Abstract
The immune system plays a critical role in exerts effects in the growth and progression of hepatocellular carcinoma (HCC), which needs interacting approaches for effective therapy. In this study, we have found that the Norcantharidin (NCTD) + Coix lacryma-jobi seed oil (CLSO) combination exhibited more potent antitumor effects in an terms of cytotoxicity and apoptotic induction in human HepG2 and HepG2/ADM cells than NCTD or CLSO alone. In vivo, administration of NCTD+CLSO combinations significantly suppressed the formation of tumor in Hepal-1 hepatoma-bearing mice. Furthermore, we found that the in vitro co-cultures of HepG2 or HepG2/ADM cells with PBMCs from healthy donors led to an increase in the number of CD4 + CD25 + T cells. This increase was down-regulated by the combination effectively. Down-regulation of FoxP3 mRNA and protein expression occurred during the combination in the co-cultures. The amount of Tregs of Hepal-1 hepatoma-bearing mice was significantly decreased in the combination treated group. The combination down-regulated the expression of FoxP3, CTLA-4 and Tregs related cytokine (TGF-β and IL-10) in the serum of tumor bearing mice. Taken together, these results suggest that the most valuable aspect of the NCTD+CLSO combined use improves the anti-tumor activity and regulates tumor infiltrating Tregs.
Subject terms: Cancer microenvironment, Drug development
Introduction
Hepatocellular carcinoma (HCC) is the fifth most frequently diagnosed cancer worldwide1. Due to the dietary habits and public hygiene, higher incidence, poor prognosis and rapid progression of HCC is reported in East Asia and sub-Saharan Africa, especially in China2, 3. Chemotherapy remains as the curative option of choice, but the efficacy of remains limited by chronic hepatitis infection and inflammation4. Simultaneously, the severe systemic side effects resulting from nonselective biodistribution still enormously restrict the clinical applications.
It has been found that CD4 + CD25 + regulatory T cells (Tregs), myeloid derived suppressor cells, and various immunosuppressive factors, including interleukin 10 (IL-10), transforming growth factor β (TGF-β), vascular endothelial growth factor, and prostaglandin E2, are frequently enriched in the tumor microenvironment and facilitate tumor immune evasion5. Antagonizing immunosuppressive mechanisms in the tumor microenvironment are a prerequisite for the translation of antitumor immune responses into therapeutic benefits6. Tregs have been shown to reduce the host antitumor responses and presence of Tregs in both the peripheral blood and tumor microenvironment has been associated with a poor patient prognosis in HCC patients7, 8. Therefore, targeting the number and function of Tregs may be a useful and effective approach for HCC therapies.
Norcantharidin (NCTD) and Coix lacryma-jobi seed oil (CLSO) have already been developed for antitumor clinical applications9–11. NCTD is derived from the dried body of blister beetle (Mylabris phalerata Pallas). CLSO is obtained from the seed of Coix lacryma-jobi. NCTD and CLSO are used to treat primary malignant tumors, including lung cancer, liver cancer, gastric cancer, and breast cancer, because of their anti-proliferation and pro-apoptotic effects on numerous tumor cell lines in vitro and tumor models in vivo 12–18.
Being an efficacious formula in treatment of HCC patients, the combined prescription of NCTD and CLSO were investigated in this study. The most valuable aspect of the combined use improves the anti-tumor activity and regulates tumor infiltrating Tregs. All the results obtained strongly supported the new combination of NCTD and CLSO shows the better therapeutic effect.
Results
Combination of NCTD and CLSO significantly inhibits cell viability in HepG2 and HepG2/ADM cells
To address the role of NCTD, CLSO and their combination on the cell viability. HCC cell lines, including HepG2 and HepG2/ADM, were treated with different concentrations of NCTD (0–12 μg/mL) or CLSO (0–160 μg/mL). As depicted in Fig. 1, NCTD or CLSO decreased cell viability both in a dose- and time - dependent manner significantly in HepG2 and HepG2/ADM cells with an IC50 value of 6.71 (HepG2), 99.39 (HepG2/ADM) μg/mL of NCTD (Fig. 1a) alone or an IC50 value of 8.85 (HepG2), 148.77 (HepG2/ADM) μg/mL of CLSO (Fig. 1b) alone for 24 h. Next, we investigated whether CLSO enhanced sensitivity of HepG2 and HepG2/ADM cells to NCTD treatment. The CDI was utilized to display the effects of interaction between these two drugs. Our results showed that combination with CLSO significantly enhanced NCTD efficacy with higher IC50 value of 3.75/ 49.98 (HepG2), 4.48/59.77 (HepG2/ADM) μg/mL (Fig. 1c) and exhibited synergistic effects for HepG2 and HepG2/ADM cells (Table 1). In addition, the bare microemulsions of CLSO showed no cytotoxicity against HepG2 or HepG2/ADM cells under the same conditions (Fig. 1d).
Figure 1.
Combination of NCTD and CLSO significantly inhibits cell viability in HepG2 and HepG2/ADM cells. (a) The percentage of viable cells as measured by the MTT assay at 24 and 48 h relative to no-drug controls and NCTD concentrations were plotted as a dose response curve (n = 6 per group). (b) Cells treated with the CLSO(n = 6 per group). (c) Cells treated with the combination of NCTD and CLSO. Cell viability was assayed (n = 6 per group). (d) Cytotoxicity of bare microemulsion formulation (containing Span-80 1 g and Sween-80 1.5 g) against HepG2 and HepG2/ADM cells. *P < 0.05; **P < 0.01, vs. control, aP < 0.05; aaP < 0.01 vs. NCTD alone, bP < 0.05; bbP < 0.01 vs. CLSO alone. One-way ANOVA, post hoc comparisons, Tukey’s test. Columns, means; error bars, SDs.
Table 1.
CDI of the combination of NCTD and CLSO in HepG2 and HepG2/ADM cells.
| Concentrations(μg/mL) | HepG2 | HepG2/ADM | |
|---|---|---|---|
| NCTD | CLSO | ||
| 1.5 | 20 | 0.982 | 0.790 |
| 3 | 40 | 0.979 | 0.767 |
| 6 | 80 | 0.891 | 0.810 |
| 12 | 160 | 0.770 | 0.602 |
CDI, coefficient of drug interaction.
