Abstract
Purpose
When postoperative ileus is not resolved after 5 days or recurs after resolution, prolonged POI (PPOI) is diagnosed. PPOI increases discomfort, morbidity and hospitalisation length, and is mainly caused by an inflammatory response following intestinal manipulation. This response can be weakened by targeting the cholinergic anti-inflammatory pathway, with nicotine as essential regulator. Chewing gum, already known to stimulate gastrointestinal motility itself, combined with nicotine is hypothesised to improve gastrointestinal recovery and prevent PPOI. This pilot study is the first to assess efficacy and safety of nicotine gum in colorectal surgery.
Methods
Patients undergoing elective oncological colorectal surgery were enrolled in this double-blind, parallel-group, controlled trial and randomly assigned to a treatment protocol with normal or nicotine gum (2 mg). Patient reported outcomes (PROMS), clinical characteristics and blood samples were collected. Primary endpoint was defined as time to first passage of faeces and toleration of solid food for at least 24 h.
Results
In total, 40 patients were enrolled (20 vs. 20). In both groups, six patients developed PPOI. Time to primary endpoint (4.50 [3.00–7.25] vs. 3.50 days [3.00–4.25], p = 0.398) and length of stay (5.50 [4.00–8.50] vs. 4.50 days [4.00–6.00], p = 0.738) did not differ significantly between normal and nicotine gum. There were no differences in PROMS, inflammatory parameters and postoperative complications.
Conclusions
We proved nicotine gum to be safe but ineffective in improving gastrointestinal recovery and prevention of PPOI after colorectal surgery. Other dosages and administration routes of nicotine should be tested in future research.
Electronic supplementary material
The online version of this article (doi:10.1007/s00384-017-2839-z) contains supplementary material, which is available to authorized users.
Keywords: Postoperative ileus, Prevention, Nicotine chewing gum, Colorectal surgery
Introduction
Postoperative ileus (POI) is a temporary inhibition of gastrointestinal motility after abdominal surgery and is usually associated with nausea, vomiting, abdominal distension and lack of flatus and defaecation [1, 2]. In more than 50% of cases, POI is not fully resolved in 4 days after the operation and when it does not resolve after 5 days or recurs after an apparent resolution, prolonged POI (PPOI) is diagnosed [3, 4]. PPOI causes an increase in patient discomfort, morbidity, hospital-acquired infections, hospitalisation days and healthcare costs [5].
The aetiology of POI is complex, with multiple factors contributing to its pathogenesis [6]. Opioid use for postoperative analgesia is known to inhibit gastrointestinal transit and prolong POI [7, 8]. However, the development of POI after abdominal surgery is mainly caused by intestinal manipulation during the surgical procedure, thereby triggering an inflammatory response and causing a sustained and generalised gastrointestinal hypomotility [7, 9, 10]. Targeting this inflammatory response is of clinical relevance, but effective strategies are not yet available in clinical practice [8].
The cholinergic anti-inflammatory pathway (CAIP) is one of the mechanisms that can be targeted for the prevention of POI. Experimental studies have shown that mediation of CAIP by vagus nerve stimulation can increase bowel motility and control inflammatory cell recruitment, by that preventing pathological changes important in the development of POI [1, 8, 11]. Moreover, nicotinic acetylcholine receptors (nAChR) play an important role in mediation of CAIP, making nicotine an essential regulator of the pathway [11–13]. Additionally, the α7-nAChR also plays a role in nicotine-induced analgesia [14] and clinical evidence shows that preoperative transdermal and intranasal administration of nicotine significantly reduced postoperative opioid use [15, 16], while reducing opioids is an important strategy of shortening POI [1, 8, 17].
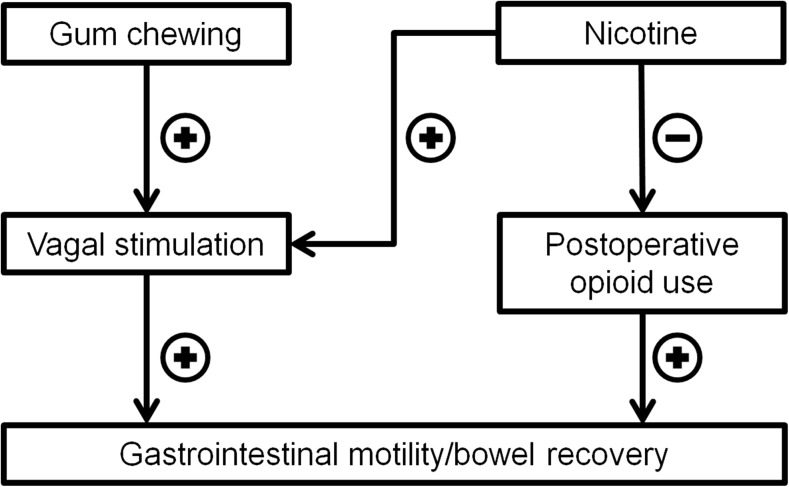
Gum chewing is another important strategy, which has already been proven to be beneficial for gastrointestinal recovery after surgery. Several systematic reviews and meta-analyses have been published, supporting postoperative gum chewing in abdominal surgery [18–23]. As a form of sham feeding, it mimics the cephalic phase of digestion and stimulates the gastrointestinal motility via neurohormonal and vagal pathways [18, 24]. Combining perioperative gum chewing with the potential beneficial effects of nicotine leads to the hypothesis that nicotine gum chewing can reduce POI and improve postoperative outcomes (e.g. less morbidity and shorter length of stay) as well as reduce medical costs [25]. The commercially available and inexpensive nicotine chewing gum may have a wide clinical application in POI prevention, by both stimulating the cephalic-vagal reflex and activating CAIP (Fig. 1). Therefore, we performed a multicenter, randomised, double-blind, controlled pilot study, comparing perioperative use of nicotine chewing gum with normal chewing gum, to assess the clinical efficacy and safety in patients undergoing colorectal surgery.
