Abstract
Non-thermal atmospheric plasma (NTP) has shown its selective anticancer effects in many types of tumors in vitro and one of the main mechanisms is that the different increase of intracellular ROS in cancer and homologous normal cells. In this study, we report that NTP treatment reduces the proliferation in triple negative breast cancer (TNBC) and normal cell lines. Simultaneously, STAT3 pathway is inhibited by NTP effects. However, it is observed that normal cells MCF10A are more sensitive to ROS toxicity induced by NTP than cancer cells MDA-MB-231. When 5 mM of ROS inhibitor N-acetyl cysteine (NAC) is employed in NTP treatments, the proliferation of normal breast cells MCF10A recovers. Meanwhile, NTP effects remain significant inhibition of MDA-MB-231 cells. Our results further reveal that NTP can induce apoptosis in MDA-MB-231 cells through inhibiting interleukin-6 receptor (IL-6R) pathway. Moreover, the mechanism of NTP anti-cancer selectivity relates to constantly HER2/Akt activation induced by NTP especially in MCF10A cells but not in MDA-MB-231 cells. Therefore, these two different cell signaling pathways induced by NTP treatments in TNBC and homologous normal cells make NTP becoming a potential tool in future therapy.
Introduction
Breast cancer is the cause of 23% of the total cancer cases and 14% of cancer deaths, and is the most frequently diagnosed cancers that causes the prominent number cancer death among females1. Breast cancer subtypes are ordinarily identified by the presence of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor type 2 (HER2). Triple-negative (TN) patients lack all three. While comparing to other breast cancer subtypes, triple-negative breast cancers (TNBC) are higher frequency and are more aggressive with high potential to metastasize and more lack of targeted treatment options. Chemotherapy is currently the only adjunctive therapy2; although triple-negative patients, when regarded as a group, have a worse long-term survival after chemotherapy than patients with other breast cancer subtypes3, 4. Toxicity and side effects come along with chemotherapy, while obtained from chemo resistance tumor cells can still maintain viability after chemotherapy5. For many women, benefit of chemotherapy is uncertain. Therefore establishing new therapies with little side effects for TNBC is imminent.
Physical plasmas are composed of reactive atoms, molecules, ions and radicals. While non-thermal plasma can allow strong non-equilibrium chemistry because of its ions and neutral species remains cold6. Also NTP can produce a mixture of highly-reactive chemical species which play important roles in cell processes7. Reactive oxygen and nitrogen species (RONS) induced by NTP that enter adjacent aqueous liquid phases are the key mechanism known to react with important biomolecules8. At present, NTP has become as a novel tool in some biomedical areas, such as sterilization, antifungal treatments, blood coagulation, dental care, wound healing and cosmetics targeted cell/tissue removal9, 10. NTP treatment has been proven effectively in inducing apoptosis in vitro of various tumor types and inhibiting tumor growth in vivo 10. Moreover, NTP effects on cells were dose dependent that low doses of plasma can stop cancer cells proliferation and high doses result in cell apoptosis and necrosis11.
Results of current studies show that NTP can induce apoptosis in vitro in various cancer types, from lung cancer, glioblastoma, prostate cancer, melanoma and cervical cancer to breast cancer12–17. Previous studies suggest that RONS generated by NTP plays an important role in cell death. In lung cancer, non-thermal plasma induced apoptotic cell death through mitochondrial damage by generating RONS in culture media18. In addition, cold atmospheric plasma can induce more intracellular RONS in A549 cells than normal cells19. ROS formation in cell culture induces high levels of DNA damage after plasma exposure, and leads to necrosis both in primary prostate epithelial cells and prostate cancer cells14. In cervical cancer Hela cells, H2O2 in plasma-treated medium is the main course of inactivation of cell viability20. Earlier study also shows that cold plasma can significantly reduce tumor size through deregulating ROS related genes21.
Recent researches are tending to focus on revealing the molecular mechanisms underlying the changes of cancer cells activity by non-thermal plasma. For example, NTP can produce exogenous ROS in cell medium that act as the elementary active species which induce cancer cell death through activating DNA damage22. In head and neck cancer, NTP induced apoptotic cell death by involving in MAPK-mediated mitochondrial ROS signaling pathways23. Inhibition of the migration and invasion in HeLa cells was induced by NTP via significant depressing MMP-9 expression in ERK1/2 and JNK signaling pathways24. NTP also led to sub-G1 arrest in oral cavity squamous cell carcinoma cells and the arrest was related with DNA damage and the ATM/p53 signaling pathways25. Furthermore, in the human glioma cell line U373MG, ROS-, JNK- and caspase-independent mechanism of cell death by NTP treatment26.
The rise of ROS induced by NTP treatment in vitro is the main cause of the cell death. It was reported that hydrogen peroxide (H2O2) generated by plasma and concentration-matched H2O2 both resulted in significant rise of intracellular ROS in T helper cells and monocytes, and reduction of cell growth27. Former researchers report that air plasma jet treatment induced mitochondrial-mediated apoptosis in HeLa cells was mainly caused by ROS generation28. Accumulation of ROS and RNS in HepG2 cells under NTP treatment was reported, because of these effects endoplasmic reticulum stress mediated apoptosis and cellular dysfunction were observed29. The exposure of A549 cells to plasma-activated medium (PAM) significantly induced the generation of intracellular ROS and causes caspase-independent apoptosis through involving the mitochondrial–nuclear network30. PAM effects on the proliferation inhibition and DNA damage in HCT-116 multicellular tumor spheroid were also reported, while H2O2 induced by PAM plays an important role in observed genotoxic effect31. H2O2 in PAM and/or •OH generated in association with iron ions also involve in the mitochondrial-nuclear network and result in A549 cell injury32. For the application of plasma-stimulated medium (PSM) in future cancer therapy, utilizing stabilizing strategies like storing PSM at 8 °C or −25 °C and adding 3-Nitro-L-tyrosine into DMEM could weaken the degradation plasma-originated reactive species33. Recent researchers reported that enhanced level of ROS in U937 cells induced by plasma treatment caused a compromised redox status obvious from reduction of GSH/GSSG ratios, rise of NADP+/NADPH levels and total endogenous antioxidant activity decreasing34.
NTP selective effects on carcinoma and normal cells were reported and were classified into three groups according to NTP effects on cancer cells: strong selectivity, weak selectivity, and negative selectivity35. It was observed that intracellular ROS rising significantly in cancer cells A549 and increased slightly in normal cells HEK293T and BEAS-2B after plasma exposure. Meanwhile, cellular viability of HEK293T and BEAS-2B cells did not show significant change compared to A549 cells36. NTP shows strong and weak selective anticancer capacity to most studied cancer cell lines, while only papilloma cells and prostate cancer cells show high resistance to NTP. It was point out that differentiation between normal cells and cancer cells may have correlation with the NTP selectivity35. However, NTP effects on the death of normal cells were often observed in some cases and as long as the cells were under NTP treatment, this phenomenon could not be avoided. For example, plasma-induced necrosis and autophagy in primary prostate epithelial cells were recorded14. Also, apoptotic cell death in mammalian breast epithelial cells induced by NTP treatment was reported37. In addition, atmospheric pressure plasma was observed to suppress cell proliferation of human aortic endothelial cells38. ROS generated by atmospheric plasma may cause the death of normal cell in these cases, because of its proverbial damage to DNA39. Furthermore, cancer cells from different tissues have quite distinct response to NTP treatment, so it is more meaningful and important to study NTP selective anticancer capacity on normal cells and homologous cancer cells35.
