Abstract
The use of psychostimulants is often associated with hypersexuality, and psychostimulant users have identified the effects of drug on sexual behavior as a reason for further use. It was previously demonstrated in male rats that methamphetamine (Meth), when administered concurrently with sexual behavior results in impairment of inhibition of sexual behavior in a conditioned sex aversion (CSA) paradigm where mating is paired with illness. This is indicative of maladaptive sex behavior following Meth and sex experience. The present study examined the neural pathways activated during inhibition of sexual behavior in male rats and the effects of concurrent Meth and sexual behavior on neural activity, using ERK phosphorylation (pERK). First, exposure to conditioned aversive stimuli in males trained to inhibit sexual behavior in the CSA paradigm increased pERK expression in medial prefrontal (mPFC), orbitofrontal cortex (OFC) and areas in striatum and amygdala. Second, effects of concurrent Meth and sex experience were tested in males that were exposed to four daily sessions of concurrent Meth (1 mg/kg) or saline and mating and subsequently exposed to CSA one week after last treatment. Meth and mating-treated males showed significant impairment of inhibition of mating, higher pERK expression under baseline conditions, and disrupted pERK induction by exposure to the conditioned aversive stimuli in mPFC and OFC. These alterations of pERK occurred in CaMKII-expressing neurons, suggesting changes in efferent projections of these areas. Altogether, these data show that concurrent Meth and mating experience causes maladapative sexual behavior that is associated with alterations in neural activation in mPFC and OFC.
INTRODUCTION
There is increased awareness that drug addiction may share ‘brain vulnerabilities’ with the compulsive pursuit of non-drug rewards, such as gambling, sex, and food (Kraus et al, 2016). Abused drugs, including cocaine (Johnson and Bruner, 2012), methamphetamine (Meth) (Zapata et al, 2008), heroin (Mitchell et al, 2012), and alcohol (Mastroleo et al, 2015) are highly co-morbid with hypersexuality (Frohmader et al, 2010b). Drug users report heightened sexual desire, arousal, and pleasure (Green and Halkitis, 2006), loss of inhibitory control of sex behavior and increased risk taking behavior (Green and Halkitis, 2006; Semple et al, 2004). These effects are reported in men and women, heterosexuals and homosexuals (Cartier et al, 2008; Johnson et al, 2015; Mausbach et al, 2009; Semple et al, 2004), and can serve as a primary motivation for drug use (Lyons et al, 2010). Thus, a better understanding of the neural basis for the association between drug use and hypersexuality may be beneficial for designing treatment options for both drug and sex addiction.
Our laboratory has shown that Meth treatment in male rats causes maladaptive sex behavior (Frohmader et al, 2010a, 2011), utilizing a conditioned sex aversion (CSA) paradigm (Davis et al, 2010) in which male rats learn to associate mating with subsequent visceral illness induced by lithium chloride (LiCl) (Peters, 1983). Following repeated pairings of mating and the illness, animals ceased to engage in copulatory behaviors (Davis et al, 2010; Frohmader et al, 2010a, 2011). However, males that received repeated experience with Meth exposure and mating behavior (ie, concurrent Meth and sex experience), continued to engage in sexual behavior in this CSA paradigm despite the learned negative consequences and weeks of drug abstinence before testing in the CSA paradigm (Frohmader et al, 2010c, 2011). This attenuating effect of Meth and sex experience was dependent on the concurrent experience of Meth and sex (Frohmader et al, 2011). These findings provide an experimental paradigm to further investigate the neural mechanisms by which concurrent Meth and mating experience causes maladaptive sexual behavior or hypersexuality.
The neural substrates of inhibition of sexual behavior are not understood. Therefore, the current study first investigated the neural circuits activated by cues associated with CSA using phosphorylation of MAP kinase, or extracellular-regulated protein kinase (pERK), as a marker for neural activation in multiple brain regions known to be involved in sexual and drug reward behavior (Frohmader et al, 2010b), and aversion learning (Longoni et al, 2011). pERK is a useful marker for neural activation by conditioned cues and its expression is critical for learning and memory during different task including conditioned taste aversion (Berman et al, 1998), fear conditioning (Schafe et al, 2000), food-reward conditioning (Ribeiro et al, 2005), and sexual behavior (Pitchers et al, 2014). Moreover, the expression of pERK is involved in the mechanisms of drug action and synaptic plasticity (Giorgi et al, 2015; Valjent et al, 2006b).
Second, the hypothesis was tested that concurrent Meth and mating experience causes alterations in the neural circuits involved in inhibition of sexual behavior, by determining if concurrent Meth and sex experience disrupted conditioned aversive cue-induced pERK expression. Finally, to identify the particular neuronal projections that demonstrate disruption of pERK activation, cortical efferents, ie, pyramidal neurons, were identified by CaMKII expression (Fanous et al, 2012) and contrasted with cortical interneurons, identified by GAD expression (Fanous et al, 2012). Altogether, these approaches were used to test the hypothesis that concurrent Meth and mating experience disrupts neural circuits involved in inhibitory control of sexual behavior.
MATERIALS AND METHODS
Subjects
Adult male Sprague–Dawley rats (Charles River, Montreal, QC, Canada (experiments 1 and 2), Wilmington, MA (experiment 2 and Supplementary Study 3; 210–250 g)) were housed in pairs in standard Plexiglas cages on a 12/12 h reversed light/dark cycle. Food and water were available ad libitum. Female rats (Charles River, 201–225 g) for sexual partners were bilaterally ovariectomized under deep anesthesia (87 mg/kg ketamine and 13 mg/kg xylazine) and received a subcutaneous implant (Dow Corning, Midland, MI) containing 5% estradiol benzoate (Sigma-Aldrich, St Louis, MO) and 95% cholesterol (Sigma-Aldrich). Sexual receptivity was induced by a subcutaneous (s.c.) injection of 500 μg progesterone (Sigma-Aldrich) in 0.1 ml sesame oil (Sigma-Aldrich) 4 h before testing. All experimental procedures were conducted during dark phase and approved by the Institutional Animal Care and Use Committees at the University of Western Ontario, the University of Michigan, and the University of Mississippi Medical Center and conform to the guidelines outlined by the Canadian Council on Animal Care and the United States National Institutes of Health.
Sexual Experience
In experiment 1, male rats (n=11) gained sexual experience during 4 twice weekly mating sessions before the experiment. During each mating session, males were allowed to mate with a sexually experienced and hormone-treated receptive female, in separate test cages (mating arenas; 60 × 45 × 50 cm3) containing clean bedding, until the display of one ejaculation or for one hour, whichever occurred first. During each mating session, all standard parameters for sexual performance were recorded, including: mount latency (ML; time from introduction of the female until the first mount), intromission latency (IL; time from introduction of the female until the first mount with vaginal penetration), ejaculation latency (EL; time from the first intromission to ejaculation), post ejaculation interval (PEI; time from ejaculation to first subsequent intromission), number of mounts (M), and number of intromissions (IM) (Frohmader et al, 2011). All animals reached criteria of experience: three or more ejaculations during the four sessions (Supplementary Table 1).
