Abstract
Background
Up to now, numerous case-control studies have reported the associations between fat mass and obesity associated (FTO) gene rs9939609 A/T polymorphism and polycystic ovary syndrome (PCOS), however, without a consistent result. Hence we performed current systematic review and meta-analysis to clarify the controversial results.
Methods
Case-control studies reporting the relationship of rs9939609 A/T polymorphism and PCOS published before April 2015 were searched in Pubmed database without language restriction. Data was analyzed by Review Manager 5.2.
Results
A total of five studies involving 5010 PCOS patients and 5300 controls were included for further meta-analysis. The results of meta-analysis showed that the FTO gene rs9939609 A/T polymorphism was significantly different between PCOS group and control group in different gene models (For AA + AT vs. TT: OR = 1.41, 95% CI = 1.28–1.55, P < 0.00001. For AA vs. AT + TT: OR = 1.54, 95% CI = 1.25–1.89, P < 0.0001. For AA vs. TT: OR = 1.74, 95% CI = 1.38–2.18, P < 0.00001. For A vs. T: OR = 1.36, 95% CI = 1.25–1.47, P < 0.00001, respectively) suggesting that A allele was a risk factor for PCOS susceptibility. Furthermore, subgroup analysis in Asian and Caucasian ethnicities also found significant association between rs9939609 A/T polymorphism and PCOS (In Asian subgroup: OR = 1.43, 95% CI = 1.29–1.59, P < 0.0001. In Caucasian subgroup: OR = 1.33, 95% CI = 1.08–1.64, P = 0.008)
Conclusion
This meta-analysis suggests that rs9939609 A/T polymorphism of FTO gene is associated with PCOS risk, and that A allele is a risk factor for PCOS susceptibility simultaneously.
Keywords: Polycystic ovary syndrome, FTO, Meta-analysis, Polymorphism
Background
Polycystic ovary syndrome (PCOS), a common endocrine metabolic disorder in women of childbearing age, is characterized by polycystic ovary (PCO), oligomenorrhea or amenorrhea, clinical or biochemical hyperandrogenism, and insulin resistance. In addition to leading to infertility, PCOS is closely associated with diabetes mellitus type 2, cardiovascular diseases and endometrial carcinoma [1–3]. It was firstly reported in 1935 by Stein et al. [4], but its etiology still remains ambiguous. Existing data suggests that genetics is an important factor in the occurrence and development of PCOS [5, 6]. In recent years, a total of more than 100 candidate genes have been set out to explore the associations between single nucleotide polymorphism (SNP) and PCOS [7], such as fat mass and obesity associated (FTO) gene.
The human FTO gene is located on the chromosome 16q12.2 with a wide expression range of almost all tissues [8, 9]. The protein coded by FTO gene belongs to 2-oxoglutarate-dependent nucleic acid demethylase, involving energy metabolism [10]. In 2007, a genome-wide association study reported that FTO is associated with body mass index (BMI) and obesity [7]. Obesity is a common characteristic in PCOS patients. It is reported that more than 50% PCOS cases are overweight/obese [11]. So it is reasonable to assume that FTO gene might play a role in the pathogenesis of PCOS via BMI and/or obesity. Therefore, based on this assumption, many studies have been conducted to research the associations between FTO gene and the risk of PCOS.
Recently, a common single nucleotide polymorphism (SNP) (rs9939609) in the first intron of FTO gene with T to A change is widely investigated in PCOS. However, the results from different studies are controversial. Some studies observed the positive association of FTO with PCOS [12–15], while others studies found the opposite relationship [16–18]. A recent meta-analysis by Cai et al. showed that FTO rs9939609 polymorphism was associated with PCOS risk in East Asians but not in overall population [19]. Imperfectly, this meta-analysis considered only one genetic model and not all included studies were involved in FTO rs9939609 polymorphism. Therefore, we carried out current systemic review and meta-analysis to clarify the associations between the SNP rs9939609 and susceptibility to PCOS.