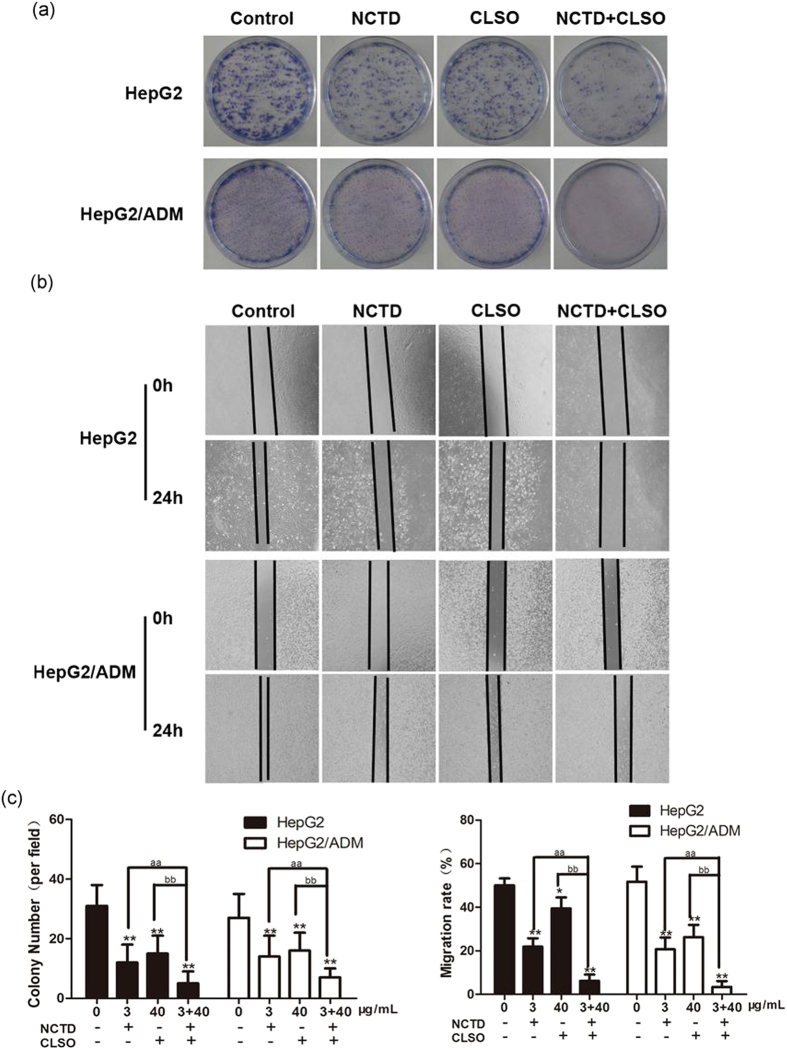
Combination of NCTD and CLSO inhibits colony-formation and migration in HepG2 and HepG2/ADM cells
We further investigated the combined effect of NCTD and CLSO on colony-formation ability and migratory potential. Our results showed that the colony-formation ability of HepG2 and HepG2/ADM cells could be affected by exposure to NCTD, CLSO alone or the combination (Fig. 2a and c). As determined by scratch motility assay, NCTD or CLSO treatment alone induced a partial inhibition of migration whereas the combined treatment notably inhibited the migratory potential of HepG2 and HepG2/ADM cells (Fig. 2b and c).
Figure 2.
Combination of NCTD and CLSO inhibits colony-formation and migration in HepG2 and HepG2/ADM cells. (a) Representative images were captured from HepG2 and HepG2/ADM cells incubated with NCTD (3 μg/mL) and CLSO (40 μg/mL)) either alone or in combination for 24 h and subjected to cell colony-formation assays. (b) Effects of NCTD (3 μg/mL), CLSO (40 μg/mL) or combined NCTD and CLSO treatment on the migratory potential of HepG2 and HepG2/ADM cells were analyzed by a scratch assay. Migration was analyzed after 24 h incubation and photographed (original magnification, ×10). (c) The above assays were quantified. Data represent three independent experiments. *P < 0.05; **P < 0.01, vs. control, aP < 0.05; aaP < 0.01 vs. NCTD alone, bP < 0.05; bbP < 0.01 vs. CLSO alone. One-way ANOVA, post hoc comparisons, Tukey’s test. Columns, means; error bars, SDs.
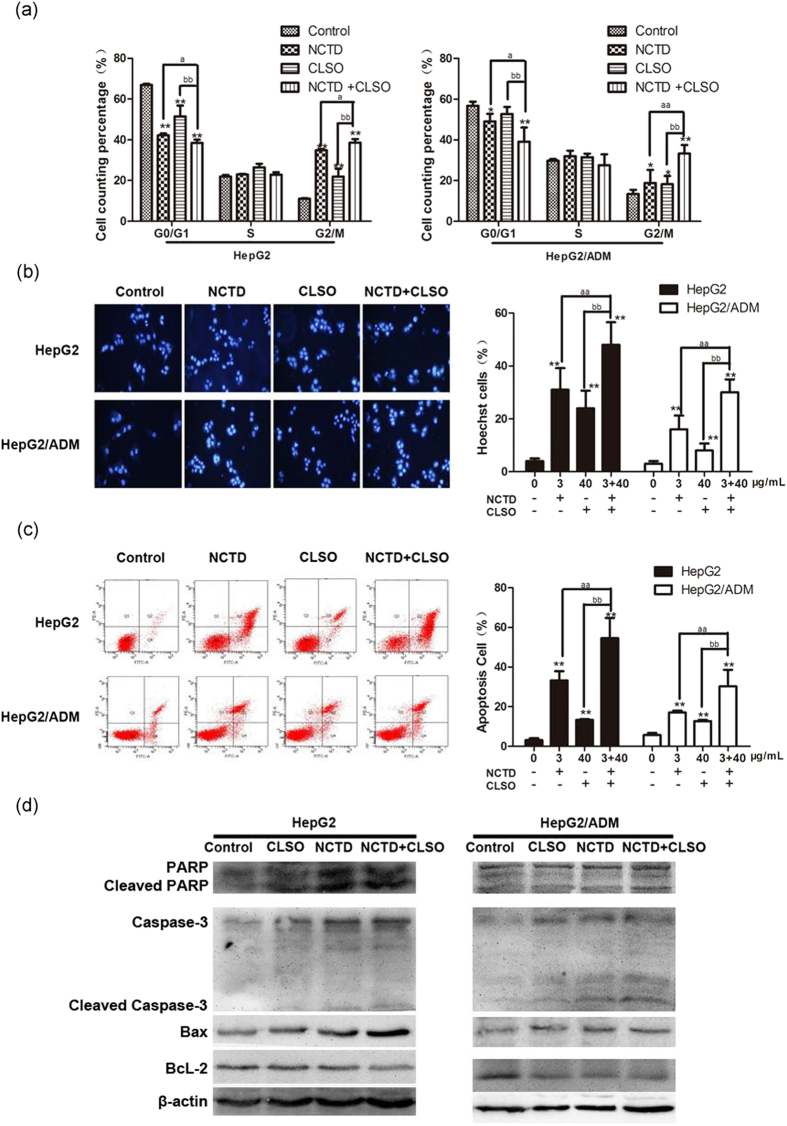
Combination of NCTD and CLSO induces cell cycle arrest and apoptosis in HepG2 and HepG2/ADM cells
Given the superior synergistic interactions observed between NCTD and CLSO, we investigated the potential effects on cell apoptosis mediated by these combinations. Our results showed that treatment of HepG2 and HepG2/ADM cells with NCTD, CLSO alone or combination for 24 h significantly increased cells in G2/M phase of cell cycle (Fig. 3a). In addition, the NCTD+CLSO combination led to enhanced accumulation of cells in the G2/M phase compared to the single agents. Hoechst 33342 staining (Fig. 3b) and Annexin V/Propidium Iodide (PI) double staining (Fig. 3c) showed that most of the cell death induced by NCTD, CLSO alone or combination can be classified as apoptosis both in HepG2 and HepG2/ADM. We also observed the NCTD+CLSO combination significantly increased apoptotic cell death compared with NCTD or CLSO as a single agent. These data revealed an additive mechanism of the NCTD+CLSO combination inducing cell death via apoptosis.
