Abstract
TREX1/DNASE III, the most abundant 3′-5′ DNA exonuclease in mammalian cells, is tail-anchored on the endoplasmic reticulum (ER). Mutations at the N-terminus affecting TREX1 DNase activity are associated with autoimmune and inflammatory conditions such as Aicardi-Goutières syndrome (AGS). Mutations in the C-terminus of TREX1 cause loss of localization to the ER and dysregulation of oligosacchryltransferase (OST) activity, and are associated with retinal vasculopathy with cerebral leukodystrophy (RVCL) and in some cases with systemic lupus erythematosus (SLE). Here we investigate mice with conditional expression of the most common RVCL mutation, V235fs, and another mouse expressing a conditional C-terminal mutation, D272fs, associated with a case of human SLE. Mice homozygous for either mutant allele express the encoded human TREX1 truncations without endogenous mouse TREX1, and both remain DNase active in tissues. The two mouse strains are similar phenotypically without major signs of retinal, cerebral or renal disease but exhibit striking elevations of autoantibodies in the serum. The broad range of autoantibodies is primarily against non-nuclear antigens, in sharp contrast to the predominantly DNA-related autoantibodies produced by a TREX1-D18N mouse that specifically lacks DNase activity. We also found that treatment with an OST inhibitor, aclacinomycin, rapidly suppressed autoantibody production in the TREX1 frame-shift mutant mice. Together, our study presents two new mouse models based on TREX1 frame-shift mutations with a unique set of serologic autoimmune-like phenotypes.
1. Introduction
Developing a full understanding of the pathogenesis of human disease as well as modes of treatment or prevention often requires animal models. This process is complicated when different mutations in a single gene result in markedly divergent phenotypes. This is the case with the 3′-5′ DNA exonuclease, TREX1. TREX1 functions as a homodimer with the N-terminal catalytic domain in the cytoplasm and the C-terminus anchoring the protein in the endoplasmic reticulum (ER). Mutations disrupting the catalytic activity of the protein drive the development of a severe pediatric neuro-inflammatory disease Aicardi-Goutières syndrome (AGS) as well as the autoimmune disease systemic lupus erythematosus (SLE) and familial chilblain lupus (FCL), a pediatric disease also associated with autoimmune manifestations [1]. Both AGS and SLE are associated with high levels of IFNα. In contrast, a third quite distinct disease, termed retinal vasculopathy with cerebral leukodystrophy (RVCL), is caused by mutations in the C-terminus that appear to have no effect on TREX1 DNase activity but result in loss of anchoring in the ER and redistribution throughout the cell [2,3]. Individuals carrying this mutation are essentially normal into their forties but then die of a progressive, debilitating neurodegenerative process associated with profound retinal changes [2,3]. However, not all C-terminal truncations are associated with RVCL with some being found in individuals diagnosed with SLE [1].
Previous efforts to understand the mechanisms contributing to the development of these distinct human diseases caused by mutations in the TREX1 gene centered on mice bearing a null mutation of the gene [4–8]. Trex1−/− mice develop a lethal inflammatory cardiomyopathy [4,5], a condition not observed in individuals carrying any of the different mutant alleles of TREX1 described previously. Humans heterozygous for another mutation obviating TREX1 catalytic activity, D18N, develop a monogenic cutaneous form of lupus termed familial chilblain lupus (FCL). A recently established knock-in mouse, TREX1-D18N, disrupting only the DNase activity, develops similar autoimmune disease phenotypes as Trex1−/− mice when expressed in the homozygous state, including inflammation in multiple tissues and elevated autoantibodies [9]. The average survival of TREX1-D18N knock-in mice (over one year) is much longer than Trex1−/− mice (8–10 weeks), suggesting that the lack of TREX1 DNase activity cannot fully explain the severe pathology observed in Trex1−/− mice. Importantly, D18N heterozygous animals exhibited normal survival and had no more inflammation than found in WT mice (9). Thus, modeling the effects in mice of a simple catalase-dead mutation found in human FCL or AGS remains unfulfilled. These observations raise the possibility that other DNase-dependent and -independent functions of TREX1 may exist, and that additional efforts to develop closer disease models, especially for the C-terminal mutations, would be important.
Here we describe two knock-in mouse models carrying human TREX1 frame-shift mutations that truncate the C-terminus of TREX1: V235fs, the most common mutation associated with RVCL [2,3]; and D272fs, identified in an early study in one patient with SLE [10]. We generated mice with the mutant human TREX1 coding exons swapped for those sequences in the mouse (TREX1 is a single exon gene in both human and mouse). Our previous study using mice bearing the V235fs mutant allele revealed dysregulation of the ER oligosaccharyltransferase (OST) complex leading to free glycan release, immune activation and autoantibody production [11]. Paralleling findings were made with cells from Trex1−/− mice and with TREX1-V235fs patient lymphoblasts validating the model [11]. The present study was undertaken to comprehensively analyze autoimmune and other phenotypes of V235fs and D272fs mice.
2. Results
2.1. Human TREX1 C-terminal truncations expressed in mice are DNase active
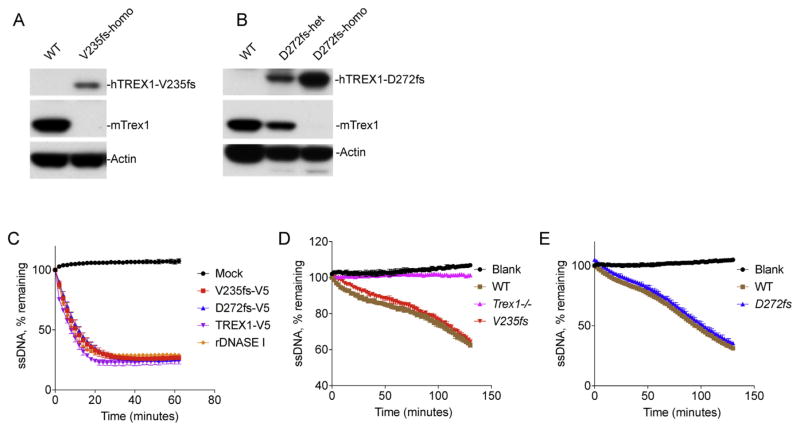
To better model the effects of human mutations associated with the development of RVCL and SLE, we generated two transgenic mouse lines expressing human TREX1-V235fs (associated with RVCL, [11]) or TREX1-D272fs (associated with SLE, new in this study) under the control of the endogenous mouse promoter. We crossed each floxed allele with Meox2-Cre or Vav1-Cre mice to generate whole body knock-ins of the human transgene (designated V235fs and D272fs, same throughout). We first validated expression of TREX1 mutants by immunoblots with specific antibodies against human or mouse TREX1. We found that both V235fs and D272fs transgenic lines successfully expressed the expected human mutant proteins and eliminated mouse Trex1 ([11], Fig. 1A and B). Both mutants are expected to remain DNase active because the frame-shift mutations only remove the C-terminal ER localization domain. Indeed, they are active when transiently expressed in HeLa cells by plasmid transfection (Fig. 1C). To determine whether the human mutants expressed in transgenic mice are also DNase active, we isolated tissue extracts from WT, Trex1−/−, homozygous V235fs, homozygous D272fs mouse spleens and examined exonuclease activity in vitro with a fluorescent assay using 30-mer ssDNA as a substrate [11]. We found that spleen extracts from WT mice are fully active in degrading the ssDNA substrate, whereas Trex1−/− spleen extracts show a complete lost of DNase activity (Fig. 1D), consistent with Trex1 being the most abundant exonuclease in animal cells and tissues [12,13]. Both V235fs and D272fs mouse spleen extracts are fully DNase active, similar to that of WT mouse (Fig. 1D and E). These data indicate that the two transgenic lines successfully express the correct TREX1 mutants and both proteins are DNase active in vivo.
