ABSTRACT
We tested the activities of ceftolozane-tazobactam and 13 other antimicrobial agents against 221 strains of Burkholderia cepacia complex and Burkholderia gladioli. Most strains (82%) were cultured from persons with cystic fibrosis, and most (85%) were recovered since 2011. The ceftolozane-tazobactam MIC was ≤8 μg/ml for 77% of the strains. However, the MIC range was broad (≤0.5 to >64 μg/ml; MIC50/90, 2/32 μg/ml). Significant differences in susceptibility to some antimicrobial agents were observed between species.
KEYWORDS: Burkholderia, ceftolozane, cystic fibrosis
TEXT
The 20 species in the Burkholderia cepacia complex (Bcc) and the closely related species Burkholderia gladioli are Gram-negative nonfermenting bacilli that cause opportunistic infections in susceptible hosts, particularly persons with cystic fibrosis (CF) or chronic granulomatous disease. Respiratory tract infection with Burkholderia cenocepacia, Burkholderia multivorans (both members of the Bcc), or B. gladioli accounts for the majority of Burkholderia infections in CF patients (1). Infection is often refractory to therapy, as most Burkholderia strains are resistant to available antimicrobial agents. Thus, there is a need to explore the utility of newer antimicrobials for their activity against Burkholderia species.
Ceftolozane-tazobactam is a combination cephalosporin-beta lactamase inhibitor approved by the Food and Drug Administration in 2014 for treatment of complicated intra-abdominal and complicated urinary tract infections. It has been shown to have good activity against antimicrobial-resistant Gram-negative pathogens, including Enterobacteriaceae and Pseudomonas aeruginosa (2–5). Studies evaluating ceftolozane-tazobactam activity against bacteria recovered from patients with CF have been limited (2, 6), and we are aware of no studies that specifically focused on activity against Burkholderia species. Further, no data have been published regarding differences in susceptibilities to ceftolozane-tazobactam between species within the Bcc.
We tested the activities of ceftolozane-tazobactam and 13 comparator antimicrobial agents against 221 Bcc and B. gladioli isolates from the strain collection of the Burkholderia cepacia Research Laboratory and Repository at the University of Michigan. All strains had been identified to the species level using genetically based methods previously described (7), and all were distinct by genotyping analyses (8). The strains were recovered from 221 patients receiving care in 122 medical centers in 91 cities in 41 U.S. states and Toronto, Ontario, Canada. Most strains (82%) were recovered from respiratory specimens from CF patients. All were recovered between 2005 and 2016, with the majority (85%) being isolated between 2011 and 2016. The distribution of Burkholderia species included in this set reflected that found in CF patients; B. cenocepacia, B. multivorans, and B. gladioli accounted for 62% of the strains (1). Ceftolozane-tazobactam MIC values were determined, in triplicate, using the reference Clinical and Laboratory Standards Institute broth microdilution method (9), and custom-dried antibiotic plates were read on a Sensititre ARIS instrument (Thermo Scientific).
The results of susceptibility testing are summarized in Table 1. Ceftolozane-tazobactam demonstrated good overall activity (MIC50, 2 μg/ml; MIC90, 32 μg/ml) against the 221 Burkholderia strains tested. Although the range of inhibitory activities was broad (MIC range, ≤0.25 to >64 μg/ml), 77% of the strains were inhibited by concentrations of ≤8 μg/ml, which was comparable to the activity demonstrated by ceftazidime, arguably the most relevant drug among the comparator agents tested; 76% of the strains were susceptible (MICs, ≤8 μg/ml) to ceftazidime. Further, ceftolozane-tazobactam was at least 2-fold more active than ceftazidime against 82% of ceftazidime-susceptible strains and at least 4-fold more active against 33% of these strains (Fig. 1). In contrast, ceftazidime was at least 2-fold more active than ceftolozane-tazobactam against only 5% of ceftazidime-susceptible strains and at least 4-fold more active against only 3% of these strains. When MIC50 values were used, ceftolozane-tazobactam showed potency equivalent to that of meropenem and minocycline (MIC50 for all three agents, 2 μg/ml). Ceftolozane-tazobactam showed good activities (MICs, ≤8 μg/ml) against 19 (76%) of the 25 multidrug-resistant (MDR) (10) strains in the test set and against 5 (25%) of the 20 extensively drug-resistant (10) strains. Other agents retaining activities against the MDR strains included ceftazidime (MIC50, 4 μg/ml; 50% susceptible), meropenem (MIC50, 4 μg/ml; 64% susceptible), piperacillin-tazobactam (MIC50, 16 μg/ml), and trimethoprim-sulfamethoxazole (MIC50, ≤1 μg/ml; 80% susceptible).
