Abstract
Visual processing starts in the retina. Within only two synaptic layers, a large number of parallel information channels emerge, each encoding a highly processed feature like edges or the direction of motion. Much of this functional diversity arises in the inner plexiform layer, where inhibitory amacrine cells modulate the excitatory signal of bipolar and ganglion cells. Studies investigating individual amacrine cell circuits like the starburst or A17 circuit have demonstrated that single types can possess specific morphological and functional adaptations to convey a particular function in one or a small number of inner retinal circuits. However, the interconnected and often stereotypical network formed by different types of amacrine cells across the inner plexiform layer prompts that they should be also involved in more general computations. In line with this notion, different recent studies systematically analysing inner retinal signalling at a population level provide evidence that general functions of the ensemble of amacrine cells across types are critical for establishing universal principles of retinal computation like parallel processing or motion anticipation. Combining recent advances in the development of indicators for imaging inhibition with large‐scale morphological and genetic classifications will help to further our understanding of how single amacrine cell circuits act together to help decompose the visual scene into parallel information channels. In this review, we aim to summarise the current state‐of‐the‐art in our understanding of how general features of amacrine cell inhibition lead to general features of computation.

Keywords: amacrine cell, computation, GABA, glycine, inhibition, neuronal network, retina, vision
Abbreviations
- AC
amacrine cell
- BC
bipolar cell
- IPL
inner plexiform layer
- PR
photoreceptor
- RGC
retinal ganglion cell
Inhibitory amacrine cells shape visual information processing in the inner retina
The vertebrate retina is an established model system in neuroscience for sensory information processing. It decomposes the visual input into parallel channels, each selective for a specific feature like motion, contrast or edges (Wässle, 2004; Masland, 2012a). Already at the first synapse, signals from single photoreceptors (PRs) are split into more than a dozen bipolar cell (BC) types (Euler et al. 2014), which relay the information to more than 30 output channels formed by the retinal ganglion cells (RGCs) (Robles et al. 2014; Sanes & Masland, 2015; Baden et al. 2016). Most computations towards retinal feature extraction are implemented in the second synaptic layer – the inner plexiform layer (IPL) (Gollisch & Meister, 2010). Here, at least 30 types of inhibitory amacrine cell (AC) form intricate connections with BCs and RGCs (Vaney, 1990) to shape and gate the excitatory pathways of the inner retina. The modulatory role of ACs is key for generating the functional diversity present in the retinal output (Masland, 2012b). While a handful of individual AC circuits have been studied at great detail, it is not clear how representative their functions are for the great majority of ACs and surprisingly little is known about more general computational principles that ACs as a whole impart on the retinal network. Here, we summarise current knowledge about how ACs contribute to information processing in the retina and speculate about more universal roles of AC function.
Alongside RGCs, ACs are the most diverse class of retinal neurons and they differ even more greatly in size and morphology than RGCs (Vaney, 1990; MacNeil & Masland, 1998; Masland, 2012b). Two major groups can be distinguished: ‘small‐field’ types mainly signal vertically across synaptic layers of the IPL and are classically thought to modulate visual signalling locally over a small region of the visual field. In contrast, ‘wide‐field’ ACs predominantly transfer information laterally within single IPL strata and can span up to several millimetres of the retinal surface (Fig. 1 A), thereby shaping visual information processing at a larger spatial scale. Although some wide‐field ACs possess axons for fast signal transmission across large distances, most types use their dendrites as both input and output structures (Euler & Denk, 2001). In mammals, small‐field ACs mainly release glycine as neurotransmitter (Menger et al. 1998), whereas wide‐field types release GABA (Fig. 1 B; Pourcho & Goebel, 1983). Additionally, many ACs co‐release a second neurotransmitter (e.g. glutamate; Lee et al. 2014) or neuromodulator (e.g. nitric oxide; Vielma et al. 2011). ACs provide type‐specific inhibitory inputs to BC axon terminals (‘presynaptic inhibitory inputs’) and RGC dendrites (‘postsynaptic inhibitory inputs’) as well as to other ACs (‘serial inhibitory inputs’) (Fig. 1 C; Eggers & Lukasiewicz, 2011; Zhang & McCall, 2012).
Figure 1. Organisation of the amacrine cell network in the inner retina.
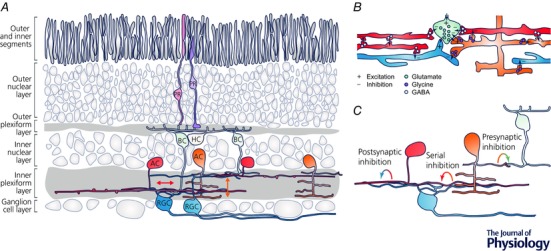
A, in the retina, photoreceptors (PRs, purple) transduce the visual input into an electrical signal and feed into bipolar cells (BCs, green) that provide input to the retina's output neurons, the retinal ganglion cells (RGCs, blue). This vertical excitatory pathway is extensively modulated by inhibitory amacrine cells (ACs) in the inner retina. Here, wide‐field ACs (red) mainly transfer information laterally within individual synaptic layers, while small‐field ACs (orange) predominantly mediate vertical signalling across synaptic layers. Arrows indicate main signal flow. HC, horizontal cells (yellow). B, ACs form a complex and dense synaptic network in the inner plexiform layer. In mammals, wide‐field ACs use GABA as neurotransmitter (light blue vesicles), while small‐field ACs use glycine (purple vesicles). Bipolar cells use glutamate (green vesicles). C, ACs provide inhibitory inputs to BC axon terminals (presynaptic inhibitory inputs), RGC dendrites (postsynaptic inhibitory inputs) and other ACs (serial inhibitory inputs).
