Abstract
Background
Cardiopulmonary injury is common in neonatal encephalopathy, but the link with cerebrovascular dysfunction is unknown. We hypothesized that cerebral autoregulation is associated with cardiopulmonary injury in neonates treated with therapeutic hypothermia (TH) for neonatal encephalopathy.
Methods
The cerebral hemoglobin volume index (HVx) from near-infrared spectroscopy was used to identify the mean arterial blood pressure (MAP) with optimal autoregulatory vasoreactivity (MAPOPT). We measured associations between MAP relative to MAPOPT and indicators of cardiopulmonary injury (duration of mechanical respiratory support and administration of inhaled nitric oxide (iNO), milrinone, or steroids).
Results
We identified associations between cerebrovascular autoregulation and cardiopulmonary injury that were often sex-specific. Greater MAP deviation above MAPOPT was associated with shorter duration of intubation in boys but longer ventilatory support in girls. Greater MAP deviation below MAPOPT related to longer intensive care stay in boys. Milrinone was associated with greater MAP deviation below MAPOPT in girls.
Conclusion
MAP deviation from MAPOPT may relate to cardiopulmonary injury after neonatal encephalopathy, and sex may modulate this relationship. Whereas MAP above MAPOPT may protect the brain and lungs in boys, it may be related to cardiopulmonary injury in girls. Future studies are needed to characterize the role of sex in these associations.
INTRODUCTION
Neonatal encephalopathy affects ~3 in 1000 births in the United States.(1, 2) Neonatal encephalopathy causes multi-organ disease that includes cardiopulmonary injury in 80% of cases despite the use of therapeutic hypothermia (TH).(1, 3, 4) Supporting cerebral autoregulation, the physiologic mechanism that maintains stable cerebral blood flow across a range of blood pressure, may protect the post-ischemic neonatal brain.(5–8) Dysfunctional cerebral autoregulation is linked to pulmonary injury during hypothermic cardiopulmonary bypass(9) and longer duration of post-operative mechanical ventilation in adults.(10) Whether strategies that support cerebral autoregulation also protect the cardiopulmonary system in neonates with neonatal encephalopathy is unknown.
The hemoglobin volume index (HVx) measures cerebral autoregulatory vasoreactivity using near infrared spectroscopy (NIRS). HVx is calculated by a continuous, moving correlation coefficient between mean arterial pressure (MAP) and the NIRS relative total tissue hemoglobin (rTHb), a surrogate measure of cerebral blood volume.(11, 12) Because the rTHb optical density is calculated as the sum of oxygenated and deoxygenated hemoglobin,(12) changes in tissue oxygen supply and metabolic rate during TH and mechanical ventilation affect HVx less than indices solely derived from oxyhemoglobin. HVx can be used to identify the range of MAP with most robust autoregulatory vasoreactivity—the optimal mean arterial blood pressure (MAPOPT)—after neonatal encephalopathy.(5, 7, 13) Blood pressure deviation below MAPOPT after neonatal encephalopathy is linked to greater brain injury on magnetic resonance imaging (MRI) at 2 weeks (5, 7) and worse neurocognitive outcomes at 2 years.(14)
Here, we investigated whether blood pressure deviation from MAPOPT is associated with cardiopulmonary injury after neonatal encephalopathy and TH. Our objective was to study the associations between deviation from MAPOPT and durations of cardiopulmonary support and stay in the neonatal intensive care unit (NICU). We analyzed all neonates and stratified the data by sex based on increasing evidence that responses to neonatal encephalopathy are sex-specific.(15–20)
METHODS
The Johns Hopkins University (JHU) Institutional Review Board (IRB) approved this study. Between September 2010 and July 2015, neonates admitted to the NICU for neonatal encephalopathy were screened using criteria reported previously.(5, 7, 14) Criteria for neonatal encephalopathy requiring TH were based on the NICHD Neonatal Research Network’s clinical trial.(21) We obtained written informed consent from parents of participants until May 2013. After that, NIRS monitoring became standard of care for neonatal encephalopathy at JHU NICU, and the IRB waived the requirement for written consent.
Clinical care
The JHU NICU whole-body TH protocol was followed as previously published.(5, 7, 14) The treating team was blinded to HVx and made all clinical decisions. For persistent pulmonary hypertension of the newborn (PPHN), the primary mode of ventilation was high-frequency oscillatory ventilation. Clinicians initiated inhaled nitric oxide (iNO) (INOmax, Mallinckrodt pharmaceuticals, St Louis, MO) by endotracheal tube for the clinical diagnosis of PPHN based on persistently low PaO2 and oxygenation index >25 despite a FiO2 of 1.0 and lung recruitment strategies. PPHN was confirmed by echocardiogram. Per clinical protocol, the iNO was started at 20 ppm and weaned down once the goal PaO2 could be sustained with FiO2≤0.60. Dopamine was the first-line inotropic agent (maximum dose 20 mcg/kg/min), and clinicians determined the neonates’ hemodynamic goals. Milrinone infusion was used for ventricular dysfunction diagnosed by echocardiogram and as part of PPHN management. All neonates received morphine intravenously (IV) per NICU TH protocol. Some neonates also received benzodiazepines or additional opiates. Occasionally clonidine was administered for shivering, and vecuronium was given for neuromuscular blockade when needed. Early hydrocortisone (20 mg/m2 IV) was used for arterial hypotension refractory to inotropes and/or adrenal insufficiency. Steroids were therefore considered a marker of severe systemic illness affecting multiple organ systems.