Fig. 1.
Simplified scheme of hypothesised effect mechanism of nicotine chewing gum
Methods
Study design and participants
This is a prospective, parallel-group, double-blind, randomised, controlled pilot study, conducted in the Havenziekenhuis, Rotterdam and the Sint Franciscus Gasthuis, Rotterdam. Adult patients who underwent an elective oncologic colorectal resection and gave written consent were included. Exclusion criteria were severe chronic cardiovascular disease or acute cardiovascular disease, severe liver- or kidney disease, oral or pharyngeal infection, esophagitis, hypersensitivity to any component of the nicotine gum, previous colorectal surgery, pregnancy, breast feeding, having an elevated risk of choking or being unable to chew gum for any reason.
Study procedures
Patients received either normal chewing gum or Nicorette® 2-mg chewing gum (2 mg/gum). This nicotine chewing gum is normally used as nicotine replacement therapy to help control craving for cigarettes and contains a low dose of nicotine. Patients had to chew the allocated chewing gum 2 h preoperatively and three times a day postoperatively, for half an hour at a time, until the first passage of faeces and tolerance of solid food for more than 24 h.
Patients were asked to fill out a questionnaire before surgery and daily after surgery, until postoperative day (POD) 6. This patient diary contained questions regarding chewing gum use, oral intake, bowel movements, defaecation, gastrointestinal symptoms and visual analogue scale (VAS) pain score.
Surgeons or surgical residents were asked to fill out case record forms (CRF) with information regarding both patient and surgical characteristics, such as age, gender, body mass index (BMI), American Society of Anesthesiologists (ASA) score, medication use, smoking, operative procedure, postoperative course and postoperative complications (e.g. anastomotic leakage (AL), surgical site infection (SSI), fascial dehiscence, urinary tract infection (UTI) and pneumonia).
Blood sample analysis
Peripheral blood samples was drawn from patients prior to the surgical procedure, and in the morning on POD1 and POD3. Measurements of plasma white blood cell count (WBC) and C-reactive protein (CRP) were performed by the hospital’s laboratory at these same time points. Blood samples were centrifuged and plasma was stored at −80 °C. Enzyme-linked immunosorbent assays (ELISAs) were performed according to instructions of the manufacturer (PeproTech Inc., Rocky Hill, USA) to quantify the concentration of the systemic inflammatory marker interleukin-6 (IL-6). A ratio of samples was calculated, through dividing the values of POD3 samples by those of the preoperative samples ().
Outcome parameters
The primary study parameter was the time from surgery until the resolution of POI, defined as passage of faeces and toleration of solid food for at least 24 h [26]. Secondary endpoints included time to first flatus, hospitalisation length, postoperative (infectious) complications, postoperative mortality, postoperative opioid use, patient reported outcomes (e.g. pain score, nausea, regurgitations, vomiting, chewing gum use), inflammatory parameters (e.g. CRP, WBC and IL-6), blood pressure, body temperature and heart rate.
PPOI was defined as POI that was not resolved after POD5 or recurrent POI after an apparent resolution of POI. Diagnosis of PPOI was not made directly by the participating surgeons, but via retrospective review of the patient diary and medical record, to ensure objectiveness of the primary endpoint.
Sample size calculation
According to Asao’s gum chewing experiment [24] and Flood’s nicotine trial [16], a sample size calculation was made, based on a mean POI time of 4.0 days in the chewing gum groups and an assumption of 2.6 days in the nicotine chewing gum group with a standard deviation of 1.5 days in both groups. In order to obtain a power of 80%, with an α level of 0.05, a number of 16 patients were needed in each group. As a dropout rate of 20% was expected, a total number of 40 patients (20 patients per group) were needed.
Patient allocation
Randomisation was done with Microsoft Excel 2010 (Microsoft Corporation, Redmond, WA, USA) and results were placed and concealed in sequentially numbered, sealed, opaque envelopes by a person who was not connected to the trial. Patients were asked to participate by surgeons or specialised nurses who were involved in the trial. Patients were preoperatively randomised in a 1:1 design to either treatment with normal chewing gum or nicotine chewing gum. The allocated treatment was given to the patients by the nursing staff. Both patients and investigators were blinded for treatment allocation.
Statistical analysis
Only patients who completed the full study period were analysed. Data analysis was carried out using the Statistical Package for the Social Sciences (SPSS Inc., Chicago, USA, version 21.0 for Windows). Demographic data were presented in n (%) and median (interquartile range [IQR]). Categorical variables were compared using the Fisher’s exact test. Continuous variables were compared using the Mann-Whitney U test.