Methods
Search strategy
A comprehensive literature research was conducted in Pubmed to identify the studies aimed at exploring the association between FTO rs9939609 polymorphism and PCOS risk. The following keywords were used: “fat mass and obesity associated gene or FTO”, “rs9939609”, “polycystic ovary syndrome or PCOS”. All researched articles were updated on 30 March 2015 without any language limitation. When one eligible article was screened, we would check the references list of this paper manually to identify additional relevant publications.
Selection of studies
Any study included in current meta-analysis must satisfy the following criteria: (a) Case-control studies aimed at investigating the association between FTO rs9939609 polymorphism and PCOS risk. (b) The number of genotypes and allele frequency in cases and controls were respectively available to calculate odds ratio (OR) with 95% confidence interval (CI). (c) Genotypes in control group meet the Hardy − Weinberg equilibrium (HWE) (d) PCOS patients accorded with one diagnose criteria of the following three: 1990 NIH, 2003 Rotterdam, and 2006 AE-PCOS Society [20–22].
Data extraction and analysis
Two investigators (Liu and Xie) independently extracted data from the qualified articles. The following information were extracted: the name of first author, publication time, ethnicity, country, sample size, diagnose criteria in PCOS and controls, genotype distribution in both case and control groups, and P value of HWE in control group. If any disagreement was generated, another author would take part in to resolve it and make a final decision.
The pooled odd ratios (ORs) and 95% confidence intervals (CIs) were used to assess the strength of association between FTO rs9939609 polymorphism and PCOS. The associations were calculated under the following four genetic models: dominant model (AA + AT vs. TT), recessive model (AA vs. AT + TT), additive model (AA vs. TT) and allele model (A vs. T). Subgroup analysis classified by ethnicity was conducted to explore whether ethnicity has a special effect on this association.
Heterogeneity among studies was examined by Q-test and I2 statistic. When P < 0.05 for Q statistic or I2 > 50%, random effects model was used, otherwise fixed model was applied. To assess the stability of single study in overall effect, sensitivity analysis was performed by excluding each study once a time. Publication bias was estimated via funnel plot. All data analyses were performed by Review Manager 5.2. P < 0.05 was considered statistically significant.
Results
Characteristics of included studies
The flow diagram of study selection was presented in Fig. 1. Based on the inclusion criteria, a total of five studies exploring the association between FTO rs9939609 and PCOS were eligible for current meta-analysis, involving 5010 PCOS patients and 5300 controls [12–16]. All of the eligible studies provided the genotypes or allele information. One study was excluded because whether the genotypes distribution in PCOS was consistent with HWE was not mentioned, leading to the number of genotypes in PCOS group uncountable. The study by Li et al. [14] was a two-stage design (a GWAS and a replication study) with two populations. Because the two populations was the same ethnicity background (Han of Chinese), we considered them as one study. Among the five studies, two studies were conducted in Caucasian population and three studies were in Asian population. The diagnosis of PCOS in all studies was based on Rotterdam criteria. All the selected studies were consistent with HWE in controls. The main characteristics of the included studies are presented in Table 1.
Fig. 1.
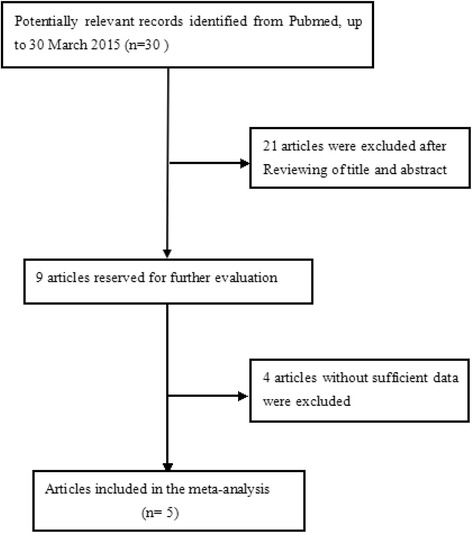
The flow chart of study selection
Table 1.
