Abstract
Tremella fuciformis polysaccharide (TFPS), which is the extract of Tremella fuciformis Berk, has previously been demonstrated to exhibit potent anti-oxidative, anti-inflammatory and anti-aging effects. However, the mechanisms underlying these protective and therapeutic effects remain to be elucidated. The aim of the present study was to investigate the protective effects of TFPS on hydrogen peroxide-induced injury of human skin fibroblasts and to elucidate the aforementioned underlying mechanisms. A hydrogen peroxide-induced human skin fibroblast injury model was firstly established. MTT and reactive oxygen species (ROS) production assays, in addition to terminal deoxynucleotidyl transferase dUTP nick end labeling, reverse transcription-quantitative polymerase chain reaction and western blotting, were performed to investigate the protective effects of TFPS. Hydrogen peroxide decreased human skin fibroblast viability with a concurrent increase in ROS generation and cell apoptosis. Treatment with 0–400 µg/ml TFPS alone for up to 48 h did not result in alteration in cell viability. Notably, TFPS pre-treatment reduced oxidative stress and cell apoptosis in hydrogen peroxide-treated skin fibroblasts. In addition, there was profound inhibition of p16, p21, p53 and caspase-3 expression, and activation of extracellular-signal regulated kinase and Akt serine/threonine kinase 1, following TFPS pre-treatment. Furthermore, it was revealed that TFPS additionally protected fibroblasts via the upregulation of SIRT1 expression, and this was abrogated by the SIRT1 inhibitor niacinamide. These results indicated that TFPS alleviated hydrogen peroxide-induced oxidative stress and apoptosis in skin fibroblasts via upregulation of SIRT1 expression, indicating that TFPS may act as a potential therapeutic agent for oxidative-stress-associated skin diseases and aging.
Keywords: Tremella fuciformis polysaccharide, human skin fibroblasts, oxidative stress, apoptosis, SIRT1
Introduction
Oxidative stress induced by free radicals, which are produced in response to redox reactions, radiation and chemical reactions, can cause DNA and protein damage. Such stress is associated with the development of many diseases, such as atherosclerosis, diabetes mellitus, articular gout, and cancer (1,2). Hydrogen peroxide (H2O2) is one of type of reactive oxygen species (ROS) produced in normal cells during cell respiration and metabolism (3). However, many studies have indicated that a high concentration of H2O2, present under pathological conditions, may induce various human degenerative diseases and aging. Furthermore, H2O2 also interacts with intracellular ions such as iron and copper, leading to a chain reaction that produces more reactive radicals and amplifies the damage, thereby causing DNA and protein damage (4). Therefore, antioxidants are very important for the treatment of oxidative-stress-related diseases and aging.
Antioxidants in food, including vitamin C, polyphenols, tocopherols and flavonoids, may prevent free-radical-induced cell damage (5). They also may attenuate oxidative-stress-induced inflammatory reactions and apoptosis. Many natural plants and fungi contain antioxidant compounds, such as various phenolic compounds, that can be used as antioxidants to alleviate the damage caused by oxidative stress, inflammation, and energy expenditure (6–9). Polyphenols are natural compounds that can be found in fruits, vegetables, red wine and cereals. Epidemiological studies have found a negative correlation between the risk of chronic human diseases and the consumption of a polyphenol-rich diet (10–14).
Tremella fuciformis Berk, belonging to the order Tremellales and the family Tremellaceae, is an edible mushroom that has been a traditional Chinese medicine for centuries. Recently, many studies have revealed that the soluble extract of the polysaccharide fraction purified from Tremella fuciformis polysaccharide (TFPS) has several pharmacological activities, including anti-oxidant, anti-inflammatory, anti-diabetic, antitumor, and anti-aging effects (10,15). TFPS has also been used clinically for cancer and anti-aging treatments in China (16). However, the protective mechanism of TFPS is not clear.
In this study, we investigated the protective effect of a water extract of TFPS against hydrogen peroxide-induced injury to human skin fibroblasts. Because TFPS has several biological functions in cells, we also investigated its downstream target molecule and the underlying signaling pathways.
Materials and methods
Reagents
Tremella fuciformis was purchased from Gutian County Yishengyuan fruit and vegetable planting cooperatives. Antibodies against p16, p21, p53, Bax, Caspase-3, ERK, phosphorylated ERK, Akt, phosphorylated Akt, SIRT1 and GAPDH were purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA); anti-rabbit secondary antibodies were also purchased from Cell Signaling Technology, Inc. Unless otherwise indicated, all chemicals were purchased from Sigma (St. Louis, MO, USA).
Preparation of hot water extract of TFPS
The dry fruiting body of Tremella fuciformis was ground into powder (40 mesh) using a food mixer. Optimal extraction conditions were obtained as follows: Solid-liquid ratio, 1:60 g/ml; extraction time, 6 h; extraction temperature, 97°C. The mixture was placed in a water bath shaker at 120 rpm for hot water extraction and cooling to room temperature and then centrifuged at 4,000 rpm at 4°C for 20 min. The supernatant was precipitated with 98% ethanol, deproteinized by the Sevag method, purified through a dialysis membrane, and freeze-dried to obtain pure TFPS, as reported previously (17).
Cell culture
Human Dermal Fibroblasts-neonatal (HDF-n) (cat. no. 2310) were obtained from Sciencell Research Laboratories (Carlsbad, CA, USA). The cells were cultured in Fibroblast Medium (cat. no. 2301), which contains 10% FBS and fibroblast growth supplement, under 10% CO2 at 37°C.
Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) and Hoechst 33342 staining
Nuclear fragmentation was detected by TUNEL staining with an apoptosis detection kit (Roche Diagnostics, Indianapolis, IN, USA) and 10 mM Hoechst 33342 as previously described (17). Each data point indicates the results from 1,600 to 2,000 cells from 4 independent experiments.
Cell viability assay
Cell viability was analyzed by MTT assay (Roche Diagnostics) according to the manufacturer's instructions. Human skin fibroblasts (5,000 cells/well) were plated onto 24-well plates, pretreated with TFPS for 1 h and then treated with hydrogen peroxide for 24 h. All assays were performed in triplicate. The cells were incubated with 0.5 mg/ml 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenylterazolium bromide for 4 h, and the absorbance was measured at 490 nm, as described previously (18,19).
In situ detection of reactive oxygen species (ROS)
For ROS production to be evaluated in situ, human skin fibroblasts were stained with 10 µmol/l DHE (Sigma) for 30 min in a dark, humidified chamber at 37°C. ROS were indicated by red fluorescence, visualized by fluorescence microscopy and analyzed with ImageJ software.
Western blotting analysis
Cell lysates were analyzed by SDS-PAGE and electrotransferred to PVDF membranes. Membranes were then blocked with 5% skim milk for 2 h and incubated with specific antibodies overnight. After five washes in TBST (containing 0.1% Tween-20 in TBS), the membranes were incubated with horseradish peroxidase-conjugated secondary antibodies in TBST at room temperature for 1 h. The bands were detected by chemiluminescence detection agents and analyzed with ImageJ software.
Statistical analysis
All of the statistical calculations were performed using the GraphPad Prism 5 software program (GraphPad Prism, San Diego, CA, USA). The data are expressed as the mean ± SEM. One-way ANOVA with Bonferroni correction was used for multiple comparisons. P<0.05 was considered to indicate a statistically significant difference.
Results
TFPS alleviates hydrogen peroxide-triggered injury of human skin fibroblasts
To assess the potential function of TFPS, we performed cytotoxicity analysis by MTT assay of cultured human skin fibroblasts treated with various concentrations of TFPS. As shown in Fig. 1A, treatment with 0–300 µg/ml TFPS for up to 48 h did not change the cell viability of human skin fibroblasts. The cell viability assay was also performed on 0–500 µM H2O2-treated human skin fibroblasts. Hydrogen peroxide induced a significant (P<0.05) concentration-dependent decrease in viability (Fig. 1B), from the 200 µM H2O2 treatment. Furthermore, treatment with 300 µM H2O2 led to a decrease in skin fibroblast viability to 59.5% of that of the control, which was a moderate amount of cell injury in vitro. Therefore, we used 300 µM H2O2 to establish a human skin fibroblast injury model for the remainder of the experiments. To analyze whether TFPS can protect human skin fibroblasts from cell injury induced by 300 µM hydrogen peroxide, we pre-treated the human skin fibroblasts with 0–300 µg/ml TFPS for 1 h before H2O2 application. The cell viability results showed that TFPS had a concentration-dependent protective effect on human skin fibroblasts and reached a peak protective effect at a concentration of 200 µg/ml (Fig. 1C). Thus, these results demonstrate that TFPS can protect human skin fibroblasts from a hydrogen peroxide-induced decrease in cell viability.
Figure 1.
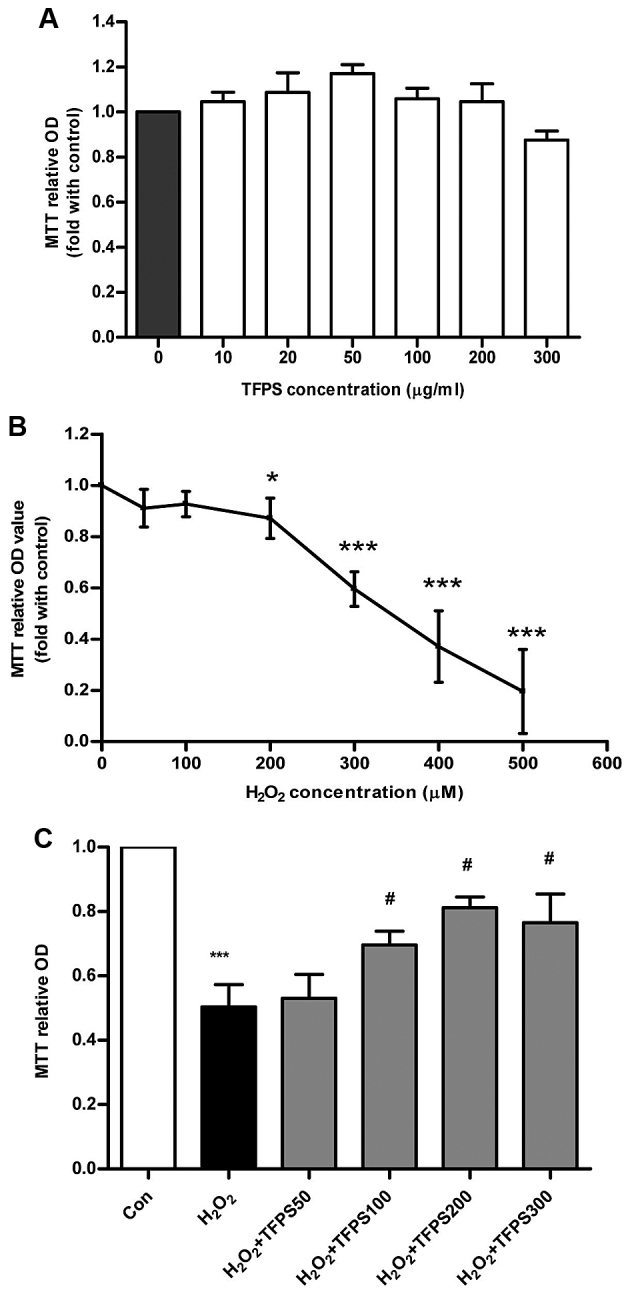
Tremella fuciformis polysaccharide (TFPS) alleviates hydrogen peroxide-triggered human skin fibroblast injury. (A) Human skin fibroblasts were cultured in the presence of TFPS (0, 10, 20, 50, 100, 200, and 300 µg/ml), for 24 h, and cell viability was measured by MTT assay (n=3). (B) Human skin fibroblasts were cultured in the presence of H2O2 (100, 200, 300, 400, 500, and 600 µM) for 24 h, and cell viability was analyzed by MTT assay (n=3). (C) Human skin fibroblasts were pre-treated with TFPS (50, 100, 200, and 300 µg/ml) for 1 h, and then 300 µM H2O2 was applied for 24 h. Cell viability was determined by MTT assay (n=4) (*P<0.05 and ***P<0.001 vs. the control group; #P<0.05 vs. the 300 µM H2O2 group).