Autoregulation monitoring
Our autoregulation monitoring methodology was previously published.(5, 7, 14) Briefly, neonatal cerebral oximetry probes were placed bilaterally on the forehead and connected to an INVOS 5100 NIRS machine (INVOS; Medtronic, Minneapolis, MN). Synchronous measurements of NIRS and arterial blood pressure signals at 100 Hz were processed with ICM+ software (Cambridge Enterprises, Cambridge, UK). We analyzed time-integrated, 10-second means of MAP and rTHb, a surrogate measure of cerebral blood volume, to filter out high-frequency waves from respiration and pulse. HVx is calculated by a continuous, moving Pearson correlation coefficient between MAP and rTHb from consecutive 10-second windows, and the average from each 300-second epoch is used to calculate one HVx value.(11, 12) HVx is a continuous variable that ranges from −1 to +1. When autoregulatory vasoreactivity is functional, HVx is negative or near-zero. A positive HVx that approaches 1 indicates dysfunctional autoregulation. Artifacts in the NIRS and MAP signals were manually filtered out, and data that comprised <1% of the recording were removed as an additional method to remove artifacts.(5, 7, 14) After ruling out unilateral intracranial lesions on MRI, right and left HVx values were averaged and sorted into 5-mmHg bins. The most negative HVx (nadir) was used to identify the MAPOPT with optimal autoregulatory vasoreactivity in each observation period (TH, rewarming, and first 6 hours of normothermia).(5, 7, 13, 14) Some neonates were coded as “unidentifiable MAPOPT” if a clear nadir could not be identified.(14) An investigator (JKL) blinded to outcome measures identified the MAPOPT values, which were corroborated by additional investigators (FJN, MG). We calculated an area under the curve (AUC) below MAPOPT to combine the extent of blood pressure deviation with the amount of time spent with blood pressure below MAPOPT. The AUC (min•mmHg/h) was calculated as time (minutes) spent with blood pressure below MAPOPT and blood pressure deviation (mmHg) below MAPOPT, and then normalized for the duration of monitoring (hours).(5, 7, 13, 14) In summary, the parameters derived from MAPOPT in each period were: (i) maximal blood pressure deviation below or above MAPOPT; (ii) duration of time spent with blood pressure below, at, or above MAPOPT as a percentage of the monitoring period; and (iii) AUC.
Demographic and perinatal data
Clinical data were retrieved from electronic medical record by investigators blinded to the autoregulation data (RC-V and MOC). (Table 1)(22)
Table 1.
Demographic and clinical characteristics of infants with an identified optimal mean arterial blood pressure.
| TOTAL | BOYS | GIRLS | P-value | ||||
|---|---|---|---|---|---|---|---|
| Demographic - antenatal data | n | n | n | ||||
| Gestational age, weeks (mean, SD) | 65 | 38 6/7 (16/7) | 38 | 38 6/7 (15/7) | 27 | 39 (16/7) | 0.61a |
| Birth weight, grams (mean, SD) | 65 | 3287 (622) | 38 | 3452 (660) | 27 | 3054 (486) | 0.007b* |
| Race, White (n, %) | 65 | 31 (48%) | 38 | 21 (55%) | 27 | 10 (37%) | 0.35c |
| Race, Black (n, %) | 65 | 24 (37%) | 38 | 12 (32%) | 27 | 12 (44%) | 0.50c |
| Ethnicity, Hispanic (n, %) | 65 | 8 (11%) | 38 | 3 (8%) | 27 | 5 (19%) | 0.32c |
| Adequate for gestational age (n, %) | 65 | 51 (78%) | 38 | 28 (74%) | 27 | 23 (85%) | 0.85c |
| In utero growth restriction (n, %) | 65 | 1 (1.5%) | 38 | 0 (0%) | 27 | 1 (3.7%) | 1.00 e |
| Maternal gestational diabetes (n, %) | 65 | 6 (9%) | 38 | 5 (13%) | 27 | 1 (4%) | 0.19c |
| Born at outside facility (n, %) | 65 | 57 (88%) | 38 | 34 (89%) | 27 | 23 (85%) | 0.60c |
| Delivery mode, c-section (n, %) | 65 | 50 (77%) | 38 | 27 (71%) | 27 | 23 (85%) | 0.18c |
| Emergency delivery (n, %) | 65 | 48 (74%) | 38 | 26 (68%) | 27 | 22 (82%) | 0.24c |
| Perinatal data | |||||||
| Apgar score, 1 min (median, IQR) | 65 | 1 (1–2) | 38 | 1 (1–3) | 27 | 1(0–2) | 0.04d* |
| Apgar score, 5 min (median, IQR) | 65 | 3 (2–6) | 38 | 4 (2–6) | 27 | 3 (2–4) | 0.11d |
| Cord pH (mean, SD) | 47 | 6.93 (0.17) | 28 | 6.93 (0.17) | 19 | 6.94 (0.19) | 0.95a |
| Cord base deficit (mean, SD) | 41 | −15.9 (7.4) | 23 | −15.8 (6.9) | 18 | −15.9 (8.3) | 0.96a |
| First hour pH (mean, SD) | 64 | 7.11 (0.15) | 38 | 7.11 (0.15) | 26 | 7.09 (0.14) | 0.45a |
| First hour base deficit (mean, SD) | 55 | −16.8 (5.4) | 34 | −16.6 (5.4) | 21 | −17.1 (5.5) | 0.69a |
| Admission Sarnat score (median, IQR) | 65 | 2 (2–2) | 38 | 2 (2–2) | 27 | 2 (2–2) | 0.78d |
| Severe encephalopathy (n, %) | 65 | 13 (20%) | 38 | 8 (21%) | 27 | 5 (19%) | 0.56c |
| Hemoglobin (g/dL) (mean, SD) | 63 | 15.5 (1.7) | 36 | 15.5 (1.5) | 27 | 15.5 (1.9) | 0.95a |
| Delta hemoglobin (g/dL) (mean, SD) | 59 | 2.5 (1.5) | 34 | 2.4 (1.5) | 25 | 2.6 (1.5) | 0.56a |
| Cardiopulmonary outcomes | |||||||
| Mech. ventilation, days (median, IQR) | 65 | 1 (1–7) | 38 | 1 (0–6) | 27 | 2 (1–7) | 0.74d |