Results
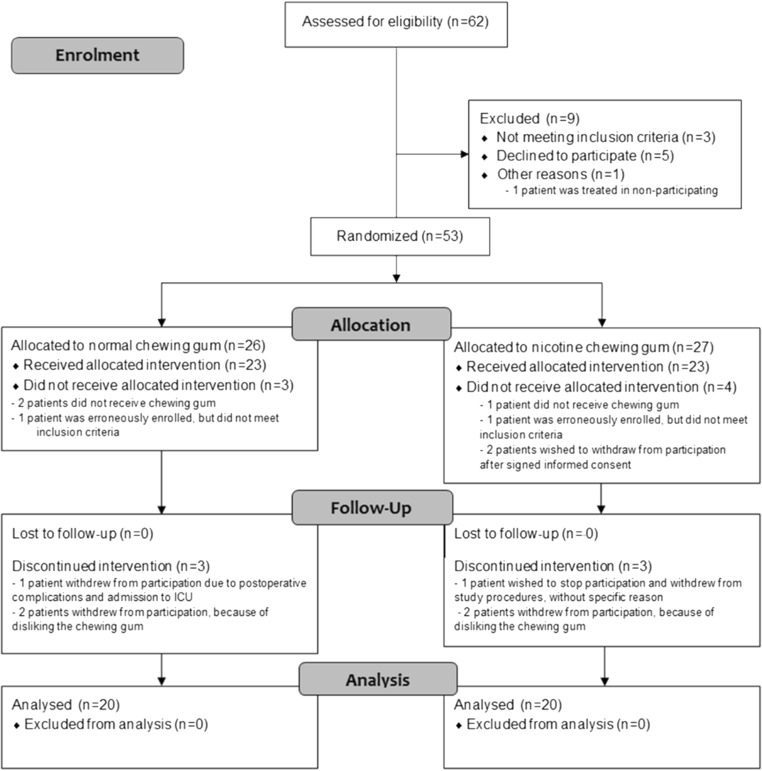
In Fig. 2, the CONSORT flow diagram of the study is shown. Between January 29, 2015 and July 14, 2016, 62 patients were assessed for eligibility. Of these patients, 53 were randomly assigned to the normal chewing gum group or the nicotine chewing gum group. Two patients in each group withdrew from participation, because of disliking the chewing gum. One patient in the normal gum group was unable to continue treatment protocol, because of postoperative complications and ICU admission. The other patients withdrew for other reasons than disliking or being unable to chew the allocated gum. In total, 40 patients were included for data analysis.
Fig. 2.
CONSORT flow diagram
Baseline patient and surgical characteristics were distributed evenly between both groups, without significant differences (Table 1). Urinary catheterisation failed in one patient in the normal gum group and one patient in the nicotine gum group required vasopressors for hemodynamic support during surgery. All patients who were admitted to the intensive care unit (ICU) directly after surgery were transferred to the surgical ward after 1 day.
Table 1.
Baseline patient and surgical characteristics in treatment groups
| Normal gum (n = 20) | Nicotine gum (n = 20) | |
|---|---|---|
| Patient characteristics | ||
| Sex | ||
| Male | 13 (65) | 14 (70) |
| Female | 7 (35) | 6 (30) |
| Age (years) | 67.50 [60.75–74.75] | 69.00 [62.50–70.00] |
| BMI (kg/m2) | 26.91 [23.77–31.61] | 25.02 [23.15–27.67] |
| Smoking | 2 (10) | 4 (20) |
| Diabetes mellitus | 1 (5) | 4 (20) |
| COPD | 2 (10) | 3 (15) |
| Cardiovascular disease | 9 (45) | 5 (25) |
| Corticosteroid use | 3 (15) | 1 (5) |
| Statin use | 4 (20) | 5 (25) |
| Neoadjuvant radiotherapy | 1 (5) | 0 |
| Neoadjuvant chemoradiotherapy | 1 (5) | 0 |
| Previous abdominal surgery | 3 (15) | 3 (15) |
| ASA classification | ||
| ASA I | 4 (20) | 3 (15) |
| ASA II | 13 (65) | 14 (70) |
| ASA III | 3 (15) | 2 (10) |
| ASA IV | 0 | 0 |
| Surgical characteristics | ||
| Type of procedure | ||
| Low anterior resection | 3 (15) | 3 (15) |
| Left hemicolectomy | 3 (15) | 2 (10) |
| Right hemicolectomy | 8 (40) | 6 (30) |
| Sigmoidectomy | 5 (25) | 6 (30) |
| Subtotal colectomy | 1 (5) | 0 |
| Transverse colon resection | 0 | 3 (15) |
| Laparoscopic approach | 20 (100) | 16 (80) |
| Anastomotic technique | ||
| End-to-end | 4 (20) | 2 (10) |
| End-to-side | 1 (5) | 1 (5) |
| Side-to-end | 5 (25) | 5 (25) |
| Side-to-side | 10 (50) | 11 (55) |
| Anastomotic configuration | ||
| Stapled | 12 (60) | 13 (65) |
| Sutured | 8 (40) | 8 (40) |
| Protective ileostomy | 2 (10) | 2 (10) |
| Nasogastric tube | 12 (60) | 13 (65) |
| Intraoperative complications | 1 (5) | 1 (5) |
| >50-mL blood loss | 5 (25) | 9 (45) |
| Duration of surgery (min) | 133 [101–176] | 117 [109–150] |
| Postoperative ICU admission | 1 (5) | 2 (10) |
Data are median [IQR] or n (%)
BMI body mass index, ASA American Society of Anesthesiologists classification, ICU intensive care unit
The time to primary endpoint (as defined earlier) as well as the time to first passage of faeces and flatus and length of stay (LOS) are given in Table 2. No statistically significant differences were found between groups. In both groups, six patients (30%) suffered from PPOI on or after POD6. Furthermore, there was no significant difference in the percentage of resolution of POI on POD1 to 5 (Table 3 and Online Resource Fig. S1). In a subgroup analysis in which all four open procedures were excluded, the time to primary endpoint in the nicotine gum group was shorter, but also not significantly different from the normal gum group (3.00 days [3.00–4.50] vs. 4.50 [3.00–7.25], p = 0.249).
