ABSTRACT
The recent Ebola virus (EBOV) epidemic in West Africa demonstrates the potential for a significant public health burden caused by filoviral infections. No vaccine or antiviral is currently FDA approved. To expand the vaccine options potentially available, we assessed protection conferred by an EBOV vaccine composed of vesicular stomatitis virus pseudovirions that lack native G glycoprotein (VSVΔG) and bear EBOV glycoprotein (GP). These pseudovirions mediate a single round of infection. Both single-dose and prime/boost vaccination regimens protected mice against lethal challenge with mouse-adapted Ebola virus (ma-EBOV) in a dose-dependent manner. The prime/boost regimen provided significantly better protection than a single dose. As N-linked glycans are thought to shield conserved regions of the EBOV GP receptor-binding domain (RBD), thereby blocking epitopes within the RBD, we also tested whether VSVΔG bearing EBOV GPs that lack GP1 N-linked glycans provided effective immunity against challenge with ma-EBOV or a more distantly related virus, Sudan virus. Using a prime/boost strategy, high doses of GP/VSVΔG partially or fully denuded of N-linked glycans on GP1 protected mice against ma-EBOV challenge, but these mutants were no more effective than wild-type (WT) GP/VSVΔG and did not provide cross protection against Sudan virus. As reported for other EBOV vaccine platforms, the protection conferred correlated with the quantity of EBOV GP-specific Ig produced but not with the production of neutralizing antibodies. Our results show that EBOV GP/VSVΔG pseudovirions serve as a successful vaccination platform in a rodent model of Ebola virus disease and that GP1 N-glycan loss does not influence immunogenicity or vaccination success.
IMPORTANCE The West African Ebola virus epidemic was the largest to date, with more than 28,000 people infected. No FDA-approved vaccines are yet available, but in a trial vaccination strategy in West Africa, recombinant, infectious VSV encoding the Ebola virus glycoprotein effectively prevented virus-associated disease. VSVΔG pseudovirion vaccines may prove as efficacious and have better safety, but they have not been tested to date. Thus, we tested the efficacy of VSVΔG pseudovirions bearing Ebola virus glycoprotein as a vaccine platform. We found that wild-type Ebola virus glycoprotein, in the context of this platform, provides robust protection of EBOV-challenged mice. Further, we found that removal of the heavy glycan shield surrounding conserved regions of the glycoprotein does not enhance vaccine efficacy.
KEYWORDS: Ebola virus, filovirus, glycoproteins, glycosylation, pseudovirion, vaccine
INTRODUCTION
Filoviruses, such as Ebola virus (EBOV) and Marburg virus (MARV), cause sporadic outbreaks of viral hemorrhagic fever throughout Central Africa. Most recently, the largest EBOV epidemic on record occurred in West Africa, a region that had not previously experienced filovirus outbreaks (1).
Good overviews of the extensive work on Ebola virus vaccines have recently been published (2–8). A number of different vaccine platforms that express the filovirus glycoprotein (GP) have proven to be effective at protecting against lethal homotypic filovirus challenge in animal models (9–19). Those platforms that have proved efficacious in at least one animal model include DNA plasmids, adenoviral vectors, virus-like particles, recombinant Venezuelan equine encephalitis virus particles, and infectious recombinant viruses, such as human parainfluenza virus type 3, rabies virus, cytomegalovirus, and vesicular stomatitis virus (VSV). These studies have led the field to conclude that immune responses against filovirus GPs are necessary and sufficient for protection. While infectious, recombinant VSV expressing the wild-type (WT) EBOV GP provides effective protection against EBOV disease (20), pseudotyping the viral glycoprotein, EBOV GP, onto VSVΔG has not been evaluated as a vaccine candidate. EBOV GP/VSVΔG pseudovirions have a number of advantages as a vaccine platform, including the absence of virus replication concerns and the robust immune stimulatory activity associated with VSV proteins, which abrogates the need for the addition of an adjuvant (21).
The highly effective EBOV immunogen GP is produced from a proprotein that is processed by furin in cells to produce GP1/GP2 heterodimers. These viral class I heterodimeric glycoproteins reside as trimers on the surfaces of infected cells and virions. Mature GP1 is cradled by GP2, which is anchored in the membrane. GP1 contains four different domains, including a base, receptor-binding domain (RBD), glycan cap and mucin-like domain (MLD). The first three domains compose the core of GP1 and are required for expression and function of the prefusion glycoprotein, whereas the MLD is not required for virion entry or GP expression (22). GP1 is extensively glycosylated, with approximately half of the mature GP mass contributed by N- and O-linked glycans (22). Fifteen N-linked glycans are found on EBOV GP1, and as many as 80 O-linked glycans are thought to be present on the MLD (22–24). The transmembrane protein GP2 contains two N-linked glycans on its ectodomain that are conserved throughout the virus family (22, 23, 25).
Glycans on viral glycoproteins have been shown to facilitate immune evasion through shielding the protein from neutralizing antibodies (26–31). For example, antibodies raised against influenza A virus hemagglutinin (HA), bearing truncated glycans, have enhanced antigen binding and neutralization of virus. Furthermore, decreasing the complexity of N-linked glycans on HA increased the efficacy of an HA subunit vaccine in mice (29). Previous work with EBOV indicated that vaccination with virus-like particles (VLPs) expressing EBOV GP lacking the MLD resulted in up to 2.5-fold-higher neutralizing-antibody titers than with wild-type (WT) GP on VLPs (32), suggesting that glycan shielding compromises EBOV GP immunogenicity. Occlusion of the GP1 RBD is thought to be conferred by N-linked glycans present on the core of GP1 and N- and O-linked glycans on the MLD. Consistent with the possibility that glycan shielding might reduce immune responses to EBOV GP, we have previously shown that removal of all N-linked glycans from EBOV GP1 increases convalescent antibody-dependent neutralization without compromising GP incorporation into VSV pseudovirions (24). By modeling, complete elimination of N-linked glycans on GP1, as is found on our GP mutant 7Gm8G, would be predicted to result in greater surface availability of the highly conserved GP1 RBD. In contrast to these studies, a previous study by Dowling, et al. hinted that loss of glycans might decrease the immune-stimulating efficiency of EBOV GP. They demonstrated that the protection offered by GP was diminished by deleting the MLD or mutation of one of the two GP2 N-glycans (33).
Here, we evaluated the efficacy of EBOV GP pseudotyped onto VSVΔG as a vaccine candidate in a mouse model of filovirus infection. We found robust protection conferred against lethal challenge with mouse-adapted EBOV (ma-EBOV). Protection was strongly associated with production of EBOV GP-specific immunoglobulins (Igs) but not with production of neutralizing antibodies. We also assessed whether removal of EBOV GP1 N-linked glycans that likely expose epitopes masked in the GP RBD leads to increased immunoglobulin and neutralizing-antibody production as well as vaccine efficacy. Further, we evaluated whether these N-linked-glycan-deficient GPs provide better cross protection against other Ebolavirus members, such as Sudan virus (SUDV). We demonstrate that these N-linked glycan site (NGS) mutants provide protection against ma-EBOV challenge equivalent to that provided by WT EBOV/VSVΔG pseudovirions and offer little to no protection against SUDV challenge.
RESULTS
EBOV GP/VSVΔG pseudovirion prime/boost provides better protection than a single dose against ma-EBOV infection.
