Keywords: nerve regeneration, diabetic encephalopathy, adult neural stem cells, olfactory function, subventricular zone, proliferation, glycogen synthase kinase 3 beta, β-catenin, differentiation, rats, insulin, type 1 diabetes mellitus, neural regeneration
Abstract
Sensitive smell discrimination is based on structural plasticity of the olfactory bulb, which depends on migration and integration of newborn neurons from the subventricular zone. In this study, we examined the relationship between neural stem cell status in the subventricular zone and olfactory function in rats with diabetes mellitus. Streptozotocin was injected through the femoral vein to induce type 1 diabetes mellitus in Sprague-Dawley rats. Two months after injection, olfactory sensitivity was decreased in diabetic rats. Meanwhile, the number of BrdU-positive and BrdU+/DCX+ double-labeled cells was lower in the subventricular zone of diabetic rats compared with age-matched normal rats. Western blot results revealed downregulated expression of insulin receptor β, phosphorylated glycogen synthase kinase 3β, and β-catenin in the subventricular zone of diabetic rats. Altogether, these results indicate that diabetes mellitus causes insulin deficiency, which negatively regulates glycogen synthase kinase 3β and enhances β-catenin degradation, with these changes inhibiting neural stem cell proliferation. Further, these signaling pathways affect proliferation and differentiation of neural stem cells in the subventricular zone. Dysfunction of subventricular zone neural stem cells causes a decline in olfactory bulb structural plasticity and impairs olfactory sensitivity in diabetic rats.
Introduction
In many mammalian species, newborn neurons continue to be integrated into the olfactory bulb. In rodents, the subventricular zone (SVZ) near the lateral ventricle wall generates newborn neurons that migrate to the olfactory bulb where they differentiate into local neurons (Whitman and Greer, 2009). Recently, several studies suggest that adult olfactory neurogenesis may be involved in regulation of olfactory behavior in rodents (Kageyama et al., 2012; Manzini, 2015). Moreno et al. (2009) reported that olfactory learning improves odor distinction and is damaged by infusion of cytosine-β-D-arabinofuranoside (AraC), which inhibits neural stem cell proliferation and survival. Additionally, a previous study showed that AraC infusion decreases short-term olfactory memory and odor detection sensitivity in mice (Breton-Provencher and Saghatelyan, 2012). Furthermore, long-term olfactory memory retention was impaired with AraC treatment, although basic olfactory functions were unaltered (Sultan et al., 2010).
A study has shown that olfactory dysfunction may be an early sign of brain changes in Alzheimer's disease or cognitive impairment because it appears to precede clinical signs. In addition, mild cognitive impairment is accompanied by olfactory dysfunction in patients (Devanand et al., 2000). Several clinical studies also revealed association between olfactory dysfunction and cognitive impairment in the older population (Wilson et al., 2006; Schubert et al., 2008). Some patients with type 2 diabetes mellitus (DM) also suffer from olfactory dysfunction (Le Floch et al., 1993; Infante-Garcia et al., 2015). Indeed, several studies have proposed that olfactory dysfunction in diabetic patients is due to, or at least aggravated by, secondary pathologies (Naka et al., 2010; Brady et al., 2013; Gouveri et al., 2014). Interestingly, epidemiological surveys suggest that diabetes is associated with increased prevalence of Alzheimer's disease (Sahay et al., 2011). Diabetic encephalopathy is characterized by brain atrophy, reactive oxygen species accumulation, reduced synaptic plasticity, and cognitive impairment. These changes are similar to those that occur during acceleration of brain ageing (Biessels et al., 2002; Baquer et al., 2009). We previously showed that aberrant metabolism following insulin deficiency (including hyperglycemia and hyperlipidemia) causes hippocampal atrophy, neurodegeneration, amyloid beta deposition, and declined dendritic spine density in streptozotocin (STZ)-induced diabetic rats (Wang et al., 2014).
Signaling molecules of the Wnt family play important roles in maintaining cellular proliferation, differentiation, migration, and axon guidance during neural development (Ille and Sommer, 2005). Increased β-catenin due to virally transduced expression of a stabilized form of this protein increases proliferation of Ascl1-expressing SVZ cells and olfactory bulb neurogenesis. As the modulator, insulin is implicated in modification of β-catenin signaling (Kim et al., 2013). Additionally, type 1 diabetes mellitus (T1DM) is characterized by absolute insulin deficiency. Therefore, in this study, we determined whether T1DM negatively affects proliferation and differentiation of neural stem cells in SVZ, and explored olfaction changes in this process.
Materials and Methods
Animals
Eight- to 10-week male Sprague-Dawley rats were obtained from the Animal Center of Lanzhou University of China (license No. SCXK (Gan) 2009-0004). Rats were fed in an animal house at 22 ± 2°C and relative humidity of 55 ± 10% on 12-hour light-dark cycle. Rats were allowed free access to food and water. Experimental procedures were approved by the Animal Ethics Committee, Lanzhou University, China. The experiment followed the National Guidelines for the Care and Use of Laboratory Animals, and Consensus Author Guidelines for Animal Use formulated by the International Association of Veterinary Editors (IAVE). The article was prepared in accordance with the Animal Research: Reporting of In Vivo Experiments Guidelines (ARRIVE Guidelines).
Overnight-fasted rats were injected once with 65 mg/kg STZ (Sigma, St. Louis, MO, USA) through the femoral vein to induce DM. Age-matched normal rats received 0.2 mL normal saline. One week after STZ injection, blood samples were collected through the tail vein, and plasma glucose levels measured by plasma glucose test films (Sinocare Inc., Changsha, China) and enzymatic diagnostic kits (Shanghai Rongsheng Biotech Co., Ltd., Shanghai, China). Rats with plasma glucose levels ≥ 300 mg/dL and symptoms of polyuria, polyphagia, and polydipsia were considered diabetic and used in the present study. Diabetic rats (DM group) and age-matched rats (normal group) were raised for 2 months.
