Abstract
An epidemic of obesity over the past three decades increases the risk of chronic and infectious diseases for adults and children alike. Within the past few years, obesity has been shown to impair the adaptive immune response to infection through alterations in T cell functioning. Growing evidence suggests that perturbations in T cell metabolism drives this stunted immune response, stemming from nutrient, hormone and adipokine dysregulation in the obese. In this review, recent findings in the fields of obesity and T cell mediated immunity demonstrate a unique relationship between altered mechanisms of T cell metabolic homeostasis and plasticity of adaptive immune responses in the obese setting.
Introduction
The immune response to infection involves a complex orchestra of diverse cell types, including dendritic cells, macrophages, natural killer cells, and B and T cells.
As one of the most prominent and critical players in the response to infection, subsets of T cells range in activity from supporting the function and activation of other immune cells, as well as T cells themselves, to producing pro and anti-inflammatory proteins. Cytotoxic T cell subsets are instrumental in the elimination of pathogen-infected host cells. This diversified set of immunologically adaptive immune cells plays a critical and central role in combating pathogens. A number of T cell subset knockout models have demonstrated that removal of T cells during an infectious challenge often results in higher rates of morbidity and mortality. For many years, T cell activation and function was believed to occur from a combination of antigen recognition, subsequent signaling cascades and micro-environmental cues [1,2]. However, recent studies have clearly demonstrated that the cellular metabolism of the T cell is also a key player affecting T cell differentiation, proliferation, function and its ultimate fate [3–8].
Reports of how metabolic fuels such as glucose, amino acids and fatty acids elicit distinct metabolic profiles depending on cell state (naïve, effector or memory) and subtype (Th1, Th2, Th17, Treg, etc.) [4,9–11], has led to a revolution in the understanding of T cell driven immunity. Furthermore, it highlights the metabolic plasticity of T cells to respond to the energetic and biosynthetic demands required to successfully fight infection. For primers on T cell metabolism, refer to the excellent reviews by MacIver et al. [4] and Buck et al. [5].
Although T cells respond to antigenic challenge by altering their metabolic state, what is not as well understood is how metabolic conditions may alter their ability to function. One such metabolic condition that may have a profound effect on T cell function is obesity. Obesity has classically been characterized as a state of excess adiposity and is associated with chronic inflammation and metabolic dysfunction such as hyperglycemia, hyperleptinemia and hormone resistance [12]. These uncontrolled metabolic conditions can lead to the development of chronic diseases such as type II diabetes, kidney disease, cardiovascular disease and specific cancers [13]. However, recent data from our lab and others demonstrates a link between obesity and increased incidences of infectious diseases, most likely through impaired cellular immune responses [13,14].
Considering recent findings on how T cell metabolism drives cellular function and survival, understanding how obesity impacts these processes in T cells remains critical. This review examines recent works in the fields of obesity and T cell mediated immunity; exploring possible mechanisms of inflammation, hormone and adipokine regulation, and senescence to understand the altered response to infection in obesity through changes in T cell metabolism.
T cell metabolism supports cell survival and function
T cells display unique metabolic flexibility unlike other cells in the body. Upon stimulation of the T cell receptor (TCR) and costimulatory receptors, T cells undergo a dramatic metabolic shift from a quiescent to activated state, highlighted by a change from primarily catabolic to anabolic metabolism (Figure 1) [4]. This shift from oxidative phosphorylation to glycolysis produces ATP and supports the generation of nucleotides and amino acids for the production of daughter cells necessary to mount an effective immune response [15]. Importantly, this glycolytic shift supports effector functions, which varies depending on T cell subtype [3,11,16]. Following clearance of the infection, the majority of effector T cells undergo apoptosis with a small subset remaining as long-lived memory T cells [17,18]. These memory T cells revert back to a relatively quiescent catabolic state. However, unlike their naïve cell counterparts, memory T cells quickly respond to antigenic challenge upon re-exposure through elevated metabolic activity, increased proliferation and production of cytokines [19,20].
Figure 1.

T cell metabolism and state of quiescent and activated T cells. Quiescent T cells utilize oxidative respiration of glucose-derived pyruvate as well as fatty acids and amino acids to produce ATP through the TCA cycle and electron transport chain. This energy production supports immune surveillance and homeostasis. Upon stimulation of the T cell receptor (TCR) and costimulatory receptors, T cells upregulate glycolysis and glutamine oxidation while reducing fatty acid oxidation to support cell growth, differentiation and the production of daughter cells. Following clearance of the pathogen, the majority of T cells undergo apoptosis with a subset surviving as memory T cells, which return to a quiescent state dependent on oxidative phosphorylation of fatty acids.
