Abstract
Understanding of the long-term clinical outcomes associated with bariatric surgery has recently been advanced. Research related to the sequelae of diabetes—in particular, long-term microvascular and macrovascular complications—in patients who undergo weight-loss surgery is imperative to this pursuit. While numerous randomized control trials have assessed glucose control with bariatric surgery compared with intensive medical therapy, bariatric surgery outcome data relating to microvascular and macrovascular complications have been limited to observational studies and nonrandomized clinical trials. As a result, whether bariatric surgery is associated with a long-term reduction in microvascular and macrovascular complications when compared with current intensive glycemic control therapy cannot be determined because the evidence is insufficient. However, the consistent salutary effects of bariatric surgery on diabetes remission and glycemic improvement support the opportunity (and need) to conduct high-quality studies of bariatric surgery versus intensive glucose control. This review provides relevant background information related to the treatment of diabetes, hyperglycemia, and long-term complications; reports clinical findings (to date) with bariatric surgery; and identifies ongoing research focusing on long-term vascular outcomes associated with bariatric surgery.
Introduction
In May 2013 the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) and the National Heart, Lung, and Blood Institute (NHLBI) convened a multidisciplinary workshop to review systematically the long-term clinical outcomes related to bariatric surgery and, perhaps more importantly, to identify research gaps and future investigative opportunities (1,2). The consensus opinion of the expert panel was that information regarding the impact of bariatric surgery on microvascular and macrovascular long-term clinical outcomes was lacking. For example, at the time of this workshop (2013) there were no outcome data from long-term observational or controlled clinical trials on microvascular complications in patients with type 2 diabetes mellitus (T2DM) who had undergone bariatric surgeries. For cardiovascular events (specifically stroke and myocardial function), data from only two observational studies had been reported (2). Furthermore, a recent review of the effects of bariatric surgery on microvascular complications related to T2DM identified only 16 studies (3), the majority of which included small numbers of patients followed for fewer than 2 years. Of the reviewed studies, only one was population-based (4), and just one included a prospective matched cohort (5). Not included in this review was a recent study by Sjöström et al. (6) analyzing the association of bariatric surgery and microvascular and macrovascular complications over a 10-year follow-up of the Swedish Obesity Subjects (SOS) study. Recognizing the sparsity of high-quality evidence, the NIDDK/NHLBI workshop experts recommended that scientific efforts be made to advance the current understanding of the long-term effects of bariatric surgery on microvascular and macrovascular comorbidities of diabetes.
In view of this important recommendation, the objective of this article is to provide relevant background information, report clinical findings to date, and identify in-progress research in relation to microvascular and macrovascular outcomes following bariatric surgery. For clarification, reference to microvascular outcomes includes complications related to renal disease and diabetes-related eye disease and neuropathy; macrovascular outcomes include coronary artery, cerebrovascular, and peripheral vascular diseases. Diabetes-related and cardiovascular causes of death are also considered outcomes that can generally be related to microvascular and macrovascular outcomes.
Background
The escalating twin pandemics of obesity and T2DM are among the most devastating health crises (7–13). Almost 400 million people worldwide are estimated to be afflicted with T2DM, and that number is expected to approach 500 million by the year 2025 (14). Use of diets, exercise, and medications to promote weight loss and control blood glucose concentrations remain the cornerstones of T2DM therapy, and tight glycemic control does reduce microvascular complications (15). However, macrovascular complications and cardiovascular mortality remain difficult to address, even with intensive glucose-lowering therapy (16). Furthermore, despite an ever-increasing collection of pharmacotherapeutics, adequate glycemic control often remains elusive, and complete remission of T2DM is rare. Moreover, some diabetes medications may promote weight gain, and using sulfonylureas and insulin to achieve tight glycemic control is associated with an increased risk of hypoglycemia.
Numerous studies have documented that bariatric surgical procedures have profound effects on glycemic control among people with T2DM (17–21). An initial meta-analysis of 621 studies (mostly case series) of Roux-en-Y gastric bypass (RYGB), laparoscopic adjustable gastric banding (LAGB), and biliopancreatic diversion (BPD)/duodenal switch (DS) procedures found that most patients with diabetes who are undergoing these procedures experienced remission of their T2DM. Remission was defined as being off of diabetes medications, with fasting blood glucose concentration <100 mg/dL or HbA1c <6.0%. However, there were important differences in remission rates by procedures: RYGB 80% remission, LAGB 57% remission, and BPD/DS 95% remission (17). A more recent evidence-based review was restricted to bariatric surgery studies in which eligibility criteria included severely obese patients (BMI ≥35 kg/m2) and availability of more than 2 years of outcome data and follow-up measures for at least 80% of the initial cohort (22). Among the 7,371 studies identified, 29 met the inclusion criteria, and reported diabetes remission rates were 66.7% for RYGB (n = 428) and 28.6% for LAGB (n = 96). Following a systematic review of bariatric surgery–related randomized clinical trials (through 2012), Gloy et al. (23) identified 11 studies that included patients with T2DM and a BMI ≥35 kg/m2 who had bariatric surgery, including RYGB, LAGB, sleeve gastrectomy (SG), and BPD. All had short-term follow-up. Using these data, Gloy et al. conducted a meta-analysis (including 796 participants) and reported that patients who had bariatric surgery had a significantly greater likelihood for T2DM remission compared with nonsurgical cohorts (relative risk 22.1; 95% CI 3.2–154.3). Four of the 11 randomized controlled trials (RCTs) are continuing long-term follow-up of their cohorts, and three have published analyses at 3 years or beyond (24–27).
The STAMPEDE study—the first RCT to report on 3 years of follow-up—assessed clinical outcomes of 150 obese patients with uncontrolled T2DM who were randomly assigned to one of three groups: intensive medical therapy (IMT) alone or IMT combined with either RYGB or SG (25). With glycated hemoglobin ≤6.0% as the study’s primary end point, 5% of the IMT group met this criteria, compared with 38% of the RYGB group (P < 0.001) and 24% of the SG group (P = 0.01) (25). Five-year follow-up of an RCT by Mingrone et al. (26), which compared medical treatment (n = 20), RYGB (n = 20), and BPD (n = 20) among severely obese patients with at least 5 years’ diabetes duration before intervention, demonstrated that 37% (n = 7) and 63% (n = 12) of the RYGB and BPD groups, respectively, maintained diabetes remission compared with none (n = 15) of the medically treated group (P = 0.0007). One additional RCT with 3 years of follow-up was reported by Courcoulas et al. (27). Sixty-one obese patients with T2DM were randomized to intense lifestyle intervention, RYGB, or LAGB; partial or complete T2DM remission was reported for 40% (n = 8) of patients undergoing RYGB, 29% (n = 6) of patients undergoing LAGB, and none of the patients receiving the intense lifestyle intervention (P = 0.004). The favorable findings of T2DM remission following bariatric surgery have lead the Centers for Medicare & Medicaid Services to conclude that BPD/DS, RYGB, and LAGB are effective methods for treating T2DM (28).
RYGB and SG are currently the most common bariatric procedures performed in the United States. Although SG has often been performed on patients with a BMI ≥50 kg/m2 as an initial procedure after the patient underwent BPD/DS, using SG as the primary bariatric surgical procedure is a relatively recent process. As a result, long-term clinical outcome data related to SGs are limited. By contrast, the long history of RYGB as a sole bariatric/metabolic procedure, with demonstrated safety and evidence of long-term survival benefit (29,30), has influenced published reviews and guidelines to concur that RYGB is an effective therapy for weight loss and diabetes control (17,31). The LAGB procedure is purely restrictive; weight loss alone explains improvements in diabetes control after LAGB. However, RYGB engages additional antidiabetes mechanisms beyond those related to reductions in food intake and body weight (31). T2DM typically resolves within days to weeks after RYGB—long before major weight loss occurs (21)—and mounting evidence indicates that the remarkable antidiabetes impact of RYGB is mediated by several weight-independent mechanisms, including changes in gut hormones (e.g., ghrelin, glucose-dependent insulinotropic polypeptide, glucagon-like peptide 1, and peptide YY) that constitute the enteroinsular axis (32–38). However, Jackness et al. (39) reported short-term (21 days) improvement in insulin sensitivity and β-cell function in severely obese patients with diabetes, as well as rapid weight loss from following a very low–calorie diet (VLCD). In that study patients with T2DM undergoing RYGB (n = 11) were mean-matched for BMI, HbA1c, and diabetes duration with patients (n = 14) following a VLCD. Following equal weight loss 21 days after therapy (i.e., surgery or VLCD), improved glycemic control was similar in the two groups (39). Much less is known about the long-term risks and benefits of other bariatric procedures in treating T2DM (i.e., BPD/DS and SG). Additional postulated mechanisms related to bariatric/metabolic surgery and T2DM remission are detailed within this special issue.