In initial studies, we assessed the efficacy of our vaccine platform as a single dose delivered subcutaneously (s.c.) versus a prime/boost regimen delivered intramuscularly (i.m.) over a range of 10-fold dilutions of VSVΔG particles bearing WT EBOV GP. The same stock of pseudovirions was used as the vaccine in these studies, and all studies were performed in the absence of an adjuvant. For single-dose vaccination studies, we vaccinated groups of 10 6-week-old C57BL/6 female mice with 2 × 103 to 2 × 107 single-round infectious particles (SRIPs) and lethally challenged with ma-EBOV 3 weeks later. In a prime/boost study, groups of 10 mice were given 2 × 103 to 2 × 107 SRIPs and boosted with the same quantity of pseudovirions 3 weeks later. In the prime/boost regimen, mice were challenged with ma-EBOV 4 weeks after the final vaccination. All phosphate-buffered saline (PBS)-treated mice that were challenged with ma-EBOV succumbed by day 6 to 7 of infection (data not shown). Administration of the higher concentrations of viral particles protected mice, with 2 × 106 to 2 × 107 SRIPs providing 90 to 100% protection by both the single-dose and the prime/boost vaccinations (Fig. 1). Prime/boost administration of 2 × 105 SRIPs also gave complete protection; however, a single dose with this same amount of SRIPs was not effective. Lower doses of either regimen provided little to no protection. Thus, the quantity of GP-containing pseudovirions strongly correlated with protection. A statistical comparison of the protection conferred by a single dose of our wild-type EBOV GP/VSVΔG SRIPs delivered s.c. versus prime/boost vaccination delivered i.m. demonstrated that the hazard ratio for prime/boost versus single dose was 0.428 (P = 0.0013), providing support that our prime/boost regimen worked significantly better at protecting from death than our single-dose regimen. Prime/boost vaccination with 2 × 107 SRIPs i.m. followed by ma-EBOV challenge was also performed in BALB/c mice, with similar protection conferred (data not shown).
FIG 1.
A prime/boost regimen provides better protection against lethal ma-EBOV challenge than a single dose of EBOV GP/VSVΔG. Groups (n = 10) of C57BL/6 mice received the indicated amounts of EBOV GP/VSVΔG SRIPs as either a single dose s.c. or a prime-plus-boost dose i.m., followed by lethal challenge with ma-EBOV. Data are presented as the percentage of survival at the indicated SRIP dose.
Somewhat surprisingly, the regimens that did not protect against death also did not extend time to death beyond that observed in the PBS-treated mice (data not shown). This stands in contrast to some, but not all, other EBOV vaccine studies, where vaccines that protect a portion of the vaccinated population may also prolong survival of those animals that do ultimately succumb to the infection (12, 13, 34, 35).
Protection is also conferred by N-linked-glycan-deficient EBOV GP/VSVΔG pseudovirions.
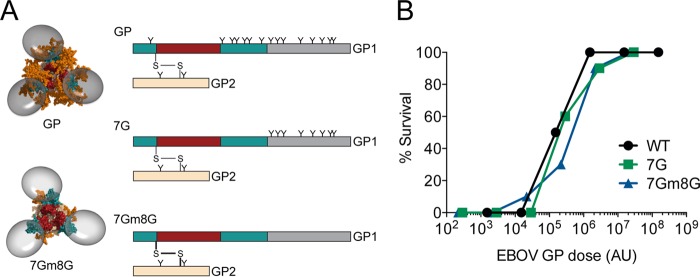
In parallel with the vaccine studies described above, we evaluated the level of protection conferred by pseudovirions bearing GP1 that was partially (7G) or fully (7Gm8G) denuded of N-linked glycans (Fig. 2A). Our previous study demonstrated that VSVΔG particles bearing these mutant GPs were more infectious but also more easily neutralized than WT particles by convalescent-phase sera from vaccinated and challenged nonhuman primates (NHPs) (24). When modeled on the structure of the trimeric GP ectodomain, N-linked glycans on the RBD and glycan cap of GP1 are shown to sterically occlude the RBD (Fig. 2A), and upon removal of these glycans, the RBD may be a more available immune target. Hence, we reasoned that VSVΔG pseudovirions bearing N-linked-glycan-deficient GPs might serve as better immunogens than fully glycosylated WT EBOV GP. The prime/boost regimens of 10-fold serial dilutions of WT, 7G, or 7Gm8G SRIPs were administered i.m. as described above. WT, 7G, and 7Gm8G VSVΔG particles when normalized for VSV matrix did not contain equivalent quantities of GP; therefore, the protection conferred against lethal challenge was assessed relative to the GP administered during the vaccinations (Fig. 2B). The protection offered by all three pseudovirion regimens was similar, with the two highest doses protecting 90 to 100% of mice against ma-EBOV challenge and the lowest doses yielding no protection. These studies support that the protection conferred by the vaccines correlates with to the amount of GP administered. The ability of equivalent doses of mutant GP pseudovirions and WT GP pseudovirions to confer protection suggests that GP1 N-linked glycosylation status has little impact on protection against lethal challenge.
FIG 2.
EBOV GP NGS mutant models and vaccination efficacy. (A) Left, top-down view of models of EBOV GP trimer (top) and a GP mutant deficient in GP1 N-linked glycans (7Gm8G) (bottom). Right, linear models of GP constructs utilized in this study. Positions of N-linked glycans are marked with “Y,” and the GP1-GP2 disulfide bond is shown. The RBD is shown in red, the glycan cap is shown in teal, and GP2 is shown in tan, N-linked glycans are shown in orange, and the MLD (not included in the crystal structure) is shown in gray and modeled as a sphere in the structure. The two conserved N-linked glycans found on GP2 were not mutated in our glycosylation mutants investigated in these studies and are shown around the base of GP2. (B) Dose-dependent protection conferred by WT or mutant EBOV GP/VSVΔG pseudovirions that are partially (7G) or fully (7Gm8G) deficient in N-linked glycans. Groups (n = 10) of C57BL/6 mice received a prime/boost of VSVΔG SRIPs bearing the indicated GP followed by lethal challenge with ma-EBOV. Data are presented as the percentage of survival at the indicated SRIP dose, normalized for EBOV GP immunogen expression as determined by quantitative Western blotting. AU, arbitrary units of GP.
To determine if exposure of highly conserved regions of GP1 by the loss of N-linked glycans led to quantitative changes in anti-EBOV GP antibodies produced against conserved regions of GP, we assessed the amount of immunoglobulin (Ig) in sera from prime/boost vaccinees. Enzyme-linked immunosorbent assays (ELISAs) were performed to detect mouse IgG and IgM that bound to EBOV GPΔMLD pseudotyped onto feline immunodeficiency virus (FIV). An EBOV GP with the MLD deleted was selected as the target in these ELISAs, since recent findings indicate that antibodies against the core of EBOV GP are sufficient for protection (36). Further, we were interested to determine whether vaccination with EBOV GPs lacking the GP1 N-linked glycans might lead to a greater quantity of antibodies targeting the RBD and glycan cap region of GP. While the quantity of anti-EBOV GP Ig produced by the vaccination treatments correlated strongly with the dose of GP administered, we did not observe greater amounts of Ig against EBOV GPΔMLD produced when we vaccinated with pseudovirions bearing the N-linked glycan-deficient GPs (Fig. 3A). Instead, the quantities of anti-EBOV GP antibodies generated with increasing doses of the three vaccines were similar. We also observed that the amount of anti-EBOV GP Ig produced by each vaccine regimen correlated with similar levels of survival (Fig. 3B). Vaccine treatments that resulted in anti-EBOV GPΔMLD Ig levels of 100 μg/ml or more were protective, independent of the glycosylation status of the GP used in the regimen. Partial protection was conferred with the generation of 30 to 60 μg/ml of Ig, and production of low levels (17 μg/ml of Ig or less) of anti-EBOV GP Igs was not protective. These findings are consistent with other reports demonstrating that the quantity of anti-EBOV GP antibodies correlated with vaccine efficacy (37) and with the idea that anti-EBOV GP Ig production serves as an excellent immune correlate for vaccine protection.
FIG 3.
Humoral responses of mice vaccinated by the prime/boost regimen. Sera were collected from vaccinated mice 3 weeks following the boost. (A) Correlation between the quantity of EBOV GP immunogen administered and Ig production. AU, arbitrary units of GP. (B) Correlation between group survival and anti-EBOV GP ΔMLD Ig in sera. The trend line for pooled results from all three groups is shown. (C) Lack of neutralizing activity present in sera from mice vaccinated with 2 × 107 SRIPs that were normalized for VSV matrix. Serum was serially diluted and incubated with EBOV GP/VSVΔG for 30 min at 37°C prior to addition to Vero cells. Data are shown as infection (GFP-positive cells) as a percentage of the value for a PBS control. (D) Passive transfer of immunity. Groups (n = 10) of C57BL/6 mice received the indicated serum 1 day following lethal challenge with ma-EBOV. Significance was determined by a Cox proportional hazards model (***, P < 0.0001).