Characteristics of included studies in this meta-analysis
| Authors | Year | Country/Ethincity | Definition | Sample size | PCOS | Control | HWE | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PCOS | Controls | PCOS | Control | AA | AT | TT | AA | AT | TT | ||||
| Barber et al. | 2008 | UK/Caucasian | Rotterdam criteria | Unknown | 463 | 1336 | 99 | 231 | 133 | 212 | 644 | 480 | 0.870 |
| Yan et al. | 2009 | China/Asian | Rotterdam criteria | Females proven fertile with normal menstrual cycle and ovarian morphology, and no family history of abnormal menses or hirsutism | 215 | 227 | 5 | 55 | 155 | 1 | 43 | 183 | 0.361 |
| Li et al. | 2013 | China/Asian | Rotterdam criteria | Regular menstrual cycle (26–35) and normal ovarian morphology | 3599 | 3082 | 67 | 867 | 2665 | 29 | 563 | 2490 | 0.650 |
| Kim et al. | 2014 | Korea/Asian | Rotterdam criteria | Women without special health problem | 552 | 559 | 7 | 118 | 427 | 8 | 106 | 445 | 0.559 |
| Ramos et al. | 2015 | Brazil/Caucasian | Rotterdam criteria | Non-hirsute,regular ovulatory cycles, no hormone drug taking at least 3 months before the study | 181 | 96 | 39 | 83 | 59 | 17 | 47 | 32 | 0.971 |
HWE Hardy-Weinberg equilibrium
Meta-analysis results
The results of association between FTO polymorphism and PCOS risk were presented in Fig. 2. We found that the FTO rs9939609 A/T polymorphism was associated with PCOS risk under four genetic models (For AA + AT vs. TT: OR = 1.41, 95%CI = 1.28–1.55, P < 0.00001. For AA vs. AT + TT: OR = 1.54, 95% CI = 1.25–1.89, P < 0.0001. For AA vs. TT: OR = 1.74, 95% CI = 1.38–2.18, P < 0.00001. For A vs. T: OR = 1.36, 95% CI = 1.25–1.47, P < 0.00001, respectively), suggesting A allele was a risk factor for PCOS risk.
Fig. 2.
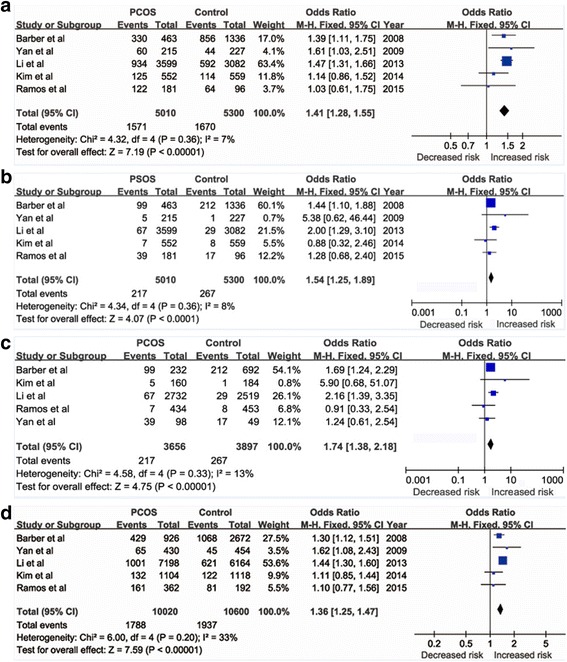
The association between FTO rs9939609 A/T polymorphism and risk of PCOS. (a) AA + AT vs. TT in overall analysis, (b) AA vs. AT + TT in overall analysis, (c) AA vs. TT in overall analysis, (d) A vs. T in overall analysis. For each study, the solid squares represent the ORs from the individual studies, horizontal lines represent corresponding 95% CIs. The pooled ORs and 95% CIs are shown by the shaded diamonds
Subgroup analysis
In subgroup analysis stratified by ethnicity, significant association between FTO 9939609 A/T and PCOS risk was observed in Caucasian and Asian populations under dominant model (OR = 1.33, 95% CI = 1.08–1.64, P = 0.008; OR = 1.43, 95%CI = 1.29–1.59, P < 0.00001, respectively). The relative results were displayed in Fig. 3.