Table 2.
Time to primary endpoint, time to first passage of faeces and flatus, length of stay in days
| Normal gum (n = 20) | Nicotine gum (n = 20) | p value | |
|---|---|---|---|
| Time to primary endpoint (days) | 4.50 [3.00–7.25] | 3.50 [3.00–4.25] | 0.398 |
| Time to first passage of faeces (days) | 3.00 [1.75–5.00] | 3.00 [1.75–4.00] | 0.414 |
| Time to first passage of flatus (days) | 1.00 [1.00–2.25] | 1.00 [1.00–1.00] | 0.454 |
| Length of stay (days) | 5.50 [4.00–8.50] | 4.50 [4.00–6.00] | 0.738 |
Data are median [IQR]
Table 3.
Resolution of POI
| Normal gum (n = 20) | Nicotine gum (n = 20) | p value | |
|---|---|---|---|
| Resolution of POI | |||
| POD1 | 0 | 0 | – |
| POD2 | 0 | 2 (10) | 0.487 |
| POD3 | 6 (30) | 9 (45) | 0.515 |
| POD4 | 11 (55) | 13 (65) | 0.748 |
| POD5 | 14 (70) | 14 (70) | 1.000 |
| POD6 or later | 20 (100) | 20 (100) | 1.000 |
Data are n (%)
Six patients in the normal gum group and six in the nicotine group required a nasogastric tube during their postoperative stay. Three patients in the normal gum group and four in the nicotine gum group required total parental nutrition (TPN). Postoperative complications, reinterventions, readmissions and mortality during the first 30 days after surgery are given in Table 4. No differences were found between both treatment groups. Only one patient in the nicotine gum group had a short period of atrial fibrillation and overall, no myocardial infarction was seen. One patient in each treatment group required blood transfusion. One patient was readmitted because of anastomotic leakage and drainage of an intra-abdominal abscess, one patient was readmitted for adhesion ileus and one for observation of fever of unknown origin. One patient died during primary hospital stay, due to severe small bowel ischemia, caused by venous mesenteric thrombosis. Subgroup analysis for cases without intra-abdominal infectious complications during primary stay showed a time to primary endpoint of 4.00 days (3.00–5.50) vs. 4.50 (4.00–6.00) (p = 0.339) and LOS of 5.00 days (4.00–8.00) vs. 4.50 (4.00–6.00) (p = 0.673), for the normal gum and nicotine gum groups, respectively.
Table 4.
Postoperative complications, reinterventions (surgical and/or radiological), readmissions and mortality (≤30 days)
| Normal gum (n = 20) | Nicotine gum (n = 20) | p value | |
|---|---|---|---|
| Atrial fibrillation | 0 | 1 (5) | 1.000 |
| Fascial dehiscence | 0 | 0 | – |
| Colorectal anastomotic leakage | 2 (10) | 0 | 0.487 |
| Intra-abdominal abscess | 1 (5) | 0 | 1.000 |
| Myocardial infarction | 0 | 0 | – |
| Pneumonia | 1 (5) | 0 | 1.000 |
| Surgical site infection | 4 (20) | 2 (10) | 0.661 |
| Urinary retention | 0 | 1 (5) | 1.000 |
| Urinary tract infection | 2 (10) | 1 (5) | 1.000 |
| Reinterventions (<30 days) | 4 (20) | 2 (10) | 0.661 |
| Readmissions (<30 days) | 3 (15) | 0 | 0.231 |
| Mortality (<30 days) | 0 | 1 (5) | 1.000 |
Data are n (%)
Postoperative opioid use
Fourteen patients in the normal gum group used oral opioids postoperatively, compared to 11 in the nicotine gum group (p = 0.514). Respectively, epidural opioids were used in 16 and 14 patients and a combination of oral and epidural opioids was used in 12 and 9 patients (p = 0.527). One patient in the normal group used a PCA pump and five patients in the nicotine gum group (p = 0.091). Patients in the nicotine gum group used epidural opioids for a significantly longer time (3.00 days [2.00–4.25] vs. 2.00 [1.00–.00], p = 0.006), but duration of oral opioid use did not differ between groups (1.00 day [0–3.50] vs. 1.00 [0–3.00], p = 0.740).
Patient reported outcomes
Fifteen patients who received normal gum filled out their diary, as compared to 16 patients who received nicotine chewing gum (p = 1.000). Pain scores (VAS) were significantly lower in the nicotine gum group on POD3 (1.40 [0.50-] vs. 2.70 [1.50-], p = 0.007), but did not differ on the other postoperative days (Online Resource Fig. S2). No differences were found in patient reported nausea, vomiting, regurgitations, abdominal distension and appetite (Online Resource Fig. S3). Treatment compliance, as based on patient reported chewing gum use, is given in Online Resource Table S1.