Individual amacrine cell microcircuits
A small number of specific AC circuits have been studied at great detail. One prominent example is the starburst AC, which provides the critical inhibition underlying direction‐selective responses of rodent and lagomorph RGCs (Vaney et al. 2012). Several mechanisms have been suggested to contribute to the computation of direction selectivity in starburst ACs, including different intrinsic mechanisms, such as active membrane conductances (Hausselt et al. 2007), and network interactions like reciprocal inhibition between neighbouring starburst ACs (Lee & Zhou, 2006; Münch & Werblin, 2006; Enciso et al. 2010; Ding et al. 2016). Probably by combining these mechanisms, each main dendritic branch of a single starburst AC is differentially tuned to stimuli moving centrifugally from the soma (Euler et al. 2002). As such, individual starburst AC dendrites are central computational units within inner retinal circuits that extract the direction of object motion. Similarly, the A17 AC joins a morphological ‘set‐up’ (the regular ∼20 μm spacing of varicosities; Grimes et al. 2010) with biophysical features, like the complement of expressed receptor types (Chávez et al. 2006; Grimes et al. 2009, 2015), such that each individual varicosity contains an independent ‘microcircuit’ that provides highly local feedback. Thereby the cell provides synapse‐specific gain control to individual rod BC axon terminals to modulate the sensitivity of the rod pathway. Further examples of well‐studied AC types and circuits include the AII (Demb & Singer, 2012), the A8 (Kolb & Nelson, 1981; Lee et al. 2015) and the glutamatergic AC (Lee et al. 2014; Tien et al. 2016) (for discussion of the role of glutamatergic outputs of ACs see Baden & Euler, 2016) as well as types of polyaxonal ACs (Baccus et al. 2008; Greschner et al. 2014; Murphy‐Baum & Taylor, 2015). One key feature that all of these AC types have in common is that they use highly specific morphological and functional adaptations to carry out specific computations in a single or at most a small number of inner retinal circuits.
General amacrine cell functions
The population of ACs across types forms a dense network connecting all strata of the IPL, suggesting that the activity in any one cell – at least in principle – might affect the activity in any other cell of the network. One striking example in support of this notion is that altering the activity in a single salamander BC in the absence of visual stimulation changes firing rates in RGCs of different types and across large distances via polysynaptic pathways involving ACs (Asari & Meister, 2014). Because of this across‐type interconnectivity, ACs might interact beyond their individual microcircuits, possibly contributing in addition to general features of inner retinal computation. Indeed, the complex synaptic arrangements the population of ACs forms in the IPL include stereotypic and repeating connectivity motifs. For example, probably all BCs receive independent reciprocal and lateral inhibition from different AC types at the same specialised output structure (Vigh et al. 2011; Grimes, 2012; Tanaka & Tachibana, 2013), the dyad synapse (Dowling & Boycott, 1966; Raviola & Dacheux, 1987). Similarly, RGCs consistently show an excess of inhibitory inputs compared to excitatory inputs across types and species and these inputs appear to be randomly distributed across the dendritic tree of individual RGCs (Freed & Sterling, 1988; Hitchcock, 1989; Kolb & Nelson, 1993). In addition, ACs form repeating and spatially extensive circuits via serial synaptic connections (Dowling & Boycott, 1966; Zhang et al. 2004) that shape the magnitude and timing of inhibition in the inner retina (Zhang et al. 1997; Roska et al. 1998; Eggers et al. 2007). One connectivity motif that is shared across all classes of inner retinal neurons – BCs (Molnar & Werblin, 2007; Rosa et al. 2016), ACs (Hsueh et al. 2008) and RGCs (Manookin et al. 2008; Cafaro & Rieke, 2013) – is crossover inhibition that connects the On and Off pathway via small‐field ACs. Therefore, while type‐specific morphological adaptations give rise to highly specific functions (see above), it seems reasonable to suggest that general characteristics of inner retinal connectivity shared across the ensemble of AC types will lead to general features of computation.
One ‘classical’ example of such a general computation is the establishment of the antagonistic centre–surround organisation of BC and RGC receptive fields (e.g. Kuffler, 1953). The inhibitory surround of individual cells is largely generated in the inner retina by lateral inhibition provided by wide‐field ACs (Flores‐Herr et al. 2001; Ichinose & Lukasiewicz, 2005; Buldyrev & Taylor, 2013; Protti et al. 2014; but see e.g. Naka & Nye, 1971; Marchiafava, 1978). However, numbers alone dictate that the surround of each of the ∼14 BC and > 30 RGC types cannot be created by an exclusive ‘partner AC’ – there simply are not enough AC types in the retina. Instead, the inhibitory surround is likely to be at least partially independent from each cell's individual microcircuitry and instead emerges from general principles of inner retinal organisation.
