Abstract
Plant defensins (PDFs) are cysteine-rich peptides that have a range of biological functions, including defence against fungal pathogens. However, little is known about their role in defence against bacteria. In this study, we showed that the protein encoded by ARABIDOPSIS THALIANA PLANT DEFENSIN TYPE 1.1 (AtPDF1.1) is a secreted protein that can chelate apoplastic iron. Transcripts of AtPDF1.1 were induced in both systemic non-infected leaves of Arabidopsis thaliana plants and those infected with the necrotrophic bacterium Pectobacterium carotovorum subsp. carotovorum (Pcc). The expression levels of AtPDF1.1 with correct subcellular localization in transgenic A. thaliana plants were positively correlated with tolerance to Pcc, suggesting its involvement in the defence against this bacterium. Expression analysis of genes associated with iron homeostasis/deficiency and hormone signalling indicated that the increased sequestration of iron by apoplastic AtPDF1.1 overexpression perturbs iron homeostasis in leaves and consequently activates an iron-deficiency-mediated response in roots via the ethylene signalling pathway. This in turn triggers ethylene-mediated signalling in systemic leaves, which is involved in suppressing the infection of necrotrophic pathogens. These findings provide new insight into the key functions of plant defensins in limiting the infection by the necrotrophic bacterium Pcc via an iron-deficiency-mediated defence response.
Introduction
Organisms have evolved multiple defence strategies for protection against pathogen attack, many of which are triggered by environmental cues. One such strategy is the innate immune response, which utilizes Toll-like or pattern-recognition receptors to perceive specific pathogen-associated molecular patterns (PAMPs) that are derived from pathogens to trigger the defence signalling pathways. In plants, this process involves the activation of the salicylic acid (SA), jasmonic acid (JA) and ethylene (ET) signaling1 pathways and the biosynthesis of defence molecules, such as antimicrobial peptides (AMPs)2, 3, which cause membrane disruption, pore formation, and lead to cell lysis and death4. Based on sequence similarity, disulphide bond patterns and characteristic structural folds, AMPs can be categorized into several groups, including defensins, thionins, hevein-like peptides, knottin, α-hairpinin and lipid transfer proteins (LTPs)5–7.
Defensins constitute a well-studied group of relatively small AMPs (34–54 amino acids) with cysteine-rich motifs, which are present in a taxonomically broad range of multicellular eukaryotic organisms8. Unlike animal defensins which mainly possess anti-bacterial properties, plant defensins (PDFs), which have a highly conserved cysteine composition but are highly variable in their primary sequence8, have primarily been associated with protection against fungi5, 8, 9 and heavy metal stress10, 11. In contrast to the well-established actions of plant PDFs in limiting fungal infection, little is known about their roles and modes of action against bacteria5.
To date, more than 300 defensin-like (DEFL) genes have been identified in the model plant Arabidopsis thaliana 12. AtPDF1.1 was originally described as encoding a seed-localized protein13, but its expression was later reported to be induced in leaves infected with the fungus Blumeria graminis f. sp. hordei 14, 15.
An association between PDFs and metal homeostasis was also indicated by our preliminary data, which showed that AtPDF1.1 expression was induced upon iron overloading but not iron deficiency (Fig. S1B,D). Iron is essential for most living organisms and is involved in many biological processes16. In plants, intracellular iron is maintained at a very low level and excess iron is sequestered and stored in the apoplast and organelles, including the vacuole and plastid, to reduce iron toxicity17. Accordingly, iron homeostasis in plants is tightly controlled at the transcriptional and posttranslational levels18, 19. For example, Fe-deficiency-induced transcription factor 1 (FIT1) has been shown to interact with other iron homeostasis responsive transcription factors, such as basic helix-loop-helix 38 and 39 (bHLH38 and bHLH 39), to activate the expression of FERRIC CHELATE REDUCTION OXIDASE 2 (FRO2) and IRON-REGULATED TRANSPORTER 1 (IRT1) in A. thaliana 20.
Iron is also important for bacterial pathogens as it is required for their metabolism, enzymatic functions and pathogenesis. In order to acquire iron, plant pathogens utilize host iron-containing compounds such as heme21, transferrin, and lactoferrin, or synthesize siderophores to compete for iron uptake with their hosts22. Moreover, genes associated with immunity and iron homeostasis are also induced in response to treatment with the iron chelators ethylenediamine- di(o-hydroxyphenylacetic acid) (EDDHA) and desferrioxamine (DFO), suggesting that iron sequestration activates an immune response23. Together these findings demonstrated the existence of a relationship between perturbation of iron distribution and immunity in plants.
Given that AtPDF1.1 confers plant tolerance to fungi and that our preliminary data revealed increased transcript levels of AtPDF1.1 following iron overloading, in this study, we provide evidence that apoplastic AtPDF1.1 perturbs iron homeostasis via iron chelation, resulting in activation of the iron-deficiency-induced ethylene signalling pathway. This response confers protection against infection by Pectobacterium carotovorum subsp. carotovorum (Pcc), a necrotrophic pathogen that causes bacterial soft rot (BSR) and serious production losses of a wide range of economically important crops worldwide24.
Results
Transcript levels of AtPDF1s increase in response to iron treatment and Pcc infection
The overexpression of AhPDF1.1 in A. thaliana and yeast has been shown to enhance tolerance to zinc10. To evaluate whether PDF1 genes in A. thaliana are similarly involved in zinc response, the expression profiles of the orthologs of AhPDF1.1, namely AtPDF1.1, AtPDF1.2a and AtPDF1.3, were monitored during ZnSO4 treatment. The results showed that the expression of AtPDF1.2a and AtPDF1.3, but not AtPDF1.1, was upregulated by ZnSO4 treatment (Fig. S1A), whereas transcript levels of these three AtPDF1 genes were upregulated by treatment with other essential divalent ions, such as FeSO4 and CuSO4 (Fig. S1B,C). This suggested that AtPDF1.1 may be involved in iron and copper homeostasis, but not in zinc homeostasis. In contrast to iron overloading, the expression of AtPDF1 gene was not dramatically affected by iron deficient treatment (Fig. S1D).
To evaluate whether these three AtPDF1 genes are involved in defence against bacterial pathogens, their transcriptional profiles were monitored in A. thaliana infected with Pcc. Time-course studies revealed that all three AtPDF1 genes were upregulated at the infection site 1 hour post infection (hpi), with maximum expression occurring at 3 hpi for AtPDF1.2 and AtPDF1.3, and at 4 hpi for AtPDF1.1 (Fig. S2A). Analysis of non-inoculated leaves from infected plants revealed that the transcript levels of AtPDF1.1, but not AtPDF1.2 or AtPDF1.3, were induced systemically at 24 hpi (Fig. S2B). In order to investigate the protein level of AtPDF1.1, the AtPDF1.1:green fluorescent protein fusion protein (AtPDF1.1:GFP) under the control of the AtPDF1.1 promoter was used to distinguish from other native defensins. When transgenic A. thaliana plants expressing AtPDF1.1:GFP were infected with Pcc, maximum protein accumulation was observed at the local site of infection at 3–4 hpi (Fig. S2C). Furthermore, in addition to a protein band of ~36 kDa, which corresponds to the predicted size of the AtPDF1.1-GFP fusion protein, a protein of ~33 kDa was also dramatically accumulated at the local site of infection at 5 hpi and in systemic leaves at 24 hpi (Fig. S2C), suggesting that AtPDF1.1 may have undergone post-translational modification, and AtPDF1.1 expression is induced both locally and systemically upon Pcc infection. Along with this notion, analysis of the predicted AtPDF1.1 protein sequence using SignalP 4.1 software (http://www.cbs.dtu.dk/services/SignalP/) revealed the presence of a putative N-terminal secretory signal peptide (Fig. S3A), suggesting that the AtPDF1.1 protein may be targeted to the secretory pathway25, 26, and the 33-kDa protein is likely resulted from the cleavage of the N-terminal signal. Consistent with this targeting prediction, the first identified plant defensin was reported to be a secreted protein released from germinated radish seeds27.
