Many hypotheses have been proposed to explain the overall marked sex bias in autoimmunity and pSS [3], including genetic and epigenetic factors [4, 5], sex hormones [6], and X-chromosome aberrances [7, 8]. However, the molecular mechanisms that drive this sex skewing still remain elusive.
Interestingly, the differences between the sexes likewise extend to the clinical manifestations, where female and male patients differ in disease presentation and severity. Despite being less prone to develop autoimmune diseases, male patients have been reported to have a more severe disease and a worse prognosis. In systemic lupus erythematosus (SLE), men present more renal disease [9] and serositis [10]. Further, Andrade et al. [11] identified male sex as a strong predictor for poorer long-term prognosis due to accelerated damage accrual, while Manger et al. [12] reported male sex as a risk factor for increased SLE mortality. Male sex is also deemed as a factor for accelerated disease progression in multiple sclerosis (MS) [13] and associated with a significantly higher prevalence of comorbidities [14] such as diabetes, epilepsy, depression, and anxiety. Although less clear, sex differences in rheumatoid arthritis (RA) severity and extra-articular manifestations have also been described; women appear more prone to present sicca symptoms and men to have erosive joint disease, rheumatoid nodules, and interstitial lung disease [15, 16].
Regarding pSS, several studies have addressed sex differences in clinical presentation [17, 18]. As reviewed by Brandt et al. [3], some authors have identified differences in extraglandular manifestations and serological markers, with a focus on female prevalence. However, there is no clear consensus on whether male sex is associated with a more severe disease. Our group has described that at diagnosis, male patients more frequently present with extraglandular manifestations, more concomitant extraglandular manifestations, and higher anti-Ro52 levels by investigation in two independent cohorts [19]. To understand if there are sex differences in the clinical presentation of pSS also after a long-standing disease and whether risk for comorbidities vary between the sexes, we assessed glandular and extraglandular manifestations, serological parameters, and comorbidities of pSS in men and women years after diagnosis in a large Scandinavian cohort.
Methods
Patients
DISSECT—“Dissecting disease mechanisms in three systemic inflammatory autoimmune diseases with an interferon signature”—is a multicenter consortium comprising the Scandinavian Sjögren’s syndrome research network, the Swedish SLE network and the Swedish Myositis network linked to the European Myositis network. All 967 patients with pSS and fulfilling the American–European consensus criteria [20] in the DISSECT cohort were included in this study. Out of these, 899 were females and 68 were males (Table 1). The patients were diagnosed and followed at the Departments of Rheumatology at the University Hospitals in Gothenburg, Skåne, Linköping, Örebro, and Uppsala, as well as the Karolinska University Hospital in Stockholm, Sweden, and the Department of Rheumatology at Haukeland University Hospital, Bergen, and the University Hospital in Stavanger, Norway. Of the 205 patients from the Karolinska University Hospital, 127 were included in a previous analysis of clinical manifestations in female and male patients at diagnosis [19]. Clinical data with regard to autoantibody status and clinical manifestations were retrieved from the patient’s medical records. This included information on sicca symptom onset, age at diagnosis, histopathological examination of minor labial salivary gland biopsies, and serological analysis of ANA, Ro/SSA, and La/SSB autoantibodies. ANA was determined by indirect immunoflourescence of Hep2 cells for the vast majority of patients, while methods for determining Ro/SSA and La/SSB autoantibodies at the respective accredited Clinical Immunology department varied over time and between the centers and included indirect immunofluorescence of transfected cells, immunoblotting, ELISA, and multiplex technologies. Information on extraglandular manifestations according to doctors’ clinical assessments included articular, pulmonary, renal, cutaneous, muscular, endocrine, and lymphoid systems. The study was approved by the local ethical committee for respective study center, and patients gave informed written consent.
Table 1.
Demographic and basic characteristics of the cohort
| Women n = 899% (frequency) | Men n = 68% (frequency) | p value | |
|---|---|---|---|
| Basic characteristics | |||
| Sex | 93% (899/967) | 7% (68/967) | <0.0001 |
| Age at symptom onset (years, mean ± SD) | 46.16 ± 14.79 | 47.88 ± 14.71 | 0.50 |
| Age at diagnosis (years, mean ± SD) | 52.65 ± 13.75 | 52.63 ± 13.52 | 0.96 |
| Follow-up time from diagnosis (years, mean ± SD) | 8.76 ± 7.62 | 8.48 ± 6.15 | 0.68 |
| Item IV. Histopathologya | |||
| Salivary gland biopsy | |||
| Performed | 87% (760/871) | 79% (52/66) | 0.06 |
| Positive (focus score ≥1) | 90% (616/685) | 86% (42/49) | 0.35 |
| Germinal centers | 21% (75/351) | 33% (8/24) | 0.20 |
Bold values indicate statistically significant findings (p < 0.05)
aAccording to the 2002 Revised American-European Consensus Group criteria for Sjögren’s syndrome
Statistical analysis
For the comparison of continuous variables, the Mann–Whitney U test was used. The chi-square test was used when analyzing categorical data, and Fisher’s exact test was employed if the observed frequency of any given cell was <5 and/or the total number of analyzed individuals in any group was <40. Data was analyzed with GraphPad Prism 6, and p values <0.05 were considered statistically significant.