Here we mainly paid attention in the NTP effects on breast carcinoma and normal cells. Previous research reported that migration and invasion of breast carcinoma MDA-MB-231 cells can be significantly inhibited by cold atmospheric plasma treatments40. Also breast cancer cells (MCF-7 and MDA-MB-231) with quite distinct range of cell confluences showed different response to the same NTP treatment time dose41. Additionally, non-thermal plasma treatment can induce apoptosis and the formation of single-stranded DNA breaks of mammalian breast epithelial cells MCF10A37 and many cancer cells due to the formation of intracellular ROS. It was reported that U373MG cells demonstrate a higher resistance against H2O2, but NTP treatments still show significantly killing effects on U373MG cells26. However, NTP selective anticancer capacity on TNBC cells has not been reported and the mechanism of NTP effects on breast cancer cells and homologous normal cells is unclear. In this study, we intend to find out the signaling pathways involved in NTP selective killing effects and avoid ROS toxicity induced by NTP to TN breast normal cells MCF10A. We investigated the cell viability and intracellular ROS change in MDA-MB-453, MDA-MB-231 and MCF10A cells under NTP treatments. Moreover, in this work we reveal the different signaling pathways regulated in MDA-MB-231 and MCF10A cells under NTP treatment.
Results
NTP treatment reduces the proliferation of both TN breast normal and carcinoma cell lines
After 48 hours, following the NTP treatment, the percentage of viable cells was quantified. Both two types of TNBC cell lines MDA-MB-453 and MDA-MB-231 observed a reduction of proliferation (Fig. 1A and B). The results showed that at 60 s NTP treatment MDA-MB-453 had no significant reduction comparing to 0 s treatment (Fig. 1C). Furthermore, at 120 s NTP treatment MDA-MB-453 percentage of viable cells was reduced to <80%, whereas MDA-MB-231 was reduced to <50% (Fig. 1C). In addition, a significant reduction in proliferation was observed in normal TN breast cell lines MCF10A (Fig. 1B). From 60 s to 120 s NTP treatment the percentage of viable cells of MCF10A was all reduced significantly to <40% (Fig. 1C). Morphology of MDA-MB-231 and MCF10A is different from MDA-MB-453 (Fig. 1B), indicating that this difference might be related to the cause of different responses. Previous study has reported that the plasma effects on the cell shape and membrane as well as cell viability points out the complex interaction between plasma and cell membrane surface42.
Figure 1.
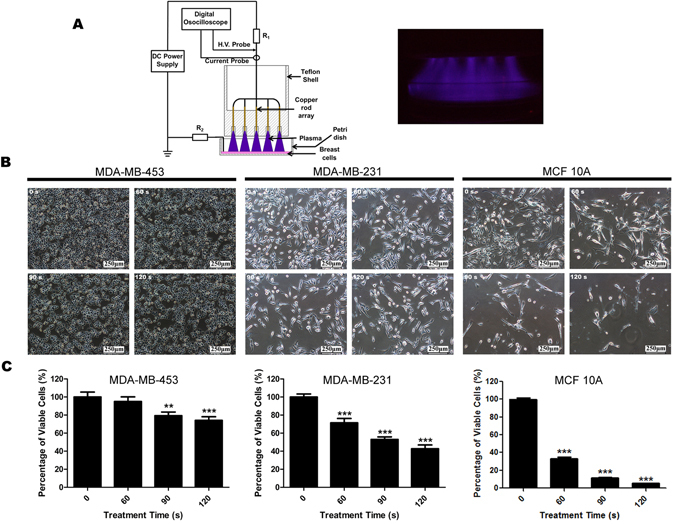
NTP treatment reduces proliferation of MDA-MB-453, MDA-MB-231 and MCF10A cells in a time dose dependent inhibition. (A) Schematic of the non-thermal atmospheric plasma set-up utilised in this study, and a photograph of the air plasma. All cells were exposed to NTP for 0, 60, 90 or 120 s. Forty-eight hours later, cell images were captured with 100 × magnification (B) and cell numbers of each dish were counted by an automatic analyzer CountStar (C). All experiments were replicated a minimum of three times. Data are presented as means ± S.D. and statistical analysis was carried out using one-way ANOVA with Tukey’s multiple comparison test (*p < 0.05, **p < 0.01, ***p < 0.001 versus control).
MDA-MB-453, MDA-MB-231 and MCF10A cells show different response under NTP treatment with or without ROS scavenger NAC
According to data shown in Fig. 1, normal cells MCF10A was observed more sensitive to the cytotoxicity induced NTP treatment compared to cancer cells. Because ROS is the main toxicity species induced by NTP, we intend to use ROS scavenger to protect this cytotoxicity effects on MCF10A cells. To determine the role of ROS induced by NTP treatment on TN breast cell lines, all cells were pre-treated for 1 h with 5 mM NAC and then washed before NTP treatment (PRE NAC) as referred to former studies26, 43. In addition, we want to use NAC as a long term co-drug for protecting normal cells from ROS toxicity; cells were also post-treated with 5 mM NAC immediately after NTP treatment (POST NAC) until harvested. After further 48 h interesting results were observed, through PRE NAC and POST NAC treatments counteract part of the 90 s NTP killing effects but NTP remaining significant inhibition of MDA-MB-231 cells (Fig. 2B). MDA-MB-453 cells gained a recovery in proliferation under 90 s and 120 s NTP treatments with the help of both PRE NAC and POST NAC treatments (Fig. 2A). On the other hand, PRE NAC and POST NAC treatments can significantly counteract 60 s, 90 s and 120 s NTP killing effects in MCF10A cells (Fig. 2C). It is indicated that normal cells MCF10A show more resistance to the NTP affection especially with POST NAC treatments. We then analyzed the rise of intracellular ROS in all cell lines after 120 s NTP treatment. All cell lines showed a significantly rise of intracellular ROS after 120 s NTP treatment (Fig. 2). With the help of NAC, intracellular ROS of MDA-MB-453 cells remained unchanged compared to control group after NTP treatments (Fig. 2A). However in MCF10A cells, POST NAC group performed more protection compared to PRE-NAC group (Fig. 2C). It is also indicated that ROS plays the key role in the cause of cell death in MCF10A and MDA-MB-453. Interestingly, strong inhibition in MDA-MB-231 cells was observed under any designed NTP treatment in spite of the fact that the rise of intracellular ROS was significantly different between NAC treated group and NTP group (Fig. 2B). POST NAC group of MCF10A cells showed more tolerable to NTP treatment than NTP treatment without NAC (Fig. 2C). On the other hand, NTP anti-cancer effects on TNBC MDA-MB-231 cells were observed uncorrelated with rise of intracellular ROS (Fig. 2B). It is also showed that post-treated NAC group can effectively protect cells form intracellular ROS cytotoxicity and recover the proliferation of MCF10A cells. These findings lead us to focus on the NTP selective effects by using NAC and revealing the mechanism that causes the divergent response between MDA-MB-231 and MCF10A.
Figure 2.
