Abstract
Abnormal expansion of hexanucleotide GGGGCC (G4C2) in the C9ORF72 gene has been associated with multiple neurodegenerative disorders, with particularly high prevalence in amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD). Repeat expansions of this type have been associated with altered pathology, symptom rate and severity, as well as psychological changes. In this study, we enrolled twenty-five patients with ALS and fifteen neurologically healthy controls in a P300 brain-computer interface (BCI) training procedure. Four of the patients were found to possess an expanded allele, which was associated with a reduction in the quality of evoked potentials that led to reduced performance on the BCI task. Our findings warrant further exploration of the relationship between brain function and G4C2 repeat length. Such a relationship suggests that personalized assessment of suitability of BCI as a communication device in patients with ALS may be feasible.
Introduction
The repeat hexanucleotide GGGGCC (G4C2) expansion in the gene C9ORF72 is the most common identified genetic link between amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD). With an upper limit of normal defined as thirty repeats1, 2, abnormal expansion of this sequence is prevalent in 50–72% of patients with familial ALS3, and roughly 7% of sporadic cases4.
ALS patients possessing the repeat expansion exhibit earlier onset, more rapid progression, and earlier death4–10. MRI studies point to structural changes common among repeat carriers that extend beyond the degeneration of the cortico-motor pathways typically involved in ALS7, 11, 12. Transcranial magnetic stimulation studies have identified a reduction in cortical inhibition attributable to this expansion found in ALS patients13, 14. In an electroencephalographic (EEG) study of seven ALS/FTD patients with the repeat expansion, two showed generalized slowing of the background activity, while another two showed intermittent abnormal temporal delta-theta activity15. Another study described a bilateral slowing of frontotemporal theta in three individuals with the expansion16.
Brain-computer interfaces (BCI), specifically the P300 speller17, enable communication without overt movement. The visual P300 spelling task relies on a user’s focus on an intended target while observing a larger group of randomly flashing letters. Reaction to flashing of the target, although producing no overt disturbance, generates a P300 evoked potential over centro-parietal electrode sites, which the computer can interpret as a selection. In the past few years, these devices have been used successfully as assistive communication tools for those with ALS18–20, but declines in performance with severe disease progression have also been documented21–23. The effect of cognitive decline on the performance of these devices has recently been shown to be a major predictor of BCI utility in this population24. The question of whether genotype, particularly in this case as it applies to expansion of C9ORF72, produces measurable changes in the performance of an EEG-based BCI task is unexplored. Our hypothesis is that the BCI task in this study - evocation of the P300 potential and related visually evoked potentials (VEPs) - is performance-dependent on the absence of C9ORF72 expansion. In this paper, we describe a relationship between this repeat expansion and EEG modulation for the purpose of brain-computer interaction.
Results
Twenty-five patients were enrolled along with 15 neurologically-healthy control participants who were age and gender matched (Table 1). One patient did not complete the ALS Cognitive Behavioral Screen (ALS-CBS) because of severe communication difficulties, and another did not complete the behavioral portion of the screen because of the lack of an available caregiver. All patients and 14/15 controls provided samples for assessment of the C9ORF72 repeat expansion. Therefore, 25 patients and 14 control participants were included in the analysis. All control participants possessed hexanucleotide repeat lengths of twelve or fewer (Fig. 1a). Four out of 25 patients (16%) carried a repeat expansion. The presence of a hexanucleotide repeat expansion was not found to be associated with ALS Functional Rating Scale (ALSFRS-R) score, age at symptom onset, nor with cognitive or behavioral scores on the ALS-CBS (Fig. 1b–e).
Table 1.
Patient and control participant demographics.
| Patients (n = 25) | Controls (n = 14) | p-val | |
|---|---|---|---|
| Age, years | 58 (45.5–74) | 55.3 (45–73.5) | 0.292 |
| Education, years | 14 (11.5–24) | 18 (12–24) | 0.005 |
| Gender, % male | 68 | 57 | 0.515 |
Statistics are given as median and range (min-max) and p-values are the result of Wilcoxon rank sum testing between control and patient samples.
Figure 1.