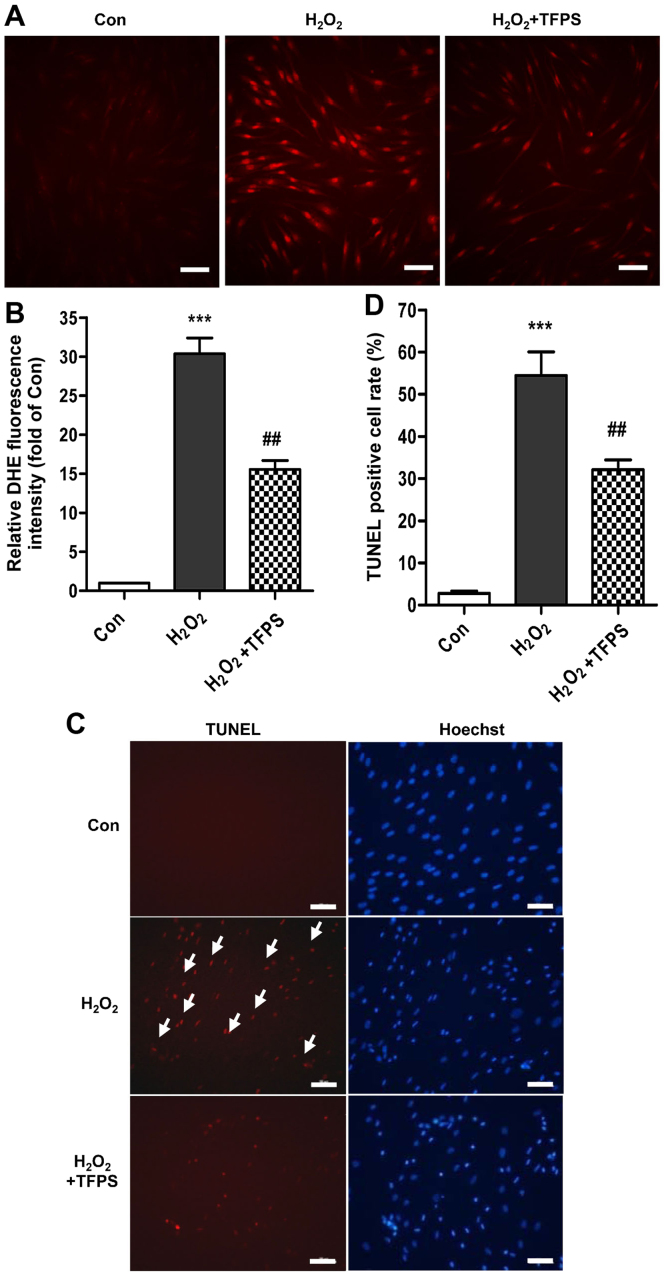
TFPS relieves hydrogen peroxide-induced human skin fibroblast injury by attenuating oxidative stress and cell apoptosis
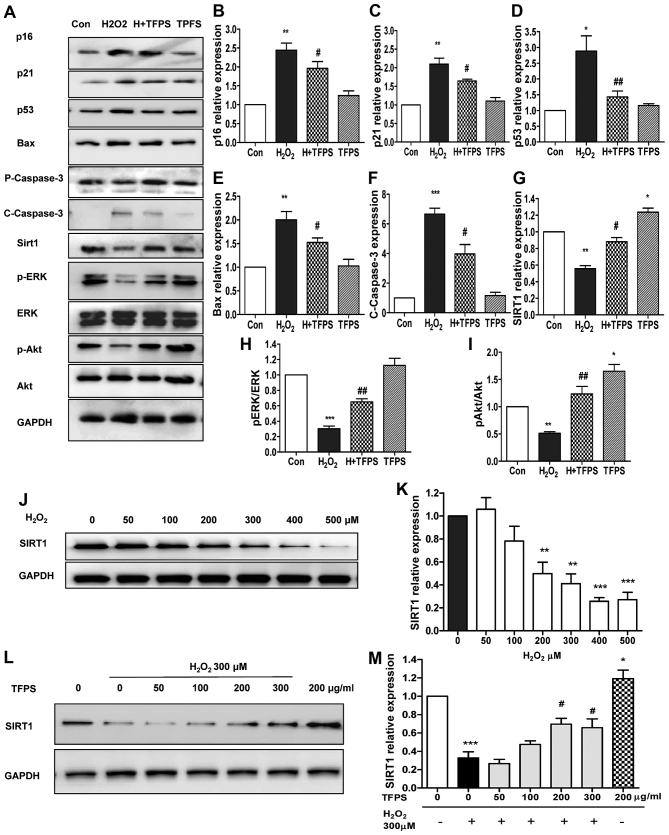
The ROS level increased to nearly 30-fold in human skin fibroblasts treated with hydrogen peroxide compared with in the control. However, human skin fibroblasts pretreated with 200 µg/ml TFPS had attenuated H2O2-induced ROS generation, with a decline of approximately 51.7% compared with fibroblasts that had not been pretreated (Fig. 2A and B). In addition, the 200 µg/ml TFPS treatment decreased human skin fibroblast apoptosis to 32.1±4.0% compared with 54.4±9.7% in the H2O2 group, as analyzed by TUNEL staining (Fig. 2C and D). Western blot analysis also showed that TFPS pretreatment clearly attenuated the H2O2-induced p16, p21, Bax and p53 upregulation and Caspase-3 activation (cleaved) (Fig. 3A-F). However, pretreatment with TFPS alone did not result in changes to the expression of p16, p21, Bax, p53, or Caspase-3 activity. Pretreatment with TFPS also upregulate SIRT1 expression and activated Akt signaling pathways (regardless of subsequent H2O2 treatment), as well as increased Erk1/2 activation with subsequent H2O2 treatment (Fig. 3A and G-I). Moreover, H2O2 led to SIRT1 downregulation in a concentration-dependent manner (Fig. 3J and K), whereas TFPS pretreatment promoted SIRT1 expression in a concentration-dependent manner after 300 µM H2O2 application (Fig. 3L and M). Interestingly, treatment with TFPS alone promoted basal SIRT1 expression in skin fibroblasts (Fig. 3L and M). Taken together, these results indicate that TFPS can suppress oxidative stress and cell injury by the inhibition of pro-apoptotic pathways and the activation of pro-survival pathways.