Durability of Diabetes Remission
Despite the mounting evidence of the glycemia-lowering effects of bariatric surgery in patients with T2DM, the longest follow-up study has shown that remission of T2DM after bariatric surgery is not durable in all patients. The SOS study, including more than 4,000 matched obese patients receiving either nonsurgical care or bariatric surgery, demonstrated that 72% experienced T2DM remission 2 years after surgery, but only 36% remained free of T2DM at the 10-year follow-up (40). While most patients in the SOS study underwent the outdated vertical banded gastroplasty, small case series have confirmed a similar rate of T2DM relapse among patients undergoing RYGB, and weight regain is significantly associated with relapse (41,42). Arterburn et al. (43) conducted a large, multisite study investigating the long-term remission and relapse of T2DM in 4,353 patients undergoing RYGB who, before their surgery, were diagnosed with uncontrolled or medication-controlled T2DM, defined as HbA1c ≥6.5% or HbA1c <6.5% while taking diabetes medication preoperatively. While 68.2% (95% CI 66–70) of patients experienced complete diabetes remission within 5 years following RYGB, 35.1% (95% CI 32–38) of patients in remission redeveloped T2DM within 5 years. In this long-term retrospective study, important predictors of T2DM relapse included poor preoperative glycemic control, insulin use, and longer T2DM duration (43). Blackstone et al. (44) conducted a retrospective analysis of prospectively collected data from patients with T2DM undergoing RYGB (n = 483). They found that diabetes remission rates (remission was defined as no diabetes medication, fasting glucose <100 mg/dL, and HbA1c <5.7%) 14 months after surgery were better among patients not taking insulin therapy (before surgery) compared with those taking insulin (53.8% vs. 13.5%; P < 0.001). In addition, among the patients who were treated with insulin before surgery (n = 126), those whose diabetes duration before surgery was less than the median duration of 4 years had significantly greater diabetes remission rates compared with patients with diabetes duration >4 years (50% vs. 9%; P < 0.001) (44). In summary, although bariatric/metabolic surgery, and RYGB in particular, seems to induce an initial remission of T2DM in most severely obese adults, many of these patients will relapse to overt T2DM at some point after their initial remission.
Studies such as the SOS and the large, multicenter findings of Arterburn et al. (43) demonstrate that RYGB does not permanently reverse T2DM. However, patients with shorter duration of T2DM, those not requiring insulin, and those with good glycemic control are most likely to achieve long-term remission of their disease and avoid relapse. This outcome likely relates to the patient’s residual β-cell function at the time of surgery.
Most of the evidence of the long-term effects of bariatric procedures on glycemic control comes from observational studies, and long-term randomized clinical trials (currently absent from the literature) are needed to confirm these findings. Despite the sometimes transient effects of bariatric surgery on T2DM remission, adults who experience a relapse of T2DM may still benefit from surgery; further study is needed to understand what effect a transient period of T2DM remission might have on insulin secretion, long-term glycemic control, medication requirements, and the subsequent incidence of microvascular complications, such as nephropathy, retinopathy, and cardiovascular disease (CVD). Two studies of IMT, the UK Prospective Diabetes Study (UKPDS) in T2DM and the Epidemiology of Diabetes Interventions and Complications (EDIC) study in type 1 diabetes mellitus, have shown that temporary periods of superior glycemic control resulted in significant long-term benefits in terms of reduced microvascular and macrovascular complications (45,46). Most importantly, these benefits persisted even though the levels of glycemic control between intervention and control subjects eventually became identical after the active intervention period ended. The prolonged beneficial effects of improved glycemic control observed in these studies have been termed the legacy effect or metabolic memory. Whether a similar legacy effect follows bariatric surgery—if glycemia first improves and then relapses—is unknown. Initial improvement in microvascular disease has yet to be firmly established by controlled clinical trials. If bariatric surgery–related observational studies hold true in future clinical trials, however, then there would be reason to believe that even a short period of intensive glycemic control induced by bariatric surgery might have lasting benefits (i.e., a legacy effect may be expected).
Conceptual Model: Microvascular and Macrovascular Complications
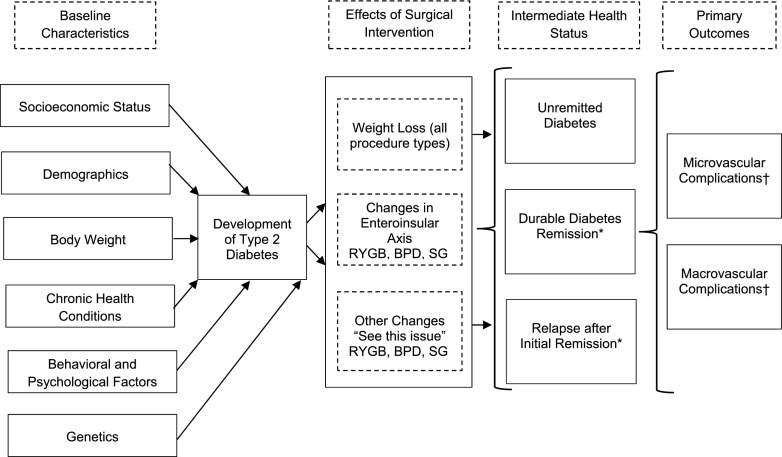
The conceptual model for long-term outcomes of surgical treatment for diabetes and severe obesity (i.e., macrovascular and microvascular outcomes as primary end points) is graphically represented in Fig. 1 and reflects numerous related studies (32–38,47–54), as well as physiological mechanistic changes after bariatric surgery (highlighted in this special issue) that may contribute to diabetes remission/relapse and eventual microvascular and macrovascular outcomes. In this model the development of diabetes and severe obesity are influenced by socioeconomic status, demographics, body weight, other chronic health conditions, genetics, and numerous behavioral and psychological factors. When a patient with T2DM and severe obesity undergoes bariatric/metabolic surgery, the type of procedure performed determines whether the improvements in glycemia are mediated by weight loss alone or through other mechanisms that are unique to the intervention itself, that is, changes in gut hormones (e.g., ghrelin, glucose-dependent insulinotropic polypeptide, glucagon-like peptide 1, and peptide YY) that constitute the enteroinsular axis or other physiological changes that occur after surgery (these are highlighted in this special issue). Current evidence suggests that the amount of time spent in each of three intermediate health states—1) unremitted diabetes, 2) durable diabetes remission, and 3) diabetes remission followed by relapse—could significantly affect the incidence of microvascular and macrovascular complications, which can in turn influence a patient’s quality of life and longevity (45,46,55).
Figure 1.
Conceptual model for outcomes of surgical treatment for diabetes and severe obesity. “See this issue” refers to physiological mechanistic changes after bariatric surgery (as highlighted in this special issue) that may contribute to diabetes remission/relapse and eventual microvascular and macrovascular outcomes. *May be associated with average glycemia duration. †Amelioration of hypertension and dyslipidemia through weight loss can also affect complication risk.