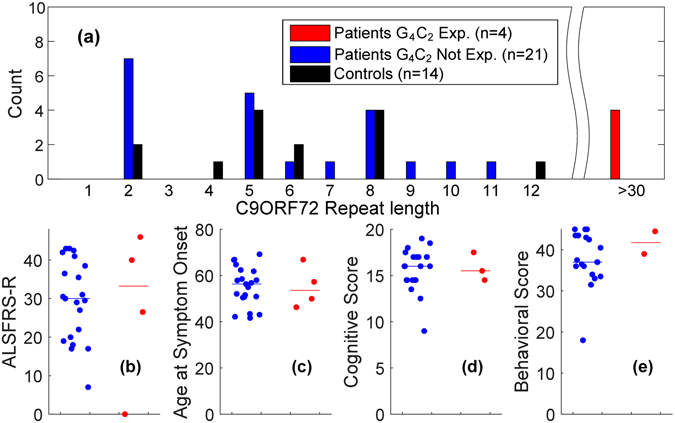
(a) Histogram of C9ORF72 allele lengths. The presence of a repeat expansion among ALS patients (horizontal line indicates group median) does not significantly associate with (b) ALSFRS-R score, (c) age at symptom onset, (d) cognitive impairment, or (e) behavioral impairment.
A VEP quality metric, independent of the BCI classifier, was calculated to represent the discriminability of target vs. non-target EEG responses in the channels Fz, Cz, and Pz. Morphology of VEP quality differed between groups (Fig. 2). Control participants displayed greater magnitude quality responses compared to patients in both early (220–250 ms, 300–370 ms) and late (580–780 ms) evoked potentials. Differences can be seen in both of these periods between patients with and without the repeat expansion, although group-wide differences in median quality exist primarily between patients with the expansion and controls, most notably during the early and late portions of the VEP. The lower half of Fig. 2 shows the time windows in which the 3 group or pairwise comparisons generated a p-value of less than 0.05 (light shading), while the darker shading represent windows retaining significance after False Discovery Rate (FDR) correction (p < 0.0076).
Figure 2.
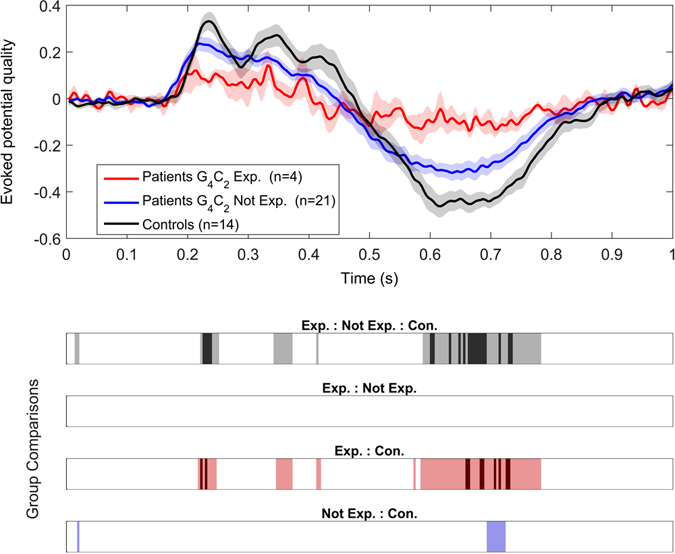
VEP quality is decreased in ALS, particularly in the presence of C9ORF72 expansion. Top: Mean quality of the evoked potential (±SEM) for controls and patients, with and without the expansion. Bottom: Windows where there exist significant differences in VEP quality between the three groups as well as three post-hoc pairwise comparisons. These differences are shown a the α = 0.05 level (light colors) as well as after FDR correction (dark colors).
Online accuracies across groups varied significantly (Fig. 3). The median online P300 spelling accuracy was 23.4% for the patients with the C9ORF72 expansion, 78.1% for patients without the expansion, and 90.6% for controls. The mean online accuracy ranks for each group were found to be significantly different (χ 2 = 10.67, p = 0.005). Post-hoc analysis showed that four patients with the expansion had lower mean rank than the control group (p = 0.005) and there was a non-significant trend to the same effect with the patients without the expansion (p = 0.095). The median offline P300 spelling accuracy was 89.7% for the patients with the C9ORF72 expansion, 100.0% for patients without the expansion, and 100.0% for controls. The accuracies calculated offline were not found to differ significantly between the three groups (χ 2 = 3.43, p = 0.18).
Figure 3.
