Abstract
Patients with unknown time of stroke onset (UOS) represent around one-third of ischemic stroke patients. These are patients with wake-up stroke (WUS) or daytime-unwitnessed stroke (DUS), often presenting outside the time-window for reperfusion therapy. UOS patients presenting between 4.5 and 12 h after time of last-seen-well were included. Clinical and imaging characteristics were compared between WUS and DUS patients. Good functional outcome was defined as a modified Rankin scale of ≤2 at follow-up. Sixty-one UOS patients were included: 42 WUS and 19 DUS patients. Stroke severity at presentation was mild to moderate with a median National Institutes of Health Stroke Scale of 5 in WUS and 6 in DUS patients. Time between last-seen-well and presentation at the hospital was shorter in patients with DUS compared to WUS (506 vs 362 min, p < 0.01). CT imaging results were similar, with a median Alberta Stroke Program Early CT Score of 10 for both WUS and DUS patients. After correction for age and NIHSS at presentation, no difference in good functional outcome was found between WUS (52%) and DUS (22%). In patients with unknown onset ischemic strokes presenting between 4.5 and 12 h after time of last-seen-well, clinical and radiological features were in large part similar between WUS and DUS. The outcome in the overall cohort was rather poor despite a favorable neuroimaging profile at presentation. These findings underscore the need for clinical trials in patients in whom stroke onset time is unknown.
Keywords: Ischemic stroke, Unknown onset, Wake up, Daytime unwitnessed
Introduction
In up to 36% of cases of ischemic stroke, the exact time of onset is unknown (unknown onset stroke, UOS). About a quarter of stroke patients notice their stroke symptoms upon awakening (wake-up stroke, WUS). These patients have worse functional outcome than patients with known onset, probably due to a lack of acute treatment options [1–5].
Patients with daytime stroke onset can also present with UOS when they are unable to communicate the time of onset and it cannot be pinpointed by a witness (daytime-unwitnessed stroke, DUS) [1]. Current standard of care for acute ischemic stroke in most countries involves intravenous tissue plasminogen activator (IVT) within 4.5 h and/or intra-arterial thrombectomy (IAT) within 6 h after onset of symptoms [6, 7]. In patients with UOS, the time when they were last-seen-well (LSW) is used as a reference for time of onset of stroke, frequently exceeding the allowed time window for acute stroke treatment. As a result, a large proportion of patients with UOS are excluded from thrombolytic therapy, although treatment might be safe [8, 9].
A study comparing the characteristics between DUS and WUS patients concluded that DUS was more likely to receive acute reperfusion therapy [1]. However, DUS patients present at the emergency department (ED) earlier after LSW compared to WUS patients, often even within the 4.5-h time window for IVT. Therefore, these patients can be treated with IVT according to the guidelines.
The aim of this study was to specifically characterise patients with UOS, either DUS or WUS, presenting at the ED outside of the time window for thrombolytic therapy based on time of LSW. We compared patient demographics, clinical and neuroimaging characteristics and functional outcome between WUS and DUS. We hypothesised the frequency of early ischemic changes on neuroimaging to be more common in patients with WUS since the time between LSW and presentation might be longer compared to patients with DUS. Since all selected patients in this study presented outside the time window for IVT, we expected the functional outcome to be similar.
Patients and methods
Patients with ischemic stroke who were admitted between January 1st, 2014, and July 31st, 2015, were extracted from our prospective stroke registry at the Haaglanden Medical Center. Patients meeting the following criteria were included:
Clinical diagnosis of acute ischemic stroke with unknown time of onset of stroke.
Last-seen-well >4.5 h (or 6 h in case of IAT-candidates) and <12 h prior to presentation.
National Institutes of Health Stroke Scale (NIHSS) at presentation of ≥2 points.
General life expectancy >90 days (exceeding time of planned follow-up).
Clinical data collected included age, gender, pre- and post-stroke modified Rankin Scale (mRS), time of LSW, time of presentation at ED, NIHSS at presentation, anterior or posterior circulation ischemia and eligibility for IVT and IAT. Patients were categorised as either WUS or DUS. Functional outcome at the 90-day follow-up was assessed using the modified Rankin Scale (mRS) which was documented in our prospective stroke registry. Good functional outcome was defined as a mRS of 0–2 at 90 days after stroke. All patients received routine clinical care in accordance with current guidelines. Eligibility for reperfusion therapy was assessed according to guidelines and current time window for acute treatment (<4.5 h for IVT, <6 h for IAT) [7, 10].