Results
Basic characteristics of the cohort
The cohort consisted of 967 pSS patients, of which 899 were women (93%) and 68 were men (7%) (Table 1). The female/male ratio was 13:1. The mean age at symptom onset for the female group was 46 years ± 14.8 (95% CI) and 48 years ± 14.7 (95% CI) for the male group. There was no significant difference with regard to the age at diagnosis between women and men (52.6 and 52.6 years, respectively), or the follow-up time from diagnosis between female and male patients (8.8 years ± 7.6, 95% CI and 8.5 years ± 6.1, 95% CI, respectively).
We also compared the histopathological parameters of the salivary gland biopsy from all the included patients (Table 1). Although women tended to more frequently undergo a salivary gland biopsy than men (p = 0.06), the histological findings revealed no significant differences in terms of a positive focus score or presence of germinal center-like structures.
Serological differences between female and male patients with pSS
Autoantibody profiles were also analyzed in a sex-specific manner (Table 2). Autoantibody positivity was defined as presenting both or either of anti-Ro/SSA or anti-La/SSB. Accordingly, 72% of the patients from this cohort were autoantibody positive; 71% of the female and 81% of the male patients had either Ro/SSA and/or La/SSB antibodies.
Table 2.
Serological characteristics in female and male patients with pSS
| Women n = 899% (frequency) | Men n = 68% (frequency) | p value | |
|---|---|---|---|
| Item VI.a Autoantibodies | |||
| Ro/SSA and/or La/SSB positive | 71% (640/899) | 81% (54/67) | 0.1 |
| Ro/SSA positive | 68% (612/897) | 76% (51/67) | 0.18 |
| La/SSB positive | 41% (367/894) | 57% (38/67) | 0.01 |
| Ro/SSA and La/SSB positive | 38% (342/892) | 52% (35/67) | 0.02 |
| ANA positive | 74% (660/895) | 87% (58/67) | 0.02 |
Italicized values indicate statistically significant findings (p < 0.05)
aAccording to the 2002 Revised American-European Consensus Group criteria for Sjögren’s syndrome
Anti-Ro/SSA positivity was observed in 68% of the women and 76% of the men; SSB autoantibodies were detected in 41% of the women and 57% of the men (p = 0.01) and positivity for both anti-Ro/SSA and anti-La/SSB was found in 38% of the women and 52% of the men (p = 0.02) (Table 2). Furthermore, ANA positivity was significantly more frequent in male patients (p = 0.02). Thus, the stratified analysis indicated that the male group presents significantly higher frequencies of positivity towards ANA, La/SSB, and Ro/SSA + La/SSB.
Sex hormones have been suggested to influence the immune system, especially in terms of antibody production [21]. To evaluate whether the number or percentage of autoantibody positive individuals diagnosed was related to menopause, we further stratified the female and male patients with and without autoantibodies based on age at diagnosis (Fig. 1). We observed an increasing number of autoantibody-positive women being diagnosed up to 60 years of age, and that at the same time, a steadily increasing number of autoantibody negative women receiving the diagnosis (Fig. 1a). The male group displayed a comparable pattern (Fig. 1b). Consistently, also when analyzed as percent autoantibody positive (Fig. 1c), the trend was similar in both the female and male groups. Already in the late thirties/early forties the percentage of autoantibody-positive patients diagnosed with pSS started to decline and did so steadily until the mid-seventies. Very few males were diagnosed after the age of 75 (n = 2), making the last point of the curve less relevant to consider. Altogether, the data show a consistent higher percentage of autoantibody-positive men. Neither the female nor male group did show any obvious change specific for age 50 or 55, which is commonly used as a proxy for menopause.
Fig. 1.
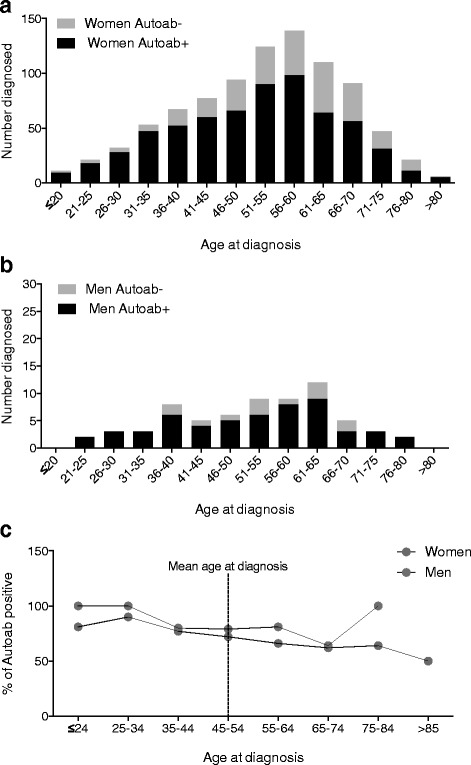
Ro/SSA- and/or La/SSB-positive and negative women and men diagnosed with Sjögren’s syndrome at different age intervals. a Number of Ro/SSA- and/or La/SSB-positive and negative women diagnosed with Sjögren’s syndrome. b Number of Ro/SSA and/or La/SSB positive and negative men diagnosed with Sjögren’s syndrome. c Percentage of Ro/SSA and/or La/SSB positive women and men diagnosed with Sjögren’s syndrome
Frequencies of clinical manifestations differ between female and male pSS patients
The presence and type of extraglandular manifestations were obtained in order to evaluate differences in frequencies among the sexes. There were significant sex differences in the frequencies of several extraglandular manifestations (Table 3). Interstitial lung disease was more frequent in male patients (p = 0.008), as well as lymphadenopathy (p = 0.04) and, notably, lymphoma (p = 0.007). There was also a tendency for men to present more often with major salivary glands swelling (p = 0.11), as well as myositis (p = 0.14), while hypothyroidism was more common in female patients, present in 24% of the women as opposed to 8% of the male patients (p = 0.009).