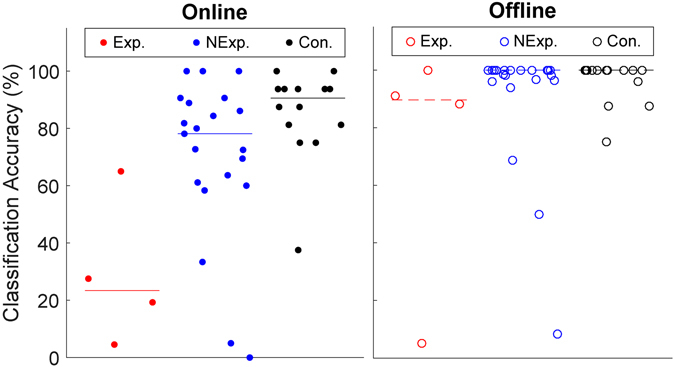
BCI accuracy for all study participants, with group medians represented by a horizontal line. Left: Median online accuracy for patients with the expansion was significantly less than controls (p = 0.005). Right: When analyzed offline, reduction in median accuracy in those with the expansion did not reach significance.
Discussion
Four of the ALS patients in this study possessed an expansion of the C9ORF72 gene, which has been linked to the presence of electrophysiological brain abnormalities, including cortical hyperexcitability13, 14, background rhythm slowing15, and epileptic activity16. While there was both a reduction in VEP quality and online BCI performance in ALS patients as a group, those with the C9ORF72 expansion demonstrated further reductions compared to those without it. This effect was not observed when comparing offline task accuracies across groups. This may be due to a classifier saturation effect; by the completion of the fourth session, patients with the expansion may have accumulated enough data to counteract the noisier signal and create a more generalizable classifier. The comparatively poor online accuracy for these patients may reflect a longer “learning period” for the classifier. In addition, offline accuracy from the control group was calculated with data from only two sessions, which may have also led to a relatively smaller difference in accuracies between patients and controls compared to online results.
The reduced discriminability of target vs. non-target EEG responses was observed most strongly in the early (220–250 ms) and late (580–780 ms) portions of the VEP. This was subclinical, in that those displaying the expansion were no different from patients without the expansion in regards to cognitive and behavioral function or symptom onset. We tried to control for other aspects of the disease which may better explain the observed differences. While we did account for age, time since symptom onset, and education, we did not measure vision or oculomotor control, which has been shown to be a critical factor in P300 speller success for those with ALS22.
Our results raise the possibility of a relationship between an expansion in the C9ORF72 gene and brain function in patients with ALS. Previous work by our group showed that intact cognition, as measured by the ALS-CBS, was the disease characteristic most strongly predictive of P300-BCI performance24. With this present finding, differences in evoked potential production and BCI performance may be at least partially explained by physiological brain changes that are dependent on G4C2 repeat number, independent of ALS-CBS.
Although the study was performed prospectively, there were a few limitations to the procedure which limit the generalizability of these findings. The patients possessing gene expansion did not have a higher incidence of FTD symptoms. Since the presence of cognitive impairment with C9ORF72 mutation is several-fold greater than those without (40–50% vs. 8–9%, respectively)3, we expected our group of four carriers to present with higher incidence of cognitive and/or behavioral impairments of FTD. We are aware, however, that our method for performing neuropsychological screening was far from comprehensive. This limitation and the small sample size may contribute to this missed association which has wide pathological and clinical backing. Better quantification of repeat expansions are also needed, in order to determine whether differences exist between individuals with intermediate repeat lengths (20–100), and large repeats (100–1000+)25. Although we are confident of our analysis leading to identification of patients harboring expansions, we did not have the ability to perform Southern Blotting for determination of precise expansion lengths.
The presence of hexanucleotide repeat expansions that affect BCI usage would have direct applicability for assessment of device utility, and thus could impact effectiveness of such devices in clinical use. Screening for repeat length in ALS patients might serve as one of several tools for personalized assessments of BCI suitability, or might guide the development of training programs for such devices. More broadly, this association raises the possibility of BCI as a diagnostic tool for early detection of non-motor manifestations of ALS.
Methods
The study was approved by the Institutional Review Board of Penn State Hershey Medical Center. All methods were performed in accordance to the guidelines and regulations of The Pennsylvania State University. All participants provided informed consent and were at least 18 years of age. Those in the patient cohort possessed a diagnosis of definite, probable, probable laboratory-supported, or possible ALS by revised El Escorial research criteria26. Those with clinically significant dementia, as determined by the ALS clinic neurologist, were excluded. The control cohort consisted of neurologically healthy adults who were age and gender matched to the patient group. Age, gender, region of onset, time since symptom onset, education level, and ALS Functional Rating Scale - Revised (ALSFRS-R)27 were recorded from each patient. Cognitive and behavioral function were assessed via the ALS-Cognitive Behavioral Screen (ALS-CBS)28.
