Abstract
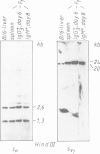
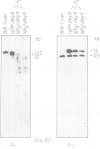
Mouse B lymphocytes can be activated polyclonally by bacterial lipopolysaccharide (LPS) to differentiate into plasmablasts. Within several days many cells perform immunoglobulin (Ig) class switching in vitro. We have purified LPS blasts expressing IgM or only IgG3 on the cell surface and analysed the DNA of these cells by Southern hybridisation blotting to detect rearrangement or deletion of CH genes. Quantitative evaluation of the Southern blots suggests that populations of surface IgG3+ (sIgG3+) cells from 6-day and sIgM+ cells from 8-day-old cultures contain only about half as many Cmu genes as spleen cells. Cmu deletion is nearly complete in populations of sIgG3+ cells from 9-day-old cultures. Therefore, upon stimulation with LPS, within a few days Cmu is deleted in most sIgG3+ cells from both chromosomes.
Full text
PDF
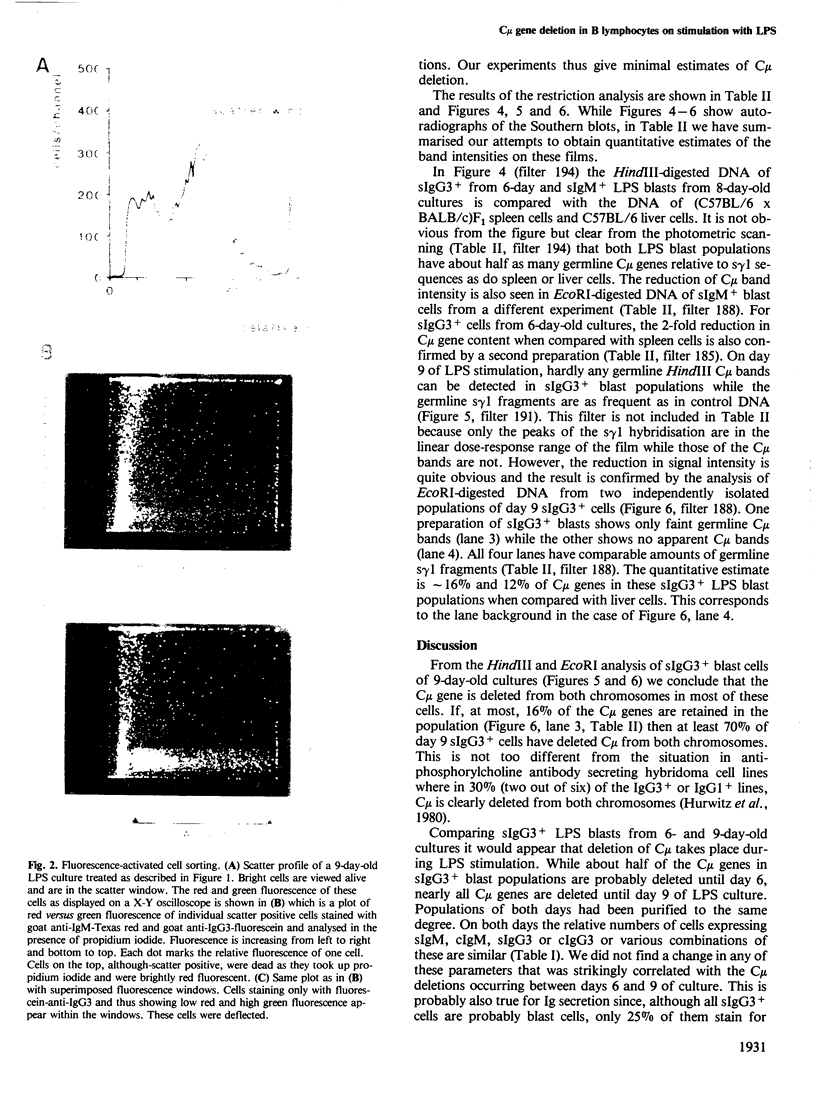




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andersson J., Coutinho A., Melchers F. The switch from IgM to IgG secretion in single mitogen-stimulated B-cell clones. J Exp Med. 1978 Jun 1;147(6):1744–1754. doi: 10.1084/jem.147.6.1744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bothwell A. L., Paskind M., Reth M., Imanishi-Kari T., Rajewsky K., Baltimore D. Heavy chain variable region contribution to the NPb family of antibodies: somatic mutation evident in a gamma 2a variable region. Cell. 1981 Jun;24(3):625–637. doi: 10.1016/0092-8674(81)90089-1. [DOI] [PubMed] [Google Scholar]
- Boyle W. An extension of the 51Cr-release assay for the estimation of mouse cytotoxins. Transplantation. 1968 Sep;6(6):761–764. doi: 10.1097/00007890-196809000-00002. [DOI] [PubMed] [Google Scholar]
- Coleclough C., Cooper D., Perry R. P. Rearrangement of immunoglobulin heavy chain genes during B-lymphocyte development as revealed by studies of mouse plasmacytoma cells. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1422–1426. doi: 10.1073/pnas.77.3.1422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coleclough C., Perry R. P., Karjalainen K., Weigert M. Aberrant rearrangements contribute significantly to the allelic exclusion of immunoglobulin gene expression. Nature. 1981 Apr 2;290(5805):372–378. doi: 10.1038/290372a0. [DOI] [PubMed] [Google Scholar]
- Cooper M. D., Kearney J. F., Lydyard P. M., Grossi C. E., Lawton A. R. Studies of generation of B-cell diversity in mouse, man, and chicken. Cold Spring Harb Symp Quant Biol. 1977;41(Pt 1):139–145. doi: 10.1101/sqb.1977.041.01.018. [DOI] [PubMed] [Google Scholar]
- Cory S., Adams J. M. Deletions are associated with somatic rearrangement of immunoglobulin heavy chain genes. Cell. 1980 Jan;19(1):37–51. doi: 10.1016/0092-8674(80)90386-4. [DOI] [PubMed] [Google Scholar]
- Cory S., Jackson J., Adams J. M. Deletions in the constant region locus can account for switches in immunoglobulin heavy chain expression. Nature. 1980 Jun 12;285(5765):450–456. doi: 10.1038/285450a0. [DOI] [PubMed] [Google Scholar]
- Cory S., Webb E., Gough J., Adams J. M. Recombination events near the immunoglobulin Cmu gene join variable and constant region genes, switch heavy-chain expression, or inactivate the locus. Biochemistry. 1981 Apr 28;20(9):2662–2671. doi: 10.1021/bi00512a047. [DOI] [PubMed] [Google Scholar]