Table 3.
Frequency of pSS-associated clinical manifestations and comorbidities in female and male patients with pSS
| Women n = 899% (frequency) | Men n = 68% (frequency) | p value | |
|---|---|---|---|
| Classificationa | |||
| Articular | |||
| Arthritis | 20% (169/859) | 14% (9/65) | 0.25 |
| Pulmonary | |||
| Interstitial lung disease | 6% (40/619) | 17% (8/48) | 0.008 |
| Renal | |||
| Interstitial nephritis | 3% (19/607) | 2% (1/48) | 1.00 |
| Cutaneous | |||
| Dermal vasculitis | 11% (91/836) | 8% (5/62) | 0.49 |
| Lymphadenopathy and lymphoma | |||
| Enlarged lymph nodes | 8% (69/825) | 16% (10/62) | 0.04 |
| Lymphoma | 4% (32/889) | 10% (7/68) | 0.007 |
| Muscular | |||
| Myositis | 0.9% (7/772) | 3% (2/61) | 0.14 |
| Glandular | |||
| Major salivary gland swelling | 29% (225/767) | 40% (21/53) | 0.11 |
| Presence of EGM | 34% (304/899) | 41% (28/68) | 0.22 |
| Number of EGM (mean + SD) |
0.44 ± 0.69 | 0.51 ± 0.74 | 0.30 |
| Other common comorbidities and clinical manifestations | |||
| Hypothyroidism | 24% (175/739) | 8% (4/51) | 0.009 |
| Raynaud’s phenomenon | 29% (247/851) | 30% (20/66) | 0.83 |
Italicized values indicate statistically significant findings (p < 0.05)
aAvailable data on the extraglandular manifestations evaluated to estimate the EULAR Sjögren’s Syndrome Disease Activity Index (ESSDAI) are included
EGM extraglandular manifestations
Considering the observation that men with pSS present an increased risk for lymphoma when compared to women (Table 3), we analyzed the difference between histopathologically verified subtypes of lymphoma. Mucosa-associated lymphoid tissue (MALT) lymphoma and diffuse large B cell lymphoma (DLBCL) were the most common subtypes, but no significant difference between the occurrence between women and men was found (Table 4).
Table 4.
Frequencies of subtypes of lymphoma in women and men with pSS
| Women n = 32% (frequency) |
Men n = 7% (frequency) |
p valuea | |
|---|---|---|---|
| MALT lymphoma | 56% (18/32) | 57% (4/7) | 1.00 |
| DLBCL | 16% (5/32) | 0% (0/7) | 0.56 |
| Follicular lymphoma | 6% (2/32) | 14% (1/7) | – |
| Myeloma | 3% (1/32) | 0% (0/7) | – |
| CLL | 3% (1/32) | 0% (0/7) | – |
| Lymphoplasmacytic lymphoma | 0% (0/32) | 14% (1/7) | – |
| Other NHL | 13% (4/32) | 14% (1/7) | – |
| Hodgkin lymphoma | 3% (1/32) | 0% (0/7) | – |
According to the WHO 2016 classification
aCalculated when n ≥ 5 for either group
MALT mucosa-associated lymphoid tissue, DLBCL diffuse large B cell lymphoma, CLL Chronic lymphatic leukemia, NHL non-Hodgkin lymphoma
Autoantibody-positive pSS female and male patients differ in terms of clinical manifestations
We further assessed whether presentation between autoantibody-positive female and male patients differed from the unstratified analysis described above, focusing on the extraglandular manifestations and other clinical manifestations that had significantly differed between female and male patients regardless of serology-status. Interstitial lung disease and lymphoma were significantly more frequent in men (p = 0.01 and p = 0.03, respectively) also when including only seropositive cases in the analysis, and hypothyroidism was more common in women (p = 0.03) (Table 5). Lymphadenopathy had a higher observed frequency among male patients (Table 3), which was however not statistically significant when considering only the autoantibody positive cases.
Table 5.
Frequency of comorbidities, extraglandular, and other common clinical manifestations in SSA- and/or SSB-positive female and male patients with pSS
| Women n = 640% (frequency) | Men n = 54% (frequency) | p value | |
|---|---|---|---|
| Interstitial lung disease | 7% (31/444) | 19% (7/37) | 0.01 |
| Enlarged lymph nodes | 10% (57/584) | 16% (8/49) | 0.15 |
| Hypothyroidism | 23% (117/517) | 8% (3/40) | 0.03 |
| Lymphoma | 4% (28/634) | 11% (6/54) | 0.03 |
Italicized values indicate statistically significant findings (p < 0.05)
Discussion
Primary Sjögren’s syndrome represents the autoimmune disease with the highest female bias, ranging from a ratio of 10–20:1 [22]. Besides the overwhelming sex bias observed in disease susceptibility, previous studies have also aimed to investigate whether the disease manifests differently between female and male patients. Earlier observations have not reached a clear consensus as to whether male pSS patients have a distinct clinical course and a more severe presentation of the disease [3]. In a recent study, though, we reported that men with pSS, from a population-based incident case cohort, displayed significantly higher levels of anti-Ro52, were diagnosed at an earlier age than their female counterparts, presented more concomitant extraglandular manifestations, and had a higher frequency of pulmonary complications and cutaneous vasculitis. Similarly, an Italian cohort revealed a significant male propensity towards extraglandular manifestation presentation [19]. These findings strongly suggest that men affected by pSS have a more severe disease at time of diagnosis. In the present study, we addressed whether differences between the sexes are also present several years after diagnosis.