| Pressure support, days (median, IQR) | 65 | 2 (1–7) | 38 | 1 (1–7) | 27 | 3 (1–7) | 0.19d |
| Oxygen support, days (median, IQR) | 65 | 9 (5–15) | 38 | 8 (5–15) | 27 | 10 (5–15) | 0.69d |
| PPHN (n, %) | 65 | 25 (39%) | 38 | 15 (40%) | 27 | 10 (37%) | 0.84c |
| iNO use (n, %) | 65 | 13 (20%) | 38 | 7 (18%) | 27 | 6 (22%) | 0.71c |
| Any steroids first 7 days of life (n, %)f | 65 | 13 (20%) | 38 | 7 (18%) | 27 | 6 (22%) | 0.71c |
| Steroid days (median, IQR) | 65 | 0 (0–3) | 38 | 0 (0–2) | 27 | 0 (0–6) | 0.77d |
| Ventricular dysfunction (n, %) | 65 | 9 (14%) | 38 | 4 (11%) | 27 | 5 (19%) | 0.36c |
| Milrinone (n, %) | 65 | 9 (14%) | 38 | 5 (13%) | 27 | 4 (15%) | 0.67c |
| NICU LOS, days (median, IQR) | 65 | 13 (9–28) | 38 | 13 (9–29) | 27 | 14 (9–22) | 0.98d |
| Confounders included in analysis | |||||||
| Vasopressor, any (n, %) | 65 | 43 (66%) | 38 | 24 (63%) | 27 | 19 (70%) | 0.54c |
| Seizures, any (n, %) | 65 | 23 (35%) | 38 | 13 (34%) | 27 | 10 (37%) | 0.81c |
| Benzodiazepine, any (n, %) | 65 | 9 (11%) | 38 | 5 (13%) | 27 | 4 (15%) | 0.85c |
| Clonidine, any (n, %) | 65 | 9 (11%) | 38 | 5 (13%) | 27 | 4 (15%) | 0.85c |
| PaCO2 All 35 – 45 (n, %) | 65 | 5 (8%) | 38 | 4 (11%) | 27 | 1 (4%) | 0.39e |
| Some < 35, all < 45 (n, %) | 65 | 13 (20%) | 38 | 7 (18%) | 27 | 6 (22%) | 0.95c |
| None < 35, some > 45 (n, %) | 65 | 26 (40%) | 38 | 14 (38%) | 27 | 12 (44%) | 0.72c |
| Some < 35, some > 45 (n, %) | 65 | 21 (32%) | 38 | 13 (34%) | 27 | 8 (30%) | 0.90c |
p<0.05;
unpaired T-test;
unpaired T-test with Levene’s test;
χ2 test;
Mann-Whitney U test;
Fisher’s exact test;
used either as outcome or confounder in modeling.
Markers of cardiopulmonary injury
The duration of invasive mechanical ventilation, pressure support, and oxygen support were defined as the number of days with an endotracheal tube, with any support other than low-flow nasal cannula, and FiO2>0.21, respectively. PaCO2 measurements classified neonates into those with:(23) (i) PaCO2 levels 35–45 mmHg; (ii) some <35 but none >45 mmHg; (iii) none <35 but some >45 mmHg; and (iv) some <35 and some >45 mmHg. Data were also collected on the presence of electrical seizures; use of iNO, inotropes, milrinone, benzodiazepines, or clonidine during HVx monitoring; and receipt of steroids.
Statistical analysis
Analyses were conducted with SASv9.2 (SAS Institute, Cary, NC). We determined associations between the autoregulation parameters (MAPOPT and blood pressure in relation to MAPOPT) in all neonates and stratified by sex. Linear regression for the logarithmic transformation of the average number of days of NICU stay, invasive mechanical ventilation, pressure support, and oxygen support was used to find both the crude (unadjusted) (data not shonw) and adjusted associations with the autoregulation parameters from each of three observation periods (TH, rewarming, and first 6 hours of normothermia). These analyses were adjusted for PaCO2 category, seizures, and receipt of a vasopressor, benzodiazepine, clonidine, or steroids. Binary outcomes (milrinone, iNO, or steroids) were analyzed as longitudinal outcomes over time, by autoregulation parameters within the same observation period. We used logistic regression for these binary measurements with generalized estimating equations to control for each measurement (milrinone, iNO, or steroid use) within the same neonate.(24) Temporal associations between autoregulation parameters and subsequent and preceding milrinone and iNO use were analyzed by logistic regression. The analysis of milrinone, iNO, and steroid use were controlled for presence of seizures, and the analysis of iNO and steroids were additionally adjusted for PaCO2 category during autoregulation monitoring.
RESULTS
We screened 122 newborns from September 27, 2010 to July 27, 2015. Forty-six neonates were ineligible for the study because of an unreliable arterial blood pressure tracing (16), parents’ refusal to consent (9), death (5), transfer to the pediatric ICU for extracorporeal membrane oxygenation (ECMO) (6), technical difficulties (5), inadequate monitoring resources (3), coagulopathy (1), complex heart disease (1), and language barriers (1). In total, 75 neonates received HVx monitoring (Supplemental Table S1, online). These patients received continuous HVx monitoring for 45.8 ±20.6 hours [mean ±SD] (n=31 girls/44 boys) during TH, 6.4±2.3 hours (n=26 girls/39 boys) during rewarming, and 5.6±0.9 hours (n=25 girls/38 boys) during normothermia. Ten neonates did not have HVx monitoring during rewarming because of ECMO (5), withdrawal of care (3), and technical difficulties (2). Two additional neonates did not have HVx monitoring during normothermia due to removal of the arterial catheter (1) or NIRS (1). Fifty-two neonates received vasopressors: 63% (33/52; 21 boys and 12 girls) received dopamine only, 28% (15/52; 7 boys and 8 girls) received dopamine and dobutamine, and 9% (5/52; 2 boys and 3 girls) received dopamine, dobutamine, and epinephrine.