All subjects provided a saliva or blood sample for genetic analysis. Samples were initially analyzed by fluorescent fragment length polymerase chain reaction (FFL-PCR) to assay for the G4C2 repeat length1. For our FFL-PCR protocol, the largest callable repeat contained 14 G4C2 repeats. Samples determined to be heterozygotes by this method required no further analysis. Homozygotes underwent an additional assay using the Repeat Primed PCR (RP-PCR) method1, which was used to determine if the sample contained a true homozygous repeat number allele as opposed to a potentially pathogenic repeat that had been masked by FFL-PCR. Pathogenic repeats were identified qualitatively by the presence of a “stutter” pattern in the pherogram. In a fourteen-site blinded study, the mean sensitivity and specificity for detection of C9ORF72 repeat expansion correctly from DNA was 95% and 98% respectively, when using this approach29. Fragment analyses were performed with GeneScan 500 ROX dye Size Standard on an ABI3130XL using Peak Scanner 2 software (Thermo Fisher Scientific).
Patients completed four sessions of EEG-BCI training over the course of 1–2 months, each with a half-hour training entailing 4–8 runs of a P300 spelling task. Control participants completed two sessions. Each run of the P300 spelling task consisted of copy spelling a 4 letter word, where each trial culminated in the selection of 1 letter after flashing each grid icon 20 times. A checkerboard-type speller with 32 targets30 was used to evoke the P300 signal from the user. Targets were highlighted for 187.5 ms, and the interstimulus interval was 62.5 ms.
During the recordings, EEG electrodes were affixed in an electrode cap at 19 locations in the 10–20 system, with ground at Fpz, and referenced to linked earlobes. Additionally, electrodes were placed on the forehead and lateral canthi for the purpose of artifact reduction. Electrode paste was used to mount the electrodes on the scalp, and impedances were generally kept below 10 kΩ. In this data set, we demonstrated previously that electrode impedance did not associate with BCI performance24. Signals were amplified and digitized with two g.USBamp amplifiers (g.tec GmbH). Data acquisition, signal processing, and feedback generation were performed by a customized program in BCI200031. Ocular artifact reduction was automated though a regression procedure32, followed by rejection of epochs which exceeded an amplitude of ±75 μV. Features forwarded to the classifier were the stimulus time-locked average EEG signals, downsampled to 20 Hz. Stepwise selection of regression coefficients was used on training data to generate a classifier for predicting intended targets online30. The classifier was generated using the stepwisefit function in MATLAB, and was updated after each feedback run.
Following all recordings, we utilized the Wilcoxon rank sum test to determine whether the presence of a repeat expansion was associated with cognitive or behavioral impairment, as well as age and ALSFRS-R score. Following this, the ‘quality’ of the BCI control signal was calculated. Signal quality acted as a classifier-independent measure of the robustness of the control signal. This metric was calculated for the BCI task as the standard distance between data belonging to the two classes of interest. As an example, Q Cz, the quality of the target-specific evoked potential in channel Cz, is the standard distance between target and non-target evoked potentials for the trials of that task,
| 1 |
| 2 |
where Cz T represent the EEG responses in channel Cz to target stimuli and Cz N represent the EEG responses to non-target stimuli, which occur at a ratio of 1:7. To compute the standard difference, the difference in means, μ, of these two groups are divided by their pooled standard deviation, σ.
Signal quality for the P300 task was averaged over channels Fz, Cz, and Pz. To determine whether there was a significant difference in the quality of the BCI control signals between groups, a series of Kruskal Wallis tests were performed at each EEG sample over one second following VEP extraction. Significant results were followed by post-hoc tests of pairwise comparisons using the Tukey-Kramer method, if necessary. To correct for multiple comparisons, the type-1 error rate threshold was modified using the False Discovery Rate method33. Finally, we reanalyzed all the EEG data offline using the same feature extraction and classification scheme to determine the offline accuracy for each individual. Data was segmented in a leave-one-trial-out manner for estimation of offline accuracy.
Acknowledgements
The authors would like to thank the patients, their caregivers, and the staff of the Penn State Hershey Medical Center ALS Clinic and Research Center. We thank the staff of the Institute for Personalized Medicine, especially Qunyan Jin, Cynthia Reed, Sue Patrick, Lisa Schneper, and Glenn Gerhard, as well as Robert Brucklacher of the Penn State Hershey Genome Sciences Core Facility. This work was supported by the endowment funds of Harvey F. Brush, the Paul and Harriet Campbell Fund for ALS Research, the ALS Association Greater Philadelphia Chapter, and many other private donations to the Penn State Hershey ALS Center.