Olfactory function evaluated by buried food pellet test
At 55 days after treatment, rats were evaluated for their ability to find food (lab regular diet) hidden underneath bedding as previously described (Montani et al., 2013). Before the test, rats were food deprived for 12 hours with free access to water. A scented pellet was placed at one corner of the clean cage and the time taken to reach the visible pellet recorded. Rats were then removed from the cage and a scented pellet buried underneath a 7 cm-thick layer of bedding. Time from introduction of the animal to the cage until the food pellet was retrieved with its front paws was measured in seconds up to a maximum of 300 seconds. Failure to find the food pellet within the allocated time was represented as 300 seconds. Time to find the buried pellet was recorded. The trial was repeated three times, separated by 10-minute intervals. Latency to find visible food in three trials was averaged.
Olfactory sensitivity test
At 55 days after treatment, rats were placed in a plastic box (40 × 50 × 20 cm3) without food, and ambient noise from the main laboratory blocked. After a 3-minute adaptation, diluted amyl acetate (1:1 and 1:50) was pipetted onto a 5 cm2 piece of filter paper that was taped to a plastic sheet (5 × 5 cm2). The stimulation protocol included a habituation phase with three consecutive purified water presentations (2 minutes each) interrupted by 1-minute inter-trial intervals, which was followed by a dishabituation phase of three consecutive diluted amyl acetate presentations (2 minutes each) separated by 1-minute inter-trial intervals. An odor discrimination response was confirmed when the rat neared its nose against the grid bars opposite the plastic containing the odor cues. The test process was recorded with a video system (HIK vision, Hangzhou, China), and the time rats spent exploring the odor stimulus recorded during 2-minute span exposures.
Injection of bromodeoxyuridine (BrdU)
The BrdU-injection protocol is shown in Figure 1A. Briefly, at 57 days after STZ or saline injection, eight rats from each group were intraperitoneally injected with BrdU (Sigma) (60 mg/kg) once daily for three consecutive days to ensure that dividing and early differentiating cells were labeled. After 4 days, the rats were intraperitoneally anesthetized with chloral hydrate (350 mg/kg) and then intracardially perfused with 0.1 M phosphate-buffered saline (PBS) and 4% paraformaldehyde. Rat brains were removed and incubated with graded sucrose. Serial coronal brain sections were prepared using a cryotome (Leica, Heidelberg, Germany) and then stored at –20°C until use.
Figure 1.
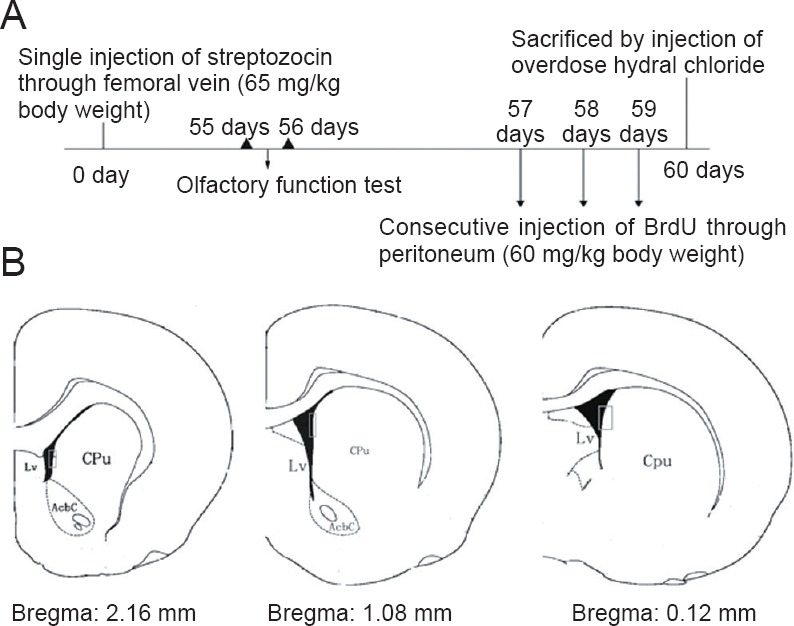
Schematic diagrams.
(A) Experimental flow chart: time of streptozotocin injection, time of behavioral testing, time course of BrdU injection. (B) Three coronal serial sections at 2.16, 1.08, and 0.12 mm from bregma (according to the Brain Atlas, 5th version) were selected from each rat brain. Rectangle frames indicate areas of cell counting in the subventricular zone. Lv: Lateral ventricle; CPu: caudate putamen (striatum); AcbC: nucleus accumbens, core; BrdU: bromodeoxyuridine.
Biochemistry assay
After 2 months, 0.2 mL blood samples were collected from the tail vein at a designated time. Blood samples were centrifuged at 13,000 r/min, and plasma isolated. Cerebrospinal fluid was collected before sacrifice. Plasma and cerebrospinal fluid glucose content were analyzed using appropriate diagnostic kits (Randox, Crumlin Co., Antrim, UK), according to the manufacturer's instructions. Plasma insulin levels were tested using an enzyme linked immunosorbent assay kit (R&D Systems Inc., Minneapolis, MN, USA). Results were calculated from standard curves. Body weight was analyzed using consecutive metabolic tests.