Figure 3.
Combination of NCTD and CLSO induces cell cycle arrest and apoptosis in HepG2 and HepG2/ADM cells. (a) Cell cycle distribution of HepG2 and HepG2/ADM cells was determined 24 h after treatment with NCTD and CLSO alone or in combination(n = 3). (b) Cells were stained with Hoechst33342 (5 μg/ml) and subjected to analysis of apoptosis population(n = 3). (c) PE-Annexin V staining of phosphatidylserine exposed on the cell surface was measured by flow cytometric analysis (n = 3). Data derived from three separate experiments are presented as the means ± S.D. (d) Total cell lysates were prepared for western blot analysis of the apoptosis regulatory proteins (n = 3). *P < 0.05; **P < 0.01, vs. control, aP < 0.05; aaP < 0.01 vs. NCTD alone, bP < 0.05; bbP < 0.01 vs. CLSO alone. One-way ANOVA, post hoc comparisons, Tukey’s test. Columns, means; error bars, SDs.
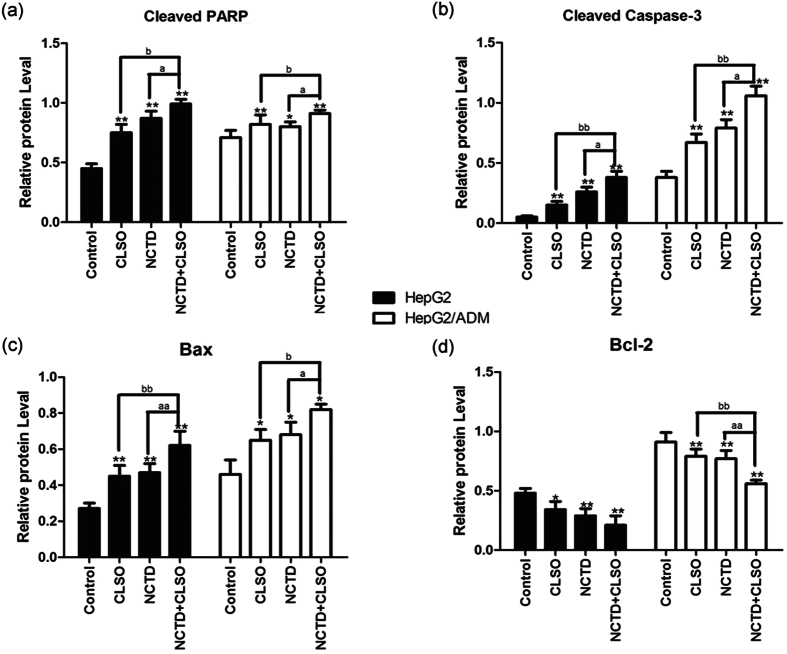
Activation of caspases is a biochemical feature of apoptosis. Immunoblotting assessment (Fig. 3d and 4) showed that the cleavage of poly (ADP) ribose polymerase (PARP) and caspase-3 was also increased in HepG2 and HepG2/ADM cells which were treated with the NCTD+CLSO combination compared to the single agents, in agreement with cell death assays.
Figure 4.
Western blotting results of PARP, Caspase-3, Bax and Bcl-2 in HepG2 and HepG2/ADM cells. The densitometric analysis bar diagram of the results. Columns represent the mean from three independent experiments and bars represent standard deviations. (a) Cleaved PARP. (b) Cleaved Caspase-3. (c) Bax. (d) Bcl-2.
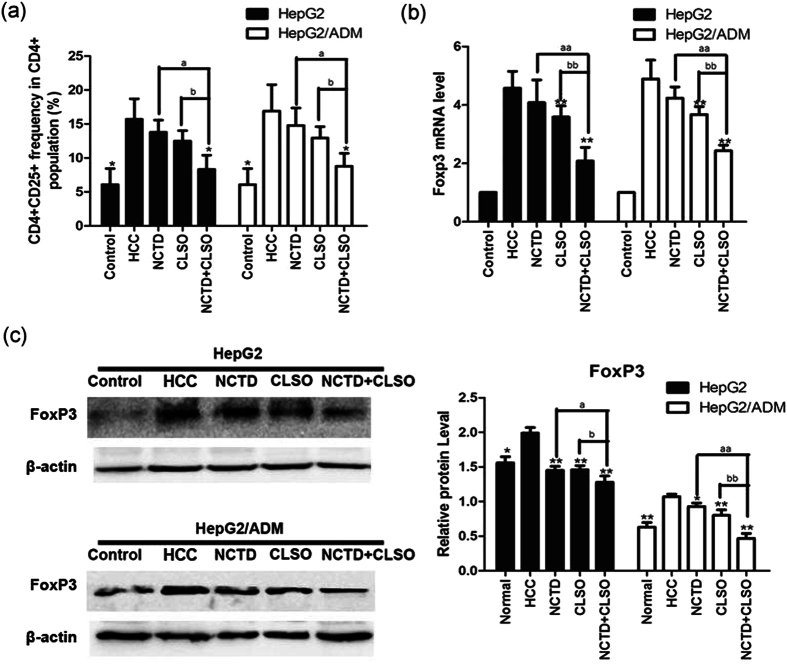
Combination of NCTD and CLSO downregulates CD4+CD25+FoxP3+ Tregs in HepG2 and HepG2/ADM cells
Cultures of hepatoma cell Lines with transwelled PBMC to evaluate further the mechanism of how NCTD, CLSO alone or combination may regulate Tregs in HCC, we established an in vitro culture system with HepG2, HepG2/ADM cells, normal hepatocytes, and PBMCs from healthy controls, using a transwell chamber to limit their interaction. To further investigate whether the combination of NCTD and CLSO regulates CD4, CD25, and FoxP3 Tregs in HCC, flow cytometric analysis of CD4 + CD25 + cells, RT-PCR, and western blotting of FoxP3 were examined. NCTD, CLSO alone or combination significantly decreased the CD4 + CD25 + T cells frequency compared with the series of PBMC co-cultures in the presence of HepG2 or HepG2/ADM cells after a 48 h culture period (Fig. 5a). RT-PCR analysis showed that NCTD + CLSO combination significantly decreased expression of FoxP3 in HCC compared with NCTD or CLSO as a single agent (Fig. 5b). NCTD+CLSO combination significantly decreased the expression of FoxP3 in HepG2 and HepG2/ADM cells (Fig. 5c). These results further confirmed that NCTD+CLSO combination suppressed the accumulation of the tumor infiltrating Tregs.