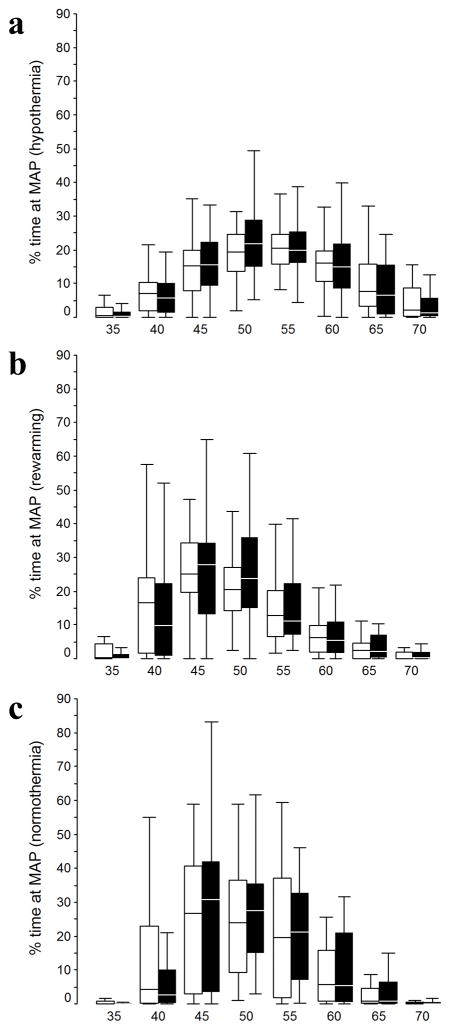
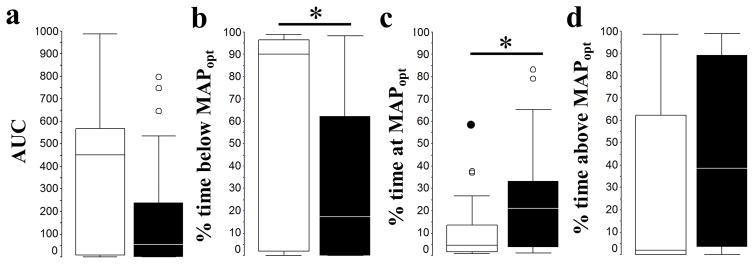
We identified MAPOPT in 65 infants (87%) during TH (MAPOPT = 45±7.5 mmHg); in 58 infants (89%) during rewarming (MAPOPT = 50±7.5 mmHg); and in 60 (95%) during normothermia (MAPOPT = 50±7.5 mmHg). Clinical data about the infants with identified MAPOPT are presented in Table 1. Ten neonates (6 or 60% male, 4 or 40% female) were excluded because their MAPOPT could not be identified. Boys were heavier than girls (mean ± SD = 3452 ± 660g vs. 3054 ± 486g, p=0.007). Although the boys had higher 1-min Apgar scores than did girls (p=0.04), the median 1-min Apgar score was 1 for both sexes (Table 1). MAP and MAPOPT were similar in both sexes in each of the observation periods (Figure 1 and Table 2). During normothermia, girls spent more time with blood pressure below MAPOPT (p=0.05) than did boys. Accordingly, girls spent less time with blood pressure at MAPOPT than did boys during normothermia (p=0.02). (Table 2 and Figure 2)
Figure 1.
Box and whisker plots of the percentage of time that girls (white boxes) and boys (black boxes) spent at each level of mean arterial blood pressure (MAP, mmHg). The observation periods were therapeutic hypothermia (a, girls [n=31], and boys [n=44]), rewarming (b, girls [n=26], and boys [n=39]), and the first 6 hours of normothermia (c, girls [n=25], and boys [n=38]). Box and whiskers plots, boxes represent the interquartile range (IQR) limited by the 25th and 75th percentile (lower and upper limit, respectively), line inside the box indicates the median and whiskers extend up the last datapoint within 1.5 times the IQR from the median. Outliers are not represented.
Table 2.
Optimal MAP (MAPOPT) and blood pressure parameters in relation to MAPOPT
| Parameter | n | TOTAL median (IQR) |
n | BOYS median (IQR) |
n | GIRLS median (IQR) |
P-valuea |
|---|---|---|---|---|---|---|---|
| MAPOPT (mmHg) | |||||||
| Hypothermia | 65 | 45 (40–55) | 38 | 45 (40–55) | 27 | 45 (40–55) | 0.97 |
| Rewarming | 58 | 50 (45–55) | 33 | 50 (45–55) | 25 | 50 (45–58) | 0.91 |
| Normothermia | 60 | 50 (45–55) | 38 | 50 (45–55) | 22 | 55 (44–60) | 0.23 |
| Area under the curve below MAPOPT (min•mmHg/h) | |||||||
| Hypothermia | 65 | 22.9 (1.3–236) | 38 | 48.6 (0.6–220) | 27 | 19.6 (3.4–287) | 0.39 |
| Rewarming | 58 | 180.3 (21.5–474.8) | 33 | 175.5 (24.8–386) | 25 | 197.9 (8.9–556) | 0.59 |
| Normothermia | 60 | 102.7 (3–482.7) | 38 | 59 (1–296) | 22 | 452 (8.1–585) | 0.07 |
| Duration of BP below MAPOPT (% of the autoregulation monitoring period) | |||||||
| Hypothermia | 65 | 6 (0.05–45.8) | 38 | 13.4 (0–45.4) | 27 | 4.5 (0.6–54.2) | 0.48 |
| Rewarming | 58 | 42.4 (4.7–85.7) | 33 | 41.3 (6.6–79.7) | 25 | 43.5 (1.7–87.9) | 0.65 |
| Normothermia | 60 | 35.9 (0–93) | 38 | 19 (0–64) | 22 | 90 (1.5–96.7) | 0.05* |
| Duration of BP at MAPOPT (% of the autoregulation monitoring period) | |||||||
| Hypothermia | 65 | 10.5 (4.5–22.5) | 38 | 12.6 (4.3–26) | 27 | 9.5 (94.8–18.8) | 0.78 |
| Rewarming | 58 | 12.5 (3.6–30.8) | 33 | 12 (3.6–32.2) | 25 | 13 (3.9–25.8) | 0.59 |
| Normothermia | 60 | 10 (2.6–30.6) | 38 | 19.9 (3.5–33.6) | 22 | 4.6 (1.6–16.6) | 0.02* |
| Duration of BP above MAPOPT (% of the autoregulation monitoring period) | |||||||
| Hypothermia | 65 | 73.6 (25.8–94.3) | 38 | 63.7 (31.8–94.6) | 27 | 79.2 (20.7–93.8) | 0.88 |
| Rewarming | 58 | 23.5 (5.4–62) | 33 | 22.4 (5.8–62.4) | 25 | 29 (3.7–61.7) | 0.89 |
| Normothermia | 60 | 26.7 (0.9–84) | 38 | 35.3 (2.7–89.9) | 22 | 2.1(0–64) | 0.06 |
| Maximal BP deviation below MAPOPT (mmHg) | |||||||
| Hypothermia | 65 | 10 (2.5–15) | 38 | 10 (0–15) | 27 | 10 (5–20) | 0.33 |
| Rewarming | 58 | 10 (5–15) | 33 | 10 (5–15) | 25 | 10 (5–20) | 0.51 |
| Normothermia | 60 | 10 (0–20) | 38 | 10 (0–15) | 22 | 15 (3.8–20) | 0.11 |
| Maximal BP deviation above MAPOPT (mmHg) | |||||||
| Hypothermia | 65 | 25 (15–35) | 38 | 22.5 (15–31.3) | 27 | 25 (20–35) | 0.18 |
| Rewarming | 58 | 15 (10–28) | 33 | 15 (10–28) | 25 | 15 (8–28) | 0.87 |
| Normothermia | 60 | 15 (5–25) | 38 | 15 (8–23) | 22 | 13 (0–25) | 0.36 |
p≤0.05;
Mann-Whitney U test.