Concurrent Meth and Sex Experience
In experiment 2, male rats received concurrent experience with Meth or saline exposure and sexual behavior. Thirty minutes before mating, males received either Meth (1 mg/kg s.c.; Meth+Sex; n=27) or saline (Sal+Sex; n=26) and were subsequently allowed to mate to 1 ejaculation or for 1 h, whichever occurred first, during 4 daily mating sessions on consecutive days. Meth treatment during these four sessions delayed initiation of mating (increased mount and intromission latencies; Supplementary Table 2), but did not affect measures of performance (ejaculation latencies; Supplementary Table 2), in agreement with our previous studies (Frohmader et al, 2010a). Moreover, groups did not differ in any measure of sexual behavior on the first day of CSA, confirming that the two groups were comparable at the onset of the CSA paradigm (Supplementary Table 2).
CSA
One (experiment 1) or two (experiment 2) weeks following the last mating session, animals were subjected to a CSA paradigm using LiCl as described in our previous papers (Frohmader et al, 2010a, 2011). Briefly, animals were exposed to seven (experiment 1) or eight (experiment 2) consecutive conditioning trials, each consisting of 2 days. On the first day, males were placed in the mating arena for a 10 min habituation period after which a receptive female was introduced, scented by swabbing almond oil on the neck and base of the tail to strengthen conditioning (Agmo, 2002). Males mated for 30 min or until one ejaculation. If intromission did not occur within the first 15 min, mating was terminated. One minute following ejaculation or trial termination all males received a 127.2 mg/kg intraperitoneal (i.p.) injection of LiCl administered at a volume of 10 ml/kg. LiCl was administered regardless of whether mating occurred. The second day of each trail, males received saline injections, but were not exposed to scented females or allowed to mate. Our previous papers have shown significant inhibition of initiation of sexual behavior in male rats following three or more conditioning trials when tested 1–2 weeks after gaining sexual experience during daily or twice weekly sessions (Davis et al, 2010; Frohmader et al, 2010a, 2011).
Final Test
Following the final conditioning trial, males were exposed to either the sex aversion conditioned cue, ie, the scented female and the mating cage environment, and perfused 10 min later to examine pERK expression, or removed from their home cage before brain collection to serve as controls. The expression of pERK is highly dynamic and occurs 5–20 min after neural activation (Frohmader et al, 2010c). Groups in experiment 1 included: exposed to Female (n=6) or Home Cage control (n=6). Groups in experiment two included: Sal+Sex Home Cage (n=12), Sal+Sex Female (n=14), Meth+Sex Home Cage (n=10), and Meth+Sex Female (n=17). Mating behavior was not displayed by any of the males exposed to the scented female during this final test. Experiment 2 was conducted in two separate replicate cohorts of animals, each containing the same experimental groups and using identical methods. This was due to relocation of the laboratory (half of each group was tested at the University of Western Ontario and half at the University of Michigan). The two replicates of the experiment did not statistically differ in any of the measured parameters (outlined below) and were combined for final analysis.
Tissue Preparation
Animals were anesthetized with pentobarbital (270 mg/kg; i.p., Bimeda-MTC, Animal Health, Cambridge, ON, Canada; Vortech, Dearborn, MI, USA) and perfused transcardially with 5 ml of saline followed by 500 ml 4% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA) in 0.1 M phosphate buffer (PB). All perfusions were performed within 4 min of pentobarbital injection to avoid any effects of pentobarbital or injection stress on pERK immunoreactivity (Takamura et al, 2008). Brains were removed and post-fixed for 1 h at room temperature in the same fixative, then immersed in 20% sucrose (Fisher Scientific, Fair Lawn, NJ) and 0.01% Sodium Azide (Sigma-Aldrich) in 0.1 M PB and stored at 4 °C. Coronal sections (35 μm) were cut on a freezing microtome (H400R, Micron, Germany), collected in four parallel series in cryoprotectant solution (30% sucrose and 30% ethylene glycol in 0.1 M PB) and stored at −20 °C until further processing.
Immunohistochemistry
All incubations were performed at room temperature with gentle agitation. Free floating sections were washed extensively with phosphate-buffered saline (0.1 M PB containing 0.9% sodium chloride; PBS) between incubations. Sections were incubated in 1% H2O2 for 10 min, then blocked in incubation solution (PBS containing 0.1% bovine serum albumin and 0.4% Triton X-100) for 1 h.
pERK
One parallel series of sections from each animal was processed to visualize pERK using immunoperoxidase technique. Sections from all experimental groups within each of the two experiments were immunoprocessed simultaneously. Sections were incubated overnight with a rabbit polyclonal antibody against p42 and p44 MAP Kinases ERK1 and ERK2 (pERK; 1:4000; Cell Signaling Technology, Danvers, MA, USA Cat # 9101), followed by a 1 h incubation with biotinylated goat anti-rabbit IgG (1:500; 1 h; Vector Laboratories, Burlingame, CA, USA) and avidin-horseradish peroxidase complex (ABC-Elite; 1:1000; Vector). Next, tissue was incubated for 10 min in a chromogen solution containing 0.02% 3,3′-diaminobenzidine tetrahydrochloride (DAB; Sigma-Aldrich) in 0.1 M PB containing 0.015% hydrogen peroxide, resulting in a reddish-brown reaction product. Sections were washed thoroughly in 0.1 M PB, mounted onto Superfrost plus glass slides (Fisher, Pittsburgh, PA, USA) with 0.3% gelatin in ddH2O. Following DAB staining, slides were dehydrated and coverslipped with dibutyl phthalate xylene (DPX; Sigma-Aldrich).
Dual fluorescence for pERK+CaMKII and pERK+GAD67
One parallel series of sections from experiment 2 containing mPFC and orbitofrontal cortex (OFC) were incubated overnight with either mouse anti-CaMKII (1:10 000; Cat# MA1-048, Pierce Biotechnology, Rockford, IL) or mouse anti-GAD67 (1:200; Cat#MAB5406, lot 2188494, Millipore, Temecula, CA) and Alexa Fluor 555-conjugated goat anti-mouse secondary (1 : 100; 30 min; Cat# A21422, Molecular Probes, Eugene, OR). Next, sections were incubated overnight with rabbit anti-pERK (1:4000; overnight; Cell Signaling, Cat# 9101) and with biotinylated goat anti-rabbit for one hour (1:500; cat. # BA-1000, Vector Laboratories, Burlingame, CA, USA), avidin-horseradish peroxidase complex (ABC-elite, 1:1000 in PBS; 1 h; cat. # PK-6100, Vector), biotinylated tyramine/tissue sample amplification (TSA; 1:250 in PBS containing 1 ul/ml of 3% H2O2 for 10 min; NEL700/700A; PerkinElmer Life Sciences, Boston, MA, USA), and Dylight 488 conjugated streptavidin (1:100 in PBS, 30 min; cat. #21832, lot PF204220, Pierce Biotech., Rockford, IL, USA). Sections were mounted on plus charged slides (Thermo Fisher Scientific, Cat# 12-550-20, Waltham, MA, USA) and coverslipped with an aqueous mounting medium (Gelvatol; (Harlow and Lane, 1988)) containing the anti-fading agent 1,4-diazabicyclo(2,2)octane (DABCO; 50 mg/ml, Sigma-Aldrich) and stored in the dark at 4 °C.
All primary antibodies have been extensively characterized in rat, omission of the primary antibodies prevented all immunoreactivity, and Western blot analysis of rat brain tissue revealed bands at the appropriate molecular weights for all antibodies (Fanous et al, 2012).
Data Analysis
SigmaPlot (Systat Software, San Jose, CA) was used for all statistical analyses.
