Abstract
Purpose
Patients with glioblastoma (GBM) have a <15 month median survival despite surgical resection, high-dose radiation and chemotherapy with temozolomide (TMZ). We previously demonstrated that targeting Cytomegalovirus (CMV) pp65 using dendritic cells (DCs) can extend survival and, in a separate study, that dose-intensified (DI) TMZ and adjuvant GM-CSF potentiates tumor-specific immune responses in patients with GBM. Here, we evaluated pp65-specific cellular responses following DI-TMZ with pp65-DCs and determined the effects on long-term progression-free survival (PFS) and overall survival (OS).
Experimental Design
Following standard of care, 11 patients with newly diagnosed GBM received DI-TMZ (100 mg/m2/day × 21 days per cycle) with at least three vaccines of pp65-lysosome-associated membrane glycoprotein (LAMP) mRNA-pulsed DCs admixed with GM-CSF on Day 23 ± 1 of each cycle. Thereafter, monthly DI-TMZ cycles and pp65-DCs were continued if patients had not progressed.
Results
Following DI-TMZ cycle 1 and three doses of pp65-DCs, pp65 cellular responses significantly increased. After DI-TMZ, both the proportion and proliferation of regulatory T-cells (TRegs) increased and remained elevated with serial DI-TMZ cycles. Median PFS and OS were 25.3 months (CI95: 11.0-∞) and 41.1 months (CI95: 21.6-∞), exceeding survival using recursive partitioning analysis and matched historical controls. Four patients remained progression-free at 59 to 64 months from diagnosis. No known prognostic factors (age, KPS, IDH-1/2 mutation, and MGMT promoter methylation) predicted more favorable outcomes for the patients in this cohort.
Conclusions
Despite increased TReg proportions following DI-TMZ, patients receiving pp65-DCs showed long-term PFS and OS, confirming prior studies targeting CMV in GBM.
Keywords: cytomegalovirus, dose-intensified temozolomide, glioblastoma, pp65
Introduction
Patients with newly diagnosed glioblastoma (GBM) have a median survival of <15 months despite maximal tumor resection, high-dose radiation and temozolomide (TMZ) chemotherapy (1, 2). Novel approaches to therapy are desperately needed. Several groups, including our own laboratory, have demonstrated that human Cytomegalovirus (CMV) proteins are expressed in over 90% of GBMs (3–5). CMV expression has not been detected in surrounding normal brain tissue (3, 4, 6, 7), which provides an unparalleled opportunity to subvert CMV antigens as tumor-specific targets. Recent evidence has also demonstrated that CMV-specific T cell immunity can be generated to recognize and effectively kill autologous GBM tumor cells expressing endogenous levels of the immunodominant pp65 antigen (8), providing compelling support for the development of CMV-directed immunotherapy for the treatment of GBM.
We have recently demonstrated, in a small randomized pilot trial, that patients who received CMV pp65-specific dendritic cells (pp65-DCs) combined with vaccine site pre-conditioning using tetanus-diphtheria toxoid showed significantly improved progression-free survival (PFS) (range 15.4 – 47.3 months) and overall survival (OS) (range 20.6 – 47.3 months) compared to controls (9). In addition, in related trials we have also demonstrated that dose-intensified (DI) temozolomide (TMZ) and adjuvant GM-CSF can enhance immune responses to tumor-specific antigens in patients with GBM (10).
In this phase I trial, our primary objective was to evaluate the safety and feasibility of vaccinating newly diagnosed patients with pp65-DCs admixed with GM-CSF following host conditioning with DI-TMZ. Secondary objectives were constructed to investigate patient cellular immune responses induced by pp65-DCs admixed with GM-CSF and to determine progression-free survival (PFS) and overall survival (OS) compared to that expected with standard of care. GM-CSF was chosen as an adjuvant to pp65-DCs based on our own experience with GM-CSF-containing DCs (VICTORI trial) (11) and peptide vaccines (ACTIVATE and ACT II trials) (10, 12) and its previously characterized effects on DC viability and differentiation (13). We chose to administer DI-TMZ in this study also based on our prior experience that profound lymphopenia following DI-TMZ can be leveraged to foster de novo expansion of vaccine-induced antigen-specific immune responses through reactive homeostatic proliferation (14, 15). Here, we demonstrate that despite profound lymphopenia and increased TReg proportions following DI-TMZ, patients with GBM receiving pp65-DCs showed expansion of antigen-specific immunity and long-term PFS and OS, confirming earlier studies targeting CMV in newly diagnosed GBM.
Materials and Methods
Patient Selection
Patients were enrolled and treated with the study drug in a separate clinical study under an overarching parent protocol. The clinical protocol and informed consent were approved by the U.S. Food and Drug Administration and Institutional Review Board (IRB) at Duke University for this study (FDA-IND-BB-12839, Duke IRB Pro00003877, NCT00639639). Eligibility criteria included adults with a histologically-confirmed, newly diagnosed WHO Grade IV GBM. Patients were eligible if they underwent a gross total resection defined as > 90% with residual contrast enhancement of < 1 cm2 on post-resection magnetic resonance imaging (MRI), had a baseline Karnofsky Performance Status (KPS) score of ≥ 80, did not require continuous steroid therapy above physiologic levels, and did not receive additional treatments aside from the study therapy. Histopathology of all specimens was initially read as GBM, and diagnosis was re-confirmed by a second board-certified neuro-pathologist. Both methyl-guanine methyltransferase (MGMT) promoter methylation and isocitrate dehydrogenase (IDH)-1 and IDH-2 mutation analyses were performed by PCR (16, 17). Given published reports showing high expression of CMV viral proteins in > 90% of sampled primary GBM specimens (3, 4, 6, 7), we elected not to include pp65 staining of tumor tissue as an eligibility criterion for patients enrolled on this trial.
Historical Controls
A cohort of historical controls at least double the sample size of our study cohort was used to compare survival rates of patients receiving our study drug with similar patients receiving other therapies. Historical controls had histopathology-confirmed primary GBM, with 23/23 negative for the IDH-1 mutation and mixed MGMT methylator phenotype (12 negative, 5 positive, 6 not available). Historical controls were treated similarly to study patients with initial gross total resection and standard six week XRT/TMZ. If no progressive disease occurred at this point, historical controls proceeded to receive monthly standard TMZ cycles (150–200 mg/m2/d × 5 days) and did not receive any other therapies until progression. Those controls who did progress after the completion of standard therapy were offered additional therapies, including bevacizumab, etoposide, irinotecan, CCNU, vorinostat, and heat-shock protein targeted therapies.
Study Design
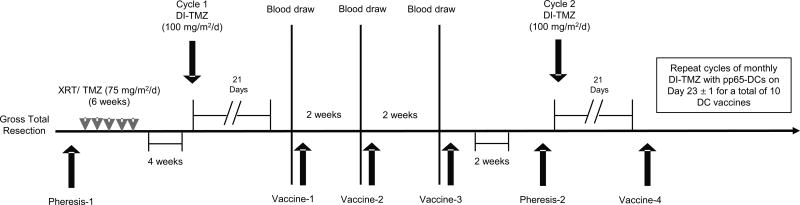
Eligible patients underwent initial leukapheresis (Pheresis-1) prior to XRT/TMZ for immunologic monitoring and subsequent ex vivo differentiation of autologous DCs. Each patient then completed a six week course of conformal external beam radiation therapy (XRT) to a dose of 60 Gray (Gy) with concurrent TMZ at a targeted daily dose of 75 mg/m2/d. Upon completion of standard chemoradiation therapy, all patients were re-imaged with MRI for evidence of progressive disease. Those with evidence of progressive disease or requiring steroid therapy in excess of physiological levels (> 2 mg/day of dexamethasone) at the time vaccination was scheduled did not continue on study. At four weeks following standard XRT/TMZ, the first DI-TMZ cycle (100 mg/m2/day) was administered over the course of 21 days of a 28-day cycle, and DC Vaccine-1 was administered on Day 23 ± 1 of that 28-day cycle. For each pp65-DC vaccine, an intradermal injection of 2 × 107 pp65 mRNA-pulsed DCs admixed with 150 µg GM-CSF in 0.4 mL of saline was administered bilaterally in the groin. Following DC Vaccine-3, patients underwent a second leukapheresis (Pheresis-2) for immunologic monitoring and subsequent ex vivo differentiation of autologous DCs. From DC Vaccine-3 and onward, patients received monthly DC vaccines in conjunction with subsequent DI-TMZ cycles every 5 ± 1 weeks for a total of 6 to 12 cycles at the discretion of the treating neuro-oncologist. DCs were given on Day 23 ± 1 of each 28-day TMZ cycle for a total of 10 vaccines unless progression occurred. Patients were imaged bi-monthly and did not receive any other prescribed anti-tumor therapy.