Given the positive association of serum neutralizing antibodies with survival against EBOV in human and animal model infections (9, 34, 38) and our observed correlation between survival and anti-EBOV GP antibody levels in sera from vaccinated mice, we evaluated the ability of sera from vaccinated mice to neutralize EBOV GP/VSVΔG pseudovirion infection of Vero cells, using the criterion of at least 50% inhibition of EBOV GP/VSVΔG infection as evidence of neutralization. Sera from mice vaccinated with even our highest concentrations of pseudovirions did not meet our criterion for neutralizing activity, despite providing robust protection against lethal challenge (Fig. 3C). These results indicate that VSVΔG pseudovirion-based vaccines against EBOV elicit protective immune responses that can be independent of the production of antibodies that neutralize viral particles in cell culture assays. Consistent with our findings, protection conferred by other EBOV vaccines does not always correlate with the production of neutralizing antibodies (12, 13, 39, 40).
To determine if the anti-EBOV GP antibodies that we detected were simply serving as immune correlates of protection or directly contributed to protection against lethal challenge, we performed passive immunization assays. Anti-EBOV GP IgG from sera given postexposure have been shown to protect NHPs against lethal challenge (41). Mice given a lethal dose of ma-EBOV were inoculated intraperitoneally (i.p.) on day 1 of infection with 0.4 ml of serum from either naive mice or mice vaccinated with WT EBOV GP/VSVΔG pseudovirions. Vaccinated sera were obtained from mice prime/boosted with 2 × 107 SRIPs of EBOV GP/VSVΔG as described above, and serum was collected 3 weeks following the boost. While all mice given naive serum succumbed to infection, 100% of the mice were protected by the passive transfer of serum from vaccinated animals (Fig. 3D). This study provides evidence that our vaccine platform successfully generates GP-specific antibodies capable of controlling ma-EBOV disease and emphasizes the importance of GP-elicited antibodies in vaccine-generated control of filovirus infection.
Vaccination with N-linked-glycan-deficient EBOV GP/VSVΔGs does not provide protection against wild-type SUDV.
EBOV GP immunogens lacking a protective N-linked glycan shield around the conserved RBD might be predicted to generate antibodies to more-conserved GP epitopes and thereby provide cross protection against more distantly related filoviruses. To test this possibility, we vaccinated mice in a prime/boost schedule with 2 × 107 7G or 7Gm8G SRIPs and challenged them with SUDV. VSVΔG pseudovirions bearing SUDV GP served as the positive control for these studies, since others have shown that vaccines containing the homotypic SUDV GP protect against SUDV challenge (42, 43). VSVΔG pseudovirions bearing the Lassa virus (LASV) glycoprotein complex (GPC) served as the negative control in these studies. C57BL/6 mice lacking the interferon alpha/beta receptor (IFNAR−/−) were used, as a mouse-adapted strain of SUDV that is lethal to immunocompetent mice is not currently available; previous work has demonstrated that IFNAR−/− mice have significant and consistent weight loss upon WT SUDV or EBOV infection, and we assessed weight loss in these SUDV-challenged mice (44). WT EBOV GP pseudovirions were not evaluated for their ability to confer protection against SUDV, as others have previously demonstrated that WT EBOV GP does not provide cross protection (44). Mice vaccinated with PBS, SUDV GP/VSVΔG, 7G GP/VSVΔG, or 7Gm8G/VSVΔG pseudovirions were challenged with 1,000 PFU of WT EBOV (Kitwik) (Fig. 4A) or WT SUDV (Boniface) (Fig. 4B) i.p., and mice were weighed daily as previously described (45, 46). While SUDV GP/VSVΔG pseudovirions protected against weight loss associated with SUDV challenge, 7G- and 7Gm8G-vaccinated mice had weight loss similar to that of mice given pseudovirions bearing Lassa virus GPC or the PBS control, indicating that our mutant EBOV GPs were unable to provide protection, despite the fact that the same vaccination dosage protected well against WT EBOV challenge.
FIG 4.
VSVΔG SRIPs bearing EBOV GP deficient in N-linked glycans do not provide protection against SUDV challenge. Groups (n = 10) of C57BL/6 IFNAR−/− mice were primed and boosted with 2 × 107 SRIPs bearing the indicated viral glycoprotein, followed by challenge with WT EBOV (A) or WT SUDV (B). The percentages of weight loss at the indicated times following infection are shown.
DISCUSSION
In the present study, we sought to determine if mice vaccinated with EBOV GP-pseudotyped VSVΔG are protected from EBOV or SUDV challenge. We found that either a single dose or our prime/boost regimen of 2 × 106 SRIPs or more of WT EBOV GP/VSVΔG was protective. A dose of 2 × 105 SRIPs was efficacious only if given under our prime/boost regimen conditions, and lower concentrations of SRIPs did not provide complete protection. These findings suggest that VSVΔG SRIP-based vaccines may provide a highly effective alternative to other EBOV vaccine platforms and identify quantities of SRIPs that confer protection in a mouse model. It should be noted that protection of mice against ma-EBOV is relatively easy to achieve, occurring more readily than that observed in other established animal models (47–49). Thus, while the inexpensive mouse model is the logical first in vivo system in which to test a new vaccine platform, additional testing in more stringent models is needed.
The VSVΔG SRIP platform has a number of advantages similar to those of the recombinant, replication-competent EBOV GP/rVSV but without the potential safety issues associated with an infectious vaccine. For instance, as with the recombinant VSV platform, protection was possible with a single dose of the pseudovirions, albeit requiring relatively high pseudovirion quantities. In addition, no additional adjuvant was needed to elicit protection. This stands in contrast to findings with virus-like particles, where protection in mice against lethal filovirus challenge requires either a regimen of a prime and two boosts or the presence of adjuvants (9, 42, 50–52). VSVΔG SRIPs bearing the glycoprotein from other highly pathogenic viruses such as Nipah virus have also been demonstrated to serve as a protective vaccine (53, 54). It is thought that one or more VSV proteins have adjuvant-like properties, thereby eliciting robust adaptive immune responses to the vaccine immunogen (21, 55, 56), although which VSV protein(s) is important for this enhancing effect has not been identified.
The single-dose regimen given s.c. required delivery of 2 × 106 particles to obtain complete protection of EBOV-challenged mice, whereas the prime/boost i.m. regime required 10-fold-lower concentrations of SRIPs to achieve the same protection. Whether the difference in protection conferred is due to the prime-only versus the prime/boost regimen or to the route of delivery remains to be determined. By comparison, others have shown that as little as 2 PFU of replication-competent EBOV GP/rVSV given i.p. provides complete protection (57), but protection studies in mice typically administer 2 × 104 PFU of EBOV GP/rVSV (57, 58). Protection conferred by these smaller doses is presumably due to the ability of infectious EBOV GP/rVSV to replicate in vivo following vaccination.
Our VSVΔG SRIP-based vaccine demonstrated strong dose-dependent antibody production against EBOV glycoprotein and protection against EBOV challenge; however, we did not examine T cell responses elicited by the vaccine. Others have shown robust VSV-based vaccine stimulation of T cell responses (55, 56, 59–61), but most of these studies have been performed with replication-competent VSV rather than VSVΔG SRIPs. To our knowledge, there is a single report on T cell responses to an antigen delivered by VSV SRIPs. That report showed that VSV SRIPs encoding HIV Env elicit T and B cell responses against Env that are as robust as those elicited by the replication-competent vaccine (59).
The detailed dose-response studies performed here provided strong experimental evidence that Ig levels against EBOV GP that lacks the MLD correlated in a dose-dependent manner with the amount of immunogen vaccinated, and this Ig level serves as a good immune marker predictive for protection. Our finding is consistent with a previous meta-analysis, demonstrating that Ig levels against the whole EBOV GP correlate strongly with the protection conferred (37). However, this robust correlation with Ig responses was not associated with evidence of serum neutralization of EBOV GP/VSVΔG pseudovirions in our in vitro assays, as others have also reported (12, 13, 62). In combination with our passive serum transfer studies, our findings provide evidence that our vaccine elicits protective humoral responses that are independent of neutralizing antibodies. Future studies are needed to define the mechanism by which these immunoglobulins mediate protection.