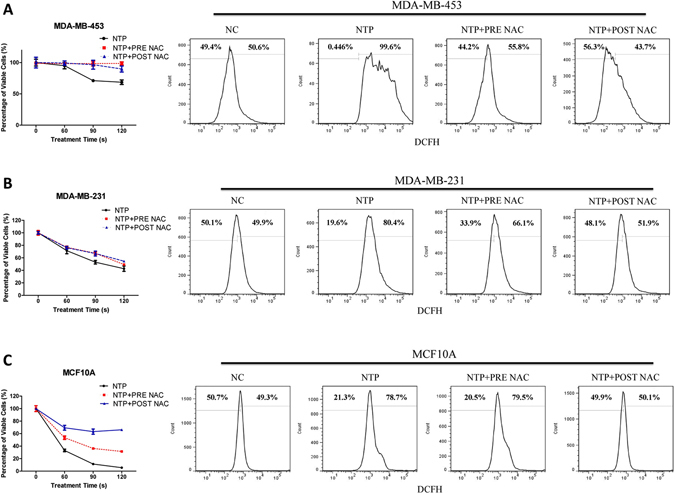
Effects of NTP treatment with or without ROS inhibitor NAC on MDA-MB-453, MDA-MB-231 and MCF10A cells proliferation. Cells were treated with (PRE NAC and POST NAC) or without NAC (NTP) exposed to NTP for 0, 60, 90 or 120 s. Forty-eight hours later, cell numbers of each dish were counted by an automatic analyzer CountStar. ROS fluorescence was analyzed by FACSVerse flow cytometer, 1 h after 120 s NTP treatment. Cells were treated with: Negative Control (NC), 120 s NTP treatment (NTP), pretreat with 5 mM NAC before 120 s NTP treatment (NTP + PRE NAC), post-treat with 5 mM NAC immediately after 120 s NTP treatment (NTP + POST NAC). For improved illustration, a guidance ratio (compared with NC group) has been indicated through histograms. All experiments were replicated a minimum of three times.
NTP treatment can induce the inhibition of activation of signal transducer and activator of transcription 3 (Stat3) in MDA-MB-231 and MCF10A cells
Constitutive activation of Stat3 protein (tyrosine-phosphorylated Stat3) has been observed in numerous kinds of tumors, including breast cancer. Previous researches have revealed that Stat3 plays an important role in oncogenesis, including enhancement of cell proliferation, inhibition of apoptosis and induction of angiogenesis44, 45.
After NTP treatment or post-treatment with ROS scavenger NAC, the expression of tyrosine-phosphorylated Stat3 (pSTAT3) in MDA-MB-231 (Fig. 3A and Supplementary Figure 1A) and MCF10A (Fig. 3B and Supplementary Figure 1B) cells was inhibited. The same mechanism was also confirmed in MDA-MB-453 (Supplementary Figure 2). This indicates that reduction of proliferation in MDA-MB-231 and MCF10A is correlated with the inhibition of activation of Stat3 protein by NTP treatment.
Figure 3.
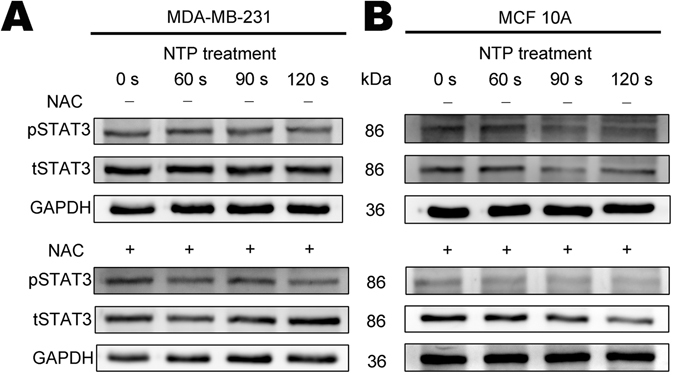
Effects of NTP on the expression of pSTAT3 and tSTAT3 in MDA-MB-231 and MCF10A cells. All cells were exposed to NTP for 0, 60, 90 or 120 s. After forty-eight hours post-treated with 5 mM N-acetyl cysteine (NAC+) or without (NAC−), proteins from total cell lysates were harvested. The expressions of pSTAT3 and STAT3 in MDA-MB-231 (A) and MCF10A (B) cells were detected by Western blot analysis. GAPDH was taken as a loading control throughout. The gels/blots were processed under the same experimental condition. Cropped gels/blots are displayed and full-length blots/gels are presented in Supplementary Figure 1.
The ROS affects the proliferation of MDA-MB-231 and MCF10A cells in a different way
To reveal the mechanism that makes MDA-MB-231 and MCF10A behavior differently under NTP treatment and to confirm the role of ROS in NTP treatments, catalase as extracellular ROS scavenger was employed in 120 s NTP treatments firstly. Catalase was adding into medium just before 0 s and 120 s NTP treatments. MDA-MB-453 cells gained resistance to NTP treatment with catalase (Fig. 4A). MCF10A cells got a recovering in proliferation (Fig. 4C). Interestingly, MDA-MB-231 still remained sensitive to NTP treatment (Fig. 4B) and which was similar to PRE NAC and POST NAC treatments (Fig. 2B). On the other hand, MCF10A cells with POST NAC treatment (Fig. 2C) can counteract more NTP killing effects than catalase treatment (Fig. 4C).
Figure 4.
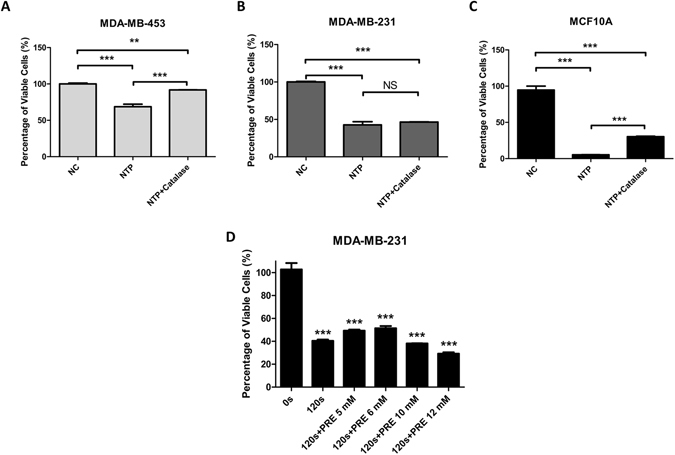
Effects of 120 s NTP treatments with extracellular and intracellular ROS inhibitor on cells’ proliferation. MDA-MB-453 (A), MDA-MB-231 (B) and MCF10A (C) cells were treated: Negative Control (NC), 120 s NTP treatment (NTP) without or with 500 U/ml catalase (NTP + Catalase). MDA-MB-231 cells (D) were treated: exposed to NTP for 0 and 120 s, pretreat with 5, 6, 10 and 12 mM NAC before 120 s NTP treatment. After forty-eight hours, cell numbers of each dish were counted by an automatic analyzer CountStar. All experiments were replicated a minimum of three times. Data are presented as means ± S.D. and statistical analysis was carried out using one-way ANOVA with Tukey’s multiple comparison test (NS = No significance, *p < 0.05, **p < 0.01, ***p < 0.001).
According to Fig. 2B results, PRE NAC treatments would counter part of intracellular ROS cytotoxicity by 120 s NTP treatment (from about 42 to 49 percent of viable cells). Furthermore, we pre-treated MDA-MB-231 cells with higher concentration of NAC to confirm that MDA-MB-231 cells are resistant to 120 s NTP treatments’ intracellular ROS cytotoxicity but not killing effects. It was showed that 10 mM and 12 mM NAC were too high to cause cytotoxicity to cells and 6 mM NAC still cannot counteract most part of the killing effect induced by NTP (Fig. 4D). It also observed that MDA-MB-231 cells pre-treated with 6 mM NAC results in about 51 percent of viable cells after 120 s NTP treatment, which is similar to 5 mM NAC results (49 percent of viable cells). Results of Fig. 4B and D showed that NTP anti-cancer capacity beyond ROS cytotoxicity is still effective on MDA-MB-231 cells.