TABLE 1.
Activities of ceftolozane-tazobactam and comparator agents against Burkholderia strains
| Species or group (na) | Antibiotic(s) | MICs (μg/ml) |
||
|---|---|---|---|---|
| Range | MIC50 | MIC90 | ||
| Burkholderia, all isolates (221) | Ceftolozane-tazobactam | ≤0.25 to >64 | 2 | 32 |
| Amikacin | ≤16 to >64 | 64 | >64 | |
| Aztreonam | ≤4 to >32 | 16 | >32 | |
| Ceftazidime | ≤0.5 to >64 | 4 | 16 | |
| Chloramphenicol | ≤8 to >32 | 16 | 32 | |
| Ciprofloxacin | ≤2 to >8 | ≤2 | >8 | |
| Doripenem | ≤1 to >8 | 4 | 8 | |
| Levofloxacin | ≤1 to >8 | ≤1 | >8 | |
| Meropenem | ≤1 to >16 | 2 | 8 | |
| Minocycline | ≤1 to >16 | 2 | 8 | |
| Piperacillin-tazobactam | ≤4 to >128 | ≤4 | 128 | |
| Tigecycline | ≤2 to 16 | ≤2 | 8 | |
| Tobramycin | ≤2 to >16 | >16 | >16 | |
| Trimethoprim-sulfamethoxazole | ≤1 to >8 | ≤1 | 2 | |
| Burkholderia multivorans (50) | Ceftolozane-tazobactam | 0.5 to >64 | 1 | >64 |
| Amikacin | ≤16 to >64 | 64 | >64 | |
| Aztreonam | ≤4 to >32 | ≤4 | >32 | |
| Ceftazidime | 1 to >64 | 2 | 64 | |
| Chloramphenicol | ≤8 to >32 | ≤8 | 32 | |
| Ciprofloxacin | ≤2 to >8 | ≤2 | >8 | |
| Doripenem | ≤1 to >8 | 4 | 8 | |
| Levofloxacin | ≤1 to >8 | ≤1 | >8 | |
| Meropenem | ≤1 to >16 | 4 | >16 | |
| Minocycline | ≤1 to 16 | <1 | 4 | |
| Piperacillin-tazobactam | ≤4 to >128 | ≤4 | >128 | |
| Tigecycline | ≤2 to 16 | ≤2 | 8 | |
| Tobramycin | ≤2 to >16 | >16 | >16 | |
| Trimethoprim-sulfamethoxazole | ≤1 to 8 | ≤1 | 2 | |
| Burkholderia cenocepacia (42) | Ceftolozane-tazobactam | ≤0.25 to >64 | 1 | 32 |
| Amikacin | ≤16 to >64 | >64 | >64 | |
| Aztreonam | ≤4 to >32 | 16 | >32 | |
| Ceftazidime | 1 to >64 | 4 | 16 | |
| Chloramphenicol | ≤8 to >32 | 16 | >32 | |
| Ciprofloxacin | ≤2 to >8 | ≤2 | >8 | |
| Doripenem | ≤1 to >8 | 4 | >8 | |
| Levofloxacin | ≤1 to >8 | 2 | >8 | |
| Meropenem | ≤1 to 16 | 2 | 8 | |
| Minocycline | ≤1 to >16 | 2 | 16 | |
| Piperacillin-tazobactam | ≤4 to >128 | 8 | 128 | |
| Tigecycline | ≤2 to 16 | ≤2 | 16 | |
| Tobramycin | 8 to >16 | >16 | >16 | |
| Trimethoprim-sulfamethoxazole | ≤1 to 8 | ≤1 | 4 | |
| Burkholderia vietnamiensis (22) | Ceftolozane-tazobactam | ≤0.25 to 32 | 1 | 4 |
| Amikacin | ≤16 to >64 | ≤16 | >64 | |
| Aztreonam | ≤4 to >32 | ≤4 | 16 | |
| Ceftazidime | 1 to 16 | 2 | 4 | |