Immunohistochemistry
After 2 months, rats were sacrificed and brain coronal sections obtained. For BrdU immunostaining, sections were obtained at 2.16, 1.08, and 0.12 mm from bregma (Figure 1B). Sections were initially incubated with 2 N HCl for 30 minutes at 45°C, rinsed in PBS buffer (0.1 M, pH 7.6), and then incubated in 1% H2O2 for 10 minutes. After washing with PBS (0.1 M, pH 7.6) for 15 minutes, sections were placed in 10% goat serum at 37°C for 1 hour and then incubated overnight with mouse monoclonal anti-BrdU antibody (Abcam, Cambridge, UK) (1:200) at 4°C. After washing, sections were incubated with Cy3-conjugated anti-mouse IgG (Beijing Zhongshan Golden Bridge Biotechnology, Beijing, China) (1:50) at 37°C for 1 hour in the dark, and then observed under a fluorescence microscope. For double staining of BrdU and doublecortin (DCX), free-floating sections were initially prepared as described above for BrdU staining. After washing, sections were treated again overnight with primary antibodies against goat polyclonal anti-DCX (Abcam, Cambridge, UK) (1:200) at 4°C. Afterwards, sections were incubated with corresponding fluorescein isothiocyanate-conjugated secondary antibodies (Beijing Zhongshan Golden Bridge Biotechnology) (1:50) prior to observation under a confocal microscope (Leica). All negative controls underwent the same treatment without primary antibodies, and with no specific staining detected. Positive cells were counted in a blind manner. Total cell numbers were obtained in a double-blind manner. In these experiments, BrdU-positive cells and BrdU+/DCX+ double-labeled cells were obtained from three SVZ sections (at 2.16, 1.08, and 0.12 mm from bregma; Figure 1B) from each rat, and manually counted under a 20× objective.
Western blot assay
Five rats from each group were decapitated, and their brains removed and placed on ice plates. Bilateral SVZ were dissected and frozen in liquid nitrogen. Total protein was extracted in lysates containing protein inhibitor cocktail. Protein (30 μg) was fractionated on 10% sodium dodecyl sulfate-polyacrylamide gels for electrophoresis, and then transferred onto polyvinylidene fluoride membranes. Membranes were blotted overnight with anti-insulin receptor β (IRβ) (1:1,000), anti-glycogen synthase kinase 3 beta (GSK3β) (1:1,000; Cell Signaling, Boston, MA, USA), anti-phospho-glycogen synthase kinase 3 beta (p-GSK3β) (1:1,000; Cell Signaling), anti-β-catenin (1:1,000; Santa Cruz Biotechnology, Santa Cruz, CA, USA), and anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (1:5,000; Santa Cruz Biotechnology) at 4°C. Membranes were washed with Tris-buffered saline containing Tween 20 and then blotted with corresponding horseradish peroxidase-conjugated secondary antibodies (1:5,000). Blotted bands were visualized by enhanced chemiluminescence and analyzed by Image J software (NIH, Bethesda, MD, USA). All western blot experiments were performed at least three times. Lanes were scanned and optical density normalized using GAPDH as an internal control.
Statistical analysis
All data are expressed as the mean ± SEM, and were analyzed with SPSS 17.0 software (SPSS, Chicago, IL, USA). One-way analysis of variance was used for multiple-group comparisons; Tukey's post-hoc analysis was performed for unpaired-group comparisons. P values < 0.05 were considered statistically significant.
Results
Metabolic parameters
Consecutive metabolic testing showed reduced body weight (Figure 2D) in diabetic rats compared with age-matched rats (P < 0.01). Consecutive plasma examination showed higher glucose levels (Figure 2A), but lower insulin (Figure 2C) in diabetic rats compared with age-matched rats (P < 0.01). Consistently, glucose levels increased in cerebrospinal fluid of diabetic rats compared with normal rats (Figure 2B) (P < 0.01).
Figure 2.
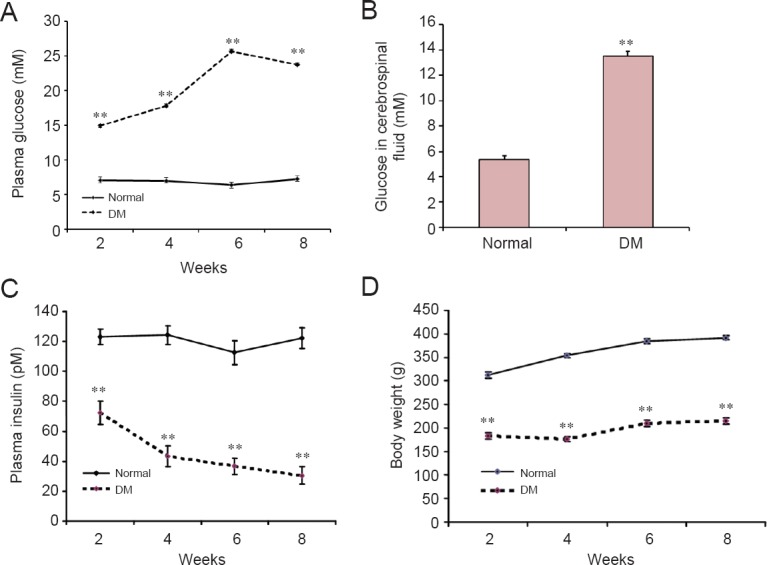
Metabolic parameters from rats in 2 consecutive months after streptozotocin injection.