We also explored whether EBOV GP pseudovirion protection was altered by partial or complete loss of N-linked glycans on GP1. We hypothesized that an immunogen with reduced GP1 glycosylation might increase immune targeting of RBD epitopes that are relatively conserved across the ebolaviruses. However, we found in our Ig ELISAs that equivalent quantities of antibodies against EBOV GPΔMLD (containing a GP1 composed solely of the base, RBD, and glycan cap) were generated in the mice vaccinated with pseudovirions bearing WT EBOV GP versus GPs partially or fully lacking N-linked glycans. Consistent with this, we found that the loss of N-linked glycans conferred similar protection against ma-EBOV challenge as our WT vaccine. Thus, our findings suggest that these glycans on EBOV GP1 neither hinder nor enhance the ability of GP to serve as an immunogen. Thus, if antibodies targeting the relatively highly conserved EBOV RBD are generated by our mutant GP immunogens, those epitopes likely are unavailable antibody targets in the WT glycoprotein. Consistent with shielding of conserved epitopes on GP1, the RBD of EBOV GP is not commonly found to be a target of anti-EBOV GP monoclonal antibodies. Recently, Hashiguchi et al. showed that a monoclonal antibody, MR78, which binds to the Marburg virus GP RBD does not bind to full-length EBOV GP and is only able to bind to the conserved epitope upon proteolytic cleavage of EBOV GP (63). This argues that structural constraints such as glycan shielding of RBD epitopes and/or conformation constraints of the WT uncleaved GP make conserved epitopes of the EBOV RBD unavailable. In addition, since our glycan mutants retained O-linked glycans on the MLD, it is possible that these smaller but highly abundant sugar chains may also shield protective epitopes on conserved regions of GP. A recent study using single-particle cryo-electron microscopy provides evidence that the MLD structure may occlude the RBD within the GP structure (64), thereby protecting potential epitopes.
These studies also demonstrated that pseudovirions bearing N-linked-glycan-denuded GP1 provide poor cross protection against SUDV challenge. Thus, our studies provide no evidence that an EBOV GP immunogen lacking NGSs on GP1 elicits novel protective immune responses to highly conserved regions of GP on the surfaces of extracellular SUDV particles. To date, the most effective broad-spectrum protection has been achieved through combinatorial vaccines that present proteins from multiple viral species to the host (11, 39, 44, 65–67). Taking this together with our results, a mixture of VSVΔG pseudotyped with representative GPs from the filovirus family could provide an efficient, tractable, and safe vaccine that warrants further investigation in other animal model systems.
MATERIALS AND METHODS
Cell lines and plasmids.
Vero cells and HEK293T cells were maintained in Dulbecco's modified Eagle medium (DMEM) with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin. The pcDNA3.1 expression plasmids for EBOV GP (accession number NP_066246) and the N-linked-glycan-deficient GP1 mutants 7G and 7Gm8G have been previously described (24). The 7G GP was mutated to eliminate the seven N-linked glycans in the core region (RBD and glycan cap) of GP1, whereas 7Gm8G eliminated all 15 N-linked glycans in GP1. All O-linked glycan sites on GP1 and the two N-linked glycans on GP2 were retained on these mutants. Codon-optimized SUDV Boniface GP (a gift from Robert Davey; accession number Q66814) containing a Q95K mutation, which we have determined increases pseudovirion titer (data not shown), was expressed from pcDNA3.1. The pCAGGS vector was used to express codon-optimized Lassa virus Josiah glycoprotein (accession number: NP_694870).
Modeling of GP N-linked glycans.
The addition of complex N-linked glycans to the prefusion EBOV GP1,2 ΔTM structure (PDB ID 3CSY) that lacks the MLD was performed as previously described (24, 25). Briefly, the published structure lacks four NGSs in GP1 due to disordered regions missing from the structure (N204 and N296) or mutations that promoted crystallization (N40 and N228) (23, 68). The EBOV GP sequence was submitted to the PHYRE2 protein fold recognition server (69), which provided a structure that contained NGSs at N40 and N228. This structure was submitted for in silico glycosylation using the GlyProt server (glycosciences.de), which produced a model containing complex N-linked glycans at all NGSs except N204 and N296, which are part of disordered regions (23). Complex glycans at these sites were modeled onto the glycosylated structure at predicted sites with PyMOL.
Production of VSVΔG/GFP pseudovirion vaccine stocks.
Pseudovirions were produced in HEK293T cells as previously described (70, 71). Briefly, HEK293T cells were transfected with the various EBOV GP-expressing constructs or Lassa virus (LASV) GPC, which served as a control in some studies. At 24 h following DNA transfection, the cells were infected (multiplicity of infection [MOI] of ∼0.5) with stock of a defective vesicular stomatitis virus that expresses the green fluorescence protein (GFP) gene but lacks the gene encoding the native G glycoprotein (VSVΔG-GFP) pseudotyped with LASV GPC. After 24 h and 48 h, cell supernatants were collected and filtered through 0.45-μm syringe filters, followed by storage at −80°C. Pseudovirions were concentrated overnight by centrifugation at 5,400 × g. Virus was resuspended in PBS, pelleted through a 20% sucrose cushion for 2 h at 83,000 × g, and resuspended in PBS treated with Detoxi-Gel endotoxin-removing resin (Thermo Scientific) to remove any endotoxin in our preparations. All virus stocks were frozen at −80°C until use.
Protein quantification to normalize pseudovirion administration.
In comparisons of the vaccine efficacy of wild-type and N-linked-glycan-deficient glycoproteins, the amount of the different pseudovirions administered in each vaccine study was adjusted for GP equivalents. To quantify this, purified pseudovirions were serially diluted, and proteins were separated by SDS-PAGE, transferred to nitrocellulose, and probed with anti-EBOV GP monoclonal antibody 21D10 (72) and VSV matrix (M) 23H12 (73). 21D10 recognizes filovirus GP residues 81 to 89, a region of GP that does not contain N-linked glycosylation and so should recognize the denatured GPs equivalently. Signals were quantified using the Odyssey Imaging Station and Image Studio software (Li-Cor). GP pixel numbers were normalized for matrix pixel number.
Single-cycle infectivity assays.
Vero cells were seeded in 48-well plates 24 h prior to transduction. Serial dilutions of WT EBOV GP/VSVΔG pseudovirion stocks were added to Vero cell monolayers. Infectivity was determined by quantification of GFP-expressing cells by flow cytometry 24 h following addition of viral particles.
Ethics statement.
Animal research at the University of Iowa was conducted under a protocol approved by the University of Iowa Institutional Animal Care and Use Committee (IACUC) in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals. Animal research at the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID) was conducted under a protocol approved by the USAMRIID IACUC in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals. Both facilities are fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International and adhere to the principles stated in the Guide for the Care and Use of Laboratory Animals (74). Challenge studies were conducted under maximum containment in the USAMRIID animal biosafety level 4 (ABSL-4) facility.
Vaccinations and filovirus challenges.
The amounts of the different pseudovirions administered in each vaccine study were normalized to equal levels of virion matrix equivalents. For each glycoprotein tested for its efficacy as a pseudovirion vaccine, a single large stock of pseudovirions was produced, characterized, aliquoted, and stored at −80°C until use. In challenge studies using mouse-adapted EBOV (ma-EBOV), two different stocks of ma-EBOV were used over the course of these studies. The challenge dose used for each stock was selected based on standard lethality studies that were completed for each new virus stock. The dose of stock 1 was 1,000 PFU intraperitoneally (i.p.) or 30,000 50% lethal doses (LD50). The lethal dose of stock 2 was 100 PFU or 3,000 LD50. At the doses administered, both stocks gave equivalent lethality.
Prime/boost ma-EBOV challenge studies.