These different metabolic states of T cells require signaling molecules to support homeostasis, effector function and survival. Cytokines such as IL-7 and IL-15 support the catabolic survival functions of naïve and memory T cells, respectively [16,21,22]. Other cytokines, such as IL-2, support proliferation upon stimulation [23], while interferon gamma (IFN-γ) promotes pro-inflammatory T cell subtypes like Th1 and cells of innate immunity [24]. T cell subsets also help to down-regulate the inflammatory response once pathogen clearance has occurred. For example, regulatory T cells (Treg) and the cytokine IL-10 help to suppress T cell activation and proliferation [24,25].
Aside from cytokine and growth factor signals, nutrients and hormone signaling also influence T cell metabolism [8,23,26]. These signals have been shown to affect regulatory pathways controlled by enzymes such as the mammalian target of rapamycin (mTOR) and AMP-activated protein kinase (AMPK) to influence cell growth/proliferation or homeostasis [27,28]. In the absence of these extrinsic signals, T cells lose their ability to maintain homeostasis and eventually succumb to apoptosis through Bcl-2 mediated cell death [7,28].
Obesity impairs the immune response to infection
As of 2014, for the first time in recorded history, obese adults outnumber underweight adults worldwide, with the global prevalence of obesity equaling 10.8% in men and 14.9% in women [29]. In the United States, obese adults comprise 36.5% of the population [30]. Obesity has been linked with increased incidences of infectious diseases, including periodontal infections, influenza, bacterial pneumonia, nosocomial and surgical site infections, among others [13,31]. Furthermore, obesity has been shown to impair immunological responses in both adults and children [32–34].
Following the first influenza pandemic of the 21st century, obesity was identified as an independent risk factor for increased morbidity and mortality from pandemic H1N1 infection [31]. Our laboratory studied the response to influenza vaccination in obese adults and found that increasing BMI was associated with greater declines in influenza-specific antibody titers one-year post vaccination [33]. In addition, influenza-stimulated T cells from obese adults were less functional and less activated compared with T cells from vaccinated healthy weight adults [35].
Obesity has been shown to reduce circulating levels of γδ T cells, as well as impair their function through reduced IL-2 receptor expression and IFN-γ production [36]. Moreover, obesity has been implicated in reduced function of Treg cells in obese patients with asthma [37]. Finally, growing evidence suggests obesity impairs immune responses to vaccines such as hepatitis B (HB), hepatitis A (HA), rabies and tetanus, as well as increases risk of several bacterial infections [38].
Despite this growing evidence supporting the notion that obesity impairs immunological responses to infection, it is worth noting that some studies have found a protective effect of obesity against infection. Roth et al., in their review of over 20 epidemiological studies, found that obesity was associated with better outcomes from infections such as tuberculosis, community-acquired pneumonia and sepsis [39]. They argue that aspects of the metabolic syndrome often found in obesity provide an advantageous niche for immune cells to fight off infection. While there is controversy between findings on obesity’s impact on the immune response to infection, it is important to note that obesity is a multifactorial condition that impacts a variety of tissue and organ systems.
Obesity alters the metabolome during infection
Studies using murine models of obesity have shown great similarity to obesity in humans, providing a translational model to study this complex condition [40]. Recent studies have shown altered metabolic profiles in diet-induced and genetically obese mice following influenza virus challenge in vivo. Using 1H NMR and global liquid chromatography-mass spectrometry, Milner et al. reported specific alterations in the metabolic profiles of influenza-infected diet-induced obese mice when compared with infected lean mice in serum, liver, lung, mesenteric white adipose tissue, urine, feces and bronchoalveolar lavage fluid [41,42]. Changes in metabolites included increased levels of fatty acid, cholesterol and phospholipids in lung tissues isolated from obese mice compared to lean mice, correlating with increased lung damage and mortality observed in the infected obese mice [42].
Notably, obese mice showed significant fold increases in glutamyl-proline, tetrahydrocortisol, 3-hydroxybutyric acid and numerous acyl-carnitine metabolites in lung tissues [42], suggesting differential metabolism in these tissues may be driving immune dysregulation in the influenza-infected lung. Additionally, diet-induced obese mice also had a 55-fold increase in p-cresol sulfate concentrations in lungs at 4 days post infection compared to lean mice [42]. P-cresol sulfate, a metabolite produced by gut microbiota during secondary metabolism of p-cresol, accumulates during kidney failure and can impact cell function [42]. This increase in secondary metabolites in obese mice suggests that obese microbiome may differ from lean, and thus contribute to differences in immune function.