Sexual behavior
Standard parameters for sexual performance (latencies to mount, intromission, ejaculation, and numbers of mounts and intromissions) were each compared between groups using a Fisher exact test. Data analysis of sexual behavior before CSA revealed no significant differences between groups in any of the parameters of sexual performance.
For the CSA paradigm, percentages of males that displayed one or more mounts, intromissions, or ejaculation during a conditioning trial were calculated and compared with conditioning trial 1 using a Fisher exact test at a significance level of 0.05. In addition, and in experiment 2, differences between groups within each of the conditioning trials were compared using the Fisher exact test.
pERK cell counts
Single-labeled DAB-stained cells for pERK were counted in the nucleus accumbens (NAc) core and shell subregions, the caudate putamen (CPu), basolateral amygdala (BLA), lateral amygdala (LA), central amygdala (CeA), the OFC, the anterior cingulate area (ACA), prelimbic (PL), and infralimbic (IL) subregions of the medial prefrontal cortex (mPFC), and the ventral tegmental area (VTA) using standard areas of analysis using a camera lucida drawing tube attached to a Leica DMRD microscope (Leica Microsystems, Wetzlar, Germany). Standard areas of analysis were: 400 × 600 μm in the NAc core and shell subregions and LA, 800 × 800 μm in the CPu, 600 × 600 μm in the lateral OFC, 600 × 800 μm in the BLA, CeA, and each subregion of the mPFC, and 800 × 1200 μm in the VTA (Supplementary Figure 1). Two sections were counted for each brain region. Cell numbers per mm2 were calculated, and the two counts were averaged per animal. Group averages were compared using a Student’s t-test at significance level of 0.05 (experiment 1) or two way ANOVA (factors: Meth vs saline, and home cage control vs female exposure; experiment 2) and Holm–Sidak pairwise post-hoc comparisons at a significance level of 0.05 (experiment 2).
pERK/CAMKII or pERK/GAD
In fluorescent-stained sections from experiment 2, cells labeled for pERK, CaMKII or GAD67, or dual were counted in standard areas of analysis as described above in experiment 2 for immunoperoxidase-stained sections in ACA, IL, PL, lateral OFC and an additional counting area ventral within the lateral OFC (600 × 600 μm), using MicroBrightField Neurolucida software (Williston, Vermont, USA). The percentages of co-expression were calculated per section and averaged per animal. Group averages were compared using two way ANOVA (factors: Meth vs saline and home cage control vs female exposure) and Holm–Sidak pairwise post-hoc comparisons at a significance level of 0.05.
RESULTS
Neural Activation of the Limbic System Following CSA
In experiment 1, conditioning for sex aversion significantly inhibited sexual behavior (Table 1 and Figure 1a), confirming our previous findings (Davis et al, 2010; Frohmader et al, 2011). Specifically, decreased percentages of LiCl-paired males that displayed mounts, intromissions, or ejaculations were first evident on the fourth conditioning day and persisted through conditioning days 5–7 (Table 1).
Table 1. Percentages of Males (n=11) in Experiment 1 Displaying One or More Mount, Intromission, or Ejaculation During Each of Seven Conditioning Sessions of the CSA Paradigm.
| CD1 | CD2 | CD3 | CD4 | CD5 | CD6 | CD7 | |
|---|---|---|---|---|---|---|---|
| % Mounting | 100 | 100 | 88.9 | 66.7a | 66.7a | 27.8a | 61.1a |
| % Intromitting | 100 | 94.4 | 88.9 | 66.7a | 66.7a | 27.8a | 61.1a |
| % Ejaculating | 94.4 | 94.4 | 88.9 | 44.4a | 50a | 11.1a | 33.3a |
Indicates significant difference from CD1 (Fisher exact; CD: conditioning day): CD4: M and I: p=0.02, E; p=0.003; CD5: M and I: p=0.02, E: p=0.007; CD6: M, I, E: p⩽0.001; and CD7: M and I: p=0.008, E; p⩽0.001.
Figure 1.
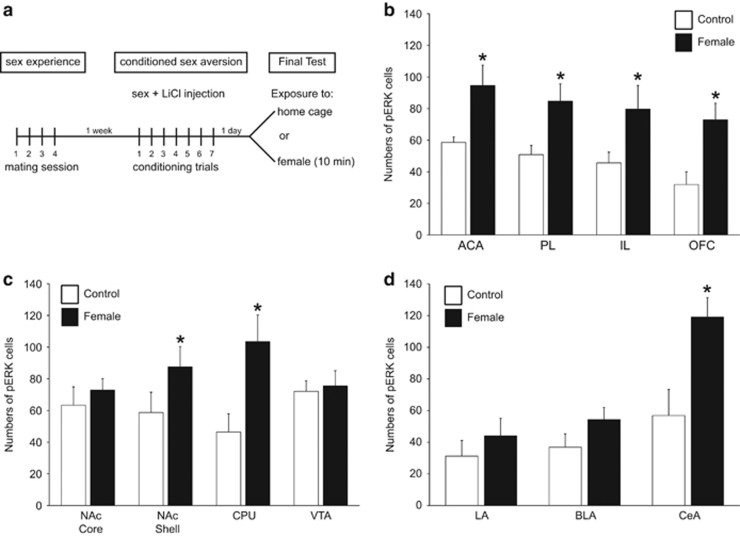
Conditioned aversive cue-induced phosphorylated MAP kinase (pERK) after conditioned sex aversion (CSA) (experiment 1). (a) Experimental design and timeline of experiment 1 showing 4 days of sex experience, followed 1 week later by 7 conditioning trials of CSA, and the final exposure to home cage or female for pERK analysis. (b–d) Numbers of pERK-immunoreactive neurons per mm2 (mean±SEM) in control males removed from home cage (control, white bars, n=5) or males exposed to the conditioned aversive cues, ie females (female, black bars, n=6). Cell counts are shown for (b) subregions of the medial prefrontal cortex (mPFC) and orbitofrontal cortex (OFC) (ACA, anterior cingulate area; PL, prelimbic cortex; IL, infralimbic cortex); (c) striatum and VTA (NAc, nucleus accumbens Core and Shell; CPU, caudate putamen; VTA, ventral tegmental area), and (d) amygdala (LA, lateral amygdala; BLA, basolateral amygdala; CeA, central amygdala). *Significant difference from home cage control group.
Analysis of numbers of pERK cells in animals from experiment 1 revealed that exposure to the conditioned cue associated with sex aversion (ie, the scented female) significantly increased pERK expression in regions of the frontal cortex, striatum, and amygdala (Figure 1b–d and Figure 2). Specifically, exposure to the scented female increased pERK expression in the ACA, PL, and IL regions of the mPFC (Figure 1b; p=0.037, p=0.036, p=0.047), OFC (p=0.020), NAc shell (p=0.045), CPu (p=0.032), and CeA (p=0.023) compared with home cage controls (Figure 1c and d). In contrast, induction of pERK immunoreactivity was not observed in the NAc core, in the lateral and basolateral amygdala, or in the ventral tegmental area (Figure 1c and d).
Figure 2.