Multimodal CT imaging, including non-contrast CT and CT angiography, was evaluated by a resident in neuroradiology, who was aware of the clinical stroke symptoms, but blinded for symptom duration. The Alberta Stroke Program Early CT Score (ASPECTS), used to assess early ischemic changes, was determined on baseline CT [11]. Furthermore, collateral supply was scored on CT angiography, either being good (collaterals filling >50% of the occluded arterial territory) or poor (<50%). Both of these imaging markers are associated with functional outcome in patients receiving reperfusion therapy and could, therefore, identify patients with UOS still eligible for therapy [12].
CT imaging protocol
Non-contrast CT was performed with contiguous 6-mm sequentially acquired axial slices. The CT angiography studies were performed on a 64 slice CT scanner (Brightspeed CT; General Electric Medical Systems, Little Chalfont, Buckinghamshire, United Kingdom) with the gantry angled to the orbitomeatal line, with 64 1-s rotations of 1.25-mm collimation and a table speed of 23 mm/s, 512 × 512 matrix, 16 cm field of view, 120 kV of variable tube current (mA) with a mean of 100 mA at the level of the circle of Willis. For CTA imaging, 50 cc of Visipaque iodine contrast material (320 mg iodine/ml) (General Electric healthcare, Little Chalfont, Buckinghamshire, United Kingdom) was injected intravenously at a rate of 6 cc/s using an automated power injector. Automated triggering of image acquisition was used at the time of contrast passage through the aortic arch, followed by a chaser bolus of saline. The CT angiographic source image data were post-processed creating coronal and axial 3-mm thick maximum intensity on a computer workstation (Advantage Workstation 4.4; Global Electronics Medical Systems).
Statistical analysis
Clinical and radiological variables between WUS and DUS groups were compared using Chi-square tests, independent t tests and logistic regression. A p value of ≤0.05 was considered statistically significant. All statistical analyses were conducted using SPSS for Windows, version 20.0 (SPSS INC., Chicago, IL).
Ethical considerations
The study protocol was approved by local ethical committee of the Haaglanden Medical Center and by the Medical Ethical Review Board of South-West Holland.
Results
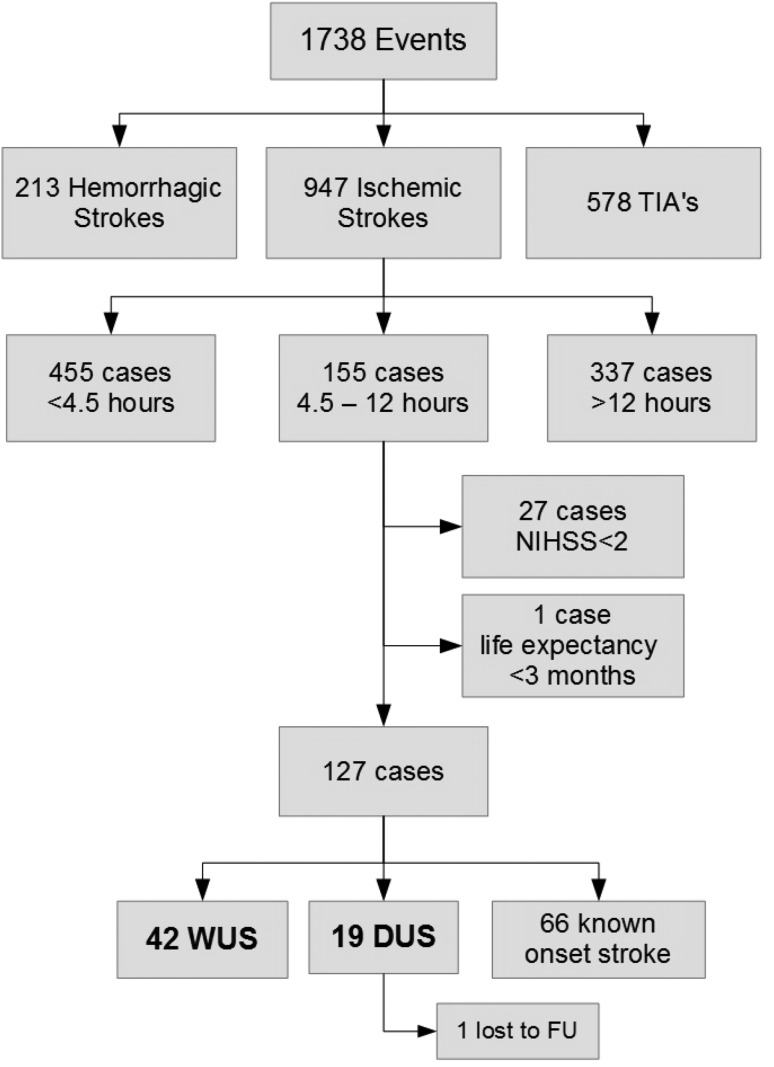
In 577 days, 1738 potential stroke events occurred: 213 (12.3%) hemorrhagic strokes, 578 (33.3%) TIAs and 947 (54.5%) ischemic strokes. One hundred fifty-five ischemic stroke patients (16.4%) presented at the ED between 4.5 h and 12 h after time of LSW. Twenty-seven patients were excluded because of NIHSS score <2 and one because of a life expectancy <3 months. Of the remaining 127 patients, 61 fulfilled the inclusion criteria of UOS. Of these, 19 were DUS and 42 WUS. One DUS patient was lost to follow-up due to emigration and excluded from further analysis (Fig. 1).