(A) Plasma glucose increased in the DM group compared with the normal group. (B) Glucose levels in cerebrospinal fluid increased in the DM group compared with the normal group at 8 weeks after treatment. (C) Enzyme linked immunosorbent assay showed decreased plasma insulin in the DM group compared with the normal group. (D) Body weight gain was less in the DM group compared with the normal group. DM group: Overnight-fasted rats were injected once with 65 mg/kg streptozotocin through the femoral vein to induce DM. Normal group: Age-matched normal rats received an equivalent volume of normal saline. **P < 0.01, vs. normal group (mean ± SEM, n = 13). One-way analysis of variance was performed for multiple-group comparisons, with Tukey's post-hoc analysis for unpaired group comparisons. Experiments were performed in triplicate. DM: Diabetes mellitus.
Diabetes impaired olfactory function
Two months after STZ injection, olfactory sensitivity was evaluated using the buried food test and odor discrimination. As shown in Figure 3A, diabetic rats showed no difference compared with normal rats in time taken to reach the visible pellet, indicating no speed difference. In the buried pellet trial, diabetic rats required a notably longer time to reach the pellet than age-matched normal rats (Figure 3B) (P < 0.01), indicating impaired olfactory performance. Time spent exploring high-concentration amyl acetate (1:1) showed significant increases compared with the water stimulus in both diabetic rats and normal rats, but the increase was smaller in the diabetic group compared with the normal group (P < 0.05; Figure 3C). Time exploring low-concentration amyl acetate (1:50) odor showed significant increases in the diabetic and normal groups, with a higher increase in the normal group compared with the diabetic group (P < 0.05; Figure 3D).
Figure 3.
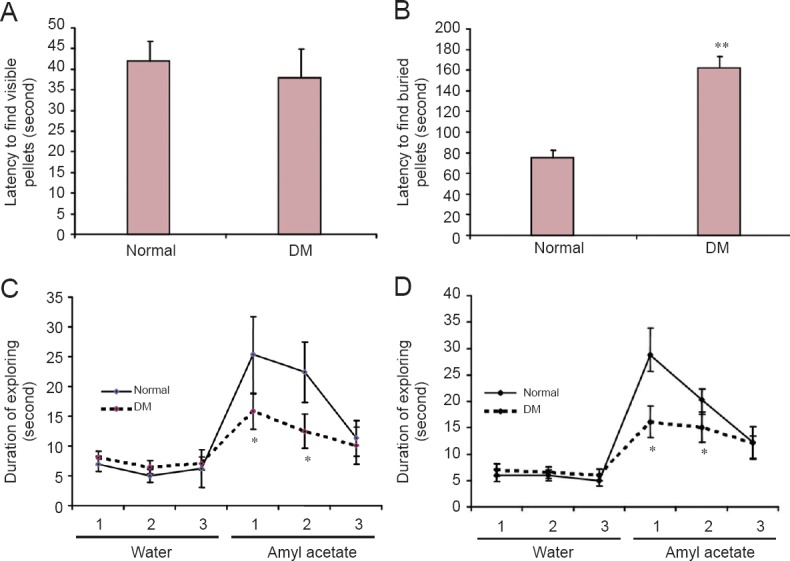
Olfactory sensitivity in normal and diabetic rats after 2 months of treatment.
(A) No difference in latency to find visible food was found between the DM group and normal group by buried food test. (B) Latency to find the buried pellet was longer in the DM group than in the normal group. (C) Olfactory sensitivity test found that the time spent exploring high-concentration amyl acetate (1:1) decreased in the DM group compared with the normal group in trials 1 and 2. (D) Time spent exploring low-concentration amyl acetate (1:50) decreased in the DM group compared with the normal group in trials 1 and 2. DM group: Overnight-fasted rats were injected once with 65 mg/kg streptozotocin through the femoral vein to induce DM. Normal group: Age-matched normal rats received an equivalent volume of normal saline. *P < 0.05, **P < 0.01, vs. normal group (mean ± SEM, n = 13). One-way analysis of variance was performed for multiple-group comparisons, with Tukey's post-hoc analysis for unpaired group comparisons. Experiments were performed in triplicate (1, 2, 3). DM: Diabetes mellitus.
Diabetes reduced proliferation and differentiation of neural stem cells in SVZ
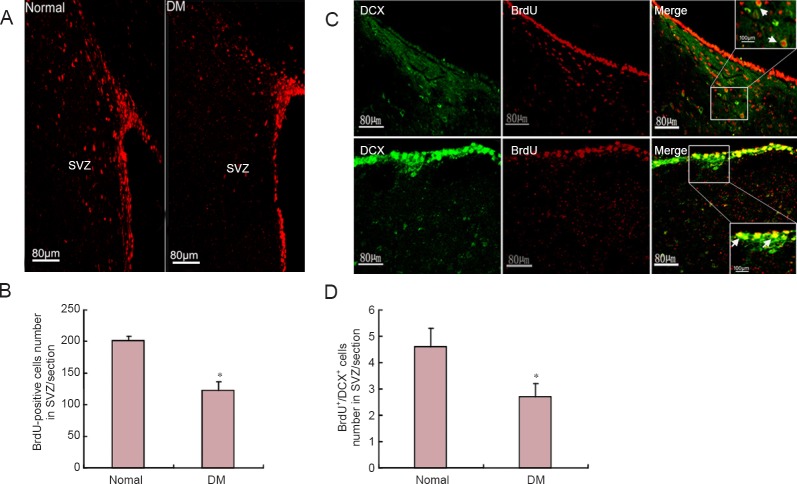
BrdU labeling was used to examine proliferation of neural stem cells in SVZ. The number of BrdU-positive cells in SVZ was lower in diabetic rats (120.3 ± 23.2) than age-matched normal rats (202.2 ± 10.8; P < 0.05; Figure 4A, B). Immuno-double labeling, cell counting, and confocal microscopy were performed to examine differentiation ability of neural precursor cells in SVZ of diabetic rats. Cells labeled with BrdU and DCX were identified as immature, newly generated neurons. The number of BrdU+/DCX+ cells in SVZ was significantly lower in diabetic rats than in age-matched rats (P < 0.05; Figure 4C, D).