AtPDF1.1 confers plant tolerance to Pcc infection
Since AtPDF1.1 transcript levels increased in response to Pcc infection (Fig. S2), we then investigated the role of AtPDF1.1 in defending against pathogenic bacteria. Suppression (RNAi) transgenic lines were generated, because AtPDF1.1 expression in the only available AtPDF1.1 T-DNA insertion mutant (SALK_029051) is unaltered compared to the wild-type plant. In addition, to determine whether the cellular localization of AtPDF1.1 is critical for its functions, transgenic plants overexpressing a truncated form of AtPDF1.1, lacking the N-terminal signal peptide (AtPDF1.1ΔSP) and AtPDF1.1 overexpression (OE) were also generated (Fig. S3B). Quantitative PCR (qPCR) analysis verified the success of AtPDF1.1 OE and RNAi in transgenic plants (Fig. S3C). The results showed that, compared with the empty vector (Ev) harbouring control plants, the AtPDF1.1 OE plants exhibited enhanced tolerance to Pcc infection, the AtPDF1.1ΔSP plants exhibited similar disease response, while the AtPDF1.1 RNAi plants exhibited increased susceptibility (Fig. 1). These results indicated that the tolerance to Pcc infection correlates well with the expression level of AtPDF1.1, and that the putative N-terminal secretory signal peptide, which could be a key determinant for the apoplastic location of AtPDF1.1, is crucial for its function in defence against Pcc infection.
Figure 1.
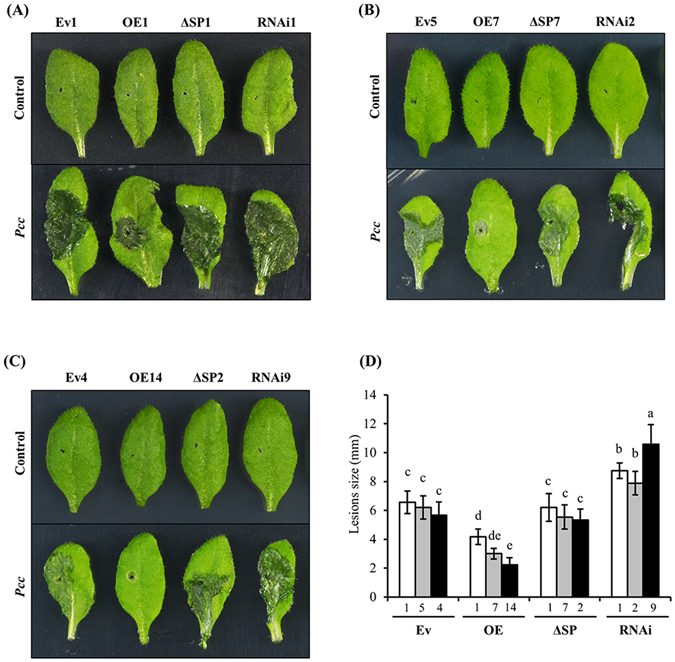
Expression levels of AtPDF1.1 correlate with protection against Pcc infection. (A,B and C) Transgenic Arabidopsis thaliana plants (Ev: line1, 5, 4; OE: line1, 7, 14; ΔSP: line1, 7, 2; RNAi: line1, 2, 9) were inoculated with Pcc or water (control) and Pcc-mediated symptoms were evaluated at 24 hpi. (D) The maximal lesion sizes (diameter; mm) on leaves, denoting bacterial soft rot severity, were determined at 24 hpi. Values are means ± standard errors from ten samples for each line and each infection in a single experiment that was repeated at least three times with similar results. Different letters above each bar indicate significant difference (Fisher’s Least Significant Difference (LSD) LSD post hoc one-way ANOVA, P < 0.05).
AtPDF1.1 is secreted into the apoplast
To determine the subcellular localization of AtPDF1.1, we generated transgenic A. thaliana plants overexpressing GFP fused to the C-terminal of full-length AtPDF1.1-encoding gene (AtPDF1.1:GFP), or a truncated AtPDF1.1-encoding gene lacking either the signal peptide (AtPDF1.1ΔSP:GFP) or the mature protein region (AtPDF1.1(SP only):GFP) (Fig. 2A). A. thaliana lines harbouring the empty expression vector pH7FWG2 (Ev-GFP) were used as a negative control (Fig. 2A). Due to the presence of the ccdB and chloramphenicol-resistance (CmR) gene cassette upstream of the GFP-encoding gene in this vector, GFP transcripts are not translated into functional protein. Confocal microscope imaging of the plasmolysed epidermis of the leaves of transgenic plants revealed that the GFP signal was localized in the apoplastic region surrounding the plasma membrane of AtPDF1.1:GFP and AtPDF1.1(SP only):GFP plants (Fig. 2B–D and H–J), while AtPDF1.1ΔSP:GFP plants showed the GFP signal in the cytosol (Fig. 2E–G). We concluded that the mature form of AtPDF1.1 is localized in the apoplast, which is consistent with the localization of other plant defensins27, 28.
Figure 2.
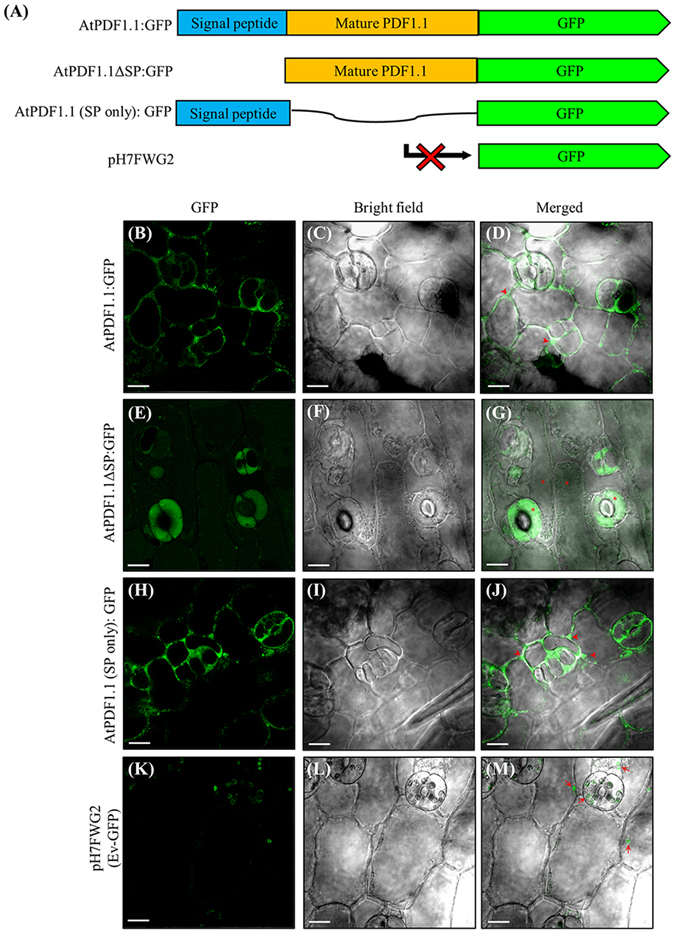
AtPDF1.1 fusion proteins are localized and accumulated in the apoplast. (A) Schematic diagram of the AtPDF1.1:GFP (green fluorescent protein), AtPDF1.1ΔSP:GFP and AtPDF1.1(SP only):GFP fusion constructs. The leaf epidermis of AtPDF1.1:GFP (B–D), AtPDF1.1ΔSP:GFP (E–G), AtPDF1.1(SP only):GFP (H–J) and Ev-GFP transgenic Arabidopsis thaliana plants (K–M) were subjected to confocal microscopy to determine the subcellular localization of AtPDF1.1 fusion proteins. Left panel, GFP fluorescence; middle panel, bright field images; right panel, merged images. Each line was repeated at least three times with similar results. Asterisks (*) denote GFP signals detected in the cytosol (asterisks), arrowheads denote GFP signals detected in the apoplastic space and arrows denote the chlorophyll fluorescence. Bars, 10 μm.
To confirm that AtPDF1.1 is secreted, levels of the recombinant fusion protein were measured in the growth medium of suspension cells generated from transgenic plants (Fig. S4A) together with levels in protein lysates from whole seedlings, by western blot analysis with an anti-GFP antibody. Two GFP fusion protein bands were detected in the seedling lysates of AtPDF1.1:GFP and AtPDF1.1(SP only):GFP plants, suggesting that AtPDF1.1 with and without the N-terminal signal peptide were expressed and that AtPDF1.1(SP only):GFP (band 3 and 4) may increase molecular weight via certain post-translational modification (Fig. S4B). In contrast, a single GFP fusion protein band was observed in the protein lysate of AtPDF1.1ΔSP:GFP seedlings. When the proteins in the media of the suspension cells were analysed, only GFP fusion proteins containing the N-terminal signal peptide were detected in the supernatants of AtPDF1.1:GFP and AtPDF1.1(SP only):GFP suspension cell cultures, and no immunoreactive proteins were detected in the growth medium of AtPDF1.1ΔSP:GFP suspension cells (Fig. S4C). These data further suggest that the N-terminal signal peptide is responsible for targeting AtPDF1.1 to the apoplast. Because the apoplast is the initial site for Pcc infection29, and the correct localization of AtPDF1.1 is critical for its function in defence against Pcc infection (Fig. 1), our results are consistent with the idea that apoplastic AtPDF1.1 can interfere with Pcc infection.