Six-week-old female C57BL/6 mice were purchased from Charles River. Tenfold serial dilutions in PBS of WT EBOV GP/VSVΔG pseudovirions (2 × 103 to 2 × 107units of single-round infectious particles [SRIPs], determined via titration of pseudovirions on Vero cells) were given intramuscularly (i.m.) as a prime and then a boost 3 weeks later (n = 10/group). The quantity of other VSVΔG pseudovirions (7G and 7Gm8G) administered was based on matrix equivalents to that of EBOV GP/VSVΔG using the same dilution series and prime/boost strategy. The findings from these studies were then adjusted for GP dosage administered via the pseudovirions. Serum was collected by retro-orbital bleed 6 weeks following initial vaccination. Vaccinated mice were shipped to USAMRIID (Fort Detrick, MD), where mice were housed in microisolator cages and provided chow and water ad libitum. At 7 weeks following initiation of the vaccination regimen, mice were challenged with 1,000 PFU of stock 1 of ma-EBOV i.p. Challenged mice were observed daily for lethality or clinical signs of disease, including, but not limited to, reduced grooming, hypoactivity, and weight loss. Observations were increased to a minimum of twice daily while mice were exhibiting signs of disease. Moribund mice were humanely euthanized based on IACUC-approved criteria.
Prime/boost SUDV versus WT EBOV challenge studies.
Four- to 8-week-old male and female mice lacking the interferon alpha/beta receptor (C57BL/6.129S2-Ifnar1tm1Agt/Mmjax; referred to here as IFNAR−/−) were purchased from Jackson Laboratories (Bar Harbor, ME) and used for all Sudan virus (SUDV) challenge experiments (n = 10/group). Upon arrival, mice were housed in microisolator cages and provided chow and water ad libitum. All vaccinated mice received the matrix equivalents of 2 × 107 SRIPs of WT EBOV GP/VSVΔG i.m. in a prime/boost regimen. Seven weeks following vaccinations, mice were challenged i.p. with 1,000 PFU of SUDV (Boniface isolate) or 1,000 PFU of WT EBOV. Wild-type strains of both SUDV and EBOV, rather than mouse-adapted strains, were used in these studies since no mouse-adapted strains of SUDV have been developed to date. Mice were monitored as described above.
Single-vaccine-dose ma-EBOV challenge studies.
Six-week-old female C57BL/6 mice were purchased from Charles River. At USAMRIID, 10-fold serial dilutions in PBS of WT EBOV GP/VSVΔG pseudovirions (2 × 103 to 2 × 107 SRIPs) were given subcutaneously (s.c.) (n = 10/group). Vaccinated mice were challenged with 100 PFU of stock 2 of ma-EBOV i.p. at 21 days following vaccination. Challenged mice were observed daily for lethality or clinical signs of disease, including, but not limited to, reduced grooming, hypoactivity, and weight loss. Observations were increased to a minimum of twice daily while mice were exhibiting signs of disease. Moribund mice were humanely euthanized based on IACUC-approved criteria.
IgG titers and neutralization assays.
Sera taken from mice in our prime/boost studies were assessed for immunoglobulin (IgG and IgM) detecting EBOV GP lacking the mucin domain by ELISA. Briefly, 2 × 105 SRIPs of purified EBOV GPΔMLD/feline immunodeficiency virus (FIV) pseudovirions (as assessed on SNB-19 cells) or a mouse IgG standard curve was applied overnight in PBS at 4°C in Immulon HB 96-well plates. FIV pseudovirions were generated as previously described (75). Serial dilutions of sera were added to plates and left overnight at 4°C. Wells were washed with PBS and 0.125% Tween 20 and incubated with 1 μg/ml horseradish peroxidase (HRP)-conjugated rabbit anti-mouse Ig secondary antibody (Pierce) and washed again. OptEIA (BD Biosciences) was used to assess HRP activity; the reaction was stopped with the addition of H2SO4 and results read at 405 nm on a microtiter plate reader.
To assess the quantity of antibodies made against just the core of EBOV GP in our prime/boosted vaccinated mice, a mucin like domain (MLD)-deleted EBOV GP on FIV pseudovirions was selected as the target in these ELISAs, since recent findings indicate that antibodies against the core of EBOV GP are sufficient for protection (36). Briefly, 2 × 105 SRIPs of purified EBOV GPΔMLD/FIV pseudovirions (as assessed on SNB-19 cells) were applied and left overnight in PBS at 4°C in an Immulon HB 96-well plate. FIV pseudovirions were generated as previously described (76). Virus was diluted in PBS. The standard curve was composed of a 2-fold dilution series of mouse Ig fraction (ImmunoReagent, Inc.). Plates were blocked for 1 h at room temperature with PBS with 2% bovine serum albumin (BSA), incubated with serial dilutions (1:1,000, 1:10,000, and 1:100,000) of serum overnight at 4°C, and probed with 1 μg/ml HRP-conjugated rabbit anti-mouse antibody (Pierce) for 1 h, and wells were assessed for HRP activity with UltraTMB (Thermo). Absorbance was read at 405 nm.
To assess the ability of our vaccinated mouse serum to neutralize EBOV GP-mediated entry, serial dilutions of serum were incubated with WT EBOV GP VSVΔG pseudovirions (WT MOI of ∼0.3), for 30 min at 37°C. The reaction mixtures were then diluted 5-fold in medium and added to confluent monolayers of Vero cells.
Passive immunizations.
All vaccinated mice were primed and then boosted 21 days later with 2 × 107 SRIPs of wild-type EBOV GP/VSVΔG. At 42 days following the initial vaccination, serum was collected. Sera from naive mice is used as a control in this study. Ten 8- to 12-week-old female C57BL/6 mice were inoculated i.p. with 100 PFU of stock 2 of ma-EBOV. On day 1 of infection, one group was treated i.p. with serum (0.4-ml volume) collected from vaccinated mice. Control mice received naive serum (0.4-ml volume) i.p. at that time. Mice were observed daily for 28 days for moribund condition. Moribund mice were promptly euthanized when they meet euthanasia criteria.
Statistical analyses.
The efficacies of the different vaccine regimens to prolong survival were compared by a Cox proportional hazards model, determining the hazard ratio. The significance of neutralization data was determined by a one-sample t test.
ACKNOWLEDGMENTS
The opinions, conclusions, interpretations, and recommendations are those of the authors and are not necessarily endorsed by the U.S. Army. The mention of trade names or commercial products does not constitute endorsement or recommendation for use by the Department of the Army or the Department of Defense.
This work was supported by funding from the National Institutes of Health (grants R01 AI077519 to W.M., T32AI007533 to support N.J.L. and R.B., and T32 AI996343 to support C.L.M.-H.) and the Department of Defense (grant CB3947 to J.M.D.).