Investigating general functional features of amacrine cells
Understanding how these general AC functions arise within the retinal network requires the systematic recording of light‐evoked activity in many different retinal neurons under the same experimental condition (e.g. adaptational state, visual stimulation protocol) in the intact tissue preparation (i.e. the whole‐mounted retina or, if possible, in vivo), where long‐range inhibitory connections are preserved. For example, Johnston & Lagnado (2015) performed electrical single cell recordings of different RGC types in the whole‐mounted goldfish retina to study the underlying mechanism of motion anticipation. Here, the visual system compensates for the temporal delay in phototransduction cascade and downstream signal transmission to accurately estimate the position of a moving object. This fundamental property of the retinal output occurs in the majority of RGC types and across species (Berry et al. 1999; Schwartz et al. 2007). In this study, they found that feedforward inhibition from ACs is critical for anticipating moving stimuli and locate this computation to the dendritic tree of individual RGCs. Since this mechanism applies to different RGC types that receive inhibitory inputs from different AC types, this study reveals a general function of the population of ACs. Similarly, Asari & Meister (2012) investigated global principles of signal transmission in the inner retina by intracellularly manipulating single BCs in the whole‐mounted salamander retina and simultaneously recording the spiking activity of many surrounding RGCs. They showed that AC function is critical for diversifying the signal properties (e.g. kinetic, adaptation) of different types of BC and RGC – another universal role of ACs.
To further investigate how inhibition from ACs shapes the visual signal in the inner retina in a general manner, one would ideally get closer to the critical site of AC interactions and monitor the activity in the cellular compartments that directly receive the inhibitory inputs. These include the axon terminals of BCs as well as dendritic processes of both ACs and RGCs. Here, optical population imaging of neuronal activity comes to shine. In contrast to electrical somatic recordings, which typically give only a limited representation of what happens in individual neuronal compartments distal to the recording site such as axon terminals (Oltedal et al. 2006) or dendrites (Poleg‐Polsky & Diamond, 2011), an optical imaging approach permits recording of the activity of many subcellular structures in parallel at micrometre resolution. Moreover, genetic targeting in combination with the spatial resolution provided by the imaging system allows unambiguous identification of the origin of the recorded signals (e.g. Duebel et al. 2006; Odermatt et al. 2012; Yonehara et al. 2013). Several recent studies drew on the opportunities provided by an optical approach to address general features of inner retinal function (e.g. characterisation of BC and RGC function in mouse and zebrafish; Dreosti et al. 2009; Odermatt et al. 2012; Baden et al. 2013, 2016; Borghuis et al. 2013a; Nikolaev et al. 2013; Rosa et al. 2016). Importantly, the diversity of available indicators (Lin and Schnitzer, 2016) selective for different neuronal events allows recording not only of neuronal activity per se but of a specific biological process. For example, since the development of the glutamate indicator iGluSnFR (Marvin et al. 2013), it is now possible to record light‐evoked glutamate release from BC axon terminals throughout the IPL (Borghuis et al. 2013b, Franke et al. 2017). Unlike presynaptic calcium, glutamate release directly corresponds to the output of BCs, accounting not only for presynaptic inhibition (e.g. Borghuis et al. 2014) but also for any release dynamics of BC ribbon synapses (Burrone & Lagnado, 2000; Cho and von Gersdorff, 2012; Nikolaev et al. 2013). Another example relevant for studying ACs is chloride indicators. They allow monitoring changes in the intracellular chloride concentration (e.g. Duebel et al. 2006) which can be directly linked to inhibitory inputs and therefore represent a promising tool for unravelling AC function in BC axon terminals and AC and RGC dendrites.
Evidence for a universal role of amacrine cells
A recent study (Rosa et al. 2016) used in vivo population imaging of calcium signals in zebrafish BC axon terminals to investigate the role of crossover inhibition on temporal signalling in the inner retina. Electrophysiological studies had shown before that crossover inhibition is involved in a variety of functions (Werblin, 2010), such as the compensation of synaptic rectification (Molnar et al. 2009). The large dataset of calcium responses across different strata of the IPL in combination with pharmacological manipulations allowed the authors to identify a novel, systematic effect of crossover inhibition on frequency tuning of BCs: while crossover inhibition shifts band‐pass synapses into low‐pass and therefore generates the sustained Off channel in the inner retina, it suppresses contrast responses in On BC terminals.