In order to differentiate between the dietary vs. obesity effects, mice lacking hypothalmic leptin receptors (LepRH−/−) were utilized in the influenza infection model. LepRH−/− knockout mice were established by crossing fully floxed leptin receptor mice with Cre transgene expressing C57BL/6J-Tg(Nkx2-1-cre)2S mice driven by the Nkx2.1 promoter. These transgenic mice lacked leptin receptor signaling in hypothalamic neurons, resulting in obesity from excess consumption of a low fat chow diet; therefore removing the influence of high fat diet on immune function. Compared to lean controls (LepRHFlox/Flox consuming an identical diet), obese mice that gained weight on the chow diet also exhibited altered metabolic profiles. Similar to high fat fed diet-induced obese mice, the LepRH−/− obese mice had altered fatty acid, cholesterol and nucleic acid metabolites in urine and lung tissues following influenza virus infection [42]. Again, these variations in metabolites correlated with greater lung pathology and inflammation, as well as reduced levels of quiescent and activated CD4+ and Treg cells in the lung and bronchoalveolar lavage fluid during the immune response to influenza virus infection [42]. These studies suggest that obesity itself, not the diet, alters metabolites both in circulation and in tissue specific regions impacted by infection. This work supports our proposal that obesity alters the metabolic landscape of the host, thus impairing T cell function, leading to increased susceptibility to infectious disease.
Adiopocytes in the obese state promote inflammatory T cell activation through altered metabolism
Another theory as to how obesity impairs the T cell response to infection involves the distorted cytokine and adipokine milieu brought about by excess adiposity. Leptin, a hormone involved in energy homeostasis, has been shown to be essential for glucose uptake in effector T cells [26]. Upregulation of Glut1 receptors by leptin signaling under normal conditions represents a bioenergetic advantage for T cells to prevent suppressed proliferative and functional responses when glucose concentrations become limited [7,23,26]. However, in obesity, when leptin secretion becomes systemic and chronic, leptin signaling may lead to altered CD4+ T cell differentiation. Leptin signaling has been shown to promote pro-inflammatory T cell subtypes, Th1 and Th17 [26,43,44], suggesting leptin plays a critical role in the development of an inflammatory adipose tissue microenvironment. Leptin’s role in modulating T cell repertoires, leading to inflammatory adipose tissue microenvironments is further supported with the identification that pro-inflammatory CD8+ T cells precede adipose tissue macrophage (ATM) infiltration in visceral adipose tissue, with marked declines in CD4+ helper T cell and Treg cell populations in obese mice compared to lean [45].
Infiltration of T cells in adipose tissue has received attention for its suggested role in the development of insulin resistance [46]. Recent findings by Morris et al. identified a mechanism by which ATMs function as antigen presenting cells (APCs) to regulate the activation of CD4+ T cells in mice [47]. This finding, reproduced by Cho et al., identified a novel MHCII-dependent activation loop between CD4+ T cells and ATMs which supports T cell driven metainflammation in adipose tissue in mice [48].
However, large human adipocytes were also shown to activate CD4+ T cells through MHCII-upregulation, thereby acting as APCs to stimulate T cell inflammatory effector functions [49]. This activation of adipose tissue associated T cells by MHCII-mediated APCs and leptin costimulation supports the notion of an inflammatory microenvironment observed in obese adipose tissue. These findings suggest the possibility that obesity leads to suppressed T cell response to infection through altered T cell populations caused by premature activation to pro-inflammatory T cell subtypes.
Adipocytes in the obese state promote T cell senescence
Senescence, typically associated with aging, is described as a fate in which cellular proliferation becomes halted but metabolic activity and function remains [50]. This cell phenotype represents a state of cellular exhaustion, supported by chronic activation of Akt and mTOR, thereby supporting cytokine production but limiting proliferation [50,51]. Recently, obesity was proposed to increase T cell senescence in visceral adipose tissue (VAT) of high fat fed obese mice [52]. These mice displayed increased accumulation of CD153+PD-1+CD44+CD4+ T cells in VAT, resembling the activity of senescent associated T cells, with increased osteopontin secretion and VAT inflammation [52]. Previously, osteopontin has been shown to be a Th1 promoting cytokine that supports pro-inflammatory function [53].
This increased production of PD-1 (programmed cell death-1), an exhaustion/tolerance marker, [54], signals an exhausted state of visceral adipose T cells. This role of PD-1 as an immune regulator of TCR activation in effector T cells is evidenced by the establishment of PD-1 expressing CD8+ effector memory T cells, and not central memory T cells [55]. Furthermore, strength of PD-1 expression is tied to signaling activation and effector function in T cells, such that high expression of PD-1 is needed for cell exhaustion but only low levels are required to disrupt some functions such as IL-2 or TNF-α secretion [56].