Fig. 3.
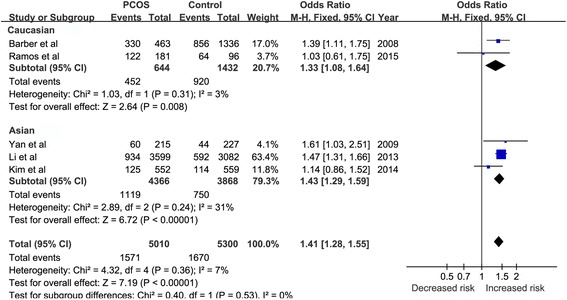
The association between FTO rs9939609 A/T polymorphism and risk of PCOS in subgroup model. The analysis was stratified by race. AA + AT vs. TT in subgroup (Caucasian and Asian) analysis of HWE > 0.05. The solid squares represent the ORs from the individual studies, horizontal lines represent corresponding 95% CIs. The pooled ORs and 95% CIs are presented as diamonds
Sensitivity analysis and publication bias
To confirm the stability of above results, a sensitivity analysis was performed by removing studies sequentially. The results showed that the corresponding pooled ORs and P value under different genetic models were not changed significantly. Publication bias of the recruited studies was evaluated using funnel plot, the shapes of the funnel plots did not show significant asymmetry (Fig. 4).
Fig. 4.
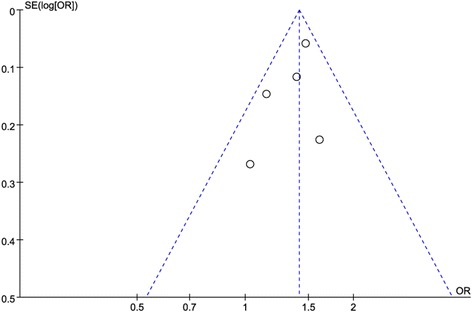
Funnel plot of publication bias in selection of studies on the FTO rs9939609 A/T polymorphism. Publication bias was assessed under dominant model (AA + AT vs. TT). SE represents standard error; OR represents odds ratio
Discussion
As one of the most common endocrine disorder, the etiology of PCOS remains incompletely unknown. However, family studies have showed that genetic basis is responsible for PCOS and that no single gene can explain this syndrome entirely [23–25]. It is reported that 40–80% of PCOS women are overweight or obese [26–29]. Therefore, based on the high prevalence of obesity in PCOS and the heritable of obesity [30], genes associated with obesity might play a role in pathogenesis of PCOS such as FTO gene. Recent years, many studies have been conducted to identify the relationships between FTO rs9939609 and PCOS susceptibility. However, the results were inconsistent, making these associations more indistinct. The current meta-analysis was aimed at clarifying the association of FTO rs9939609 and PCOS.
In our meta-analysis, we found that FTO rs9939609 polymorphism was associated with PCOS under different genetic models. This finding is consistent with the study by Li et al. They performed a two stage study involving 3599 PCOS and 3082 control subjects in Chinese population [14]. They found that rs9939609 polymorphism of FTO is associated with PCOS in Chinese women, no matter the PCOS women are obese or non-obese. This indicated that FTO might interact with PCOS directly. However, because of insufficient data, this potential interaction way can not be re-analyzed in our meta-analysis. Contrary to our result, a case-control study by Kim et al. observed that FTO polymorphisms of rs9939609 are not major determinants of PCOS. But for women with PCOS, the variant allele of FTO rs9939609 was associated with increased BMI, suggesting FTO polymorphism contributes to PCOS risk through affecting BMI indirectly [16]. Therefore, the associations among PCOS, BMI/obese and FTO polymorphism still remain indistinct. FTO might indirectly play a role in PCOS risk via BMI/obese. Also, FTO might contribute to PCOS by directly interacting or via the combined direct and indirect ways. More studies are needed to demonstrate it.