Figure 4.
Effect of DM on neural stem cell status in SVZ.
Representative images of BrdU staining and immunofluorescence histochemistry were observed by fluorescence microscope. (B) BrdU-positive cells were counted. Positive cell number decreased in the DM group compared with the normal group. (C) Representative images of BrdU and DCX double-labeling, with immunofluorescence staining observed by confocal microscopy. Arrows indicate double-labeled cells. Squares in right images are magnified in upper/lower corners. (D) BrdU+/DCX+ cells decreased in the DM group compared with the normal group. DM group: Overnight-fasted rats were injected once with 65 mg/kg streptozotocin through the femoral vein to induce DM. Normal group: Age-matched normal rats received an equivalent volume of normal saline. *P < 0.05, vs. normal group (mean ± SEM, n = 8). One-way analysis of variance was performed for multiple-group comparisons, with Tukey's post-hoc analysis performed for unpaired group comparisons. Experiments were performed in triplicate. BrdU: Bromodeoxyuridine; DCX: doublecortin; DM: diabetes mellitus; SVZ: subventricular zone.
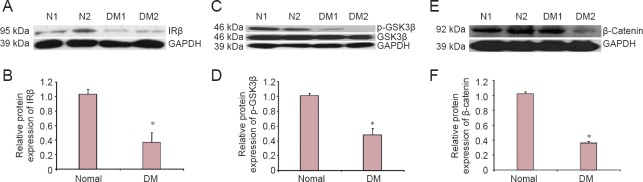
Changes in insulin/GSK3β/β-catenin signaling in SVZ of diabetic rats
Insulin signals play important roles in maintaining energy balance and neuronal survival in the central nervous system. In the central nervous system, most insulin is produced by pancreatic islets and transferred into the central nervous system across the blood-brain barrier (Schwartz et al., 1991). T1DM is characterized by an absolute insulin deficit throughout the whole body, including the brain. To determine whether insulin signaling is impaired in SVZ, IRβ protein levels were tested by western blot assay. Our results show that IRβ expression levels in SVZ were lower by approximately 60% in diabetic rats compared with age-matched normal rats (P < 0.05; Figure 5A, B). Many downstream signals are regulated by insulin including GSK3β and β-catenin. Cell cycle and proliferation are regulated by β-catenin, and β-catenin activity is negatively regulated by GSK3β. Thus, we measured GSK3β activity. Our results show that GSK3β phosphorylation (at lysine 9) was significantly lower in SVZ of diabetic rats than age-matched normal rats (P < 0.05; Figure 5C, D), suggesting increased GSK3β activity. Consistently, β-catenin expression levels were lower in SVZ of diabetic rats than age-matched normal rats (P < 0.05; Figure 5E, F).
Figure 5.
Effect of DM on IRβ, p-GSK3β, and β-catenin expression in SVZ 8 weeks after treatment (western blot assay).
(A) Representative IRβ immunoblots. (B) Relative IRβ expression decreased in the DM group compared with the normal group. (C) Representative p-GSK3β immunoblots. (D) Relative p-GSK3β expression decreased in the DM group compared with the normal group. (E) Representative β-catenin immunoblots. (F) Relative β-catenin expression decreased in the DM group compared with the normal group. DM group: Overnight-fasted rats were injected once with 65 mg/kg streptozotocin through the femoral vein to induce DM. Normal group: Age-matched normal rats received an equivalent volume of normal saline. N1, 2: Normal groups, age-matched normal rats received an equivalent volume of normal saline; DM1, 2: DM groups, overnight-fasted rats were injected once with 65 mg/kg streptozotocin through the femoral vein to induce DM. *P < 0.05, vs. normal group (mean ± SD, n = 5). One-way analysis of variance was performed for multiple-group comparisons, with Tukey's post-hoc analysis for unpaired group comparisons. Experiments were performed in triplicate. DM: Diabetes mellitus; IRβ: insulin receptor β; GSK3β: glycogen synthase kinase 3 beta; p-GSK3β: phospho-glycogen synthase kinase 3 beta; GAPDH: glyceraldehyde phosphate dehydrogenase; SVZ: subventricular zone.
Discussion
Here, we show that proliferation of adult neural stem cells is markedly lower in SVZ of diabetic rats compared with normal rats. Particularly, the number of BrdU-positive cells located in the lateral ventricle margin was significantly reduced. It has been shown that the lateral ventricle wall is principally comprised of ependymal cells, which possess neural stem cell characteristics (Tong et al., 2014). In our experiments, SVZ cell types were identified by transmission electron microscopy. We found that in DM rats, proliferated SVZ cells are mainly ependymal cells located along the lateral ventricle. Nevertheless, in the normal group, proliferated SVZ cells include ependymal cells, astrocytes, and neuroblasts. Our results suggest that differential localization of proliferated cells between normal and DM groups may contribute to divergent cell types. DCX is a microtubule-associated protein implicated in neuronal migration during development and adulthood. DCX expression is transitory during adult neurogenesis, dropping off with the emergence of mature neuronal markers, and primarily localized to areas of continuous neurogenesis and rarely elsewhere (Brown et al., 2003; Keays, 2007; von Bohlen und Halbach, 2011). Dramatically, compared with other markers (such as nestin and GFAP), DCX is particular to the neuronal lineage. Our results show that BrdU/DCX double-positive cells located in SVZ decrease significantly in DM rats compared with the normal group. Similarly, BrdU/GFAP double-positive cells also decreased in diabetic rats. This suggests that progenitor cell differentiation in SVZ is impaired under the diabetic condition.