Fig. 1.
Flowchart of included patients
Comparison of patients with WUS vs DUS
No major differences were observed in baseline characteristics although patients with DUS tended to be older compared to the group with WUS (78 vs 70 years; p = 0.06). As expected, the time between LSW and presentation at ED was shorter in DUS (6.0 h) compared with WUS patients (8.5 h; p < 0.01). Although this could suggest that patients with WUS presented later after stroke onset, no difference in early ischemic changes based on ASPECTS was identified. Collateral status was good in the majority of patients and not different between patients with WUS and DUS. At 90 day follow-up, 52% of WUS and 22% of DUS patients had good functional outcome (p = 0.05 for difference), defined as mRS 0–2 (Table 1). In multivariate analysis, no difference in functional outcome was found for WUS vs. DUS (p = 0.21) after correction for age (p = 0.01) and NIHSS at presentation (p = 0.02).
Table 1.
Characteristics of WUS and DUS patients
| All cases (n = 61) | WUS (n = 42) | DUS (n = 19) | P value for differences DUS–WUS | |
|---|---|---|---|---|
| Age, median (SD) | 72 (15) | 70 (15) | 78 (14) | p = 0.06 (t) |
| Female sex, n (%) | 35 (57%) | 23 (55%) | 12 (63%) | p = 0.59 (Χ) |
| Prestroke mRS ≥3, n (%) | 4 (7%) | 3 (7%) | 1 (5%) | p = 1.0 (Χ) |
| Posterior circulation ischemia, n (%) | 8 (13%) | 5 (12%) | 3 (16%) | p = 0.70 (Χ) |
| Time between LSW and presentation at ED in minutes, mean (SD) | 461 (137) | 506 (135) | 362 (75) | p < 0.01 ( t ) |
| NIHSS at presentation, median (SD) | 5 (3.7) | 5 (3.9) | 6 (3.3) | p = 0.61 (t) |
| Imaging characteristics for anterior circulation ischemia (n = 53) | ||||
| ASPECTS, median (SD) | 10 (1.7) | 10 (1.4) | 10 (2.2) | p = 0.21 (t) |
| Collateral supply <50%, n (%) | 3 (7%) | 2 (7%) | 1 (8%) | p = 1.0 (Χ) |
| Therapy eligibility | ||||
| Eligible for IVTa, n (%) | 49 (80%) | 34 (81%) | 15 (79%) | |
| Eligible for IATa, n (%) | 9 (15%) | 7 (17%) | 2 (11%) | |
| IAT performed, n | 4 (44%) | 3 (43%) | 1 (50%) | |
| Outcome | ||||
| Good outcome (mRS ≤2) after 90 days, n (%) | 26 (43%) | 22 (52%) | 4 (22%) | p = 0.05 ( Χ ) |
Significant p values <0.05 are shown in bold
aEligibility is assessed using the currently accepted criteria excluding the criterion of time window <4.5 h (IVT) or <6 h (IAT)
(t) independent t test, (Χ) Chi-square test
Eligibility for reperfusion therapy
All patients were ineligible for IVT based on presentation at ED in the time window of 4.5 h after LSW. Other exclusion criteria for IVT were only present in 12 patients (20%), similar in patients with WUS vs DUS. Nine patients (15%) were diagnosed with a large vessel occlusion >6 h after LSW. In four of these (44%) IAT was performed nonetheless (Table 1).