Microvascular and Macrovascular Complications: Review
Because long-term bariatric/metabolic studies (i.e., ≥5 years) whose primary outcomes are microvascular and macrovascular complications are primarily observational and do not include randomized control trials, whether bariatric surgery ameliorates these outcomes when compared with current conventional therapy cannot, unfortunately, be determined because the evidence is insufficient. However, favorable 3-year follow-up results from the STAMPEDE randomized clinical trial (25) encourage the opportunity (and need) to conduct studies (i.e., RCTs and large, retrospectively matched studies) to determine whether bariatric surgical procedures reduce microvascular and macrovascular complications compared with nonsurgical intensive glucose therapy. In support of this proposed research, this section briefly reviews both bariatric/metabolic surgery and nonsurgical studies related to microvascular and macrovascular complications.
IMT (Nonsurgical)
A recent review related to advances in the diagnosis and treatment of diabetes (56) supported evidence that medical treatment strategies for achieving long-term glycemic control are safe and show a consistent reduction in the risk of microvascular disease (15,45,57–61). These studies, involving large populations of patients with diabetes who are randomly assigned to various glucose control therapies (i.e., standard vs. intensive glucose control) have included the Diabetes Control and Complications Trial (57) and its long-term follow-up in the Epidemiology of Diabetes Interventions and Complications study (61), the UKPDS (15,45), the glycemic arm of the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial (59), and the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) trial (60). Clinical benefits resulting from intensive treatment aimed at achieving near-normal glycemia seem to persist well beyond the actual treatment period (i.e., the previously mentioned metabolic memory or legacy effect) (45,62). Furthermore, intensive glycemic management of T2DM has resulted in varied cardiovascular outcomes (56): two studies demonstrated a reduction (15–17%) in CVD (45,63), one study reported no benefit in CVD (60), and another suggested increased mortality as a result of treatment (64). While the incidence of diabetes has increased worldwide, this careful review of nonsurgical diabetes diagnosis and treatment concludes that efforts to reduce risk factors are increasing (65), whereas overall renal disease and CVD occurring among the population with diabetes “have been reduced substantially in the past decade” (56,66).
Bariatric/Metabolic Surgical Therapy
Highlighted in Table 1 are bariatric surgery–related studies that have reported short- and long-term changes in microvascular and macrovascular outcomes. Abstracted data related to patient population, inclusion of control participants, and microvascular and macrovascular outcomes are identified in this table. With the exception of the STAMPEDE trial (25), a 3-year RCT that reports on renal outcomes, the remainder of the referenced studies are prospective cohort trials (5,6,67,68), observational prospective trials (69–71), prospective pilot studies (72), retrospective cohort studies (4,73–88), case-control trials (89), or case reports (90–92).
Table 1.
Review of studies related to bariatric surgery and microvascular- and macrovascular-related outcomes, including long-term mortality specifically related to diabetes and cardiovascular disease
| Reference | Type | Surgical patients, n (M/F) | Control subjects, n (M/F) | Follow-up (years) |
Type(s) of surgery | Microvascular and macrovascular results | Comments | |
|---|---|---|---|---|---|---|---|---|
| Surgery | Control | |||||||
| Eliasson et al. (96) |
Observational prospective study |
6,132 (2,364/3,758) |
6,132 (2,364/3,758) |
3.5 |
3.5 |
RYGB |
All-cause mortality (HR 0.42 [95% CI 0.30–0.57]); CV-caused mortality (HR 0.41 [95% CI 0.19–0.90]) |
All patients with diabetes before bariatric surgery and matched nonsurgical controls with diabetes; surgeries in Sweden |
| Carlsson et al. (5) |
Prospective cohort |
2,010 (590/1,420); 1,498 without baseline albuminuria |
2,037 (590/1,447); 1,610 without baseline albuminuria |
10.0 (median) |
10.0 (median) |
VBG, band, RYGB |
Primary outcome: incidence of albuminuria (surgical vs. controls, HR 0.37 [95% CI 0.30–0.47]) |
Only prospective long-term study; pre- and postclinical data available; analysis included surgical and control patients without baseline albuminuria; surgeries in Sweden |
| Chen et al. (73) |
Retrospective cohort |
173; data on 78 patients (16/62) |
80 (17/63) |
11.0 (median) |
11.0 (median) |
RYGB |
Surgical vs. controls: microvascular complications, 46.3% vs. 11.5% (P < 0.01); macrovascular complications, 20.3% vs. 5% (P < 0.01) |
All patients (surgery and control) had diagnosed diabetes; control patients were matched for BMI, age, race, and severity of diabetes; surgeries in the U.S. |
| Schauer et al. (25) |
Randomized clinical trial |
97 (baseline: RYGB, 58% female; SG, 78% female) |
40 (baseline: 62% female); 2,037 (590/1,447) |
3.0 |
3.0 |
RYGB (n = 48), SG (n = 49) |
Outcome: UACR and change in albuminuria. Surgical vs. controls: reduction in the UACR (P < 0.04; both surgery groups); of patients with baseline albuminuria, 62% (8 of 13; P = 0.04 for within-group comparison), 80% (8 of 10; P = 0.11), and 25% (1 of 4; P = 1.00) for the RYGB, SG, and IMT groups, respectively, returned to normal values |
All patients (surgery and control) had uncontrolled diabetes; controls underwent IMT; surgeries in the U.S. |
| Sjöström et al. (6) |
Prospective cohort |
2,010 (590/1,420); 343 (141/202) with diabetes at baseline |
2,037 (590/1,447); 260 (104/156) with diabetes at baseline |
17.6 (median) |
18.1 (median) |
VBG, band, RYGB |
Surgical vs. controls: microvascular, adjusted HR 0.44 (95% CI 0.34–0.56); macrovascular, adjusted HR 0.68 (95% CI 0.54–0.85) |
Only prospective long-term study; pre- and postclinical data available; analysis included surgical and control patients with baseline diabetes; surgeries in Sweden |
| Johnson et al. (4) |
Population-based, retrospective cohort |
2,580 (593/1,987) |
13,371 (4,359/9,012) |
13.0 |
13.0 |
RYGB, band, VBG, BD, SG |
Surgical vs. controls: microvascular, adjusted HR: 0.22 (95% CI 0.09–0.49); macrovascular adjusted HR 0.39 (95% CI 0.29–0.51); other vascular events, adjusted HR 0.25 (95% CI 0.19–0.32) |
All patients (surgery and control) had diabetes before surgery; controls had moderate to severe obesity; surgeries in the U.S. |
| Scott et al. (74) |
Population-based, retrospective cohort |
4,747 (808/3,939) |
4,393 (1,020/3,373) |
12.0 |
12 |
RYGB, band |
Primary outcome: time-to-occurrence of MI, stroke, or death. BAR vs. orthopedic surgery controls: HR 0.72 (95% CI 0.58–0.89); BAR vs. GI surgery controls: HR 0.48 (95% CI 0.39–0.61) |
Patients excluded if previous MI or stroke; control patients had previously undergone either orthopedic (n = 3,066) or nonbariatric GI (n = 1,327) surgery; surgeries in the U.S. |
| Amor et al. (69) |
Observational prospective study |
255 (74/181) |
No control patients |
1.0 and 2.0 |
— |
RYGB, SG |
Primary outcome: UACR. In patients with diabetes, UACR reduced 42.2 ±142.8 mg/g (P < 0.005) at 1 year, with no additional change at 2 years |
Of bariatric surgical patients, 37.6% (n = 96) had diabetes before surgery; surgeries in Spain |
| Fenske et al. (70) |
Observational prospective study |
34 (5/29) |
— |
1.0 |
— |
RYGB, band, SG |