Recently, Franke et al. (2017) systematically recorded glutamate release of > 13,000 BC axon terminals across the whole IPL to investigate how AC circuits help to decompose the visual scene into the parallel channels carried by the BCs in mouse. By applying light stimuli of different spatial scales, they compared centre (‘local’) responses – dominated by the excitatory input from PRs – with centre–surround (‘full‐field’) responses – additionally including the inhibitory inputs from ACs. The authors found that the functional diversity among BCs critically relies on inhibitory inputs from ACs: while local responses are highly similar across BC types of the same response polarity (Fig. 2 A), additional surround stimulation significantly increases the functional differences (decorrelates) between BCs (Fig. 2 B). Previously, several studies demonstrated the effect of axonal inhibition provided by ACs for modulating temporal and spatial properties of BCs (Eggers et al. 2007; Purgert & Lukasiewicz, 2015). However, so far the importance of these axonal inputs for diversifying BC responses has been underestimated compared to dendritic mechanisms like the expression of specific glutamate receptors (DeVries et al. 2006; Lindstrom et al. 2014). By using pharmacological manipulations they showed that GABAergic wide‐field ACs provide the decorrelating inhibition to BC axon terminals. In contrast, glycinergic small‐field ACs mainly shape BC function indirectly by gating the spatially extensive GABAergic network, challenging the classical view that small‐field ACs are predominantly involved in local signal processing. Together these findings suggest that the two major groups of AC of the mouse retina act together to set the ratio of excitation and inhibition. This cooperative action increases functional diversity in the inner retina. The study also demonstrates a general and AC group‐specific effect of GABAergic and glycinergic ACs across different BC types and IPL strata. Since ACs also provide inhibition to RGCs, there might be an additional step of signal decorrelation at the level of RGC dendrites (see Asari & Meister, 2012).
Figure 2. Example of a general function of amacrine cells.
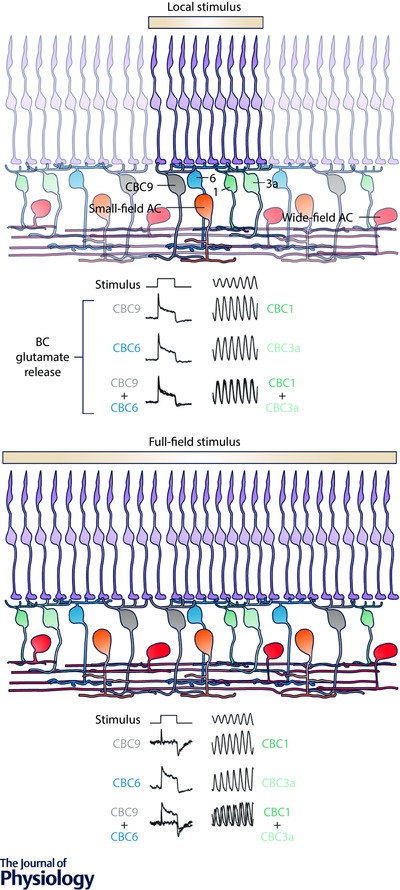
A, a local stimulus (100 μm diameter, yellow) mainly activates retinal pathways directly beneath the stimulus including PRs and BCs as well as small‐field ACs. The glutamatergic output of individual BCs in response to such a local light step (left) and contrast flicker (right), is nearly independent of the specific BC type recorded from (of the same polarity) – here exemplarily shown for two On (CBC9 and CBC6) and Off (CBC1 and CBC3a) BC types (Franke et al. 2017). The last row of traces represents an overlay of the previous two rows. Colour intensity used to indicate expected stimulus‐driven activity level of individual neurons. B, full‐field stimulation (600 μm diameter) additionally recruits wide‐field ACs that provide inhibitory GABAergic inputs to BC axon terminals and AC dendrites. This lateral inhibition decorrelates the BC responses, thereby increasing the functional diversity across BC types (Franke et al. 2017). Because the response diversification is observed for all BC types, which receive inputs from different AC types, this effect illustrates an example of a general role of ACs in inner retinal signalling.
Amacrine cell function – specific or general?
What is the functional significance of the diverse population of ACs? Does every AC type have an isolated and specific function in one distinct circuit? Conversely, does every AC circuit contribute to more general effects of inner retinal computation? The truth probably lies somewhere in the middle. Clearly, at least some AC types have developed highly specific morphological and functional adaptations to perform particular computations (e.g. starburst or A17 AC, see above). Despite these selective adaptations, the functions of individual AC types may still combine to serve a common goal. There is a rapidly growing body of literature (e.g. Asari & Meister, 2012; Johnston & Lagnado, 2015; Franke et al. 2017; Rosa et al. 2016) arguing that general functions of ACs across types are critical for establishing universal principles of retinal signalling like parallel processing or motion anticipation.
Future directions
To get a better understanding of how individual and general AC functions are organised will require a comprehensive characterisation of morphological and functional aspects of ACs, both at high resolution and at the population level. In particular, for verifying the concept of a general mode of action of ACs, it will be important to explore to what extent general computations performed by the population of ACs are independent of individual AC types. This might for example be achieved by deactivating single genetically defined AC populations. Here, transcriptional analysis (Macosko et al. 2015) in combination with large‐scale electron microscopy (EM) datasets (e.g. Helmstaedter et al. 2013) will serve to identify the number of AC types and make them more accessible for targeted genetic manipulations. In parallel, advances in the development of sensors for imaging inhibition (e.g. Paredes et al. 2016) promise to provide a more direct approach for studying the role and integration of inhibitory inputs in inner retinal circuits at subcellular resolution in the near future. However, doing these types of experiments in a single species (like the mouse) may prove to be insufficient. After all, the degree of generality of AC function might vary for different species. To therefore truly probe to what extent general AC actions are a universal feature of inner retinal signalling it will be critical to compare results across species. Taken together, combining these different approaches will provide new insights into how the single AC circuits act together to shape information processing in the early visual system in a general manner.