Discussion
Our study shows that in patients with UOS presenting between 4.5 and 12 h after LSW, clinical and neuroimaging features were in large part similar between WUS and DUS. The rate of good functional outcome was 43% which is comparable to results in the placebo arms of IVT trials [13]. In our study, no patients were treated with IVT although other exclusion criteria were only present in 20% of patients. These findings underscore the need for assessment of reperfusion treatments for these patients in whom exact stroke onset time is unknown in randomised controlled trials, such as the DAWN, WAKE-UP and EXTEND-trials [14–16]. In addition, studies investigating expansion of time window for endovascular treatment are ongoing, perhaps providing opportunities for acute treatment in this group of patients as well [14, 17].
Imaging markers, such as small infarct core, core–penumbra mismatch, diffusion-weighted imaging (DWI)/fluid-attenuated inversion recovery (FLAIR) mismatch, perfusion/diffusion mismatch, CT perfusion–target mismatch and good collateral status, may identify patients most likely to benefit from reperfusion therapies. These selection criteria have been studied to potentially extend the time window for intravenous thrombolysis [9, 18–21] and endovascular stroke treatment [22–26]. Some of these parameters require magnetic resonance imaging (MRI) as imaging modality to identify patients who might potentially benefit from reperfusion: DWI/FLAIR mismatch and perfusion/DWI mismatch; although CT Perfusion can also be reliable on identifying patients with small core and salvageable tissue [26, 27]. In our center, CT imaging is mostly used for acute radiological assessment because this modality is more commonly used in clinical practice since it is fast, non-invasive, inexpensive, widely available and more practical than MR imaging [28]. CT Perfusion was not routinely used because until recently it was considered controversial in guiding selection of patients for stroke treatment because of equivocal results [29, 30].
Previous studies have suggested that in a large proportion of patients with WUS, stroke onset occurred only shortly before waking up, since imaging patterns were similar to known onset strokes presenting within a few hours after onset of symptoms [1, 4, 31–35]. It is, therefore, hypothesised that ischemia might be disrupting the sleeping brain, leading to awakening of the patient. These patients would probably present themselves at the ED quite shortly after onset of ischemia and thus within the treatment window of IVT. In our study, time between LSW and presentation at ED was significantly longer for WUS than for DUS patients. However, despite this 2.5-h time difference, the ASPECT score did not differ between these patients and did not reveal early ischemic changes in most patients. This suggests that onset of stroke in WUS patients might indeed be around the time of waking up potentially even caused the awakening itself. As hypothesised, no association between type of UOS (WUS vs DUS) and functional outcome was found after correction for age and NIHSS at presentation.
Our study has limitations. First, the sample size is modest with only 61 included patients. However, we had decided to only analyse patients presenting in the time window of 4.5–12 h after LSW. This enabled the specific evaluation of patients ineligible for IVT, since stroke onset time was unknown and time since LSW was more than 4.5 h, as well as the comparison of WUS vs DUS. This approach seemed appropriate to comment on findings in patients who are currently excluded from IVT. Second, some patients were treated with IAT. Although this percentage was rather low, the outcome of these four patients has potentially been modified by this intervention. Therefore, the outcome reported might not completely reflect the expected clinical course of patients with UOS ineligible for reperfusion therapy. Third, ASPECTS and collateral status were used to determine tissue status and potential benefit of reperfusion therapy. However, diffusion-weighted imaging/perfusion-weighted imaging might have been more appropriate to determine salvageable tissue in this study population.