BP, blood pressure; MAP, mean arterial pressure.
Figure 2.
Box and whisker plots of the area under the curve (AUC) below MAPOPT (a), and percent time spent below MAPOPT (b), at MAPOPT (c), and above MAPOPT (d) for girls (white boxes) and boys (black boxes) during 6 hours of normothermia (girls n=22, and boys n=38). Box and whiskers plots, boxes represent the interquartile range (IQR) limited by the 25th and 75th percentile (lower and upper limit, respectively), line inside the box indicates the median and whiskers extend up the last datapoint within 1.5 times the IQR from the median. Outliers (open circles) are those datapoints within 1.5 to 3 times the IQR from the median and extremes (closed circles) are those datapoint beyond 3 times the IQR form the median. *, p<0.05. Analysis performed by Mann Whitney U Test.
Sex-specific associations between cerebral autoregulation and duration of respiratory support
Greater maximal blood pressure deviation above MAPOPT during normothermia was associated with 13% shorter duration of mechanical ventilation (days) among boys (β = –0.142, p = 0.018) (Supplemental Table S2, online). In girls, however, more time spent with blood pressure above MAPOPT during rewarming was associated with 3% more days of positive pressure support (β=0.033, p = 0.044). Greater AUC (β = –0.004, p = 0.046) and greater duration with blood pressure below MAPOPT (β = –0.036, p= 0.024) during rewarming were associated with 1% and 4% fewer days of positive pressure support in girls, respectively. (Supplemental Table S3, online). Greater AUC (β = –0.003, p = 0.007), longer duration with blood pressure below MAPOPT (β = –0.022, p = 0.005), and greater maximal blood pressure deviation below MAPTOPT (β = −0.081, p = 0.019) during normothermia were also associated with fewer days of oxygen support in girls (Supplemental Table S4, online).
Blood pressure below MAPOPT is associated with length of NICU stay
The median duration of NICU stay was 13 days (IQR: 9, 28) and was similar between boys and girls (Table 1). Among boys, more time spent with blood pressure above MAPOPT during rewarming and normothermia (β = −0.008 for both, p = 0.016 and 0.004, respectively) was associated with a shorter NICU stay. Greater AUC (β=0.001, p = 0.018), longer duration of blood pressure below MAPOPT (β=0.007, p = 0.004), and greater maximal blood pressure deviation below MAPOPT (β=0.024, p = 0.018) during normothermia were associated with longer NICU stays in boys. In contrast, greater AUC (β = −0.001, p = 0.036), longer duration of blood pressure below MAPOPT (β = −0.006, p = 0.012), and greater maximal blood pressure deviation below MAPOPT (β = −0.024, p = 0.024) during normothermia were associated with shorter NICU stays in girls (Supplemental Table S5, online).
Cerebral blood pressure autoregulation may be associated with milrinone use
Nine neonates (14%; 5 boys and 4 girls) received milrinone for ventricular dysfunction or PPHN (Table 1). Nine (12%) received milrinone during TH, 6 (8%) during rewarming, and 4 (5%) during the first 6 hours of normothermia. Milrinone use was associated with the maximal blood pressure deviation below MAPOPT in the same time period in girls (β=0.157; p 0.005); Supplemental Table S6, online). Longitudinal analysis of autoregulation and milrinone use in preceding or subsequent periods were not associated (data not shown).
The cerebral autoregulation parameters were not associated with clinical use of iNO or steroids
Twenty-nine infants received iNO for clinical diagnoses of PPHN. Nineteen neonates received iNO during HVx monitoring (32% of girls and 21% of boys, Supplemental Table S1, online), and 13 of these neonates had an identified MAPOPT (22% of girls and 18% of boys, Table 1). The iNO was initiated at 20 ppm (maximum dose) in all infants and weaned per clinical protocol. iNO was started within the first 24 h of life in 84% of neonates (16/19), between 24 and 48h of life in 5% (1/19), and soon after rewarming in 11% (2/19). Among neonates who started iNO during TH, 88% (15/17) received iNO for at least the first 7 days of life, and the other 12% (2/17) were weaned off iNO before the end of TH. Thus, 23% of neonates undergoing HVx monitoring received iNO during TH and rewarming and 27% during normothermia. Blood pressure parameters in relation to MAPOPT were not associated with the simultaneous and clinical use of iNO for PPHN (Supplemental Table S6, online). Similarly, longitudinal analysis of autoregulation and iNO in preceding or subsequent periods were not associated (data not shown).
Girls and boys received steroids for a similar length of time. Twelve (16%) neonates received steroids during TH, 15 (20%) during rewarming, and 16 (21%) during the first 6 hours of normothermia. Thirteen infants (20%; 6 girls and 7 boys) with an identified MAPOPT received steroids during the first 7 days of life (Table 1). Blood pressure in relation to MAPOPT was not associated with steroid use (Supplemental Table S6, online).
DISCUSSION
We investigated the association between cerebral autoregulatory vasoreactivity measured by HVx and cardiopulmonary outcomes in neonates treated with TH for neonatal encephalopathy. Although girls and boys had similar MAPOPT values, the relationships between blood pressure autoregulation and lung injury were often sex-specific. While blood pressure above MAPOPT during normothermia was associated with shorter duration of intubation in boys, it was associated with longer durations of intubated or noninvasive respiratory pressure support in girls. We previously demonstrated that blood pressure below MAPOPT is associated with more severe brain injury on MRI (5, 7) at 2 weeks and worse neurocognitive outcomes at 2 years.(14) Our current findings suggest that maintaining blood pressure above MAPOPT protects both the brain and lungs in boys but not in girls. Additionally, blood pressure deviation below MAPOPT during normothermia was associated with a longer NICU stay in boys but shorter stay in girls. These findings provide new evidence for sex differences in the relationships between cerebral autoregulation and cardiopulmonary injury with neonatal encephalopathy.