AtPDF1.1 localized to the apoplast is involved in iron distribution
The apoplast is a primary infection site for bacterial pathogens of plants, and pathogens such as P. syringae rely on extracellular nutrients30. The iron status of A. thaliana has been reported to be critical for the pathogenicity of the necrotrophic bacterium Dickeya dadantii 31. Under host iron limiting conditions, bacteria produce siderophores to enhance their iron uptake32. Since AtPDF1.1 expression is responsive to both iron overloading (Fig. S1B) and Pcc infection (Fig. S2), we hypothesized that AtPDF1.1 may function as an iron chelator to limit the availability of iron for Pcc as part of an iron-withholding defence system33. To determine whether apoplastic iron affects Pcc pathogenesis, exogenous iron was supplemented to plants and disease response was assessed. The results showed that extra iron supplement to the leaves increased the susceptibility of all transgenic plants to Pcc invasion (Fig. S5), supporting the importance of apoplastic iron for Pcc infection. However, even with extra iron supplement, the AtPDF1.1 OE plants still displayed the lowest susceptibility to Pcc infection, further supporting a positive role of AtPDF1.1 in plant defence against Pcc.
To evaluate whether AtPDF1.1 is involved in chelating/sequestering iron, we first determined whether it has the capacity to bind metal ions. In a metal affinity assay (Fig. S6), AtPDF1.1ΔSP showed binding affinity to iron ions. Since the resins derivatized from iminodiacetic acid exhibited different degrees of affinity to metal ions, with a preference for transition metals, such as cobalt (Co), nickel (Ni), copper (Cu) and zinc (Zn)34–36, we deduced that fewer Fe3+-conjugated resins are available for AtPDF1.1ΔSP protein to bind. This may explain the relatively weak protein band detected, and so the intensity of detection signal does not correlate with the binding affinity between the metal ions and AtPDF1.1ΔSP protein. Overall, the data suggest that AtPDF1.1ΔSP protein is able to bind to iron ions.
To further evaluate the iron binding affinity of the AtPDF1.1 protein in planta, transgenic plants were grown under hydroponic conditions and their iron acquisition capabilities were compared. As shown in Fig. 3A, Perls staining revealed a high abundance of iron ions in the intercellular regions of the root epidermis of AtPDF1.1 OE plants, but only weak staining was observed in the Ev, AtPDF1.1ΔSP and AtPDF1.1 RNAi transgenic plants. Furthermore, sections of roots and leaves from transgenic plants were subjected to Perls/diaminobenzidine (DAB)/H2O2 staining and then examined by confocal microscopy to spot regions with iron deposits. Consistent with previous studies of A. thaliana wild-type plants37, the apoplastic regions of the cortex of roots were stained in all sections (Fig. 3B), indicating extracellular iron accumulation. However, compared to the Ev, AtPDF1.1ΔSP and AtPDF1.1RNAi transgenic plants, more heavily stained zones were observed in the apoplast of the root epidermis and cortex of AtPDF1.1 OE plants. Similarly, substantial amounts of iron aggregates were observed in the leaf apoplast of AtPDF1.1 OE plants (Fig. 3C), while only low levels of iron aggregates were present in the Ev, AtPDF1.1ΔSP and AtPDF1.1 RNAi plants. Since the amounts of iron aggregates accumulated in the apoplast, where AtPDF1.1 is localized (Fig. 2), correlated well with the expression levels of AtPDF1.1 (Fig. 3) and AtPDF1.1 can bind to iron (Fig. S6), we projected that AtPDF1.1 can chelate/sequester iron.
Figure 3.

Iron aggregate is accumulated in the apoplast by AtPDF1.1 protein. The roots of four-week-old hydroponically grown transgenic A. thaliana plants were harvested. (A) The root epidermis was subjected to Perls Prussian Blue staining, and iron distribution was examined. Bars, 50 μm. Transverse sections of roots (B) and leaves (C) were subjected to Perls/DAB/H2O2 staining and iron distribution was examined. Each line was repeated at least three times with similar results. Arrows indicate the presence of iron accumulating in the apoplast; e, c and x denote the epidermis, cortex and xylem, respectively. Bars, 10 μm.
We next determined whether apoplastic sequestration of iron by AtPDF1.1 might affect the intracellular iron content. The results showed that AtPDF1.1 OE and AtPDF1.1 RNAi plants, respectively, had the highest and lowest apoplastic iron content in the roots and leaves (Fig. S7A,B), and that the levels of apoplastic iron were inversely correlated with intracellular iron content (Fig. S7C,D). Given that roots are the site for iron uptake38, we concluded that the binding of iron by AtPDF1.1 in the root apoplast might limit the amount of intracellular iron, hence accounting for the low levels of iron in the root and leaves of the AtPDF1.1 OE plants.
We next evaluated the effect of Pcc infection on systemic iron content. As shown in Fig. S7A and C, Pcc infection caused a decrease in root apoplastic iron content and an increase in root intracellular iron content. In contrast, an increase in leaf apoplastic iron content after Pcc infection was observed in all transgenic plants, except the AtPDF1.1 OE lines (Fig. S7B), while the leaf intracellular iron content was not significantly changed (Fig. S7D). AtPDF1.1 overexpression seemed to restrict iron in leaf apoplasts without significantly affecting the leaf intracellular iron content. Because Pcc infection is mainly confined to leaves during the duration of our experiments, these results suggested that Pcc infection results in root apoplastic iron depletion and induces a change in root-to-shoot translocation of iron.
When transgenic plants were grown under long-term-hydroponic conditions, slight chlorosis was observed in the leaves of the AtPDF1.1 OE plants, but chlorophyll content was higher and leaves were greener in the AtPDF1.1 RNAi plants (Fig. S8). Iron is involved in the formation of δ-aminolevulinic acid, which is required for chlorophyll formation, and loss of chlorophyll is associated with a lack of iron availability39. Since the iron distribution of the leaf was aggregated in the apoplast of AtPDF1.1 transgenic OE plants (Figs 3C and 7B), these results are consistent with the observation that the AtPDF1.1 OE plant had lower chlorophyll content than the others (Fig. S8).
Figure 7.
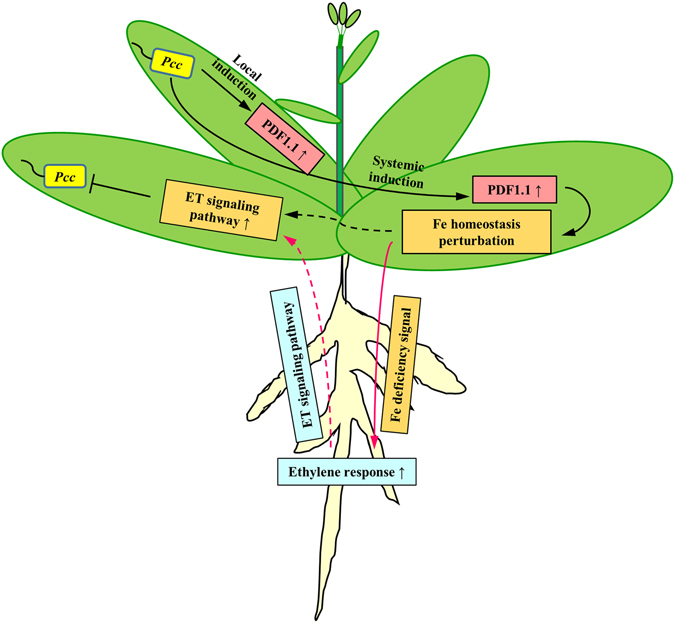
A proposed model of AtPDF1.1-mediated defence response to the infection by Pcc. During Pcc infection, transcription of AtPDF1.1 is upregulated in both the local infected and systemic non-infected leaves. The production of AtPDF1.1 protein leads to the chelation/restriction of iron availability in the apoplast, and the consequent perturbation of iron homeostasis. This results in the activation of an iron deficiency response in leaves and the subsequent activation of the ethylene (ET) biosynthesis and signalling pathway in roots. This in turn causes the systemic activation of a jasmonic acid (JA)/ET-mediated immune response in leaves, which promotes tolerance to the infection of necrotrophic pathogens.