Conclusion
In patients with unknown onset ischemic strokes presenting between 4.5 and 12 h after time of last-seen-well, clinical and radiological features were in large part similar between WUS and DUS. Considering the favourable neuroimaging profile at presentation, a reasonable percentage of these patients might benefit from IVT. This underscores the need to identify (neuroimaging) criteria to select patients who can still benefit and assess the effect of IVT in this subgroup of patients as is currently being studied in randomised controlled trials.
Acknowledgements
The authors wish to specifically thank our local stroke nurses for the extensive and accurate registration of all stroke data in the registry.
Abbreviations
- ASPECTS
Alberta Stroke Program Early CT Score
- DUS
Daytime-unwitnessed stroke
- DWI
Diffusion-weighted imaging
- ED
Emergency department
- IAT
Intra-arterial therapy
- IVT
Intravenous therapy
- LSW
Last-seen-well
- mRS
modified Rankin Scale
- NIHSS
National Institutes of Health Stroke Scale
- UOS
Unknown onset of stroke
- WUS
Wake-up stroke
Author contributions
IvdW, JB and RL conceptualised the study. LD collected cases, carried out the statistical analysis and drafted the manuscript. HH performed the imaging analysis. All authors helped in the revision of the manuscript and approved the final version.
Compliance with ethical standards
Conflict of interest
RL is a senior clinical investigator of FWO Flanders. None of the other authors had a conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
This is a non-invasive, retrospective data analysis study. The local ethical committee of the Haaglanden Medical Center as well as the Medical Ethical Review Board (METC) of South-West Holland approved this study.
Informed consent
Due to the nature of this study, no informed consent was needed for the conduction of this study.
Guarantor
LD.
References
- 1.Kim YJ, Kim BJ, Kwon SU, Kim JS, Kang DW. Unclear-onset stroke: daytime-unwitnessed stroke vs. wake-up stroke. Int J Stroke. 2016;11(2):212–220. doi: 10.1177/1747493015616513. [DOI] [PubMed] [Google Scholar]
- 2.Rimmele DL, Thomalla G. Wake-up stroke: clinical characteristics, imaging findings, and treatment option—an update. Front Neurol. 2014;5:35. doi: 10.3389/fneur.2014.00035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kang DW, Kwon JY, Kwon SU, Kim JS. Wake-up or unclear-onset strokes: are they waking up to the world of thrombolysis therapy? Int J Stroke. 2012;7(4):311–320. doi: 10.1111/j.1747-4949.2012.00779.x. [DOI] [PubMed] [Google Scholar]
- 4.Fink JN, Kumar S, Horkan C, et al. The stroke patient who woke up; clinical and radiological features, including diffusion and perfusion MRI. Stroke. 2002;33(4):988–993. doi: 10.1161/01.STR.0000014585.17714.67. [DOI] [PubMed] [Google Scholar]
- 5.Lago A, Geffner D, Tembl J, Landete L, Valero C, Baquero M. Circadian variation in acute ischemic stroke. A hospital-based study. Stroke. 1998;29(9):1873–1875. doi: 10.1161/01.STR.29.9.1873. [DOI] [PubMed] [Google Scholar]
- 6.Jauch EC, Saver JL, Adams HP, et al. Guidelines for the early management of patients with acute ischemic stroke. A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(3):870–947. doi: 10.1161/STR.0b013e318284056a. [DOI] [PubMed] [Google Scholar]
- 7.Fiehler J, Cognard C, Gallitelli M, et al. European recommendations on organisation of interventional care in acute stroke (EROICAS) Int J Stroke. 2016;11(6):701–716. doi: 10.1177/1747493016647735. [DOI] [PubMed] [Google Scholar]
- 8.Barreto AD, Fanale CV, Alexandrov AV, Wake-Up Stroke Investigators et al. Prospective, open-label safety study of intravenous recombinant tissue plasminogen activator in wake-up stroke. Ann Neurol. 2016;80(2):211–218. doi: 10.1002/ana.24700. [DOI] [PubMed] [Google Scholar]