Inflammatory parameters
No significant differences were observed in IL-6 levels and white blood cell counts in preoperative samples and POD1 and three samples (Online Resource Fig. S4). CRP levels differed on POD1 in the normal and nicotine gum groups, respectively (71.50 mg/L [35.00–92.75] vs. 94.50 mg/L [58.50–128.25], p = 0.017), but no differences were found in preoperative and POD3 samples (Online resource Fig. S4b). None of the calculated ratios showed differences between both groups (Table 5). On none of the postoperative days, a statistically significant difference was found in systolic and diastolic blood pressure, and heart rate (Online Resource Fig. S5).
Table 5.
Inflammatory parameters (Interleukin-6 (IL-6), C-reactive protein (CRP) and white blood cell (WBC) count
| Normal gum (n = 20) | Nicotine gum (n = 20) | p value | |
|---|---|---|---|
| IL-6 (pg/mL) | |||
| Preoperative | 1088.95 [529.65–1680.70] | 1108.40 [547.18–1732.38] | 0.663 |
| POD1 | 881.80 [516.90–2138.70] | 1047.65 [752.53–1930.10] | 0.883 |
| POD3 | 959.00 [648.90–2043.60] | 987.40 [518.38–2139.75] | 0.940 |
| Ratio | 1.13 [0.99–1.54] | 1.12 [0.89–1.24] | 0.517 |
| CRP (mg/L) | |||
| Preoperative | 2.60 [1.00–4.75] | 3.70 [2.25–23.38] | 0.089 |
| POD1 | 71.50 [35.00–92.75] | 94.50 [58.50–128.25] | 0.017 |
| POD3 | 99.50 [76.25–179.50] | 151.00 [101.75–188.50] | 0.180 |
| Ratio | 45.83 [19.70–83.68] | 33.92 [6.59–79.10] | 0.180 |
| WBC count (×109/L) | |||
| Preoperative | 7.10 [3.90–9.60] | 6.60 [6.25–9.10] | 0.477 |
| POD1 | 12.30 [7.65–15.45] | 12.30 [10.85–13.85] | 0.865 |
| POD3 | 8.50 [4.95–10.60] | 9.00 [7.15–11.85] | 0.583 |
| Ratio | 1.08 [0.95–1.59] | 1.15 [0.99–1.77] | 0.734 |
Data are median [IQR]
Discussion
This study was the first to investigate the role of nicotine chewing gum for the prevention of postoperative ileus by assessing its clinical efficacy and safety in patients undergoing elective colorectal surgery. By performing this parallel-group, double-blind, randomised, controlled pilot study, it was not possible to prove the beneficial effect of nicotine chewing gum, as compared to normal chewing gum. We hypothesised that the combination of perioperative gum chewing, with the potential beneficial effects of nicotine, could improve the resolution of POI, but although the median time to primary endpoint seemed shorter in the nicotine gum group (3.50 days vs. 4.50), the difference was not statistically significant. Moreover, an equal number of patients (n = 6) in each group suffered from PPOI and LOS did not differ significantly between both groups. Open procedures are known to worsen POI outcomes in colorectal surgery as compared to the laparoscopic approach [27]. Since all four open procedures in this study were in the nicotine gum group, a subgroup analysis was performed in which these four procedures were excluded. This showed an improvement in median time to primary endpoint (3.50 vs. 3.00 days) and length of stay (4.50 vs. 4.00 days) in the nicotine gum group, but although these open procedures influenced the outcomes, they did not provide a complete explanation for the lack of efficacy, since a significant difference between both groups was still not found.
A limitation of this study might be the relatively small sample size of 40 patients in total. However, our sample size calculation was based on the results of Asao et al. [24] who showed significant effects of chewing gum in a total of 19 patients. With a larger sample size than we initially calculated, we might have had more power to make a better distinguishment between the effects of sham feeding with normal chewing gum and the hypothesised additional effects of sham feeding with nicotine chewing gum.
Experimental studies have shown that a specific α7-nAChR agonist (AR-R17779) ameliorates POI in rats and that stimulation of the α7-nAChR improves survival of sepsis in rats [11, 28]. Nevertheless, as clinical results of nicotine use for POI after colorectal surgery were still lacking, the second aim of this pilot study was to evaluate the safety of nicotine chewing gum for the purpose of preventing POI. Because of concerns of systemic effects induced by nicotine administration, particularly cardiovascular complications [29], we decided to use Nicorette® 2 mg. This relatively low dose might have potentially been another reason for the lack of efficacy of the nicotine chewing gum in this study. However, the use of Nicorette® 2 mg in a perioperative setting of elective colorectal surgery, which has not been described in the literature before, seems to be safe.
No myocardial infarctions were registered in this study and only one patient had one short period of atrial fibrillation. This patient did receive nicotine chewing gum, but was known to have had previous episodes of paroxysmal atrial fibrillation. These findings are consistent with a previously published Cochrane Review, concluding that there is no evidence that nicotine replacement therapy increases the risk of heart attacks [30]. Moreover, apart from cardiovascular complications, no differences were found in major and minor postoperative complications, reinterventions, readmissions and mortality between the normal and nicotine chewing gum groups. Overall, only one patient—enrolled in the nicotine chewing gum group—died during primary hospital stay on the 15th postoperative day. We did not consider usage of the nicotine chewing gum related to the cause of death, which was a result of intestinal ischemia caused by venous mesenteric thrombosis.