Additionally, H2O2 multi-dose experiments were examined. Under high concentration of H2O2 treatments, the proliferation of both MDA-MB-231 and MCF10A cells observed a significantly decreasing (Fig. 5A and B). We also examined H2O2 multi-dose experiments with 5 mM NAC and it was showed that 5 mM NAC could counteract part of 50 μM and 100 μM H2O2 cytotoxicity but not when the concentration of H2O2 reaches 150 μM (Supplementary Figure 3A). As we measured the H2O2 concentration of 120 s NTP treated medium is 185 μM (Supplementary Figure 3B). And it was observed that all three cell lines treated with catalase can counteract 185 μM H2O2 cytotoxicity. Meanwhile 5 mM NAC could not protect cells from cytotoxicity induced by such high H2O2 concentration (Supplementary Figure 3C), although 5 mM NAC would consume most H2O2 in 3 hours and eliminate all H2O2 in 24 hours (Supplementary Figure 3D). Interestingly in MDA-MB-231 cells, low concentration and high concentration of H2O2 can affect the proliferation in opposite directions (Fig. 5A). MCF10A cells showed no resistance to H2O2 treatment (Fig. 5B and Supplementary Figure 3) just similar to the situation under NTP treatment (Fig. 1B).
Figure 5.
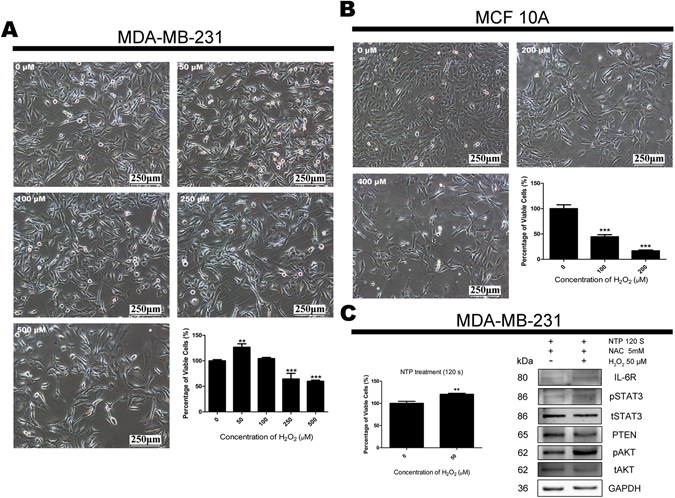
Effects of H2O2 on the proliferation of MDA-MB-231 and MCF10A cells. MCF10A (A) and MDA-MB-231 (B) cells were incubated with multi-dose H2O2. Forty-eight hours later, cell images were captured with 100 × magnification and cell numbers of each dish were counted by an automatic analyzer CountStar. All experiments were replicated a minimum of three times. Data are presented as means ± S.D. and statistical analysis was carried out using one-way ANOVA with Tukey’s multiple comparison test. MDA-MB-231 cells were post-treated with 5 mM NAC and 0/50 μM H2O2 immediately after 120 s NTP treatment (C). Cell numbers of each dish were counted by an automatic analyzer CountStar and proteins from total cell lysates were harvested after forty-eight hours. Data are presented as means ± S.D. for three independent experiments and analyzed by Student’s t-test. *p < 0.05, **p < 0.01, ***p < 0.001 versus control. The expressions of IL-6R, pSTAT3, tSTAT3, PTEN, pAkt and tAkt were detected by Western blot analysis. GAPDH was taken as a loading control throughout. The gels/blots were processed under the same experimental condition. Cropped gels/blots are displayed and full-length blots/gels are presented in Supplementary Figure 4.
Taken together, it is indicated that NTP shows selective anticancer effects when employed with 5 mM POST NAC treatments and ROS cytotoxicity plays an important role in NTP treatment to those who are sensitive to ROS. Further more, it is important to find out which signaling pathways are affected by NTP treatments.
Low concentration of H2O2 recovers the induction of proliferation in MDA-MB-231 cells under 120 s NTP treatment with post-treated NAC by effects Interleukin-6 receptor (IL-6R)/Stat3 pathway
The IL-6/IL-6 receptor complex performs as upstream regulatory of Stat3 pathway46 and has important roles in the proliferation of cancer cells47. Both of the IL-6 and IL-6R are expressed in MDA-MB-23148. Depending upon the results shown in Fig. 5A, MDA-MB-231 cells were post-treated with 5 mM NAC and 0/50 μM H2O2 immediately after 120 s NTP treatment. The NTP killing effects on both treated groups were still significantly (data not shown), but a significant recovery of proliferation and IL-6R pathway upregulation in low concentration of H2O2 treated cells was observed (Fig. 5C and Supplementary Figure 4). It indicated that low concentration of H2O2 can recover the expression of IL-6R and tyrosine-phosphorylated Stat3. Under this situation, cell proliferation related PTEN/Akt pathway was also biased through IL-6R/pSTAT3 affection (Fig. 5C and Supplementary Figure 4). Taken together, the IL-6R expression plays an important role in proliferation of MDA-MB-231 cells and NTP can down regulate IL-6R pathway.
The mechanism of NTP selective effects on TN normal breast cells MCF10A and TNBC cells MDA-MB-231 with POST NAC treatment
48 h after NTP treatment post-treated with or without NAC, we examined the activation of IL-6R/STAT3 and PTEN/Akt pathways in MDA-MB-231 and MCF10A cells. Along with growing exposure time dose under NTP treatment, the expression of IL-6R and pSTAT3 showed significant decrease while in contrary PTEN increased markedly due to the decline of the former in all cases (Fig. 6A and B). Since NAC eliminated the protection of ROS on Akt activation, the expression of pAkt in MDA-MB-231 is down regulated by NTP induced PTEN increasing (Fig. 6A and Supplementary Figure 5A). It is interesting that Akt activation constantly increased in MCF10A cells which was induced by NTP with POST NAC treatment (Fig. 6B and Supplementary Figure 5B). Previous studies revealed that Akt activation promotes proliferation in MCF10A cells49 and HER2 is an upstream regulator of Akt activation50, 51. Surprisingly, the expression of HER2 recovered and increased significantly in this TN normal cell MCF10A (Fig. 6B and Supplementary Figure 5B) after NTP treatment with or without NAC. Under the same situation, TNBC cells MDA-MB-231 remain lack of HER2 expression (Fig. 6A and Supplementary Figure 5A). These results suggest that selective effects of NTP treatments on HER2 expression caused the divergent response in MDA-MB-231 and MCF10A after post-treated with NAC.
Figure 6.