- 9.Kang DW, Sohn SI, Hong KS, et al. Reperfusion therapy in unclear-onset stroke based on MRI evaluation (RESTORE): a prospective multicenter study. Stroke. 2012;43(12):3278–3283. doi: 10.1161/STROKEAHA.112.675926. [DOI] [PubMed] [Google Scholar]
- 10.European Stroke Organisation Guidelines for management of ischaemic stroke and transient ischaemic attack 2008. Cerebrovasc Dis. 2008;25(5):457–507. doi: 10.1159/000131083. [DOI] [PubMed] [Google Scholar]
- 11.Barber PA, Demchuk AM, Zhang J, Buchan AM, ASPECTS Study Group Validity and reliability of a quantitative computed tomography score in predicting outcome of hyperacute stroke before thrombolytic therapy. Lancet. 2000;355(9216):1670–1674. doi: 10.1016/S0140-6736(00)02237-6. [DOI] [PubMed] [Google Scholar]
- 12.Tan IY, Demchuk AM, Zhang L, et al. CT angiography clot burden score and collateral score: correlation with clinical and radiologic outcomes in acute middle cerebral artery infarct. Am J Neuroradiol. 2009;30(3):525–531. doi: 10.3174/ajnr.A1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lees KR, Emberson J, Blackwell L, Stroke Thrombolysis Trialists’ Collaborators Group et al. Effects of alteplase for acute stroke on the distribution of functional outcomes: a pooled analysis of 9 trials. Stroke. 2016;47(9):2373–2379. doi: 10.1161/STROKEAHA.116.013644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jovin TG, Nogueira RG. Dawn in full daylight (abstract) Eur Stroke J. 2017;2:494. [Google Scholar]
- 15.Thomalla G, Fiebach JB, Østergaard L, WAKE-UP investigators et al. A multicenter, randomized, double-blind, placebo-controlled trial to test efficacy and safety of magnetic resonance imaging-based thrombolysis in wake-up stroke (WAKE-UP) Int J Stroke. 2014;9(6):829–836. doi: 10.1111/ijs.12011. [DOI] [PubMed] [Google Scholar]
- 16.Amiri H, Bluhmki E, Bendszus M, et al. European cooperative acute stroke study-4: extending the time for thrombolysis in emergency neurological deficits ECASS-4: ExTEND. Int J Stroke. 2016;11(2):260–267. doi: 10.1177/1747493015620805. [DOI] [PubMed] [Google Scholar]
- 17.ClinicalTrials.gov (Internet). Albers GW (2015) endovascular therapy following imaging evaluation for ischemic stroke 3 (DEFUSE 3). ClinicalTrials.gov identifier: NCT02586415. https://clinicaltrials.gov/ct2/show/NCT02586415. Accessed 12 July 2017
- 18.Albers GW, Thijs VN, Wechsler L, DEFUSE Investigators et al. Magnetic resonance imaging profiles predict clinical response to early reperfusion: the diffusion and perfusion imaging evaluation for understanding stroke evolution (DEFUSE) study. Ann Neurol. 2006;60(5):508–517. doi: 10.1002/ana.20976. [DOI] [PubMed] [Google Scholar]
- 19.Ogata T, Christensen S, Nagakane Y, EPITHET and DIFUSE Investigators et al. The effects of alteplase 3 to 6 hours after stroke in the EPITHET-DEFUSE combined dataset: post hoc case-control study. Stroke. 2013;44(1):87–93. doi: 10.1161/STROKEAHA.112.668301. [DOI] [PubMed] [Google Scholar]
- 20.Davis SM, Donnan GA, Parsons MW, EPITHET investigators et al. Effects of alteplase beyond 3 h after stroke in the echoplanar imaging thrombolytic evaluation trial (EPITHET): a placebo-controlled randomised trial. Lancet Neurol. 2008;7(4):299–309. doi: 10.1016/S1474-4422(08)70044-9. [DOI] [PubMed] [Google Scholar]
- 21.Albers GW, von Kummer R, Truelsen T, DIAS-3 Investigators et al. Safety and efficacy of desmoteplase given 3-9 h after ischaemic stroke in patients with occlusion or high-grade stenosis in major cerebral arteries (DIAS-3): a double-blind, randomised, placebo-controlled phase 3 trial. Lancet Neurol. 2015;14(6):575–584. doi: 10.1016/S1474-4422(15)00047-2. [DOI] [PubMed] [Google Scholar]