Finally, CD4+ T cell metabolism was shown to alter PD-1 expression in mice cultured with glucose versus galactose [57]. This alteration in T cell metabolism from aerobic glycolysis to respiration resulted in differences in glyceride 3-phosphate dehydrogenase expression, correlating with PD-1 expression and subsequent impairment in IFN-γ production [57]. This connection between aerobic glycolysis of CD4+ T cells and PD-1 expression demonstrates a novel mechanism through which T cell function is influenced by nutrition. How long-term exposure to obesogenic conditions impact gene expression remains unresolved. However, some recent studies suggest that obesity can lead to the hypermethylation of lymphocyte DNA in humans and animals [58,59], thereby proposing an epigenetic component that supports the conditions of obesity associated T cell senescence.
Conclusion
Despite the lack of direct evidence linking obesity and altered T cell metabolism with impaired immune response to infection, foundations from several studies over recent years support the hypothesis that obesity disrupts T cell metabolism, resulting in impaired function. Altered metabolism of the obese host has been shown both in the steady state and during infection. Hormone resistance, inflammation and alterations in nutrient levels all influence T cell activation, function and survival. A surfeit of nutrients such as glucose and fatty acids, along with excess leptin production in the obese state, may elicit the activation of T cells in the absence of specific pathogens, thus, skewing naïve and memory cells towards inflammatory Th1 & Th17 subtypes, while reducing anti-inflammatory Treg repertories (Figure 2). The response to infection in an obesogenic environment most likely would result in disrupted T cell metabolism through increased glycolytic and oxidative flux and cause impaired T cell response by promoting inflammation and reducing anti-inflammatory immune surveillance. How chronic exposure to excess nutrients, hormones and inflammation influences gene regulation in lymphocytes remains unknown. Obesity as a complex multifactorial condition represents a growing and serious health problem, warranting further study.
Figure 2.
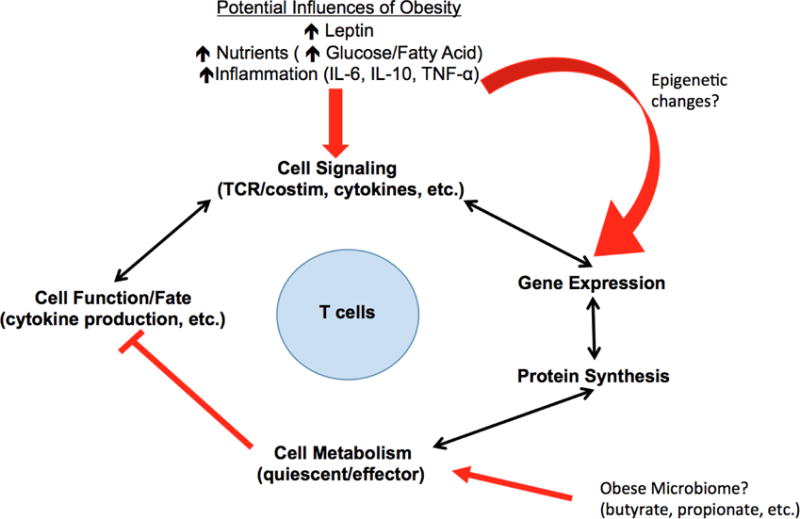
Mechanism of impairment of normal T cell signaling and response to infection in obesity. Nutrients, leptin and inflammation signals cause a disruption of normal T cell metabolism, resulting in impaired cellular functions. Long-term exposure to these extrinsic signals alters the metabolic profile of T cells, impacting their feedback of cytokine secretion and functional response to infection. Whether or not long-term exposure results in epigenetic changes, or metabolite influence from obese microbiota has yet to be determined.
Highlights.
Obesity impairs immune response through altered T cell activation and function
Obese adipocytes promote pro-inflammatory milieu altering T cell repertoire
Excess nutrients and adipokines may activate T cells in pathogen-free environments
Acknowledgments
Funding: This work was supported by the National Institutes of Health [R01 A1078090].
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflicts of Interest: The authors have declared no conflicts of interest.