Safety and Adverse Events
All patients were monitored for treatment-related toxicity. Adverse events (AEs) and serious adverse events (SAEs) were graded according to the National Cancer Institute's Common Terminology Criteria for AEs (Version 3.0). Safety checkpoints were defined to halt the study if any two patients experienced a drug-related Grade IV or irreversible Grade III toxicity.
Dendritic Cell Vaccine Generation
Autologous DCs were generated using the method of Romani et al. (18, 19). After harvest, the cells were frozen and assessed for contamination and lineage purity as previously published (20). The 1.932 kB pp65 full-length cDNA insert was obtained from Dr. Bill Britt (University of Alabama-Birmingham, Birmingham, Alabama), and RNA was generated and transfected as previously reported (9).
Peripheral Blood Mononuclear Cell Processing
In addition to leukaphereses, blood draws for pp65 ELISpot assays were performed just prior to vaccination with pp65-DCs. Peripheral blood mononuclear cells (PBMCs) were separated from blood collected in ACD tubes within 4 hours of blood collection using Histopaque (Sigma) and stored in liquid nitrogen. On the day of testing, patient PMBCs were rapidly thawed, washed, rested overnight in RPMI-1640 with 10% FBS and additives (21) and processed for absolute cell count and viability on a Guava easyCyte flow cytometer (Merck KGaA, Darmstadt, Germany).
Gamma Interferon Enzyme-Linked ImmunoSpot
Patient pp65 responses were measured ex vivo by direct IFN-γ Enzyme-Linked ImmunoSpot (ELISpot). PBMCs were stimulated overnight with a pool of synthetic peptides spanning CMV pp65 (15-mers overlapping by 11 amino acids with > 95% purity), kindly provided by Dr. Robert A. Olmsted (Alphavax, Inc., Research Triangle Park, NC). ELISpot plates coated with mouse IgG1 anti-human IFN-γ monoclonal antibody were incubated overnight at 37°C, 5% CO2, washed with PBS/Tween-20, incubated with biotinylated mouse IgG1 anti-human IFN-γ for 1 hour at room temperature, washed with PBS, incubated with avidin-peroxidase complex for 1 hour at room temperature, washed, and incubated with substrate (3-amino-9-ethylcarbazole) for 4 minutes at room temperature. Spot enumeration was performed in a blinded fashion by Zellnet Consulting, Inc. (Fort Lee, NJ) using a KS ELISpot reader (Zeiss Inc., Thornwood, NY), software version KS 4.9.16 using established guidelines (22). Results were expressed as the mean spot-forming cells (SFC)/106 PBMC after subtraction of background counts from PBMCs cultured without peptide. Negative values were raised to zero for mean calculations. Positive and negative control PBMCs for pp65 (from Dr. Robert A. Olmsted) were qualified and validated by precision and intermediate precision testing using pp65 standardized peptide pools. The criteria for a valid assay required control wells with CMV pp65 positive control > 692 SFC/106 PBMC and negative control < 9 SFC/106 PBMC.
TReg Analysis
PBMC surface antigens were stained with CD4-FITC (RPA-T4), CD25-APC (MA251), Ki67-PE (B56), and CD127-PE (hIL-7R-M21) (BD Bioscience, San Diego, CA). After washing to remove unbound antibody, cells were incubated on ice for 30 minutes in fixation/permeabilization buffer (eBioscience, San Diego, CA). Cells were washed again with 1X permeabilization buffer (eBioscience), pelleted, and stained with FOXP3-APC (PCH101, eBioscience). Samples were acquired on BD FACS Calibur (BD, San Diego, CA) and analyzed with FlowJo (TreeStar, Ashland, OR). TRegs were defined as CD25+FOXP3+ of CD4+ lymphocytes.
Disease Progression
Progressive disease was defined radiographically according to the RANO criteria and defined as 1) at least a 25% increase in the longest diameter on an axial image of any enhancing tumor on consecutive CT or MRI images or 2) the appearance of a new radiographically demonstrable lesion measuring ≥ 1cm in any two perpendicular axial planes (23). Upon tumor progression, patients could undergo stereotactic biopsy or resection for confirmation with additional consent.
Statistical Analysis
For sample size estimation, patients in this Phase I study were recruited in a single arm fashion to evaluate safety and feasibility as the primary endpoint. No a priori power calculations were used for secondary endpoints. Therefore, a final sample size matching that of the prior study (9) was calculated to ensure at least six evaluable subjects. For paired comparisons between two time points and for fold change comparisons, a Wilcoxon signed rank test was used to determine statistical significance. Patients were enrolled over the course of two years, and a lock date 4.25 years after the last patient enrolled was applied for survival analysis of censored data (patients who had not progressed and were alive at the time of analysis). Progression-free survival (PFS) was defined as the time from histologic diagnosis at surgery until radiographic or clinical progression and was censored at the lock date if the patient remained alive without disease progression at the time of analysis. Overall survival (OS) was defined as the time from histologic diagnosis until death and was censored at the lock date if the patient remained alive at the time of analysis. Median PFS and OS were estimated using Kaplan-Meier methods, and comparisons with historical controls used the log-rank test. Predicted survival utilized known prognostic factors under the RPA classification (24), with observed-expected survival calculated for each patient.
Results
Patient Population
A total of 14 patients were initially enrolled in the study (schema shown in Fig 1). The study therapy was defined as completion of DI-TMZ cycle 1 and DC Vaccines 1 to 3 without additionally prescribed therapies. Two patients were excluded from immune response analyses as they were not eligible for study participation due to treatment with bevacizumab at outside hospitals. One patient discontinued protocol treatment prior to DC Vaccine-3 due to disease progression. Primary immune response analysis is based on the 11 patients who received at least 3 pp65-DC vaccinations.
Figure 1.
Schema of ATTAC-GM trial. Following standard of care with gross total resection (> 90%), external beam radiation (RT) and temozolomide (TMZ), patients received DI-TMZ cycle 1 (100 mg/m2/d) for 21 days of a 28-day cycle. DC vaccines consisted of 2×107 mature pp65- lysosome-associated membrane glycoprotein (LAMP) mRNA-pulsed DCs (pp65-DCs) admixed with 150 µg GM-CSF. Vaccination with pp65-DCs occurred on Day 23 ± 1 of the 28-day cycle with the first three DC vaccines administered two weeks apart. Following DI-TMZ cycle 2 and DC Vaccine-4, patients then received monthly DC vaccines administered on DI-TMZ cycle Day 23 ± 1 for a total of 10 vaccines in conjunction with monthly DI-TMZ cycles for a total of 6 to 12 cycles unless progression occurred. Patients were imaged bi-monthly without receiving any other prescribed anti-tumor therapy. For immune monitoring of pp65 responses, peripheral blood mononuclear cells were sampled at Pheresis-1 and Pheresis-2, along with blood draws just prior to vaccination with pp65-DCs.
Patients on study had a median age of 55 and median KPS of 90 at diagnosis (Table 1).To account for the possibility that certain prognostic factors could have selected for more favorable patient outcomes, we analyzed primary GBM specimens for IDH-1 and IDH-2 mutations (16) and promoter methylation of MGMT (17). All patient specimens that could be sampled (10/11) were negative for IDH mutations. No significant differences in survival were seen in patients with MGMT promoter methylation (5/11), although this study was not powered to assess this variable as a prognostic marker for survival. Recursive partitioning analysis (RPA) class incorporating KPS and age at diagnosis was used to evaluate expected survival for each patient. Observed-expected survival rates based upon the RPA showed a gain in survival for each of the 11 patients receiving at least three vaccines, with a median gain of 30 months for the entire cohort (Table 1).