To combine the potential benefits of sham feeding with chewing gum and nicotine, we chose to use nicotine chewing gum in this pilot study. The reason being that this facilitated the possibility for a simple blinded comparison with normal chewing gum. However, a limitation of nicotine administration through chewing gum is that a sufficient release of nicotine is dependent on treatment compliance of the patient. All patients in this study were asked to report their use of chewing gum in the patient diary and it can be concluded that compliance to chewing the allocated gum is decreasing in the first three postoperative days. Conceivably, a more constant way of nicotine administration, such as the nicotine patches which Habib et al. [15] used in their study, might have given a more continuous release of nicotine.
If indeed the effective dose of nicotine would be too low in some patients, either due to low administered dose, insufficient exposure to the nicotine chewing gum or both, this might account for the absence of significant differences between the measured clinical and inflammatory parameters in both groups.
Il-6 and CRP levels, as well as WBC, were analysed in venous blood samples, as markers of the immune response, because it was hypothesised that this response would be less pronounced in patients in the nicotine chewing gum group. Overall lower levels of any of the three inflammatory parameters (IL-6, CRP and WBC) were not seen in this group and no differences were found, when compared to the normal chewing gum group, except for a significant difference of CRP levels on POD1. Sparreboom et al. [31] have concluded in their meta-analysis that levels of pro-inflammatory cytokines, such as IL-6, were higher in peritoneal samples as compared to serum samples after colorectal surgery, which might also explain why significant changes and differences were not detected in our serum samples. Moreover, postoperative infectious complications, such as surgical site infections and pneumonia, could have affected the levels of these inflammatory parameters in both groups.
No differences in patient reported outcomes, such as nausea, vomiting, regurgitations, abdominal distension and appetite, were found. However, patient reported pain scores were significantly lower in the nicotine gum group as compared to the normal gum group on POD3 (1.40 [0.50-] vs. 2.70 [1.50-], p = 0.007). Although promising, this difference could partially be explained by the fact that patients in this group received epidural opioids for a significantly longer period of time (3.00 days [2.00–4.25] vs. 2.00 [1.00–.00], p = 0.006). However, the exact reason for a longer use of epidural opioids remains uncertain, since the decision to stop epidural anaesthesia in this study was made by the anaesthesiologist, who was blinded for patient allocation, with the aim to stop as early as possible, and preferably on or before POD2–3. These decisions were not registered prospectively. Furthermore, no significant differences in patient outcomes were found between both groups which might explain the extended requirement for epidural opioids in the nicotine gum group.
In conclusion, this study is the first to evaluate the potential beneficial role of nicotine chewing gum for another purpose than NRT in a randomised and double-blind clinical setting. Although the hypothesised potential benefits of nicotine chewing gum, as a cheap and readily available treatment option, seemed promising, no evident beneficial effects were found. This might be attributed to the sample size, the dose of the nicotine chewing gum and insufficient patient compliance to the allocated chewing gum. People in the nicotine chewing gum group seemed to experience less pain in the first three postoperative days, but a difference could only be proven on POD3. Therefore, more data on the effects of nicotine gum on bowel recovery after surgery are awaited [32]. Furthermore, this study provides positive new insights on the safety of nicotine chewing in the setting of patients undergoing elective colorectal surgery. Future research should focus on other means of nicotine administration to patients undergoing colorectal surgery (e.g. patches), whether or not combined with normal chewing gum, and in higher doses, to further assess its effects on gastrointestinal recovery after colorectal surgery.
Electronic supplementary material
(DOCX 82 kb)
Resolution of POI (a, b) (GIF 2 kb)
Postoperative patient reported pain scores (Visual Analogue Scale) (GIF 1 kb)
Postoperative patient reported outcomes: (a) Nausea, (b) Vomiting, (c) Regurgitations, (d) Abdominal distension, (e) Appetite (GIF 3 kb)
Inflammatory parameters (a) Interleukin-6 (IL-6) levels, (b) C-reactive protein (CRP) levels, (c) white blood cell count (GIF 4 kb)
Postoperative outcomes: (a) Systolic blood pressure, (b) Diastolic blood pressure, (c) Heart rate. Normal gum = ● (dots), nicotine gum = ■ (squares) (GIF 2 kb)
Acknowledgements
We thank all patients for their participation in this study, and all involved staff members for their efforts, in particular, A. de Boer, I. Sendar, A. van der Spek, W. Lee and A. van Duuren. This pilot study was funded by Stichting Coolsingel (Rotterdam, The Netherlands).
Compliance with ethical standards
In accordance with the Dutch law on medical research in humans, this study was approved by the Institutional Review Board of the Erasmus University Medical Center, Rotterdam, The Netherlands and the Institutional Review Boards of both participating hospitals. Patients gave their written consent after receiving oral and written information. This study was investigator initiated. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Source of funding
Stichting Coolsingel, Schiekade 75, 3033 BD, Rotterdam, The Netherlands, E-mail: secretariaat@stichtingcoolsingel.nl.
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Electronic supplementary material
The online version of this article (doi:10.1007/s00384-017-2839-z) contains supplementary material, which is available to authorized users.