Additional information
Competing interests
None declared.
Author contributions
K.F.: conception and design; administrative support; provision of study materials or patients; collection and assembly of data; data analysis and interpretation; manuscript writing. T.B.: conception and design; financial support; administrative support; provision of study materials or patients; collection and assembly of data; data analysis and interpretation; manuscript writing. Both authors have approved the final version of the manuscript and agree to be accountable for all aspects of the work. All persons designated as authors qualify for authorship, and all those who qualify for authorship are listed.
Funding
This work was supported by the European Commission H2020 (ERC StG 677687 “NeuroVisEco” to T.B.) and funding to Thomas Euler (who is not an author) from the National Institutes of Neurological Disorders and Stroke as well as the National Eye Institute (U01NS090562 and 1R01EY023766) of the National Institutes of Health was used to support K.F. This work is part of the research program of the Bernstein Center for Computational Neuroscience, Tübingen, funded by the German Federal Ministry of Education and Research and the Max Planck Society (BMBF, FKZ: 01GQ1002; MPG M.FE.AKYBE0004).
Acknowledgements
We thank Thomas Euler for critical feedback on the manuscript.
Biographies
Katrin Franke did her PhD with Thomas Euler and Tom Baden at the Centre for Integrative Neuroscience at the University of Tübingen. Since January 2017 she has been a Junior Research Group Leader at the Bernstein Centre for Computational Neuroscience at the University of Tübingen.

Tom Baden did his PhD with Berthold Hedwig (Zoology, Cambridge, UK) and his postdocs with Leon Lagnado (LMB‐MRC, Cambridge, UK) and Thomas Euler (CIN, Tübingen, Germany) before returning to the UK to take up his current group leader position at the University of Sussex. They share a common interest in understanding circuit computations in neuronal networks with particular focus on the early visual system of vertebrates.
This review was presented at the symposium “Phototransduction and synaptic transmission” which took place at the Phototransduction UK workshop, Sheffield, 31 August – 2 September 2016.
References
- Asari H & Meister M (2012). Divergence of visual channels in the inner retina. Nat Neurosci 15, 1581–1589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asari H & Meister M (2014). The projective field of retinal bipolar cells and its modulation by visual context. Neuron 81, 641–652. [DOI] [PubMed] [Google Scholar]
- Baccus SA, Olveczky BP, Manu M & Meister M (2008). A retinal circuit that computes object motion. J Neurosci 28, 6807–6817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baden T, Berens P, Bethge M & Euler T (2013). Spikes in mammalian bipolar cells support temporal layering of the inner retina. Curr Biol 23, 48–52. [DOI] [PubMed] [Google Scholar]
- Baden T, Berens P, Franke K, Román Rosón M, Bethge M & Euler T (2016). The functional diversity of retinal ganglion cells in the mouse. Nature 529, 345–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baden T & Euler T (2016). Dispatches retinal physiology: non‐bipolar‐cell excitatory drive in the inner retina. Curr Biol 26, 706–708. [DOI] [PubMed] [Google Scholar]
- Berry MJ, Brivanlou IH, Jordan TA & Meister M (1999). Anticipation of moving stimuli by the retina. Nature 398, 334–338. [DOI] [PubMed] [Google Scholar]
- Borghuis BG, Looger LL, Tomita S & Demb JB (2014). Kainate receptors mediate signaling in both transient and sustained off bipolar cell pathways in mouse retina. J Neurosci 34, 6128–6139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borghuis BG, Marvin JS, Looger LL & Demb JB (2013a). Two‐photon imaging of nonlinear glutamate release dynamics at bipolar cell synapses in the mouse retina. J Neurosci 33, 10972–10985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borghuis BG, Marvin JS, Looger LL & Demb JB (2013b). Two‐photon imaging of nonlinear glutamate release dynamics at bipolar cell synapses in the mouse retina. J Neurosci 33, 10972–10985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buldyrev I & Taylor WR (2013). Inhibitory mechanisms that generate centre and surround properties in ON and OFF brisk‐sustained ganglion cells in the rabbit retina. J Physiol 591, 303–325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burrone J & Lagnado L (2000). Synaptic depression and the kinetics of exocytosis in retinal bipolar cells. J Neurosci 20, 568–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cafaro J & Rieke F (2013). Regulation of spatial selectivity by crossover inhibition. J Neurosci 33, 6310–6320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chávez AE, Singer JH & Diamond JS (2006). Fast neurotransmitter release triggered by Ca influx through AMPA‐type glutamate receptors. Nature 443, 705–708. [DOI] [PubMed] [Google Scholar]