UACR deceased, 4.1 ± 0.3 (baseline) to 0.9 ± 0.04 (P < 0.01); eGFR increased, 67.4 ± 1.0 to 85.0 ± 2.0 mL/min/1.73 m2 (P < 0 0.001); serum CR decreased, 74.4 ± 2.2 to 60 ± 1.1 µmol/L (P < 0.001) |
All patients followed at baseline, 1 month, and 12 months; patients excluded for significant chronic kidney disease (eGFR <60 mL/min/1.73 m2); surgeries in Europe |
| Brethauer et al. (75) |
Retrospective cohort |
217 (58/159) |
— |
6.0 (median) |
— |
RYGB, band, SG |
Outcomes: UACR and serum CR. Change in CR, 0.05 ± 0.2 mg/dL (P < 0.039); DN regressed in 10 patients (53%) and remained stable in 9 (47%) |
All patients diagnosed with diabetes (before surgery); renal-related outcomes were analyzed in 59 patients; DN data relate to RYGB patients; surgeries in the U.S. |
| Stephenson et al. (76) |
Retrospective cohort |
23 (12/11) |
— |
3.0 |
— |
Band |
Among patients with baseline microalbuminuria (n = 16), 9 reverted to normoalbuminuria, 1 progressed from micro- to macroalbuminuria |
All patients diagnosed with diabetes (before surgery); patients presented with micro- (n = 16) or macroalbuminuria (n = 7); surgeries in Australia |
| Jose et al. (77) |
Retrospective cohort |
25 (8/17) |
— |
3.9 |
— |
BD |
Outcomes: serum CR and eGFR. Serum CR decreased 16.23 ± 19.57 µmol/L (P < 0.001); eGFR increased 10.6 ± 15.45 mL/min/1.73 m2 (P = 0.048) |
Patients with extreme BMI (≥40 kg/m2); surgeries in the U.K. |
| Hou et al. (78) |
Retrospective cohort |
233 (91/142) |
— |
1.0 |
— |
RYGB, band, SG, miniGBP |
Primary outcome: GFR. GFR decreased to 133.9 ± 25.7 mL/min in the hyperfiltration group (P < 0.05), increased to 114.2 ± 22.2 mL/min in the normal group, increased to 93.3 ± 20.4 mL/min in the CKD stage 2 group (P < 0.05), increased to 66.8 ± 19.3 mL/min in the CKD stage 3 group (P < 0.05) |
90% of patients diagnosed with diabetes (before surgery); patients stratified by GFR: hyperfiltration (GFR >125 mL/min), normal (GFR 125–90 mL/min), CKD stage 2 (GFR 89–60 mL/min), and CKD stage 3 (GFR 59–30 mL/min); surgeries in China |
| Heneghan et al. (79) |
Retrospective cohort |
52 (13/39) |
— |
5.5 |
— |
RYGB, band, SG |
Primary outcome: DN, indicated by micro- (30–300 mg/g) or macroalbuminuria (>300 mg/g). DN resolved in 58.3%; non-DN incidence in 25% |
All patients diagnosed with diabetes (before surgery), with mean duration of 8.6 years, and 37.6% with albuminuria before surgery; surgeries in the U.S. |
| Johnson et al. (97) |
Retrospective cohort |
349 (123/226) |
903 (334/569) |
2.3 |
2.9 |
RYGB, band |
Primary outcome: all-cause mortality HR 0.60 (95% CI 0.36–0.99) |
All surgical and nonsurgical subjects with diagnosed CV event of MI or stroke (before surgery); control data extracted from hospital records; surgeries in the U.S. |
| Sjöström et al. (67) |
Prospective cohort |
2,010 (590/1,420) |
2,037 (590/1,447) |
14.7 |
14.7 |
VBG, band, RYGB |
Surgical vs. controls: CV-caused mortality (HR 0.47 [95% CI 0.29–0.76]); first time (fatal or nonfatal) CV event (MI or stroke): adjusted HR 0.67 (95% CI, 0.54–0.83) |
Only prospective long-term mortality study; pre- and postclinical data available; surgeries in Sweden |
| MacLaughlin et al. (80) |
Retrospective review |
9 (3/6) |
— |
0.5 |
— |
SG |
4 of 5 hemodialysis patients added to kidney transplantation wait list because of weight loss |
All patients diagnosed with CKD before surgery, with 5 undergoing hemodialysis; surgeries in the U.K. |
| Varadhan et al. (81) |
Retrospective cohort (pilot) |
119 (sex not listed) |
— |
1.0 |
— |
RYGB, SG |
Primary outcome: DR. 38 patients had DR before surgery; 2 had DR incidence, 13 had no DR before and after surgery, 2 had no regression of DR after surgery |
All patients diagnosed with diabetes (before surgery); DR classified as background, preproliferative, or proliferative with or without maculopathy; surgeries in the U.K. |
| Lammert et al. (71) |
Observational prospective study |
30 (11/19) |
— |
0.8 |
— |
RYGB, SG, band, BD |
Primary outcome: ED in retinal vessels. Retinal AVR improved (+0.04; P < 0.0001), flicker response to light showed no significant change |
ED measured by AVR for evaluation of vascular pathology; flicker response to light also measured for ED; surgeries in Germany |
| Miras et al. (82) |
Retrospective cohort |
84 (sex not listed) |
— |
1.5 |
— |
RYGB, SG, band |
Primary outcomes: RD, DR, and renal complications. RD improved (n = 5), worsened (n = 1), and did not change (n = 61); DR improved (n = 5), worsened (n = 1), and did not change (n = 22); retinopathy scores decreased, 4.7 ± 0.6 to 3.3 ± 0.5 (P < 0.004); UACR improved, 7.6 (4.7–24.5) to 2.2 (1.0–17.3) mg/mmol (P < 0.001) |
All patients diagnosed with diabetes (before surgery); measures included retinal photographs (two-field) and UACRs; before surgery, 67 patients had full RD data and 28 patients had DR; 32 patients had preoperative albuminuria; surgeries in the U.K. |
| Iaconelli et al. (89) |
Unblinded, case-control trial |
22 (10/12) |
28 (12/16) |
10.0 |
10.0 |
BD |
Primary outcome: GFR. GFR variation was −45.7 ± 18.8% in the medical arm and 13.6 ± 24.5% in the surgical arm (P < 0.001) |
Consecutive sample of 110 severely obese patients with newly diagnosed diabetes (study completed by 50 patients); controls received conventional therapy; surgeries in Italy |
| Schuster et al. (83) |
Retrospective cohort |
813 (187/626) |
— |
∼2.0 |
— |
RYGB |
Primary outcome: serum CR. 76.7% of patients with impaired CR (n = 56) at ∼2 years had normal CR, 12.5% had a CR concentration 1.3–1.6 mg/dL, and 10.7% had CR >1.6 mg/dL |
Baseline CR groups: normal (<1.3 mg/dL; n = 757), mild impairment (1.3–1.6 mg/dL; n = 40), moderate impairment (>1.6 mg/dL; n = 16); 68% of patients with impaired CR had diabetes; surgeries in the U.S. |
| Navaneethan et al. (72) |
Prospective pilot |
14 (8/6) |
— |
0.5 |
— |
RYGB (n = 9), SG (n = 4), band (n = 2) |
Outcome: UACR and adipokine and insulin sensitivity. Pre- to post-surgery: RYGB showed only UACR reduction (median, 36 mg/g [7–94] vs. 27 mg/g [5.5–42.5]) |
All bariatric surgical patients had diabetes before surgery; surgeries in the U.S. |
| Szomstein et al. (90) |
Retrospective case report |
5 (0/5) |
— |
2.0 |
— |
RYGB, SG |
No postoperative bariatric surgery complications in any patient; no dosage alteration of immunosuppressant drugs recorded after bariatric surgery |
Patients who had previously had a kidney transplant subsequently underwent bariatric surgery; 2 patients had diabetes; surgeries in the U.S. |
| Agrawal et al. (84) |
Retrospective cohort |
62 (11/51) |
— |
1.3 |
— |
RYGB |
Primary outcome: UACR and hsCRP. UACR reduced 47.7% (interquartile range, −74.0 to 0); hsCRP reduced 57.3% (± 32.5) |
Patients has no overt CV or renal disease; 40% had diabetes before surgery; surgeries in the U.S. |
| Alexander et al. (85) |
Retrospective cohort |
45 (sex not listed) |
— |
2.0–5.0 |
— |
RYGB |
14 patients proceeded to kidney transplant; of 31 without transplant, 9 patients had resolution, improvement, or stabilization of their kidney function; 2 of these 9 patients were already receiving, or were ready for, dialysis |
At baseline, all patients had not had transplants but had established renal disease and had undergone RYGB; 22 patients already required dialysis; surgeries in the U.S. |
| Navaneethan and Yehnert (86) |
Retrospective cohort |
25 (7/18) |
— |
1.0–2.0 |
— |
Restrictive or malabsorptive |
Primary outcome: renal function using eGFR. At 1 year, eGFR improved, 47.9 to 61.6 mL/min/1.73 m2 (P < 0.001) |
Patients who had bariatric surgery had stage 3 CKD (GFR 30–59 mL/min/1.73 m2); postoperative acute renal failure patients excluded; surgeries in the U.S. |