Figure 2.
TFPS relieves hydrogen peroxide-induced human skin fibroblast injury by attenuating oxidative stress and apoptosis. (A and B) DHE staining was performed in control, H2O2-treated (300 µM) and H2O2-treated, TFPS pretreated (200 µg/ml) skin fibroblasts, and an average of the data is shown (n=4, scale bar: 100 µM, ***P<0.001 vs. control, ##P<0.01 vs. the H2O2 group). (C and D) Skin fibroblast apoptosis was detected by TUNEL staining, and an average of the data in control, H2O2-treated (300 µM) and H2O2-treated, TFPS pretreated (200 µg/ml) skin fibroblasts is shown (n=4, scale bar: 100 µM, ***P<0.001 vs. control, ##P<0.01 vs. H2O2 group).
Figure 3.
TFPS inhibits pro-apoptotic pathways and activates pro-survival pathways in human skin fibroblasts. (A-I) p16, p21, Bax, p53, SIRT1, Caspase-3, p-Akt, Akt, p-ERK, ERK and GAPDH protein expression in control, H2O2-treated (300 µM), H2O2-treated, TFPS pretreated (200 µg/ml), and TFPS (200 µg/ml) pretreated in skin fibroblasts as assayed by western blotting (n=4, *P<0.05, **P<0.01, and ***P<0.001 vs. control; #P<0.05, ##P<0.01 vs. H2O2 group). (J and K) SIRT1 and GAPDH protein expression in the presence of 0–500 µM H2O2 for 24 h, as assayed by western blotting (n=3, *P<0.05, **P<0.01, ***P<0.001 vs. control). (L and M) SIRT1 and GAPDH protein expression with and without 0–300 µg/ml TFPS pre-treatment for 1 h and then application of 300 µM H2O2 for 24 h, as assayed by Western blotting (n=4, *P<0.05 and ***P<0.001 vs. control; #P<0.05 vs. 300 µM H2O2 group).