Figure 5.
Combination of NCTD and CLSO downregulates CD4 + CD25 + FoxP3 + Tregs in HepG2 and HepG2/ADM cells. (a) Mean CD4+ CD25+ frequency in CD4+ population from PBMCs before and after co-culture with HCC. (b) Gene expression analysis of FoxP3 of PBMC co-cultures in the presence of HepG2 or HepG2/ADM cells by Quantitative Real-time PCR. The relative quantification value, fold difference, is expressed as 2−ΔΔCt. (c) The expression levels of FoxP3 were detected by Western blot analysis. Results represent mean values of three experiments ± SD. *P < 0.05; **P < 0.01, vs. HCC, aP < 0.05; aaP < 0.01 vs. NCTD alone, bP < 0.05; bbP < 0.01 vs. CLSO alone.
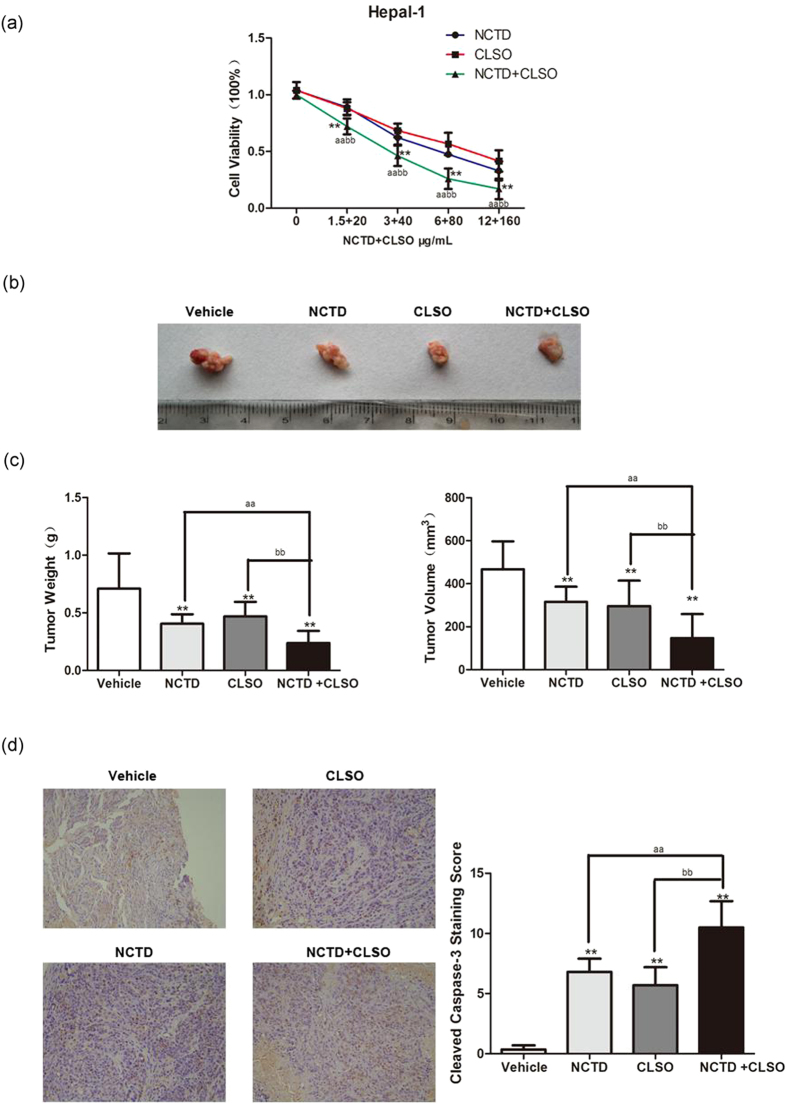
Combination of NCTD with CLSO arrests tumor growth in vivo
As shown in Fig. 6a, the NCTD+CLSO combination significantly increased Hepal-1 cell death compared with NCTD or CLSO as a single agent in vitro. To evaluate the anti-HCC activity of the NCTD+CLSO combination in vivo, the hepatoma-bearing mice model was established. After administration of NCTD, CLSO alone or combination in hepatoma-bearing mice by i.p., the NCTD+CLSO combination treatment exerted marked antitumor activity in Hepal-1 tumors compared to the single agents (Fig. 6b). The NCTD+CLSO combination treatment significantly decreased the weight and volume of tumors (Fig. 6c). Furthermore, we used immunohistochemistry to analyze the hepatoma-bearing mice model. The results suggested that the NCTD+CLSO combination treatment notably activated the cleaved caspase-3, a biochemical feature of apoptosis, in Hepal-1 cell-derived tumors (Fig. 6d). In conclusion, our data revealed that the NCTD+CLSO combination treatment significantly arrests tumor growth in vivo via the apoptosis regulatory mechanisms.
Figure 6.
Combination of NCTD with CLSO arrests tumor growth in vivo. (a) Hepal-1 cells treated with the combination of NCTD and CLSO. Cell viability was assayed (n = 6 per group). (b) Microscopic view of HCC tumor tissue in mice. (c) Isolated tumor volume and Tumor weight from the Hepal-1 mouse HCC model. (d) Cleaved caspase-3 in tumor tissue lysates from vehicle and NCTD, CLSO alone or combination-treated mice were detected by immunohistochemistry analysis. *P < 0.05; **P < 0.01, vs. Vehicle, aP < 0.05; aaP < 0.01 vs. NCTD alone, bP < 0.05; bbP < 0.01 vs. CLSO alone. One-way ANOVA, post hoc comparisons, Tukey’s test. Columns, means; error bars, SDs.