Fig. 1.
V235fs and D272fs mutants are DNase active in mice. (A, B) Immunoblots showing expression of transgenic human TREX1 truncations and endogenous mouse Trex1. Actin serves as a loading control. Mice spleen of indicated genotype (top) were used for immunoblots. V235fs in A and D272fs in B. (C–E) DNase activity assay. Hela cells were transfected with plasmids expressing indicated V5-tagged TREX1 wild type or mutants (C). Spleen was isolated from V235fs mice (D) or D272fs mice (E). DNase activity of cell lysate was measured 24 h after transfection (C) or directly with spleen extract (D, E). Mock, mock transfected cells. rDNASE I, recombinant DNASE I (a positive control, NEB). Data are representative of at least three independent experiments.
2.2. V235fs and D272fs mice do not develop major retinal or neurological disease
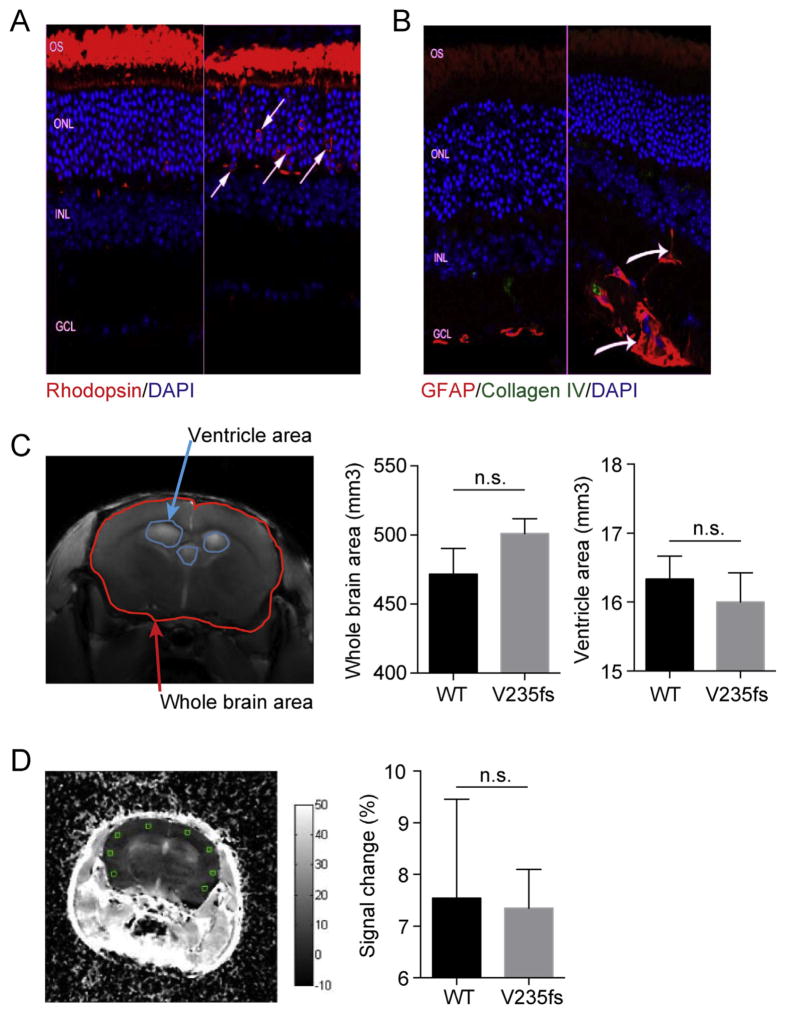
In humans, vascular retinopathy presenting as decreased acuity and field defects is often the earliest manifestation of RVCL. Visual changes are associated with hemorrhages, microvascular abnormalities and/or cotton wool spots. These were not identified by fluorescence angiography (data not shown). To identify areas of retinal pathology, cryosections were prepared, immunolabeled with antibodies to photoreceptor, glial and vascular markers and examined by confocal microscopy. Only rare areas suggestive of pathology were seen and then in only some of the retinas. Rhodopsin, a rod photoreceptor-specific protein, is concentrated in light-sensitive cylindrical organelles called outer segments (OS). With photoreceptor degeneration, outer segments often shorten and become disorganized, frequently accompanied by elevated rhodopsin immunoreactivity in photoreceptor cell bodies of the outer nuclear layer (ONL). Most samples from V235fs and D272fs mice showed normal OS morphology and a normal pattern of opsin immunolabeling. The thickness of the ONL, another metric of photoreceptor viability, was also normal in most samples analyzed. One out of eight V235fs mice had focal areas of photoreceptor disruption/degeneration (Fig. 2A). None of the 8 age-matched littermate no-Cre control mice demonstrated any detectable abnormalities.
Fig. 2.
V235fs and D272fs mice do not develop major retinal or neurological pathology. (A, B) Confocal microscopic images of retinas of two different V235fs mice, with areas of normal retina for each mouse shown on the left side of each panel and areas of suggestive of focal pathology on the right side. A, immunolabeling with antibody to Rhodopsin (red). Arrows denote the location of individual rods with elevated opsin immunolabeling. B, immunolabeling with antibodies to glial fibrillary acidic protein (GFAP, red) and collagen IV (green). (C) Magnetic Resonance Imaging (MRI) of the in vivo mouse brain. Image on the left shows a representative transverse image depicting the selection of Regions Of Interests (ROI) in the entire brain and the ventricles. Plots on the right show the total brain volume and ventricular areas, calculated over all MRI transverse slices, for WT (n = 3 [11, 13, 15 months old]) and V235fs homozygous mice (n = 8 [7–21 months old]). (D) A transverse image slice that illustrate the ROIs selected in the cortical region (total of 53 in the entire brain) in order to quantitatively evaluate possible breaching of the blood brain barrier (BBB). Relative signal intensity (calculated as {[pre contrast intensity – post contrast intensity]/pre contrast intensity}x100) upon administering an MR contrast agent, Gd DTPA, in WT and V235fs mice. ns, not significant. Student's t-test. Error bars, SEM.