We identified significant sex differences in terms of serological parameters and frequencies of some organ involvement. Our results indicate that the humoral response between women and men is different; particularly, men present more often with La/SSB, Ro/SSA + La/SSB and ANA positivity. This increased immune activity observed among the male patients is of special interest since in a healthy state, men mount a lower antibody response in comparison with women [23–25]. Although the pathogenic effect of autoantibodies has not been clearly established, the presence of certain autoantibodies has been associated with organ manifestations. Noteworthy, SSA antibodies are related to pulmonary diseases [26, 27], an extraglandular manifestation we observed overrepresented in the male patients from our cohort. Further, a recent study proposed that anti-La/SSB antibodies are a risk factor associated with increased mortality in pSS patients [28]. Thus, even though the pathogenic role of pSS-associated autoantibodies remains unknown, seropositivity has a strong correlation with organ involvement and worse prognosis, supporting the conclusion that the disease course is more severe in male patients than in female patients.
Interstitial lung disease has been extensively studied in the context of pSS [29, 30]. Male sex is widely recognized as a risk factor for developing interstitial lung disease [26, 31]. In accordance with more recent studies [19], our extended cohort shows that male patients with pSS are indeed more prone to develop interstitial lung disease. The reasons for this male preponderance are poorly understood; however, this might be due to increased seropositivity, environmental exposure to certain pollutants [32] and smoking [31] in the male pSS group. In fact, idiopathic pulmonary fibrosis, which represents a usual interstitial pneumonia histopathological pattern, is one of the most common forms of interstitial lung disease detected in pSS [29] and has a higher prevalence in men, as epidemiological studies have previously described [33, 34]. In other words, regardless of pSS diagnosis, men in general are more frequently affected by a type of interstitial lung disease that is associated with a worse prognosis. This susceptibility, thus, might be augmented in pSS, driven by other pathophysiological factors that enhance this propensity to develop pulmonary disease.
It is well known that pSS patients have an increased risk for developing non-Hodgkin lymphoma [35–38]. Sex-specific risk for lymphoma development in patients with rheumatic disease has been seldom studied, mainly due to the inclusion of mostly female patients. Nevertheless, Ansell et al. have reported a significantly higher incidence of lymphoma in male RA patients. Despite the increased association of autoimmune diseases and lymphoma in men [39], earlier studies of sex differences in lymphoproliferative malignancies in pSS have not shown a clear sex-specific predominance [40, 41]. In contrast, our present study is the largest pSS cohort to report a significantly increased risk for male patients to present lymphoma in comparison with female patients. This is in accordance with the results from a smaller patient sample from which an increased risk for men affected with pSS, SLE, RA, and autoimmune hemolytic anemia to develop lymphoma was reported [42].
Since the male bias observed in pSS-associated malignancies has only recently been described, it is not fully understood. However, considering the reported predictive factors of lymphoma development, an increased risk of lymphoma in male patients is logical. As reviewed by Nocturne and Mariette [43], the main clinical manifestations and parameters associated with this type of cancer are swelling of salivary glands, lymphadenopathy, palpable purpura, cryoglobulinemia, lymphopenia, low complement levels, and a monoclonal component in serum or urine. Interestingly, the male pSS patients from our cohort presented more frequently with lymphadenopathy. Although not included in the data analysis due to the high amount of missing data, cryoglobulinemia was also more commonly observed in the male patients (20/151 in females vs 3/7 in males, p = 0.03).
The only clinical manifestation that was more significantly represented in the female pSS patients from our cohort was hypothyroidism. Endocrine problems are not uncommon in patients with autoimmune disorders [44], and the female bias towards thyroid diseases has been extensively documented [45–48]. Furthermore, since the predominantly female incidence of hypothyroidism corresponds with the female susceptibility to autoimmune diseases, the thyroid gland has been proposed as a decisive organ to explain the sex skewness in autoimmune diseases. The effect of adipokines, which comprise a number of different cytokines including, e.g., leptins, adiponectins, TNF-α, and IL-6, on thyroid tissue has been suggested as a triggering mechanism for autoimmune thyroiditis, which probably precedes or coincides with the diagnosis of another systemic autoimmune disease such as pSS and SLE. [44]. This emphasizes the need for a comprehensive screening and close monitoring of thyroid function in suspected patients because it might be an important marker for autoimmune disease development. As for the thyroid gland being responsible for female-preponderant diseases, further studies should be performed to clarify its role.
The studied cohort offers a valuable large group of clinically carefully characterized patients with pSS, allowing for analysis of parameters that differ between men and women affected by the syndrome. The long follow-up time is essential for identifying clinical manifestations at different time points of the disease course. However, the patients included in this cohort were mostly included at tertiary referral centers of university hospital clinics. A possible limitation of the study is that the study population might therefore not mirror the general pSS patient population and that the patients described in this study represent cases with an overall more severe disease phenotype, both female and male patients. A further possibility is that male patients with mild symptoms and less severe disease are less often referred, as the primary health care doctor may not be as likely to suspect Sjögren’s syndrome due to its rarity in men, resulting in only men with more severe disease being included in the study. As the evaluation of extraglandular manifestations was dependent on doctors’ clinical assessments and did not include specific laboratory or physiologic tests unless the patient had symptoms, it is also possible that subclinical extraglandular manifestations may have been missed. However, the mean number of extraglandular manifestations diagnosed did not differ significantly between centers, nor did the proportion of men and women contributed. A further limitation is the lack of EULAR primary Sjögren’s syndrome disease activity (ESSDAI) and patient-reported (ESSPRI) indexes [49] at diagnosis, as well as information on other common extraglandular manifestations such as neurological diseases.