REFERENCES
- 1.Incident Management System Ebola Epidemiology Team CDC, Guinea Interministerial Committee for Response Against the Ebola Virus, World Health Organization, CDC Guinea Response Team, Liberia Ministry of Health and Social Welfare, CDC Liberia Response Team, Sierra Leone Ministry of Health, CDC Sierra Leone Response Team, Viral Special Pathogens Branch, National Center for Emerging and Zoonotic Infectious Diseases, CDC. 2015. Update: Ebola virus disease epidemic—West Africa, January 2015. MMWR Morb Mortal Wkly Rep 64:109–110. [PMC free article] [PubMed] [Google Scholar]
- 2.Friedrich BM, Trefry JC, Biggins JE, Hensley LE, Honko AN, Smith DR, Olinger GG. 2012. Potential vaccines and post-exposure treatments for filovirus infections. Viruses 4:1619–1650. doi: 10.3390/v4091619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hoenen T, Groseth A, Feldmann H. 2012. Current ebola vaccines. Expert Opin Biol Ther 12:859–872. doi: 10.1517/14712598.2012.685152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sridhar S. 2015. Clinical development of Ebola vaccines. Ther Adv Vaccines 3:125–138. doi: 10.1177/2051013615611017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Medaglini D, Harandi AM, Ottenhoff TH, Siegrist CA, Consortium VS-E. 2015. Ebola vaccine R&D: filling the knowledge gaps. Sci Transl Med 7:317ps324. doi: 10.1126/scitranslmed.aad3106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ohimain EI. 2016. Recent advances in the development of vaccines for Ebola virus disease. Virus Res 211:174–185. doi: 10.1016/j.virusres.2015.10.021. [DOI] [PubMed] [Google Scholar]
- 7.Mire CE, Geisbert TW, Feldmann H, Marzi A. 2016. Ebola virus vaccines—reality or fiction? Expert Rev Vaccines 15:1421–1430. doi: 10.1080/14760584.2016.1178068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Henao-Restrepo AM, Preziosi MP, Wood D, Moorthy V, Kieny MP, WHO Ebola Research Development Team. 2016. On a path to accelerate access to Ebola vaccines: the WHO's research and development efforts during the 2014-2016 Ebola epidemic in West Africa. Curr Opin Virol 17:138–144. doi: 10.1016/j.coviro.2016.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Warfield KL, Bosio CM, Welcher BC, Deal EM, Mohamadzadeh M, Schmaljohn A, Aman MJ, Bavari S. 2003. Ebola virus-like particles protect from lethal Ebola virus infection. Proc Natl Acad Sci U S A 100:15889–15894. doi: 10.1073/pnas.2237038100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jones SM, Feldmann H, Stroher U, Geisbert JB, Fernando L, Grolla A, Klenk HD, Sullivan NJ, Volchkov VE, Fritz EA, Daddario KM, Hensley LE, Jahrling PB, Geisbert TW. 2005. Live attenuated recombinant vaccine protects nonhuman primates against Ebola and Marburg viruses. Nat Med 11:786–790. doi: 10.1038/nm1258. [DOI] [PubMed] [Google Scholar]
- 11.Pratt WD, Wang D, Nichols DK, Luo M, Woraratanadharm J, Dye JM, Holman DH, Dong JY. 2010. Protection of nonhuman primates against two species of Ebola virus infection with a single complex adenovirus vector. Clin Vaccine Immunol 17:572–581. doi: 10.1128/CVI.00467-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rao M, Bray M, Alving CR, Jahrling P, Matyas GR. 2002. Induction of immune responses in mice and monkeys to Ebola virus after immunization with liposome-encapsulated irradiated Ebola virus: protection in mice requires CD4(+) T cells. J Virol 76:9176–9185. doi: 10.1128/JVI.76.18.9176-9185.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pushko P, Bray M, Ludwig GV, Parker M, Schmaljohn A, Sanchez A, Jahrling PB, Smith JF. 2000. Recombinant RNA replicons derived from attenuated Venezuelan equine encephalitis virus protect guinea pigs and mice from Ebola hemorrhagic fever virus. Vaccine 19:142–153. doi: 10.1016/S0264-410X(00)00113-4. [DOI] [PubMed] [Google Scholar]
- 14.Vanderzanden L, Bray M, Fuller D, Roberts T, Custer D, Spik K, Jahrling P, Huggins J, Schmaljohn A, Schmaljohn C. 1998. DNA vaccines expressing either the GP or NP genes of Ebola virus protect mice from lethal challenge. Virology 246:134–144. doi: 10.1006/viro.1998.9176. [DOI] [PubMed] [Google Scholar]
- 15.Sullivan NJ, Geisbert TW, Geisbert JB, Xu L, Yang ZY, Roederer M, Koup RA, Jahrling PB, Nabel GJ. 2003. Accelerated vaccination for Ebola virus haemorrhagic fever in non-human primates. Nature 424:681–684. doi: 10.1038/nature01876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sullivan NJ, Sanchez A, Rollin PE, Yang ZY, Nabel GJ. 2000. Development of a preventive vaccine for Ebola virus infection in primates. Nature 408:605–609. doi: 10.1038/35046108. [DOI] [PubMed] [Google Scholar]
- 17.Bukreyev A, Rollin PE, Tate MK, Yang L, Zaki SR, Shieh WJ, Murphy BR, Collins PL, Sanchez A. 2007. Successful topical respiratory tract immunization of primates against Ebola virus. J Virol 81:6379–6388. doi: 10.1128/JVI.00105-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tsuda Y, Caposio P, Parkins CJ, Botto S, Messaoudi I, Cicin-Sain L, Feldmann H, Jarvis MA. 2011. A replicating cytomegalovirus-based vaccine encoding a single Ebola virus nucleoprotein CTL epitope confers protection against Ebola virus. PLoS Negl Trop Dis 5:e1275. doi: 10.1371/journal.pntd.0001275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Papaneri AB, Bernbaum JG, Blaney JE, Jahrling PB, Schnell MJ, Johnson RF. 2015. Controlled viral glycoprotein expression as a safety feature in a bivalent rabies-Ebola vaccine. Virus Res 197:54–58. doi: 10.1016/j.virusres.2014.11.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Henao-Restrepo AM, Camacho A, Longini IM, Watson CH, Edmunds WJ, Egger M, Carroll MW, Dean NE, Diatta I, Doumbia M, Draguez B, Duraffour S, Enwere G, Grais R, Gunther S, Gsell PS, Hossmann S, Watle SV, Konde MK, Keita S, Kone S, Kuisma E, Levine MM, Mandal S, Mauget T, Norheim G, Riveros X, Soumah A, Trelle S, Vicari AS, Rottingen JA, Kieny MP. 23 December 2016. Efficacy and effectiveness of an rVSV-vectored vaccine in preventing Ebola virus disease: final results from the Guinea ring vaccination, open-label, cluster-randomised trial (Ebola Ca Suffit!). Lancet doi: 10.1016/S0140-6736(16)32621-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cobleigh MA, Bradfield C, Liu Y, Mehta A, Robek MD. 2012. The immune response to a vesicular stomatitis virus vaccine vector is independent of particulate antigen secretion and protein turnover rate. J Virol 86:4253–4261. doi: 10.1128/JVI.05991-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jeffers SA, Sanders DA, Sanchez A. 2002. Covalent modifications of the ebola virus glycoprotein. J Virol 76:12463–12472. doi: 10.1128/JVI.76.24.12463-12472.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee JE, Fusco ML, Hessell AJ, Oswald WB, Burton DR, Saphire EO. 2008. Structure of the Ebola virus glycoprotein bound to an antibody from a human survivor. Nature 454:177–182. doi: 10.1038/nature07082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lennemann NJ, Rhein BA, Ndungo E, Chandran K, Qiu X, Maury W. 2014. Comprehensive functional analysis of N-linked glycans on Ebola virus GP1. mBio 5:e00862-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lennemann NJ, Walkner M, Berkebile AR, Patel N, Maury W. 2015. The role of conserved N-linked glycans on Ebola virus glycoprotein 2. J Infect Dis 212(Suppl 2):S204–S209. doi: 10.1093/infdis/jiv201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wei X, Decker JM, Wang S, Hui H, Kappes JC, Wu X, Salazar-Gonzalez JF, Salazar MG, Kilby JM, Saag MS, Komarova NL, Nowak MA, Hahn BH, Kwong PD, Shaw GM. 2003. Antibody neutralization and escape by HIV-1. Nature 422:307–312. doi: 10.1038/nature01470. [DOI] [PubMed] [Google Scholar]