Glial fibrillary acidic protein (GFAP) is expressed by glial cells. In the retina, GFAP is abundantly expressed by retinal astrocytes and at lower levels in a class of radial glia, known as Muller cells. Increased expression of GFAP in response to injury is a hallmark of reactive gliosis and an indicator of focal and widespread pathology. Most samples from V235fs and D272fs mice showed normal intensity and distribution of GFAP in both astrocytes and Muller glia. One V235fs mouse (a different mouse from the same cohort as above) showed evidence consistent with focal pathology (Fig. 2B). None of the 8 age-matched littermate no-Cre control mice show any detectable abnormalities.
We also performed neuropathologic examinations on brains of wild type and both mutant mice, including H&E staining and magnetic resonance imaging (MRI). No remarkable abnormalities were found by MRI (Fig. 2C and D) or histopathology. We conclude that mice expressing either of the two truncated human TREX1 proteins did not develop any of the major retinal or neurological findings regularly seen at autopsy in patients with RVCL.
2.3. V235fs and D272fs mice show similar transcriptome alterations
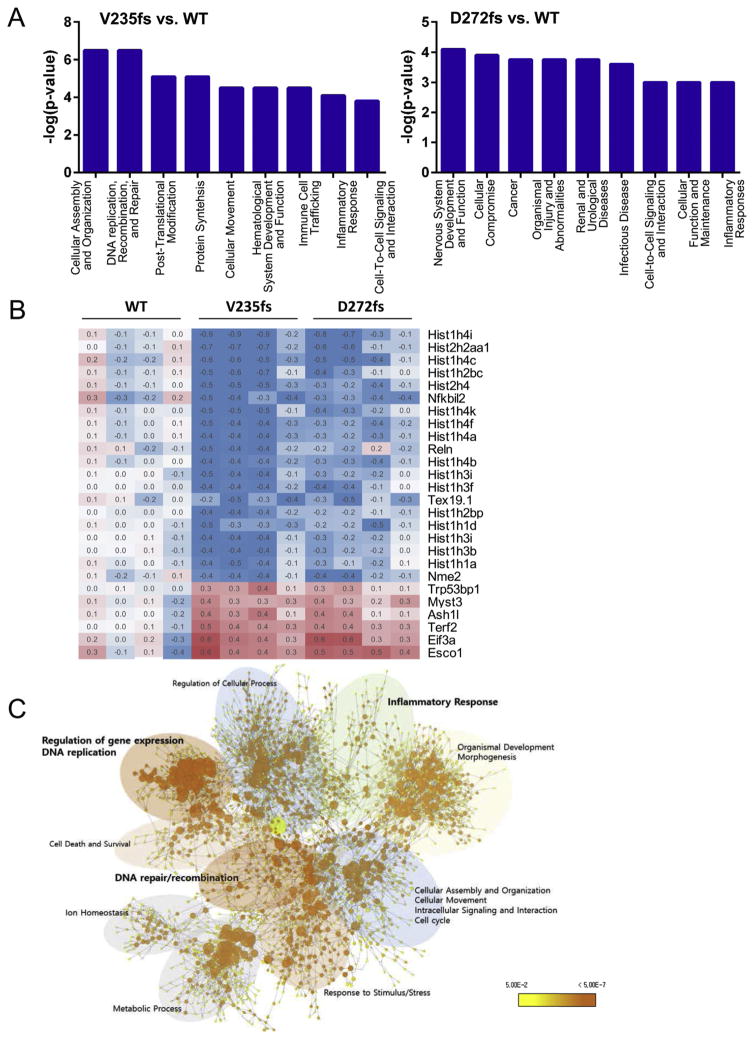
To develop insights into molecular alterations that might result from the V235fs and D272fs mutations, we used microarrays to evaluate similarities and differences in gene expression. The fact that TREX1 is normally expressed at the highest levels in monocytes and dendritic cells prompted us to study spleen cells, a rich source of both populations, from WT, V235fs and D272fs mice (all homozygous on C57BL/6 background). The results of these studies identified 990 genes that were differentially expressed in comparisons of samples from D272fs vs. WT, V235fs vs. WT and D272fs vs. V235fs mice. Fisher's exact test was performed to identify significantly enriched biological categories among the genes using Ingenuity Pathway Analysis (IPA). Both strains of mutant mice had enriched categories in common including cell-to-cell signaling and interaction and inflammatory responses (Fig. 3A). To better understand the features of genes that distinguish expression patterns in the mutant and WT mice, we compared those that were up or down-regulated in each of the enriched categories. As exemplified by the category of DNA replication, recombination and repair, the patterns of up and down regulated genes were strikingly similar for both mutant strains (Fig. 3B). This suggests that the V235fs and D272fs mutations, both of which cause truncations of the TREX1 C-terminus, have strikingly similar molecular effects that reflect common consequences of truncation rather than separate manifestations of SLE for D272fs mice and RVCL for V235fs mice. A more global perspective of the relative distributions of genes among the biologic categories for V235fs mice emphasized the concentrations of highly distinguishing genes in the categories of regulation of gene expression, DNA replication and metabolic processes (Fig. 3C). Future studies will be directed at expression profiling of purified microglia from retina and brain.
Fig. 3.
V235fs and D272fs mice show similar transcriptome alterations. (A) Top 9 significant biological categories modulated by V235fs (left panel) and D272fs (right panel). Total mRNA was isolated from mouse spleen and subjected to transcriptome analysis. Data analyzed by Ingenuity Pathway Analysis (IPA). (B) Differentially expressed genes in the category of DNA Replication, Recombination and Repair. (C) Functional network of gene sets of differentially expressed genes in V235fs mice. Each node represents a GO term, and its color and size indicate the p-value and the number of genes in the cluster, respectively. Microarray analyses were performed on spleens of 4 individual wild type mice or mice with either mutation.