Author Contributions
A.G., Z.S., and S.J.S. conceived the experiment, A.G. conducted the BCI experiment, K.E.S. and J.R.B. collected and processed genetic samples for C9ORF72 allele lengths, and A.G. analysed the results. All authors reviewed the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.DeJesus-Hernandez M, et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011;72:245–256. doi: 10.1016/j.neuron.2011.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Renton AE, et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72:257–268. doi: 10.1016/j.neuron.2011.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sabatelli M, et al. New als-related genes expand the spectrum paradigm of amyotrophic lateral sclerosis. Brain Pathology. 2016;26:266–275. doi: 10.1111/bpa.12354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cooper-Knock J, et al. Clinico-pathological features in amyotrophic lateral sclerosis with expansions in C9ORF72. Brain. 2012;135:751–764. doi: 10.1093/brain/awr365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beck J, et al. Large C9orf72 hexanucleotide repeat expansions are seen in multiple neurodegenerative syndromes and are more frequent than expected in the UK population. The American Journal of Human Genetics. 2013;92:345–353. doi: 10.1016/j.ajhg.2013.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gijselinck I, et al. A C9orf72 promoter repeat expansion in a flanders-belgian cohort with disorders of the frontotemporal lobar degeneration-amyotrophic lateral sclerosis spectrum: a gene identification study. The Lancet Neurology. 2012;11:54–65. doi: 10.1016/S1474-4422(11)70261-7. [DOI] [PubMed] [Google Scholar]
- 7.Byrne S, et al. Cognitive and clinical characteristics of patients with amyotrophic lateral sclerosis carrying a C9orf72 repeat expansion: a population-based cohort study. The Lancet Neurology. 2012;11:232–240. doi: 10.1016/S1474-4422(12)70014-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Irwin DJ, et al. Cognitive decline and reduced survival in C9orf72 expansion frontotemporal degeneration and amyotrophic lateral sclerosis. Journal of Neurology, Neurosurgery & Psychiatry. 2013;84:163–169. doi: 10.1136/jnnp-2012-303507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hübers A, et al. Polymerase chain reaction and Southern blot-based analysis of the C9orf72 hexanucleotide repeat in different motor neuron diseases. Neurobiology of Aging. 2014;35:1214.e1–e6. doi: 10.1016/j.neurobiolaging.2013.11.034. [DOI] [PubMed] [Google Scholar]
- 10.van Blitterswijk M, et al. Association between repeat sizes and clinical and pathological characteristics in carriers of C9ORF72 repeat expansions (Xpansize-72): a cross-sectional cohort study. The Lancet Neurology. 2013;12:978–988. doi: 10.1016/S1474-4422(13)70210-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bede P, et al. Multiparametric MRI study of ALS stratified for the C9orf72 genotype. Neurology. 2013;81:361–369. doi: 10.1212/WNL.0b013e31829c5eee. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Simón-Sánchez J, et al. The clinical and pathological phenotype of C9ORF72 hexanucleotide repeat expansions. Brain. 2012;135:723–735. doi: 10.1093/brain/awr353. [DOI] [PubMed] [Google Scholar]
- 13.Geevasinga N, et al. Cortical function in asymptomatic carriers and patients with c9orf72 amyotrophic lateral sclerosis. JAMA Neurology. 2015;72:1268–1274. doi: 10.1001/jamaneurol.2015.1872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schanz O, et al. Cortical hyperexcitability in patients with c9orf72 mutations: Relationship to phenotype. Muscle & Nerve. 2016;54:264–269. doi: 10.1002/mus.25047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kaivorinne A-L, et al. Clinical characteristics of C9ORF72 -linked frontotemporal lobar degeneration. Dementia and Geriatric Cognitive Disorders. 2013;3:251–262. doi: 10.1159/000351859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Capasso, M., Anzellotti, F., Giacomo, R. D. & Onofrj, M. Epilepsy and electroencephalographic abnormalities in c9orf72 repeat expansion. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration0, 1–2, doi:10.1080/21678421.2016.1231825, PMID: 27652840 (2016). [DOI] [PubMed]