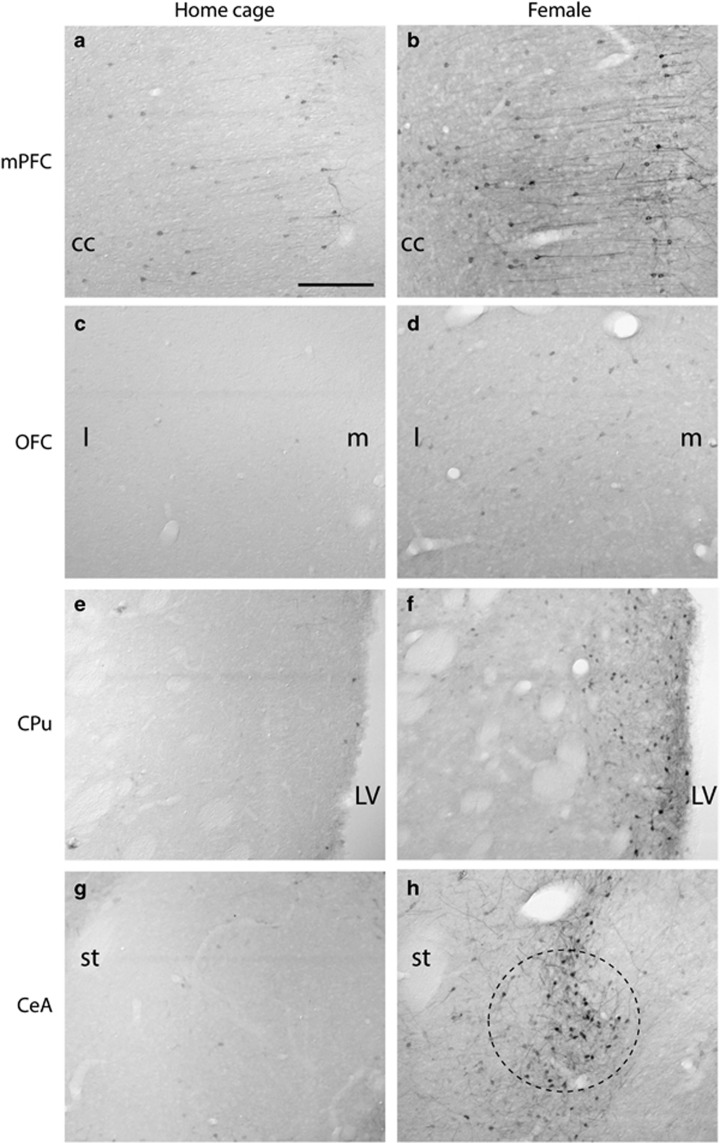
Representative images of pERK expression in home cage control males (a,c,e,g) and following exposure to the conditioned aversive cue, ie, the female (b,d,f,h) in experiment 1. CeA, central amygdala; cc, corpus callosum; CPu, caudate putamen; LV, lateral ventricle; mPFC, medial prefrontal cortex; OFC, orbitofrontal cortex (l: lateral; m: medial); st, stria terminalis. Circle in h indicates location of CeA. Scale bar indicates 200 μm.
Meth and Sex Pretreatment Attenuates Neural Activation Following CSA and Increases Baseline Neural Activation
In experiment 2, concurrent Meth and sex experience attenuated CSA (Table 2 and Figure 3a) confirming our previous observations (Frohmader et al, 2010a, 2011). Significantly higher percentages of males pre-exposed to Meth and sex displayed mounts, intromissions, and ejaculations compared with males treated with saline and sex (Table 2). Group differences were first observed on the fifth conditioning day, and persisted through conditioning days 6, 7, and 8. In addition, saline and sex-pretreated animals showed a significant inhibition on and after conditioning day 6, whereas Meth and sex-pretreated animals showed significant inhibition only during conditioning day 8 (Table 2).
Table 2. Percentages of Males Displaying Mating Behavior During Sex Aversion Conditioning After Saline (n=26) or Meth (n=27) Pretreatment in Experiment 2.
| CD1 | CD2 | CD3 | CD4 | CD5 | CD6 | CD7 | CD8 | |
|---|---|---|---|---|---|---|---|---|
| Saline | ||||||||
| % Mounting | 92.3 | 96.2 | 92.3 | 84.6 | 73.1 | 42.3a | 53.8a | 46.2a |
| % Intromitting | 92.3 | 96.2 | 92.3 | 84.6 | 73.1 | 42.3a | 53.8a | 46.2a |
| % Ejaculating | 84.6 | 80.8 | 92.3 | 76.9 | 61.5 | 34.6a | 46.2a | 34.6a |
| Meth | ||||||||
| % Mounting | 92.6 | 100 | 92.6 | 100 | 96.3b | 100b | 88.9b | 66.7a |
| % Intromitting | 88.9 | 100 | 92.6 | 100 | 92.6 | 96.3b | 88.9b | 66.7 |
| % Ejaculating | 81.5 | 85.2 | 92.6 | 92.6 | 66.7 | 81.5b | 81.5b | 55.6a |
Indicates significant difference from CD1 within group (Fisher exact; CD: conditioning day): CD6 saline, M, I, and E: p<0.001; CD7 saline, M: p=0.004, I: p=0.004, E: p=0.008; CD8 saline, M, I, and E: p<0.001; CD8 Meth, M: p=0.039, E: p=0.047.
Indicates significant difference from saline-pretreated males for each conditioning day (Fisher exact): CD5, M: p=0.024; CD6, M, I, and E: p<0.001; CD7, M and I: p=0.006, E: p=0.010.
Figure 3.
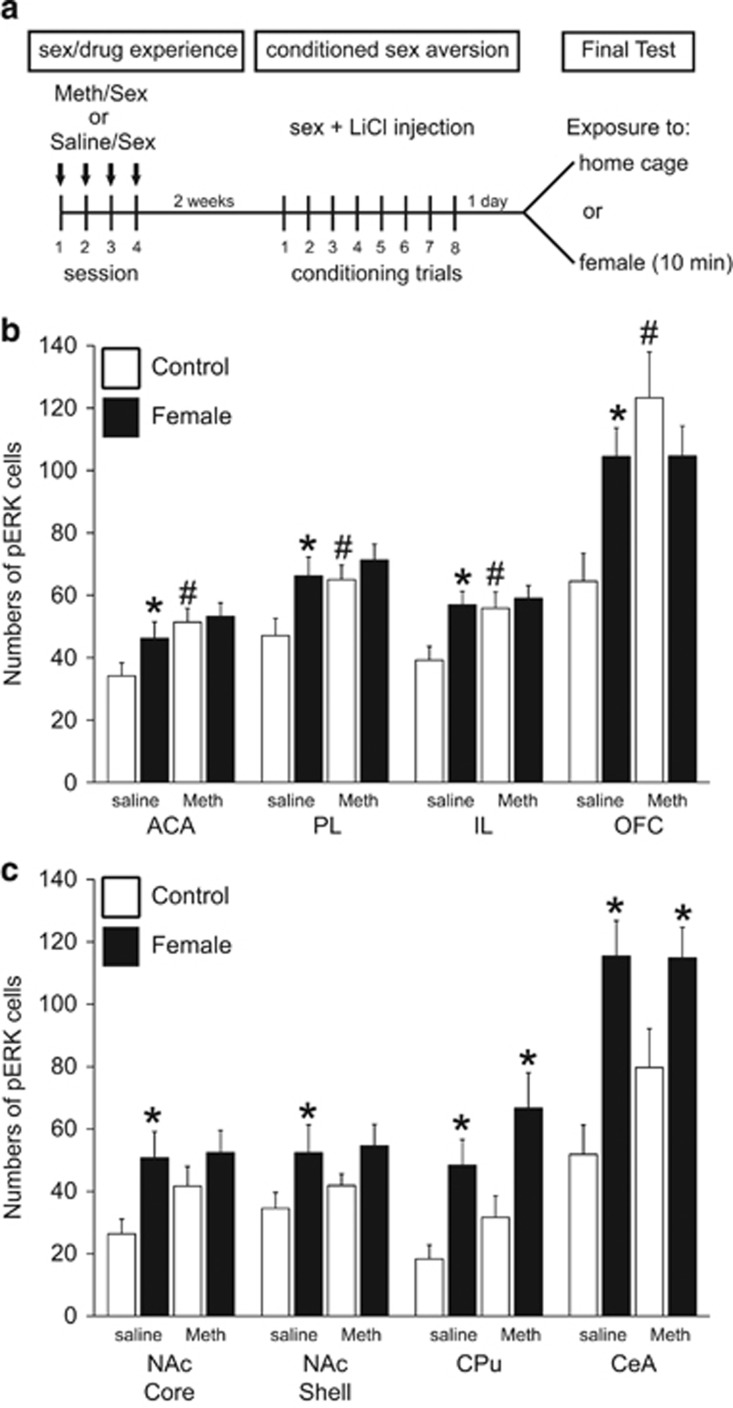
Effect of Meth/sex pretreatment on conditioned cue-induced pERK. (a) Experimental design and timeline of experiment 2, showing 4 days of Saline/sex or Meth/sex treatment, followed 2 weeks later by 8 conditioning trials of conditioned sex aversion (CSA), and the final exposure to home cage or female for phosphorylated MAP kinase (pERK) analysis. (b,c) Numbers of pERK-immunoreactive neurons per mm2 (mean±SEM) in males pretreated with Saline/sex (saline), or Meth/sex (Meth) and removed from home cage (control; white bars; n=12 saline and n=10 Meth) or exposed to the female (female; black bars; n=14 saline and n=17 Meth). (b) pERK in mPFC subregions and OFC, and (c) in NAc, CPU and CeA. ACA, anterior cingulate area; PL, prelimbic cortex; IL, infralimbic cortex; OFC, orbitofrontal cortex; NAc, nucleus accumbens; CPU, caudate putamen; VTA, ventral tegmental area; LA, lateral amygdala; BLA, basolateral amygdala; CeA, central amygdala. *Significant difference from home cage control group, #Significant difference from Saline/sex-pretreated home cage control group.
Analysis of pERK in animals from experiment 2 showed that there were main effects of exposure to the conditioned aversive stimulus on pERK in the PL and IL subregions of the mPFC (Figure 3; F(1,49)=5.1 and 5.2; p=0.028 and 0.027), NAc core (F(1,47)=5.8; p=0.020) and shell (F(1,47)=4.5; p=0.039), CeA (F(1,49)=20.3; p<0.001), and CPu (F(1,49)=12.3; p<0.001). In addition, there was a main effect of Meth pretreatment in all subregions of the mPFC (Figure 3b; F(1,49)=1.1–4.209; p=0.014–0.046), and OFC (F(1,48)=7.7; p=0.008), and an interaction in OFC (F(1,48)=7.6; p=0.008).