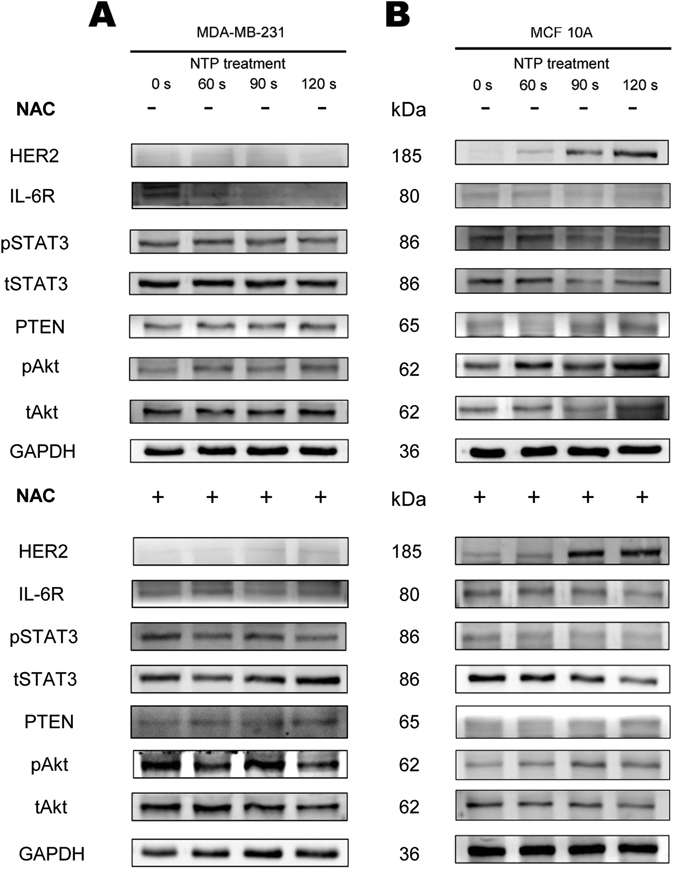
Effects of NTP on the IL-6R/pSTAT3 and HER2/pAkt pathway in MDA-MB-231 and MCF10A cells. MDA-MB-231 (A) and MCF10A (B) cells were exposed to NTP for 0, 60, 90 or 120 s. After forty-eight hours post-treated with 5 mM N-acetyl cysteine (NAC+) or without (NAC−), proteins from total cell lysates were harvested. The expressions of HER2, L-6R, pSTAT3, tSTAT3, PTEN, pAkt and tAkt were detected by Western blot analysis. GAPDH was taken as a loading control throughout. The gels/blots were processed under the same experimental condition. Cropped gels/blots are displayed and full-length blots/gels are presented in Supplementary Figure 5.
Discussion
In recent years, NTP has been demonstrated to be a potential tool in anticancer therapy. The mechanism of NTP effects on cancer cells in vitro has been mainly revealed in several aspects, including the toxicity induced by chemical essence of NTP, NTP affects the change of cellular shape and membrane, the transmembrane diffusion of NTP generated reactive species, the change of intracellular ROS and redox balance, causing DNA damage and influencing apoptosis pathways52. Previous studies also reported that NTP treatment can induce apoptotic cell death in various tumors by ROS generation intracellular and extracellular. Mechanism of these ROS-dependent cell deaths by NTP was discovered to have been associated with several cellular signaling pathways such as: NTP treatment induced p53-dependent apoptosis in colon carcinoma cells53, 54. PARP1-mediated DNA damage is also an extensively revealed mechanism that explains the mammalian and carcinoma cell death induced by non-thermal plasma37, 55. Additionally, several groups have reported that activation of ERK and JNK signaling pathway is involved in immune cells, lung and cervical cancer cells apoptosis by non-thermal plasma15, 24, 56. Taken together, these studies reveal that the NTP associated mechanisms is complicated and specific in different types of cells. It is notable that to reveal mechanisms specifically in such subgroups of cancer under non-thermal plasma treatment is critical for further precision therapy.
Patients with TNBC, a subgroup of breast cancer, lack specific therapy and have a comparatively poor outcome57. Especially subtypes of TNBC like MDA-MB-231 cells are more invasive and metastasize because of constantly activated IL-6 signaling58. IL-6 receptor plays a critical role in IL-6 signaling in human mammary carcinoma cell lines48. Depending on our results, NTP treatment can significantly inhibit IL-6R/pSTAT3 activation in vitro (Fig. 7B). Based on this conclusion, it is highly possible that NTP could be a promising therapy for cancer related with IL-6 signaling.
Figure 7.
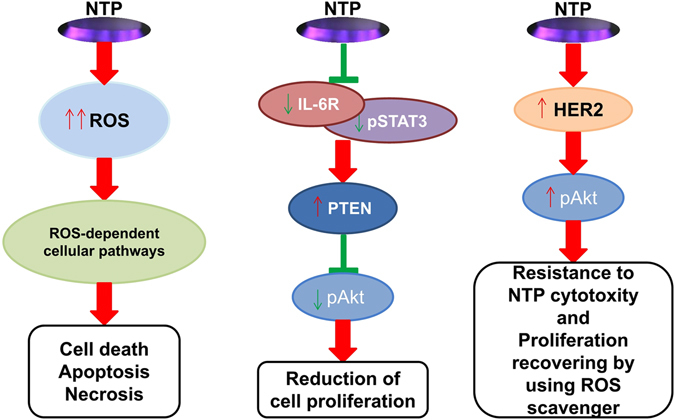
A general survey of NTP treatment affection on triple-negative breast carcinoma and normal cells through different cellular pathways.(A) NTP treatment inhibits proliferation of MDA-MB-453 and MCF10A cells through ROS-dependent cellular pathway (not discussed in this article). (B) NTP treatment effects on reduction of proliferation through IL-6R/pSTAT3 pathway in MDA-MB-231 and MCF10A cells. (C) NTP treatment effects on recovering the proliferation especially in normal breast MCF10A cells with post-treated 5 mM NAC.
Activated Akt gets involved in the regulation of cellular growth and cell survival. In cancer cells, Akt activation has been associated with increased resistance to apoptosis. On the other hand, Akt can be activated by some antitumor cytokine59. Recent study reported that pAkt in thyroid papillary cancer cells can be decreased by NTP treatment60. We also confirmed that Akt activation can be reduced after NTP treatment with NAC (Fig. 7B). In addition, we observed that Akt can be constantly activated especially in normal breast MCF10A cells through NTP effects (Fig. 7C). We discover this selective effect of NTP on Akt inactivation and activation was observed.
Non-thermal atmospheric plasma treatment is widely reported to cause cytotoxic effects in cells by inducing RONS to the cell culture43, 61, 62. Our data showed that the inhibition of proliferation in normal breast cell MCF10A cells induced by 120 s NTP treatment was similar to the cytotoxicity of 200 μM H2O2 treatment. In order to avoid ROS cytotoxicity on normal cells MCF10A induced by NTP, we have employed 5 mM NAC post-treated with cells after NTP treatments that could recover MCF10A’s cell viability from about 5% to 65% (120 s NTP) and a significant prevention of intracellular ROS (Fig. 2C). Results of Fig. 2C also showed that POST NAC treatments would recover more proliferation of MCF10A cells than PRE NAC treatments. It was reported that NAC as a derivative of cysteine would strongly react with ROS41, 63, 64. Our results showed that POST NAC treatments could protect more MCF10A cells than extracellular ROS scavenger catalase under 120 s NTP treatments (Figs 2C and 4C), but POST NAC treatments could not save cells after 185 μM H2O2 treatments (Supplementary Figure 3C). In Supplementary Figure 3A, it was observed that post-treated MCF10A cells with 5 mM NAC could counteract part of the cytotoxicity induced by 50 and 100 μM H2O2 but not 150 μM H2O2. It may be the case that high concentration H2O2 (more than 100 μM) could cause permanent damage in a short period of time before NAC consume it. Taken together, these findings indicated that HER2/Akt activation induced by NTP treatments which plays a crucial role in proliferation recovery of MCF10A cells. Interestingly, TNBC MDA-MB-231 cells show different responses to H2O2 under low and high concentrations. The similar results are observed in C. elegans 65 and Hepatic cells mice66 through different cell signaling. Our findings showed that low concentration of H2O2 can counteract part of NTP killing effects on TNBC MDA-MB-231 cells via IL-6R/pSTAT3 pathway. Based on these findings above, generation of ROS accompany with NTP treatment may have an influence of unexpected to some TNBC patients.