Combination of NCTD with CLSO downregulates peripheral blood CD4+CD25+FoxP3+ Tregs in mice
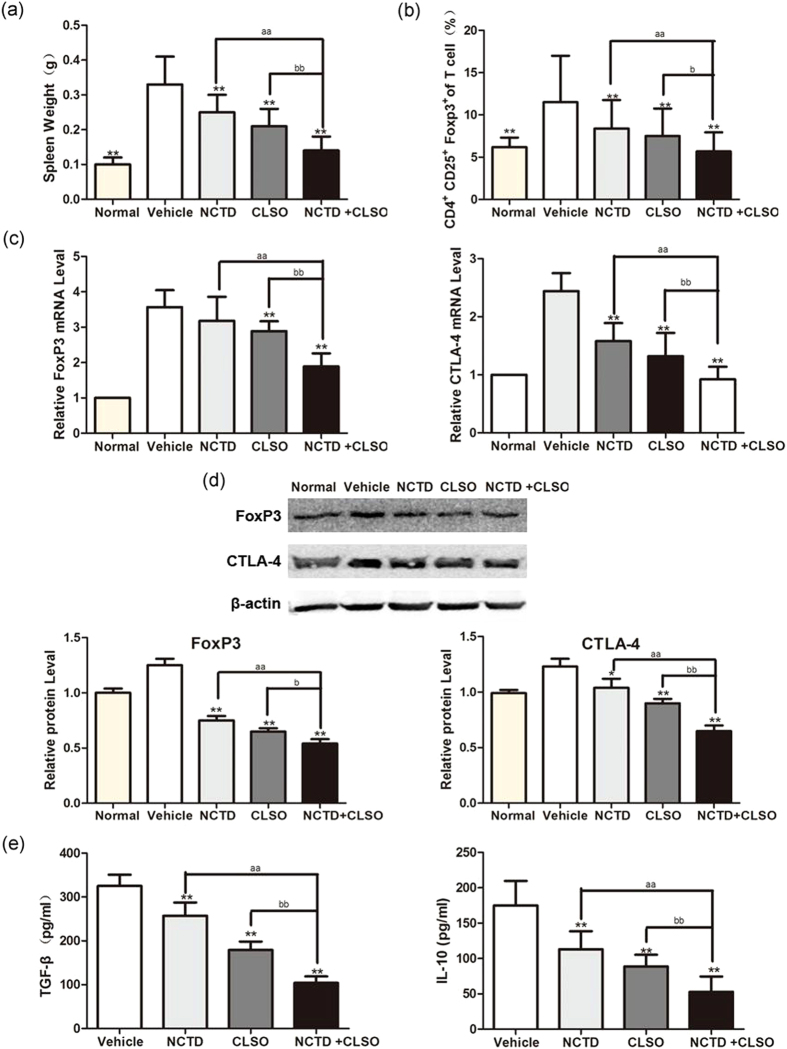
Apart from developing a large number of intestinal polyps, hepatoma-bearing mice also developed a massive splenomegaly. The NCTD+CLSO combination treatment decreased the weight of the spleen in Hepal-1 mice compared with NCTD or CLSO as a single agent (Fig. 7a). We next examined the effect of NCTD, CLSO alone or combination on Tregs in the HCC mice by evaluating the percentage of CD4 + CD25 + FoxP3 + cells in the peripheral blood. The NCTD+CLSO combination significantly decreased the ratio of Tregs compared with NCTD or CLSO as a single agent (Fig. 7b).
Figure 7.
Combination of NCTD with CLSO downregulates peripheral blood CD4 + CD25 + FoxP3 + Tregs in mice. (a) Isolated spleen weight from the Hepal-1 hepatoma-bearing model. (b) The peripheral blood cells were harvested, and the percentage of CD4 + CD25 + FoxP3 + cells was determined using flow cytometry. The data from the peripheral blood of mice are shown. (c) Gene expression analysis of FoxP3 and CTLA-4 of the peripheral blood of Hepal-1 mice by Quantitative Real-time PCR. The relative quantification value, fold difference, is expressed as 2−ΔΔCt. (d) The expression levels of FoxP3 in the peripheral blood samples were detected by Western blot analysis. Results represent mean values of three experiments ± SD. (e) The concentrations of TGF-β and IL-10 in the serum of mice were measured by ELISA. Data are representative of three independent experiments.*P < 0.05; **P < 0.01, vs. Vehicle, aP < 0.05; aaP < 0.01 vs. NCTD alone, bP < 0.05; bbP < 0.01 vs. CLSO alone. One-way ANOVA, post hoc comparisons, Tukey’s test. Columns, means; error bars, SDs.
Naturally occurring FoxP3+ regulatory T cells (Tregs) constitutively express CTLA-4. Antibody blockade of CTLA-4 abrogates Tregs activity in some in vitro and in vivo settings. To further investigate whether the NCTD+CLSO combination regulates CTLA-4 and FoxP3 Tregs in the peripheral blood of Hepal-1 hepatoma-bearing mice, immunohistochemical and western blotting of FoxP3 and CTLA-4 were examined. RT-PCR analysis showed the NCTD+CLSO combination significantly decreased expression of FoxP3 and CTLA-4 in the peripheral blood of Hepal-1 mice compared with that of vehicle mice, and better than NCTD or CLSO as a single agent (Fig. 7c). The NCTD+CLSO combination significantly decreased the expression of FoxP3 in hepatoma-bearing mice (Fig. 7d). As shown in Fig. 7e, NCTD+CLSO combination treatment significantly down-regulated Tregs related cytokine TGF-β and IL-10 in the serum of tumor bearing mice compared with NCTD or CLSO as a single agent. These results further confirmed that Tregs play an essential role in the carcinogenesis of hepatoma-bearing mice and the NCTD+CLSO combination reduced the formation of tumor through regulating the percentage of Tregs in hepatoma-bearing mice.
Discussion
Hepatocellular carcinoma (HCC), a hypervascular tumor type with characteristic of high levels of neovascularization and angiogenesis19. The immune system plays a critical role inexerts effects in the growth and progression which needs interacting approaches for effective therapy20–22. Due to the association of single agents with treatment resistance, we considered that the combination therapy increased the lethality in HCC. Norcantharidin (NCTD) and Coix lacryma-jobi seed oil (CLSO) has been demonstrated to exert antitumor effects in various types of tumors including HCC. Thus, in the present study, we investigated whether the combinations of NCTD+CLSO led to more superior antitumor effects than NCTD or CLSO alone in HCC cells. This study demonstrated that either NCTD or CLSO alone reduce cell viability, metastasis, and colony formation and induce cell cycle arrest and apoptosis. However, the NCTD+CLSO combination exhibited more potent antitumor effects in terms of cytotoxicity and apoptotic induction in human HepG2 and HepG2/ADM cells. In vivo, administration of NCTD+CLSO combinations significantly suppressed the formation of tumor in Hepal-1 hepatoma-bearing mice. NCTD+CLSO combinations not only reduced the incidence of HCC, but also up-regulated cleaved caspase-3. The combinations might be a potent alternative regimen for HCC.