Post-hoc analyses showed that in saline-pretreated males, exposure to the female increased pERK in ACA, (p=0.040), PL (p=0.010) and IL (p=0.004), OFC (p=0.005), CeA (p<0.001), NAc core (p=0.010) NAc shell (p=0.041), and CPu (p=0.013) compared with saline-pretreated home cage controls (Figure 3b and c), confirming the findings of experiment 1. This pattern of activation of pERK by conditioned aversive cues was different in males that received pretreatment of Meth and sex experience, as exposure to the aversive cues did not induce pERK in the NAc core or shell, mPFC subregions, or OFC compared with Meth-pretreated home cage controls. In contrast, cue exposure in Meth-pretreated males did induce pERK in CPu (p=0.011) and CeA (p=0.029) compared with Meth-treated home cage controls (Figure 3c).
In addition, Meth pretreatment increased baseline (home cage) pERK expression in the mPFC (p=0.022, p=0.042 and 0.021; ACA, PL, and IL) and OFC (p<0.001) compared with saline-treated home cage controls (Figure 3b). This effect did not reach statistical significance in the NAc core and shell, CPu or CeA (Figure 3c).
To rule out effects of exposure to the CSA paradigm on this increased baseline pERK expression, experiment 2 was replicated in a separate study in which males received concurrent or non-concurrent Meth and sex exposure without testing in the CSA paradigm (Supplementary Materials: experiment 3). In this separate study, concurrent Meth and sex pretreatment increased pERK expression under baseline conditions (Supplementary Figure 2), replicating the findings of experiment 2. Therefore, exposure to CSA paradigm was not the cause for the baseline increase. Moreover, this effect was not evident in males that received non-concurrent Meth and sex experience (Supplementary Figure 2), indicating that concurrent Meth and sex experience is essential for this alteration in baseline activity and that the effect appears to be synergistic, rather than additive. Finally, supplementary analysis of pERK was conducted in experiment 2 in the portion of saline-treated animals that showed higher rates of initiation (ie, persistence) of mating behavior despite learned negative consequences (Supplementary Data, analysis experiment 2), confirming an increased pERK baseline expression in animals that show maladaptive sex behavior without Meth pretreatment (Supplementary Table 3).
pERK Expression in CaMKII, But Not GAD65 Cells
To next determine if pERK is expressed in mPFC and OFC pyramidal neurons or interneurons, analysis of dual fluorescent-stained sections for pERK and CaMKII (expressed in pyramidal cells (Fanous et al, 2012)) or GAD (expressed in interneurons (Fanous et al, 2012)) was performed. First, the findings above for pERK expression were confirmed using the fluorescent analysis (Figure 4a: Main effects of Meth: ACA: F(1,11)=5.8; p=0.034, PL: F(1,10)=18.7; p=0.002, IL: F(1,10)=8.6; p=0.015, OFC: F(1,14)=10.8; p=0.005; Main effect of female: OFC: F(1,14)=6.5, p=0.023; and interaction effects: ACA: F(1,11)=7.4, p=0.020; PL: F(1,10)=6.6, p=0.028; IL: F(1,10)=8.6, p=0.015; OFC: F(1,14)=16.5, p=0.001). Post-hoc analyses showed that exposure to the female increased pERK in saline-pretreated control males compared with home cage controls (Figure 4a; ACA: p=0.027; PL: p=0.033; IL: p=0.045; OFC: p=0.002). Meth-pretreated males had increased pERK expression under baseline conditions (ACA: p=0.009, PL: p=0.001, IL: p=0.004, OFC: p=0.001 compared with saline home cage controls) and exposure to the female did not induce further pERK expression (Figure 4a).
Figure 4.