The mechanisms underlying neural progenitor cell determination are complicated. Insulin signaling in the brain is implicated in modification of proliferation, differentiation, and survival of neural progenitor cells. Insulin deficiency is important to T1DM. Moreover, IR expression significantly decreased in SVZ of diabetic rats, suggesting lower insulin signaling activity in diabetic rats than in age-matched normal rats. Insulin signaling activity can phosphorylate PI3K/PKB and FOXO to suppress FOXO-target gene transcription, and thereby improve neuronal survival and inhibit neuronal apoptosis (Xu et al., 2012). In addition, insulin signaling activates the Ras-MAPK signaling pathway, which promotes E2F translocation into the nucleus and facilitates cell duplication and proliferation (Real et al., 2011). Active PKB can phosphorylate GSK3β, which is an effector of the Wnt signaling pathway. Phosphorylated GSK3β inhibits β-catenin degradation, enhances β-catenin aggregation, and improves β-catenin translocation into the nucleus (Li et al., 2014). β-Catenin located in the nucleus enhances cell proliferation by regulating cyclin-dependent kinases (Davidson and Niehrs, 2010). Enhancement of β-catenin promotes neural progenitor cell proliferation in SVZ of adult mouse (Adachi et al., 2007). Additionally, active GSK3β modifies neurite and growth cone development, and also alters synaptic plasticity (Hall et al., 2002). In our present experiments, GSK3β phosphorylation levels decreased in SVZ of diabetic rats. Similarly, β-catenin levels were lower in SVZ of diabetic rats than age-matched normal rats. We inferred that proliferation and differentiation of adult neural stem cells in SVZ are modified by the insulin/GSK3β/β-catenin signaling axis. Le Floch et al. (1993) reported that olfactory recognition is impaired in patients with DM. Olfactory dysfunction is associated with age and degenerative complications of diabetes (Le Floch et al., 1993). Evaluation of olfactory function usually consists of the threshold assay of lowest detectable odorant concentration, discrimination ability, and identification between odorants (Eibenstein et al., 2005). Recently, several studies reported that decline of olfactory identification and discrimination, but not olfactory threshold, are strongly associated with risk of higher cognitive impairment (Wilson et al., 2006; Schubert et al., 2008; Sohrabi et al., 2009). The pathology of diabetic encephalopathy is involved in insulin deficiency and reactive oxygen species accumulation, which decrease glucose utilization in the brain. These pathologies are strongly associated with cognitive impairment, which is the main symptom of clinical diabetic encephalopathy. In clinical studies, incidence rates of brain atrophy, enlarged lateral ventricles, and cognitive impairment are higher in the diabetic elderly population than the nondiabetic elderly population (Biessels et al., 2002; Korf et al., 2006). Diabetic mice exhibit reduced hippocampal synaptic plasticity and impaired cognition (Arum et al., 2014). These pathologies and behaviors due to DM are similar to acceleration of brain ageing. Ageing is considered a critical negative factor in maintenance of adult stem cell status. Proliferation ability of neural precursors in the subgranular zone and SVZ are dramatically lower in aged mice than in young mice (Arum et al., 2014). Our findings show that proliferation and survival of SVZ precursors are decreased in diabetic rats, suggesting that DM is a negative factor in neurogenesis, accelerates brain ageing, and causes complicated neural symptoms including mild cognitive impairment, depression, and olfactory deficiency. Likewise, in this study, olfactory sensitivity was also reduced in DM rats compared with normal rats. Even so, olfactory bulb structure showed no obvious change in diabetic rats. Furthermore, BrdU-positive cells did not decrease in the diabetic olfactory bulb compared with normal rats.
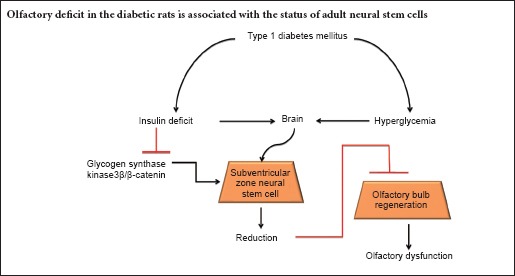
In brief, the SVZ niche is changed in DM, exhibiting disturbed status of neural stem cells, which decrease the proliferation and differentiation abilities of adult neural stem cells in SVZ. The mechanisms underlying DM-induced abnormalities of adult neural stem cells are involved in deregulation of GSK3β and β-catenin signals. Diabetes-induced olfactory deficits are partly associated with neural stem cell impairments in SVZ.
Footnotes
Funding: This work is partly supported by the National Natural Science Foundation of China, No. 81370448, 81570725.
Conflicts of interest: None declared.
Research ethics: The study protocol was approved by the Animal Ethics Committee, Lanzhou University, China. The experimental procedure followed the United States National Institutes of Health Guide for Care and Use of Laboratory Animals (NIH Publication No. 85-23, revised 1986), and “Consensus Author Guidelines on Animal Ethics and Welfare” produced by the International Association for Veterinary Editors (IAVE). All efforts were made to minimize the suffering and number of animals used in this study. The article was prepared in accordance with the “Animal Research: Reporting of In Vivo Experiments Guidelines” (ARRIVE Guidelines).
Contributor agreement: A statement of “Publishing Agreement” has been signed by an authorized author on behalf of all authors prior to publication.