Even though boys and girls had similar MAPOPT during TH, rewarming, and the first 6 hours of normothermia, girls had greater blood pressure deviation below MAPOPT than did boys during normothermia. Boys also spent more time with blood pressure within MAPOPT during normothermia, which indicates that boys had better autoregulatory function than girls in this pilot study cohort. The reasons behind this sex difference are not clear. We controlled for vasopressor use in the analysis given the confounding effects of vasopressors on autoregulation. Although MAP and thus time spent above MAPOPT are affected by vasopressor and inotropic medications, boys and girls with identified MAPOPT were treated in equal proportions with these agents (63% vs. 70%, respectively, p=0.54). The type of vasopressor used should be considered given potential sex-dependent differences in autoregulatory responses to different vasopressors. For example, phenylephrine and norepinephrine protect autoregulation only in female piglets with traumatic brain injury, (25, 26) whereas dopamine preserves autoregulation in both sexes.(27) In our study, vasopressor treatment was similar between boys and girls. Dopamine was universally used as the first-line agent, and dobutamine was added for boys and girls in equal proportions. The reciprocal interactions between cerebral and cardiopulmonary injuries are not well studied in neonatal encephalopathy, and the influence of different vasopressors on this complex relationship deserves further study.
The relationships between cerebral autoregulation and cardiopulmonary outcomes were often sex-specific. In boys, higher blood pressure above MAPOPT was associated with fewer days of intubation, and each 1 mmHg increase in maximal MAP above MAPOPT during normothermia was associated with a 13% decrease in days of intubation. In contrast, more time with blood pressure above MAPOPT during rewarming related to longer periods of intubation or noninvasive mechanical ventilation in girls. More blood pressure deviation below MAPOPT during normothermia corresponded to fewer days of supplemental oxygen in girls but not boys. Blood pressure deviation below MAPOPT is associated with more severe brain injury on MRI at 2 weeks(5, 7) and worse neurocognitive outcomes at 2 years(14) in neonatal encephalopathy. Hence, it appears that maintaining blood pressure at or above MAPOPT in boys may protect both the brain and lungs, whereas blood pressure above MAPOPT could worsen pulmonary injury and prolong the need for respiratory support in girls. Raising MAP to support cerebral perfusion pressure may cause cardiogenic strain with pulmonary edema or lung injury as previously reported in a preclinical model of neonatal encephalopathy.(28) However, it is unclear why girls might be more susceptible to these mechanisms than boys.
Blood pressure in relation to MAPOPT did not affect the likelihood of receiving iNO for PPHN or ventricular dysfunction. While it is possible that cerebral delivery of nitrite from iNO(29) may cause cerebral vasodilation,(30) iNO did not affect MAPOPT or increase blood pressure deviation below MAPOPT during clinical use for PPHN in our cohort. The iNO dosing by endotracheal tube was determined by cardiopulmonary clinical indications rather than for potential cerebral vasodilation. Therefore, it is possible that higher doses of iNO or nasal administration could affect cerebral vasoactivity. Milrinone may decrease systemic blood pressure or dilate the cerebral vasculature.(31) In this pilot study, milrinone use was associated with greater maximal decrease in blood pressure below MAPOPT among girls. Further studies are needed to determine the influence of milrinone on cerebral blood pressure autoregulation.
Our prior research in neonatal encephalopathy identified important relationships between autoregulation during TH and rewarming and neurologic outcomes.(5, 7, 14) In the current study, we identified several significant associations between the cardiopulmonary outcomes and blood pressure in relation to MAPOPT during rewarming and normothermia. Many of these relationships were observed solely in one sex but not the other. The absence of associations during hypothermia may be related to our methods of examining blood pressure in relation to MAPOPT during the entire hypothermic period which, while permitting direct comparisons between hypothermia, rewarming and normothermia, may sacrifice some granularity in the data across time.
Our study had several limitations. A causal relationship between blood pressure autoregulation and cardiopulmonary outcomes cannot be determined with the current design. Selection bias may have led to inclusion of less-ill neonates because parents of the sickest babies and those deemed likely to die were not consented for the study during the initial period of data collection, and HVx was not monitored in neonates who died, were withdrawn from life-sustaining treatments, or were transferred for possible ECMO. Larger studies are required to further define the multifactorial nature of the complex interactions between cerebral autoregulation and the cardiopulmonary system. Our observational data suggest that sex may play a role in this relationship. We did not measure HVx during the first 24 h of life in many neonates, and we did not evaluate the trajectory of MAPOPT across time in short epochs. Lastly, we acknowledge that multiple testing of outcome data derived from single patients may introduce statistical limitations in the interpretation of the results.
In conclusion, we report the potential use of cerebral autoregulation monitoring with HVx to identify blood pressures that support cerebral and cardiopulmonary recovery during and after TH for neonatal neonatal encephalopathy. MAPOPT measurements from HVx may provide information about the risk of lung injury, and sex may modulate cerebral vascular-pulmonary interactions to some degree in neonatal encephalopathy. Maintaining blood pressure above MAPOPT may protect both the brain and lungs in boys, but it is associated with respiratory compromise in girls. Girls who receive milrinone may also be more likely to have blood pressure deviation below MAPOPT. Growing pre-clinical and clinical data suggest sex-dimorphism in physiological responses to neonatal HI injury. Understanding these potentially divergent injury mechanisms in relation to autoregulation would improve treatment strategies in neonatal encephalopathy. Furthermore, our results support the need for large, prospective studies that are powered to evaluate sex-differences in pathophysiologic responses to neonatal encephalopathy as well as the interactions between neurologic and non-neurologic systems in neonates.
Supplementary Material
Acknowledgments
We are grateful to the Johns Hopkins Hospital nursing staff for their constant support.