References and recommended reading
Papers of interest published within the period of review are denoted as:
• of special interest
•• of outstanding interest
- 1.van Panhuys N, Klauschen F, Germain RN. T-cell-receptor-dependent signal intensity dominantly controls CD4(+) T cell polarization In Vivo. Immunity. 2014;41:63–74. doi: 10.1016/j.immuni.2014.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brownlie RJ, Zamoyska R. T cell receptor signalling networks: branched, diversified and bounded. Nat Rev Immunol. 2013;13:257–269. doi: 10.1038/nri3403. [DOI] [PubMed] [Google Scholar]
- 3.Michalek RD, Gerriets VA, Jacobs SR, Macintyre AN, MacIver NJ, Mason EF, Sullivan SA, Nichols AG, Rathmell JC. Cutting edge: distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J Immunol. 2011;186:3299–3303. doi: 10.4049/jimmunol.1003613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.MacIver NJ, Michalek RD, Rathmell JC. Metabolic regulation of T lymphocytes. Annu Rev Immunol. 2013;31:259–283. doi: 10.1146/annurev-immunol-032712-095956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5•.Buck MD, O’Sullivan D, Pearce EL. T cell metabolism drives immunity. J Exp Med. 2015;212:1345–1360. doi: 10.1084/jem.20151159. The authors provide a concise and expansive sumary of current knowledge on T cell metabolism and its contribution to the adaptive immune response. Topics discussed include metabolic states of T cells by different activation states and subsets as well as substrate utilization and function. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pearce EL, Poffenberger MC, Chang CH, Jones RG. Fueling immunity: insights into metabolism and lymphocyte function. Science. 2013;342:1242454. doi: 10.1126/science.1242454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.MacIver NJ, Jacobs SR, Wieman HL, Wofford JA, Coloff JL, Rathmell JC. Glucose metabolism in lymphocytes is a regulated process with significant effects on immune cell function and survival. Journal of Leukocyte Biology. 2008;84:949–957. doi: 10.1189/jlb.0108024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jacobs SR, Herman CE, MacIver NJ, Wofford JA, Wieman HL, Hammen JJ, Rathmell JC. Glucose Uptake Is Limiting in T Cell Activation and Requires CD28-Mediated Akt-Dependent and Independent Pathways. The Journal of Immunology. 2008;180:4476–4486. doi: 10.4049/jimmunol.180.7.4476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pearce EL, Pearce EJ. Metabolic pathways in immune cell activation and quiescence. Immunity. 2013;38:633–643. doi: 10.1016/j.immuni.2013.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lochner M, Berod L, Sparwasser T. Fatty acid metabolism in the regulation of T cell function. Trends Immunol. 2015;36:81–91. doi: 10.1016/j.it.2014.12.005. [DOI] [PubMed] [Google Scholar]
- 11.Macintyre AN, Gerriets VA, Nichols AG, Michalek RD, Rudolph MC, Deoliveira D, Anderson SM, Abel ED, Chen BJ, Hale LP, et al. The glucose transporter Glut1 is selectively essential for CD4 T cell activation and effector function. Cell Metab. 2014;20:61–72. doi: 10.1016/j.cmet.2014.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gregor MF, Hotamisligil GS. Inflammatory mechanisms in obesity. Annu Rev Immunol. 2011;29:415–445. doi: 10.1146/annurev-immunol-031210-101322. [DOI] [PubMed] [Google Scholar]
- 13.Huttunen R, Syrjanen J. Obesity and the risk and outcome of infection. Int J Obes (Lond) 2013;37:333–340. doi: 10.1038/ijo.2012.62. [DOI] [PubMed] [Google Scholar]
- 14.Milner JJ, Beck MA. The impact of obesity on the immune response to infection. Proc Nutr Soc. 2012;71:298–306. doi: 10.1017/S0029665112000158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Park BV, Pan F. Metabolic regulation of T cell differentiation and function. Mol Immunol. 2015;68:497–506. doi: 10.1016/j.molimm.2015.07.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jacobs SR, Michalek RD, Rathmell JC. IL-7 is essential for homeostatic control of T cell metabolism in vivo. J Immunol. 2010;184:3461–3469. doi: 10.4049/jimmunol.0902593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Weinberg SE, Chandel NS. Futility sustains memory T cells. Immunity. 2014;41:1–3. doi: 10.1016/j.immuni.2014.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.O’Sullivan D, van der Windt GJ, Huang SC, Curtis JD, Chang CH, Buck MD, Qiu J, Smith AM, Lam WY, DiPlato LM, et al. Memory CD8(+) T cells use cell-intrinsic lipolysis to support the metabolic programming necessary for development. Immunity. 