- Cho S & von Gersdorff H (2012). Ca2+ influx and neurotransmitter release at ribbon synapses. Cell Calcium 52, 208–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demb JB & Singer JH (2012). Intrinsic properties and functional circuitry of the AII amacrine cell. Vis Neurosci 29, 51–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeVries SH, Li W & Saszik S (2006). Parallel processing in two transmitter microenvironments at the cone photoreceptor synapse. Neuron 50, 735–748. [DOI] [PubMed] [Google Scholar]
- Ding H, Smith RG, Poleg‐Polsky A, Diamond JS & Briggman KL (2016). Species‐specific wiring for direction selectivity in the mammalian retina. Nature 535,105–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dowling JE & Boycott BB (1966). Organization of the primate retina: electron microscopy. Proc R Soc Lond B Biol Sci 166, 80–111. [DOI] [PubMed] [Google Scholar]
- Dreosti E, Odermatt B, Dorostkar MM & Lagnado L (2009). A genetically encoded reporter of synaptic activity in vivo. Nat Methods 6, 883–889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duebel J, Haverkamp S, Schleich W, Feng G, Augustine GJ, Kuner T & Euler T (2006). Two‐photon imaging reveals somatodendritic chloride gradient in retinal on‐type bipolar cells expressing the biosensor clomeleon. Neuron 49, 81–94. [DOI] [PubMed] [Google Scholar]
- Eggers ED & Lukasiewicz PD (2011). Multiple pathways of inhibition shape bipolar cell responses in the retina. Vis Neurosci 28, 95–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eggers ED, Mccall MA & Lukasiewicz PD (2007). Presynaptic inhibition differentially shapes transmission in distinct circuits in the mouse retina. J Physiol 582, 569–582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enciso GA, Rempe M, Dmitriev AV, Gavrikov KE, Terman D & Mangel SC (2010). A model of direction selectivity in the starburst amacrine cell network. J Comput Neurosci 28, 567–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Euler T & Denk W (2001). Dendritic processing. Curr Opin Neurobiol 11, 415–422. [DOI] [PubMed] [Google Scholar]
- Euler T, Detwiler PB & Denk W (2002). Directionally selective calcium signals in dendrites of starburst amacrine cells. Nature 418, 845–852. [DOI] [PubMed] [Google Scholar]
- Euler T, Haverkamp S, Schubert T & Baden T (2014). Retinal bipolar cells: elementary building blocks of vision. Nat Rev Neurosci 15, 507–519. [DOI] [PubMed] [Google Scholar]
- Flores‐Herr N, Protti DA & Wässle H (2001). Synaptic currents generating the inhibitory surround of ganglion cells in the mammalian retina. J Neurosci 21, 4852–4863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franke K, Berens P, Schubert T, Bethge M, Euler T & Baden T (2017). Interplay of excitation and inhibition decorrelates visual feature representation in the mammalian inner retina. Biorxiv, doi:10.1101/040642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freed MA & Sterling P (1988). The ON‐alpha ganglion cell of the cat retina and its presynaptic cell types. J Neurosci 8, 2303–2320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gollisch T & Meister M (2010). Eye smarter than scientists believed: neural computations in circuits of the retina. Neuron 65, 150–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greschner M, Field GD, Li PH, Schiff ML, Gauthier JL, Ahn D, Sher A, Litke AM & Chichilnisky EJ (2014). A polyaxonal amacrine cell population in the primate retina. J Neurosci 34, 3597–3606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimes WN (2012). Amacrine cell‐mediated input to bipolar cells: variations on a common mechanistic theme. Vis Neurosci 29, 41–49. [DOI] [PubMed] [Google Scholar]
- Grimes WN, Li W, Chávez AE & Diamond JS (2009). BK channels modulate pre‐ and postsynaptic signaling at reciprocal synapses in retina. Nat Neurosci 12, 585–592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimes WN, Zhang J, Graydon CW, Kachar B & Diamond JS (2010). Retinal parallel processors: more than 100 independent microcircuits operate within a single interneuron. Neuron 65, 873–885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimes WN, Zhang J, Tian H, Graydon CW, Hoon M, Rieke F & Diamond JS (2015). Complex inhibitory microcircuitry regulates retinal signaling near visual threshold. J Neurophysiol 114, 341–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hausselt SE, Euler T, Detwiler PB & Denk W (2007). A dendrite‐autonomous mechanism for direction selectivity in retinal starburst amacrine cells. PLoS Biol 5, 1474–1493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helmstaedter M, Briggman KL, Turaga SC, Jain V, Seung HS & Denk W (2013). Connectomic reconstruction of the inner plexiform layer in the mouse retina. Nature 500, 168–174. [DOI] [PubMed] [Google Scholar]
- Hitchcock PF (1989). Morphology and distribution of synapses onto a type of large field ganglion cell in the retina of the goldfish. J Comp Neurol 283, 177–188. [DOI] [PubMed] [Google Scholar]