- Coutinho A., Forni L. Intraclonal diversification in immunoglobulin isotype secretion: an analysis of switch probabilities. EMBO J. 1982;1(10):1251–1257. doi: 10.1002/j.1460-2075.1982.tb00021.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis M. M., Calame K., Early P. W., Livant D. L., Joho R., Weissman I. L., Hood L. An immunoglobulin heavy-chain gene is formed by at least two recombinational events. Nature. 1980 Feb 21;283(5749):733–739. doi: 10.1038/283733a0. [DOI] [PubMed] [Google Scholar]
- Davis M. M., Kim S. K., Hood L. E. DNA sequences mediating class switching in alpha-immunoglobulins. Science. 1980 Sep 19;209(4463):1360–1365. doi: 10.1126/science.6774415. [DOI] [PubMed] [Google Scholar]
- Early P., Huang H., Davis M., Calame K., Hood L. An immunoglobulin heavy chain variable region gene is generated from three segments of DNA: VH, D and JH. Cell. 1980 Apr;19(4):981–992. doi: 10.1016/0092-8674(80)90089-6. [DOI] [PubMed] [Google Scholar]
- Hurwitz J. L., Cebra J. J. Rearrangements between the immunoglobulin heavy chain gene JH and C mu regions accompany normal B lymphocyte differentiation in vitro. Nature. 1982 Oct 21;299(5885):742–744. doi: 10.1038/299742a0. [DOI] [PubMed] [Google Scholar]
- Hurwitz J. L., Coleclough C., Cebra J. J. CH gene rearrangements in IgM-bearing B cells and in the normal splenic DNA component of hybridomas making different isotypes of antibody. Cell. 1980 Nov;22(2 Pt 2):349–359. doi: 10.1016/0092-8674(80)90345-1. [DOI] [PubMed] [Google Scholar]
- Kataoka T., Kawakami T., Takahashi N., Honjo T. Rearrangement of immunoglobulin gamma 1-chain gene and mechanism for heavy-chain class switch. Proc Natl Acad Sci U S A. 1980 Feb;77(2):919–923. doi: 10.1073/pnas.77.2.919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kearney J. F., Cooper M. D., Lawton A. R. B cell differentiation induced by lipopolysaccharide. IV. Development of immunoglobulin class restriction in precursors of IgG-synthesizing cells. J Immunol. 1976 Nov;117(5 Pt 1):1567–1572. [PubMed] [Google Scholar]
- Kearney J. F., Klein J., Bockman D. E., Cooper M. D., Lawton A. R. B cell differentiation induced by lipopolysaccharide. V. Suppression of plasma cell maturation by anti-mu: mode of action and characteristics of suppressed cells. J Immunol. 1978 Jan;120(1):158–166. [PubMed] [Google Scholar]
- Kearney J. F., Lawton A. R. B lymphocyte differentiation induced by lipopolysaccharide. I. Generation of cells synthesizing four major immunoglobulin classes. J Immunol. 1975 Sep;115(3):671–676. [PubMed] [Google Scholar]
- Loken M. R., Herzenber L. A. Analysis of cell populations with a fluorescence-activated cell sorter. Ann N Y Acad Sci. 1975 Jun 30;254:163–171. doi: 10.1111/j.1749-6632.1975.tb29166.x. [DOI] [PubMed] [Google Scholar]
- Maki R., Traunecker A., Sakano H., Roeder W., Tonegawa S. Exon shuffling generates an immunoglobulin heavy chain gene. Proc Natl Acad Sci U S A. 1980 Apr;77(4):2138–2142. doi: 10.1073/pnas.77.4.2138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcu K. B., Banerji J., Penncavage N. A., Lang R., Arnheim N. 5' flanking region of immunoglobulin heavy chain constant region genes displays length heterogeneity in germlines of inbred mouse strains. Cell. 1980 Nov;22(1 Pt 1):187–196. doi: 10.1016/0092-8674(80)90167-1. [DOI] [PubMed] [Google Scholar]