TFPS attenuates hydrogen peroxide-induced cell oxidative stress and apoptosis by upregulation of the SIRT1 pathway
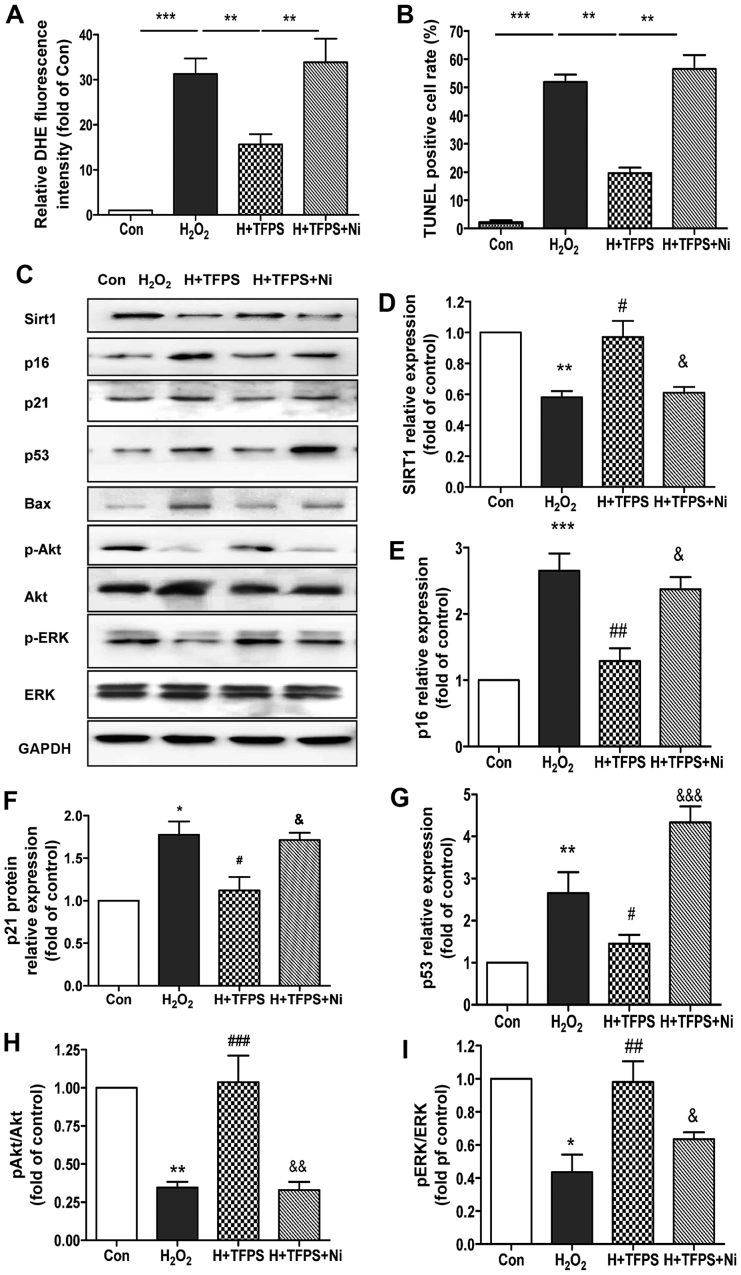
Next, to determine whether TFPS-induced SIRT1 upregulation is necessary for human skin fibroblast protection, niacinamide (a SIRT1 inhibitor) was applied in the H2O2-induced cell injury model (Fig. 4). Fig. 4A and B illustrates that TFPS attenuated H2O2-induced ROS generation and apoptosis, whereas the SIRT1 inhibitor niacinamide significantly reversed the protective effect of TFPS. We then checked several important cell survival and death signaling pathways in the presence and absence of the SIRT1 inhibitor niacinamide. The results showed that, after niacinamide treatment, the inhibitory effects of TFPS on SIRT1, p16, p21, Bax, p53 and Caspase-3 were reversed (Fig. 4C and E-G), and the effects of TFPS on the activation of ERK and Akt were blocked (Fig. 4C, H and I). These results indicate that TFPS-induced SIRT1 upregulation is necessary for the protection of human skin fibroblasts from H2O2-induced oxidative stress, ROS production and apoptosis.
Figure 4.
SIRT1 plays a central role in TFPS-mediated human skin fibroblast protection. (A) DHE staining in control, H2O2-treated (300 µM), 200 µg/ml TFPS pretreated + H2O2-treated, and 200 µg/ml TFPS + niacinamide pretreated + H2O2-treated skin fibroblasts (n=4, **P<0.01, ***P<0.001). (B) Skin fibroblast apoptosis in control, H2O2-treated (300 µM), 200 µg/ml TFPS pretreated + H2O2-treated, and 200 µg/ml TFPS + niacinamide pretreated + H2O2-treated skin fibroblasts, as detected by TUNEL staining (n=4, **P<0.01, ***P<0.001). (C-I) p16, p21, Bax, p53, SIRT1, Caspase-3, p-Akt, Akt, p-ERK, ERK and GAPDH protein expression in control, H2O2-treated (300 µM), 200 µg/ml TFPS pretreated + H2O2-treated, and 200 µg/ml TFPS + niacinamide pretreated + H2O2-treated skin fibroblasts for 24 h, as assayed by western blotting. An average of the data is shown. (n=3, *P<0.05, **P<0.01, ***P<0.001 vs. control; #P<0.05, ##P<0.01, ###P<0.001 vs. H2O2 group; &P<0.05, &&P<0.01, &&&P<0.001 vs. H2O2 + TFPS group).
Discussion
Hydrogen peroxide triggers human skin fibroblast cell apoptosis
It has been shown that hydrogen peroxide, like other free radicals, plays an important role in the development of skin injury and aging. Treatment of human skin fibroblasts with 0–500 µM H2O2 induced a decrease in cell viability in a concentration-dependent manner. Furthermore, 300 µM hydrogen peroxide caused a 40.5% cell viability decrease, which corresponds to a moderate skin fibroblast injury in this study.
We also examined H2O2-induced ROS production and apoptosis in human skin fibroblasts. Treatment of cells with 300 µM H2O2 for 24 h significantly increased ROS levels, by approximately 30.37-fold, compared with those of the control groups, confirming previous data showing the decrease in viability of fibroblasts (18,19). Furthermore, the proportion of apoptotic cells increased significantly, from 2.78% in the control group to 54.45% in the 300 µM H2O2 group. Thus, our data confirm that hydrogen peroxide can induce skin fibroblast injury (20–22).