2.4. V235fs and D272fs mice exhibit altered B cell subset populations
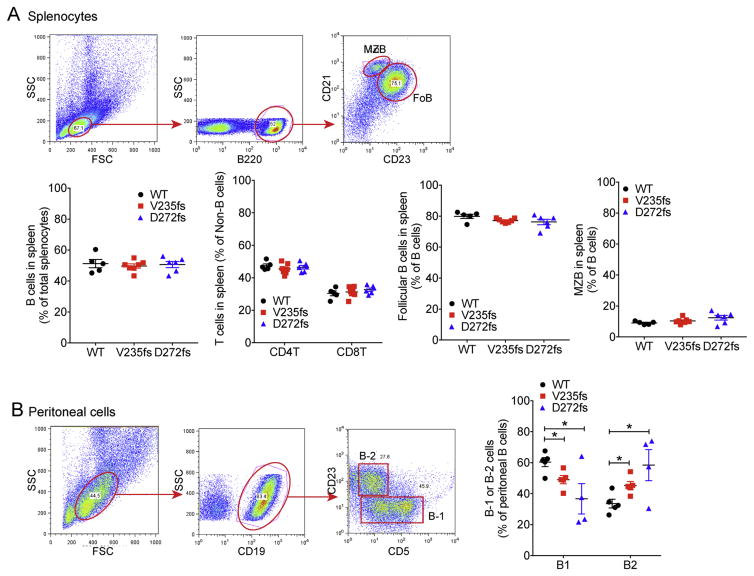
We next analyzed major and minor immune cell populations in wild type, V235fs and D272fs mice by FACS (Fig. 4A and B). Total B cells, follicular and marginal zone B cells in the spleen did not differ amongst all three genotypes nor did splenic CD4+ and CD8+ T cells (Fig. 4A). When we analyzed peritoneal B cells subsets, we found that the percentages of B-1 cells are significantly decreased in both mutant strains compared with WT mice, and the percentages of B-2 cells are significantly increased (Fig. 4B). These changes were more pronounced in spleens of D272fs mice.
Fig. 4.
V235fs and D272fs mice exhibit altered peritoneal B-1 and B-2 population. (A) FACS analysis of major B and T cell populations in the spleen. Top, gating strategy for follicle (FoB) and marginal zone (MZB) B cells. Gating strategy for CD4 and CD8 T cells is not shown. Bottom, quantitation of each cell population (as indicated) from WT, V235fs and D272fs mice. (B) FACS analysis of peritoneal B-1 and B-2 cells. Gating strategy on the left and quantitation on the right. *, p<0.05. Error bar, SEM. Data are representative of at least two independent experiments.
2.5. V235fs and D272fs mice produce increased levels of autoantibodies
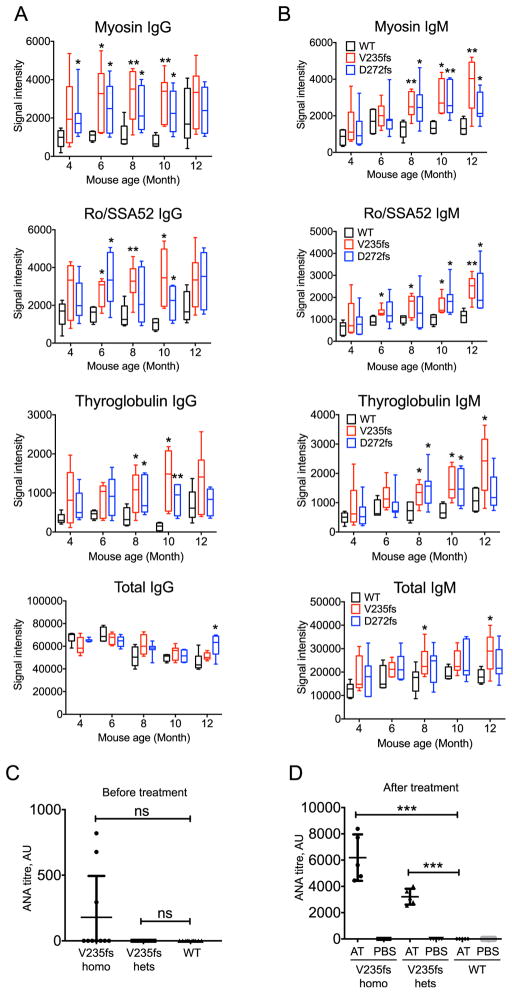
We showed previously that V235fs mice produce increased serum autoantibodies [11]. To determine in-depth the onset and kinetics of autoantibody production in V235fs and D272fs mice, we measured both IgG and IgM autoantibodies in mouse serum using a 128-autoantigen array. These antigens were selected based on published studies and have been proven to be targeted by auto-antibodies in various immune related-diseases (see Methods). We found that both V235fs and D272fs mice have significantly increased serum levels of many autoantibodies from as early as 6 months of age for some autoantigens (e.g. Myosin, Ro/SSA52) that remain significantly elevated compared to WT mice through 10 months of age for IgGs and up to 12 months of age for IgMs (Fig. 5A and B). We also observed a trend of age-dependent increases in IgM autoantibodies in both V235fs and D272fs mice compared to age-matched littermate WT controls.
Fig. 5.
V235fs and D272fs mice produce increased levels of broad range of autoantibodies. (A, B) Mouse serum autoantibody analysis. Representative autoantibodies from autoantibody array analysis are shown. IgG in A and IgM in B. n = 5. Statistical analysis was performed using multiple t-test comparing WT vs V235fs or WT vs D272 fs at each time point. (C, D) Mouse serum antinuclear antibody (ANA) titer before (C) and after (D) injection of apoptotic thymocytes (AT). n = 5. Statistical analysis was performed using t-test comparing WT vs V235fs or WT vs D272fs before treatment (C) and only AT-treated samples in D. *, p < 0.05. **, p < 0.01. ***, p < 0.001. ns, not significant. Error bar, SEM. Data are representative of at least two independent experiments.
We also examined whether we could enhance other features of RVCL-like disease in V235fs mice by stressing the immune system with exposure to autoantigens. To do this, we injected V235fs mice i.v. with dexamethasone-treated apoptotic thymocytes on twice weekly basis for 8 weeks beginning at 12 weeks of age. Serum antinuclear antibody (ANA) levels were quantified by ELISA before and at the end of treatment. We found that only a few homozygous V235fs mice had low levels of ANA before treatment while heterozygous V235fs and WT mice did not (Fig. 5C). By the end of treatment, both homozygous and heterozygous V235fs mice had substantially increased levels of ANA with the homozygous mutant mice having significantly higher levels than the heterozygotes (Fig. 5D). Sera from groups of control mice injected with PBS rather than thymocytes were all negative for ANA. Collectively, these data suggest that both TREX1 frame-shift mutant mice are predisposed to development of autoantibodies but that immune stress did not elicit classic manifestations of RVCL.