In subgroup analysis, the significant association between FTO polymorphism and PCOS was found not only in Caucasian but also in Asian, suggesting ethnicity was not the potential source of heterogeneity. Partly in line with our result, a case-control study by Cai et al. observed that FTO rs9939609 polymorphism (or its proxy) was associated with PCOS only in East Asians, but not in Caucasians [19]. In Caucasians subgroup of study by Cai et al., one of the two included studies was about FTO rs9939609 and another was about FTO rs8050136. So, we inferred this point might the source of different conclusion compared with our subgroup result.
Several limitations of this meta-analysis should be acknowledged. First, the number of studies included in our meta-analysis was relatively small, thus leading to smaller studies in subgroup analysis and weaken statistical power. Second, potential sources of heterogeneity in this meta-analysis should include other factors such as BMI, waist-to-hip ratio (WHR), obese. However, because of limited data, we could not explore these factors in current meta-analysis. Finally, it is well known that the etiology of PCOS involving in gene-gene, and gene-environment interactions. However these interactions could not be investigated in our meta-analysis due to insufficient information.
Conclusions
In conclusion, this meta-analysis provided evidence that the FTO rs9939609 polymorphism was significantly associated with risk of PCOS risk not only in Asians but also in Caucasians. Further large studies are needed to clarify the associations among the FTO rs9939609 polymorphism, BMI/obese and PCOS risk in the future.
This meta-analysis indicates that rs9939609 A/T polymorphism of FTO gene is associated with PCOS susceptibility, and that A allele is a risk factor for PCOS simultaneously.
Acknowledgments
Dr. Kai Li provided greatly technical help and writing assistance.
Funding
This research was supported by National Natural Science Foundation of China (NSFC, No. 31371277) and the Natural Science Foundation of Hunan Province (2016JJ6126).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- BMI
Body mass index
- CI
Confidence interval
- FTO
Fat mass and obesity associated
- HWE
Hardy − Weinberg equilibrium
- OR
Odds ratio
- PCO
Polycystic ovary
- PCOS
Polycystic ovary syndrome
- SNP
Single nucleotide polymorphism
- WHR
Waist-to-hip ratio
Authors’ contributions
ALL carried out writing paper and collecting data, HJX and HYX participated in paper design and data analysis. JL, JY and JSH performed extracting and checking the data, reviewing the manuscript and exerting additional genetic testing help. CYP contributed to checking and designing paper, and acquisition of funding. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The manuscript has been read and approved by all the authors. The research adhered to the tenets of The Declaration of Helsinki. The Ethics Committee of Nanhua-Xinghui Hospital approved the protocol. Written informed consent was obtained from all participants among these cited references (12–16).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Ai Ling Liu, Email: LiuAiLing917@outlook.com.
Hui Jun Xie, Email: 65465126@qq.com.
Hong Yan Xie, Email: 19785642678@qq.com.
Jun Liu, Email: nhuliujun@qq.com.
Jie Yin, Email: 13309624@qq.com.
Jin Song Hu, Email: 522371193@qq.com.
Cui Ying Peng, Phone: +86-0734 8281224, Email: pengcuiying2004@126.com.