TFPS can protect human skin fibroblasts from oxidative stress and apoptosis caused by hydrogen peroxide
ROS, such as H2O2, superoxide anions (O2−) and hydroxyl radicals (HO−), are continuously produced endogenously during normal cellular respiration and metabolism. At low concentrations, they are important in the regulation of cellular functions such as immune responses and cell signal transduction (23,24). However, at high concentrations, ROS can cause damage to DNA and proteins, which has been associated with atherosclerosis, diabetes mellitus, and carcinogenesis (25). Very high concentration of ROS, which can be generated during inflammation, UV radiation, and aging, are also involved in the pathogenesis of various skin diseases (10,26,27). Therefore, ROS scavengers, such as vitamins, herbal extracts, and antioxidants, are very important in the treatment of ROS-induced pathological changes.
Tremella fuciformis is a traditional nutritional food in China and is used as a traditional Chinese medicine and dietary supplement. TFPS has also been used in the clinic in China for cancer patients who receive radio- and chemotherapy, to enhance their immune function, and for chronic gastritis and chronic hepatitis patients (14,28–30). Recent studies have indicated that the medicinal and tonic properties of Tremella fuciformis are due to its polysaccharides (21,31,32), which have several biological activities such as anti-inflammation, anti-diabetic, anti-cancer, and immunoprotective properties.
In the present study, we verified that TFPS at concentrations ranging from 100 to 200 µg/ml had excellent antioxidative and cell protective functions. TFPS markedly reduced H2O2-induced skin fibroblast injury through suppression of ROS production and apoptosis and is a potential alternative herbal food supplement and pharmacotherapy for treating skin injury upon aging.
SIRT1 plays a central role in the TFPS-mediated protection effect on human skin fibroblasts
Aging is a time-dependent progression of biochemical and physiological loss of function, and it is associated with increased risk of morbidity and mortality. There is a growing evidence indicating that oxidative stress plays a key role in the aging process and in various degenerative diseases, including atherosclerosis, diabetes, inflammation and cancer (33). Many studies have suggested that age-related biochemical and physiological decline is associated with an imbalance in the production of free radical and intracellular antioxidants, which leads to cell death and loss of function.
Sirtuins (SIRTs) are a family of NAD(+)-dependent enzymes that have a primarily protective function in the development of many age-related diseases, including cardiovascular disease, neurodegeneration and cancer. Recent studies have suggested that the regulation of mammalian lifespan by sirtuins has important therapeutic implications for age-related diseases (34,35).
SIRT1 is a member of a highly conserved gene family encoding nicotinamide adenine dinucleotide (NAD)(+)-dependent deacetylases. SIRT1 expression leads to increased DNA stability and prolonged survival in organisms from yeast to mammals (36,37). SIRT1 has been found to regulate many cellular processes, including cellular senescence, apoptosis, glucose homeostasis, aging and longevity. In this study, we found that H2O2 induced oxidative stress and cell injury in skin fibroblasts, with significant downregulation of SIRT1, in a concentration-dependent manner. However, TFPS pre-treatment significantly increased SIRT1 expression during H2O2 treatment of cultured human skin fibroblasts, and the SIRT inhibitor nicotinamide reversed this protective effect. Furthermore, TFPS promoted fibroblast SIRT1 expression and then regulated cell aging and survival via decreased expression of p16, p21, p53 and Capsase-3 and activation of ERK and Akt. Our data provide new insight into the protective mechanisms of TFPS during H2O2-induced skin injury and suggest that TFPS functions as a SIRT1 activator and could be used as a new anti-skin aging agent.
TFPS and clinical relevance to skin injury and aging
We demonstrated that pretreatment with TFPS suppressed H2O2-induced oxidative stress and apoptosis in skin fibroblasts in a concentration-dependent manner and possessed excellent antioxidative properties. Therefore, our present findings may provide a theoretical basis for TFPS as an alternative herbal food supplement and potential pharmacotherapy for skin injury and aging.
However, given that TFPS contains several polysaccharides and has a variety of pharmacological effects, our study, which focused on oxidative stress inhibition and anti-apoptotic effects, has limitations. Further studies are required to define the potential protective mechanisms of TFPS in H2O2-induced skin injury.
In summary, we revealed that TFPS exhibits protective properties against hydrogen peroxide-induced oxidative stress and apoptosis mainly by upregulating SIRT1 and promoting downstream signaling, suggesting that TFPS is a potential therapeutic agent for oxidative stress-related skin diseases and skin aging.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (nos. 81470427, 81200221 and 81270887), the Quality Inspection Public Welfare Scientific Research Foundation of China (nos. 201310132 and 201410019), the Beijing Natural Science Foundation (no. 7142142) and the Beijing Hospital Nova project (no. BJ-2016-045).