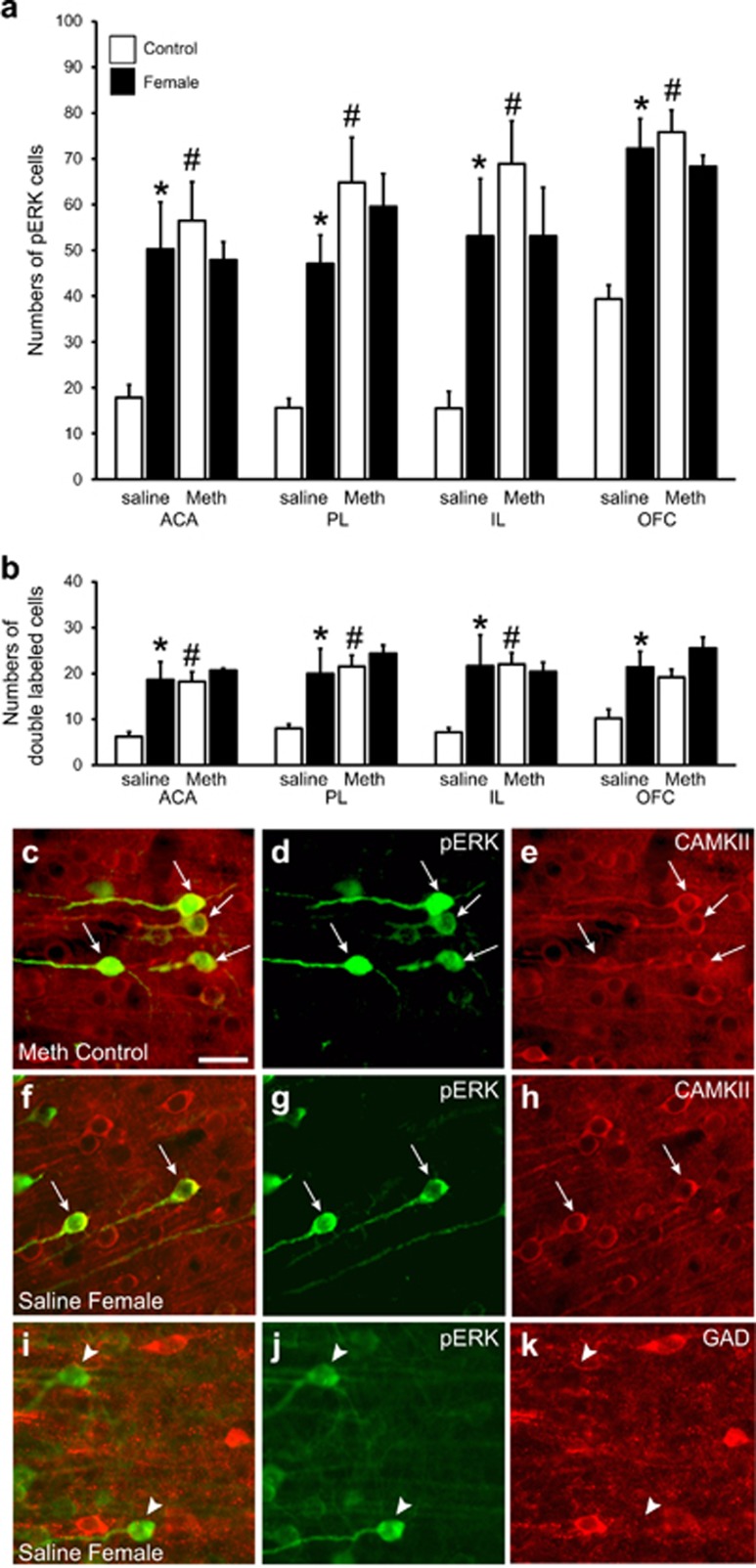
Phosphorylated MAP kinase (pERK) is localized in CaMKII neurons in medial prefrontal cortex (mPFC) and orbitofrontal cortex (OFC). (a) Numbers of pERK-immunofluorescent neurons per mm2 (mean±SEM) in mPFC subregions (ACA, PL, IL) and OFC in males (from experiment 2) pretreated with Saline/sex (saline), or Meth/sex (Meth) and removed from home cage (control; white bars; n=4 saline and n=4 Meth) or exposed to the female (female; black bars; n=4 saline and n=3 Meth). (b) Numbers of neurons co-labeled for pERK and CaMKII per mm2 (mean±SEM). (c–k) Representative images illustrating the co-localization (c, f; arrows indicate dual labeled neurons) of pERK (green; d,g,j) and CaMKII (red; e,h) and the lack of co-localization (i; arrow heads indicate pERK single-labeled neurons) of pERK (green; j) and GAD (red; k) in PL of representative Meth+Sex Home Cage control (c–e), or Saline/sex exposed to female (f–k) males. Scale bar indicates 50 μm.
Moreover, analysis of dual labeling showed that mPFC pERK was expressed in CaMKII neurons in all groups in both mPFC and OFC (Figure 4b and c–h). Analysis of pERK expression in CaMKII neurons mirrored that of pERK alone (Figure 4b; Main effects of Meth: F(1 : 10–14=5.3–18.1, p=0.002–0.045; Main effects of female: F(1 : 10–14)=8.7–12.3, p=0.005–0.028)). Post-hoc analyses showed that in the ACA, PL, and IL subregions of the mPFC, pERK was increased in CaMKII cells by exposure to the female in saline and sex-pretreated males (p=0.012, 0.007, 0.019), but not in Meth and sex-pretreated males. Meth and sex-pretreated males had significantly higher pERK expression in CaMKII cells in the ACA, PL, and IL subregions of the mPFC under baseline conditions compared with saline and sex-treated males (Figure 4b; p=0.015, 0.002, 0.010). In the OFC, pERK was increased in CaMKII cells by exposure to the female in saline and sex-pretreated males (p=0.046), but not in Meth and sex-pretreated males. Finally, in all groups pERK-immunoreactive cells were observed that were not co-labeled for CaMKII: 33–50% of pERK cells in mPFC subregions and 17–22% of pERK cells in OFC co-expressed CaMKII, regardless of home cage control or female exposure. However, single-labeled pERK cells did have the morphological appearance of pyramidal cells, suggesting that CaMKII staining failed to identify all pyramidal neurons (Figure 4c–e and f–h). Finally, pERK cells did not co-express GAD (0–0.3%) in any of the groups (Figure 4i–k).
DISCUSSION
These findings indicate that concurrent Meth and sex experience may cause maladaptive sexual behavior by increasing baseline neural activity in mPFC and OFC, and by attenuating neural activation in mPFC, OFC, and NAc induced by conditioned cues signaling aversive outcomes of sex behavior. These findings provide new insights into the neural circuits that are affected in drug-associated hypersexuality. Moreover, neural activity changes occurred in CaMKII neurons in the mPFC and OFC and may thus influence the efferent connections of these cortical areas.
First, it was demonstrated that exposure to cues associated with conditioned inhibition of sex behavior resulted in significant pERK activation in mPFC subregions, OFC, NAc, CPu, and CeA. This neural activation is distinct from that induced by exposure to a receptive female or mating behavior (Veening and Coolen, 2014) and thus appears to be involved in conditioned inhibition rather than initiation of sex behavior. The current findings expand previous reports showing pERK in NAc and mPFC during expression and acquisition of conditioned taste aversion, respectively (Marotta et al, 2014). The current findings suggest that mPFC is involved in the expression of CSA, having an extended role in regulation of mating inhibition beyond a role in learning (Calu et al, 2013; Riga et al, 2014; Rosen et al, 2015). Indeed, lesions of the mPFC result in expression of maladaptive sex behavior, without disruption of aversive learning or the expression of sexual behavior and reward (Davis et al, 2010).
Moreover, the current findings show that Meth and sex experience prevented the induction of neural activation by CSA-related cues in the mPFC and OFC, further demonstrating the potential involvement of these frontal cortices in the effects of Meth and sex experience on maladaptive sex behavior. This is consistent with roles for OFC and mPFC in cue-outcome prediction and expectancy (OFC) (Schoenbaum and Shaham, 2008), and decision making and associative learning (mPFC) (Riga et al, 2014).