Chelation of iron in the leaf by apoplastic AtPDF1.1 induces an iron deficiency response
Given that the apoplastic iron content is positively associated with AtPDF1.1 expression level, we next examined whether the activation of the regulatory pathways involved in iron homeostasis correlated with AtPDF1.1 expression. Analysis of the transcriptional profiles of the iron homeostasis transcription factor genes, FIT1, bHLH38, bHLH39, bHLH100 and bHLH101 40 showed that, under non-infected condition, these genes were significantly upregulated in the leaves of AtPDF1.1 OE plants, and, with the exception of FIT1, bHLH38 and bHLH101, were downregulated in the AtPDF1.1 RNAi plants, (Fig. 4). Similarly, transcript levels of the iron-deficiency-responsive genes, IRT1, FRO2, NAS1 and NAS4 41, 42 were higher and lower, respectively, in the roots of AtPDF1.1 OE and AtPDF1.1 RNAi plants compared with the Ev plants (Fig. 4). This suggests that the high levels of AtPDF1.1 protein present in the apoplast result in a perturbation of iron homeostasis and consequently a state of intracellular iron deficiency.
Figure 4.

Iron deficiency-associated genes are upregulated in AtPDF1.1 OE plants. The leaves and roots were harvested from hydroponically grown transgenic A. thaliana plants (Ev: line 1, 5, 4; OE: line1, 7, 14; ΔSP: line 1, 7, 2; RNAi: line 1, 2, 9) and the expression levels of genes associated with iron homeostasis (FIT1, bHLH38, bHLH39, bHLH100 and bHLH101) in the leaves, and iron deficiency response (IRT1, FRO2, NAS1 and NAS4) in the roots were monitored by qPCR and compared with expression in the empty vector control plants (Ev4, defined value of 1). Transcript levels were normalized to those of ACTIN2. Values are means ± standard errors from twelve samples for each line in a single experiment that was repeated at least three times with similar results. Different letters above each bar indicate significant difference (LSD post hoc one-way ANOVA, P < 0.05).
When plants were subjected to Pcc infection, transcript levels of the iron homeostasis transcription factor genes, bHLH38, bHLH39 and bHLH100 and the iron-deficiency-responsive genes, IRT1, FRO2, and NAS4, were increased in the systemic leaves or roots of all transgenic plants (Fig. S9). This indicated that Pcc infection induces an iron deficiency response that is independent of AtPDF1.1 transcript levels.
Ferric chelate reductase (FCR) is required to reduce Fe3+ to Fe2+ in the rhizoshere to allow iron uptake into the cell, and its activity is highly activated in response to iron deficiency43. As shown in Fig. S10, FCR activity was the highest and lowest, respectively, in the roots of AtPDF1.1 OE and AtPDF1.1 RNAi plants, suggesting that the AtPDF1.1 OE plants were experiencing greater iron deficiency. In addition, FCR activity was further enhanced by Pcc infection, further confirming that Pcc infection can lead to an iron deficiency response (Fig. S10). The observation that Pcc infection caused an increase in FCR activity in all transgenic plants was consistent with the decreased root apoplastic iron content and the increased root intracellular iron content, respectively (Fig. S7A,C).
A defence response is activated upon the induction of an iron deficiency response
To examine whether iron aggregation in the apoplast increased as a result of Pcc infection, both locally infected and systemic leaves were harvested at 4 and 24 hpi and subjected to Perls/DAB/H2O2 staining analysis. In the absence of Pcc infection, iron aggregates were only detected in the leaves of the AtPDF1.1 OE plants. When plants were infected with Pcc, enhanced accumulation of iron aggregates was observed in the local and systemic leaf tissues of all transgenic plants, particularly near to the vascular bundles (Fig. S11). This suggested that Pcc infection induced iron accumulation both locally and systemically, and that the accumulation of iron aggregates was further enhanced by the presence of AtPDF1.1. AtPDF1.1-mediated enhancement on Pcc-induced iron accumulation would account for the overall perturbation of iron homeostasis and elicitation of the iron deficiency response observed in transgenic lines (Figs S9 and S10).
Studies by Alia Dellagi et al.33 revealed that the sequestration of iron by synthetic iron chelators or bacterial siderophores can induce the expression of genes associated with iron homeostasis and the subsequent activation of immune responses, including callose deposition, hydrogen peroxide production and a SA-mediated response23. The transcriptional profiles of JA/ET- and SA-responsive genes were evaluated to investigate whether the AtPDF1.1-mediated activation of iron homeostasis genes (Fig. 4) also triggered a plant defence response23. As shown in Fig. 5A, expression of the JA/ET-responsive genes, ERF1, ERF2, PR3, PR4 and AtPDF1.2a, was upregulated in the AtPDF1.1 OE plants. However, the expression of VSP2 and MYC2, which are in the MYC-branch of the JA response pathway44, was not induced in any of transgenic plants. This suggests that ET signalling, but not JA signalling, may respond to altered AtPDF1.1 expression. In contrast, most of the tested genes associated with the SA signalling pathway (WRKY46, WRKY53 and WRKY70 and PR1) were vaguely downregulated and upregulated, respectively, in the AtPDF1.1 OE and AtPDF1.1 RNAi plants (Fig. 5B). Upon Pcc infection, genes associated with the JA/ET responses were upregulated in all transgenic plants, with the highest induced levels in the AtPDF1.1 OE plants (Fig. S12A). In contrast, the expression profiles of genes associated with the SA response were inconsistent and not correlate with AtPDF1.1 expression levels (Fig. S12B). These results suggest that AtPDF1.1 action and Pcc infection promote the JA-/ET-mediated signalling. Since the JA/ET signalling pathway is associated with defence against necrotrophic pathogens, including Pcc 45, these results may explain the enhanced tolerance to Pcc infection in the AtPDF1.1 OE plants (Fig. 1), but not to the hemibiotrophic bacterium Pst DC3000 (Fig. S13).
Figure 5.
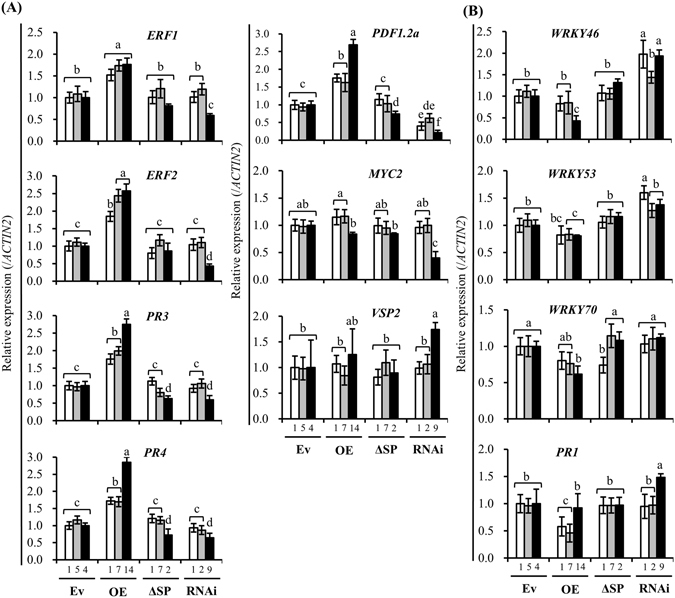
Expression levels of downstream genes of ERF1/2 are increased in AtPDF1.1 OE plants. The leaves were harvested from hydroponically grown transgenic A. thaliana plants (Ev: line 1, 5, 4; OE: line 1, 7, 14; ΔSP: line 1, 7, 2; RNAi: line 1, 2, 9) and the expression levels of JA/ET-responsive genes (ERF1, ERF2, PR3, PR4, PDF1.2a, MYC2 and VSP2; (A)) and SA-responsive genes (WRKY46, WRKY53, WRKY70 and PR1; (B)) were monitored by qPCR and compared with expression in empty vector control plants (Ev4, defined value of 1). Transcript levels were normalized to those of ACTIN2. Values are means ± standard errors from twelve samples for each line in a single experiment that was repeated at least three times with similar results. Different letters above each bar indicate significant difference (LSD post hoc one-way ANOVA, P < 0.05).
The state of iron deficiency leads to the induction of ethylene biosynthesis in the roots46, 47. Since iron deficiency response occurred in the leaves and roots of AtPDF1.1 OE plants (Figs 4 and S10), we evaluated whether the state of iron deficiency induced by AtPDF1.1 overexpression also led to activation of ethylene signalling in the roots. As shown in Fig. 6, the ET-biosynthesis genes, SAM1, ACS6 and ACO2 (Fig. 6A) and ET-signalling genes, ETR1 and EIN2 (Fig. 6B) were expressed at higher levels in APDF1.1 OE plants compared to the other transgenic plants. The activation of ET signalling in the AtPDF1.1 OE plants may explain why the JA-/ET-responsive ERF1/2 and their downstream genes, but not the JA-responsive MYC2 and VSP2, were upregulated (Fig. 5A)48. Furthermore, the expression of JA/SA biosynthesis genes was not significantly affected by AtPDF1.1 (Fig. 6C), but the expression of ET/JA biosynthesis genes and ET signalling genes in the roots was increased at varied levels after Pcc infection (Fig. S14). To further verify whether ethylene signalling is involved in AtPDF1.1-mediated tolerance to Pcc, the ethylene inhibitor 1-methylcyclopropene (1-MCP) was applied to transgenic plants, followed by inoculation with Pcc. With the 1-MCP treatment, the soft rot symptom caused by Pcc was enhanced in all of transgenic plants, including the AtPDF1.1 OE plants (Fig. S15). This result further points to the involvement of ET signalling in AtPDF1.1-mediated tolerance to Pcc infection.