References
- 1.Lubbers T, Buurman W, Luyer M. Controlling postoperative ileus by vagal activation. World J Gastroenterol. 2010;16(14):1683–1687. doi: 10.3748/wjg.v16.i14.1683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gero D, Gié O, Hübner M, Demartines N, Hahnloser D. Postoperative ileus: in search of an international consensus on definition, diagnosis, and treatment. Langenbeck’s Arch Surg. 2016;402:149–158. doi: 10.1007/s00423-016-1485-1. [DOI] [PubMed] [Google Scholar]
- 3.Wolff BG, Michelassi F, Gerkin TM, Techner L, Gabriel K, Du W, Wallin BA, Alvimopan Postoperative Ileus Study G Alvimopan, a novel, peripherally acting mu opioid antagonist: results of a multicenter, randomized, double-blind, placebo-controlled, phase III trial of major abdominal surgery and postoperative ileus. Ann Surg. 2004;240(4):728–734. doi: 10.1097/01.sla.0000141158.27977.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Artinyan A, Nunoo-Mensah JW, Balasubramaniam S, Gauderman J, Essani R, Gonzalez-Ruiz C, Kaiser AM, Beart RW., Jr Prolonged postoperative ileus-definition, risk factors, and predictors after surgery. World J Surg. 2008;32(7):1495–1500. doi: 10.1007/s00268-008-9491-2. [DOI] [PubMed] [Google Scholar]
- 5.Schuster R, Grewal N, Greaney GC, Waxman K. Gum chewing reduces ileus after elective open sigmoid colectomy. Arch Surg. 2006;141(2):174–176. doi: 10.1001/archsurg.141.2.174. [DOI] [PubMed] [Google Scholar]
- 6.Luckey A, Livingston E, Tache Y. Mechanisms and treatment of postoperative ileus. Arch Surg. 2003;138(2):206–214. doi: 10.1001/archsurg.138.2.206. [DOI] [PubMed] [Google Scholar]
- 7.Bauer AJ, Boeckxstaens GE. Mechanisms of postoperative ileus. Neurogastroenterol Motil. 2004;16(Suppl 2):54–60. doi: 10.1111/j.1743-3150.2004.00558.x. [DOI] [PubMed] [Google Scholar]
- 8.Boeckxstaens GE, de Jonge WJ. Neuroimmune mechanisms in postoperative ileus. Gut. 2009;58(9):1300–1311. doi: 10.1136/gut.2008.169250. [DOI] [PubMed] [Google Scholar]
- 9.Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg. 1998;228(5):652–663. doi: 10.1097/00000658-199811000-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Turler A, Moore BA, Pezzone MA, Overhaus M, Kalff JC, Bauer AJ. Colonic postoperative inflammatory ileus in the rat. Ann Surg. 2002;236(1):56–66. doi: 10.1097/00000658-200207000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.The FO. Boeckxstaens GE, Snoek SA, Cash JL, Bennink R, Larosa GJ, van den Wijngaard RM, Greaves DR, de Jonge WJ. Activation of the cholinergic anti-inflammatory pathway ameliorates postoperative ileus in mice. Gastroenterology. 2007;133(4):1219–1228. doi: 10.1053/j.gastro.2007.07.022. [DOI] [PubMed] [Google Scholar]
- 12.van der Zanden EP, Snoek SA, Heinsbroek SE, Stanisor OI, Verseijden C, Boeckxstaens GE, Peppelenbosch MP, Greaves DR, Gordon S, De Jonge WJ. Vagus nerve activity augments intestinal macrophage phagocytosis via nicotinic acetylcholine receptor alpha4beta2. Gastroenterology. 2009;137(3):1029–1039. doi: 10.1053/j.gastro.2009.04.057. [DOI] [PubMed] [Google Scholar]
- 13.Wang H, Yu M, Ochani M, Amella CA, Tanovic M, Susarla S, Li JH, Wang H, Yang H, Ulloa L, Al-Abed Y, Czura CJ, Tracey KJ. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature. 2003;421(6921):384–388. doi: 10.1038/nature01339. [DOI] [PubMed] [Google Scholar]
- 14.Damaj MI, Meyer EM, Martin BR. The antinociceptive effects of alpha7 nicotinic agonists in an acute pain model. Neuropharmacology. 2000;39(13):2785–2791. doi: 10.1016/S0028-3908(00)00139-8. [DOI] [PubMed] [Google Scholar]
- 15.Habib AS, White WD, El Gasim MA, Saleh G, Polascik TJ, Moul JW, Gan TJ. Transdermal nicotine for analgesia after radical retropubic prostatectomy. Anesth Analg. 2008;107(3):999–1004. doi: 10.1213/ane.0b013e31816f2616. [DOI] [PubMed] [Google Scholar]
- 16.Flood P, Daniel D. Intranasal nicotine for postoperative pain treatment. Anesthesiology. 2004;101(6):1417–1421. doi: 10.1097/00000542-200412000-00023. [DOI] [PubMed] [Google Scholar]
- 17.Behm B, Stollman N. Postoperative ileus: etiologies and interventions. Clin Gastroenterol Hepatol. 2003;1(2):71–80. doi: 10.1053/cgh.2003.50012. [DOI] [PubMed] [Google Scholar]
- 18.Vasquez W, Hernandez AV, Garcia-Sabrido JL. Is gum chewing useful for ileus after elective colorectal surgery? A systematic review and meta-analysis of randomized clinical trials. J Gastrointest Surg. 2009;13(4):649–656. doi: 10.1007/s11605-008-0756-8. [DOI] [PubMed] [Google Scholar]