| Chloramphenicol | ≤8 to >32 | 16 | >32 | |
| Ciprofloxacin | ≤2 to >8 | ≤2 | 4 | |
| Doripenem | ≤1 to 8 | ≤1 | 4 | |
| Levofloxacin | ≤1 to >8 | 2 | 8 | |
| Meropenem | ≤1 to 4 | ≤1 | 4 | |
| Minocycline | ≤1 to 8 | 2 | 4 | |
| Piperacillin-tazobactam | ≤4 to 128 | ≤4 | 16 | |
| Tigecycline | ≤2 to >16 | ≤2 | 4 | |
| Tobramycin | ≤2 to >16 | 8 | >16 | |
| Trimethoprim-sulfamethoxazole | ≤1 to >8 | ≤1 | 8 | |
| Burkholderia cepacia (22) | Ceftolozane-tazobactam | 0.5 to 4 | 1 | 4 |
| Amikacin | 32 to >64 | 32 | >64 | |
| Aztreonam | 8 to >32 | 16 | >32 | |
| Ceftazidime | 2 to 16 | 4 | 8 | |
| Chloramphenicol | ≤8 to >32 | 16 | 32 | |
| Ciprofloxacin | ≤2 to 4 | ≤2 | ≤2 | |
| Doripenem | 4 to 8 | 4 | 8 | |
| Levofloxacin | ≤1 to 8 | 2 | 4 | |
| Meropenem | 2 to 8 | 4 | 4 | |
| Minocycline | ≤1 to 16 | 2 | 4 | |
| Piperacillin-tazobactam | ≤4 to 128 | 8 | 32 | |
| Tigecycline | ≤2 to 16 | ≤2 | 4 | |
| Tobramycin | 16 to >16 | >16 | >16 | |
| Trimethoprim-sulfamethoxazole | ≤1 | ≤1 | ≤1 | |
| Other Bcc spp.b (39) | Ceftolozane-tazobactam | ≤0.25 to >64 | 2 | 16 |
| Amikacin | ≤16 to >64 | 64 | >64 | |
| Aztreonam | ≤4 to >32 | 16 | >32 | |
| Ceftazidime | ≤0.5 to >64 | 4 | 16 | |
| Chloramphenicol | ≤8 to >32 | 16 | 32 | |
| Ciprofloxacin | ≤2 to >8 | ≤2 | 4 | |
| Doripenem | ≤1 to >8 | 4 | 8 | |
| Levofloxacin | ≤1 to >8 | ≤1 | 4 | |
| Meropenem | ≤1 to >16 | 4 | 8 | |
| Minocycline | ≤1 to >16 | 2 | 8 | |
| Piperacillin-tazobactam | ≤4 to 128 | 8 | 128 | |
| Tigecycline | ≤2 to 16 | ≤2 | 8 | |
| Tobramycin | ≤2 to >16 | >16 | >16 | |
| Trimethoprim-sulfamethoxazole | ≤1 to 4 | ≤1 | ≤1 | |
| Burkholderia gladioli (46) | Ceftolozane-tazobactam | 2 to 32 | 8 | 16 |
| Amikacin | ≤16 to 64 | ≤16 | ≤16 | |
| Aztreonam | 16 to >32 | 32 | >32 | |
| Ceftazidime | 1 to 64 | 16 | 16 | |
| Chloramphenicol | ≤8 to >32 | 32 | 32 | |
| Ciprofloxacin | ≤2 to >8 | ≤2 | ≤2 | |
| Doripenem | ≤1 to 4 | ≤1 | 2 | |
| Levofloxacin | ≤1 to >8 | ≤1 | 2 | |
| Meropenem | ≤1 to 8 | ≤1 | 2 | |
| Minocycline | ≤1 to 16 | 2 | 4 | |
| Piperacillin-tazobactam | ≤4 to 64 | ≤4 | ≤4 | |
| Tigecycline | ≤2 to 16 | ≤2 | ≤2 | |
| Tobramycin | ≤2 to >16 | ≤2 | ≤2 | |
| Trimethoprim-sulfamethoxazole | ≤1 to 2 | ≤1 | ≤1 | |
Number tested in each category.