Figure 6.
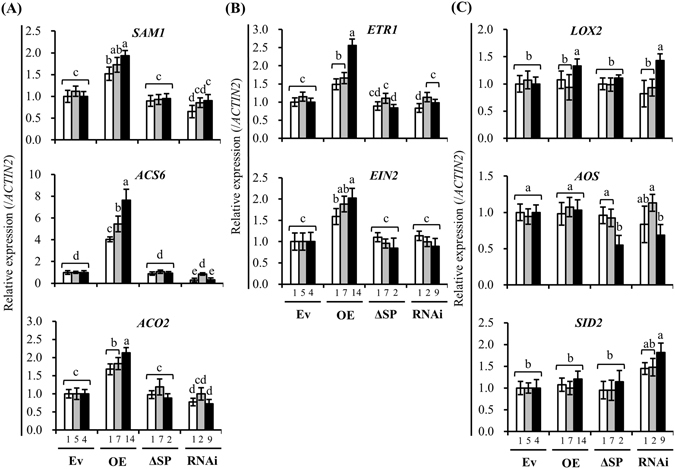
ET-signalling genes are upregulated in AtPDF1.1 OE plant. The roots from hydroponically-grown transgenic plants (Ev: line1, 5, 4; OE: line1, 7, 14; ΔSP: line1, 7, 2; RNAi: line1, 2, 9) were harvested and transcript levels of ET-biosynthesis genes (SAM1, ACS6 and ACO2; (A)), ET-signalling genes (ETR1 and EIN2; (B)) JA-/SA-biosynthesis genes (LOX2, AOS and SID2; (C)) were monitored by qPCR and compared with expression in empty vector control plants (Ev4, defined value of 1). Transcript levels were normalized to ACTIN2. Values are means ± standard errors from twelve samples for each line in a single experiment that was repeated at least three times with similar results. Different letters above each bar indicate significant difference (LSD post hoc one-way ANOVA, P < 0.05).
Discussion
Iron is an essential nutrient for hosts and pathogens21 and so the iron status of a plant can affect the pathogenesis of pathogens31. One of the first lines of defence against bacterial infection is to sequester and withhold iron in an iron-storage compartment, limiting bacterial growth and pathogenicity49. In this study, we found that transcription levels of AtPDF1.1 are elevated during Pcc infection (Fig. S2) and that the AtPDF1.1 protein is secreted into the apoplast to sequester iron (Figs 2 and 3 and S11). We propose that this process serves as a key defence mechanism, limiting iron availability and thereby compromising the pathogenesis of Pcc. This hypothesis was supported by the increased sensitivity to Pcc invasion of the AtPDF1.1 OE plants after iron supplementation (Fig. S5).
The state of iron deficiency in AtPDF1.1 OE plants leads to the induction of ET signalling in the roots (Fig. 6)46, 47, which can then activate the ERF branch, but not the MYC branch, of the JA/ET-mediated response in systemic leaves (Fig. 5). Since the JA/ET signalling pathway is involved in the defence against Pcc 44, 45, the ability of AtPDF1.1 to systemically induce an iron-deficiency-mediated ET response would be one of its functions in defending off Pcc. The fact that blocking ET signalling led to increased susceptibility to Pcc infection (Fig. S15) further reinforces the involvement of ET signalling in AtPDF1.1-mediated tolerance to Pcc infection.
Our study showed that AtPDF1.1 could chelate iron (Figs 3 and S11) and its overexpression caused an iron deficiency response (Fig. 4), which correlates well with the promoted tolerance to Pcc infection (Figs 1 and S5). These results mirror previous studies which demonstrated that iron deficiency triggers defence responses and prompts resistance against pathogenic microbes. For instance, Koen et al.50 showed that treatment of β-aminobutyric acid (BABA), a nonprotein amino acid able to chelate iron, led to a transient iron deficiency response which is linked with a protective effect against microbial pathogens. In addition, siderophore pyoverdine, which is produced by the beneficial bacterium Pseudomonas fluorescens, restored the growth of A. thaliana under iron-deficient conditions by repressing defence pathways and activating iron acquisition/redistribution signalling51, further demonstrating the link between iron deficiency and immune responses.
During Pcc infection, genes associated with both JA/ET- and SA-mediated defence responses are activated52. In this study, we showed that plants ectopically expressing AtPDF1.1 had pre-existing protection against Pcc infection (Fig. 1) and exhibited an iron deficiency state (Figs 4 and S8). In addition, the iron-deficiency-mediated JA/ET response is further enhanced upon Pcc infection (Fig. S12A), suggesting that AtPDF1.1 has a synergistic effect on the Pcc-mediated iron deficiency response, and is capable of amplifying the ET defence response. Pcc infection also led to decreased iron content in root apoplasts (Fig. S7A) and activated JA/ET signalling (Fig. S12A). Therefore, exposing plants to AtPDF1.1 prior to pathogen infection may have a protective and amplifying effect on their disease defence responses.
SA-mediated defence responses are involved in preventing infection by the hemibiotrophic bacterial pathogen Pst DC300053. Bacterial siderophores induce an iron-deficiency-mediated SA immune response to defend against Pst DC3000 in A. thaliana by restricting Fe3+ availability in the leaf cell wall. This restriction results in a perturbation of iron distribution and activates an iron deficiency response23. Although AtPDF1.1 also has an iron-sequestration action, and is capable of inducing an iron-deficiency-mediated response. The expression of AtPDF1.1 does not affect defence against Pst DC3000 infection (Fig. S13) nor the activation of genes involved in SA signalling (Fig. 5B). Interestingly, siderophore pyoverdine produced by the beneficial bacterium Pseudomonas fluorescens has an opposite effect, in which pyoverdine promoted Arabidopsis growth under iron-deficient conditions by repressing defence pathways, including SA and abscisic acid, leading to increased susceptibility to Botrytis cinerea 52. Taken together, these results indicate that the iron-deficiency-mediated signalling triggered by AtPDF1.1 specifically enhances the ET-mediated defence response. The differences in the immune response pathways activated by the AtPDF1.1 protein and siderophores may be due to their different properties. First, the cysteine-rich AtPDF1.1 utilizes sulphur (S) atoms as an electron donor to bind Fe2+ and Fe3+ ions (Fig. S6B), whereas siderophores predominantly chelate Fe3+ via their oxygen (O) ligands22, 23, 54, 55. In addition, AtPDF1.1 is induced systemically during Pcc infection (Fig. S2B), whereas siderophores are not proteins and are secreted by pathogens at the infection site22.
The fact that AtPDF1.1 overexpression resulted in high levels of apoplastic iron accumulation (Fig. 3), which was accompanied by a decrease in intracellular iron content, and reduced AtPDF1.1 expression gave an opposite effect (Fig. S7) suggests that the chelation of iron by AtPDF1.1 in the apoplast influences intracellular iron levels. It has been reported that coumarin phenolic compounds and nicotianamine can chelate apoplastic iron ions and are required for iron uptake from roots41, 56, 57. We postulate that high levels of AtPDF1.1 proteins present in the apoplast may compete for iron ions with coumarin phenolic compounds and nicotianamine, resulting in the sequestration of iron in the apoplast (Figs 3 and S11). In addition, siderophore-mediated-upregulation of AtFer1 has been identified as an iron-withholding defence against the proliferation of the necrotrophic bacterium D. dadantii 33. We propose that AtPDF1.1 may similarly compete with bacterial siderophores for iron binding and thereby mediate an iron-withholding defence against Pcc invasion.
In addition to its known antifungal function58, this study shows that AtPDF1.1 possesses an iron-withholding defence function to limit the growth of the necrotrophic bacterial pathogen Pcc via the activation of the ET defence pathway (Fig. 7). During Pcc infection, signals induced by the pathogen may trigger the translocation of iron ions from the root apoplast to the leaf apoplast for bacterial utilization and proliferation. As a defence strategy, plants elevate AtPDF1.1 expression, and apoplast-localized AtPDF1.1 chelates iron, thereby iron homeostasis is perturbed to activate an iron deficiency response in the leaf. In addition, this may potentially lead to the activation of ET signalling and biosynthesis in the roots, and subsequently trigger an ET-mediated defence response in systemic leaves to counteract the further attack of pathogens45, 59. The pre-existing ET defence response mediated by AtPDF1.1 in the AtPDF1.1 OE plants may account for the increased tolerance to Pcc infection, while suppressing AtPDF1.1 expression reduces defence against Pcc infection.