- Hsueh HA, Molnar A & Werblin FS (2008). Amacrine‐to‐amacrine cell inhibition in the rabbit retina. J Neurophysiol 100, 2077–2088. [DOI] [PubMed] [Google Scholar]
- Ichinose T & Lukasiewicz PD (2005). Inner and outer retinal pathways both contribute to surround inhibition of salamander ganglion cells. J Physiol 565, 517–535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston J & Lagnado L (2015). General features of the retinal connectome determine the computation of motion anticipation. Elife 2015, 1–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolb H & Nelson R (1981). Amacrine cells of the cat retina. Vision Res 21, 1625–1633. [DOI] [PubMed] [Google Scholar]
- Kolb H & Nelson R (1993). OFF‐alpha and OFF‐beta ganglion cells in cat retina: II. Neural circuitry as revealed by electron microscopy of HRP stains. J Comp Neurol 329, 85–110. [DOI] [PubMed] [Google Scholar]
- Kuffler SW (1953). Discharge patterns and functional organization of mammalian retina. J Neurophysiol 16, 37–68. [DOI] [PubMed] [Google Scholar]
- Lee S, Chen L, Chen M, Ye M, Seal RP & Zhou ZJ (2014). An unconventional glutamatergic circuit in the retina formed by vGluT3 amacrine cells. Neuron 84, 708–715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S & Zhou ZJ (2006). The synaptic mechanism of direction selectivity in distal processes of starburst amacrine cells. Neuron 51, 787–799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee SCS, Meyer A, Schubert T, Hüser L, Dedek K & Haverkamp S (2015). Morphology and connectivity of the small bistratified A8 amacrine cell in the mouse retina. J Comp Neurol 523, 1529–1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin MZ & Schnitzer MJ (2016). Genetically encoded indicators of neuronal activity. Nat Neurosci 19, 1142–1153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindstrom SH, Ryan DG, Shi J & DeVries SH (2014). Kainate receptor subunit diversity underlying response diversity in retinal Off bipolar cells. J Physiol 592, 1457–1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacNeil MA & Masland RH (1998). Extreme diversity among amacrine cells: Implications for function. Neuron 20, 971–982. [DOI] [PubMed] [Google Scholar]
- Macosko EZ, Basu A, Satija R, Nemesh J, Shekhar K, Goldman M, Tirosh I, Bialas AR, Kamitaki N, Martersteck EM, Trombetta JJ, Weitz DA, Sanes JR, Shalek AK, Regev A & McCarroll SA (2015). Highly parallel genome‐wide expression profiling of individual cells using nanoliter droplets. Cell 161, 1202–1214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manookin MB, Beaudoin DL, Ernst ZR, Flagel LJ & Demb JB (2008). Disinhibition combines with excitation to extend the operating range of the off visual pathway in daylight. J Neurosci 28, 4136–4150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchiafava PL (1978). Horizontal cells influence membrane potential of bipolar cells in the retina of the turtle. Nature 275, 141–142. [DOI] [PubMed] [Google Scholar]
- Marvin JS, Borghuis BG, Tian L, Cichon J, Harnett MT, Akerboom J, Gordus A, Renninger SL, Chen T‐W, Bargmann CI, Orger MB, Schreiter ER, Demb JB, Gan W‐B, Hires SA & Looger LL (2013). An optimized fluorescent probe for visualizing glutamate neurotransmission. Nat Methods 10, 162–170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masland RH (2012a). The neuronal organization of the retina. Neuron 76, 266–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masland RH (2012b). The tasks of amacrine cells. Vis Neurosci 29, 3–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menger N, Pow DV & Wässle H (1998). Glycinergic amacrine cells of the rat retina. J Comp Neurol 401, 34–46. [DOI] [PubMed] [Google Scholar]
- Molnar A, Hsueh HA, Roska B & Werblin FS (2009). Crossover inhibition in the retina: Circuitry that compensates for nonlinear rectifying synaptic transmission. J Comput Neurosci 27, 569–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molnar A & Werblin F (2007). Inhibitory feedback shapes bipolar cell responses in the rabbit retina. J Neurophysiol 98, 3423–3435. [DOI] [PubMed] [Google Scholar]
- Münch TA & Werblin FS (2006). Symmetric interactions within a homogeneous starburst cell network can lead to robust asymmetries in dendrites of starburst amacrine cells. J Neurophysiol 96, 471–477. [DOI] [PubMed] [Google Scholar]
- Murphy‐Baum BL & Taylor WR (2015). The synaptic and morphological basis of orientation selectivity in a polyaxonal amacrine cell of the rabbit retina. J Neurosci 35, 13336–13350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naka KI & Nye PW (1971). Role of horizontal cells in organization of the catfish retinal receptive field. J Neurophysiol 34, 785–801. [DOI] [PubMed] [Google Scholar]
- Nikolaev A, Leung K‐M, Odermatt B & Lagnado L (2013). Synaptic mechanisms of adaptation and sensitization in the retina. Nat Neurosci 16, 934–941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Odermatt B, Nikolaev A & Lagnado L (2012). Encoding of luminance and contrast by linear and nonlinear synapses in the retina. Neuron 73, 758–773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oltedal L, Morkve SH, Veruki ML & Hartveit E (2006). Patch‐clamp investigations and compartmental modeling of rod bipolar axon terminals in an in vitro thin‐slice preparation of the mammalian retina. J Neurophysiol 97, 1171–1187. [DOI] [PubMed] [Google Scholar]