Conclusions
In summary, our findings provide compelling evidence that the clinical presentation of pSS differs between women and men. The sex-specific preference of some clinical manifestations hints at divergent pathophysiological mechanisms between women and men with pSS. Consequently, management of the disease will benefit from sex-specific tailored clinical programs to address complications that are common or expected in the respective sex.
Acknowledgements
DISSECT consortium members:
Agneta Zickert, Stockholm; Albin Björk, Stockholm; Anders A. Bengtsson, Lund; Andreas Jönsen, Lund; Andrei Alexsson, Uppsala; Anna Tjärnlund, Stockholm; Ann-Christine Syvänen, Uppsala; Antonella Notarnicola, Stockholm; Argyri Mathioudaki, Uppsala; Åsa Karlsson, Uppsala; Carin Backlin, Uppsala; Christine Bengtsson, Umeå; Christopher Sjöwall, Linköping; Dag Leonard, Uppsala; Daniel Hammenfors, Bergen; Elisabet Svenungsson, Stockholm; Elke Theander, Malmö; Eva Baecklund, Uppsala; Eva Murén, Uppsala; Fabiana Farias, Uppsala; Gerli Pielberg, Uppsala; Guðný Ella Thorlacius, Stockholm; Gunnel Nordmark, Uppsala; Hector Chinoy, Manchester; Helena Andersson, Oslo; Helena Enocsson, Linköping; Helena Forsblad-d’Elia, Gothenburg; Ingrid E. Lundberg, Stockholm; Iva Gunnarsson, Stockholm; Janine Lamb, Manchester; Jennifer Meadows, Uppsala; Jessika Nordin, Uppsala; Johan G Brun, Bergen; Johanna Dahlqvist, Uppsala; Johanna K. Sandling, Uppsala; John Mo, Gothenburg; Jonas Carlsson Almlöf, Uppsala; Jonas Wetterö, Linköping; Jorge I. Ramírez Sepúlveda, Stockholm; Juliana Imgenberg-Kreuz, Uppsala; Karin Bolin, Uppsala; Karin Hjorton, Uppsala; Karl A. Brokstad, Bergen; Karolina Tandre, Uppsala; Kathrine Skarstein, Bergen; Katrine Brække Norheim, Stavanger; Kerstin Lindblad-Toh, Uppsala; Lars Rönnblom, Uppsala; Leonid Padyukov, Stockholm; Lilian Vasaitis, Uppsala; Lina Hultin-Rosenberg, Uppsala; Louise Pyndt Diederichsen, Odense; Maija-Leena Eloranta, Uppsala; Malin V. Jonsson, Bergen; Marie Wahren-Herlenius, Stockholm; Marika Kvarnström, Stockholm; Maryam Dastmalchi, Stockholm; Matteo Bianchi, Uppsala; Niklas Hagberg, Uppsala; Outi Vaarala, Gothenburg; Øyvind Molberg, Oslo; Per Eriksson, Linköping; Roald Omdal, Stavanger; Robert G. Cooper, Liverpool; Roland Jonsson, Bergen; Sara Magnusson Bucher, Örebro; Sergey Kozyrev, Uppsala; Silke Appel, Bergen; Simon Rothwell, Manchester; Solbritt Rantapää-Dahlqvist, Umeå; Svein Joar Johnsen, Stavanger; Thomas Mandl, Malmö.
Funding
The study was supported by grants from the Swedish Research Council, the Heart-Lung Foundation, the Stockholm County Council, Karolinska Institutet, the Swedish Rheumatism association, and the King Gustaf the Vth 80-year foundation. The DISSECT study is supported by an AstraZeneca Science for Life Laboratory Research Collaboration grant.
Availability of data and materials
Not applicable.
Authors’ contributions
JIRS and MWH conceived and designed the study. DISSECT consortium: MK, PE, KBN, SJJ, DH, MVJ, KS, HFdE, EB, SMB, ET, TM, RO, RJ, and GN managed the study participant recruitment and clinical data acquisition. Data was analyzed by JIRS and GN. JIRS and MWH wrote the first draft of the manuscript, and all authors participated in the revision until its final stage. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The study was approved by the regional ethical committee for respective study center, and patients gave informed written consent.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Jorge I. Ramírez Sepúlveda, Email: jorge.ramirez@ki.se
Marika Kvarnström, Email: marika.kvarnstrom@karolinska.se.
Per Eriksson, Email: per.eriksson@regionostergotland.se.
Thomas Mandl, Email: thomas.mandl@med.lu.se.
Katrine Brække Norheim, Email: katnorheim@gmail.com.
Svein Joar Johnsen, Email: svein.joar.auglend.johnsen@sus.no.
Daniel Hammenfors, Email: daniel.hammenfors@gmail.com.
Malin V. Jonsson, Email: Malin.Jonsson@uib.no
Kathrine Skarstein, Email: Kathrine.Skarstein@uib.no.
Johan G. Brun, Email: Johan.Brun@uib.no
Lars Rönnblom, Email: lars.ronnblom@medsci.uu.se.