2.6. V235fs and D18N mice present distinct autoantibody profiles
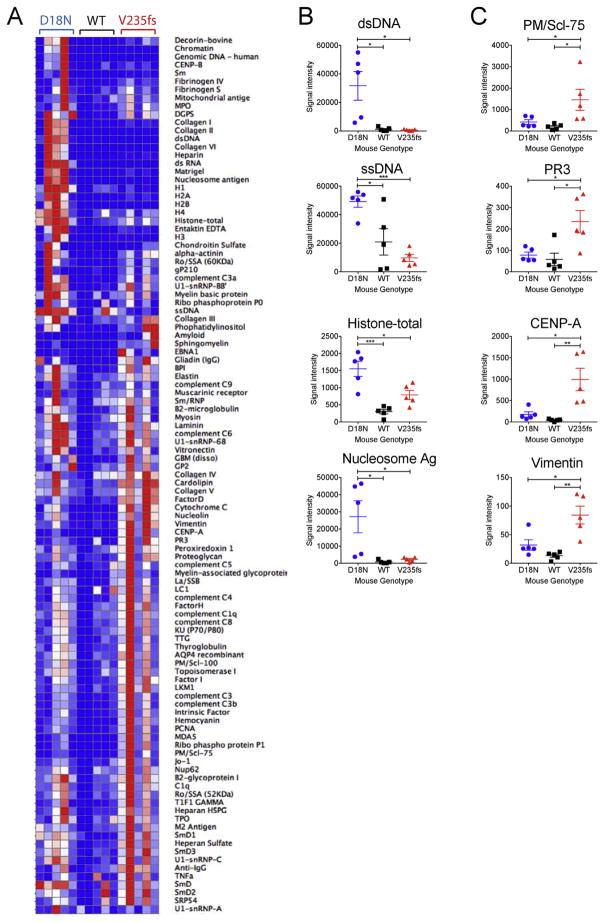
Two major functions of TREX1 are DNase activity associated with the N-terminus and ER localization/OST regulation associated with the C-terminus [11]. The D18N mutation disrupts DNase activity without affecting ER localization whereas V235fs mutation remains DNase active while dysregulating OST activity and glycan catabolism. We next asked whether disrupting either function of TREX1 in mice would lead to distinct autoantibody profiles. Both V235fs and D18N mice have increased levels of a broad range of autoantibodies but with distinct patterns (Fig. 6A and B). The D18N autoantibody profile shows strong associations with DNA-related autoantibodies, such as those against chromatin, dsDNA, nucleosomes and histones, consistent with the underlying defective DNase activity (Fig. 6A and B). In contrast, the V235fs autoantibody profile shows little or no elevation of DNA-related autoantibodies; Instead, a broader range of autoantibodies against non-nuclear antigens are found in these mice (Fig. 6A and B). These data strongly support the distinct molecular defects of the two mutations. It also raises the possibility of using the autoantibody profile as a biomarker for stratifying patients with TREX1 mutations.
Fig. 6.
D18N and V235fs mice exhibit distinct profiles of autoantibodies. (A) A heatmap of IgG autoantibody array. Mouse sera (n = 5, 6–8 month old) were analyzed by autoantibody array. Mouse genotypes are indicated on top. (B) Representative DNA-related autoantibody measurement from A. (C) Representative non-DNA-related autoantibody measurement from A. Statistical analysis was performed using t-test comparing V235fs vs D18N or V235fs vs WT. *, p < 0.05. **, p < 0.01. ***, p < 0.001. Error bar, SEM. Data are representative of at least two independent experiments.
2.7. Aclacinomycin treatment suppresses autoantibody production in V235fs and D272fs mice
Aclacinomycin A (Aclarubicin) (ACM) is an anthracycline drug. We recently showed that the molecular defect of TREX1 frame-shift mutant-associated autoimmune disease is dysregulated activity of OST, an endoplasmic reticulum (ER) enzyme that transfers oligo-saccharides to nascent proteins [11]. In that study we also showed that OST normally partners with TREX1 in the ER, that mutations involving the carboxyterminus of TREX1 enhance the activity of OST and that the drug ACM reduces free glycan and immune gene signatures of Trex1−/− mice and TREX1-V235fs patient cells. We next sought to determine if treatment of TREX1 frame-shift mice with ACM would alter another manifestation of the mutations, the expression of autoantibodies.
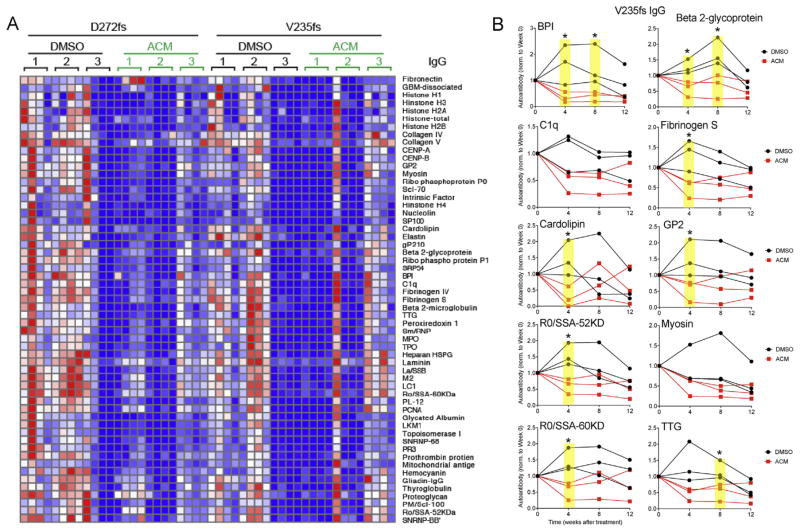
We treated 12-month old mice with DMSO or ACM for 12 weeks and measured autoantibody profiles every 4 weeks. DMSO-treated mutant mice produce gradually increasing amounts of autoantibodies during the 12-week course of treatment, similar to that of untreated mice (Fig. 7A and B). In contrast, ACM-treated mutant mice show decreased autoantibody production. We found significant differences in autoantibody levels between samples from DMSO- and ACM-treated mice in 7 out of 10 autoantibodies analyzed at the 4 week time point post treatment. Three out of 10 autoantibodies tested at the 8-week point showed statistical significance while none at 12-weeks were significantly different. This analysis suggests that ACM treatment is most effective soon after initiation of treatment, and that treatment efficacy diminishes at later stages due to reasons that are presently unclear. These data also imply that serologic autoimmunity can be a dynamic parameter of TREX1 frame-shift induced alterations in mice responsive to treatment.
Fig. 7.