- 19.Chan MK, Law WL. Use of chewing gum in reducing postoperative ileus after elective colorectal resection: a systematic review. Dis Colon Rectum. 2007;50(12):2149–2157. doi: 10.1007/s10350-007-9039-9. [DOI] [PubMed] [Google Scholar]
- 20.Purkayastha S, Tilney HS, Darzi AW, Tekkis PP. Meta-analysis of randomized studies evaluating chewing gum to enhance postoperative recovery following colectomy. Arch Surg. 2008;143(8):788–793. doi: 10.1001/archsurg.143.8.788. [DOI] [PubMed] [Google Scholar]
- 21.Li S, Liu Y, Peng Q, Xie L, Wang J, Qin X. Chewing gum reduces postoperative ileus following abdominal surgery: a meta-analysis of 17 randomized controlled trials. J Gastroenterol Hepatol. 2013;28(7):1122–1132. doi: 10.1111/jgh.12206. [DOI] [PubMed] [Google Scholar]
- 22.Noble EJ, Harris R, Hosie KB, Thomas S, Lewis SJ. Gum chewing reduces postoperative ileus? A systematic review and meta-analysis. Int J Surg. 2009;7(2):100–105. doi: 10.1016/j.ijsu.2009.01.006. [DOI] [PubMed] [Google Scholar]
- 23.Short V, Herbert G, Perry R, Atkinson C, Ness AR, Penfold C, Thomas S, Andersen HK, Lewis SJ. Chewing gum for postoperative recovery of gastrointestinal function. Cochrane Database Syst Rev. 2015;2:CD006506. doi: 10.1002/14651858.CD006506.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Asao T, Kuwano H, Nakamura J, Morinaga N, Hirayama I, Ide M. Gum chewing enhances early recovery from postoperative ileus after laparoscopic colectomy. J Am Coll Surg. 2002;195(1):30–32. doi: 10.1016/S1072-7515(02)01179-1. [DOI] [PubMed] [Google Scholar]
- 25.Wu Z, Boersema GS, Jeekel J, Lange JF. Nicotine gum chewing: a novel strategy to shorten duration of postoperative ileus via vagus nerve activation. Med Hypotheses. 2014;83(3):352–354. doi: 10.1016/j.mehy.2014.06.011. [DOI] [PubMed] [Google Scholar]
- 26.van Bree SH, Bemelman WA, Hollmann MW, Zwinderman AH, Matteoli G, El Temna S, The FO. Vlug MS, Bennink RJ, Boeckxstaens GE. Identification of clinical outcome measures for recovery of gastrointestinal motility in postoperative ileus. Ann Surg. 2014;259(4):708–714. doi: 10.1097/SLA.0b013e318293ee55. [DOI] [PubMed] [Google Scholar]
- 27.Holte K, Kehlet H. Postoperative ileus: a preventable event. Br J Surg. 2000;87(11):1480–1493. doi: 10.1046/j.1365-2168.2000.01595.x. [DOI] [PubMed] [Google Scholar]
- 28.Wang H, Liao H, Ochani M, Justiniani M, Lin X, Yang L, Al-Abed Y, Wang H, Metz C, Miller EJ, Tracey KJ, Ulloa L. Cholinergic agonists inhibit HMGB1 release and improve survival in experimental sepsis. Nat Med. 2004;10(11):1216–1221. doi: 10.1038/nm1124. [DOI] [PubMed] [Google Scholar]
- 29.Thomas GA, Rhodes J, Mani V, Williams GT, Newcombe RG, Russell MA, Feyerabend C. Transdermal nicotine as maintenance therapy for ulcerative colitis. N Engl J Med. 1995;332(15):988–992. doi: 10.1056/NEJM199504133321503. [DOI] [PubMed] [Google Scholar]
- 30.Stead LF, Perera R, Bullen C, Mant D, Hartmann-Boyce J, Cahill K, Lancaster T. Nicotine replacement therapy for smoking cessation. Cochrane Database Syst Rev. 2012;11:CD000146. doi: 10.1002/14651858.CD000146.pub4. [DOI] [PubMed] [Google Scholar]
- 31.Sparreboom CL, Wu Z, Dereci A, Boersema GS, Menon AG, Ji J, Kleinrensink GJ, Lange JF. Cytokines as early markers of colorectal anastomotic leakage: a systematic review and meta-analysis. Gastroenterol Res Pract. 2016;2016:3786418. doi: 10.1155/2016/3786418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Weiss E Cleveland clinic florida does nicotine gum enhance bowel recovery after colorectal surgery? In: In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2016 Dec 30] Available from: https://clinicaltrials.gov/ct2/show/NCT01662115 NLM Identifier: NCT01662115
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX 82 kb)
Resolution of POI (a, b) (GIF 2 kb)
Postoperative patient reported pain scores (Visual Analogue Scale) (GIF 1 kb)
Postoperative patient reported outcomes: (a) Nausea, (b) Vomiting, (c) Regurgitations, (d) Abdominal distension, (e) Appetite (GIF 3 kb)
Inflammatory parameters (a) Interleukin-6 (IL-6) levels, (b) C-reactive protein (CRP) levels, (c) white blood cell count (GIF 4 kb)
Postoperative outcomes: (a) Systolic blood pressure, (b) Diastolic blood pressure, (c) Heart rate. Normal gum = ● (dots), nicotine gum = ■ (squares) (GIF 2 kb)