Regulatory T cells characterized by the expression of the transcription factor FoxP3 play a pivotal role in immune homeostasis and suppress function of effector cells such as CD4+ T cells, CD8+ T cells, and natural killer (NK) T cells23. Among stromal cells infiltrating tumors, Tregs represented one of the most important players mediating immunosuppression. So, radically resected patients showed significantly reduced Tregs frequencies24, 25. Previous study has demonstrated that an abundant accumulation of Treg cells was found in tumor regions compared with nontumor regions in HCC patients26. We then used an in vitro HCC transwell model to understand the underlying mechanisms responsible for the upregulation of Tregs observed. The co-cultures of HepG2 or HepG2/ADM cells with PBMCs from healthy donors in vitro led to an increase in the number of CD4 + CD25 + T cells, suggesting that cancer-derived soluble factors result in the expansion of Tregs. This increase was down-regulated by the NCTD+CLSO combination effectively. However, a recent study describes the existence of CD45RA+ Tregs in the periphery of humans that express high levels of FoxP3 and manifests equivalent suppressive activity in vitro when compared to their CD45RO+ Tregs counterparts27. FoxP3+ Tregs may contribute to tumoral immune escape and disease progression28. It has been reported that increased tumor-related expression of the transcription factor FoxP3 is associated with adverse prognosis29. The expression of FoxP3 was positively correlated with Dukes staging and lymph node metastasis30. Down-regulation of FoxP3 mRNA and protein expression occurred during the NCTD+CLSO combination in the co-cultures of PBMCs with HepG2 or HepG2/ADM cells supporting that the decreased CD4 + CD25 + T cells in the cultures represent Tregs. Our study also found that the amount of CD4 + CD25 + FoxP3 + regulatory T cells in tumor tissue of Hepal-1 hepatoma-bearing mice was significantly decreased in the NCTD+CLSO combination treated group. The NCTD+CLSO combination reduced the expression of FoxP3 which in turn decreased the carcinogenesis in the HCC, and down-regulated the expression of CTLA-4 and Treg related cytokine (TGF-β and IL-10) in the serum of tumor bearing mice31–33. This result confirmed the view that the NCTD+CLSO combination can inhibit tumor cells to recruit Tregs to inhibit the efficiency of cancer immunotherapy.
In conclusion, these data demonstrated that the NCTD+CLSO combination treatment notably reduced cell viability and induced apoptosis in human HepG2 and HepG2/ADM cells, and effectively suppressed tumor growth in hepatoma-bearing mice. The decrease of Tregs accumulation and inactivation was associated with the NCTD+CLSO combination administration. Overall, these findings have revealed the molecular mechanisms of the NCTD+CLSO combination treatment with HCC, which provide a basis for and warrant future study to investigate the combination therapy for the treatment of drug resistant tumors with targeted therapy. Further studies will be conducted to investigate how the NCTD+CLSO combination regulates activation and recruitment of Tregs and their underlying mechanism in regulating carcinogenesis.
Materials and Methods
Cell culture, animals and Reagents
Human Human hepatocellular cancer (HCC) cells (HepG2 and HepG2/ADM) and the mouse hepatoma Hepal-1 cell line were obtained from American Type Culture Collection (ATCC, Rockville, MD). All cell lines were incubated in RPMI-1640 (Invitrogen) and DMEM (Invitrogen), supplemented with 10% (v/v) fetal bovine serum (Invitrogen) and 1% (v/v) penicillin–streptomycin (Invitrogen) at 37 °C in a humidified atmosphere of 5% CO2. C57BL/6 male mice (5–6 weeks old, 18–22 g) were purchased from the Animal Supplier Center of Binzhou Medical University (No. 37009200006098). All procedures involving laboratory animals were in accordance with the guidelines of the Instituted Animal Care and Use Committee of Binzhou Medical University. All protocols were submitted and validated by the Animal Care Ethics Committee of Binzhou Medical University.
The NCTD was purchased from Sigma-Aldrich (St. Louis, MO, USA), dissolved in dimethylsulfoxide (DMSO; Sigma-Aldrich, St. Louis, MO, USA) at 50 mM stock and diluted immediately before each experiment. CLSO was obtained from Guangzhou Masson Pharmaceutical Ltd. (Guangzhou, China) and prepared using supercritical CO2 extraction technology, with the O/W microemulsion formulation (10 g:100 ml) mainly containing CLSO as oil phase, Span-80 and Sween-80.
Measurement of cell viability
Cell viability assays were performed using the Cell Counting Kit-8 (CCK-8; Dojindo Molecular Technologies, Japan). Cells were seeded with culture medium onto 96-well plates and incubated at 37 °C or 24 h. After adaptation, cells were treated with either NCTD or CLSO, or in combination for 24 h and 48 h. Then the culture medium was replaced with fresh medium containing 10 ml of CCK-8 solution. The optical density (OD) at 490 nm was assayed following cell incubation at 37 °C for 2 h.
The coefficient of drug interaction (CDI) analysis for evaluating effects of drug combinations was calculated according to Cao et al.34 using the equation: CDI = AB / (A × B). A or B is the ratio of the single agent group to the control group and AB is the ratio of the combination groups to the control group. A CDI of ≤ or >1 indicates synergy, additivity or antagonism, respectively. A CDI < 0.7 shows that the drugs are significantly synergistic.
Colony formation assay
Cells were plated in 6 cm dishes and incubated in RPMI 1640 or DMEM with 10% FBS at 37 °C. Two weeks later, the cells were fixed and stained with 0.1% crystal violet. The number of visible colonies was counted manually.
Cell scratch assay
A marker was used to draw lines across the back of culture plates. Cells (5 × 105) were added to each plate of the corresponding experimental groups. Sterile pipette tips (200 µl) were used to make a scratch in the cell monolayer across the bottom of the well. Cells were then allowed to grow in RPMI 1640 or DMEM containing 5% calf serum. The cell migration in the wounded area was observed.
Cell cycle analysis and Apoptosis assay by flow cytometry
Cells were treated with NCTD or CLSO or in combination for 24 h and then the cells were performed by cell cycle analysis. In brief, 5 × 104 cells were suspended in 0.5 ml of PI solution, and incubated 30 min in the dark according to the manufacturer’s instructions. Cell cycle distribution was analyzed by FACS flow cytometry.
Cells were stained with Annexin V-FITC apoptosis detection kit (BD Biosciences, San Jose, CA, USA). According to the manufacturer’s instructions, the cells were incubated with 5 ml of Annexin V and 5 ml of propidium iodide (PI) for 15 min at room temperature, and then the stained cells were analyzed on a FACS flow cytometer.
Flow cytometric analysis of Tregs
Peripheral blood cells were stained with a FITC-conjugated anti-mouse CD4 mAb (BD, New jersey, Franklin, USA), anti-mouse CD25 mAb (BD, New jersey, Franklin, USA), and a PE-Cy5-conjugated anti-mouse/rat FoxP3 mAb (BD, New jersey, USA), and the samples were fixed with 1% paraformaldehyde/PBS and analyzed by using a FACSCalibur flow cytometer and CellQuest software (Becton Dickinson Japan, Tokyo, Japan).