References
- 1.Hardiman P, Pillay OC, Atiomo W. Polycystic ovary syndrome and endometrial carcinoma. Lancet. 2003;361(9371):1810–1812. doi: 10.1016/S0140-6736(03)13409-5. [DOI] [PubMed] [Google Scholar]
- 2.Homburg R. The management of infertility associated with polycystic ovary syndrome. Reprod Biol Endocrinol. 2003;1:109. doi: 10.1186/1477-7827-1-109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Diamanti-Kandarakis E, Piperi C, Spina J, Argyrakopoulou G, Papanastasiou L, Bergiele A, et al. Polycystic ovary syndrome: the influence of environmental and genetic factors. Hormones (Athens) 2006;5(1):17–34. doi: 10.14310/horm.2002.11165. [DOI] [PubMed] [Google Scholar]
- 4.Stein IF, Leventhal ML. Amenorrhea associated with bilateral poly-cystic ovaries. Am J Obstet Gynecol. 1935;29:181–191. doi: 10.1016/S0002-9378(15)30642-6. [DOI] [Google Scholar]
- 5.Segars JH, DeCherney AH. Is there a genetic basis for polycystic ovary syndrome? J Clin Endocrinol Metab. 2010;95(5):2058–2060. doi: 10.1210/jc.2010-0518. [DOI] [PubMed] [Google Scholar]
- 6.Vink JM, Sadrzadeh S, Lambalk CB, Boomsma DI. Heritability of polycystic ovary syndrome in a Dutch twin-family study. J Clin Endocrinol Metab. 2006;91(6):2100–2104. doi: 10.1210/jc.2005-1494. [DOI] [PubMed] [Google Scholar]
- 7.Demirci H, Yilmaz M, Ergun MA, Yurtcu E, Bukan N, Ayvaz G. Frequency of adiponectin gene polymorphisms in polycystic ovary syndrome and the association with serum adiponectin, androgen levels, insulin resistance and clinical parameters. Gynecol Endocrinol. 2010;26(5):348–355. doi: 10.3109/09513590903367051. [DOI] [PubMed] [Google Scholar]
- 8.Hubacek JA, Stanek V, Gebauerová M, Pilipcincová A, Dlouhá D, Poledne R, et al. A FTO variant and risk of acute coronary syndrome. Clin Chim Acta. 2010;411(15–16):1069–1072. doi: 10.1016/j.cca.2010.03.037. [DOI] [PubMed] [Google Scholar]
- 9.Frayling TM, Timpson NJ, Weedon MN, Zeggini E, Freathy RM, Lindgren CM, et al. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science. 2007;316(5826):889–894. doi: 10.1126/science.1141634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Willer CJ, Speliotes EK, Loos RJF, Li S, Lindgren CM, Heid IM, et al. Six new loci associated with body mass index highlight a neuronal influence on body weight regulation. Nat Genet. 2009;41(1):25–34. doi: 10.1038/ng.287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shi Y, Guo M, Yan J, Sun W, Zhang X, Geng L, et al. Analysis of clinical characteristics in large-scale Chinese women with polycystic ovary syndrome. Neuro Endocrinol Lett. 2007;28(6):807–810. [PubMed] [Google Scholar]
- 12.Yan Q, Hong J, Gu W, Zhang Y, Liu Q, Su Y, et al. Association of the common rs9939609 variant of FTO gene with polycystic ovary syndrome in Chinese women. Endocrine. 2009;36(3):377–382. doi: 10.1007/s12020-009-9257-0. [DOI] [PubMed] [Google Scholar]
- 13.Barber TM, Bennett AJ, Groves CJ, Sovio U, Ruokonen A, Martikainen H, et al. Association of variants in the fat mass and obesity associated (FTO) gene with polycystic ovary syndrome. Diabetologia. 2008;51(7):1153–1158. doi: 10.1007/s00125-008-1028-6. [DOI] [PubMed] [Google Scholar]
- 14.Li T, Wu K, You L, Xing X, Wang P, Cui L, et al. Common variant rs9939609 in gene FTO confers risk to polycystic ovary syndrome. PLoS One. 2013;8(7):e66250. doi: 10.1371/journal.pone.0066250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ramos RB, Spritzer PM. FTO gene variants are not associated with polycystic ovary syndrome in women from southern Brazil. Gene. 2015;560(1):25–29. doi: 10.1016/j.gene.2015.01.012. [DOI] [PubMed] [Google Scholar]