2014;41:75–88. doi: 10.1016/j.immuni.2014.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.van der Windt GJ, Everts B, Chang CH, Curtis JD, Freitas TC, Amiel E, Pearce EJ, Pearce EL. Mitochondrial respiratory capacity is a critical regulator of CD8+ T cell memory development. Immunity. 2012;36:68–78. doi: 10.1016/j.immuni.2011.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.van der Windt GJ, O’Sullivan D, Everts B, Ching-Cheng Huang S, Buck MD, Curtis JD, Chang C, Smith AM, Ai T, Faubert B, Jones RG, Pearce EJ, Pearce EL. CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. PNAS. 2013;110:14336–14341. doi: 10.1073/pnas.1221740110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cui G, Staron MM, Gray SM, Ho PC, Amezquita RA, Wu J, Kaech SM. IL-7-Induced Glycerol Transport and TAG Synthesis Promotes Memory CD8+ T Cell Longevity. Cell. 2015;161:750–761. doi: 10.1016/j.cell.2015.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dong W, Ding T, Wu L, Ren X, Epling-Burnette PK, Yang L. Effect of IL-7 and IL-15 on T cell phenotype in myelodysplastic syndromes. Oncotarget. 2016;7:27479–27488. doi: 10.18632/oncotarget.8459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23•.Renner K, Geiselhoringer AL, Fante M, Bruss C, Farber S, Schonhammer G, Peter K, Singer K, Andreesen R, Hoffmann P, et al. Metabolic plasticity of human T cells: Preserved cytokine production under glucose deprivation or mitochondrial restriction, but 2-deoxy-glucose affects effector functions. Eur J Immunol. 2015;45:2504–2516. doi: 10.1002/eji.201545473. The authors demonstrate alterations in human CD4 and CD8 T cell proliferative and fucntional responses when challeneged with glycolytic and respiratory restriction. This investigation provides evidence linking metabolic flexibility of human T cells to effector functions, like cytokine production, and highlights translational insights from murine models. [DOI] [PubMed] [Google Scholar]
- 24.Battaglia A, Buzzonetti A, Baranello C, Fanelli M, Fossati M, Catzola V, Scambia G, Fattorossi A. Interleukin-21 (IL-21) synergizes with IL-2 to enhance T-cell receptor-induced human T-cell proliferation and counteracts IL-2/transforming growth factor-beta-induced regulatory T-cell development. Immunology. 2013;139:109–120. doi: 10.1111/imm.12061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sheng J, Chen W, Zhu HJ. The immune suppressive function of transforming growth factor-beta (TGF-beta) in human diseases. Growth Factors. 2015;33:92–101. doi: 10.3109/08977194.2015.1010645. [DOI] [PubMed] [Google Scholar]
- 26••.Saucillo DC, Gerriets VA, Sheng J, Rathmell JC, Maciver NJ. Leptin metabolically licenses T cells for activation to link nutrition and immunity. J Immunol. 2014;192:136–144. doi: 10.4049/jimmunol.1301158. Here, the authors provide a novel link between lymphocyte activation and nutrition by illucidating leptin’s impact on effector T cell metabolism. This finding provides an essential role for leptin in T cell mediated immunity. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pollizzi KN, Powell JD. Integrating canonical and metabolic signalling programmes in the regulation of T cell responses. Nat Rev Immunol. 2014;14:435–446. doi: 10.1038/nri3701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Blagih J, Coulombe F, Vincent EE, Dupuy F, Galicia-Vazquez G, Yurchenko E, Raissi TC, van der Windt GJ, Viollet B, Pearce EL, et al. The energy sensor AMPK regulates T cell metabolic adaptation and effector responses in vivo. Immunity. 2015;42:41–54. doi: 10.1016/j.immuni.2014.12.030. [DOI] [PubMed] [Google Scholar]
- 29.Collaboration NRF. Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19.2 million participants. The Lancet. 2016;387:1377–1396. doi: 10.1016/S0140-6736(16)30054-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ogden CL, Carroll MD, Fryar CD, Flegal KM. Prevalence of Obesity Among Adults and Youth-United States, 2011–2014. NCHS data brief. 2015;(219) [PubMed] [Google Scholar]
- 31.Louie JK, Acosta M, Samuel MC, Schechter R, Vugia DJ, Harriman K, Matyas BT, California Pandemic Working G A novel risk factor for a novel virus: obesity and 2009 pandemic influenza A (H1N1) Clin Infect Dis. 2011;52:301–312. doi: 10.1093/cid/ciq152. [DOI] [PubMed] [Google Scholar]