Gene and protein expression analysis
Gene expression was analyed by Quantitative Real-time PCR. Total RNA was extracted with TRIZOL reagent (Invitrogen, California, Carlsbad, USA) according to the manufacturer’s instructions. The first-strand cDNA synthesis was performed with cDNA synthesis kit (TaKaRa, Dalian, China). Quantitative real-time PCR was performed using the SYBR Green real-time PCR kit (TaKaRa, Dalian, China). The fold changes were calculated by the delta–delta Ct method. All experiments were performed in three biological replicates. Primers for CTLA-4 and FoxP3 were constructed as described previously.17
Protein expression was analyed by western blot. Total protein (50 μg) was separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. After protein transfer to polyvinylidene fluoride microporous membranes Bio-Rad), the membranes were blocked with 5% nonfat dry milk and incubated sequentially with the primary antibodies [anti-PARP, anti-Caspase-3, anti-Bax, or anti- Bcl-2, anti-FoxP3 (FoxP3 of mouse and human, 1:500 dilution, Abcom, England), anti- CTLA-4 (CTLA-4 of mouse and human, 1:500 dilution, Santa Cruz, USA)], followed by incubation with the fluorescein-linked anti-mouse (anti-rabbit) IgG (1:1000) and then incubation with anti-fluorescein alkaline phosphatase-conjugated antibody (1:5000). The immune complexes were detected with the enhanced chemiluminescence reagent. For quantification, signals were densitometrically normalized to β-actin by Quantity One image analysis software.
Measurement of in vivo activity
Under sterile conditions, Hepal-1 cells (5 × 106 cells/animal) were subcutaneously inoculated into the C57BL/6 mice. The inoculated mice were randomly divided into five groups, with six mice in each group; the body weight difference between groups was not significant. In the normal and vehicle group, an intraperitoneal injection of saline (10 ml/kg) was performed every other day. In group 3 and 4, an intraperitoneal injection of NCTD (2 mg/kg) or CLSO (30 mg/kg) was administered every other day, respectively. In experimental group 5 (the combination group), NCTD (2 mg/kg) and CLSO (30 mg/kg) were administered by injection every other day. The mice were sacrificed five weeks after tumor implantation. The weights and tumor volumes of the nude mice were recorded every second day until the animals were sacrificed.
Immunohistochemistry
Paraffin-embedded colon sections were dewaxed, rehydrated, and pre-treated with hydrogen peroxidase in PBS buffer. Heat-induced antigen retrieval was performed. Sections were incubated with anti-β-catenin (clone 6B3, 1:100 dilution, Cell Signaling Technology (CST), Boston, USA) and anti-cleaved caspase-3 (1:100 dilution, CST, Boston, USA). After incubation with HRP-conjugated secondary antibody and tyramide amplification followed by streptavidin-HRP, positive signals were visualized by DAB kit. Section were examined at a magnification of 400× and analyzed using NIS-Elements. The positive content was calculated using the following formula: positive content (PC) = mean optical density × positive area.
Measurement of TGF-β and IL-10 in the serum of mice by ELISA
The mice of control and treatment groups were sacrificed, and the serum of mice were collected. The concentrations of TGF-β and IL-10 in the serum were measured by ELISA using ELISA kits (Ebiosciences-Easy-Set-Go) according to the manufacturer’s protocols.
Statistical Analysis
Each experiment was repeated at least three times. Data were presented as mean ± SD. All data were analyzed using the SPSS statistical package (version 16.0, SPSS Inc, Chicago, IL, USA). Data between two groups were compared with 2-independent samples tests. Mean values of data from more than 3 groups were compared with one-way analysis of variance (ANOVA) and multi-comparison was performed. A value of P < 0.05 was considered as statistically significant.
Data availability
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.
Acknowledgements
We thank Dr. Zhang of LUYE PHARMA Group Ltd. for HepG2 and HepG2/ADM cell lines. This research was supported by Shandong Provincial Natural Science Foundation (No. 2016ZRB14353, No. 2014ZRA06063), the Scientific Research Foundation of Binzhou Medical University (BY2015KYQD02) and the Dominant Disciplines’ Talent Team Developmen Scheme of Higher Education of Shandong Province.
Author Contributions
D.W., C.Y., Z.W., Y.Y., D.L., X.D., W.X., and Q.Z. designed the study. D.W., C.Y., Z.W., and Y.Y. performed experiments, prepared figures. D.W., C.Y., Z.W., Y.Y., D.L., X.D., W.X., and Q.Z. analysed the data. D.W., C.Y., W.X., and Q.Z. wrote and discussed all sections of the manuscript. All authors reviewed the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
This article has been retracted. Please see the retraction notice for more detail: 10.1038/s41598-024-57766-9
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Dan Wang and Chendong Yang contributed equally to this work.
Change history
3/26/2024
This article has been retracted. Please see the Retraction Notice for more detail: 10.1038/s41598-024-57766-9
Contributor Information
Wenjuan Xu, Email: xwjycd@126.com.
Qiusheng Zheng, Email: zqsyt@sohu.com.