Table 1. Clinical trial patient characteristics.
Demographic and prognostics factors for patients with newly-diagnosed GBM and corresponding PFS and OS from the time of surgery (histologic diagnosis). Patients shown were those able to complete the predefined study therapy (completion of DI-TMZ cycle 1 and DC Vaccines 1 to 3). Observed and predicted survival times are expressed in months. RPA O-E: The difference between the observed survival and the survival predicted based upon the Curran et al. recursive partition analysis (RPA). For Class III, the predicted median survival is 17.9 months, whereas for Class IV, the predicted median survival is 11.1 months.
| Patient | Sex | Age | Race | KPS | WHO score |
IDH-1/2 mutation |
MGMT promoter methylation |
PFS (diagnosis) |
OS (diagnosis) |
RPA Predicted Class |
RPA O-E |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | M | 55 | W | 90 | 1 | - | - | 10.1 | 21.7 | IV | 10.6 |
| 2 | M | 55 | W | 80 | 1 | - | - | 64.0a | 64.0b | IV | 52.9 |
| 3 | M | 53 | W | 90 | 1 | N/A | - | 61.9a | 61.9b | IV | 50.8 |
| 4 | M | 47 | H | 100 | 0 | - | + | 60.7a | 60.7b | III | 42.8 |
| 5 | F | 60 | W | 100 | 0 | - | + | 59.0a | 59.0b | IV | 47.9 |
| 6 | F | 55 | W | 90 | 1 | - | - | 20.0 | 33.4 | IV | 22.3 |
| 7 | M | 67 | W | 90 | 1 | - | + | 25.3 | 46.5 | IV | 35.4 |
| 8 | F | 63 | W | 80 | 1 | - | + | 11.6 | 24.2 | IV | 13.1 |
| 9 | M | 57 | W | 90 | 1 | - | + | 29.2 | 41.1 | IV | 30 |
| 10 | M | 55 | W | 80 | 1 | - | - | 14.3 | 21.6 | IV | 10.5 |
| 11 | M | 59 | W | 80 | 1 | - | - | 11.0 | 19.7 | IV | 8.6 |
|
| |||||||||||
| Median | 55 | 90 | 1 | 25.3 | 41.1 | IV | 30 | ||||
No progression
Alive
W, White; H, Hispanic; KPS, Karnofsky Performance Status; WHO, World Health Organization; IDH1, isocitrate dehydrogenase type 1; MGMT, O6-Methylguanine-DNA methyltransferase; PFS, progression-free survival; OS, overall survival; O-E, observed – expected survival months; RPA, recursive partitioning analysis; NA, tissue not available.
Toxicity and Adverse Events
Patients on study were monitored for toxicity and adverse events (AEs) defined according to the National Cancer Institute's Common Toxicity Criteria (Version 3.0). No patients experienced AEs related to the cellular portion of the pp65-DC vaccine, yet one single AE was noted and attributable to GM-CSF administration. This was classified as a severe (Grade 3) vaccine-related immunologic reaction, which occurred shortly after Vaccine-8. Immunologic workup for this patient revealed sensitization to the GM-CSF component of the vaccine and the production of high levels of anti-GM-CSF autoantibodies during vaccination (25). Removal of GM-CSF from the DC vaccine allowed continued vaccination (total of 10 vaccines) without incident for this patient. No other study drug AEs were detected.
Patient Survival
Patients in this study administered at least three vaccines of pp65-DCs with concomitant DI-TMZ showed significantly increased PFS (Fig 2A) and OS (Fig 2B) compared to historical controls (n = 23) matched for age, gender, tumor, and standard of care treatment. To counter any potential for selection bias of historical controls, a sample size at least double our study cohort was randomly selected from a large database of patients who had resection and were treated contemporaneously over the course of two years at our institution. Selection criteria for these controls included identical demographics (age, gender, histopathology-confirmed GBM) and tumor molecular pathology characteristics (IDH-1-negative and mixed MGMT promoter methylation) as well as identical standard of care treatment, including gross total resection and standard six week XRT/TMZ. Historical controls did not receive any other therapies until progression was documented. Those with progressive disease after the completion of standard therapy were offered additional therapies. Apart from this caveat of additional therapies, patients on our study still showed markedly prolonged PFS and OS compared to matched historical controls (median PFS 25.3 vs. 8.0 months, P = 0.0001, median OS 41.1 vs. 19.2 months, P = 0.0001, log-rank test).
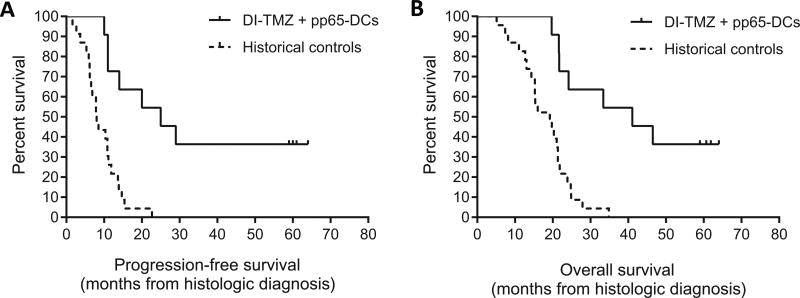
Figure 2.
Survival rates in patients receiving pp65-DCs and DI-TMZ compared to historical controls. A, PFS and B, OS of study patients (n = 11) with newly-diagnosed GBM receiving DI-TMZ conditioning and GM-CSF-containing pp65-DC vaccines compared to matched historical controls (n = 23) with newly diagnosed GBM treated with standard of care and additional therapies after disease progression. Kaplan Meier survival curves represent observed rates for DI-TMZ + pp65 DC patients who completed the predefined study therapy. Of all 11 patients, four had not progressed and were alive at the time of survival analysis (DI-TMZ + pp65-DCs median PFS = 25.3 months [CI95: 11.0-∞] vs. Historical controls median PFS = 8.0 months [CI95: 6.2–10.8], P = 0.0001; DI-TMZ + pp65-DCs median OS = 41.1 months [CI95: 21.6-∞] vs. Historical controls median OS = 19.2 months [CI95: 14.3–21.3], P = 0.0001, log-rank test).
When compared to RPA Class predicted median survival (24), we found that 100% of these patients exceeded expected median survival, with a median gain in survival of 30 months (Table 1). In addition, 4 of these 11 patients remained progression-free 59–64 months from initial surgery. The one enrolled patient who did not receive at least 3 vaccinations progressed at 5.3 months and died at 9.4 months from diagnosis. With inclusion of this patient in survival analyses, the median PFS and OS were 20 months and 33.4 months, respectively.
Patient CMV pp65 Immune Reponses
Of the 11 patients treated with DI-TMZ and at least three vaccines of pp65-DCs, all but one demonstrated an increase in pp65 IFN-γ ELISpot between baseline and Pheresis-2 that occurred after the third pp65-DC vaccine (P = 0.019, Fig 3A). Following reintroduction of DI-TMZ cycle 2 after Pheresis-2, functional (IFN-γ secreting) pp65 responses had diminished towards baseline levels and remained suppressed throughout DI-TMZ cycles 3–6 (Fig 3B).
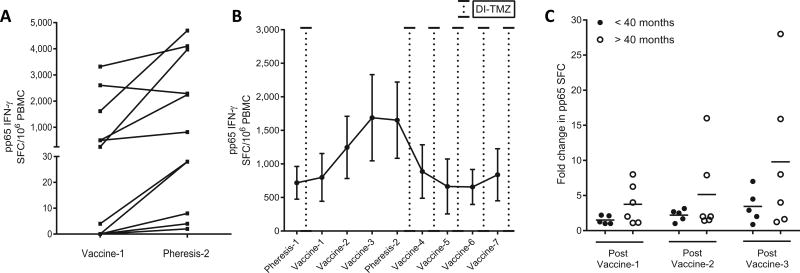
Figure 3.