Helena Forsblad-d’Elia, Email: helena.forsblad.delia@umu.se.
Sara Magnusson Bucher, Email: sara.magnusson-bucher@regionorebrolan.se.
Eva Baecklund, Email: eva.baecklund@medsci.uu.se.
Elke Theander, Email: Elke.Theander@med.lu.se.
Roald Omdal, Email: roald.omdal@lyse.net.
Roland Jonsson, Email: roland.jonsson@uib.no.
Gunnel Nordmark, Email: gunnel.nordmark@medsci.uu.se.
Marie Wahren-Herlenius, Phone: +46-8-51773431, Email: Marie.Wahren@ki.se.
the DISSECT consortium:
Agneta Zickert, Albin Björk, Anders A. Bengtsson, Andreas Jönsen, Andrei Alexsson, Anna Tjärnlund, Ann-Christine Syvänen, Antonella Notarnicola, Argyri Mathioudaki, Åsa Karlsson, Carin Backlin, Christine Bengtsson, Christopher Sjöwall, Dag Leonard, Daniel Hammenfors, Elisabet Svenungsson, Elke Theander, Eva Baecklund, Eva Murén, Fabiana Farias, Gerli Pielberg, Guðný Ella Thorlacius, Gunnel Nordmark, Hector Chinoy, Helena Andersson, Helena Enocsson, Helena Forsblad-d’Elia, Ingrid E. Lundberg, Iva Gunnarsson, Janine Lamb, Jennifer Meadows, Jessika Nordin, Johan G. Brun, Johanna Dahlqvist, Johanna K. Sandling, John Mo, Jonas Carlsson Almlöf, Jonas Wetterö, Jorge I. Ramírez Sepúlveda, Juliana Imgenberg-Kreuz, Karin Bolin, Karin Hjorton, Karl A. Brokstad, Karolina Tandre, Kathrine Skarstein, Katrine Brække Norheim, Kerstin Lindblad-Toh, Lars Rönnblom, Leonid Padyukov, Lilian Vasaitis, Lina Hultin-Rosenberg, Louise Pyndt Diederichsen, Maija-Leena Eloranta, Malin V. Jonsson, Marie Wahren-Herlenius, Marika Kvarnström, Maryam Dastmalchi, Matteo Bianchi, Niklas Hagberg, Outi Vaarala, Øyvind Molberg, Per Eriksson, Roald Omdal, Robert G. Cooper, Roland Jonsson, Sara Magnusson Bucher, Sergey Kozyrev, Silke Appel, Simon Rothwell, Solbritt Rantapää-Dahlqvist, Svein Joar Johnsen, and Thomas Mandl
References
- 1.Whitacre CC. Sex differences in autoimmune disease. Nat Immunol. 2001;2:777–780. doi: 10.1038/ni0901-777. [DOI] [PubMed] [Google Scholar]
- 2.Kvarnstrom M, Ottosson V, Nordmark B, Wahren-Herlenius M. Incident cases of primary Sjogren's syndrome during a 5-year period in Stockholm County: a descriptive study of the patients and their characteristics. Scand J Rheumatol. 2015;44:135–142. doi: 10.3109/03009742.2014.931457. [DOI] [PubMed] [Google Scholar]
- 3.Brandt JE, Priori R, Valesini G, Fairweather D. Sex differences in Sjogren's syndrome: a comprehensive review of immune mechanisms. Biol Sex Differ. 2015;6:19. doi: 10.1186/s13293-015-0037-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lessard CJ, Li H, Adrianto I, et al. Variants at multiple loci implicated in both innate and adaptive immune responses are associated with Sjogren's syndrome. Nat Genet. 2013;45:1284–1292. doi: 10.1038/ng.2792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Imgenberg-Kreuz J, Sandling JK, Almlöf JC, Nordlund J, Signér L, Norheim KB, Omdal R, Rönnblom L, Eloranta ML, Syvänen AC, Nordmark G. Genome-wide DNA methylation analysis in multiple tissues in primary Sjögren's syndrome reveals regulatory effects at interferon-induced genes. Ann Rheum Dis. 2016;75(11):2029–36. doi:10.1136/annrheumdis-2015-208659. [DOI] [PMC free article] [PubMed]
- 6.Cutolo M, Capellino S, Sulli A, et al. Estrogens and autoimmune diseases. Ann N Y Acad Sci. 2006;1089:538–547. doi: 10.1196/annals.1386.043. [DOI] [PubMed] [Google Scholar]
- 7.Liu K, Kurien BT, Zimmerman SL, et al. X chromosome dose and sex bias in autoimmune diseases: increased prevalence of 47,XXX in systemic lupus erythematosus and Sjogren's syndrome. Arthritis Rheumatol. 2016;68:1290–1300. doi: 10.1002/art.39560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Harris VM, Sharma R, Cavett J, et al. Klinefelter's syndrome (47,XXY) is in excess among men with Sjogren's syndrome. Clin Immunol. 2016;168:25–29. doi: 10.1016/j.clim.2016.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Boodhoo KD, Liu S, Zuo X. Impact of sex disparities on the clinical manifestations in patients with systemic lupus erythematosus: a systematic review and meta-analysis. Medicine (Baltimore) 2016;95:e4272. doi: 10.1097/MD.0000000000004272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Voulgari PV, Katsimbri P, Alamanos Y, Drosos AA. Gender and age differences in systemic lupus erythematosus. A study of 489 Greek patients with a review of the literature. Lupus. 2002;11:722–729. doi: 10.1191/0961203302lu253oa. [DOI] [PubMed] [Google Scholar]
- 11.Andrade RM, Alarcon GS, Fernandez M, et al. Accelerated damage accrual among men with systemic lupus erythematosus: XLIV. Results from a multiethnic US cohort. Arthritis Rheum. 2007;56:622–630. doi: 10.1002/art.22375. [DOI] [PubMed] [Google Scholar]