References
- 1.Toyokuni S. The origin and future of oxidative stress pathology: From the recognition of carcinogenesis as an iron addiction with ferroptosis-resistance to non-thermal plasma therapy. Pathol Int. 2016;66:245–259. doi: 10.1111/pin.12396. [DOI] [PubMed] [Google Scholar]
- 2.Singh R, Devi S, Gollen R. Role of free radical in atherosclerosis, diabetes and dyslipidaemia: Larger-than-life. Diabetes Metab Res Rev. 2015;31:113–126. doi: 10.1002/dmrr.2558. [DOI] [PubMed] [Google Scholar]
- 3.Barbosa MR, Sampaio IH, Teodoro BG, Sousa TA, Zoppi CC, Queiroz AL, Passos MA, Alberici LC, Teixeira FR, Manfiolli AO, et al. Hydrogen peroxide production regulates the mitochondrial function in insulin resistant muscle cells: Effect of catalase overexpression. Biochim Biophys Acta. 2013;1832:1591–1604. doi: 10.1016/j.bbadis.2013.04.029. [DOI] [PubMed] [Google Scholar]
- 4.Jomova K, Valko M. Advances in metal-induced oxidative stress and human disease. Toxicology. 2011;283:65–87. doi: 10.1016/j.tox.2011.03.001. [DOI] [PubMed] [Google Scholar]
- 5.Wahlqvist ML. Antioxidant relevance to human health. Asia Pac J Clin Nutr. 2013;22:171–176. doi: 10.6133/apjcn.2013.22.2.21. [DOI] [PubMed] [Google Scholar]
- 6.Skulachev MV, Skulachev VP. New data on programmed aging-slow phenoptosis. Biochemistry (Mosc) 2014;79:977–993. doi: 10.1134/S0006297914100010. [DOI] [PubMed] [Google Scholar]
- 7.Pandey KB, Rizvi SI. Plant polyphenols as dietary antioxidants in human health and disease. Oxid Med Cell Longev. 2009;2:270–278. doi: 10.4161/oxim.2.5.9498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yang G, Xue Y, Zhang H, Du M, Zhu MJ. Favourable effects of grape seed extract on intestinal epithelial differentiation and barrier function in IL10-deficient mice. Br J Nutr. 2015;114:15–23. doi: 10.1017/S0007114515001415. [DOI] [PubMed] [Google Scholar]
- 9.Yang G, Wang H, Kang Y, Zhu MJ. Grape seed extract improves epithelial structure and suppresses inflammation in ileum of IL-10-deficient mice. Food Funct. 2014;5:2558–2563. doi: 10.1039/C4FO00451E. [DOI] [PubMed] [Google Scholar]
- 10.Han CK, Chiang HC, Lin CY, Tang CH, Lee H, Huang DD, Zeng YR, Chuang TN, Huang YL. Comparison of immunomodulatory and anticancer activities in different strains of Tremella fuciformis berk. Am J Chin Med. 2015;43:1637–1655. doi: 10.1142/S0192415X15500937. [DOI] [PubMed] [Google Scholar]
- 11.Xu W, Shen X, Yang F, Han Y, Li R, Xue D, Jiang C. Protective effect of polysaccharides isolated from Tremella fuciformis against radiation-induced damage in mice. J Radiat Res. 2012;53:353–360. doi: 10.1269/jrr.11073. [DOI] [PubMed] [Google Scholar]
- 12.Gao QP, Jiang RZ, Chen HQ, Jensen E, Seljelid R. Characterization and cytokine stimulating activities of heteroglycans from Tremella fuciformis. Planta Med. 1996;62:297–302. doi: 10.1055/s-2006-957888. [DOI] [PubMed] [Google Scholar]
- 13.Wang ZC, Yang S, Li LX, Zhou FM, Wang RD. Studies on the effects of Tremella fuciformis Berk preparation on immunity and blood formation in rhesus monkeys. J Tradit Chin Med. 1983;3:13–16. [PubMed] [Google Scholar]
- 14.Jin Y, Hu X, Zhang Y, Liu T. Studies on the purification of polysaccharides separated from Tremella fuciformis and their neuroprotective effect. Mol Med Rep. 2016;13:3985–3992. doi: 10.3892/mmr.2016.5026. [DOI] [PubMed] [Google Scholar]
- 15.Liao WC, Hsueh CY, Chan CF. Antioxidative activity, moisture retention, film formation, and viscosity stability of Auricularia fuscosuccinea, white strain water extract. Biosci Biotechnol Biochem. 2014;78:1029–1036. doi: 10.1080/09168451.2014.912113. [DOI] [PubMed] [Google Scholar]
- 16.Duan C, Wang C. Optimization of extracting Tremella fuciformis polysaccharide and detection of its toxicity to cells of human body. Chin Surfact Detergent Cosmetic. 2016;46:101–105. [Google Scholar]
- 17.Shen T, Yang C, Ding L, Zhu Y, Ruan Y, Cheng H, Qin W, Huang X, Zhang H, Man Y, et al. Tbx20 functions as an important regulator of estrogen-mediated cardiomyocyte protection during oxidative stress. Int J Cardiol. 2013;168:3704–3714. doi: 10.1016/j.ijcard.2013.06.018. [DOI] [PubMed] [Google Scholar]
- 18.Chwa M, Atilano SR, Reddy V, Jordan N, Kim DW, Kenney MC. Increased stress-induced generation of reactive oxygen species and apoptosis in human keratoconus fibroblasts. Invest Ophthalmol Vis Sci. 2006;47:1902–1910. doi: 10.1167/iovs.05-0828. [DOI] [PubMed] [Google Scholar]