Methods
Plant materials and growth conditions
The growth conditions and selection of Arabidopsis are described in Method S1.
Generation of transgenic plants
The constructions of AtPDF1.1 OE (OE), AtPDF1.1ΔSP (ΔSP), AtPDF1.1 RNAi, AtPDF1.1:GFP, AtPDF1.1ΔSP:GFP, AtPDF1.1 (SP only):GFP and Arabidopsis transformation are described in Method S2.
Gene expression analysis following metal treatments and Pcc infection
Ten-day old seedlings of Ev plants were cultured on ½ MS medium supplemented with 600 μM ZnSO4, 600 μM FeSO4, 75 μM CuSO4 or 300 μM ferrozine (Aldrich,160601) for 0 to 5 days. Four-week old soil-grown Ev plants were punctured with a 5 ml syringe needle and inoculated with or without 3 μl liquid culture of 5 × 106 cfu ml−1 of Pcc (three rosette leaves/plant) and local and systemic leaves were harvested at 1–5 hours or 4–24 hours post inoculation (hpi). Total RNA was isolated using the Trizol RNA isolation reagent (Invitrogen), according to the manufacturer’s instructions. cDNAs were synthesized using the M-MLV reverse transcriptase cDNA synthesis kit (Promega) according to the supplier’s instructions. Actin2 (AT3G18780) or UBC21 (AT5G25760) were used as internal normalization controls. qPCR was performed as described by Ko et al.60. The primers used to analyse AhPDF1s have already been described by Shahzad et al.11. Primers used for gene expression studies are listed in Table S2.
Plant pathogen inoculation and disease response assay
For the Pcc infection assay, bacteria were grown at 28 °C overnight in Luria-Bertani (LB) medium (Difco, 244620). Three rosette leaves from each four-week old soil-grown plant were punctured with a 5 ml syringe needle and inoculated with 3 μl liquid culture of 5 × 106 cfu ml−1 of Pcc or water (control). Bacterial soft rot symptoms were evaluated periodically. The maximal lesion sizes (diameter; mm) on leaves were recorded at 24 hpi using the protocol described by Li et al.61. For the P. syringae pv. tomato (Pst) DC3000 infection assay, the bacteria were grown at 28 °C overnight in King’s B medium containing 50 μg ml−1 rifampicin62. Plants were sprayed with liquid culture of 2 × 108 cfu mL−1 of Pst DC3000 containing 0.02% Silwet L-77 or 10 mM MgSO4 (control). Disease symptoms were evaluated periodically and in planta bacterial CFU counts in leaves were measured at 3 dpi.
Subcellular protein localization
Ten-day-old transgenic plants were immersed in 1× phosphate-buffered saline (pH 7.4; MDBio) for 30 min to induce plasmolysis and to stabilize EGFP fusion proteins in the apoplast63, 64. The true leaves were removed and imaging was performed using a Zeiss confocal microscope LSM 710 (Zeiss Axio Observer Z.1) with a Plan-Apochromat 100×/1.40 Oil DIC M27 objective lens. For detection of GFP signals, a 488 nm excitation wavelength and a 500–550 nm emission wavelength were used.
Detection of secreted proteins
Suspension cells of transgenic plants were induced as described by May and Leaver65. The culture media were harvested weekly and combined. Each 200 ml of culture media was centrifuged at 8,000 g for 10 min to remove debris, and filtered through Miracloth (Calbiochem) and two layers of Whatman grade 1 qualitative filter paper. The filtrates were lyophilized using a freeze dryer (Uniss), and dissolved in 10 ml milli-Q H2O and then desalted and concentrated to 10-fold volume using Vivaspin 6 centrifugal filters (Sartorius). The protein concentrations in the different samples were measured by protein assay dye reagent (Bio-Rad [5000006]) and adjusted to the same value with SDS sample buffer66 and 20 µg protein per sample was used for immunoblot analysis.
Immunoblot analysis
Experimental procedures are described in Method S3.
Metal binding assay
The experimental design and procedure are described in Method S4.
Detection of iron distribution
For Perls staining, roots were vacuum infiltrated with Perls staining solution containing equal volumes of 4% (v/v) HCl and 4% (w/v) K-ferrocyanide for 15 min and incubated at room temperature for 30 min67. The formation of blue pigments, denoting iron accumulation, was observed using light microscopy. For Perls/DAB/H2O2 staining, leaves and roots were vacuum infiltrated with fixation solution and dehydrated as described by Roschzttardtz et al.68. Embedding and sectioning were performed as described by Lin et al.69. Sections for infected leaves (7 mm from the inoculation site), systemic leaves and roots were subjected to Perls/DAB/H2O2 staining68 and imaging using a Zeiss LSM 710 confocal microscope fitted with an EC Plan-Neofluar 20×/0.50 M27 lens, under transmitted light (bright field).
Measurement of metal concentration
The experimental design and procedure are described in Method S5.
FCR activity assays
FCR activity was measured in four-week-old transgenic plants grown in hydroponic medium. Three leaves from each plant were punctured and inoculated with 3 μl liquid culture of 5 × 106 cfu ml−1 of Pcc or water (control). The roots were harvested at 24 hpi and a modified FCR activity assay was performed as previously described70.
Iron infiltration and treatment of ethylene inhibitor
The 2.5 mM MES buffer, pH 5.6 containing with or without FeSO4 was infiltrated into the Arabidopsis leaves using 1 ml syringes without a needle, and followed by inoculation of Pcc 1 hour after iron infiltration.
Four-week-old soil-grown transgenic plants were treated with ethylene inhibitor, 1-methylcyclopropene (1-MCP, Lytone Enterprise, one tablet containing 2 mg 1-MCP was dissolved in 15 ml milli-Q H2O) in a 150 L sealed chamber for 24 hours, followed by inoculation with Pcc.
Accession numbers
The sequences of genes used in this article were obtained from The Arabidopsis Information Resource (TAIR, http://www.arabidopsis.org/index.jsp). The accession numbers of the genes are described in Method S6.
Electronic supplementary material
Acknowledgements
We are grateful to research assistants; Mr. Chin-Lin Chung at the Confocal Microscope Core Facility, Biotechnology Experimental Center in Southern Taiwan for his assistance with confocal microscopy; Ms. Fu-Hui Wu, Mr. Chen-Tran Hsu and Ms. Shu-Chen Shen at the Transformation Core Facility and the Confocal Microscope Core Facility, respectively, of the Agricultural Biotechnology Research Center, Academia Sinica, Taiwan for their assistance with the transformation and confocal microscopy; Dr. Kuo-Chen Yeh at the Agricultural Biotechnology Research Center, Academia Sinica, Taiwan for providing comments on the manuscript; and to Ms. Miranda Loney for English editing and providing comments and suggestions. We thank PlantScribe (www.plantscribe.com) for editing this manuscript. This work was supported by the Academia Sinica (to M.T.C.) and the Ministry of Science and Technology, Taiwan (103-2311-B-002-010-, 104-2311-B-002-009, 105-2311-B-002-002, to C.P.C.).
Author Contributions
P.Y.H. performed the experiments, interpreted data, generated figures and wrote the main part of the manuscript; C.P.C. and M.T.C. conceived and supervised the project, contributed to the writing and were involved in data interpretation; K.W.K. assisted in writing the manuscript and data interpretation; M.T.C. wrote the manuscript with contributions from all the authors.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-08497-7
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Chiu-Ping Cheng, Email: chiupingcheng@ntu.edu.tw.
Ming-Tsair Chan, Email: mbmtchan@gate.sinica.edu.tw.