Includes (number of isolates) Burkholderia contaminans (11), Burkholderia dolosa (10), Burkholderia ambifaria (9), and Burkholderia stabilis (9).
FIG 1.
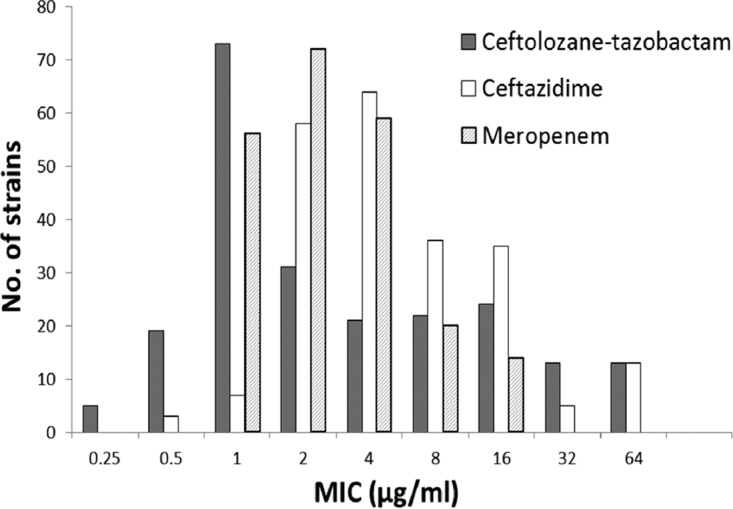
Distribution of MICs of ceftolozane-tazobactam, ceftazidime, and meropenem for 221 Burkholderia strains.
The activities of ceftolozane-tazobactam varied between species within the Bcc. Most notable were the at least 8-fold-poorer activities against B. cenocepacia (MIC90, 32 μg/ml) and B. multivorans (MIC90, >64 μg/ml) relative to those against all other Bcc species tested (MIC90, 4 μg/ml). Other noteworthy differences of the comparator drugs against strains in the test panel included (i) the greater activities of ceftazidime and piperacillin-tazobactam against Burkholderia vietnamiensis strains (MIC90s, 4 μg/ml and 16 μg/ml, respectively) relative to those against all other Bcc species (MIC90s, 16 μg/ml and >128 μg/ml, respectively) and (ii) the greater activities (based on MIC90 values) of piperacillin-tazobactam, tigecycline, and the quinolone and carbapenem antibiotics against B. gladioli compared to those against the Bcc species. The activities of the aminoglycoside antibiotics against B. gladioli, which are in stark contrast to the very poor activities of this class of antibiotics against Bcc species, are well-known distinguishing features of B. gladioli (11).
Although interpretive criteria for ceftolozane-tazobactam susceptibility testing of Burkholderia spp. are not yet established, the MICs of this combination antimicrobial were in a range comparable to those of other agents used to treat Burkholderia infections. It demonstrated marginally superior activity over that of ceftazidime against ceftazidime-susceptible strains and retained activity against most (60%) multidrug-resistant and extensively drug-resistant strains in the test set. Although the Food and Drug Administration does not include infection with Burkholderia in its list of indications for ceftolozane-tazobactam, these results suggest that this combination agent offers a potentially effective additional therapy for the management of Burkholderia infection in CF patients.