- 27.Aguilar HC, Matreyek KA, Filone CM, Hashimi ST, Levroney EL, Negrete OA, Bertolotti-Ciarlet A, Choi DY, McHardy I, Fulcher JA, Su SV, Wolf MC, Kohatsu L, Baum LG, Lee B. 2006. N-Glycans on Nipah virus fusion protein protect against neutralization but reduce membrane fusion and viral entry. J Virol 80:4878–4889. doi: 10.1128/JVI.80.10.4878-4889.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Biering SB, Huang A, Vu AT, Robinson LR, Bradel-Tretheway B, Choi E, Lee B, Aguilar HC. 2012. N-Glycans on the Nipah virus attachment glycoprotein modulate fusion and viral entry as they protect against antibody neutralization. J Virol 86:11991–12002. doi: 10.1128/JVI.01304-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang CC, Chen JR, Tseng YC, Hsu CH, Hung YF, Chen SW, Chen CM, Khoo KH, Cheng TJ, Cheng YS, Jan JT, Wu CY, Ma C, Wong CH. 2009. Glycans on influenza hemagglutinin affect receptor binding and immune response. Proc Natl Acad Sci U S A 106:18137–18142. doi: 10.1073/pnas.0909696106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Helle F, Vieyres G, Elkrief L, Popescu CI, Wychowski C, Descamps V, Castelain S, Roingeard P, Duverlie G, Dubuisson J. 2010. Role of N-linked glycans in the functions of hepatitis C virus envelope proteins incorporated into infectious virions. J Virol 84:11905–11915. doi: 10.1128/JVI.01548-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kong L, Giang E, Robbins JB, Stanfield RL, Burton DR, Wilson IA, Law M. 2012. Structural basis of hepatitis C virus neutralization by broadly neutralizing antibody HCV1. Proc Natl Acad Sci U S A 109:9499–9504. doi: 10.1073/pnas.1202924109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Martinez O, Tantral L, Mulherkar N, Chandran K, Basler CF. 2011. Impact of Ebola mucin-like domain on antiglycoprotein antibody responses induced by Ebola virus-like particles. J Infect Dis 204(Suppl 3):S825–S832. doi: 10.1093/infdis/jir295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dowling W, Thompson E, Badger C, Mellquist JL, Garrison AR, Smith JM, Paragas J, Hogan RJ, Schmaljohn C. 2007. Influences of glycosylation on antigenicity, immunogenicity, and protective efficacy of Ebola virus GP DNA vaccines. J Virol 81:1821–1837. doi: 10.1128/JVI.02098-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Konduru K, Bradfute SB, Jacques J, Manangeeswaran M, Nakamura S, Morshed S, Wood SC, Bavari S, Kaplan GG. 2011. Ebola virus glycoprotein Fc fusion protein confers protection against lethal challenge in vaccinated mice. Vaccine 29:2968–2977. doi: 10.1016/j.vaccine.2011.01.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Aviles J, Bello A, Wong G, Fausther-Bovendo H, Qiu X, Kobinger G. 2015. Optimization of prime-boost vaccination strategies against mouse-adapted ebolavirus in a short-term protection study. J Infect Dis 212(Suppl 2):S389–S397. doi: 10.1093/infdis/jiv175. [DOI] [PubMed] [Google Scholar]
- 36.Murin CD, Fusco ML, Bornholdt ZA, Qiu X, Olinger GG, Zeitlin L, Kobinger GP, Ward AB, Saphire EO. 2014. Structures of protective antibodies reveal sites of vulnerability on Ebola virus. Proc Natl Acad Sci U S A 111:17182–17187. doi: 10.1073/pnas.1414164111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sullivan NJ, Martin JE, Graham BS, Nabel GJ. 2009. Correlates of protective immunity for Ebola vaccines: implications for regulatory approval by the animal rule. Nat Rev Microbiol 7:393–400. doi: 10.1038/nrmicro2129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sun Y, Carrion R Jr, Ye L, Wen Z, Ro YT, Brasky K, Ticer AE, Schwegler EE, Patterson JL, Compans RW, Yang C. 2009. Protection against lethal challenge by Ebola virus-like particles produced in insect cells. Virology 383:12–21. doi: 10.1016/j.virol.2008.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Marzi A, Ebihara H, Callison J, Groseth A, Williams KJ, Geisbert TW, Feldmann H. 2011. Vesicular stomatitis virus-based Ebola vaccines with improved cross-protective efficacy. J Infect Dis 204(Suppl 3):S1066–S1074. doi: 10.1093/infdis/jir348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Phoolcharoen W, Dye JM, Kilbourne J, Piensook K, Pratt WD, Arntzen CJ, Chen Q, Mason HS, Herbst-Kralovetz MM. 2011. A nonreplicating subunit vaccine protects mice against lethal Ebola virus challenge. Proc Natl Acad Sci U S A 108:20695–20700. doi: 10.1073/pnas.1117715108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Dye JM, Herbert AS, Kuehne AI, Barth JF, Muhammad MA, Zak SE, Ortiz RA, Prugar LI, Pratt WD. 2012. Postexposure antibody prophylaxis protects nonhuman primates from filovirus disease. Proc Natl Acad Sci U S A 109:5034–5039. doi: 10.1073/pnas.1200409109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Warfield KL, Dye JM, Wells JB, Unfer RC, Holtsberg FW, Shulenin S, Vu H, Swenson DL, Bavari S, Aman MJ. 2015. Homologous and heterologous protection of nonhuman primates by Ebola and Sudan virus-like particles. PLoS One 10:e0118881. doi: 10.1371/journal.pone.0118881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Geisbert TW, Geisbert JB, Leung A, Daddario-DiCaprio KM, Hensley LE, Grolla A, Feldmann H. 2009. Single-injection vaccine protects nonhuman primates against infection with Marburg virus and three species of Ebola virus. J Virol 83:7296–7304. doi: 10.1128/JVI.00561-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Herbert AS, Kuehne AI, Barth JF, Ortiz RA, Nichols DK, Zak SE, Stonier SW, Muhammad MA, Bakken RR, Prugar LI, Olinger GG, Groebner JL, Lee JS, Pratt WD, Custer M, Kamrud KI, Smith JF, Hart MK, Dye JM. 2013. Venezuelan equine encephalitis virus replicon particle vaccine protects nonhuman primates from intramuscular and aerosol challenge with ebolavirus. J Virol 87:4952–4964. doi: 10.1128/JVI.03361-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen G, Koellhoffer JF, Zak SE, Frei JC, Liu N, Long H, Ye W, Nagar K, Pan G, Chandran K, Dye JM, Sidhu SS, Lai JR. 2014. Synthetic antibodies with a human framework that protect mice from lethal Sudan ebolavirus challenge. ACS Chem Biol 9:2263–2273. doi: 10.1021/cb5006454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Brannan JM, Froude JW, Prugar LI, Bakken RR, Zak SE, Daye SP, Wilhelmsen CE, Dye JM. 2015. Interferon alpha/beta receptor-deficient mice as a model for Ebola virus disease. J Infect Dis 212(Suppl 2):S282–S294. doi: 10.1093/infdis/jiv215. [DOI] [PubMed] [Google Scholar]
- 47.Oswald WB, Geisbert TW, Davis KJ, Geisbert JB, Sullivan NJ, Jahrling PB, Parren PW, Burton DR. 2007. Neutralizing antibody fails to impact the course of Ebola virus infection in monkeys. PLoS Pathog 3:e9. doi: 10.1371/journal.ppat.0030009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Parren PW, Geisbert TW, Maruyama T, Jahrling PB, Burton DR. 2002. Pre- and postexposure prophylaxis of Ebola virus infection in an animal model by passive transfer of a neutralizing human antibody. J Virol 76:6408–6412. doi: 10.1128/JVI.76.12.6408-6412.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jahrling PB, Geisbert TW, Geisbert JB, Swearengen JR, Bray M, Jaax NK, Huggins JW, LeDuc JW, Peters CJ. 1999. Evaluation of immune globulin and recombinant interferon-alpha2b for treatment of experimental Ebola virus infections. J Infect Dis 179(Suppl 1):S224–S234. doi: 10.1086/514310. [DOI] [PubMed] [Google Scholar]
- 50.Ye L, Lin J, Sun Y, Bennouna S, Lo M, Wu Q, Bu Z, Pulendran B, Compans RW, Yang C. 2006. Ebola virus-like particles produced in insect cells exhibit dendritic cell stimulating activity and induce neutralizing antibodies. Virology 351:260–270. doi: 10.1016/j.virol.2006.03.021. [DOI] [PubMed] [Google Scholar]