| Takata et al. (87) |
Retrospective review |
7 (0/7) |
— |
1.5 |
— |
RYGB |
All 7 patients went on to qualify for transplantation after surgery |
Patients had end-stage renal disease before RYGB; 3 patients had diabetes; surgeries in the U.S. |
| Koshy et al. (91) |
Case report |
3 (2/1) |
— |
∼2.0 |
— |
Band |
After banding, all patients lost sufficient weight to qualify for kidney transplantation; all patients subsequently had successful uncomplicated kidney transplantations |
Patients had end-stage renal disease prior to band surgery; surgeries in Australia |
| Sjöström et al. (98) |
Prospective cohort |
2,010 (590/1,420) |
2,037 (590/1,447) |
10.9 |
10.9 |
VBG, band, RYGB |
Surgical vs. controls: all-cause mortality (HR 0.76 [95% CI 0.59–0.99]; P = 0.04); MI most common cause of death: 13 deaths in the surgery group, 25 deaths in the control group |
Only prospective long-term mortality study; pre- and postclinical data available; surgeries in Sweden |
| Adams et al. (30) |
Retrospective cohort |
7,925 (1,268/6,657) |
7,925 (1,268/6,657) |
7.1 |
7.1 |
RYGB |
Surgical vs. controls: diabetes (HR 0.10 [95% CI 0.02–0.46]); coronary artery disease (HR 0.41 [95% CI 0.20–0.65]) |
Control data extracted from drivers license applications; surgeries in the U.S. |
| Navarro-Díaz et al. (68) |
Prospective cohort |
61 (24/37) |
24 (9/15) |
2.0 |
— |
RYGB |
Outcomes: renal parameters and renal function. At 1 year compared with baseline, serum CR, CR clearance, proteinuria, albuminuria, and microhematuria were all significantly improved in the surgical group |
Bariatric surgical patients were extremely obese and had normal serum CR and no renal disease; controls were normal-weight (only baseline data for controls); surgeries in Spain |
| Newcombe et al. (92) |
Case reports |
3 (3/0) |
— |
2.0 |
— |
Band |
Following weight loss after surgery, all 3 patients underwent successful renal transplantation |
Before bariatric surgery, 2 patients had chronic renal failure and 1 had end-stage renal failure; two had diabetes; surgeries in Israel |
| Schauer et al. (88) |
Retrospective cohort |
191 (47/144) |
— |
1.8 |
— |
RYGB |
Before surgery patients had DN (25%); CAD (12%); retinopathy (10%); and nephropathy (7%); after surgery occurred DN symptom improvement in 50% of affected patients |
All patients had diabetes before surgery; 54% of patients were “super obese;” surgeries in the U.S. |
| MacDonald et al. (95) | Retrospective cohort | 154 (36/118) | 78 (21/57) | 9.0 | 6.2 | RYGB | Surgical mortality rate 9% vs. 28% nonsurgical rate | All patients (surgery and control) had diabetes before surgery; all seeking surgery; surgeries in the U.S. |
AVR, arteriole-to-venule ratio; band, gastric band (adjustable or not); BAR, bariatric; CAD, coronary artery disease; CKD, chronic kidney disease; CR, creatinine; CV, cardiovascular; DN, diabetic nephropathy; DR, diabetic retinopathy; ED, endothelial dysfunction; eGFR, estimated glomerular filtration rate; GFR, glomerular filtration rate; GI, gastrointestinal; HR, hazard ratio; MI, myocardial infarction; miniGPB, mini gastric bypass; RD, retinal disease; UACR, urinary albumin-to-creatinine ratio; VBG, vertical banded gastroplasty.
As highlighted by Johnson et al. (4), noted in Table 1, and referenced in a review article of bariatric surgery and microvascular complications by Jackson et al. (3), a number of recently reported studies (many of which were small and uncontrolled) demonstrated favorable outcomes in renal function markers following bariatric/metabolic surgery in patients with or without the presence of impaired renal function before their bariatric surgery (68,70,75–78,83–86). Finally, patients who are waiting for or who have had renal transplantation experienced improved renal function following bariatric surgery (80,87,90–92), and a few bariatric surgery studies demonstrated varying degrees of improvement in neuropathy and ophthalmic outcomes (4,71,79,81,82,88,93).
Long-term Mortality (Macrovascular and Microvascular Related)
Deaths caused by diabetes and CVD have been considered as primary microvascular and macrovascular disease outcomes. In addition to mortality findings, bariatric studies have specifically reported macrovascular end points such as myocardial infarction or stroke. Based on a recent review, 28 studies with at least a 2-year follow-up have reported mortality rates following bariatric surgery (94). Of these studies, two in particular investigated the long-term mortality of bariatric surgery with patients with T2DM before surgery compared with control patients who also had diabetes (95,96) (Table 1).
One long-term mortality study was unique in that all patients undergoing bariatric surgery and severely obese control subjects had established CVD (97) (Table 1). Sjöström et al. (98) reported myocardial infarction to be the most common cause of death in the SOS study (13 deaths in the surgery group, 25 deaths in the control group) (Table 1). Finally, long-term mortality analysis in a Utah study reported a 56% reduction in coronary artery disease among the RYGB group when compared with control subjects (30) (Table 1).
Microvascular and Macrovascular Outcome Studies in Progress
At least two large, retrospective studies being conducted specifically focus on the long-term impact of bariatric surgery on microvascular and macrovascular outcomes. The largest study is a National Institutes of Health/NIDDK–funded study conducted by Arterburn and colleagues. That study is using existing electronic medical databases to examine long-term microvascular and macrovascular outcomes among severely obese patients with T2DM who underwent RYGB surgery under one of four health plans from 2000–2011 (approximately 6,000 patients with T2DM). The collaborative effort of this research is derived from the Health Care Systems Research Network, a consortium of integrated research divisions representing multiple health systems in the United States; four of these systems are participating in this bariatric surgery study that is now being conducted by Arterburn and his colleagues.
The second ongoing study is funded by Ethicon Endo-Surgery (Johnson & Johnson) and the Intermountain Research and Medical Foundation (Intermountain Healthcare). Using data from the electronic medical records of two large Utah health systems and the Utah Population Database, this retrospective study is examining microvascular and macrovascular outcomes of approximately 9,000 patients who had previously undergone RYGB surgery as well as nonoperated control groups with various BMI ranges (no bariatric surgery). While the cohort is not limited to patients who had T2DM before their bariatric surgery, subgroup analysis will be conducted using the T2DM cohort.
Finally, the Alliance of Randomized Trials of Medicine vs. Metabolic Surgery in Type 2 Diabetes (ARMMS-T2D) is an ongoing prospective study examining the long-term outcomes of four National Institutes of Health–funded RCTs comparing bariatric surgery versus intensive medical/lifestyle intervention in patients with T2DM. This follow-up study is funded by Ethicon Inc. and Covidien Ltd., as well as by the NIDDK, and is prospectively assessing the impact of bariatric procedures on T2DM in over 300 patients with mild to moderate obesity (BMI 27–45 kg/m2) over 7 years. The main study outcomes will be differences in the long-term rates of T2DM remission and relapse, changes in glycemic control, and rates of microvascular and macrovascular disease among the surgical and nonsurgical groups.