References
- 1.Torre LA, et al. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Ma L, Chua MS, Andrisani O, So S. Epigenetics in hepatocellular carcinoma: an update and future therapy perspectives. World J Gastroenterol. 2014;20:333–345. doi: 10.3748/wjg.v20.i2.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ruan J, et al. Overexpression of cathepsin B in hepatocellular carcinomas predicts poor prognosis of HCC patients. Mol Cancer. 2016;15:17. doi: 10.1186/s12943-016-0503-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lu X, et al. Alkylating Agent Melphalan Augments the Efficacy of Adoptive Immunotherapy Using Tumor-Specific CD4+ T Cells. J Immunol. 2015;194:2011–2021. doi: 10.4049/jimmunol.1401894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–1022. doi: 10.1002/hep.24199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zou W. Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat Rev Cancer. 2005;5:263–274. doi: 10.1038/nrc1586. [DOI] [PubMed] [Google Scholar]
- 7.Chen X, Du Y, Huang Z. CD4+ CD25+ Treg derive from hepatocellular carcinoma mice inhibits tumor immunity. Immunol Lett. 2012;148:83–89. doi: 10.1016/j.imlet.2012.09.002. [DOI] [PubMed] [Google Scholar]
- 8.Guo CL, et al. Associations between infiltrating lymphocyte subsets and hepatocellular carcinoma. Asian Pac J Cancer Prev. 2012;13:5909–5913. doi: 10.7314/APJCP.2012.13.11.5909. [DOI] [PubMed] [Google Scholar]
- 9.Chen YJ, et al. Norcantharidin induces anoikis through Jun-N-terminal kinase activation in CT26 colorectal cancer cells. Anticancer Drugs. 2008;19:55–64. doi: 10.1097/CAD.0b013e3282f18826. [DOI] [PubMed] [Google Scholar]
- 10.Chen YN, et al. Effector mechanisms of norcantharidin-induced mitotic arrest and apoptosis in human hepatoma cells. Int J Cancer. 2002;100:158–165. doi: 10.1002/ijc.10479. [DOI] [PubMed] [Google Scholar]
- 11.Ye L, Yan L, Xing T, Fu D. Formulation, Preparation and Evaluation of an Intravenous Emulsion Containing Brucea Javanica Oil and Coix Seed Oil for Anti-tumor Application.Biol Pharm Bull. 2008;31:673–680. doi: 10.1248/bpb.31.673. [DOI] [PubMed] [Google Scholar]
- 12.Yang PY, et al. Norcantharidin induces apoptosis of breast cancer cells: involvement of activities of mitogen activated protein kinases and signal transducers and activators of transcription. Toxicol In Vitro. 2011;25:699–707. doi: 10.1016/j.tiv.2011.01.011. [DOI] [PubMed] [Google Scholar]
- 13.Yeh CB, et al. Antimetastatic effects of norcantharidin on hepatocellular carcinoma by transcriptional inhibition of MMP-9 through modulation of NF-kB activity. PLoS One. 2012;7:e31055. doi: 10.1371/journal.pone.0031055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zheng J, et al. Norcantharidin induces growth inhibition and apoptosis of glioma cells by blocking the Raf/MEK/ERK pathway. World J Surg Oncol. 2014;12:207. doi: 10.1186/1477-7819-12-207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kok SH, et al. Norcantharidin-induced apoptosis in oral cancer cells is associated with an increase of proapoptotic to antiapoptotic protein ratio. Cancer Letter. 2005;217:43–52. doi: 10.1016/j.canlet.2004.07.045. [DOI] [PubMed] [Google Scholar]
- 16.Ding Q, et al. Octanoyl galactose ester-modified microemulsion system self-assembled by coix seed components to enhance tumor targeting and hepatoma therapy. International Journal of Nanomedicine. 2017;12:2045–2059. doi: 10.2147/IJN.S125293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ding Q, et al. Triterpene-loaded microemulsion using Coix lacryma-jobi seed extract as oil phase for enhanced antitumor efficacy: preparation and in vivo evaluation. International Journal of Nanomedicine. 2014;9:109–119. doi: 10.2147/IJN.S54796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yijia W, et al. Kanglaite sensitizes colorectal cancer cells to Taxol via NF-κB inhibition and connexin 43 upregulation. Sci rep. 2017;7:1280–1289. doi: 10.1038/s41598-017-01480-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Geis T, et al. HIF-2alpha-dependent PAI-1 induction contributes to angiogenesis in hepatocellular carcinoma. Exp Cell Res. 2015;331:46–57. doi: 10.1016/j.yexcr.2014.11.018. [DOI] [PubMed] [Google Scholar]
- 20.Ormandy LA, et al. Increased populations of regulatory T cells in peripheral blood of patients with hepatocellular carcinoma. Cancer Res. 2005;65:2457–2464. doi: 10.1158/0008-5472.CAN-04-3232. [DOI] [PubMed] [Google Scholar]
- 21.Behboudi S, Boswell S, Williams R. Cell-mediated immune responses to alpha-fetoprotein and other antigens in hepatocellular carcinoma. Liver Int. 2010;30:521–526. doi: 10.1111/j.1478-3231.2009.02194.x. [DOI] [PubMed] [Google Scholar]
- 22.Korangy F, Höchst B, Manns MP, Greten TF. Immune responses in hepatocellular carcinoma. Dig Dis. 2010;28:150–154. doi: 10.1159/000282079. [DOI] [PubMed] [Google Scholar]
- 23.Lee WC, et al. The impact of CD4+ CD25+ T cells in the tumor microenvironment of hepatocellular carcinoma. Surgery. 2012;151:213–222. doi: 10.1016/j.surg.2011.07.029. [DOI] [PubMed] [Google Scholar]
- 24.Sellitto A, et al. Behavior of circulating CD4+CD25+Foxp3+ regulatory T cells in colon cancer patients undergoing surgery. J Clin Immunol. 2011;31:1095–1104. doi: 10.1007/s10875-011-9585-8. [DOI] [PubMed] [Google Scholar]
- 25.Chaput N, et al. Identification of CD8+CD25+Foxp3+ suppressive T cells in colorectal cancer tissue. GUT. 2009;58:520–529. doi: 10.1136/gut.2008.158824. [DOI] [PubMed] [Google Scholar]
- 26.Greten TF, et al. Low-dose cyclophosphamide treatment impairs regulatory T cells and unmasks AFP-specific CD4+ T-cell responses in patients with advanced HCC. J Immunother. 2010;33:211–218. doi: 10.1097/CJI.0b013e3181bb499f. [DOI] [PubMed] [Google Scholar]
- 27.Seddiki N, et al. Persistence of naive CD45RA+ regulatory T cells in adult life. Blood. 2006;107:2830–2838. doi: 10.1182/blood-2005-06-2403. [DOI] [PubMed] [Google Scholar]
- 28.Kryczek I, et al. FOXP3 defines regulatory T cells in human tumor and autoimmune disease. Cancer Res. 2009;69:3995–4000. doi: 10.1158/0008-5472.CAN-08-3804. [DOI] [PubMed] [Google Scholar]
- 29.Ramsdell F. FOXP3 and natural regulatory T cells: key to a cell lineage? Immunity. 2003;19:165–168. doi: 10.1016/S1074-7613(03)00207-3. [DOI] [PubMed] [Google Scholar]
- 30.Baecher-Allan C, Viglietta V, Hafler DA. Human CD4+ CD25+ regulatory T cells. Semin Immunol. 2004;26:89–97. doi: 10.1016/j.smim.2003.12.005. [DOI] [PubMed] [Google Scholar]
- 31.Son CH, et al. CTLA-4 blockade enhances antitumor immunity of intratumoral injection of immature dendritic cells into irradiated tumor in a mouse colon cancer model. J Immunol. 2014;37:1–7. doi: 10.1097/CJI.0000000000000007. [DOI] [PubMed] [Google Scholar]
- 32.Chen ZM, et al. IL-10 and TGF-beta induce alloreactive CD4+ CD25− T cells to acquire regulatory cell function. Blood. 2003;101:5076–5083. doi: 10.1182/blood-2002-09-2798. [DOI] [PubMed] [Google Scholar]
- 33.Grossman WJ, et al. Human T regulatory cells can use the perforin pathway to cause autologous target cell death. Immunity. 2004;21:589–601. doi: 10.1016/j.immuni.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 34.Cao SS, Zhen YS. Potentiation of antimetabolite antitumor activity in vivo by dipyridamole and amphotericin B. Cancer Chemother Pharmacol. 1989;24:181–186. doi: 10.1007/BF00300240. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.