Imaging studies using functional fMRI in humans with compulsive sexual behavior have shown dorsal anterior cingulate area activation by exposure to sexually explicit cues, suggesting a disruption of PFC functionality (Voon et al, 2014). This disruption is associated with enhanced attentional bias towards sexually explicit cues (Mechelmans et al, 2014), consistent with a role for PFC in attentional shifting (Morris et al, 2016). Moreover, this disruption mostly affected frontal-striatal circuitries (Mechelmans et al, 2014; Morris et al, 2016). In the current study, Meth and sex experience also significantly attenuated pERK induction by conditioned cues in the NAc, an area critical for goal-directed behavior (NAc) (Cruz et al, 2014; Penner and Mizumori, 2012), but not in the CPu, an area involved in acquisition and storage of motor habits (Atallah et al, 2007; Balleine et al, 2009). Therefore, in rat, maladaptive sex behavior associated with Meth/sex experience appears to affect a frontal-striatal substrate that includes mPFC, OFC, and NAc. Finally, CeA expressed pERK induced by aversive conditioned cues, but was not altered by Meth/sex experience, consistent with the finding that conditioned aversion learning per se is not disrupted by Meth/sex experience (Frohmader et al, 2011). Instead, pERK in the CeA may be reflective of conditioned stress responses (Phelps and LeDoux, 2005).
A third main finding of the present study was the chronically increased baseline pERK expression after Meth/sex pre-exposure. This effect was dependent on concurrent exposure to Meth/sex and associated with maladaptive sex behavior even in animals that were not pretreated with Meth. It is currently unclear what the exact consequences are of the increased baseline ERK activity, but pERK was expressed predominantly in CaMKII-expressing neurons, thereby presumably influencing mPFC output. The mPFC in turn suppresses dopamine transmission in the NAc thereby modulating motivated behaviors (Deisseroth, 2014). Hence, dysfunction of the mPFC output may contribute to altered dopamine function in the NAc, disrupted expression of learned responses, and increasing maladaptive goal-directed behaviors. Finally, the causes of the chronic elevation in ERK activation are unknown. Drugs alter neurotransmitter signaling in the mPFC, including serotonin (Muller et al, 2007), dopamine (Wise, 1998), and glutamate (Kalivas, 2009), as well as growth factors like BDNF (Grimm et al, 2003), but the role of these neurotransmitters in the effects of concurrent Meth and sex on maladaptive sex behavior are unknown. Finally, increased baseline pERK expression may have occurred in the same mPFC and OFC neurons as those responding to aversive cues, thereby contributing to the attenuation of further activation of these neurons and subsequent disruption of behavioral inhibition.
Together, these findings demonstrate a discrete neural substrate involved in inhibition of sexual behavior, which is functionally altered in animals with maladaptive sex behavior triggered by concurrent Meth and sex experience. Chronic alterations of ERK activity appear to be critical to these effects. Previous studies have shown that MEK inhibitors can prevent cue-induced drug seeking and increase extinction learning (Lu et al, 2006; Papale et al, 2016; Valjent et al, 2006a). Therefore, MEK inhibitors may be promising avenues for treatment of drug-induced compulsive sex behavior.
Funding and disclosure
This research was supported by a grant from the Intramural Research Support Program at the University of Mississippi Medical Center. The authors declare no conflict of interest.
Acknowledgments
We thank Mr S. Rollins and Ms T. Smith for their technical support.
Footnotes
Supplementary Information accompanies the paper on the Neuropsychopharmacology website (http://www.nature.com/npp)
Supplementary Material
References
- Agmo A (2002). Copulation-contingent aversive conditioning and sexual incentive motivation in male rats: evidence for a two-stage process of sexual behavior. Physiol Behav 77: 425–435. [DOI] [PubMed] [Google Scholar]
- Atallah HE, Lopez-Paniagua D, Rudy JW, O’Reilly RC (2007). Separate neural substrates for skill learning and performance in the ventral and dorsal striatum. Nat Neurosci 10: 126–131. [DOI] [PubMed] [Google Scholar]
- Balleine BW, Liljeholm M, Ostlund SB (2009). The integrative function of the basal ganglia in instrumental conditioning. Behav Brain Res 199: 43–52. [DOI] [PubMed] [Google Scholar]
- Berman DE, Hazvi S, Rosenblum K, Seger R, Dudai Y (1998). Specific and differential activation of mitogen-activated protein kinase cascades by unfamiliar taste in the insular cortex of the behaving rat. J Neurosci 18: 10037–10044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calu DJ, Kawa AB, Marchant NJ, Navarre BM, Henderson MJ, Chen B et al (2013). Optogenetic inhibition of dorsal medial prefrontal cortex attenuates stress-induced reinstatement of palatable food seeking in female rats. J Neurosci 33: 214–226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cartier JJ, Greenwell L, Prendergast ML (2008). The persistence of HIV risk behaviors among methamphetamine-using offenders. J Psychoactive Drugs 40: 437–446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruz FC, Babin KR, Leao RM, Goldart EM, Bossert JM, Shaham Y et al (2014). Role of nucleus accumbens shell neuronal ensembles in context-induced reinstatement of cocaine-seeking. J Neurosci 34: 7437–7446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis JF, Loos M, Di Sebastiano AR, Brown JL, Lehman MN, Coolen LM (2010). Lesions of the medial prefrontal cortex cause maladaptive sexual behavior in male rats. Biol Psychiatry 67: 1199–1204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deisseroth K (2014). Circuit dynamics of adaptive and maladaptive behaviour. Nature 505: 309–317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fanous S, Goldart EM, Theberge FR, Bossert JM, Shaham Y, Hope BT (2012). Role of orbitofrontal cortex neuronal ensembles in the expression of incubation of heroin craving. J Neurosci 32: 11600–11609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frohmader KS, Bateman KL, Lehman MN, Coolen LM (2010. a). Effects of methamphetamine on sexual performance and compulsive sex behavior in male rats. Psychopharmacology 212: 93–104. [DOI] [PubMed] [Google Scholar]
- Frohmader KS, Lehman MN, Laviolette SR, Coolen LM (2011). Concurrent exposure to methamphetamine and sexual behavior enhances subsequent drug reward and causes compulsive sexual behavior in male rats. J Neurosci 31: 16473–16482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frohmader KS, Pitchers KK, Balfour ME, Coolen LM (2010. b). Mixing pleasures: review of the effects of drugs on sex behavior in humans and animal models. Horm Behav 58: 149–162. [DOI] [PubMed] [Google Scholar]
- Frohmader KS, Wiskerke J, Wise RA, Lehman MN, Coolen LM (2010. c). Methamphetamine acts on subpopulations of neurons regulating sexual behavior in male rats. Neuroscience 166: 771–784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giorgi O, Corda MG, Sabariego M, Giugliano V, Piludu MA, Rosas M et al (2015). Differential effects of cocaine on extracellular signal-regulated kinase phosphorylation in nuclei of the extended amygdala and prefrontal cortex of psychogenetically selected Roman high- and low-avoidance rats. J Neurosci Res 93: 714–721. [DOI] [PubMed] [Google Scholar]
- Green AI, Halkitis PN (2006). Crystal methamphetamine and sexual sociality in an urban gay subculture: an elective affinity. Cult Health Sex 8: 317–333. [DOI] [PubMed] [Google Scholar]