Conclusions
Although bariatric surgery (RYGB and BPD in particular) seems to induce initial remission of T2DM in most severely obese adults, many of these patients relapse to overt T2DM at some point after their initial remission. While numerous RCTs have compared the results of standard versus intensive glucose control in patients with diabetes, outcomes data on microvascular and macrovascular complications after bariatric surgery are limited to observational studies and long-term nonrandomized controlled studies. For this reason, whether bariatric surgery is associated with a long-term reduction in microvascular and macrovascular complications when compared with current medical interventions is unknown. Although early findings of one RCT (3-year follow-up data) have suggested microvascular benefits of bariatric procedures and numerous nonrandomized controlled studies have demonstrated significant diabetes remission or glycemic improvement following bariatric surgery, the relative effects of bariatric procedures on microvascular and cardiovascular complications compared with modern-day diabetes therapy must still be established. High-quality RCTs are necessary to define the impact of bariatric surgery on long-term microvascular and macrovascular outcomes.
Article Information
Funding. T.D.A. has received research funding from the National Institutes of Health and the NIDDK. D.E.A. has received institutional research grants and payments for travel expenses related to bariatric surgery from the National Institutes of Health, the Patient-Centered Outcomes Research Institute, the Department of Veterans Affairs, and the Informed Medical Decisions Foundation (now a division of Healthwise).
Duality of Interest. T.D.A. reports research funding from Ethicon Endo-Surgery, a subsidiary of Johnson & Johnson. No other potential conflicts of interest relevant to this article were reported.
Footnotes
References
- 1.Courcoulas AP, Yanovski SZ, Bonds D, et al. Long-term outcomes of bariatric surgery: a National Institutes of Health symposium. JAMA Surg 2014;149:1323–1329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Courcoulas AP, Yanovski SZ, Bonds D, et al. Long-term outcomes of bariatric surgery: a National Institutes of Health symposium. JAMA Surg 2014;149:1323–1329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jackson S, le Roux CW, Docherty NG. Bariatric surgery and microvascular complications of type 2 diabetes mellitus. Curr Atheroscler Rep 2014;16:453. [DOI] [PubMed] [Google Scholar]
- 4.Johnson BL, Blackhurst DW, Latham BB, et al. Bariatric surgery is associated with a reduction in major macrovascular and microvascular complications in moderately to severely obese patients with type 2 diabetes mellitus. J Am Coll Surg 2013;216:545–556; discussion 556–558 [DOI] [PubMed] [Google Scholar]
- 5.Carlsson LM, Romeo S, Jacobson P, et al. The incidence of albuminuria after bariatric surgery and usual care in Swedish Obese Subjects (SOS): a prospective controlled intervention trial. Int J Obes (Lond) 2015;39:169–175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sjöström L, Peltonen M, Jacobson P, et al. Association of bariatric surgery with long-term remission of type 2 diabetes and with microvascular and macrovascular complications. JAMA 2014;311:2297–2304 [DOI] [PubMed] [Google Scholar]
- 7.Arterburn DE, Alexander GL, Calvi J, et al. Body mass index measurement and obesity prevalence in ten U.S. health plans. Clin Med Res 2010;8:126–130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Arterburn DE, Crane PK, Sullivan SD. The coming epidemic of obesity in elderly Americans. J Am Geriatr Soc 2004;52:1907–1912 [DOI] [PubMed] [Google Scholar]
- 9.Flegal KM, Carroll MD, Ogden CL, Curtin LR. Prevalence and trends in obesity among US adults, 1999–2008. JAMA 2010;303:235–241 [DOI] [PubMed] [Google Scholar]
- 10.Flegal KM, Carroll MD, Ogden CL, Johnson CL. Prevalence and trends in obesity among US adults, 1999–2000. JAMA 2002;288:1723–1727 [DOI] [PubMed] [Google Scholar]
- 11.Bray GA, Bellanger T. Epidemiology, trends, and morbidities of obesity and the metabolic syndrome. Endocrine 2006;29:109–117 [DOI] [PubMed] [Google Scholar]
- 12.Cohen JW, Krauss NA. Spending and service use among people with the fifteen most costly medical conditions, 1997. Health Aff (Millwood) 2003;22:129–138 [DOI] [PubMed] [Google Scholar]
- 13.Mokdad AH, Bowman BA, Ford ES, Vinicor F, Marks JS, Koplan JP. The continuing epidemics of obesity and diabetes in the United States. JAMA 2001;286:1195–1200 [DOI] [PubMed] [Google Scholar]
- 14.International Diabetes Federation. IFD Diabetes Atlas, Sixth Edition. Brussels, Belgium, International Diabetes Federation, 2013
- 15.Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998;352:837–853 [PubMed] [Google Scholar]
- 16.American Diabetes Association Standards of medical care in diabetes--2010. Diabetes Care 2010;33(Suppl. 1):S11–S61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Buchwald H, Estok R, Fahrbach K, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med 2009;122:248–256.e5 [DOI] [PubMed] [Google Scholar]
- 18.Pories WJ, Dohm LG, Mansfield CJ. Beyond the BMI: the search for better guidelines for bariatric surgery. Obesity (Silver Spring) 2010;18:865–871 [DOI] [PubMed] [Google Scholar]
- 19.Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA 2004;292:1724–1737 [DOI] [PubMed] [Google Scholar]
- 20.Pories WJ, MacDonald KG Jr, Morgan EJ, et al. Surgical treatment of obesity and its effect on diabetes: 10-y follow-up. Am J Clin Nutr 1992;55(Suppl.):582S–585S [DOI] [PubMed] [Google Scholar]
- 21.Pories WJ, Swanson MS, MacDonald KG, et al. Who would have thought it? An operation proves to be the most effective therapy for adult-onset diabetes mellitus. Ann Surg 1995;222:339–350; discussion 350–352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Puzziferri N, Roshek TB 3rd, Mayo HG, Gallagher R, Belle SH, Livingston EH. Long-term follow-up after bariatric surgery: a systematic review. JAMA 2014;312:934–942 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gloy VL, Briel M, Bhatt DL, et al. Bariatric surgery versus non-surgical treatment for obesity: a systematic review and meta-analysis of randomised controlled trials. BMJ 2013;347:f5934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.The Cleveland Clinic. Alliance of randomized trials of medicine vs metabolic surgery in type 2 diabetes (ARMMS-T2D). In: ClinicalTrials.gov [Internet]. Bethesda, MD, National Library of Medicine, 2016. Available from https://clinicaltrials.gov/ct2/show/NCT02328599. NLM identifier: NCT02328599. Accessed 21 March 2016
- 25.Schauer PR, Bhatt DL, Kirwan JP, et al.; STAMPEDE Investigators . Bariatric surgery versus intensive medical therapy for diabetes--3-year outcomes. N Engl J Med 2014;370:2002–2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric-metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet 2015;386:964–973 [DOI] [PubMed] [Google Scholar]
- 27.Courcoulas AP, Belle SH, Neiberg RH, et al. Three-year outcomes of bariatric surgery vs lifestyle intervention for type 2 diabetes mellitus treatment: a randomized clinical trial. JAMA Surg 2015;150:931–940 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Phurrough S, Salive ME, Brechner RJ, Meltzer A. Decision memo for surgery for diabetes (CAG-00397N) [Internet], 2009. Baltimore, MD, Centers for Medicare & Medicaid Services. Available from https://www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=219&NCDId=326&ncdver=1&NcaName=Surgery+for+Diabetes&IsPopup=y&bc=AAAAAAAACAAAAA%3D%3D&. Accessed 21 March 2016 [Google Scholar]