- 17.Farwell L, Donchin E. Talking off the top of your head: toward a mental prosthesis utilizing event-related brain potentials. Electroencephalography and Clinical Neurophysiology. 1988;70:510–523. doi: 10.1016/0013-4694(88)90149-6. [DOI] [PubMed] [Google Scholar]
- 18.Silvoni S, et al. P300-based brain-computer interface communication: Evaluation and follow-up in amyotrophic lateral sclerosis. Frontiers in neuroscience. 2009;3 doi: 10.3389/neuro.20.001.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kaufmann T, et al. Face stimuli effectively prevent brain–computer interface inefficiency in patients with neurodegenerative disease. Clinical Neurophysiology. 2013;124:893–900. doi: 10.1016/j.clinph.2012.11.006. [DOI] [PubMed] [Google Scholar]
- 20.Mak JN, et al. Eeg correlates of p300-based brain–computer interface (bci) performance in people with amyotrophic lateral sclerosis. Journal of Neural Engineering. 2012;9 doi: 10.1088/1741-2560/9/2/026014. [DOI] [PubMed] [Google Scholar]
- 21.Kübler A, Birbaumer N. Brain-computer interfaces and communication in paralysis: Extinction of goal directed thinking in completely paralysed patients? Clinical Neurophysiology. 2008;119:2658–2666. doi: 10.1016/j.clinph.2008.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.McCane LM, et al. Brain-computer interface (BCI) evaluation in people with amyotrophic lateral sclerosis. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration. 2014;15:207–215. doi: 10.3109/21678421.2013.865750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chaudhary, U., Birbaumer, N. & Ramos-Murguialday, A. Chapter 5 - Brain–computer interfaces in the completely locked-in state and chronic stroke. In Coyle, D. (ed.) Brain-Computer Interfaces: Lab Experiments to Real-World Applications, vol. 228 of Progress in Brain Research, 131–161 (Elsevier, 2016). [DOI] [PubMed]
- 24.Geronimo A, Simmons Z, Schiff SJ. Performance predictors of brain–computer interfaces in patients with amyotrophic lateral sclerosis. Journal of Neural Engineering. 2016;13 doi: 10.1088/1741-2560/13/2/026002. [DOI] [PubMed] [Google Scholar]
- 25.Rohrer JD, et al. {C9orf72} expansions in frontotemporal dementia and amyotrophic lateral sclerosis. The Lancet Neurology. 2015;14:291–301. doi: 10.1016/S1474-4422(14)70233-9. [DOI] [PubMed] [Google Scholar]
- 26.Brooks BR, Miller RG, Swash M, Munsat TL. El escorial revisited: Revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotrophic Lateral Sclerosis & Other Motor Neuron Disorders. 2000;1:293–299. doi: 10.1080/146608200300079536. [DOI] [PubMed] [Google Scholar]
- 27.Cedarbaum JM, et al. The ALSFRS-R: a revised ALS functional rating scale that incorporates assessments of respiratory function. Journal of the Neurological Sciences. 1999;169:13–21. doi: 10.1016/S0022-510X(99)00210-5. [DOI] [PubMed] [Google Scholar]
- 28.Woolley SC, et al. Detecting frontotemporal dysfunction in ALS: Utility of the ALS cognitive behavioral screen (ALS-CBS) Amyotrophic Lateral Sclerosis. 2010;11:303–311. doi: 10.3109/17482961003727954. [DOI] [PubMed] [Google Scholar]
- 29.Akimoto C, et al. A blinded international study on the reliability of genetic testing for ggggcc-repeat expansions in c9orf72 reveals marked differences in results among 14 laboratories. Journal of Medical Genetics. 2014;51:419–424. doi: 10.1136/jmedgenet-2014-102360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Townsend G, et al. A novel P300-based brain-computer interface stimulus presentation paradigm: moving beyond rows and columns. Clinical Neurophysiology. 2010;121:1109–1120. doi: 10.1016/j.clinph.2010.01.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schalk, G. et al. BCI2000: A General-Purpose Brain-Computer Interface (BCI) System, IEEE Trans Biomed Eng, 51(6), 1034–1043 (2004). [DOI] [PubMed]
- 32.Schlögl A, et al. A fully automated correction method of EOG artifacts in EEG recordings. Clinical Neurophysiology. 2007;118:98–104. doi: 10.1016/j.clinph.2006.09.003. [DOI] [PubMed] [Google Scholar]
- 33.Benjamini Y, Hochberg Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. Journal of the Royal Statistical Society. Series B (Methodological) 1995;57:289–300. [Google Scholar]