Financial support: National Institutes of Health (Bethesda, MD, USA) R01HD070996 (FJN), R01HD074593 (FJN), K08NS080984 (JKL), KO8NS096115 (RCV), and R21HD072845 (JKL), by Johns Hopkins University (Baltimore, MD, USA) Clinician Scientist Award (JKL), by the American Heart Association (Dallas, TX, USA) Grant-in-Aid (JKL), and the Sutland-Pakula Endowment for Neonatal Research (RC-V). Medtronic (Minneapolis, MN, USA) provided partial funding for this study, but had no involvement in study design, analysis, interpretation and writing of this manuscript.
Footnotes
Disclosure: The authors declare no financial ties to products in the study or conflicts of interest.
References
- 1.Graham EM, Ruis KA, Hartman AL, Northington FJ, Fox HE. A systematic review of the role of intrapartum hypoxia-ischemia in the causation of neonatal encephalopathy. Am J Obstet Gynecol. 2008;199:587–595. doi: 10.1016/j.ajog.2008.06.094. [DOI] [PubMed] [Google Scholar]
- 2.Dammann O, Ferriero D, Gressens P. Neonatal encephalopathy or hypoxic-ischemic encephalopathy? Appropriate terminology matters. Pediatr Res. 2011;70:1–2. doi: 10.1203/PDR.0b013e318223f38d. [DOI] [PubMed] [Google Scholar]
- 3.Hankins GD, Koen S, Gei AF, Lopez SM, Van Hook JW, Anderson GD. Neonatal organ system injury in acute birth asphyxia sufficient to result in neonatal encephalopathy. Obstet Gynecol. 2002;99:688–691. doi: 10.1016/s0029-7844(02)01959-2. [DOI] [PubMed] [Google Scholar]
- 4.Shankaran S, Barnes PD, Hintz SR, Laptook AR, Zaterka-Baxter KM, McDonald SA, Ehrenkranz RA, Walsh MC, Tyson JE, Donovan EF, Goldberg RN, Bara R, Das A, Finer NN, Sanchez PJ, Poindexter BB, Van Meurs KP, Carlo WA, Stoll BJ, Duara S, Guillet R, Higgins RD Eunice Kennedy Shriver National Institute of Child H, Human Development Neonatal Research N. Brain injury following trial of hypothermia for neonatal hypoxic-ischaemic encephalopathy. Arch Dis Child Fetal Neonatal Ed. 2012;97:F398–404. doi: 10.1136/archdischild-2011-301524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Howlett JA, Northington FJ, Gilmore MM, Tekes A, Huisman TA, Parkinson C, Chung SE, Jennings JM, Jamrogowicz JJ, Larson AC, Lehmann CU, Jackson E, Brady KM, Koehler RC, Lee JK. Cerebrovascular autoregulation and neurologic injury in neonatal hypoxic-ischemic encephalopathy. Pediatr Res. 2013;74:525–535. doi: 10.1038/pr.2013.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Massaro AN, Govindan RB, Vezina G, Chang T, Andescavage NN, Wang Y, Al-Shargabi T, Metzler M, Harris K, du Plessis AJ. Impaired cerebral autoregulation and brain injury in newborns with hypoxic-ischemic encephalopathy treated with hypothermia. J Neurophysiol. 2015;114:818–824. doi: 10.1152/jn.00353.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tekes A, Poretti A, Scheurkogel MM, Huisman TA, Howlett JA, Alqahtani E, Lee JH, Parkinson C, Shapiro K, Chung SE, Jennings JM, Gilmore MM, Hogue CW, Martin LJ, Koehler RC, Northington FJ, Lee JK. Apparent diffusion coefficient scalars correlate with near-infrared spectroscopy markers of cerebrovascular autoregulation in neonates cooled for perinatal hypoxic-ischemic injury. AJNR Am J Neuroradiol. 2015;36:188–193. doi: 10.3174/ajnr.A4083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chalak LF, Tian F, Tarumi T, Zhang R. Cerebral Hemodynamics in Asphyxiated Newborns Undergoing Hypothermia Therapy: Pilot Findings Using a Multiple-Time-Scale Analysis. Pediatr Neurol. 2016;55:30–36. doi: 10.1016/j.pediatrneurol.2015.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ono M, Joshi B, Brady K, Easley RB, Zheng Y, Brown C, Baumgartner W, Hogue CW. Risks for impaired cerebral autoregulation during cardiopulmonary bypass and postoperative stroke. Br J Anaesth. 2012;109:391–398. doi: 10.1093/bja/aes148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ono M, Brady K, Easley RB, Brown C, Kraut M, Gottesman RF, Hogue CW., Jr Duration and magnitude of blood pressure below cerebral autoregulation threshold during cardiopulmonary bypass is associated with major morbidity and operative mortality. J Thorac Cardiovasc Surg. 2014;147:483–489. doi: 10.1016/j.jtcvs.2013.07.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Larson AC, Jamrogowicz JL, Kulikowicz E, Wang B, Yang ZJ, Shaffner DH, Koehler RC, Lee JK. Cerebrovascular autoregulation after rewarming from hypothermia in a neonatal swine model of asphyxic brain injury. J Appl Physiol (1985) 2013;115:1433–1442. doi: 10.1152/japplphysiol.00238.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee JK, Kibler KK, Benni PB, Easley RB, Czosnyka M, Smielewski P, Koehler RC, Shaffner DH, Brady KM. Cerebrovascular reactivity measured by near-infrared spectroscopy. Stroke. 2009;40:1820–1826. doi: 10.1161/STROKEAHA.108.536094. [DOI] [PubMed] [Google Scholar]
- 13.Lee JK, Brady KM, Chung SE, Jennings JM, Whitaker EE, Aganga D, Easley RB, Heitmiller K, Jamrogowicz JL, Larson AC, Lee JH, Jordan LC, Hogue CW, Lehmann CU, Bembea MM, Hunt EA, Koehler RC, Shaffner DH. A pilot study of cerebrovascular reactivity autoregulation after pediatric cardiac arrest. Resuscitation. 2014;85:1387–1393. doi: 10.1016/j.resuscitation.2014.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Burton VJ, Gerner G, Cristofalo E, Chung SE, Jennings JM, Parkinson C, Koehler RC, Chavez-Valdez R, Johnston MV, Northington FJ, Lee JK. A pilot cohort study of cerebral autoregulation and 2-year neurodevelopmental outcomes in neonates with hypoxic-ischemic encephalopathy who received therapeutic hypothermia. BMC Neurol. 2015;15:209. doi: 10.1186/s12883-015-0464-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ahnstedt H, Mostajeran M, Blixt FW, Warfvinge K, Ansar S, Krause DN, Edvinsson L. U0126 attenuates cerebral vasoconstriction and improves long-term neurologic outcome after stroke in female rats. J Cereb Blood Flow Metab. 2015;35:454–460. doi: 10.1038/jcbfm.2014.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Breu M, Zhang J, Porambo M, Pletnikov MV, Goeral K, Kakara M, Johnston MV, Fatemi A. Diffusion Tensor Imaging Abnormalities in the Cerebral White Matter Correlate with Sex-Dependent Neurobehavioral Deficits in Adult Mice with Neonatal Ischemia. Dev Neurosci. 2016 doi: 10.1159/000442943. [DOI] [PubMed] [Google Scholar]