- 29.Flum DR, Belle SH, King WC, et al.; Longitudinal Assessment of Bariatric Surgery (LABS) Consortium . Perioperative safety in the longitudinal assessment of bariatric surgery. N Engl J Med 2009;361:445–454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med 2007;357:753–761 [DOI] [PubMed] [Google Scholar]
- 31.Rubino F, Kaplan LM, Schauer PR, Cummings DE; Diabetes Surgery Summit Delegates . The Diabetes Surgery Summit consensus conference: recommendations for the evaluation and use of gastrointestinal surgery to treat type 2 diabetes mellitus. Ann Surg 2010;251:399–405 [DOI] [PubMed] [Google Scholar]
- 32.Cummings DE. Gastric bypass and nesidioblastosis--too much of a good thing for islets? N Engl J Med 2005;353:300–302 [DOI] [PubMed] [Google Scholar]
- 33.Cummings DE. Endocrine mechanisms mediating remission of diabetes after gastric bypass surgery. Int J Obes (Lond) 2009;33(Suppl. 1):S33–S40 [DOI] [PubMed] [Google Scholar]
- 34.Cummings DE, Flum DR. Gastrointestinal surgery as a treatment for diabetes. JAMA 2008;299:341–343 [DOI] [PubMed] [Google Scholar]
- 35.Thaler JP, Cummings DE. Minireview: Hormonal and metabolic mechanisms of diabetes remission after gastrointestinal surgery. Endocrinology 2009;150:2518–2525 [DOI] [PubMed] [Google Scholar]
- 36.Cummings DE, Foster-Schubert KE, Carlson MJ, Shannon MH, Overduin J. Possible hormonal mechanisms regulating the effects of bariatric surgery. In Obesity Surgery: Principle and Practice. Pitombo C, Ed. New York, McGraw-Hill, 2007, p. 137–147 [Google Scholar]
- 37.Cummings DE, Overduin J, Foster-Schubert KE. Gastric bypass for obesity: mechanisms of weight loss and diabetes resolution. J Clin Endocrinol Metab 2004;89:2608–2615 [DOI] [PubMed] [Google Scholar]
- 38.Cummings DE, Overduin J, Foster-Schubert KE, Carlson MJ. Role of the bypassed proximal intestine in the anti-diabetic effects of bariatric surgery. Surg Obes Relat Dis 2007;3:109–115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jackness C, Karmally W, Febres G, et al. Very low-calorie diet mimics the early beneficial effect of Roux-en-Y gastric bypass on insulin sensitivity and β-cell function in type 2 diabetic patients. Diabetes 2013;62:3027–3032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sjöström L, Lindroos AK, Peltonen M, et al.; Swedish Obese Subjects Study Scientific Group . Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med 2004;351:2683–2693 [DOI] [PubMed] [Google Scholar]
- 41.Chikunguwo SM, Wolfe LG, Dodson P, et al. Analysis of factors associated with durable remission of diabetes after Roux-en-Y gastric bypass. Surg Obes Relat Dis 2010;6:254–259 [DOI] [PubMed] [Google Scholar]
- 42.DiGiorgi M, Rosen DJ, Choi JJ, et al. Re-emergence of diabetes after gastric bypass in patients with mid- to long-term follow-up. Surg Obes Relat Dis 2010;6:249–253 [DOI] [PubMed] [Google Scholar]
- 43.Arterburn DE, Bogart A, Sherwood NE, et al. A multisite study of long-term remission and relapse of type 2 diabetes mellitus following gastric bypass. Obes Surg 2013;23:93–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Blackstone R, Bunt JC, Cortés MC, Sugerman HJ. Type 2 diabetes after gastric bypass: remission in five models using HbA1c, fasting blood glucose, and medication status. Surg Obes Relat Dis 2012;8:548–555 [DOI] [PubMed] [Google Scholar]
- 45.Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008;359:1577–1589 [DOI] [PubMed] [Google Scholar]
- 46.Nathan DM, Cleary PA, Backlund JY, et al.; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group . Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 2005;353:2643–2653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Arterburn D. Bariatric surgery. BMJ 2008;337:a755. [DOI] [PubMed] [Google Scholar]
- 48.Arterburn D, Livingston EH, Schifftner T, Kahwati LC, Henderson WG, Maciejewski ML. Predictors of long-term mortality after bariatric surgery performed in Veterans Affairs medical centers. Arch Surg 2009;144:914–920 [DOI] [PubMed] [Google Scholar]
- 49.Arterburn D, Schauer DP, Wise RE, et al. Change in predicted 10-year cardiovascular risk following laparoscopic Roux-en-Y gastric bypass surgery. Obes Surg 2009;19:184–189 [DOI] [PubMed] [Google Scholar]
- 50.Arterburn DE, Westbrook EO, Bogart TA, Sepucha KR, Bock SN, Weppner WG. Randomized trial of a video-based patient decision aid for bariatric surgery. Obesity (Silver Spring) 2011;19:1669–1675 [DOI] [PubMed] [Google Scholar]
- 51.Maciejewski ML, Livingston EH, Smith VA, et al. Survival among high-risk patients after bariatric surgery. JAMA 2011;305:2419–2426 [DOI] [PubMed] [Google Scholar]
- 52.Maciejewski ML, Livingston EH, Kahwati LC, Henderson WG, Kavee AL, Arterburn DE. Discontinuation of diabetes and lipid-lowering medications after bariatric surgery at Veterans Affairs medical centers. Surg Obes Relat Dis 2010;6:601–607 [DOI] [PubMed] [Google Scholar]
- 53.Maciejewski ML, Smith VA, Livingston EH, et al. Health care utilization and expenditure changes associated with bariatric surgery. Med Care 2010;48:989–998 [DOI] [PubMed] [Google Scholar]
- 54.Buse JB, Caprio S, Cefalu WT, et al. How do we define cure of diabetes? Diabetes Care 2009;32:2133–2135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Writing Team for the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA 2003;290:2159–2167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Nathan DM. Diabetes: advances in diagnosis and treatment. JAMA 2015;314:1052–1062 [DOI] [PubMed] [Google Scholar]
- 57.The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 1993;329:977–986 [DOI] [PubMed] [Google Scholar]
- 58.Ohkubo Y, Kishikawa H, Araki E, et al. Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: a randomized prospective 6-year study. Diabetes Res Clin Pract 1995;28:103–117 [DOI] [PubMed] [Google Scholar]
- 59.Ismail-Beigi F, Craven T, Banerji MA, et al.; ACCORD trial group . Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet 2010;376:419–430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Patel A, MacMahon S, Chalmers J, et al.; ADVANCE Collaborative Group . Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008;358:2560–2572 [DOI] [PubMed] [Google Scholar]
- 61.Nathan DM, Bayless M, Cleary P, et al.; DCCT/EDIC Research Group . Diabetes control and complications trial/epidemiology of diabetes interventions and complications study at 30 years: advances and contributions. Diabetes 2013;62:3976–3986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive therapy. The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. N Engl J Med 2000;342:381–389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hayward RA, Reaven PD, Wiitala WL, et al.; VADT Investigators . Follow-up of glycemic control and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2015;372:2197–2206 [DOI] [PubMed] [Google Scholar]
- 64.Action to Control Cardiovascular Risk in Diabetes Study Group, Gerstein HC, Miller ME, Byington RP, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008;358:2545–2559 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ali MK, Bullard KM, Saaddine JB, Cowie CC, Imperatore G, Gregg EW. Achievement of goals in U.S. diabetes care, 1999–2010. N Engl J Med 2013;368:1613–1624 [DOI] [PubMed] [Google Scholar]