References
- 1.Pieterse CMJ, Leon-Reyes A, Van der Ent S, Van Wees SCM. Networking by small-molecule hormones in plant immunity. Nat. Chem. Biol. 2009;5:308–316. doi: 10.1038/nchembio.164. [DOI] [PubMed] [Google Scholar]
- 2.Raub JA, Mathieu-Nolf M, Hampson NB, Thom SR. Carbon monoxide poisoning–a public health perspective. Toxicology. 2000;145:1–14. doi: 10.1016/S0300-483X(99)00217-6. [DOI] [PubMed] [Google Scholar]
- 3.Janeway CA., Jr. Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb. Symp. Quant. Biol. 1989;54(Pt 1):1–13. doi: 10.1101/SQB.1989.054.01.003. [DOI] [PubMed] [Google Scholar]
- 4.De Coninck B, Cammue BPA, Thevissen K. Modes of antifungal action and in planta functions of plant defensins and defensin-like peptides. Fungal Biol. Rev. 2013;26:109–120. doi: 10.1016/j.fbr.2012.10.002. [DOI] [Google Scholar]
- 5.Dias RO, Franco OL. Cysteine-stabilized alphabeta defensins: From a common fold to antibacterial activity. Peptides. 2015;72:64–72. doi: 10.1016/j.peptides.2015.04.017. [DOI] [PubMed] [Google Scholar]
- 6.Marmiroli N, Maestri E. Plant peptides in defence and signalling. Peptides. 2014;56:30–44. doi: 10.1016/j.peptides.2014.03.013. [DOI] [PubMed] [Google Scholar]
- 7.Tam J, Wang S, Wong K, Tan W. Antimicrobial peptides from plants. Pharmaceuticals. 2015;8:711–757. doi: 10.3390/ph8040711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Vriens K, Cammue BP, Thevissen K. Antifungal plant defensins: mechanisms of action and production. Molecules. 2014;19:12280–12303. doi: 10.3390/molecules190812280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kaur J, Sagaram US, Shah D. Can plant defensins be used to engineer durable commercially useful fungal resistance in crop plants? Fungal Biol. Rev. 2011;25:128–135. doi: 10.1016/j.fbr.2011.07.004. [DOI] [Google Scholar]
- 10.Mirouze M, et al. A putative novel role for plant defensins: a defensin from the zinc hyper-accumulating plant, Arabidopsis halleri, confers zinc tolerance. Plant J. 2006;47:329–342. doi: 10.1111/j.1365-313X.2006.02788.x. [DOI] [PubMed] [Google Scholar]
- 11.Shahzad Z, et al. Plant Defensin type 1 (PDF1): protein promiscuity and expression variation within the Arabidopsis genus shed light on zinc tolerance acquisition in Arabidopsis halleri. New Phytol. 2013;200:820–833. doi: 10.1111/nph.12396. [DOI] [PubMed] [Google Scholar]
- 12.Silverstein KA, Graham MA, Paape TD, VandenBosch KA. Genome organization of more than 300 defensin-like genes in Arabidopsis. Plant Physiol. 2005;138:600–610. doi: 10.1104/pp.105.060079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Terras FR, et al. A new family of basic cysteine-rich plant antifungal proteins from Brassicaceae species. FEBS Lett. 1993;316:233–240. doi: 10.1016/0014-5793(93)81299-F. [DOI] [PubMed] [Google Scholar]
- 14.Zimmerli L, Stein M, Lipka V, Schulze-Lefert P, Somerville S. Host and non-host pathogens elicit different jasmonate/ethylene responses in Arabidopsis. Plant J. 2004;40:633–646. doi: 10.1111/j.1365-313X.2004.02236.x. [DOI] [PubMed] [Google Scholar]
- 15.Nguyen NN, et al. Evolutionary tinkering of the expression of PDF1s suggests their joint effect on zinc tolerance and the response to pathogen attack. Front. Plant Sci. 2014;5:70. doi: 10.3389/fpls.2014.00070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kobayashi T, Nishizawa NK. Iron uptake, translocation, and regulation in higher plants. Annu. Rev. Plant Biol. 2012;63:131–152. doi: 10.1146/annurev-arplant-042811-105522. [DOI] [PubMed] [Google Scholar]
- 17.Briat JF, Curie C, Gaymard F. Iron utilization and metabolism in plants. Curr. Opin. Plant Biol. 2007;10:276–282. doi: 10.1016/j.pbi.2007.04.003. [DOI] [PubMed] [Google Scholar]
- 18.Shin LJ, et al. IRT1 degradation factor1, a ring E3 ubiquitin ligase, regulates the degradation of iron-regulated transporter1 in Arabidopsis. Plant Cell. 2013;25:3039–3051. doi: 10.1105/tpc.113.115212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Connolly EL, Fett JP, Guerinot ML. Expression of the IRT1 metal transporter is controlled by metals at the levels of transcript and protein accumulation. Plant Cell. 2002;14:1347–1357. doi: 10.1105/tpc.001263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yuan Y, et al. FIT interacts with AtbHLH38 and AtbHLH39 in regulating iron uptake gene expression for iron homeostasis in Arabidopsis. Cell Res. 2008;18:385–397. doi: 10.1038/cr.2008.26. [DOI] [PubMed] [Google Scholar]
- 21.Costa A, et al. AIR12, a b-type cytochrome of the plasma membrane of Arabidopsis thaliana is a negative regulator of resistance against Botrytis cinerea. Plant Sci. 2015;233:32–43. doi: 10.1016/j.plantsci.2015.01.004. [DOI] [PubMed] [Google Scholar]
- 22.Winkelmann G. Ecology of siderophores with special reference to the fungi. Biometals. 2007;20:379–392. doi: 10.1007/s10534-006-9076-1. [DOI] [PubMed] [Google Scholar]
- 23.Aznar A, et al. Scavenging iron: a novel mechanism of plant immunity activation by microbial siderophores. Plant Physiol. 2014;164:2167–2183. doi: 10.1104/pp.113.233585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kim HS, et al. Overexpression of the Brassica rapa transcription factor WRKY12 results in reduced soft rot symptoms caused by Pectobacterium carotovorum in Arabidopsis and Chinese cabbage. Plant Biol. (Stuttg.) 2014;16:973–981. doi: 10.1111/plb.12149. [DOI] [PubMed] [Google Scholar]
- 25.Bendtsen JD, Nielsen H, von Heijne G, Brunak S. Improved prediction of signal peptides: SignalP 3.0. J. Mol. Biol. 2004;340:783–795. doi: 10.1016/j.jmb.2004.05.028. [DOI] [PubMed] [Google Scholar]
- 26.Oomen RJ, et al. Plant defensin AhPDF1.1 is not secreted in leaves but it accumulates in intracellular compartments. New Phytol. 2011;192:140–150. doi: 10.1111/j.1469-8137.2011.03792.x. [DOI] [PubMed] [Google Scholar]
- 27.Terras FR, et al. Small cysteine-rich antifungal proteins from radish: their role in host defence. Plant Cell. 1995;7:573–588. doi: 10.1105/tpc.7.5.573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Amien S, et al. Defensin-like ZmES4 mediates pollen tube burst in maize via opening of the potassium channel KZM1. PLoS Biol. 2010;8:e1000388. doi: 10.1371/journal.pbio.1000388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Toth IK, et al. Evaluation of phenotypic and molecular typing techniques for determining diversity in Erwinia carotovora subspp. atroseptica. J. Appl. Microbiol. 1999;87:770–781. doi: 10.1046/j.1365-2672.1999.00929.x. [DOI] [PubMed] [Google Scholar]
- 30.Yu X, et al. Transcriptional analysis of the global regulatory networks active in Pseudomonas syringae during leaf colonization. MBio. 2014;5:e01683–01614. doi: 10.1128/mBio.01683-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kieu NP, et al. Iron deficiency affects plant defence responses and confers resistance to Dickeya dadantii and Botrytis cinerea. Mol. Plant Pathol. 2012;13:816–827. doi: 10.1111/j.1364-3703.2012.00790.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Franza T, Mahe B, Expert D. Erwinia chrysanthemi requires a second iron transport route dependent of the siderophore achromobactin for extracellular growth and plant infection. Mol. Microbiol. 2005;55:261–275. doi: 10.1111/j.1365-2958.2004.04383.x. [DOI] [PubMed] [Google Scholar]
- 33.Dellagi A, et al. Siderophore-mediated upregulation of Arabidopsis ferritin expression in response to Erwinia chrysanthemi infection. Plant J. 2005;43:262–272. doi: 10.1111/j.1365-313X.2005.02451.x. [DOI] [PubMed] [Google Scholar]
- 34.Oshima T, Oshima C, Baba Y. Selective extraction of histidine derivatives by metal affinity with a copper (II)-chelating ligand complex in an aqueous two-phase system. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2015;990:73–79. doi: 10.1016/j.jchromb.2015.03.023. [DOI] [PubMed] [Google Scholar]
- 35.Kavaklı PA, Kavaklı C, Güven O. Preparation and characterization of Fe (III)-loaded iminodiacetic acid modified GMA grafted nonwoven fabric adsorbent for anion adsorption. Radiat. Phys. Chem. 2014;94:105–110. doi: 10.1016/j.radphyschem.2013.07.003. [DOI] [Google Scholar]