Patient pp65 ELISpot responses following DI-TMZ and sequential pp65-DC vaccination. A, pp65 antigen-specific T-cell responses as measured by IFN-γ ELISpot ex vivo. Before-and-after pp65 ELISpot following three vaccinations of pp65-DCs from Vaccine-1 to Pheresis-2 (mean ± sem spot-forming cells (SFC) per 106 PBMC) in all patients (n = 11) are shown after stimulation with 138 15-mer peptides overlapping by 11 amino acids spanning the entire pp65 gene (P = 0.019 Wilcoxon signed rank). B, Kinetics of pp65 ELISpot throughout continuous pp65-DC vaccination and intervening DI-TMZ cycles. Timing of DI-TMZ cycles are shown as detached lines. C, Fold changes in functional pp65 ELISpot from baseline pp65 reactivity prior to Vaccine-1. Fold increases stratified by patient OS > 40 months (n = 6) and OS < 40 months (n = 5). Post Vaccine-1: mean 1.51 vs. 3.75 (P = 0.031), Post Vaccine-2: mean 2.20 vs. 5.14 (P = 0.031), Post Vaccine-3: mean 3.45 vs. 9.79 (P = 0.031 Wilcoxon signed rank). ELISpot 0 values normalized to [0 + 1] for calculation of fold change from baseline.
All 11 patients received at least seven vaccines of pp65-DCs and were eligible to receive a maximum of 10 total vaccines if they had not progressed. Monthly DI-TMZ cycles starting from cycle 2 to a total of 12 cycles were administered at the discretion of the treating neuro-oncologist. The four long-term survivors in this study received 10 vaccines, and each demonstrated an expansion in pp65 responses following DC Vaccines 1–3 when DI-TMZ was held. IFN-γ activity stimulated by pp65 then diminished for these patients once DI-TMZ cycles 2 and on were resumed (Supplementary Fig S1).
Moreover, the extent of pp65 IFN-γ increases early on from DC Vaccines 1–3 seem to be important for clinical responses in these long-term survivors, as those with extended OS > 40 months showed a much more significant expansion in pp65 responses from baseline (prior to Vaccine-1) to Pheresis-2 (after three vaccines) compared to those with OS < 40 months (Post Vaccine-3, P = 0.031; Fig 3C). The two patients with the greatest fold changes (open circles) showed prolonged OS at 59 months (censored at the time of analysis) and at 41.1 months.
A tetramer analysis was performed for six patients, using commercially available pp65 tetramer for available HLA types. The percentage of pp65 tetramer-positive CD8+ T cells began to increase following DI-TMZ cycle 1, likely due to the homeostatic expansion of several antigen-specific populations (Supplementary Fig S2). Tetramer positivity did remain elevated after three DC vaccinations to Pheresis-2. Tetramer-positive CD8+ T cells then decreased at Vaccine-4 after reinitiating DI-TMZ with cycle 2, similar to the decline in functional pp65-ELISpot responses in Fig 3B. Because tetramer detection more so indicates cellular phenotype rather than function, we chose to optimize the pp65 ELISpot assay to monitor pp65 functional responses by directly stimulating PBMCs with the pp65 epitope and measuring IFN-γ output with background subtraction of non-specific IFN-γ-secreting cells. Using the pp65 IFN-γ ELISpot as a more informative and encompassing assay to detect the expansion and contraction of pp65 responses, we observed that functional responses against the pp65 antigen were boosted by pp65-DC Vaccines 1–3 and subsequently diminished by the continuation of monthly DI-TMZ cycles.
Radiographic Changes to MRI Scans
MRI scans were performed for all patients within three weeks following completion of standard XRT/TMZ (75 mg/m2/d). We performed a retrospective assessment, with secondary review by a radiation-oncologist and neuro-oncologist, of MRI scans for patients with shorter survival times. Overall, serial MRI scans for these patients demonstrated the usual trend in progressive features, including increasing FLAIR signal from baseline post-XRT/TMZ scans and increased nodular enhancement on T1-weighted contrast images within the resection cavity, both of which were concerning for progressive disease.
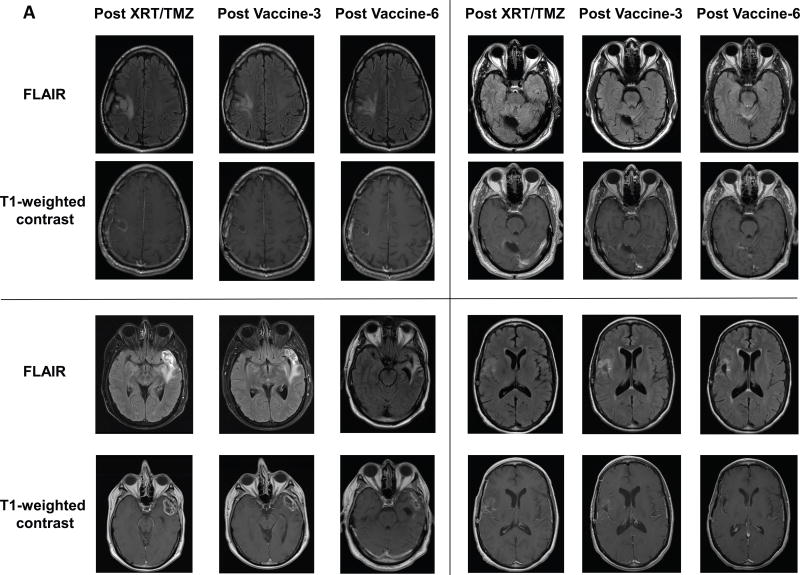
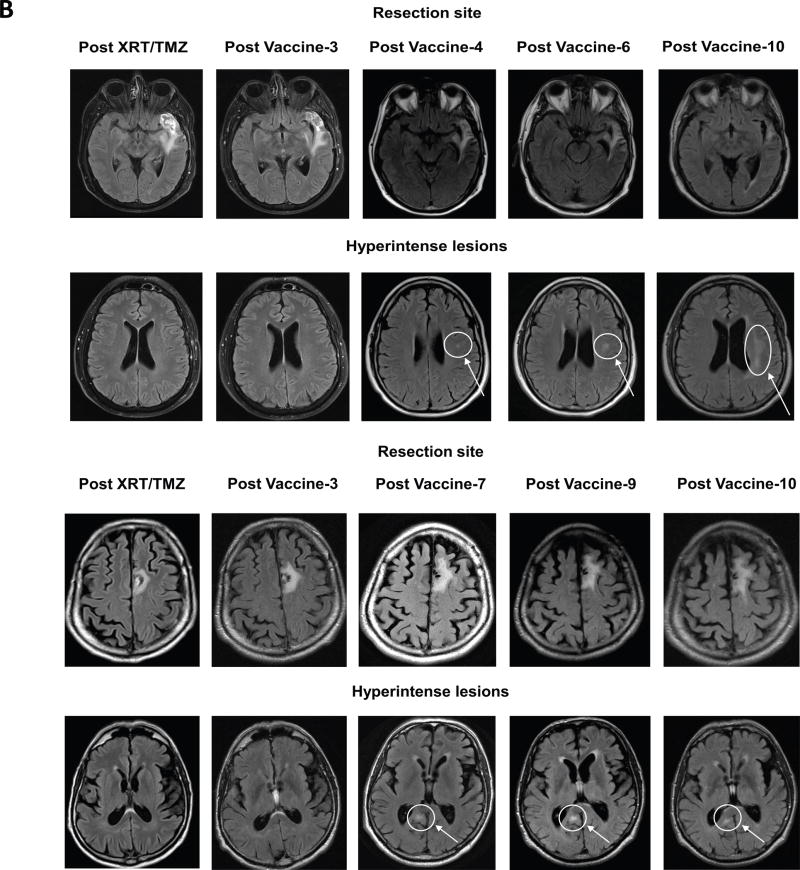
For the four long-term survivors, MRI scans at baseline post XRT/TMZ, post Vaccine-3, and post Vaccine-6 showed a trend of 1) stable or steadily FLAIR signal and edema during recovery from XRT and standard six-week TMZ therapy, 2) no new T1 contrast enhancement along resection margins or additional sites and 3) gradual collapse of the resection cavity (Fig 4A). Interestingly, in two patients with extended OS at 60.7 and 46.5 months (Patients 4 and 7), after repeated DC vaccination, satellite hyperintensities that did not enhance with contrast appeared a considerable distance from the resection site (Fig 4B). Notably, these lesions did not appear in any of the patients with < 40 month OS, were not originally present following XRT/TMZ, and were calculated to be outside of XRT high-dose radiation fields, thus unlikely to represent radiation-induced damage. Due to limitations of this retrospective analysis, the clinical significance of these lesions was not determined, whether these changes represent satellite sites of immune-activation related to DC vaccination or present as coincidental age-related changes in this population. Nonetheless, they remain an interesting feature of patients receiving repeated vaccination with pp65-DCs.