ACKNOWLEDGMENTS
This study was supported in part by a research grant from the Investigator Initiated Studies Program of Merck & Co., Inc., Kenilworth, NJ, USA.
The opinions expressed in this paper are those of the authors and do not necessarily represent those of Merck & Co., Inc.
REFERENCES
- 1.LiPuma JJ. 2010. The changing microbial epidemiology in cystic fibrosis. Clin Microbiol Rev 23:299–323. doi: 10.1128/CMR.00068-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Grohs P, Taieb G, Morand P, Kaibi I, Podglajen I, Lavollay M, Mainardi JL, Compain F. 17 January 2017. In vitro activity of ceftolozane-tazobactam against multidrug-resistant non-fermenting Gram-negative bacilli isolated from patients with cystic fibrosis. Antimicrob Agents Chemother doi: 10.1128/AAC.02688-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Snydman DR, McDermott LA, Jacobus NV. 2014. Activity of ceftolozane-tazobactam against a broad spectrum of recent clinical anaerobic isolates. Antimicrob Agents Chemother 58:1218–1223. doi: 10.1128/AAC.02253-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Farrell DJ, Flamm RK, Sader HS, Jones RN. 2013. Antimicrobial activity of ceftolozane-tazobactam tested against Enterobacteriaceae and Pseudomonas aeruginosa with various resistance patterns isolated in U.S. hospitals (2011-2012). Antimicrob Agents Chemother 57:6305–6310. doi: 10.1128/AAC.01802-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Craig WA, Andes DR. 2013. In vivo activities of ceftolozane, a new cephalosporin, with and without tazobactam against Pseudomonas aeruginosa and Enterobacteriaceae, including strains with extended-spectrum β-lactamases, in the thighs of neutropenic mice. Antimicrob Agents Chemother 57:1577–1582. doi: 10.1128/AAC.01590-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kuti JL, Pettit RS, Neu N, Cies JJ, Lapin C, Muhlebach MS, Novak KJ, Nguyen KJ, Nguyen ST, Saiman L, Nicolau DP. 2015. Microbiologic activity of ceftolozane/tazobactam, ceftazidime, meropenem, and piperacillin/tazobactam against Pseudomonas aeruginosa isolated from children with cystic fibrosis. Diagn Microbiol Infect Dis 83:53–55. doi: 10.1016/j.diagmicrobio.2015.04.012. [DOI] [PubMed] [Google Scholar]
- 7.Payne GW, Vandamme P, Morgan SH, LiPuma JJ, Coenye T, Weightman AJ, Jones TH, Mahenthiralingam E. 2005. Development of a recA gene-based identification approach for the entire Burkholderia genus. Appl Environ Microbiol 71:3917–3927. doi: 10.1128/AEM.71.7.3917-3927.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Coenye T, Spilker T, Martin A, LiPuma JJ. 2002. Comparative assessment of genotyping methods for epidemiologic study of Burkholderia cepacia genomovar III. J Clin Microbiol 40:3300–3307. doi: 10.1128/JCM.40.9.3300-3307.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Clinical and Laboratories Standards Institute. 2016. Performance standards for antimicrobial susceptibility testing; 26th ed CLSI Guideline M100. Clinical and Laboratories Standards Institute, Wayne, PA. [Google Scholar]
- 10.Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, Harbarth S, Hindler JF, Kahlmeter G, Olsson-Liljequist B, Paterson DL, Rice LB, Stelling J, Struelens MJ, Vatopoulos A, Weber JT, Monnet DL. 2012. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect 18:268–281. doi: 10.1111/j.1469-0691.2011.03570.x. [DOI] [PubMed] [Google Scholar]
- 11.Segonds C, Clavel-Batut P, Thouverez M, Grenet D, Le Coustumier A, Plésiat P, Chabanon G. 2009. Microbiological and epidemiological features of clinical respiratory isolates of Burkholderia gladioli. J Clin Microbiol 47:1510–1516. doi: 10.1128/JCM.02489-08. [DOI] [PMC free article] [PubMed] [Google Scholar]