- 16.Kim JJ, Choi YM, Hong MA, Kim JM, Hwang SS, Lee GH, et al. Gene dose effect between a fat mass and obesity-associated polymorphism and body mass index was observed in Korean women with polycystic ovary syndrome but not in control women. Fertil Steril. 2014;102(4):1143–1148. doi: 10.1016/j.fertnstert.2014.07.004. [DOI] [PubMed] [Google Scholar]
- 17.Saxena R, Welt CK. Polycystic ovary syndrome is not associated with genetic variants that mark risk for type 2 diabetes. Acta Diabetol. 2013;50(3):451–457. doi: 10.1007/s00592-012-0383-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ewens KG, Jones MR, Ankener W, Stewart DR, Urbanek M, Dunaif A, et al. FTO and MC4R gene variants are associated with obesity in polycystic ovary syndrome. PLoS One. 2011;6(1):e16390. doi: 10.1371/journal.pone.0016390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cai X, Liu C, Mou S. Association between fat mass- and obesity- associated (FTO) gene polymorphism and polycystic ovary syndrome: a meta-analysis. PLoS One. 2014;9(1) doi: 10.1371/journal.pone.0086972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zawadzki JK, Dunaif A. Diagnostic criteria for polycystic ovary syndrome: towards a rational approach. In: Dunaif A, Givens JR, Haseltine, Merriam GR, editors. Polycystic Ovary Syndrome. Boston: Blackwell Scientific Publications; 1992. pp. 377–384. [Google Scholar]
- 21.Rotterdam. ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril. 2004;81(1):19–25. doi: 10.1016/j.fertnstert.2003.10.004. [DOI] [PubMed] [Google Scholar]
- 22.Azziz R, Carmina E, Dewailly D, Diamanti-Kandarakis E, Escobar-Morreale HF, Futterweit W, et al. Positions statement: criteria for defining polycystic ovary syndrome as a predominantly hyperandrogenic syndrome: an androgen excess society guideline. J Clin Endocrinol Metab. 2006;91(11):4237–4245. doi: 10.1210/jc.2006-0178. [DOI] [PubMed] [Google Scholar]
- 23.Franks S, Gharani N, McCarthy M. Candidate genes in polycystic ovary syndrome. Hum Reprod Update. 2001;7(4):405–410. doi: 10.1093/humupd/7.4.405. [DOI] [PubMed] [Google Scholar]
- 24.Escobar-Morreale HF, Luque-Ramirez M, San Millán JL. The molecular-genetic basis of functional hyperandrogenism and the polycystic ovary syndrome. Endocr Rev. 2005;26(2):251–282. doi: 10.1210/er.2004-0004. [DOI] [PubMed] [Google Scholar]
- 25.Urbanek M, Woodroffe A, Ewens KG, Diamanti-Kandarakis E, Legro RS, Strauss JF, et al. Candidate gene region for polycystic ovary syndrome on chromosome 19p13.2. J Clin Endocrinol Metab. 2005;90(12):6623–6629. doi: 10.1210/jc.2005-0622. [DOI] [PubMed] [Google Scholar]
- 26.Legro RS, Kunselman AR, Dodson WC, Dunaif A. Prevalence and predictors of risk for type 2 diabetes mellitus and impaired glucose tolerance in polycystic ovary syndrome: a prospective, controlled study in 254 affected women. J Clin Endocrinol Metab. 1999;84(1):165–169. doi: 10.1210/jcem.84.1.5393. [DOI] [PubMed] [Google Scholar]
- 27.Barber TM, McCarthy MI, Wass JA, Franks S. Obesity and polycystic ovary syndrome. Clin Endocrinol. 2006;65(2):137–145. doi: 10.1111/j.1365-2265.2006.02587.x. [DOI] [PubMed] [Google Scholar]
- 28.Wehr E, Schweighofer N, Möller R, Giuliani A, Pieber TR, Obermayer-Pietsch B, et al. Association of FTO gene with hyperandrogenemia and metabolic parameters in women with polycystic ovary syndrome. Metabolism. 2010;59(4):575–580. doi: 10.1016/j.metabol.2009.08.023. [DOI] [PubMed] [Google Scholar]
- 29.Barber TM, Golding SJ, Alvey C, Wass JA, Karpe F, Franks S, et al. Global adiposity rather than abnormal regional fat distribution characterizes women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93(3):999–1004. doi: 10.1210/jc.2007-2117. [DOI] [PubMed] [Google Scholar]
- 30.Walley AJ, Blakemore AI, Froguel P. Genetics of obesity and the prediction of risk for health. Hum Mol Genet. 2006;15:R124–R130. doi: 10.1093/hmg/ddl215. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.