- 51.Swenson DL, Warfield KL, Kuehl K, Larsen T, Hevey MC, Schmaljohn A, Bavari S, Aman MJ. 2004. Generation of Marburg virus-like particles by co-expression of glycoprotein and matrix protein. FEMS Immunol Med Microbiol 40:27–31. doi: 10.1016/S0928-8244(03)00273-6. [DOI] [PubMed] [Google Scholar]
- 52.Warfield KL, Posten NA, Swenson DL, Olinger GG, Esposito D, Gillette WK, Hopkins RF, Costantino J, Panchal RG, Hartley JL, Aman MJ, Bavari S. 2007. Filovirus-like particles produced in insect cells: immunogenicity and protection in rodents. J Infect Dis 196(Suppl 2):S421–S429. doi: 10.1086/520612. [DOI] [PubMed] [Google Scholar]
- 53.Lo MK, Bird BH, Chattopadhyay A, Drew CP, Martin BE, Coleman JD, Rose JK, Nichol ST, Spiropoulou CF. 2014. Single-dose replication-defective VSV-based Nipah virus vaccines provide protection from lethal challenge in Syrian hamsters. Antiviral Res 101:26–29. doi: 10.1016/j.antiviral.2013.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Whitt MA, Geisbert TW, Mire CE. 2016. Single-vector, single-injection recombinant vesicular stomatitis virus vaccines against high-containment viruses. Methods Mol Biol 1403:295–311. doi: 10.1007/978-1-4939-3387-7_16. [DOI] [PubMed] [Google Scholar]
- 55.Tober R, Banki Z, Egerer L, Muik A, Behmuller S, Kreppel F, Greczmiel U, Oxenius A, von Laer D, Kimpel J. 2014. VSV-GP: a potent viral vaccine vector that boosts the immune response upon repeated applications. J Virol 88:4897–4907. doi: 10.1128/JVI.03276-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wu F, Fan X, Yue Y, Xiong S, Dong C. 2014. A vesicular stomatitis virus-based mucosal vaccine promotes dendritic cell maturation and elicits preferable immune response against coxsackievirus B3 induced viral myocarditis. Vaccine 32:3917–3926. doi: 10.1016/j.vaccine.2014.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jones SM, Stroher U, Fernando L, Qiu X, Alimonti J, Melito P, Bray M, Klenk HD, Feldmann H. 2007. Assessment of a vesicular stomatitis virus-based vaccine by use of the mouse model of Ebola virus hemorrhagic fever. J Infect Dis 196(Suppl 2):S404–S412. doi: 10.1086/520591. [DOI] [PubMed] [Google Scholar]
- 58.Wong G, Audet J, Fernando L, Fausther-Bovendo H, Alimonti JB, Kobinger GP, Qiu X. 2014. Immunization with vesicular stomatitis virus vaccine expressing the Ebola glycoprotein provides sustained long-term protection in rodents. Vaccine 32:5722–5729. doi: 10.1016/j.vaccine.2014.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Publicover J, Ramsburg E, Rose JK. 2005. A single-cycle vaccine vector based on vesicular stomatitis virus can induce immune responses comparable to those generated by a replication-competent vector. J Virol 79:13231–13238. doi: 10.1128/JVI.79.21.13231-13238.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhang M, Dong C, Xiong S. 2017. Heterologous boosting with recombinant VSV-846 in BCG-primed mice confers improved protection against Mycobacterium infection. Hum Vaccin Immunother 13:816–822. doi: 10.1080/21645515.2016.1261229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Qiu X, Fernando L, Alimonti JB, Melito PL, Feldmann F, Dick D, Stroher U, Feldmann H, Jones SM. 2009. Mucosal immunization of cynomolgus macaques with the VSVDeltaG/ZEBOVGP vaccine stimulates strong ebola GP-specific immune responses. PLoS One 4:e5547. doi: 10.1371/journal.pone.0005547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Schmaljohn A, Lewis GK. 2016. Cell-targeting antibodies in immunity to Ebola. Pathog Dis 74:ftw021. doi: 10.1093/femspd/ftw021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hashiguchi T, Fusco ML, Bornholdt ZA, Lee JE, Flyak AI, Matsuoka R, Kohda D, Yanagi Y, Hammel M, Crowe JE Jr, Saphire EO. 2015. Structural basis for Marburg virus neutralization by a cross-reactive human antibody. Cell 160:904–912. doi: 10.1016/j.cell.2015.01.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Beniac DR, Booth TF. 2017. Structure of the Ebola virus glycoprotein spike within the virion envelope at 11 A resolution. Sci Rep 7:46374. doi: 10.1038/srep46374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hensley LE, Mulangu S, Asiedu C, Johnson J, Honko AN, Stanley D, Fabozzi G, Nichol ST, Ksiazek TG, Rollin PE, Wahl-Jensen V, Bailey M, Jahrling PB, Roederer M, Koup RA, Sullivan NJ. 2010. Demonstration of cross-protective vaccine immunity against an emerging pathogenic Ebolavirus species. PLoS Pathog 6:e1000904. doi: 10.1371/journal.ppat.1000904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mire CE, Geisbert JB, Marzi A, Agans KN, Feldmann H, Geisbert TW. 2013. Vesicular stomatitis virus-based vaccines protect nonhuman primates against Bundibugyo ebolavirus. PLoS Negl Trop Dis 7:e2600. doi: 10.1371/journal.pntd.0002600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mire CE, Geisbert JB, Versteeg KM, Mamaeva N, Agans KN, Geisbert TW, Connor JH. 2015. A single-vector, single-injection trivalent filovirus vaccine: proof of concept study in outbred guinea pigs. J Infect Dis 212(Suppl 2):S384–S388. doi: 10.1093/infdis/jiv126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lee JE, Fusco ML, Abelson DM, Hessell AJ, Burton DR, Saphire EO. 2009. Techniques and tactics used in determining the structure of the trimeric ebolavirus glycoprotein. Acta Crystallogr D Biol Crystallogr 65:1162–1180. doi: 10.1107/S0907444909032314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kelley LA, Sternberg MJ. 2009. Protein structure prediction on the Web: a case study using the Phyre server. Nat Protoc 4:363–371. doi: 10.1038/nprot.2009.2. [DOI] [PubMed] [Google Scholar]
- 70.Kondratowicz AS, Lennemann NJ, Sinn PL, Davey RA, Hunt CL, Moller-Tank S, Meyerholz DK, Rennert P, Mullins RF, Brindley M, Sandersfeld LM, Quinn K, Weller M, McCray PB Jr, Chiorini J, Maury W. 2011. T-cell immunoglobulin and mucin domain 1 (TIM-1) is a receptor for Zaire Ebolavirus and Lake Victoria Marburgvirus. Proc Natl Acad Sci U S A 108:8426–8431. doi: 10.1073/pnas.1019030108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Takada A, Robison C, Goto H, Sanchez A, Murti KG, Whitt MA, Kawaoka Y. 1997. A system for functional analysis of Ebola virus glycoprotein. Proc Natl Acad Sci U S A 94:14764–14769. doi: 10.1073/pnas.94.26.14764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Holtsberg FW, Shulenin S, Vu H, Howell KA, Patel SJ, Gunn B, Karim M, Lai JR, Frei JC, Nyakatura EK, Zeitlin L, Douglas R, Fusco ML, Froude JW, Saphire EO, Herbert AS, Wirchnianski AS, Lear-Rooney CM, Alter G, Dye JM, Glass PJ, Warfield KL, Aman MJ. 2015. Pan-ebolavirus and pan-filovirus mouse monoclonal antibodies: protection against Ebola and Sudan viruses. J Virol 90:266–278. doi: 10.1128/JVI.02171-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lyles DS, Puddington L, McCreedy BJ Jr. 1988. Vesicular stomatitis virus M protein in the nuclei of infected cells. J Virol 62:4387–4392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.National Research Council. 2011. Guide for the care and use of laboratory animals, 8th ed National Academies Press, Washington, DC. [Google Scholar]
- 75.Brindley MA, Hunt CL, Kondratowicz AS, Bowman J, Sinn PL, McCray PB Jr, Quinn K, Weller ML, Chiorini JA, Maury W. 2011. Tyrosine kinase receptor Axl enhances entry of Zaire ebolavirus without direct interactions with the viral glycoprotein. Virology 415:83–94. doi: 10.1016/j.virol.2011.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hunt CL, Kolokoltsov AA, Davey RA, Maury W. 2011. The tyro3 receptor kinase axl enhances macropinocytosis of Zaire ebolavirus. J Virol 85:334–347. doi: 10.1128/JVI.01278-09. [DOI] [PMC free article] [PubMed] [Google Scholar]