- 66.Gregg EW, Li Y, Wang J, et al. Changes in diabetes-related complications in the United States, 1990-2010. N Engl J Med 2014;370:1514–1523 [DOI] [PubMed] [Google Scholar]
- 67.Sjöström L, Peltonen M, Jacobson P, et al. Bariatric surgery and long-term cardiovascular events. JAMA 2012;307:56–65 [DOI] [PubMed] [Google Scholar]
- 68.Navarro-Díaz M, Serra A, Romero R, et al. Effect of drastic weight loss after bariatric surgery on renal parameters in extremely obese patients: long-term follow-up. J Am Soc Nephrol 2006;17(Suppl. 3):S213–S217 [DOI] [PubMed] [Google Scholar]
- 69.Amor A, Jiménez A, Moizé V, et al. Weight loss independently predicts urinary albumin excretion normalization in morbidly obese type 2 diabetic patients undergoing bariatric surgery. Surg Endosc 2013;27:2046–2051 [DOI] [PubMed] [Google Scholar]
- 70.Fenske WK, Dubb S, Bueter M, et al. Effect of bariatric surgery-induced weight loss on renal and systemic inflammation and blood pressure: a 12-month prospective study. Surg Obes Relat Dis 2013;9:559–568 [DOI] [PubMed] [Google Scholar]
- 71.Lammert A, Hasenberg T, Kräupner C, Schnülle P, Hammes HP. Improved arteriole-to-venule ratio of retinal vessels resulting from bariatric surgery. Obesity (Silver Spring) 2012;20:2262–2267 [DOI] [PubMed] [Google Scholar]
- 72.Navaneethan SD, Kelly KR, Sabbagh F, Schauer PR, Kirwan JP, Kashyap SR. Urinary albumin excretion, HMW adiponectin, and insulin sensitivity in type 2 diabetic patients undergoing bariatric surgery. Obes Surg 2010;20:308–315 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chen Y, Corsino L, Shantavasinkul PC, et al. Gastric bypass surgery leads to long-term remission or improvement of type 2 diabetes and significant decrease of microvascular and macrovascular complications. Ann Surg. 13 Oct 2015. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 74.Scott JD, Johnson BL, Blackhurst DW, Bour ES. Does bariatric surgery reduce the risk of major cardiovascular events? A retrospective cohort study of morbidly obese surgical patients. Surg Obes Relat Dis 2013;9:32–39 [DOI] [PubMed] [Google Scholar]
- 75.Brethauer SA, Aminian A, Romero-Talamas H, et al. Can diabetes be surgically cured? Long-term metabolic effects of bariatric surgery in obese patients with type 2 diabetes mellitus. Ann Surg 2013;258:628–636; discussion 636–637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Stephenson DT, Jandeleit-Dahm K, Balkau B, Cohen N. Improvement in albuminuria in patients with type 2 diabetes after laparoscopic adjustable gastric banding. Diab Vasc Dis Res 2013;10:514–519 [DOI] [PubMed] [Google Scholar]
- 77.Jose B, Ford S, Super P, Thomas GN, Dasgupta I, Taheri S. The effect of biliopancreatic diversion surgery on renal function--a retrospective study. Obes Surg 2013;23:634–637 [DOI] [PubMed] [Google Scholar]
- 78.Hou CC, Shyu RS, Lee WJ, Ser KH, Lee YC, Chen SC. Improved renal function 12 months after bariatric surgery. Surg Obes Relat Dis 2013;9:202–206 [DOI] [PubMed] [Google Scholar]
- 79.Heneghan HM, Cetin D, Navaneethan SD, Orzech N, Brethauer SA, Schauer PR. Effects of bariatric surgery on diabetic nephropathy after 5 years of follow-up. Surg Obes Relat Dis 2013;9:7–14 [DOI] [PubMed] [Google Scholar]
- 80.MacLaughlin HL, Hall WL, Patel AG, Macdougall IC. Laparoscopic sleeve gastrectomy is a novel and effective treatment for obesity in patients with chronic kidney disease. Obes Surg 2012;22:119–123 [DOI] [PubMed] [Google Scholar]
- 81.Varadhan L, Humphreys T, Walker AB, Cheruvu CV, Varughese GI. Bariatric surgery and diabetic retinopathy: a pilot analysis. Obes Surg 2012;22:515–516 [DOI] [PubMed] [Google Scholar]
- 82.Miras AD, Chuah LL, Lascaratos G, et al. Bariatric surgery does not exacerbate and may be beneficial for the microvascular complications of type 2 diabetes. Diabetes Care 2012;35:e81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Schuster DP, Teodorescu M, Mikami D, Foreman K, Rogers P, Needleman BJ. Effect of bariatric surgery on normal and abnormal renal function. Surg Obes Relat Dis 2011;7:459–464 [DOI] [PubMed] [Google Scholar]
- 84.Agrawal V, Krause KR, Chengelis DL, Zalesin KC, Rocher LL, McCullough PA. Relation between degree of weight loss after bariatric surgery and reduction in albuminuria and C-reactive protein. Surg Obes Relat Dis 2009;5:20–26 [DOI] [PubMed] [Google Scholar]
- 85.Alexander JW, Goodman HR, Hawver LR, Cardi MA. Improvement and stabilization of chronic kidney disease after gastric bypass. Surg Obes Relat Dis 2009;5:237–241 [DOI] [PubMed] [Google Scholar]
- 86.Navaneethan SD, Yehnert H. Bariatric surgery and progression of chronic kidney disease. Surg Obes Relat Dis 2009;5:662–665 [DOI] [PubMed] [Google Scholar]
- 87.Takata MC, Campos GM, Ciovica R, et al. Laparoscopic bariatric surgery improves candidacy in morbidly obese patients awaiting transplantation. Surg Obes Relat Dis 2008;4:159–164; discussion 164–165 [DOI] [PubMed] [Google Scholar]
- 88.Schauer PR, Burguera B, Ikramuddin S, et al. Effect of laparoscopic Roux-en Y gastric bypass on type 2 diabetes mellitus. Ann Surg 2003;238:467–484; discussion 484–485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Iaconelli A, Panunzi S, De Gaetano A, et al. Effects of bilio-pancreatic diversion on diabetic complications: a 10-year follow-up. Diabetes Care 2011;34:561–567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Szomstein S, Rojas R, Rosenthal RJ. Outcomes of laparoscopic bariatric surgery after renal transplant. Obes Surg 2010;20:383–385 [DOI] [PubMed] [Google Scholar]
- 91.Koshy AN, Coombes JS, Wilkinson S, Fassett RG. Laparoscopic gastric banding surgery performed in obese dialysis patients prior to kidney transplantation. Am J Kidney Dis 2008;52:e15–e17 [DOI] [PubMed] [Google Scholar]
- 92.Newcombe V, Blanch A, Slater GH, Szold A, Fielding GA. Laparoscopic adjustable gastric banding prior to renal transplantation. Obes Surg 2005;15:567–570 [DOI] [PubMed] [Google Scholar]
- 93.Thomas RL, Prior SL, Barry JD, et al. Does bariatric surgery adversely impact on diabetic retinopathy in persons with morbid obesity and type 2 diabetes? A pilot study. J Diabetes Complications 2014;28:191–195 [DOI] [PubMed] [Google Scholar]
- 94.Adams TD, Mehta TS, Davidson LE, Hunt SC. All-Cause and cause-specific mortality associated with bariatric surgery: a review. Curr Atheroscler Rep 2015;17:74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.MacDonald KG Jr, Long SD, Swanson MS, et al. The gastric bypass operation reduces the progression and mortality of non-insulin-dependent diabetes mellitus. J Gastrointest Surg 1997;1:213–220; discussion 220 [DOI] [PubMed] [Google Scholar]
- 96.Eliasson B, Liakopoulos V, Franzén S, et al. Cardiovascular disease and mortality in patients with type 2 diabetes after bariatric surgery in Sweden: a nationwide, matched, observational cohort study. Lancet Diabetes Endocrinol 2015;3:847–854 [DOI] [PubMed] [Google Scholar]
- 97.Johnson RJ, Johnson BL, Blackhurst DW, et al. Bariatric surgery is associated with a reduced risk of mortality in morbidly obese patients with a history of major cardiovascular events. Am Surg 2012;78:685–692 [PubMed] [Google Scholar]
- 98.Sjöström L, Narbro K, Sjöström CD, et al.; Swedish Obese Subjects Study . Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med 2007;357:741–752 [DOI] [PubMed] [Google Scholar]