Figure 4.
MRI changes in long-term survivors with sequential pp65-DC vaccination. A, Sequential MRI scans (FLAIR and T1-weighted with contrast) of four long term survivors receiving pp65-DCs and DI-TMZ (Patient 2, Patient 3, Patient 4, and Patient 5). Repeat MRI scans demonstrate steadily decreasing FLAIR hyperintensity and stable or decreasing contrast enhancement with collapse of the resection cavity. B, Satellite FLAIR hyperintense lesions appearing after several vaccinations with pp65-DCs in two patients with prolonged OS (Patient 4 and Patient 7). These lesions were not originally present at the post-XRT/TMZ scan and were calculated to be outside the range of XRT high-dose radiation fields. Presentation of these lesions was first detected after Vaccine-4 and Vaccine-7, and their signal persisted through Vaccine-10.
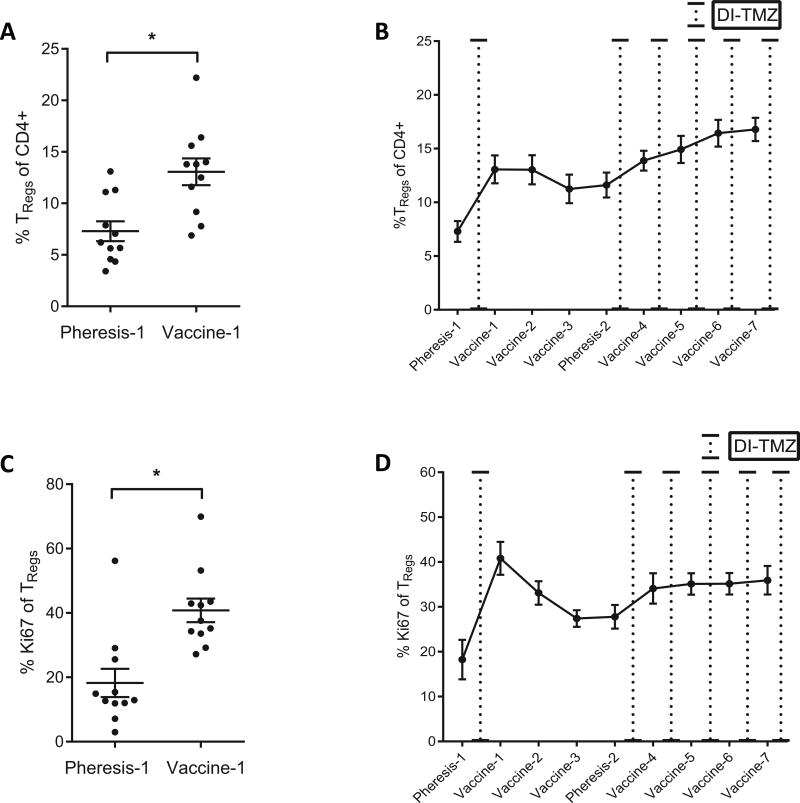
TReg Reconstitution with TMZ-Induced Lymphopenia
Following DI-TMZ cycle 1, the proportion of TRegs among CD4+ T cells increased (P = 0.002, Fig 5A). TReg proportions also steadily increased from Pheresis-2 to Vaccine-7 (Fig 5B). Interestingly, TReg proliferation, as assessed by Ki67+ staining, increased following the initiation of DI-TMZ cycle 1 (P = 0.002, Fig 5C) and then progressively declined between Vaccine-1 and Pheresis-2 when DI-TMZ was held. Following lymphodepletion, TRegs have been shown to reconstitute quite early in response to an increased pool of homeostatic cytokines (26–28). Here, TRegs exhibited proliferation following DI-TMZ lymphodepletion. Once DI-TMZ cycle 2 was initiated, a steady rate of proliferation was noted and persisted with subsequent DI-TMZ cycles (Fig 5D).
Figure 5.
TReg responses following DI-TMZ. A, TReg proportions increase from Pheresis-1 (7.3% ± 0.96, range 3.4–13.1) to Vaccine-1 (13.1% ± 1.3, range 6.9–22.2) following DI-TMZ cycle 1 (mean ± sem, P = 0.001 Wilcoxon signed rank). B, Repeated vaccination and reintroduction of DI-TMZ cycles are compounded by steadily increasing TReg proportions from Pheresis-2 to Vaccine-7). C, TReg proliferation by Ki67 from Pheresis-1 (18.3% ± 4.4, range 2.96–56.2) to Vaccine-1 (40.8% ± 3.7, range 27.2–69.9) following DI-TMZ cycle 1 (mean ± sem, P = 0.002 Wilcoxon signed rank). D, TReg proliferation initially increases following DI-TMZ cycle 1 but remain steady in proliferative capacity following continuous DI-TMZ cycles.
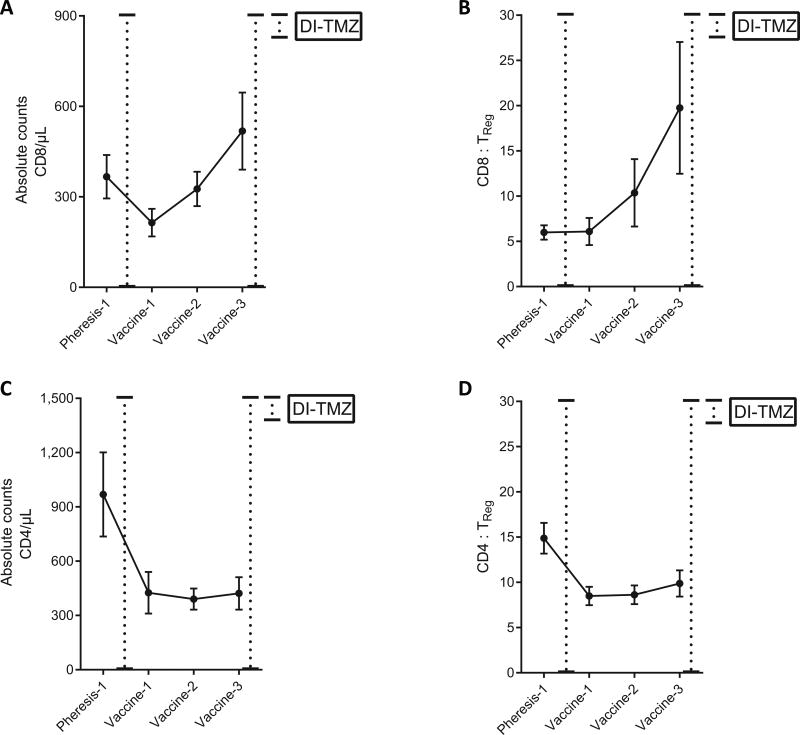
As TReg proportions increased significantly following DI-TMZ cycle 1 (Fig 5), both peripheral CD4+ and CD8+ numbers decreased (Fig 6A and 6C). Although TReg proportions remained elevated throughout sequential pp65-DCs from Vaccine-1 to Vaccine-3, CD8+ numbers and CD8 : TReg ratios steadily increased amidst this high proportion of TRegs (Fig 6A and 6B). Conventional CD4+ and CD4 : TReg ratios were seemingly unaffected by sequential pp65-DCs (Fig 6C and 6D).