- 19.Kang KA, Zhang R, Piao MJ, Lee KH, Kim BJ, Kim SY, Kim HS, Kim DH, You HJ, Hyun JW. Inhibitory effects of glycitein on hydrogen peroxide induced cell damage by scavenging reactive oxygen species and inhibiting c-Jun N-terminal kinase. Free Radic Res. 2007;41:720–729. doi: 10.1080/10715760701241618. [DOI] [PubMed] [Google Scholar]
- 20.Ada S Dantas, Day A, Ikeh M, Kos I, Achan B, Quinn J. Oxidative stress responses in the human fungal pathogen, Candida albicans. Biomolecules. 2015;5:142–165. doi: 10.3390/biom5010142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kovacs I, Horváth M, Lányi Á, Petheő GL, Geiszt M. Reactive oxygen species-mediated bacterial killing by B lymphocytes. J Leukoc Biol. 2015;97:1133–1137. doi: 10.1189/jlb.4AB1113-607RR. [DOI] [PubMed] [Google Scholar]
- 22.Simeoni L, Bogeski I. Redox regulation of T-cell receptor signaling. Biol Chem. 2015;396:555–568. doi: 10.1515/hsz-2014-0312. [DOI] [PubMed] [Google Scholar]
- 23.Bauer G. Signaling and proapoptotic functions of transformed cell-derived reactive oxygen species. Prostaglandins Leukot Essent Fatty Acids. 2002;66:41–56. doi: 10.1054/plef.2001.0332. [DOI] [PubMed] [Google Scholar]
- 24.Ivanovas B, Zerweck A, Bauer G. Selective and non-selective apoptosis induction in transformed and non-transformed fibroblasts by exogenous reactive oxygen and nitrogen species. Anticancer Res. 2002;22:841–856. [PubMed] [Google Scholar]
- 25.Amber KT, Shiman MI, Badiavas EV. The use of antioxidants in radiotherapy-induced skin toxicity. Integr Cancer Ther. 2014;13:38–45. doi: 10.1177/1534735413490235. [DOI] [PubMed] [Google Scholar]
- 26.Ukai S, Hirose K, Kiho T, Hara C, Irikura T. Antitumor activity on sarcoma 180 of the polysaccharides from Tremella fuciformis Berk. Chem Pharm Bull (Tokyo) 1972;20:2293–2294. doi: 10.1248/cpb.20.1347. [DOI] [PubMed] [Google Scholar]
- 27.Shin DI, Song KS, Park HS. Oral vaccination of mice with Tremella fuciformis yeast-like conidium cells expressing HBsAg. Biotechnol Lett. 2015;37:539–544. doi: 10.1007/s10529-014-1720-1. [DOI] [PubMed] [Google Scholar]
- 28.Liu J, Meng CG, Yan YH, Shan YN, Kan J, Jin CH. Structure, physical property and antioxidant activity of catechin grafted Tremella fuciformis polysaccharide. Int J Biol Macromol. 2016;82:719–724. doi: 10.1016/j.ijbiomac.2015.11.027. [DOI] [PubMed] [Google Scholar]
- 29.Zhang Z, Wang X, Zhao M, Qi H. Free-radical degradation by Fe2+/Vc/H2O2 and antioxidant activity of polysaccharide from Tremella fuciformis. Carbohydr Polym. 2014;112:578–582. doi: 10.1016/j.carbpol.2014.06.030. [DOI] [PubMed] [Google Scholar]
- 30.Zhu H, Yuan Y, Liu J, Zheng L, Chen L, Ma A. Comparing the sugar profiles and primary structures of alkali-extracted water-soluble polysaccharides in cell wall between the yeast and mycelial phases from Tremella fuciformis. J Microbiol. 2016;54:381–386. doi: 10.1007/s12275-016-5533-x. [DOI] [PubMed] [Google Scholar]
- 31.Long YC, Tan TM, Takao I, Tang BL. The biochemistry and cell biology of aging: Metabolic regulation through mitochondrial signaling. Am J Physiol Endocrinol Metab. 2014;306:E581–E591. doi: 10.1152/ajpendo.00665.2013. [DOI] [PubMed] [Google Scholar]
- 32.Poljsak B, Šuput D, Milisav I. Achieving the balance between ROS and antioxidants: When to use the synthetic antioxidants. Oxid Med Cell Longev. 2013;2013:956792. doi: 10.1155/2013/956792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Corbi G, Conti V, Scapagnini G, Filippelli A, Ferrara N. Role of sirtuins, calorie restriction and physical activity in aging. Front Biosci (Elite Ed) 2012;4:768–778. doi: 10.2741/e417. [DOI] [PubMed] [Google Scholar]
- 34.Poulose N, Raju R. Sirtuin regulation in aging and injury. Biochim Biophys Acta. 2015;1852:2442–2455. doi: 10.1016/j.bbadis.2015.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Imai S, Yoshino J. The importance of NAMPT/NAD/SIRT1 in the systemic regulation of metabolism and ageing. Diabetes Obes Metab. 2013;15(Suppl 3):S26–S33. doi: 10.1111/dom.12171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ruan Y, Dong C, Patel J, Duan C, Wang X, Wu X, Cao Y, Pu L, Lu D, Shen T, Li J. SIRT1 suppresses doxorubicin-induced cardiotoxicity by regulating the oxidative stress and p38MAPK pathways. Cell Physiol Biochem. 2015;35:1116–1124. doi: 10.1159/000373937. [DOI] [PubMed] [Google Scholar]
- 37.Shen T, Ding L, Ruan Y, Qin W, Lin Y, Xi C, Lu Y, Dou L, Zhu Y, Cao Y, et al. SIRT1 functions as an important regulator of estrogen-mediated cardiomyocyte protection in angiotensin II-induced heart hypertrophy. Oxid Med Cell Longev. 2014;2014:713894. doi: 10.1155/2014/713894. [DOI] [PMC free article] [PubMed] [Google Scholar]