Aclacinomyin treatment suppresses autoantibody production in V235fs and D272fs mice. (A) A heatmap of IgG autoantibody array. Mouse genotypes and treatment are indicated on top. Numbers indicate individual mouse (n = 3). Each mouse was analyzed at 0, 4, 8 and 12 wks after treatment (4 columns from left to right). (B) Ten representative IgG autoantibodies measured from A. Each line represents one mouse. Autoantibody values are normalized to week 0 value for each mouse to show fold-changes after treatment. Statistical analysis was performed using t-test comparing DMSO vs ACM for each time point. *, p < 0.05 (highlighted in yellow). Data are representative of at least two independent experiments.
3. Discussion
We established two mouse models of human diseases associated with C-terminal truncations of human TREX1 - V235fs knock-in mice for a model of human RVCL and D272fs knock-in mice for a model of human SLE. Previous studies of the V235fs mice showed that their cells shared several biochemical abnormalities with cells from RVCL patients, suggesting they might provide a suitable model for the disease. We present data here to show that both frame-shift mutant mice develop serologic autoimmunity, and that these mice had few if any of the retinal and cerebral manifestations characteristic of individuals with RVCL.
Using the 128-autoantigen array, we determined that the serum autoantibody profile of V235fs mouse is distinct from that of the D18N mouse; specifically, V235fs mice lack most DNA-related autoantibodies. This observation reinforces the notion that the V235fs mutation (and likely other TREX1 C-terminal mutations) affect a DNA-independent function of TREX1 [11]. The broad array of autoantibodies produced by V235fs mice against cytoplasmic antigens is uncommon for classical lupus. However, this unique observation is consistent with the known underlying molecular defect in OST activity in these frame-shift mice [11]. One possibility is that dysregulated OST could lead to under-glycosylation of cytoplasmic proteins. The exposed protein peptide could then be recognized by the immune system as foreign, thus stimulating autoantibody production. We also found significant changes in peritoneal B cell subsets, with decreased percentages of B-1 cells and increased percentages of B-2 cells. Major populations of T and B cells in the spleen remain unchanged. B-1 cells are considered innate-like immune cells that produce natural IgM and IgA encoded by germline sequences, where B-2 cells are continually generated from the bone marrow and can undergo class switch and somatic hypermutation. The altered frequencies of B-1 and B-2 cells could be due to stimuli that induce egress of B-1 cells from the peritoneal cavity and relocalization to spleen and other tissues where they could differentiate to plasma cells producing natural and autoantibodies [14]. This model is consistent with elevated levels of autoantibodies against specific antigens and not total IgGs or IgMs. Further investigation is needed to determine how the OST defect could contribute to altered peritoneal B-1 and B-2 populations and define which subset is responsible for the unique profile of autoantibodies observed in TREX1 frame-shift mice.
We found that the biochemical property, transcriptome and disease phenotypes of V235fs and D272fs mice on the common C57BL/6 background are generally indistinguishable. How then is it that these two mutants are associated with two different diseases in humans? One possibility is based on the 2-factor model: the TREX1 frame-shift mutation alone elevates autoantibody production and predisposition to disease. Depending on secondary genetic or environmental factors that are currently unknown, individuals with frame-shift mutations could then diverge into clinically distinct manifestations. This 2-factor model could potentially explain the identical phenotypes with both V235fs and D272fs mutations, and lack of disease pathology in mice. It is important to note that the D272fs mutation has been identified only once in patients with SLE. In view of the recent review of RVCL in which patients with fs mutations in this portion of TREX1 develop only RVCL [3]. The diagnosis in the one patient may need to be revisited.
Based on our findings that TREX1 frame-shift mutations induce autoantibodies against mostly non-nuclear antigens, patients carrying these mutations are likely be ANA negative [3]. Indeed, elevated levels of ANA are extremely rare among patients with RVCL. Thus, we predict that diseases related to these mutations maybe under- or mis-diagnosed. Future analyses of autoantibody profiles in serum from patients with TREX1 frame-shift mutations will be essential. Autoantibody production by V235fs mice was successfully suppressed by treatment with the OST inhibitor, aclacinomycin, mostly notably at 4 weeks, validating the potential therapeutic benefit of this drug for treating V235fs mutation-incurred immune disease phenotypes. Together, our results demonstrate that both the V235fs and D272fs mutations affect DNase-independent functions of TREX1 and induce serologic autoimmunity and possibly predisposition to autoimmune disease in mice. Although these mouse models do not reproduce the same clinical spectra seen with human RVCL, they do reveal important implications that will be useful for understanding the molecular function of TREX1 frame-shift mutations and pathogenesis of associated diseases.
4. Materials and methods
4.1. Mice
C57BL/6J (B6) V235fs conditional knock-in mice and the methods used in their generation were described previously [11]. The identical approach was used to knock-in a human coding exon bearing the D272fs mutation. Meox2-Cre or Vav1-Cre from Jackson Laboratory was used to generate whole body knock-in. D18N knock-in mice was described previously [9]. Experiments involving human and mouse materials were approved by the ICUAC and IRB of UT Southwestern Medical Center, the Wake Forest School of Medicine and NIAID (LIG-16E).
4.2. Treatment of mice with apoptotic thymocytes
Single cell suspensions, prepared from mouse thymi, were incubated in RPMI1640 supplemented with 1% BSA and 10 μM dexamethasone for 6 h at 37 °C under 5% CO2. After washing, 1 × 107 cells in 0.2 ml saline were injected i.v. twice weekly for 8 wks. Sera collected before treatment and 8 wks after treatment were tested for antinuclear antibody levels using an ANA ELISA kit (Alpha diagnostics International) according to the manufacturer's protocol.
4.3. Treatment of mice with aclacinomycin and autoantibody array analysis
Aclacinomycin (Santa Cruz Biotechnology) was injected i.p. using 1 ml syringe with 25G needle at 5 mg/kg in mice or with DMSO in PBS as a control twice weekly for 12 wks. We harvested sera before treatment and at 4, 8 and 12 wks after treatment. Sera were analyzed using an autoantigen array for the presence of autoantibodies.
The autoantibody array analysis was developed at UT Southwestern Medical Center Microarray Core. These 128 antigens were selected based on published studies and are targeted by autoantibodies in various immune related disease, such as Systemic Lupus Erythematosus (SLE), mixed connective tissue disease, Sjøgrens Syndrome, scleroderma, polymyositis, dermatomyositis, and rheumatoid arthritis, etc. These autoantigen arrays have been used and published by many investigators from different institutions [15–17]. Briefly, peptides of selected autoantigens were synthesized and immobilized in array format on glass microslides. Mouse serum was diluted 1:5 and incubated with the array. Then, secondary antibodies against IgG or IgM (with two different fluorophores) were used to determine the respective autoantibody levels.