- 32.Park HL, Shim SH, Lee EY, Cho W, Park S, Jeon HJ, Ahn SY, Kim H, Nam JH. Obesity-induced chronic inflammation is associated with the reduced efficacy of influenza vaccine. Hum Vaccin Immunother. 2014;10:1181–1186. doi: 10.4161/hv.28332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sheridan PA, Paich HA, Handy J, Karlsson EA, Hudgens MG, Sammon AB, Holland LA, Weir S, Noah TL, Beck MA. Obesity is associated with impaired immune response to influenza vaccination in humans. Int J Obes (Lond) 2012;36:1072–1077. doi: 10.1038/ijo.2011.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Carolan E, Hogan AE, Corrigan M, Gaotswe G, O’Connell J, Foley N, O’Neill LA, Cody D, O’Shea D. The impact of childhood obesity on inflammation, innate immune cell frequency, and metabolic microRNA expression. J Clin Endocrinol Metab. 2014;99:E474–478. doi: 10.1210/jc.2013-3529. [DOI] [PubMed] [Google Scholar]
- 35.Paich HA, Sheridan PA, Handy J, Karlsson EA, Schultz-Cherry S, Hudgens MG, Noah TL, Weir SS, Beck MA. Overweight and obese adult humans have a defective cellular immune response to pandemic H1N1 influenza A virus. Obesity (Silver Spring) 2013;21:2377–2386. doi: 10.1002/oby.20383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Costanzo AE, Taylor KR, Dutt S, Han PP, Fujioka K, Jameson JM. Obesity impairs gammadelta T cell homeostasis and antiviral function in humans. PLoS One. 2015;10:e0120918. doi: 10.1371/journal.pone.0120918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mascitelli L, Pezzetta F, Goldstein MR. Leptin and regulatory T cells in obese patients with asthma. Thorax. 2008;63:659. [PubMed] [Google Scholar]
- 38.Tagliabue C, Principi N, Giavoli C, Esposito S. Obesity: impact of infections and response to vaccines. Eur J Clin Microbiol Infect Dis. 2016;35:325–331. doi: 10.1007/s10096-015-2558-8. [DOI] [PubMed] [Google Scholar]
- 39.Roth J, Sahota N, Patel P, Mehdi SF, Wiese MM, Mahboob HB, Bravo M, Eden DJ, Bashir MA, Kumar A, et al. Obesity paradox, obesity orthodox, and the metabolic syndrome: An approach to unity. Mol Med. 2016;22 doi: 10.2119/molmed.2016.00211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nilsson C, Raun K, Yan FF, Larsen MO, Tang-Christensen M. Laboratory animals as surrogate models of human obesity. Acta Pharmacol Sin. 2012;33:173–181. doi: 10.1038/aps.2011.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Milner JJ, Wang J, Sheridan PA, Ebbels T, Beck MA, Saric J. 1H NMR-based profiling reveals differential immune-metabolic networks during influenza virus infection in obese mice. PLoS One. 2014;9:e97238. doi: 10.1371/journal.pone.0097238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42••.Milner JJ, Rebeles J, Dhungana S, Stewart DA, Sumner SC, Meyers MH, Mancuso P, Beck MA. Obesity Increases Mortality and Modulates the Lung Metabolome during Pandemic H1N1 Influenza Virus Infection in Mice. J Immunol. 2015;194:4846–4859. doi: 10.4049/jimmunol.1402295. The authors demonstrate that obesity increases mortaltiy and lung inflammation in obese mice during Pandemic H1N1 Influenza Virus infection. Furthermore, they provide a link between alterated metabolic profiles in obese mice and impaired cellular immune response. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Reis BS, Lee K, Fanok MH, Mascaraque C, Amoury M, Cohn LB, Rogoz A, Dallner OS, Moraes-Vieira PM, Domingos AI, et al. Leptin receptor signaling in T cells is required for Th17 differentiation. J Immunol. 2015;194:5253–5260. doi: 10.4049/jimmunol.1402996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Deng T, Lyon CJ, Minze LJ, Lin J, Zou J, Liu JZ, Ren Y, Yin Z, Hamilton DJ, Reardon PR, et al. Class II major histocompatibility complex plays an essential role in obesity-induced adipose inflammation. Cell Metab. 2013;17:411–422. doi: 10.1016/j.cmet.2013.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nishimura S, Manabe I, Nagasaki M, Eto K, Yamashita H, Ohsugi M, Otsu M, Hara K, Ueki K, Sugiura S, et al. CD8+ effector T cells contribute to macrophage recruitment and adipose tissue inflammation in obesity. Nat Med. 2009;15:914–920. doi: 10.1038/nm.1964. [DOI] [PubMed] [Google Scholar]