- 36.Das A. Stabilities of ternary complexes of cobalt (II), nickel (II), copper (II) and zinc (II) involving aminopolycarboxylic acids and heteroaromatic N-bases as primary ligands and benzohydroxamic acid as a secondary ligand. Transit. Metal. Chem. 1990;15:399–402. doi: 10.1007/BF01177470. [DOI] [Google Scholar]
- 37.Lei GJ, et al. Abscisic acid alleviates iron deficiency by promoting root iron reutilization and transport from root to shoot in Arabidopsis. Plant Cell Environ. 2014;37:852–863. doi: 10.1111/pce.12203. [DOI] [PubMed] [Google Scholar]
- 38.Bienfait HF, van den Briel W, Mesland-Mul NT. Free space iron pools in roots: generation and mobilization. Plant Physiol. 1985;78:596–600. doi: 10.1104/pp.78.3.596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pushnik JC, Miller GW, Manwaring JH. The role of iron in higher plant chlorophyll biosynthesis, maintenance and chloroplast biogenesis. J. Plant Nutr. 1984;7:733–758. doi: 10.1080/01904168409363238. [DOI] [Google Scholar]
- 40.Sivitz AB, Hermand V, Curie C, Vert G. Arabidopsis bHLH100 and bHLH101 control iron homeostasis via a FIT-independent pathway. PLoS One. 2012;7:e44843. doi: 10.1371/journal.pone.0044843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Koen E, et al. Arabidopsis thaliana nicotianamine synthase 4 is required for proper response to iron deficiency and to cadmium exposure. Plant Sci. 2013;209:1–11. doi: 10.1016/j.plantsci.2013.04.006. [DOI] [PubMed] [Google Scholar]
- 42.Wu H, et al. Co-overexpression FIT with AtbHLH38 or AtbHLH39 in Arabidopsis-enhanced cadmium tolerance via increased cadmium sequestration in roots and improved iron homeostasis of shoots. Plant Physiol. 2012;158:790–800. doi: 10.1104/pp.111.190983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Robinson NJ, Procter CM, Connolly EL, Guerinot ML. A ferric-chelate reductase for iron uptake from soils. Nature. 1999;397:694–697. doi: 10.1038/17800. [DOI] [PubMed] [Google Scholar]
- 44.Anderson JP, et al. Antagonistic interaction between abscisic acid and jasmonate-ethylene signalling pathways modulates defence gene expression and disease resistance in Arabidopsis. Plant Cell. 2004;16:3460–3479. doi: 10.1105/tpc.104.025833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pre M, et al. The AP2/ERF domain transcription factor ORA59 integrates jasmonic acid and ethylene signals in plant defence. Plant Physiol. 2008;147:1347–1357. doi: 10.1104/pp.108.117523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Garcia MJ, Suarez V, Romera FJ, Alcantara E, Perez-Vicente R. A new model involving ethylene, nitric oxide and Fe to explain the regulation of Fe-acquisition genes in Strategy I plants. Plant Physiol. Biochem. 2011;49:537–544. doi: 10.1016/j.plaphy.2011.01.019. [DOI] [PubMed] [Google Scholar]
- 47.Garcia MJ, Lucena C, Romera FJ, Alcantara E, Perez-Vicente R. Ethylene and nitric oxide involvement in the up-regulation of key genes related to iron acquisition and homeostasis in Arabidopsis. J. Exp. Bot. 2010;61:3885–3899. doi: 10.1093/jxb/erq203. [DOI] [PubMed] [Google Scholar]
- 48.Walley JW, et al. The chromatin remodeler SPLAYED regulates specific stress signalling pathways. PLoS Pathog. 2008;4:e1000237. doi: 10.1371/journal.ppat.1000237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fones H, Preston GM. The impact of transition metals on bacterial plant disease. FEMS Microbiol. Rev. 2013;37:495–519. doi: 10.1111/1574-6976.12004. [DOI] [PubMed] [Google Scholar]
- 50.Trapet P, et al. The Pseudomonas fluorescens siderophore pyoverdine weakens Arabidopsis thaliana defense in favor of growth in iron-deficient conditions. Plant Physiol. 2016;171(1):675–93. doi: 10.1104/pp.15.01537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Koen E, et al. β-Aminobutyric acid (BABA)-induced resistance in Arabidopsis thaliana: link with iron homeostasis. Mol. Plant Microbe Interact. 2014;27(11):1226–40. doi: 10.1094/MPMI-05-14-0142-R. [DOI] [PubMed] [Google Scholar]
- 52.Po-Wen C, Singh P, Zimmerli L. Priming of the Arabidopsis pattern-triggered immunity response upon infection by necrotrophic Pectobacterium carotovorum bacteria. Mol. Plant Pathol. 2013;14:58–70. doi: 10.1111/j.1364-3703.2012.00827.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nawrath C, Metraux JP. Salicylic acid induction-deficient mutants of Arabidopsis express PR-2 and PR-5 and accumulate high levels of camalexin after pathogen inoculation. Plant Cell. 1999;11:1393–1404. doi: 10.1105/tpc.11.8.1393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Miethke M, Marahiel MA. Siderophore-based iron acquisition and pathogen control. Microbiol. Mol. Biol. Rev. 2007;71:413–451. doi: 10.1128/MMBR.00012-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Alvarez-Fernandez A, Diaz-Benito P, Abadia A, Lopez-Millan AF, Abadia J. Metal species involved in long distance metal transport in plants. Front. Plant. Sci. 2014;5:105–124. doi: 10.3389/fpls.2014.00105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Fourcroy P, et al. Involvement of the ABCG37 transporter in secretion of scopoletin and derivatives by Arabidopsis roots in response to iron deficiency. New Phytol. 2014;201:155–167. doi: 10.1111/nph.12471. [DOI] [PubMed] [Google Scholar]
- 57.Fourcroy P, Tissot N, Gaymard F, Briat JF, Dubos C. Facilitated Fe nutrition by phenolic compounds excreted by the Arabidopsis ABCG37/PDR9 transporter requires the IRT1/FRO2 high-affinity root Fe2+ transport system. Mol. Plant. 2016;9:485–488. doi: 10.1016/j.molp.2015.09.010. [DOI] [PubMed] [Google Scholar]
- 58.De Coninck BM, et al. Arabidopsis thaliana plant defensin AtPDF1.1 is involved in the plant response to biotic stress. New Phytol. 2010;187:1075–1088. doi: 10.1111/j.1469-8137.2010.03326.x. [DOI] [PubMed] [Google Scholar]
- 59.Glazebrook J. Contrasting mechanisms of defence against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol. 2005;43:205–227. doi: 10.1146/annurev.phyto.43.040204.135923. [DOI] [PubMed] [Google Scholar]
- 60.Ko SS, et al. The bHLH142 transcription factor coordinates with TDR1 to modulate the expression of EAT1 and regulate pollen development in rice. Plant Cell. 2014;26:2486–2504. doi: 10.1105/tpc.114.126292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Li CW, et al. Tomato RAV transcription factor is a pivotal modulator involved in the AP2/EREBP-mediated defence pathway. Plant Physiol. 2011;156:213–227. doi: 10.1104/pp.111.174268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.King EO, Ward MK, Raney DE. Two simple media for the demonstration of pyocyanin and fluorescin. J. Lab. Clin. Med. 1954;44:301–307. [PubMed] [Google Scholar]
- 63.Yu, Q., Tang, C. & Kuo, J. A critical review on methods to measure apoplastic pH in plants. Plant Soil219, 29–40 (2000).
- 64.Patterson GH, Knobel SM, Sharif WD, Kain SR, Piston DW. Use of the green fluorescent protein and its mutants in quantitative fluorescence microscopy. Biophys. J. 1997;73:2782–2790. doi: 10.1016/S0006-3495(97)78307-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.May MJ, Leaver CJ. Oxidative stimulation of glutathione synthesis in Arabidopsis thaliana suspension cultures. Plant Physiol. 1993;103:621–627. doi: 10.1104/pp.103.2.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- 67.Soeters R, Aus C. Hazards of injectable therapy. Trop. Doct. 1989;19:124–126. doi: 10.1177/004947558901900310. [DOI] [PubMed] [Google Scholar]
- 68.Roschzttardtz H, et al. New insights into Fe localization in plant tissues. Front. Plant Sci. 2013;4:350. doi: 10.3389/fpls.2013.00350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lin HY, et al. Genome-wide annotation, expression profiling, and protein interaction studies of the core cell-cycle genes in Phalaenopsis aphrodite. Plant Mol. Biol. 2014;84:203–226. doi: 10.1007/s11103-013-0128-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yi Y, Guerinot ML. Genetic evidence that induction of root Fe (III) chelate reductase activity is necessary for iron uptake under iron deficiency. Plant J. 1996;10:835–844. doi: 10.1046/j.1365-313X.1996.10050835.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