4.4. Tissue exonuclease activity assay
TREX1 exonuclease activity was performed mostly as described in Ref. [11] with modifications. Briefly, one half of the spleen isolated from a mouse was homogenized to single cell suspension in PBS using a Gentle MACS Dissociator C-tube (Miltenyi Biotec). Cells were lysed in 50 μl of DNase lysis buffer (20 mM Tris-HCl [pH 7.4], 5 mM MgCl2, 2 mM DTT, 100 mg/ml BSA, 0.5% NP-40 and 1x protease inhibitor) and centrifuged at 20,000 × g for 20 min at 4 °C. The supernatant protein concentration was determined using Pierce BCA protein Assay (Thermo) and samples of 10 μl containing 50 μg of total protein were prepared. Each sample was treated with 100U of RNAse 1 (Ambion) for 30 min at 37 °C. Each sample was then mixed with 90 μl of pre-warmed to 37 °C DNase reaction buffer (20 mM Tris-HCl [pH 7.4], 5 mM MgCl2, 2 mM DTT, 100 mg/ml BSA, 1/1200 SYBR Green, and 10 ng/ml ssDNA) followed by real-time quantification of the DNA/SYBR complex using a SynergyHT microplate reader (Biotek).
4.5. Western blotting
Proteins were extracted from mouse spleen or cultured cells using NP-40 cell lysis buffer (Invitrogen) with 1x protease inhibitor (Roche). Protein concentration of each sample was measured by bicinchoninic assay (BCA) and equal amounts of protein were loaded onto 4–12% gradient Bis-Tris gels (NuPAGE, Invitrogen) and separated. Proteins were then transferred to a polyvinylidene difluoride membrane (PVDF, Invitrogen). Membranes were blocked with 5% non-fat dry milk powder in 1x PBS with 0.1% Tween-20 and incubated overnight at 4 °C in primary antibody against hTREX1 (diluted 1:1000, Washington University, or diluted 1:500, Novus NBP1-89202), mTREX1 (diluted 1:2000, BD BioSciences Cat#611987), Actin (diluted 1:2000, Sigma Aldrich A2066), washed and incubated with HRP-conjugated secondary antibody (Santa Cruz) for 1 h at room temperature. Specific protein signals were detected using ECL reagents (ThermoScientific) and autoradiography films (Denville).
4.6. Neuropathology and magnetic resonance imaging
Brains collected at necropsy were fixed in neutral buffered formalin. Formalin fixed paraffin embedded sections of brains stained with H&E were examined microscopically for histologic features. MRI experiments were performed on a 9.4-T, 30 cm horizontal Bruker Avance scanner. The mice were positioned in a stereotaxic holder and anesthetized with 1.5% isofluorane. The body core temperature was maintained at 37 °C using circulating water and monitored by means of a rectal temperature probe. A line through the tail vein was placed for SPIO infusion. The mouse head was centered in an 86 mm transmit 4 element array mouse head receive coil ensemble. Three mutually perpendicular scout images were acquired. The scout images were used to acquire 14 axial slices of T2 weighted (Repetition Time [TR]/Echo Time [TE] = 2500/12 ms, in-plane resolution = 79 × 76 μm, slice thickness = 1 mm, number of averages = 8, echo number = 8), fast spin echo and T1-weighted (TR/TE = 300/4 ms, in-plane resolution = 79 × 76 μm, slice thickness = 1 mm, number of averages = 10). Gradient echo images encompassing the whole brain. T1-weighted scans were repeated 5 min after administration of a bolus of Gd-DTPA (0.2 cm3/kg body weight). Post Gd signal variation (expressed as relative contrast − [{Pre Gd- post Gd}/pre Gd] x 100) for a series of selected anatomical areas, and temporal variation of signal in dynamic contrast images (Fig. 2D) were calculated using MATLAB routines (Mathworks Inc., Natick, MA). In order to assess the relative breaching of the BBB, multiple sites in the brain was evaluated with regions of interest (ROI) placed in the cortices of both hemispheres.
4.7. Retinal histology and immunohistochemistry
Mice were sacrificed by asphyxiation with CO2 under normal room illumination. Following enucleation, eyes were immersed for 10 min in 4% paraformaldehyde in 1x phosphate-buffered saline (PBS) pH 7.3. The cornea of each eye was incised with a scalpel to facilitate penetration of fixative. Eyes were returned to fixative for 4 h at 4 °C. The anterior segment and lens were dissected and eyecups washed in 1x PBS then cryoprotected at 4 °C in PBS containing increasing sucrose concentrations (10%–30%). Eyes were embedded in OCT (Sakura) and 8 μm frozen sections collected on charged slides (SuperFrost Plus, Fisher). Sections were dried under house vacuum in a desiccator for 40 min prior to immunolabeling. A hydrophobic barrier (ImmunEdge, Vector Labs) encircled each section. Sections were blocked for 20 min in ICC buffer (0.1M PBS, 0.5% BSA, 0.2% Tween-20, 0.05% sodium azide, pH 7.3) containing 5% normal serum. Sections were labeled with primary antibodies to rhodopsin (mab5356, Millipore), glial fibrillary acidic protein (Z0334, Dako) or collagen IV (Col94, Abcam) overnight at 4 °C. Sections were washed in ICC buffer, incubated in fluorescently-conjugated secondary antibodies for 1 h at RT. DAPI (1 ug/ml) was included to label nuclei. Following repeated washing in ICC buffer, sections were mounted in antifade compound (Fluoro-Gel, EM Sciences) and coverslipped. Primary antibodies were omitted from negative controls. Sections were imaged on a LSM780 confocal microscope (Carl Zeiss Microscopy LLC, Thornwood, NY).
4.8. Gene expression profiling
Total RNA prepared from spleen cells of four female WT and V235fs and D272fs mice were applied to Agilent (Santa Clara, CA) National Institute of Allergy and Infectious Disease customized mouse gene expression arrays with scanned images analyzed as detailed previously [18]. Raw data were normalized with LIMMA package software version 2.9.17 in R software version 2.4.1 (http://www.r-project.org, last accessed September 13, 2013). Differentially expressed genes were identified with one-way analysis of variance analysis with 5% false-discovery rate. The microarray data are deposited in National Center for Biotechnology Information's Gene Expression Omnibus (http://www.ncbi.nlm.nih.gov/geo; accession number GSE). Differentially expressed genes were categorized based upon their biological functions and the statistical significance for the enrichment was examined by Fisher's exact test. The biological pathways and the interaction networks were integrated and analyzed by the Cytoscape software (www.cytoscape.org).
Acknowledgments
We thank members of Yan and Morse laboratories for helpful discussions. This work is supported by the NIH (AR067135 to N.Y., AI116725 to F.W.P.), Alliance for Lupus Foundation (N.Y.), the Robert G. Clark Family and Clayco Corporation, and in part by the Intramural Research Program of the NIH, NIAID. Private donations from Cure CRV Research, Energy 4A Cure Foundation, the Robert G. Clark Family and Clayco Corporation (P.H.K., J.P.A.) and NIH/NHLB1 HL083822 (PHK). None of the funders had a role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript.