Figure 6.
Sequential pp65 DC vaccination expands peripheral CD8+ T cells and CD8 : TReg ratios but does not affect conventional CD4+ counts or CD4 : TReg ratios. A, CD8+ T cell counts in the peripheral blood of patients decrease following DI-TMZ cycle 1 from Pheresis-1 to Vaccine-1 (P = 0.020) and steadily increase with sequential vaccination of pp65-DCs from Vaccine-1 to Vaccine-3 (P = 0.012). B, CD8 : TReg ratios steadily increase following sequential pp65-DCs (Vaccine-1 to Vaccine-3, P = 0.004) C, Conventional CD4+ T cell counts in the peripheral blood of patients dramatically diminish following DI-TMZ cycle 1 (P = 0.037) and do not increase following sequential pp65 DC vaccination. D, CD4 : TReg ratios decrease following DI-TMZ cycle 1 (P = 0.002) but are not affected by sequential pp65-DCs (A-D, mean ± sem, Wilcoxon signed rank).
Discussion
This study corroborates prior studies targeting CMV pp65 in newly diagnosed GBM and demonstrates that CMV DC vaccines can increase pp65 immunity despite lymphodepleting and even dose-intensified TMZ while leading to unexpectedly prolonged PFS and OS. In our prior study, we demonstrated that patients randomized to pp65-DCs with tetanus-diphtheria (Td) toxoid vaccine site pre-conditioning had significantly improved PFS and OS compared to the control cohort (OSTd = 20.6 – 47.3 months vs. OSunpulsed DC = 13.8 – 41.3 months, P = 0.013) (9). In the current study, targeting CMV with pp65-DCs and DI-TMZ again resulted in long-term PFS and OS for patients with newly diagnosed GBM, with 4 of 11 patients who received at least 3 vaccinations remaining progression-free at 59–64 months following surgery. This outcome exceeded observed survival in both arms of our prior small randomized trial (9), observed PFS and OS of a matched historical control cohort, and expected median survival using RPA Class (24). Furthermore, the survival outcomes in these patients are unlikely to be strictly attributable to DI-TMZ, as the recent phase III RTOG 0525 study demonstrated no survival benefit of the same DI-TMZ regimen over standard (STD)-TMZ across all subgroups of patients with newly-diagnosed GBM including RPA Class, MGMT status, and extent of resection (29).
Our study suggests that DI-TMZ is more efficacious if used in concert with antigen-specific vaccination for GBM. T cell responses were expanded by pp65-DC vaccines in an antigen-specific manner following DI-TMZ. From Vaccine-1 to Vaccine-3, all but one patient showed significant expansion in pp65 immunity. Suspending repetitive DI-TMZ cycles during these first three pp65-DC vaccines proved beneficial, as pp65-specific immune responses continued to increase during this period. The extent of pp65 IFN-γ increases early on from DC Vaccines 1–3 was relevant to patient survival, as those with extended OS > 40 months showed a much more significant expansion in pp65 responses after three DC vaccines compared to those with OS < 40 months. However, with the reintroduction of DI-TMZ after Pheresis-2, pp65 responses diminished, elucidating that repeated monthly DI-TMZ cycles may be detrimental to maintaining IFN-γ activity with later vaccination of pp65-DCs. This can most notably be appreciated via the kinetics of pp65 responses in the four long-term survivors from Vaccine-4 to Vaccine-10.
This is not unexpected given the known effects of TMZ on peripheral lymphocyte counts in the blood (10, 30). Thus, our results highlight that the timing of vaccination with pp65-DCs in relation to DI-TMZ or any lymphodepleting drug is an important consideration in generating robust immune responses.
All 11 patients received at least seven vaccines of pp65-DCs. The kinetics of pp65 vaccine responses showed a significant decline at Vaccine-4. This diminished response may have been a result of 1) natural contraction in pp65 reactivity following a one month latency between Vaccine-3 and Vaccine-4 or 2) a possible detrimental effect of DI-TMZ cycle 2 depleting pp65-specific effector T-cells. Following Vaccine-5, when monthly DI-TMZ and monthly pp65-DCs were administered, we observed a slight upward trend in mean pp65 reactivity at Vaccine-7, mostly driven by increases in five of the 11 patients. Since all patients received at least seven vaccines, this mean increase was not a result of patient dropout. In addition, at this stage of the vaccination schedule, the memory repertoire of patient pp65-specific T-cells may have expanded and adopted a more resilient phenotype to DI-TMZ lymphodepletion. The recruitment of DNA damage repair mechanisms have been described in memory CD8+ T-cell populations expressing the DNA repair enzyme methyl-guanine methyltransferase (MGMT) (31). Such expression may have played a role in memory T-cell kinetics at this point in the vaccination schedule amidst monthly DI-TMZ. Nonetheless, studies are underway to investigate mechanisms underlying such memory T-cell resistance to TMZ.
Our previous studies characterize how increased TReg fractions govern cellular immune defects in patients with GBM and underscore the deleterious effects of TRegs on vaccine-mediated immunotherapy (32, 33). During recovery from lymphodepleted states, remnant T-cell pools undergo homeostatic expansion through proliferation (14, 27, 28, 34). TRegs represent a portion of the depleted T cell repertoire and not only proliferate early in this recovery but do so rapidly in response to high levels of the host cytokine interleukin (IL)-2 (26–28, 35). In the present study, patients exhibited an increase in TReg proportions as a result of increased proliferation following a single cycle of DI-TMZ. This is not surprising, as previous studies demonstrate increased TReg proportions in patients with GBM following STD-TMZ (35) and DI-TMZ regimens (10). After the initial peak, TReg proliferation gradually declined from Vaccine-1 to Pheresis-2 while pp65 responses simultaneously increased, indicating that while DI-TMZ still resulted in elevated TReg fractions, this did not appear to limit the induction of pp65 antigen-specific cellular responses. Elevated TReg proportions throughout sequential pp65-DC vaccination did not seem to affect peripheral CD8+ numbers, as CD8+ counts and CD8 : TReg ratios steadily increased during this interval. Expansion of CD8+ T cell numbers and functional pp65 responses may have been resilient to TReg suppression immediately following this peak, as provision of an antigen-specific vaccine in the context of lymphodepletion might have superseded homeostatically expanding TRegs (36–38). However, our findings raise the question if targeting TRegs during points of maximal proliferation could further enhance pp65 T-cell responses. A potential strategy previously published by our group employs monoclonal antibody blockade of the IL-2 receptor α (IL-2R α /CD25) to effectively deplete TRegs without impairing effector pp65 T-cell responses, and this is under further investigation in our two arm phase I study (NCT00626483) (35).
In conclusion, our study results confirm our prior findings that CMV pp65 represents a targetable axis in newly diagnosed GBM resulting in patient survival far exceeding that of predicted outcomes and observed rates in historical controls. Patients in our study showed notably extended median PFS (25.3 months) and OS (41.1 months) compared to age-matched patients in the RTOG 0525 trial (n = 422) who similarly received 21-day DI-TMZ cycles following the six week standard chemoradiation therapy (median PFS and OS, 6.7 and 14.9 months). Although eligibility criteria in our study differed from those in the RTOG trial (partial and total resection, KPS ≥ 60), patients shared similar characteristics with respect to age, MGMT methylation status, and RPA Class. We recognize that in our study, the efficacy of DI-TMZ alone was not directly compared with DI-TMZ and pp65-DC vaccination, but given the results from the RTOG trial, DI-TMZ used independently is unlikely to improve outcomes in this patient population according to recent data (29). However, our prior clinical study demonstrated that DI-TMZ induced lymphopenia, when provided with antigen-specific vaccination, resulted in superior immune responses compared to STD-TMZ dosing (10). Here, using pp65 as a different antigen in GBM, we were able to validate these prior findings, demonstrating that DI-TMZ is best utilized in concert with a vaccine to generate robust antigen-specific immunity. What remains to be determined is whether this combinatorial regimen is strictly dependent on antigen-specificity, a question that is best suited for future randomization studies. Overall, our results strengthen prior findings from other trials targeting CMV and provide evidence for the association between pp65 targeting in GBM and long-term survival.