- Grimm JW, Lu L, Hayashi T, Hope BT, Su TP, Shaham Y (2003). Time-dependent increases in brain-derived neurotrophic factor protein levels within the mesolimbic dopamine system after withdrawal from cocaine: implications for incubation of cocaine craving. J Neurosci 23: 742–747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harlow E, Lane D (1988) Antibodies, A Laboratory Manual. Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA. [Google Scholar]
- Johnson MW, Bruner NR (2012). The sexual discounting task: HIV risk behavior and the discounting of delayed sexual rewards in cocaine dependence. Drug Alcohol Depend 123: 15–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson MW, Johnson PS, Herrmann ES, Sweeney MM (2015). Delay and probability discounting of sexual and monetary outcomes in individuals with cocaine use disorders and matched controls. PLoS One 10: e0128641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalivas PW (2009). The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci 10: 561–572. [DOI] [PubMed] [Google Scholar]
- Kraus SW, Voon V, Potenza MN (2016). Neurobiology of compulsive sexual behavior: emerging science. Neuropsychopharmacology 41: 385–386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longoni R, Spina L, Vinci S, Acquas E (2011). The MEK inhibitor SL327 blocks acquisition but not expression of lithium-induced conditioned place aversion: a behavioral and immunohistochemical study. Psychopharmacology 216: 63–73. [DOI] [PubMed] [Google Scholar]
- Lu L, Koya E, Zhai H, Hope BT, Shaham Y (2006). Role of ERK in cocaine addiction. Trends Neurosci 29: 695–703. [DOI] [PubMed] [Google Scholar]
- Lyons T, Chandra G, Goldstein J, Ostrow DG (2010). Breaking the bond between stimulant use and risky sex: a qualitative study. Subst Abus 31: 224–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marotta R, Fenu S, Scheggi S, Vinci S, Rosas M, Falqui A et al (2014). Acquisition and expression of conditioned taste aversion differentially affects extracellular signal regulated kinase and glutamate receptor phosphorylation in rat prefrontal cortex and nucleus accumbens. Front Behav Neurosci 8: 153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mastroleo NR, Operario D, Barnett NP, Colby SM, Kahler CW, Monti PM (2015). Prevalence of heavy drinking and risky sexual behaviors in adult emergency department patients. Alcohol Clin Exp Res 39: 1997–2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mausbach BT, Semple SJ, Strathdee SA, Patterson TL (2009). Predictors of safer sex intentions and protected sex among heterosexual HIV-negative methamphetamine users: an expanded model of the theory of planned behavior. AIDS Care 21: 17–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mechelmans DJ, Irvine M, Banca P, Porter L, Mitchell S, Mole TB et al (2014). Enhanced attentional bias towards sexually explicit cues in individuals with and without compulsive sexual behaviours. PLoS One 9: e105476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell SG, Kelly SM, Brown BS, O’Grady KE, Schwartz RP (2012). HIV sex-risk behaviors among in- versus out-of-treatment heroin-addicted adults. Am J Drug Alcohol Abuse 38: 328–333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris LS, Kundu P, Dowell N, Mechelmans DJ, Favre P, Irvine MA et al (2016). Fronto-striatal organization: defining functional and microstructural substrates of behavioural flexibility. Cortex 74: 118–133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller CP, Carey RJ, Huston JP, De Souza Silva MA (2007). Serotonin and psychostimulant addiction: focus on 5-HT1A-receptors. Prog Neurobiol 81: 133–178. [DOI] [PubMed] [Google Scholar]
- Papale A, Morella IM, Indrigo MT, Eugene Bernardi R, Marrone L, Marchisella F et al (2016). Impairment of cocaine-mediated behaviours in mice by clinically relevant Ras-ERK inhibitors. Elife 5: e17111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penner MR, Mizumori SJ (2012). Neural systems analysis of decision making during goal-directed navigation. Prog Neurobiol 96: 96–135. [DOI] [PubMed] [Google Scholar]
- Peters RH (1983). Learned aversions to copulatory behaviors in male rats. Behav Neurosci 97: 140–145. [DOI] [PubMed] [Google Scholar]
- Phelps EA, LeDoux JE (2005). Contributions of the amygdala to emotion processing: from animal models to human behavior. Neuron 48: 175–187. [DOI] [PubMed] [Google Scholar]
- Pitchers KK, Coppens CM, Beloate LN, Fuller J, Van S, Frohmader KS et al (2014). Endogenous opioid-induced neuroplasticity of dopaminergic neurons in the ventral tegmental area influences natural and opiate reward. J Neurosci 34: 8825–8836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ribeiro MJ, Schofield MG, Kemenes I, O’Shea M, Kemenes G, Benjamin PR (2005). Activation of MAPK is necessary for long-term memory consolidation following food-reward conditioning. Learn Mem 12: 538–545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riga D, Matos MR, Glas A, Smit AB, Spijker S, Van den Oever MC (2014). Optogenetic dissection of medial prefrontal cortex circuitry. Front Syst Neurosci 8: 230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen LG, Sun N, Rushlow W, Laviolette SR (2015). Molecular and neuronal plasticity mechanisms in the amygdala-prefrontal cortical circuit: implications for opiate addiction memory formation. Front Neurosci 9: 399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafe GE, Atkins CM, Swank MW, Bauer EP, Sweatt JD, LeDoux JE (2000). Activation of ERK/MAP kinase in the amygdala is required for memory consolidation of pavlovian fear conditioning. J Neurosci 20: 8177–8187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoenbaum G, Shaham Y (2008). The role of orbitofrontal cortex in drug addiction: a review of preclinical studies. Biol Psychiatry 63: 256–262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semple SJ, Patterson TL, Grant I (2004). The context of sexual risk behavior among heterosexual methamphetamine users. Addict Behav 29: 807–810. [DOI] [PubMed] [Google Scholar]
- Takamura H, Ichisaka S, Watanabe K, Toigawa M, Hata Y (2008). Effects of anesthesia on immunohistochemical detection of phosphorylated extracellular signal-regulated kinase in cerebral cortex. J Neurosci Methods 170: 300–304. [DOI] [PubMed] [Google Scholar]
- Valjent E, Corbille AG, Bertran-Gonzalez J, Herve D, Girault JA (2006. a). Inhibition of ERK pathway or protein synthesis during reexposure to drugs of abuse erases previously learned place preference. Proc Natl Acad Sci USA 103: 2932–2937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valjent E, Corvol JC, Trzaskos JM, Girault JA, Herve D (2006. b). Role of the ERK pathway in psychostimulant-induced locomotor sensitization. BMC Neurosci 7: 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veening JG, Coolen LM (2014). Neural mechanisms of sexual behavior in the male rat: emphasis on ejaculation-related circuits. Pharmacol Biochem Behav 121: 170–183. [DOI] [PubMed] [Google Scholar]
- Voon V, Mole TB, Banca P, Porter L, Morris L, Mitchell S et al (2014). Neural correlates of sexual cue reactivity in individuals with and without compulsive sexual behaviours. PLoS One 9: e102419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wise RA (1998). Drug-activation of brain reward pathways. Drug Alcohol Depend 51: 13–22. [DOI] [PubMed] [Google Scholar]
- Zapata LB, Hillis SD, Marchbanks PA, Curtis KM, Lowry R (2008). Methamphetamine use is independently associated with recent risky sexual behaviors and adolescent pregnancy. J Sch Health 78: 641–648. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.