Plagiarism check: This paper has been checked twice with duplication-checking software iThenticate.
Peer review: A double-blind and stringent peer review process has been performed to ensure the integrity, quality and significance of this paper.
Copyedited by Wang J, Li CH, Qiu Y, Song LP, Zhao M
References
- 1.Adachi K, Mirzadeh Z, Sakaguchi M, Yamashita T, Nikolcheva T, Gotoh Y, Peltz G, Gong L, Kawase T, Alvarez-Buylla A, Okano H, Sawamoto K. Beta-catenin signaling promotes proliferation of progenitor cells in the adult mouse subventricular zone. Stem Cells. 2007;25:2827–2836. doi: 10.1634/stemcells.2007-0177. [DOI] [PubMed] [Google Scholar]
- 2.Arum O, Boparai RK, Saleh JK, Wang F, Dirks AL, Turner JG, Kopchick JJ, Liu JL, Khardori RK, Bartke A. Specific suppression of insulin sensitivity in growth hormone receptor gene-disrupted (GHR-KO) mice attenuates phenotypic features of slow aging. Aging Cell. 2014;13:981–1000. doi: 10.1111/acel.12262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baquer NZ, Taha A, Kumar P, McLean P, Cowsik SM, Kale RK, Singh R, Sharma D. A metabolic and functional overview of brain aging linked to neurological disorders. Biogerontology. 2009;10:377–413. doi: 10.1007/s10522-009-9226-2. [DOI] [PubMed] [Google Scholar]
- 4.Biessels GJ, van der Heide LP, Kamal A, Bleys RL, Gispen WH. Ageing and diabetes: implications for brain function. Eur J Pharmacol. 2002;441:1–14. doi: 10.1016/s0014-2999(02)01486-3. [DOI] [PubMed] [Google Scholar]
- 5.Brady S, Lalli P, Midha N, Chan A, Garven A, Chan C, Toth C. Presence of neuropathic pain may explain poor performances on olfactory testing in diabetes mellitus patients. Chem Senses. 2013;38:497–507. doi: 10.1093/chemse/bjt013. [DOI] [PubMed] [Google Scholar]
- 6.Breton-Provencher V, Saghatelyan A. Newborn neurons in the adult olfactory bulb: unique properties for specific odor behavior. Behav Brain Res. 2012;227:480–489. doi: 10.1016/j.bbr.2011.08.001. [DOI] [PubMed] [Google Scholar]
- 7.Brown JP, Couillard-Despres S, Cooper-Kuhn CM, Winkler J, Aigner L, Kuhn HG. Transient expression of doublecortin during adult neurogenesis. J Comp Neurol. 2003;467:1–10. doi: 10.1002/cne.10874. [DOI] [PubMed] [Google Scholar]
- 8.Davidson G, Niehrs C. Emerging links between CDK cell cycle regulators and Wnt signaling. Trends Cell Biol. 2010;20:453–460. doi: 10.1016/j.tcb.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 9.Devanand DP, Michaels-Marston KS, Liu X, Pelton GH, Padilla M, Marder K, Bell K, Stern Y, Mayeux R. Olfactory deficits in patients with mild cognitive impairment predict Alzheimer's disease at follow-up. Am J Psychiatry. 2000;157:1399–1405. doi: 10.1176/appi.ajp.157.9.1399. [DOI] [PubMed] [Google Scholar]
- 10.Eibenstein A, Fioretti AB, Lena C, Rosati N, Amabile G, Fusetti M. Modern psychophysical tests to assess olfactory function. Neurol Res. 2005;26:147–155. doi: 10.1007/s10072-005-0452-3. [DOI] [PubMed] [Google Scholar]
- 11.Gouveri E, Katotomichelakis M, Gouveris H, Danielides V, Maltezos E, Papanas N. Olfactory dysfunction in type 2 diabetes mellitus: an additional manifestation of microvascular disease? Angiology. 2014;65:869–876. doi: 10.1177/0003319714520956. [DOI] [PubMed] [Google Scholar]
- 12.Hall AC, Brennan A, Goold RG, Cleverley K, Lucas FR, Gordon-Weeks PR, Salinas PC. Valproate regulates GSK-3-mediated axonal remodeling and synapsin I clustering in developing neurons. Mol Cell Neurosci. 2002;20:257–270. doi: 10.1006/mcne.2002.1117. [DOI] [PubMed] [Google Scholar]
- 13.Ille F, Sommer L. Wnt signaling: multiple functions in neural development. Cell Mol Life Sci. 2005;62:1100–1108. doi: 10.1007/s00018-005-4552-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Infante-Garcia C, Ramos-Rodriguez JJ, Garcia-Alloza M. Prediabetes and type 2 diabetes implication in central proliferation and neurogenesis. Neural Regen Res. 2015;10:28–29. doi: 10.4103/1673-5374.150646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kageyama R, Imayoshi I, Sakamoto M. The role of neurogenesis in olfaction-dependent behaviors. Behav Brain Res. 2012;227:459–463. doi: 10.1016/j.bbr.2011.04.038. [DOI] [PubMed] [Google Scholar]
- 16.Keays DA. Neuronal migration: unraveling the molecular pathway with humans, mice, and a fungus. Mamm Genome. 2007;18:425–430. doi: 10.1007/s00335-007-9034-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim MH, Hong SH, Lee MK. Insulin receptor-overexpressing beta-cells ameliorate hyperglycemia in diabetic rats through Wnt signaling activation. PLoS One. 2013;8:e67802. doi: 10.1371/journal.pone.0067802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Korf ES, White LR, Scheltens P, Launer LJ. Brain aging in very old men with type 2 diabetes: the Honolulu-Asia Aging Study. Diabetes Care. 2006;29:2268–2274. doi: 10.2337/dc06-0243. [DOI] [PubMed] [Google Scholar]