References
- 1.Crow YJ, Manel N. Aicardi-Goutières syndrome and the type I interferonopathies. Nat Rev Immunol. 2015;15:429–440. doi: 10.1038/nri3850. http://dx.doi.org/10.1038/nri3850. [DOI] [PubMed] [Google Scholar]
- 2.Richards A, van den Maagdenberg AMJM, Jen JC, Kavanagh D, Bertram P, Spitzer D, et al. C-terminal truncations in human 3′-5′ DNA exonuclease TREX1 cause autosomal dominant retinal vasculopathy with cerebral leukodystrophy. Nat Genet. 2007;39:1068–1070. doi: 10.1038/ng2082. http://dx.doi.org/10.1038/ng2082. [DOI] [PubMed] [Google Scholar]
- 3.Stam AH, Kothari PH, Shaikh A, Gschwendter A, Jen JC, Hodgkinson S, et al. Retinal vasculopathy with cerebral leukoencephalopathy and systemic manifestations. Brain. 2016:aww217. doi: 10.1093/brain/aww217. http://dx.doi.org/10.1093/brain/aww217. [DOI] [PMC free article] [PubMed]
- 4.Morita M, Stamp G, Robins P, Dulic A, Rosewell I, Hrivnak G, et al. Gene-targeted mice lacking the Trex1 (DNase III) 3′-5′ DNA exonuclease develop inflammatory myocarditis. Mol Cell Biol. 2004;24:6719–6727. doi: 10.1128/MCB.24.15.6719-6727.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stetson DB, Ko JS, Heidmann T, Medzhitov R. Trex1 prevents cell-intrinsic initiation of autoimmunity. Cell. 2008;134:587–598. doi: 10.1016/j.cell.2008.06.032. http://dx.doi.org/10.1016/j.cell.2008.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gall A, Treuting P, Elkon KB, Loo YM, Gale M, Barber GN, et al. Auto-immunity initiates in nonhematopoietic cells and progresses via lymphocytes in an interferon-dependent autoimmune disease. Immunity. 2012;36:120–131. doi: 10.1016/j.immuni.2011.11.018. http://dx.doi.org/10.1016/j.immuni.2011.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang Y, Lindahl T, Barnes DE. Trex1 exonuclease degrades ssDNA to prevent chronic checkpoint activation and autoimmune disease. Cell. 2007;131:873–886. doi: 10.1016/j.cell.2007.10.017. [DOI] [PubMed] [Google Scholar]
- 8.Hasan M, Koch J, Rakheja D, Pattnaik AK, Brugarolas J, Dozmorov I, et al. Trex1 regulates lysosomal biogenesis and interferon-independent activation of antiviral genes. Nat Immunol. 2013;14:61–71. doi: 10.1038/ni.2475. http://dx.doi.org/10.1038/ni.2475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Grieves JL, Fye JM, Harvey S, Grayson JM, Hollis T, Perrino FW. Exonuclease TREX1 degrades double-stranded DNA to prevent spontaneous lupus-like inflammatory disease. Proc Natl Acad Sci U S A. 2015 doi: 10.1073/pnas.1423804112. http://dx.doi.org/10.1073/pnas.1423804112. [DOI] [PMC free article] [PubMed]
- 10.Lee-Kirsch MA, Gong M, Chowdhury D, Senenko L, Engel K, Lee YA, et al. Mutations in the gene encoding the 3′-5′ DNA exonuclease TREX1 are associated with systemic lupus erythematosus. Nat Genet. 2007;39:1065–1067. doi: 10.1038/ng2091. http://dx.doi.org/10.1038/ng2091. [DOI] [PubMed] [Google Scholar]
- 11.Hasan M, Fermaintt CS, Gao N, Sakai T, Miyazaki T, Jiang S, et al. Cytosolic nuclease TREX1 regulates oligosaccharyltransferase activity independent of nuclease activity to suppress immune activation. Immunity. 2015;43:1–13. doi: 10.1016/j.immuni.2015.07.022. http://dx.doi.org/10.1016/j.immuni.2015.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Robins P, Naven TJ, Pappin DJ, Lindahl T. A human DNA editing enzyme homologous to the Escherichia coli DnaQ/MutD protein. EMBO J. 1999;18:3868–3875. doi: 10.1093/emboj/18.13.3868. http://dx.doi.org/10.1093/emboj/18.13.3868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mazur DJ, Perrino FW. Identification and expression of the TREX1 and TREX2 cDNA sequences encoding mammalian 3′-5′ exonucleases. J Biol Chem. 1999;274:19655–19660. doi: 10.1074/jbc.274.28.19655. [DOI] [PubMed] [Google Scholar]
- 14.Ha SA, Tsuji M, Suzuki K, Meek B, Yasuda N, Kaisho T, et al. Regulation of B1 cell migration by signals through Toll-like receptors. J Exp Med. 2006;203:2541–2550. doi: 10.1084/jem.20061041. http://dx.doi.org/10.1084/jem.20061041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hasan M, Dobbs N, Khan S, White MA, Wakeland EK, Li QZ, et al. Cutting edge: inhibiting TBK1 by compound II ameliorates autoimmune disease in mice. J Immunol. 2015 doi: 10.4049/jimmunol.1500162. http://dx.doi.org/10.4049/jimmunol.1500162. [DOI] [PMC free article] [PubMed]
- 16.Baum R, Sharma S, Carpenter S, Li QZ, Busto P, Fitzgerald KA, et al. Cutting edge: AIM2 and endosomal TLRs differentially regulate arthritis and autoantibody production in DNase II-deficient mice. J Immunol. 2015;194:873–877. doi: 10.4049/jimmunol.1402573. http://dx.doi.org/10.4049/jimmunol.1402573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Subramanian S, Tus K, Li QZ, Wang A, Tian XH, Zhou J, et al. A Tlr7 translocation accelerates systemic autoimmunity in murine lupus. Proc Natl Acad Sci U S A. 2006;103:9970–9975. doi: 10.1073/pnas.0603912103. http://dx.doi.org/10.1073/pnas.0603912103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shin DM, Shaffer DJ, Wang H, Roopenian DC, Morse HC. NOTCH is part of the transcriptional network regulating cell growth and survival in mouse plasmacytomas. Cancer Res. 2008;68:9202–9211. doi: 10.1158/0008-5472.CAN-07-6555. http://dx.doi.org/10.1158/0008-5472.CAN-07-6555. [DOI] [PMC free article] [PubMed] [Google Scholar]