- Paredes JM, Idilli AI, Mariotti L, Losi G, Arslanbaeva LR, Sato SS, Artoni P, Szczurkowska J, Cancedda L, Ratto GM, Carmignoto G & Arosio D (2016). Synchronous bioimaging of intracellular pH and chloride based on LSS fluorescent protein. ACS Chem Biol 11, 1652–1660. [DOI] [PubMed] [Google Scholar]
- Poleg‐Polsky A & Diamond JS (2011). Imperfect space clamp permits electrotonic interactions between inhibitory and excitatory synaptic conductances, distorting voltage clamp recordings. PLoS One 6, e19463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pourcho RG & Goebel DJ (1983). Neuronal subpopulations in cat retina which accumulate the GABA agonist, (3H)muscimol: a combined Golgi and autoradiographic study. J Comp Neurol 219, 25–35. [DOI] [PubMed] [Google Scholar]
- Protti DA, Di Marco S, Huang JY, Vonhoff CR, Nguyen V & Solomon SG (2014). Inner retinal inhibition shapes the receptive field of retinal ganglion cells in primate. J Physiol 592, 49–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purgert RJ & Lukasiewicz PD (2015). Differential encoding of spatial information among retinal on cone bipolar cells. J Neurophysiol 114, 1757–1772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raviola E & Dacheux RF (1987). Excitatory dyad synapse in rabbit retina. Proc Natl Acad Sci USA 84, 7324–7328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robles E, Laurell E & Baier H (2014). The retinal projectome reveals brain‐area‐specific visual representations generated by ganglion cell diversity. Curr Biol 24, 2085–2096. [DOI] [PubMed] [Google Scholar]
- Rosa JM, Ruehle S, Ding H, Lagnado L, Rosa JM, Ruehle S, Ding H & Lagnado L (2016). Crossover inhibition generates sustained visual responses in the inner retina. Neuron 90, 308–319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roska B, Nemeth E & Werblin FS (1998). Response to change is facilitated by a three‐neuron disinhibitory pathway in the tiger salamander retina. J Neurosci 18, 3451–3459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanes JR & Masland RH (2015). The types of retinal ganglion cells: current status and implications for neuronal classification. Annu Rev Neurosci 38, 221–246. [DOI] [PubMed] [Google Scholar]
- Schwartz G, Taylor S, Fisher C, Harris R & Berry MJ (2007). Synchronized firing among retinal ganglion cells signals motion reversal. Neuron 55, 958–969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka M & Tachibana M (2013). Independent control of reciprocal and lateral inhibition at the axon terminal of retinal bipolar cells. J Physiol 591, 3833–3851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tien NW, Kim T & Kerschensteiner D (2016). Target‐specific glycinergic transmission from VGluT3‐expressing amacrine cells shapes suppressive contrast responses in the retina. Cell Rep 15, 1369–1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaney DI (1990). The mosaic of amacrine cells in the mammalian retina. Prog Retin Res 9, 49–100. [Google Scholar]
- Vaney DI, Sivyer B & Taylor WR (2012). Direction selectivity in the retina: symmetry and asymmetry in structure and function. Nat Rev Neurosci 13, 194–208. [DOI] [PubMed] [Google Scholar]
- Vielma AH, Retamal MA & Schmachtenberg O (2011). Nitric oxide signaling in the retina: What have we learned in two decades? Brain Res 1430, 112–125. [DOI] [PubMed] [Google Scholar]
- Vigh J, Vickers E & von Gersdorff H (2011). Light‐evoked lateral GABAergic inhibition at single bipolar cell synaptic terminals is driven by distinct retinal microcircuits. J Neurosci 31, 15884–15893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wässle H (2004). Parallel processing in the mammalian retina. Nat Rev Neurosci 5, 747–757. [DOI] [PubMed] [Google Scholar]
- Werblin FS (2010). Six different roles for crossover inhibition in the retina: correcting the nonlinearities of synaptic transmission. Vis Neurosci 27, 1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yonehara K, Farrow K, Ghanem A, Hillier D, Balint K, Teixeira M, Jüttner J, Noda M, Neve RL, Conzelmann K‐K & Roska B (2013). The first stage of cardinal direction selectivity is localized to the dendrites of retinal ganglion cells. Neuron 79, 1078–1085. [DOI] [PubMed] [Google Scholar]
- Zhang C & McCall MA (2012). Receptor targets of amacrine cells. Vis Neurosci 29, 11–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Jung CS & Slaughter MM (1997). Serial inhibitory synapses in retina. Vis Neurosci 14, 553–563. [DOI] [PubMed] [Google Scholar]
- Zhang J, Wang H‐H & Yang C‐Y (2004). Synaptic organization of GABAergic amacrine cells in the salamander retina. Vis Neurosci 21, 817–825. [DOI] [PubMed] [Google Scholar]