To comprehend the mechanism underlying the selective effects of non-thermal atmospheric plasma are essential in utilizing it to clinical TNBC therapy. In conclusion, our experiments have demonstrated here that the proliferation of TNBC cells can be significantly reduced after NTP treatment in two different ways. TNBC like MDA-MB-453 cells, who are lacking of autocrine IL-6, can be affected through ROS cytotoxicity induced by NTP treatment (Fig. 7A). TNBC like MDA-MB-231 cells, who are not sensitive to ROS and combined with high expression autocrine IL-6, can be inhibited through IL-6R/pSTAT3 pathway under NTP treatment (Fig. 7B). With the help of ROS scavengers, constantly HER2/Akt activation in normal TN cells like MCF10A making cells survive through selective effects of NTP treatment (Fig. 7C). These surprising fundamental results suggest that the selective effects by restricted NTP treatment can be further developed into a novel approach for urgently needed TNBC therapies.
Our results indicate that NTP remains significant in inhibition of TNBC MDA-MB-231 cells even with the ROS blockader NAC. In addition, we reported that the mechanism of NTP anti-cancer selectivity relates to constantly HER2/Akt activation induced by NTP especially in MCF10A cells but not in MDA-MB-231 cells. This aspect of NTP effects makes it possible to become a novel tool in non-invasive anticancer therapy. The mechanism of how NTP treatment affects the tumor is still not fully understood. Our experiments demonstrate ROS generation of NTP treatment is important in TNBC subtype like MDA-MB-453 cells therapy, but ROS scavenger employed in NTP treatments should be considered much more important in TNBC subtype like MDA-MB-231 cells therapy. Thus precision therapy for various carcinoma subtypes should be developing further in NTP treatment.
Methods
Cell culture
Human breast cancer cell lines MDA-MB-453, MDA-MB-231 and mammalian breast epithelial cell line MCF10A were purchased from the American Type Culture Collection (ATCC). MDA-MB-453 and MDA-MB-231 cells were cultured in L-15 medium supplemented with 10% fetal bovine serum at 37 °C incubator. MCF10A cells were cultured in high glucose DMEM/F12 50:50 mixture supplemented with 5% horse serum, Epidermal Growth Factor (100 mg/ml), Hydrocortisone (1 mg/ml), Cholera Toxin (1 mg/ml) and Insulin (10 mg/ml) at 37 °C and 5% CO2 incubator. N-Acetyl L-cysteine (NAC) and catalase was used as ROS scavenger. NAC cytotoxicity to all cell lines was tested and 5 mM NAC was chosen to do further experiments (Supplementary Figure 6A). Catalase cytotoxicity to all cell lines was tested and 500 U/ml catalase was chosen to do further experiments (Supplementary Figure 6B).
NTP device and treatment
The air plasma is generated between the high voltage electrode (an array with 16 fine copper rods) and culture medium which is used as the ground electrode. The high voltage electrode is driven by 10 kV and the discharge current is about 5 mA being restricted by two ballast resistors R1 and R2 (both 25 mΩ). The gas temperature of the plasma is below 300 K because of its low power consumption. Thus the air plasma could be touched safely and directly by human skin. (Fig. 1A).
4 × 105 cells were seeded into 60 mm diameter petri dishes for plasma exposures. The medium was replaced with 5 ml new complete culture medium just before exposures. The distance between the copper rods and the surface of the medium was fixed at 20 mm. The cells were then exposed to NTP for 0, 60, 90 or 120 s. After 48 hours, the cells were harvested for further experiments.
Cell counting
Cell viability was determined by trypan blue staining. Cell numbers of each dish were counted by an automatic analyzer (Countstar, China).
ROS scavenger studies
Cells were pre-treated for 1 h with 5 mM NAC and will be washed before the NTP treatment (PRE NAC), or post-treated with 5 mM NAC immediately after NTP treatment (POST NAC) until harvested for further experiments. As indicated in Fig. 2.
Detection of intracellular ROS
Intracellular ROS was determined using an oxidation-sensitive fluorescent probe (DCFH-DA) (Beyotime, China). Cells were preloaded with 10 μM DCFH-DA for 30 min and will be washed before NTP treatment. One hour after treatment, cells were harvested and fluorescence was analyzed by FACSVerse flow cytometer (Becton Dickinson, USA). For each sample 30 000 events were collected.
Western blot analysis
4 × 105 cells of all TN breast cell lines were seeded into 60 mm dishes with 5 mL complete culture medium and then exposed to NTP for 0, 60, 90 and 120 s with or without 5 mM NAC, and 120 s NTP treatment with or without 50 μM H2O2. After 48 h incubation, the total protein was extracted by modified RIPA lysis buffer (pH 7.4, 1% NP-40, 0.25% Na-deoxycholate, 1 mM EDTA, 150 mM NaCl, 50 mM Tris, protease inhibitor cocktail (Roche)). Proteins from total cell lysates were uploaded and separated by 10% SDS-PAGE, and then electro-transferred to polyvinylidenedifluouride (PVDF) membrane (Millipore Corp, Atlanta, GA, US). The membranes were blocked in 5% non-fat dry milk in PBS/Tween-20 at room temperature for 1 h, and then blotted with primary antibodies overnight at 4 °C, followed by HRP-conjugated secondary antibodies at room temperature for 1 h. The signals were developed using the ECL (enhanced chemiluminescence) reagents (Pierce, Rockford, IL, USA) and read on ImageQuant LAS4000 (GE Healthy).
Statistical analysis
All experiments were repeated at least three times and all data were presented as mean ± SD (standard deviation). The difference between two groups was analyzed by Student’s t-test, and the difference between more than two groups was analyzed using one-way ANOVA test with Tukey’s multiple comparison test. Any case p < 0.05 was regarded as significant.
Electronic supplementary material
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China under Grant No. 11475174, No. 11675177, No. 81472494, No. 81502282 and No. 8130232, Science Foundation of Institute of Plasma Physics, Chinese Academy of Sciences under Grant No. DSJJ-14-YY02, The Natural Science Foundation of Anhui Province Grant No. 1708085MA13, The National Key Scientific Programme of China under Grant No. 2016YFC1302305, The ministry of Education under Grant No. 20133402120032. The authors would like to thank Shulou Qian (University of Science and Technology of China, CHINA) for his assistance in the graphic design and Travis Bhandari (Anhui Medical University, CHINA) for his assistance in the improvement of English writing.
Author Contributions
Y.L., C.C. and W.X. led the project and supervised all experiments. Y.L. S.T., H.Z., X.K., L.D. J.S. and Y.L. conducted experiments and measurements. Z.T., C.C. and W.X. co-led data analysis and physical interpretations. All authors discussed the results. Y.L., Z.T., C.C. and W.X. co-wrote the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-08792-3
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Cheng Cheng, Email: chengcheng@ipp.ac.cn.
Tao Zhu, Email: zhut@ustc.edu.cn.
Weidong Xia, Email: xiawd@ustc.edu.cn.