- 19.Le Floch JP, Le Lievre G, Labroue M, Paul M, Peynegre R, Perlemuter L. Smell dysfunction and related factors in diabetic patients. Diabetes care. 1993;16:934–937. doi: 10.2337/diacare.16.6.934. [DOI] [PubMed] [Google Scholar]
- 20.Li CL, Sathyamurthy A, Oldenborg A, Tank D, Ramanan N. SRF phosphorylation by glycogen synthase kinase-3 promotes axon growth in hippocampal neurons. J Neurosci. 2014;34:4027–4042. doi: 10.1523/JNEUROSCI.4677-12.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Manzini I. From neurogenesis to neuronal regeneration: the amphibian olfactory system as a model to visualize neuronal development in vivo. Neural Regen Res. 2015;10:872–874. doi: 10.4103/1673-5374.158334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Montani G, Tonelli S, Sanghez V, Ferrari PF, Palanza P, Zimmer A, Tirindelli R. Aggressive behaviour and physiological responses to pheromones are strongly impaired in mice deficient for the olfactory G-protein -subunit G8. J Physiol. 2013;591:3949–3962. doi: 10.1113/jphysiol.2012.247528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Moreno MM, Linster C, Escanilla O, Sacquet J, Didier A, Mandairon N. Olfactory perceptual learning requires adult neurogenesis. PNAS. 2009;106:17980–17985. doi: 10.1073/pnas.0907063106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Naka A, Riedl M, Luger A, Hummel T, Mueller CA. Clinical significance of smell and taste disorders in patients with diabetes mellitus. Eur Arch Otorhinolaryngol. 2010;267:547–550. doi: 10.1007/s00405-009-1123-4. [DOI] [PubMed] [Google Scholar]
- 25.Real S, Meo-Evoli N, Espada L, Tauler A. E2F1 regulates cellular growth by mTORC1 signaling. PLoS One. 2011;6:e16163. doi: 10.1371/journal.pone.0016163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sahay A, Scobie KN, Hill AS, O’Carroll CM, Kheirbek MA, Burghardt NS, Fenton AA, Dranovsky A, Hen R. Increasing adult hippocampal neurogenesis is sufficient to improve pattern separation. Nature. 2011;472:466–470. doi: 10.1038/nature09817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schubert CR, Carmichael LL, Murphy C, Klein BE, Klein R, Cruickshanks KJ. Olfaction and the 5-year incidence of cognitive impairment in an epidemiological study of older adults. J Am Geriatr Soc. 2008;56:1517–1521. doi: 10.1111/j.1532-5415.2008.01826.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schwartz MW, Bergman RN, Kahn SE, Taborsky GJ, Jr, Fisher LD, Sipols AJ, Woods SC, Steil GM, Porte D., Jr Evidence for entry of plasma insulin into cerebrospinal fluid through an intermediate compartment in dogs. Quantitative aspects and implications for transport. J Clin Invest. 1991;88:1272–1281. doi: 10.1172/JCI115431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sohrabi HR, Bates KA, Rodrigues M, Taddei K, Laws SM, Lautenschlager NT, Dhaliwal SS, Johnston AN, Mackay-Sim A, Gandy S, Foster JK, Martins RN. Olfactory dysfunction is associated with subjective memory complaints in community-dwelling elderly individuals. J Alzheimers Dis. 2009;17:135–142. doi: 10.3233/JAD-2009-1020. [DOI] [PubMed] [Google Scholar]
- 30.Sultan S, Mandairon N, Kermen F, Garcia S, Sacquet J, Didier A. Learning-dependent neurogenesis in the olfactory bulb determines long-term olfactory memory. FASEB J. 2010;24:2355–2363. doi: 10.1096/fj.09-151456. [DOI] [PubMed] [Google Scholar]
- 31.Tong CK, Chen J, Cebrian-Silla A, Mirzadeh Z, Obernier K, Guinto CD, Tecott LH, Garcia-Verdugo JM, Kriegstein A, Alvarez-Buylla A. Axonal control of the adult neural stem cell niche. Cell Stem Cell. 2014;14:500–511. doi: 10.1016/j.stem.2014.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.von Bohlen und Halbach O. Immunohistological markers for proliferative events, gliogenesis, and neurogenesis within the adult hippocampus. Cell Tissue Res. 2011;345:1–19. doi: 10.1007/s00441-011-1196-4. [DOI] [PubMed] [Google Scholar]
- 33.Wang JQ, Yin J, Song YF, Zhang L, Ren YX, Wang DG, Gao LP, Jing YH. Brain aging and AD-like pathology in streptozotocin-induced diabetic rats. J Diabetes Res. 2014;2014:796840. doi: 10.1155/2014/796840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Whitman MC, Greer CA. Adult neurogenesis and the olfactory system. Prog Neurobiol. 2009;89:162–175. doi: 10.1016/j.pneurobio.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wilson RS, Arnold SE, Tang Y, Bennett DA. Odor identification and decline in different cognitive domains in old age. Neuroepidemiology. 2006;26:61–67. doi: 10.1159/000090250. [DOI] [PubMed] [Google Scholar]
- 36.Xu F, Gu JH, Qin ZH. Neuronal autophagy in cerebral ischemia. Neurosci Bull. 2012;28:658–666. doi: 10.1007/s12264-012-1268-9. [DOI] [PMC free article] [PubMed] [Google Scholar]