- 17.Chavez-Valdez R, Martin LJ, Razdan S, Gauda EB, Northington FJ. Sexual dimorphism in BDNF signaling after neonatal hypoxia-ischemia and treatment with necrostatin-1. Neuroscience. 2014;260:106–119. doi: 10.1016/j.neuroscience.2013.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jia J, Davis CM, Zhang W, Edin ML, Jouihan S, Jia T, Bradbury JA, Graves JP, DeGraff LM, Lee CR, Ronnekleiv O, Wang R, Xu Y, Zeldin DC, Alkayed NJ. Sex- and isoform-specific mechanism of neuroprotection by transgenic expression of P450 epoxygenase in vascular endothelium. Exp Neurol. 2016;279:75–85. doi: 10.1016/j.expneurol.2016.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Milerova M, Drahota Z, Chytilova A, Tauchmannova K, Houstek J, Ostadal B. Sex difference in the sensitivity of cardiac mitochondrial permeability transition pore to calcium load. Mol Cell Biochem. 2016;412:147–154. doi: 10.1007/s11010-015-2619-4. [DOI] [PubMed] [Google Scholar]
- 20.Xue Q, Xiao D, Zhang L. Estrogen Regulates Angiotensin II Receptor Expression Patterns and Protects the Heart from Ischemic Injury in Female Rats. Biol Reprod. 2015;93:6. doi: 10.1095/biolreprod.115.129619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shankaran S, Laptook AR, Ehrenkranz RA, Tyson JE, McDonald SA, Donovan EF, Fanaroff AA, Poole WK, Wright LL, Higgins RD, Finer NN, Carlo WA, Duara S, Oh W, Cotten CM, Stevenson DK, Stoll BJ, Lemons JA, Guillet R, Jobe AH National Institute of Child H, Human Development Neonatal Research N. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N Engl J Med. 2005;353:1574–1584. doi: 10.1056/NEJMcps050929. [DOI] [PubMed] [Google Scholar]
- 22.Sarnat HB, Sarnat MS. Neonatal encephalopathy following fetal distress. A clinical and electroencephalographic study. Arch Neurol. 1976;33:696–705. doi: 10.1001/archneur.1976.00500100030012. [DOI] [PubMed] [Google Scholar]
- 23.Pappas A, Shankaran S, Laptook AR, Langer JC, Bara R, Ehrenkranz RA, Goldberg RN, Das A, Higgins RD, Tyson JE, Walsh MC Eunice Kennedy Shriver National Institute of Child H, Human Development Neonatal Research N. Hypocarbia and adverse outcome in neonatal hypoxic-ischemic encephalopathy. J Pediatr. 2011;158:752–758. e751. doi: 10.1016/j.jpeds.2010.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zeger SL, Liang KY. Longitudinal data analysis for discrete and continuous outcomes. Biometrics. 1986;42:121–130. [PubMed] [Google Scholar]
- 25.Armstead WM, Kiessling JW, Riley J, Kofke WA, Vavilala MS. Phenylephrine infusion prevents impairment of ATP- and calcium-sensitive potassium channel-mediated cerebrovasodilation after brain injury in female, but aggravates impairment in male, piglets through modulation of ERK MAPK upregulation. J Neurotrauma. 2011;28:105–111. doi: 10.1089/neu.2010.1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Armstead WM, Riley J, Vavilala MS. Preferential Protection of Cerebral Autoregulation and Reduction of Hippocampal Necrosis With Norepinephrine After Traumatic Brain Injury in Female Piglets. Pediatr Crit Care Med. 2016;17:e130–137. doi: 10.1097/PCC.0000000000000603. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 27.Armstead WM, Riley J, Vavilala MS. Dopamine prevents impairment of autoregulation after traumatic brain injury in the newborn pig through inhibition of Up-regulation of endothelin-1 and extracellular signal-regulated kinase mitogen-activated protein kinase. Pediatr Crit Care Med. 2013;14:e103–111. doi: 10.1097/PCC.0b013e3182712b44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Arruza L, Pazos MR, Mohammed N, Escribano N, Lafuente H, Santos M, Alvarez-Diaz FJ, Martinez-Orgado J. Hypoxic-ischemic brain damage induces distant inflammatory lung injury in newborn piglets. Pediatr Res. 2015 doi: 10.1038/pr.2015.87. [DOI] [PubMed] [Google Scholar]
- 29.Ibrahim YI, Ninnis JR, Hopper AO, Deming DD, Zhang AX, Herring JL, Sowers LC, McMahon TJ, Power GG, Blood AB. Inhaled nitric oxide therapy increases blood nitrite, nitrate, and s-nitrosohemoglobin concentrations in infants with pulmonary hypertension. J Pediatr. 2012;160:245–251. doi: 10.1016/j.jpeds.2011.07.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Truong GT, Schroder HJ, Liu T, Zhang M, Kanda E, Bragg S, Power GG, Blood AB. Role of nitrite in regulation of fetal cephalic circulation in sheep. J Physiol. 2014;592:1785–1794. doi: 10.1113/jphysiol.2013.269340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nishiguchi M, Ono S, Iseda K, Manabe H, Hishikawa T, Date I. Effect of vasodilation by milrinone, a phosphodiesterase III inhibitor, on vasospastic arteries after a subarachnoid hemorrhage in vitro and in vivo: effectiveness of cisternal injection of milrinone. Neurosurgery. 2010;66:158–164. doi: 10.1227/01.NEU.0000363153.62579.FF. discussion 164. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.