- 12.Manger K, Manger B, Repp R, et al. Definition of risk factors for death, end stage renal disease, and thromboembolic events in a monocentric cohort of 338 patients with systemic lupus erythematosus. Ann Rheum Dis. 2002;61:1065–1070. doi: 10.1136/ard.61.12.1065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tremlett H, Paty D, Devonshire V. Disability progression in multiple sclerosis is slower than previously reported. Neurology. 2006;66:172–177. doi: 10.1212/01.wnl.0000194259.90286.fe. [DOI] [PubMed] [Google Scholar]
- 14.Marrie RA, Patten SB, Tremlett H, et al. Sex differences in comorbidity at diagnosis of multiple sclerosis: a population-based study. Neurology. 2016. [Epub ahead of print] [DOI] [PMC free article] [PubMed]
- 15.Weyand CM, Schmidt D, Wagner U, Goronzy JJ. The influence of sex on the phenotype of rheumatoid arthritis. Arthritis Rheum. 1998;41:817–822. doi: 10.1002/1529-0131(199805)41:5<817::AID-ART7>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 16.Gossec L, Baro-Riba J, Bozonnat MC, et al. Influence of sex on disease severity in patients with rheumatoid arthritis. J Rheumatol. 2005;32:1448–1451. [PubMed] [Google Scholar]
- 17.Gondran G, Fauchais A, Lambert M, et al. Primary Sjogren's syndrome in men. Scand J Rheumatol. 2008;37:300–305. doi: 10.1080/03009740802001426. [DOI] [PubMed] [Google Scholar]
- 18.Horvath IF, Szodoray P, Zeher M. Primary Sjogren's syndrome in men: clinical and immunological characteristic based on a large cohort of Hungarian patients. Clin Rheumatol. 2008;27:1479–1483. doi: 10.1007/s10067-008-0944-7. [DOI] [PubMed] [Google Scholar]
- 19.Ramirez Sepulveda JI, Kvarnstrom M, Brauner S, Baldini C, Wahren-Herlenius M. Difference in clinical presentation between women and men in incident primary Sjogren's syndrome. Biol Sex Differ. 2017;8:16. doi: 10.1186/s13293-017-0137-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Vitali C, Bombardieri S, Jonsson R, et al. Classification criteria for Sjogren's syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann Rheum Dis. 2002;61:554–558. doi: 10.1136/ard.61.6.554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sakiani S, Olsen NJ, Kovacs WJ. Gonadal steroids and humoral immunity. Nat Rev Endocrinol. 2013;9:56–62. doi: 10.1038/nrendo.2012.206. [DOI] [PubMed] [Google Scholar]
- 22.Garcia-Carrasco M, Mendoza-Pinto C, Jimenez-Hernandez C, et al. Serologic features of primary Sjogren's syndrome: clinical and prognostic correlation. Int J Clin Rheumtol. 2012;7:651–659. doi: 10.2217/ijr.12.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Klein SL, Jedlicka A, Pekosz A. The Xs and Y of immune responses to viral vaccines. Lancet Infect Dis. 2010;10:338–349. doi: 10.1016/S1473-3099(10)70049-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Furman D. Sexual dimorphism in immunity: improving our understanding of vaccine immune responses in men. Expert Rev Vaccines. 2015;14:461–471. doi: 10.1586/14760584.2015.966694. [DOI] [PubMed] [Google Scholar]
- 25.Klein SL, Marriott I, Fish EN. Sex-based differences in immune function and responses to vaccination. Trans R Soc Trop Med Hyg. 2015;109:9–15. doi: 10.1093/trstmh/tru167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Boitiaux JF, Debray MP, Nicaise-Roland P, et al. Idiopathic interstitial lung disease with anti-SSA antibody. Rheumatology (Oxford) 2011;50:2245–2250. doi: 10.1093/rheumatology/ker267. [DOI] [PubMed] [Google Scholar]
- 27.Ghillani P, Andre C, Toly C, et al. Clinical significance of anti-Ro52 (TRIM21) antibodies non-associated with anti-SSA 60kDa antibodies: results of a multicentric study. Autoimmun Rev. 2011;10:509–513. doi: 10.1016/j.autrev.2011.03.004. [DOI] [PubMed] [Google Scholar]
- 28.Singh AG, Singh S, Matteson EL. Rate, risk factors and causes of mortality in patients with Sjogren's syndrome: a systematic review and meta-analysis of cohort studies. Rheumatology (Oxford) 2016;55:450–460. doi: 10.1093/rheumatology/kew291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mira-Avendano IC, Abril A. Pulmonary manifestations of Sjogren syndrome, systemic lupus erythematosus, and mixed connective tissue disease. Rheum Dis Clin N Am. 2015;41:263–277. doi: 10.1016/j.rdc.2015.01.001. [DOI] [PubMed] [Google Scholar]