- 22.Lansberg MG, Straka M, Kemp S, DEFUSE 2 Study Investigators et al. Magnetic resonance imaging profile and response to endovascular reperfusion: results of the DEFUSE 2 prospective cohort study. Lancet Neurol. 2012;11(10):860–867. doi: 10.1016/S1474-4422(12)70203-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wouters A, Lemmens R, Christensen S, AXIS 2 and DEFUSE 2 study Investigators et al. Magnetic resonance imaging-based endovascular versus medical stroke treatment for symptom onset up to 12 h. Int J Stroke. 2016;11(1):127–133. doi: 10.1177/1747493015607503. [DOI] [PubMed] [Google Scholar]
- 24.Mishra NK, Albers GW, Christensen S, et al. Comparison of magnetic resonance imaging mismatch criteria to select patients for endovascular stroke therapy. Stroke. 2014;45(5):1369–1374. doi: 10.1161/STROKEAHA.114.004772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Goyal M, Demchuk AM, Menon BK BK, ESCAPE Trial Investigators et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015;372(11):1019–1030. doi: 10.1056/NEJMoa1414905. [DOI] [PubMed] [Google Scholar]
- 26.Lansberg MG, Christensen S, Kemp S, CRISP Investigators et al. Computed tomographic perfusion to predict response to recanalization in ischemic stroke. Ann Neurol. 2017;81(6):849–856. doi: 10.1002/ana.24953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Campbell BC, Christensen S, Levi CR, et al. Comparison of computed tomography perfusion and magnetic resonance imaging perfusion-diffusion mismatch in ischemic stroke. Stroke. 2012;43(10):2648–2653. doi: 10.1161/STROKEAHA.112.660548. [DOI] [PubMed] [Google Scholar]
- 28.Singer OC, de Rochemont RDM, Neumann-Haefelin T. Practical limitations of acute stroke MRI due to patient related problems. Neurology. 2004;62(10):1848–1849. doi: 10.1212/01.WNL.0000125320.53244.FA. [DOI] [PubMed] [Google Scholar]
- 29.Gonzalez RG. Low signal, high noise and large uncertainty make CT perfusion unsuitable for acute ischemic stroke patient selection for endovascular therapy. J Neurointerv Surg. 2012;4(4):242–245. doi: 10.1136/neurintsurg-2012-010404. [DOI] [PubMed] [Google Scholar]
- 30.Goyal M, Menon BK, Derdeyn CP. Perfusion imaging in acute ischemic stroke: let us improve the science before changing clinical practice. Radiology. 2013;266(1):16–21. doi: 10.1148/radiol.12112134. [DOI] [PubMed] [Google Scholar]
- 31.Kim BJ, Lee SH, Shin CW, Ryu WS, Kim CK, Yoon BW. Ischemic stroke during sleep: its association with worse early functional outcome. Stroke. 2011;42(7):1901–1906. doi: 10.1161/STROKEAHA.110.602243. [DOI] [PubMed] [Google Scholar]
- 32.Serena J, Dávalos A, Segura T, Mostacero E, Castillo J. Stroke on awakening: looking for a more rational management. Cerebrovasc Dis. 2003;16(2):128–133. doi: 10.1159/000070592. [DOI] [PubMed] [Google Scholar]
- 33.Todo K, Moriwaki H, Saito K, Tanaka M, Oe H, Naritomi H. Early CT findings in unknown-onset and wake-up strokes. Cerebrovasc Dis. 2006;21(5–6):367–371. doi: 10.1159/000091545. [DOI] [PubMed] [Google Scholar]
- 34.Silva GS, Lima FO, Camargo ECS, et al. Wake-up stroke: clinical and neuroimaging characteristics. Cerebrovasc Dis. 2010;29(4):336–342. doi: 10.1159/000278929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Roveri L, La Gioia S, Ghidinelli C, Anzalone N, De Filippis C, Comi G. Wake-up stroke within 3 hours of symptom awareness: imaging and clinical features compared to standard recombinant tissue plasminogen activator treated stroke. J Stroke Cerebrovasc Dis. 2013;22(6):703–708. doi: 10.1016/j.jstrokecerebrovasdis.2011.10.003. [DOI] [PubMed] [Google Scholar]