References
- 1.Jemal A, et al. Global cancer statistics. Ca A Cancer Journal for Clinicians. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Billar JAY, et al. Triple-Negative Breast Cancers: Unique Clinical Presentations and Outcomes. Annals of Surgical Oncology. 2010;17(3):384–390. doi: 10.1245/s10434-010-1260-4. [DOI] [PubMed] [Google Scholar]
- 3.Liedtke C, et al. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. Journal of Clinical Oncology. 2008;26:1275–1281. doi: 10.1200/JCO.2007.14.4147. [DOI] [PubMed] [Google Scholar]
- 4.Tan DSP, et al. Triple negative breast cancer: molecular profiling and prognostic impact in adjuvant anthracycline-treated patients. Breast Cancer Research & Treatment. 2008;111:27–44. doi: 10.1007/s10549-007-9756-8. [DOI] [PubMed] [Google Scholar]
- 5.O’Reilly EA, et al. The fate of chemoresistance in triple negative breast cancer (TNBC) Bba Clinical. 2015;3:257–275. doi: 10.1016/j.bbacli.2015.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Woedtke TV, Reuter S, Masur K, Weltmann KD. Plasmas for medicine. Physics Reports. 2013;530:291–320. doi: 10.1016/j.physrep.2013.05.005. [DOI] [Google Scholar]
- 7.Lu X, et al. Reactive species in non-equilibrium atmospheric-pressure plasmas: Generation, transport, and biological effects. Physics Reports. 2016;630:1–84. doi: 10.1016/j.physrep.2016.03.003. [DOI] [Google Scholar]
- 8.Graves DB. Reactive Species from Cold Atmospheric Plasma: Implications for Cancer Therapy. Plasma Processes & Polymers. 2014;11:1120–1127. doi: 10.1002/ppap.201400068. [DOI] [Google Scholar]
- 9.Fridman G, et al. Applied Plasma Medicine. Plasma Processes & Polymers. 2008;5:503–533. doi: 10.1002/ppap.200700154. [DOI] [Google Scholar]
- 10.Keidar M. Plasma for cancer treatment. Plasma Sources Science and Technology. 2015;24 doi: 10.1088/0963-0252/24/3/033001. [DOI] [Google Scholar]
- 11.Schlegel J, Köritzer J, Boxhammer V. Plasma in cancer treatment. Clinical Plasma Medicine. 2013;1:2–7. doi: 10.1016/j.cpme.2013.08.001. [DOI] [Google Scholar]
- 12.Park SB, et al. Differential Epigenetic Effects of Atmospheric Cold Plasma on MCF-7 and MDA-MB-231 Breast Cancer Cells. Plos One. 2015;10 doi: 10.1371/journal.pone.0129931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cheng X, et al. The Effect of Tuning Cold Plasma Composition on Glioblastoma Cell Viability. Plos One. 2014;9 doi: 10.1371/journal.pone.0098652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hirst AM, et al. Low-temperature plasma treatment induces DNA damage leading to necrotic cell death in primary prostate epithelial cells. British Journal of Cancer. 2015;112:1536–1545. doi: 10.1038/bjc.2015.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hou J, et al. Non-thermal plasma treatment altered gene expression profiling in non-small-cell lung cancer A549 cells. Bmc Genomics. 2014;16:1–12. doi: 10.1186/s12864-015-1644-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yokoyama M, Johkura K, Sato T. Gene expression responses of HeLa cells to chemical species generated by an atmospheric plasma flow. Biochemical & Biophysical Research Communications. 2014;450:1266–1271. doi: 10.1016/j.bbrc.2014.06.116. [DOI] [PubMed] [Google Scholar]
- 17.Ishaq M, et al. Atmospheric gas plasma-induced ROS production activates TNF-ASK1 pathway for the induction of melanoma cancer cell apoptosis. Molecular Biology of the Cell. 2014;25:1523–1531. doi: 10.1091/mbc.E13-10-0590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Panngom K, et al. Preferential killing of human lung cancer cell lines with mitochondrial dysfunction by nonthermal dielectric barrier discharge plasma. Cell Death & Disease. 2013;4:585–599. doi: 10.1038/cddis.2013.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sun JK, Chung TH. Cold atmospheric plasma jet-generated RONS and their selective effects on normal and carcinoma cells. Scientific Reports. 2016;6 doi: 10.1038/srep20332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sato T, Yokoyama M, Johkura K. A key inactivation factor of HeLa cell viability by a plasma flow. Journal of Physics D Applied Physics. 2011;44:372001–372005. doi: 10.1088/0022-3727/44/37/372001. [DOI] [Google Scholar]
- 21.Keidar M, et al. Cold plasma selectivity and the possibility of a paradigm shift in cancer therapy. British Journal of Cancer. 2011;105:1295–1301. doi: 10.1038/bjc.2011.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ishaq M, Evans M, Ostrikov K. Effect of atmospheric gas plasmas on cancer cell signaling. International Journal of Cancer Journal International Du Cancer. 2014;134:1517–1528. doi: 10.1002/ijc.28323. [DOI] [PubMed] [Google Scholar]
- 23.Kang SU, et al. Nonthermal plasma induces head and neck cancer cell death: the potential involvement of mitogen-activated protein kinase-dependent mitochondrial reactive oxygen species. Cell Death & Disease. 2014;5 doi: 10.1038/cddis.2014.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li W, et al. Non-thermal plasma inhibits human cervical cancer HeLa cells invasiveness by suppressing the MAPK pathway and decreasing matrix metalloproteinase-9 expression. Scientific Reports. 2016;6 doi: 10.1038/srep19720. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 25.Chang JW, et al. Non-thermal atmospheric pressure plasma induces apoptosis in oral cavity squamous cell carcinoma: Involvement of DNA-damage-triggering sub-G 1 arrest via the ATM/p53 pathway. Archives of biochemistry and biophysics. 2014;545:133–140. doi: 10.1016/j.abb.2014.01.022. [DOI] [PubMed] [Google Scholar]
- 26.Conway GE, et al. Non-thermal atmospheric plasma induces ROS-independent cell death in U373MG glioma cells and augments the cytotoxicity of temozolomide. British Journal of Cancer. 2016;114:435–443. doi: 10.1038/bjc.2016.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bekeschus S, et al. Hydrogen peroxide: A central player in physical plasma-induced oxidative stress in human blood cells. Free Radical Research. 2014;48:542–549. doi: 10.3109/10715762.2014.892937. [DOI] [PubMed] [Google Scholar]
- 28.Ahn HJ, et al. Atmospheric-pressure plasma jet induces apoptosis involving mitochondria via generation of free radicals. Plos One. 2011;6 doi: 10.1371/journal.pone.0028154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhao S, et al. Atmospheric pressure room temperature plasma jets facilitate oxidative and nitrative stress and lead to endoplasmic reticulum stress dependent apoptosis in HepG2 cells. Plos One. 2013;8 doi: 10.1371/journal.pone.0073665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Adachi T, et al. Plasma-activated medium induces A549 cell injury via a spiral apoptotic cascade involving the mitochondrial-nuclear network. Free Radical Biology and Medicine. 2015;79:28–44. doi: 10.1016/j.freeradbiomed.2014.11.014. [DOI] [PubMed] [Google Scholar]
- 31.Judée F, et al. Short and long time effects of low temperature Plasma Activated Media on 3D multicellular tumor spheroids. Scientific Reports. 2016;6 doi: 10.1038/srep21421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Adachi T, et al. Iron stimulates plasma-activated medium-induced A549 cell injury. Scientific Reports. 2016;6 doi: 10.1038/srep20928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yan D, et al. Stabilizing the cold plasma-stimulated medium by regulating medium’s composition. Scientific Reports. 2016;6 doi: 10.1038/srep26016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kaushik N, Kumar N, Kim CH, Kaushik NK, Choi EH. Dielectric Barrier Discharge Plasma Efficiently Delivers an Apoptotic Response in Human Monocytic Lymphoma. Plasma Processes & Polymers. 2015;11:1175–1187. doi: 10.1002/ppap.201400102. [DOI] [Google Scholar]