- 30.Flament T, Bigot A, Chaigne B, et al. Pulmonary manifestations of Sjogren's syndrome. Eur Respir Rev. 2016;25:110–123. doi: 10.1183/16000617.0011-2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yazisiz V, Arslan G, Ozbudak IH, et al. Lung involvement in patients with primary Sjogren's syndrome: what are the predictors? Rheumatol Int. 2010;30:1317–1324. doi: 10.1007/s00296-009-1152-8. [DOI] [PubMed] [Google Scholar]
- 32.Johannson KA, Balmes JR, Collard HR. Air pollution exposure: a novel environmental risk factor for interstitial lung disease? Chest. 2015;147:1161–1167. doi: 10.1378/chest.14-1299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ley B, Collard HR, King TE., Jr Clinical course and prediction of survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2011;183:431–440. doi: 10.1164/rccm.201006-0894CI. [DOI] [PubMed] [Google Scholar]
- 34.Hopkins RB, Burke N, Fell C, Dion G, Kolb M. Epidemiology and survival of idiopathic pulmonary fibrosis from national data in Canada. Eur Respir J. 2016;48:187–195. doi: 10.1183/13993003.01504-2015. [DOI] [PubMed] [Google Scholar]
- 35.Kassan SS, Thomas TL, Moutsopoulos HM, et al. Increased risk of lymphoma in sicca syndrome. Ann Intern Med. 1978;89:888–892. doi: 10.7326/0003-4819-89-6-888. [DOI] [PubMed] [Google Scholar]
- 36.Theander E, Henriksson G, Ljungberg O, et al. Lymphoma and other malignancies in primary Sjogren's syndrome: a cohort study on cancer incidence and lymphoma predictors. Ann Rheum Dis. 2006;65:796–803. doi: 10.1136/ard.2005.041186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dias C, Isenberg DA. Susceptibility of patients with rheumatic diseases to B-cell non-Hodgkin lymphoma. Nat Rev Rheumatol. 2011;7:360–368. doi: 10.1038/nrrheum.2011.62. [DOI] [PubMed] [Google Scholar]
- 38.Nishishinya MB, Pereda CA, Munoz-Fernandez S, et al. Identification of lymphoma predictors in patients with primary Sjogren's syndrome: a systematic literature review and meta-analysis. Rheumatol Int. 2015;35:17–26. doi: 10.1007/s00296-014-3051-x. [DOI] [PubMed] [Google Scholar]
- 39.Ansell P, Simpson J, Lightfoot T, et al. Non-Hodgkin lymphoma and autoimmunity: does gender matter? Int J Cancer. 2011;129:460–466. doi: 10.1002/ijc.25680. [DOI] [PubMed] [Google Scholar]
- 40.Mellemkjaer L, Pfeiffer RM, Engels EA, et al. Autoimmune disease in individuals and close family members and susceptibility to non-Hodgkin's lymphoma. Arthritis Rheum. 2008;58:657–666. doi: 10.1002/art.23267. [DOI] [PubMed] [Google Scholar]
- 41.Smedby KE, Askling J, Mariette X, Baecklund E. Autoimmune and inflammatory disorders and risk of malignant lymphomas--an update. J Intern Med. 2008;264:514–527. doi: 10.1111/j.1365-2796.2008.02029.x. [DOI] [PubMed] [Google Scholar]
- 42.Fallah M, Liu X, Ji J, et al. Hodgkin lymphoma after autoimmune diseases by age at diagnosis and histological subtype. Ann Oncol. 2014;25:1397–1404. doi: 10.1093/annonc/mdu144. [DOI] [PubMed] [Google Scholar]
- 43.Nocturne G, Mariette X. Sjogren syndrome-associated lymphomas: an update on pathogenesis and management. Br J Haematol. 2015;168:317–327. doi: 10.1111/bjh.13192. [DOI] [PubMed] [Google Scholar]
- 44.Merrill SJ, Mu Y. Thyroid autoimmunity as a window to autoimmunity: an explanation for sex differences in the prevalence of thyroid autoimmunity. J Theor Biol. 2015;375:95–100. doi: 10.1016/j.jtbi.2014.12.015. [DOI] [PubMed] [Google Scholar]
- 45.Lu MC. Yin WY, Tsai TY, Koo M. Lai NS Increased risk of primary Sjogren's syndrome in female patients with thyroid disorders: a longitudinal population-based study in Taiwan PLoS One. 2013;8:e77210. doi: 10.1371/journal.pone.0077210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mavragani CP, Fragoulis GE, Moutsopoulos HM. Endocrine alterations in primary Sjogren's syndrome: an overview. J Autoimmun. 2012;39:354–358. doi: 10.1016/j.jaut.2012.05.011. [DOI] [PubMed] [Google Scholar]
- 47.Appenzeller S, Pallone AT, Natalin RA, Costallat LT. Prevalence of thyroid dysfunction in systemic lupus erythematosus. J Clin Rheumatol. 2009;15:117–119. doi: 10.1097/RHU.0b013e31819dbe4c. [DOI] [PubMed] [Google Scholar]
- 48.Jara LJ, Navarro C, Brito-Zeron Mdel P, et al. Thyroid disease in Sjogren's syndrome. Clin Rheumatol. 2007;26:1601–1606. doi: 10.1007/s10067-007-0638-6. [DOI] [PubMed] [Google Scholar]
- 49.Seror R, Bootsma H, Saraux A, et al. Defining disease activity states and clinically meaningful improvement in primary Sjogren's syndrome with EULAR primary Sjogren's syndrome disease activity (ESSDAI) and patient-reported indexes (ESSPRI) Ann Rheum Dis. 2016;75:382–389. doi: 10.1136/annrheumdis-2014-206008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.