Supplementary Material
Translational Relevance.
The highly aggressive and therapeutically resistant nature of glioblastoma (GBM) is evidenced by a median survival of <15 months. More precise and efficacious therapies are desperately needed. Several groups have demonstrated that Cytomegalovirus (CMV) proteins are expressed in over 90% of sampled GBMs. Moreover, CMV antigen expression is restricted to glioma cells and not surrounding normal brain, providing the opportunity to subvert CMV proteins as tumor-specific immunotherapy targets. In this study, we targeted the CMV antigen pp65 using dendritic cells (DCs) in combination with dose-intensified temozolomide (TMZ) and evaluated patient antitumor immune responses and survival. Despite increases in regulatory T-cell proportions after TMZ, patients treated with pp65-DCs showed increased pp65 immunity and long-term survival extending beyond predicted rates, fortifying prior studies that target CMV antigens in GBM. Randomized studies on the prevention of generated regulatory T-cells in the context of TMZ treatment and CMV targeting are under way.
Acknowledgments
The authors thank the staff who supported this study, including D. Lally-Goss, S. McGehee-Norman, B. Perry, S. Shipes and R. Edward Coleman.
Financial Support:
This work was supported by the National Institutes of Health (NIH) SPORE in Brain Cancer (P50-CA190991-02, John Sampson), Small Business Technology Transfer (STTR) with Annias Immunotherapeutics, Inc. (R42-CA153845-02, John Sampson), Duke Comprehensive Cancer Core Grant (P30-CA14236-42, John Sampson), as well as NIH R01 (R01-CA177476-04, R01-NS085412-04, R01-NS086943-03, John Sampson; R01-NS067037, R01-CA134844, Duane Mitchell; 1R01CA175517-01A1, John Sampson and Duane Mitchell), P01 (P01-CA154291-05, Darell Bigner and John Sampson), F30 NRSA (F30-CA180411-01, Kristen Batich), and R25 (R25-NS065731-07, John Sampson) funding sources.
Footnotes
Disclosure of Potential Conflicts of Interest
J.H. Sampson holds stock ownership and is on the Board of Directors with Annias Immunotherapeutics, serves as a consultant and advisory board member for Celldex Therapeutics, and reports honoraria for Celldex Therapeutics, Bristol-Myers Squibb, and Brainlab. D.A. Mitchell is a consultant for Schering Plough North American Investigators Advisory Board. S.K Nair is a co-inventor on a patent that has been licensed by Argos Therapeutics (Durham, NC) through Duke University. S.K. Nair has no financial interests in Argos Therapeutics and is not compensated by Argos Therapeutics. JH. Sampson and D.A. Mitchell are co-inventors on a patent describing the immunologic targeting of CMV antigens in cancer. J.H. Sampson, D.A. Mitchell, and K.A. Batich are co-inventors on a patent for improving the immunogenicity of dendritic cell vaccines. No other potential conflicts of interest were disclosed by the other authors.
Authors’ Contributions
Conception and design: John H. Sampson, Gary E. Archer, Duane A. Mitchell
Development of methodology: John H. Sampson, Gary E. Archer, Smita K. Nair, Weihua Xie, Elizabeth A. Reap, Duane A. Mitchell
Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): Kristen A. Batich, Gary E. Archer, Elizabeth A. Reap, Robert Schmittling, Pam Norberg, Gordana Vlahovic, Duane A. Mitchell, John H. Sampson
Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): Kristen A. Batich, Gary E. Archer, Elizabeth A. Reap, Pam Norberg, Luis Sanchez-Perez, Robert Schmittling, James E. Herndon II, Patrick Healy, Gordana Vlahovic, Duane A. Mitchell, John H. Sampson
Writing, review, and/or revision of the manuscript: Kristen A. Batich, John H. Sampson
Administrative, technical, or material support (i.e., reporting or organizing data, constructing databases): Kristen A. Batich, John H. Sampson, Gary E. Archer, Gordana Vlahovic, Duane A. Mitchell
Study supervision: John H. Sampson and Duane A. Mitchell
References
- 1.Imperato JP, Paleologos NA, Vick NA. Effects of treatment on long-term survivors with malignant astrocytomas. Annals of Neurology. 1990;28:818–22. doi: 10.1002/ana.410280614. [DOI] [PubMed] [Google Scholar]
- 2.Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJB, et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. New England Journal of Medicine. 2005;352:987–96. doi: 10.1056/NEJMoa043330. [DOI] [PubMed] [Google Scholar]
- 3.Mitchell DA, Xie W, Schmittling R, Learn C, Friedman A, McLendon RE, et al. Sensitive detection of human cytomegalovirus in tumors and peripheral blood of patients diagnosed with glioblastoma. Neuro Oncol. 2008;10:10–8. doi: 10.1215/15228517-2007-035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cobbs CS, Harkins L, Samanta M, Gillespie GY, Bharara S, King PH, et al. Human cytomegalovirus infection and expression in human malignant glioma. Cancer Research. 2002;62:3347–50. [PubMed] [Google Scholar]
- 5.Prins RM, Cloughesy TF, Liau LM. Cytomegalovirus immunity after vaccination with autologous glioblastoma lysate. N Engl J Med. 2008;359:539–41. doi: 10.1056/NEJMc0804818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dziurzynski K, Chang SM, Heimberger AB, Kalejta RF, McGregor Dallas SR, Smit M, et al. Consensus on the role of human cytomegalovirus in glioblastoma. Neuro Oncol. 2012;14:246–55. doi: 10.1093/neuonc/nor227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ranganathan P, Clark PA, Kuo JS, Salamat MS, Kalejta RF. Significant association of multiple human cytomegalovirus genomic Loci with glioblastoma multiforme samples. J Virol. 2012;86:854–64. doi: 10.1128/JVI.06097-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nair SK, De Leon G, Boczkowski D, Schmittling R, Xie W, Staats J, et al. Recognition and Killing of Autologous, Primary Glioblastoma Tumor Cells by Human Cytomegalovirus pp65-Specific Cytotoxic T Cells. Clinical Cancer Research. 2014;20:2684–94. doi: 10.1158/1078-0432.CCR-13-3268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mitchell DA, Batich KA, Gunn MD, Huang MN, Sanchez-Perez L, Nair SK, et al. Tetanus toxoid and CCL3 improve dendritic cell vaccines in mice and glioblastoma patients. Nature. 2015;519:366–9. doi: 10.1038/nature14320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sampson JH, Aldape KD, Archer GE, Coan A, Desjardins A, Friedman AH, et al. Greater chemotherapy-induced lymphopenia enhances tumor-specific immune responses that eliminate EGFRvIII-expressing tumor cells in patients with glioblastoma. Neuro Oncol. 2011;13:324–33. doi: 10.1093/neuonc/noq157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sampson JH, Archer GE, Mitchell DA, Heimberger AB, Herndon JE, 2nd, Lally-Goss D, et al. An epidermal growth factor receptor variant III-targeted vaccine is safe and immunogenic in patients with glioblastoma multiforme. Mol Cancer Ther. 2009;8:2773–9. doi: 10.1158/1535-7163.MCT-09-0124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sampson JH, Heimberger AB, Archer GE, Aldape KD, Friedman AH, Friedman HS, et al. Immunologic escape after prolonged progression-free survival with epidermal growth factor receptor variant III peptide vaccination in patients with newly diagnosed glioblastoma. J Clin Oncol. 2010;28:4722–9. doi: 10.1200/JCO.2010.28.6963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Slingluff CLJ, Petroni GR, Yamshchikov GV, Barnd DL, Eastham S, Galavotti H, et al. Clinical and immunologic results of a randomized phase II trial of vaccination using four melanoma peptides either administered in granulocyte-macrophage colony-stimulating factor in adjuvant or pulsed on dendritic cells. Journal of Clinical Oncology. 2003;21:4016–26. doi: 10.1200/JCO.2003.10.005. [DOI] [PubMed] [Google Scholar]