- 35.Yan D, et al. Toward understanding the selective anticancer capacity of cold atmospheric plasma—A model based on aquaporins (Review) Biointerphases. 2015;10 doi: 10.1116/1.4938020. [DOI] [PubMed] [Google Scholar]
- 36.Sun JK, Joh HM, Chung TH. Production of intracellular reactive oxygen species and change of cell viability induced by atmospheric pressure plasma in normal and cancer cells. Applied Physics Letters. 2013;103 doi: 10.1063/1.4824986. [DOI] [Google Scholar]
- 37.Kalghatgi, S. et al. Effects of Non-Thermal Plasma on Mammalian Cells. Plos One6, e16270 (2011). [DOI] [PMC free article] [PubMed]
- 38.Gweon B, et al. Suppression of angiogenesis by atmospheric pressure plasma in human aortic endothelial cells. Applied Physics Letters. 2014;104 doi: 10.1063/1.4870623. [DOI] [Google Scholar]
- 39.Wiseman H, Halliwell B. Damage to DNA by reactive oxygen and nitrogen species: role in inflammatory disease and progression to cancer. Biochemical Journal. 1996;313(Pt 1):17–29. doi: 10.1042/bj3130017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang M, et al. Cold Atmospheric Plasma for Selectively Ablating Metastatic Breast Cancer Cells. Plos One. 2013;8 doi: 10.1371/journal.pone.0073741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yan D, et al. Principles of using Cold Atmospheric Plasma Stimulated Media for Cancer Treatment. Scientific Reports. 2015;5 doi: 10.1038/srep18339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Recek N, et al. Effect of Cold Plasma on Glial Cell Morphology Studied by Atomic Force Microscopy. Plos One. 2015;10 doi: 10.1371/journal.pone.0119111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ahn HJ, et al. Targeting Cancer Cells with Reactive Oxygen and Nitrogen Species Generated by Atmospheric-Pressure Air Plasma. Plos One. 2014;9 doi: 10.1371/journal.pone.0086173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bowman T, Garcia R, Turkson J, Jove R. STATs in oncogenesis. Oncogene. 2000;19:2474–2488. doi: 10.1038/sj.onc.1203527. [DOI] [PubMed] [Google Scholar]
- 45.Yu H, Jove R. The STATs of cancer–new molecular targets come of age. Nature Reviews Cancer. 2004;4:97–105. doi: 10.1038/nrc1275. [DOI] [PubMed] [Google Scholar]
- 46.Kishimoto T, Akira S, Narazaki M, Taga T. Interleukin-6 family of cytokines and gp130. Blood. 1995;86:1243–1254. [PubMed] [Google Scholar]
- 47.Jones SA, Scheller J, Rosejohn S. Therapeutic strategies for the clinical blockade of IL-6/gp130 signaling. Journal of Clinical Investigation. 2011;121:3375–3383. doi: 10.1172/JCI57158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chiu JJ, Sgagias MK, Cowan KH. Interleukin 6 acts as a paracrine growth factor in human mammary carcinoma cell lines. Clinical Cancer Research. 1996;2:215–221. [PubMed] [Google Scholar]
- 49.Debnath J, Walker SJ, Brugge JS. Akt activation disrupts mammary acinar architecture and enhances proliferation in an mTOR-dependent manner. The Journal of Cell Biology. 2003;163:315–326. doi: 10.1083/jcb.200304159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Antonoff G, Conan RJ. The discovery of receptor tyrosine kinases: targets for cancer therapy. Nature Reviews Cancer. 2004;4:361–370. doi: 10.1038/nrc1360. [DOI] [PubMed] [Google Scholar]
- 51.Geer PV, T Hunter A, Lindberg RA. Receptor protein-tyrosine kinases and their signal transduction pathways. Annual Review of Cell Biology. 1994;10:251–337. doi: 10.1146/annurev.cb.10.110194.001343. [DOI] [PubMed] [Google Scholar]
- 52.Yan D, Sherman JH, Keidar M. Cold atmospheric plasma, a novel promising anti-cancer treatment modality. Oncotarget. 2016;8:15977–15995. doi: 10.18632/oncotarget.13304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ma Y, et al. Non-Thermal Atmospheric Pressure Plasma Preferentially Induces Apoptosis in p53-Mutated Cancer Cells by Activating ROS Stress-Response Pathways. Plos One. 2014;9 doi: 10.1371/journal.pone.0091947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tuhvatulin AI, et al. Non-thermal Plasma Causes p53-Dependent Apoptosis in Human ColonCarcinoma Cells. Acta Naturae. 2012;4:82–87. [PMC free article] [PubMed] [Google Scholar]
- 55.Choi JY, et al. Non-thermal plasma-induced apoptosis is modulated by ATR- and PARP1-mediated DNA damage responses and circadian clock. Oncotarget. 2016;7:32980–32989. doi: 10.18632/oncotarget.9087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bundscherer L, et al. Impact of non-thermal plasma treatment on MAPK signaling pathways of human immune cell lines. Immunobiology. 2013;218:1248–1255. doi: 10.1016/j.imbio.2013.04.015. [DOI] [PubMed] [Google Scholar]
- 57.Foulkes WD, Smith IE, Reisfilho JS. Triple-Negative Breast Cancer. New England journal of medicine. 2010;363:1938–1948. doi: 10.1056/NEJMra1001389. [DOI] [PubMed] [Google Scholar]
- 58.Hartman ZC, et al. Growth of triple-negative breast cancer cells relies upon coordinate autocrine expression of the proinflammatory cytokines IL-6 and IL-8. Cancer Research. 2013;73:3470–3480. doi: 10.1158/0008-5472.CAN-12-4524-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Vara JÁF, et al. PI3K/Akt signalling pathway and cancer. Cancer Treatment Reviews. 2004;30:193–204. doi: 10.1016/j.ctrv.2003.07.007. [DOI] [PubMed] [Google Scholar]
- 60.Chang JW, et al. Non-thermal atmospheric pressure plasma inhibits thyroid papillary cancer cell invasion via cytoskeletal modulation, altered MMP-2/-9/uPA activity. Plos One. 2014;9 doi: 10.1371/journal.pone.0092198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kaushik N, et al. Responses of Solid Tumor Cells in DMEM to Reactive Oxygen Species Generated by Non-Thermal Plasma and Chemically Induced ROS Systems. Scientific Reports. 2015;5 doi: 10.1038/srep08587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Boehm D, Heslin C, Cullen PJ, Bourke P. Cytotoxic and mutagenic potential of solutions exposed to cold atmospheric plasma. Scientific Reports. 2016;6 doi: 10.1038/srep21464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Aruoma OI, Halliwell B, Hoey BM, Butler J. The antioxidant action of N-acetylcysteine: its reaction with hydrogen peroxide, hydroxyl radical, superoxide, and hypochlorous acid. Free Radical Biology & Medicine. 1989;6:593–597. doi: 10.1016/0891-5849(89)90066-X. [DOI] [PubMed] [Google Scholar]
- 64.Takai E, et al. Chemical modification of amino acids by atmospheric-pressure cold plasma in aqueous solution. Journal of Physics D: Applied Physics. 2014;47 doi: 10.1088/0022-3727/47/28/285403. [DOI] [Google Scholar]
- 65.Yee C, Yang W, Hekimi S. The Intrinsic Apoptosis Pathway Mediates the Pro-Longevity Response to Mitochondrial ROS in C.elegans. Cell. 2014;157:897–909. doi: 10.1016/j.cell.2014.02.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Gurzov E, et al. Hepatic Oxidative Stress Promotes Insulin-STAT-5 Signaling and Obesity by Inactivating Protein Tyrosine Phosphatase N2. Cell Metabolism. 2014;20:85–102. doi: 10.1016/j.cmet.2014.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