- 46.McLaughlin T, Liu LF, Lamendola C, Shen L, Morton J, Rivas H, Winer D, Tolentino L, Choi O, Zhang H, et al. T-cell profile in adipose tissue is associated with insulin resistance and systemic inflammation in humans. Arterioscler Thromb Vasc Biol. 2014;34:2637–2643. doi: 10.1161/ATVBAHA.114.304636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Morris DL, Cho KW, DelProposto JL, Oatmen KE, Geletka LM, Martinez-Santibanez G, Singer K, Lumeng CN. Adipose Tissue Macrophages Function As Antigen-Presenting Cells and Regulate Adipose Tissue CD4+ T Cells in Mice. Diabetes. 2013;62:2762–2772. doi: 10.2337/db12-1404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cho KW, Morris DL, DelProposto JL, Geletka L, Zamarron B, Martinez-Santibanez G, Meyer KA, Singer K, O’Rourke RW, Lumeng CN. An MHC II-dependent activation loop between adipose tissue macrophages and CD4+ T cells controls obesity-induced inflammation. Cell Rep. 2014;9:605–617. doi: 10.1016/j.celrep.2014.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49•.Xiao L, Yang X, Lin Y, Li S, Jiang J, Qian S, Tang Q, He R, Li X. Large adipocytes function as antigen-presenting cells to activate CD4(+) T cells via upregulating MHCII in obesity. Int J Obes (Lond) 2016;40:112–120. doi: 10.1038/ijo.2015.145. Here, the authors show that large adipocytes from obese humans highly express MHCII, causing the activation of IFNγ producing CD4+ T cells. This finding links obesity to adipose tissue inflammation and changes in T cell activation and populations. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang CY, Kim HH, Hiroi Y, Sawada N, Salomone S, Benjamin LE, Moskowitz MA, Liao JK. Obesity Increases Vascular Senescence and Susceptibility to Ischemic Injury Through Chronic Activation of Akt and mTOR. Science Signaling. 2009;2:1–11. doi: 10.1126/scisignal.2000143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Palmer AK, Tchkonia T, LeBrasseur NK, Chini EN, Xu M, Kirkland JL. Cellular Senescence in Type 2 Diabetes: A Therapeutic Opportunity. Diabetes. 2015;64:2289–2298. doi: 10.2337/db14-1820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52•.Shirakawa K, Yan X, Shinmura K, Endo J, Kataoka M, Katsumata Y, Yamamoto T, Anzai A, Isobe S, Yoshida N, et al. Obesity accelerates T cell senescence in murine visceral adipose tissue. J Clin Invest. 2016;126:4626–4639. doi: 10.1172/JCI88606. The authors describe a senescent T cell phenotype in murine visceral adipose tissue (VAT) found in obese mice but not lean controls. This novel population of cells was shown to drive VAT inflammation through osteopontin production, connecting adipose tissue with immune aging. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lund SA, Giachelli CM, Scatena M. The role of osteopontin in inflammatory processes. J Cell Commun Signal. 2009;3:311–322. doi: 10.1007/s12079-009-0068-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jin HT, Ahmed R, Okazaki T. Role of PD-1 in regulating T-cell immunity. Curr Top Microbiol Immunol. 2011;350:17–37. doi: 10.1007/82_2010_116. [DOI] [PubMed] [Google Scholar]
- 55.Charlton JJ, Chatzidakis I, Tsoukatou D, Boumpas DT, Garinis GA, Mamalaki C. Programmed death-1 shapes memory phenotype CD8 T cell subsets in a cell-intrinsic manner. J Immunol. 2013;190:6104–6114. doi: 10.4049/jimmunol.1201617. [DOI] [PubMed] [Google Scholar]
- 56.Wei F, Zhong S, Ma Z, Kong H, Medvec A, Ahmed R, Freeman GJ, Krogsgaard M, Riley JL. Strength of PD-1 signaling differentially affects T-cell effector functions. PNAS. 2013:E2480–E2489. doi: 10.1073/pnas.1305394110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chang CH, Curtis JD, Maggi LB, Jr, Faubert B, Villarino AV, O’Sullivan D, Huang SC, van der Windt GJ, Blagih J, Qiu J, et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell. 2013;153:1239–1251. doi: 10.1016/j.cell.2013.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Simar D, Versteyhe S, Donkin I, Liu J, Hesson L, Nylander V, Fossum A, Barres R. DNA methylation is altered in B and NK lymphocytes in obese and type 2 diabetic human. Metabolism. 2014;63:1188–1197. doi: 10.1016/j.metabol.2014.05.014. [DOI] [PubMed] [Google Scholar]
- 59.Jacobsen MJ, Mentzel CM, Olesen AS, Huby T, Jorgensen CB, Barres R, Fredholm M, Simar D. Altered Methylation Profile of Lymphocytes Is Concordant with Perturbation of Lipids Metabolism and Inflammatory Response in Obesity. J Diabetes Res. 2016;2016:8539057. doi: 10.1155/2016/8539057. [DOI] [PMC free article] [PubMed] [Google Scholar]