- 14.Dudley ME, Wunderlich JR, Robbins PF, Yang JC, Hwu P, Schwartzentruber DJ, et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science. 2002;298:850–4. doi: 10.1126/science.1076514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Asavaroengchai W, Kotera Y, Mule JJ. Tumor lysate-pulsed dendritic cells can elicit an effective antitumor immune response during early lymphoid recovery. Proc Natl Acad Sci U S A. 2002;99:931–6. doi: 10.1073/pnas.022634999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Balss J, Meyer J, Mueller W, Korshunov A, Hartmann C, von Deimling A. Analysis of the IDH1 codon 132 mutation in brain tumors. Acta Neuropathol. 2008;116:597–602. doi: 10.1007/s00401-008-0455-2. [DOI] [PubMed] [Google Scholar]
- 17.Vlassenbroeck I, Califice S, Diserens AC, Migliavacca E, Straub J, Di Stefano I, et al. Validation of real-time methylation-specific PCR to determine O6-methylguanine-DNA methyltransferase gene promoter methylation in glioma. J Mol Diagn. 2008;10:332–7. doi: 10.2353/jmoldx.2008.070169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nair S, Archer GE, Tedder TF. Isolation and generation of human dendritic cells. Current Protocols in Immunology. 2012:1–23. doi: 10.1002/0471142735.im0732s99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Romani N, Gruner S, Brang D, Kampgen E, Lenz A, Trockenbacher B, et al. Proliferating dendritic cell progenitors in human blood. The Journal of experimental medicine. 1994;180:83–93. doi: 10.1084/jem.180.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Thurner B, Roder C, Dieckmann D, Heuer M, Kruse M, Glaser A, et al. Generation of large numbers of fully mature and stable dendritic cells from leukapheresis products for clinical application.[erratum appears in J Immunol Methods 1999 Apr 22;224(1–2):211] Journal of Immunological Methods. 1999;223:1–15. doi: 10.1016/s0022-1759(98)00208-7. [DOI] [PubMed] [Google Scholar]
- 21.Bernstein DI, Reap EA, Katen K, Watson A, Smith K, Norberg P, et al. Randomized, double-blind, Phase 1 trial of an alphavirus replicon vaccine for cytomegalovirus in CMV seronegative adult volunteers. Vaccine. 2009;28:484–93. doi: 10.1016/j.vaccine.2009.09.135. [DOI] [PubMed] [Google Scholar]
- 22.Janetzki S, Price L, Schroeder H, Britten CM, Welters MJ, Hoos A. Guidelines for the automated evaluation of Elispot assays. Nat Protoc. 2015;10:1098–115. doi: 10.1038/nprot.2015.068. [DOI] [PubMed] [Google Scholar]
- 23.Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, et al. Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol. 2010;28:1963–72. doi: 10.1200/JCO.2009.26.3541. [DOI] [PubMed] [Google Scholar]
- 24.Curran WJ, Jr, Scott CB, Horton J, Nelson JS, Weinstein AS, et al. Recursive partitioning analysis of prognostic factors in three Radiation Therapy Oncology Group malignant glioma trials. Journal of the National Cancer Institute. 1993;85:704–10. doi: 10.1093/jnci/85.9.704. [DOI] [PubMed] [Google Scholar]
- 25.Mitchell DA, Sayour EJ, Reap E, Schmittling R, DeLeon G, Norberg P, et al. Severe adverse immunologic reaction in a patient with glioblastoma receiving autologous dendritic cell vaccines combined with GM-CSF and dose-intensified temozolomide. Cancer Immunol Res. 2015;3:320–5. doi: 10.1158/2326-6066.CIR-14-0100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sanchez-Perez LA, Choi BD, Archer GE, Cui X, Flores C, Johnson LA, et al. Myeloablative temozolomide enhances CD8+ T-cell responses to vaccine and is required for efficacy against brain tumors in mice. PLoS One. 2013;8:e59082. doi: 10.1371/journal.pone.0059082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Setoguchi R, Hori S, Takahashi T, Sakaguchi S. Homeostatic maintenance of natural Foxp3(+) CD25(+) CD4(+) regulatory T cells by interleukin (IL)-2 and induction of autoimmune disease by IL-2 neutralization. Journal of Experimental Medicine. 2005;201:723–35. doi: 10.1084/jem.20041982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Neujahr DC, Chen C, Huang X, Markmann JF, Cobbold S, Waldmann H, et al. Accelerated memory cell homeostasis during T cell depletion and approaches to overcome it. Journal of Immunology. 2006;176:4632–9. doi: 10.4049/jimmunol.176.8.4632. [DOI] [PubMed] [Google Scholar]
- 29.Mehta MP, Wang M, Aldape K, Stupp R, Jaeckle KA, Blumenthal D, et al. RTOG 0525: Exploratory Subset Analysis from a Randomized Phase III Trial Comparing Standard (std) Adjuvant Temozolomide (TMZ) with a Dose-dense (dd) Schedule for Glioblastoma (GBM) International journal of radiation oncology, biology, physics. 2011;81:S128–S9. [Google Scholar]
- 30.Fadul CE, Fisher JL, Gui J, Hampton TH, Côté AL, Ernstoff MS. Immune modulation effects of concomitant temozolomide and radiation therapy on peripheral blood mononuclear cells in patients with glioblastoma multiforme. Neuro Oncol. 2011;13:393–400. doi: 10.1093/neuonc/noq204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Galgano A, Barinov A, Vasseur F, de Villartay JP, Rocha B. CD8 Memory Cells Develop Unique DNA Repair Mechanisms Favoring Productive Division. PLoS One. 2015;10:e0140849. doi: 10.1371/journal.pone.0140849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fecci PE, Mitchell DA, Whitesides JF, Xie W, Friedman AH, Archer GE, et al. Increased regulatory T-cell fraction amidst a diminished CD4 compartment explains cellular immune defects in patients with malignant glioma. Cancer Research. 2006;66:3294–302. doi: 10.1158/0008-5472.CAN-05-3773. [DOI] [PubMed] [Google Scholar]
- 33.Fecci PE, Ochiai H, Mitchell DA, Grossi PM, Sweeney AE, Archer GE, et al. Systemic CTLA-4 blockade ameliorates glioma-induced changes to the CD4+ T cell compartment without affecting regulatory T-cell function. Clin Cancer Res. 2007;13:2158–67. doi: 10.1158/1078-0432.CCR-06-2070. [DOI] [PubMed] [Google Scholar]
- 34.Dummer W, Niethammer AG, Baccala R, Lawson BR, Wagner N, Reisfeld RA, et al. T cell homeostatic proliferation elicits effective antitumor autoimmunity. Journal of Clinical Investigation. 2002;110:185–92. doi: 10.1172/JCI15175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mitchell DA, Cui X, Schmittling RJ, Sanchez-Perez L, Snyder DJ, Congdon KL, et al. Monoclonal antibody blockade of IL-2 receptor alpha during lymphopenia selectively depletes regulatory T cells in mice and humans. Blood. 2011;118:3003–12. doi: 10.1182/blood-2011-02-334565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Prlic M, Jameson SC. Homeostatic expansion versus antigen-driven proliferation: common ends by different means? Microbes Infect. 2002;4:531–7. doi: 10.1016/s1286-4579(02)01569-1. [DOI] [PubMed] [Google Scholar]
- 37.Kieper WC, Jameson SC. Homeostatic expansion and phenotypic conversion of naïve T cells in response to self peptide/MHC ligands. Proc Natl Acad Sci U S A. 1999;96:13306–11. doi: 10.1073/pnas.96.23.13306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang LX, Li R, Yang G, Lim M, O'Hara A, Chu Y, et al. Interleukin-7-dependent expansion and persistence of melanoma-specific T cells in lymphodepleted mice lead to tumor regression and editing. Cancer Res. 2005;65:10569–77. doi: 10.1158/0008-5472.CAN-05-2117. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.