- Melchers F., Von Boehmer H., Phillips R. A. B-lymphocyte subpopulations in the mouse. Organ distribution and ontogeny of immunoglobulin-synthesizing and of mitogen-sensitive cells. Transplant Rev. 1975;25:26–58. doi: 10.1111/j.1600-065x.1975.tb00725.x. [DOI] [PubMed] [Google Scholar]
- Mohr R., Krawinkel U. Helper T-cell kinetics and investigation of antigen receptor expression on early and memory T-helper cells. Immunology. 1976 Aug;31(2):249–255. [PMC free article] [PubMed] [Google Scholar]
- Pernis B., Forni L., Luzzati A. L. Synthesis of multiple immunoglobulin classes by single lymphocytes. Cold Spring Harb Symp Quant Biol. 1977;41(Pt 1):175–183. doi: 10.1101/sqb.1977.041.01.023. [DOI] [PubMed] [Google Scholar]
- Rabbitts T. H., Forster A., Dunnick W., Bentley D. L. The role of gene deletion in the immunoglobulin heavy chain switch. Nature. 1980 Jan 24;283(5745):351–356. doi: 10.1038/283351a0. [DOI] [PubMed] [Google Scholar]
- Raff M. C. Development and modulation of B lymphocytes: studies on newly formed B cells and their putative precursors in the hemopoietic tissues of mice. Cold Spring Harb Symp Quant Biol. 1977;41(Pt 1):159–162. doi: 10.1101/sqb.1977.041.01.020. [DOI] [PubMed] [Google Scholar]
- Sakano H., Maki R., Kurosawa Y., Roeder W., Tonegawa S. Two types of somatic recombination are necessary for the generation of complete immunoglobulin heavy-chain genes. Nature. 1980 Aug 14;286(5774):676–683. doi: 10.1038/286676a0. [DOI] [PubMed] [Google Scholar]
- Shimizu A., Takahashi N., Yaoita Y., Honjo T. Organization of the constant-region gene family of the mouse immunoglobulin heavy chain. Cell. 1982 Mar;28(3):499–506. doi: 10.1016/0092-8674(82)90204-5. [DOI] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Titus J. A., Haugland R., Sharrow S. O., Segal D. M. Texas Red, a hydrophilic, red-emitting fluorophore for use with fluorescein in dual parameter flow microfluorometric and fluorescence microscopic studies. J Immunol Methods. 1982;50(2):193–204. doi: 10.1016/0022-1759(82)90225-3. [DOI] [PubMed] [Google Scholar]
- Wahl G. M., Stern M., Stark G. R. Efficient transfer of large DNA fragments from agarose gels to diazobenzyloxymethyl-paper and rapid hybridization by using dextran sulfate. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3683–3687. doi: 10.1073/pnas.76.8.3683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaoita Y., Honjo T. Deletion of immunoglobulin heavy chain genes from expressed allelic chromosome. Nature. 1980 Aug 28;286(5776):850–853. doi: 10.1038/286850a0. [DOI] [PubMed] [Google Scholar]
- Yaoita Y., Kumagai Y., Okumura K., Honjo T. Expression of lymphocyte surface IgE does not require switch recombination. Nature. 1982 Jun 24;297(5868):697–699. doi: 10.1038/297697a0. [DOI] [PubMed] [Google Scholar]
- Yeh C. J., Hsi B. L., Faulk W. P. Propidium iodide as a nuclear marker in immunofluorescence. II. Use with cellular identification and viability studies. J Immunol Methods. 1981;43(3):269–275. doi: 10.1016/0022-1759(81)90174-5. [DOI] [PubMed] [Google Scholar]