Abstract
Background
Antiangiogenic therapies for malignant gliomas often result in transient response, and recurrent disease is characterized by adoption of invasive and hypoxic phenotype. The notch signaling pathway is activated in gliomas, and augments cell migration and hypoxic response. Here we report a clinical study of the combination of bevacizumab and RO4929097, an inhibitor of the notch signaling cascade.
Methods
A phase I clinical trial was conducted through the Adult Brain Tumor Consortium in subjects with recurrent malignant glioma. Primary objectives were to assess safety and to define the maximum tolerated dose of RO4929097 in combination with bevacizumab. Secondary objectives were to determine overall survival, progression free survival, radiographic response, pharmacokinetic evaluation, and tissue biomarker analysis.
Results
Thirteen subjects were enrolled. Of the three subjects treated with the highest dose of RO4929097, one grade 3 toxicity and one grade 2 toxicity were observed. Definitive maximum tolerated dose of RO4929097 in combination with bevacizumab was not identified due to manufacturer’s decision to halt drug production. Two of 12 evaluable subjects demonstrated radiographic response; one subject experienced CR and the second PR. The median overall survival was 10.9 months with a median progression-free survival of 3.7 months. Two subjects remained free of disease progression at 6 months from treatment initiation. PK evaluation did not identify clinically significant drug-drug interactions. All analyzed tissue specimens revealed activation of notch signaling.
Conclusions
Combination of RO4929097 and bevacizumab was well-tolerated. Given the compelling scientific rationale, additional studies of antiangiogenic and notch signaling inhibitors should be considered.
Keywords: glioma, notch, gamma secretase, bevacizumab, clinical trial
Introduction
Malignant gliomas (MGs) account for approximately 70% of the 22,500 new cases of malignant primary brain tumors in the United States each year. Despite treatment, the median survival is only 15 months for patients with glioblastomas (GBM) and 2 to 5 years for patients with anaplastic astrocytomas (AA).[1] Chemotherapy has shown only modest efficacy against MGs. Better agents are needed to improve the outcome of patients with MG.
Notch, a cell surface protein receptor involved in transmitting growth and proliferation signals is often deregulated in cancer.[2, 3] Two membrane bound Notch families, Jagged and Delta, have been described with multiple ligands identified to date. After ligand binding, two successive proteolytic cleavage steps occur. The resulting active form Notch Intracellular Domain (NICD) translocates to the nucleus, where it binds a transcriptional repressor, thus activating target genes.[4] Blocking Notch signaling via γ-secretase inhibition produces a slower growing, less transformed cancer cells.[4, 5]
Several studies highlight the association between Notch signaling and tumorigenesis. Inappropriate activation of Notch signaling has been shown to result in proliferation, dedifferentiation, and prevention of apoptosis in a variety of cancers.[6–10] Inhibition of Notch1 in embryonal brain tumor depletes the cancer stem cell population and induces differentiation.[5] In infiltrating gliomas, Notch1 signaling was found to enhance nestin expression, suggesting that it exerts a direct effect on maintenance of the undifferentiated phenotype, and thus may contribute to glioma growth.[11, 12]
RO4929097, a γ-secretase inhibitor (GSI), is an orally administered antagonist of Notch signaling by preventing a necessary intramembranous cleavage that liberates NICD, a transcriptional co-activator. Although bevacizumab, a monoclonal antibody directed against vascular endothelial growth factor, has been utilized in the treatment of infiltrating gliomas, its use has been associated with adoption of an invasive phenotype. Here we studied the combination of GSI and bevacizumab in bevacizumab-naïve MG subjects. Notch promotes tumor cell invasion and is also involved in pathologic angiogenesis.[13, 14] Thus, the rationale is that inhibition of Notch signaling pathway may provide direct anti-tumor activity, mitigate invasive effect, and enhance antiangiogenic effect via promotion of “nonfunctional” tumor angiogenesis.
Materials and Methods
Protocol Objectives
The primary objective of this study was to assess the safety profile of RO4929097 in combination with bevacizumab and to determine a recommended Phase II dose of RO4929097 in combination with bevacizumab in patients with recurrent MGs. The secondary objectives were to describe the toxicity associated with this combination regimen, to assess the pharmacokinetics of RO4929097 in combination with bevacizumab, to assess the proportion of patients alive and progression free at 6 months (PFS6), to assess the median overall survival (OS) of the trial population, and to explore potential prognostic biomarkers from glioma tissue at baseline and potential association with Notch pathway inhibition.
Patient Eligibility
This protocol (ClinicalTrials.gov number NCT01189240) was IRB approved, and all study patients signed a written informed consent. Study patients, 18 years of age or older, must have had histologically proven MG which was progressive following radiation therapy and temozolomide chemotherapy. Patients must have had measurable contrast-enhancing recurrent malignant glioma by MRI within two weeks of starting treatment. They must have recovered from severe toxicity of prior therapy. An interval of at least 28 days must have elapsed since any prior surgery. Study patients must have had a Karnofsky Performance Status ≥ 60. They must have had adequate bone marrow and organ function. Women of childbearing potential were required to have a negative pregnancy test within 10–14 days.
Exclusion criteria include serious concurrent infection or medical illness, prior treatment with γ-secretase inhibitor or bevacizumab, serologic positivity for Hepatitis A, B or C, or those who had cirrhosis, or pregnant women or those who were breastfeeding. The remaining exclusion criteria were similar to those of previous brain tumor therapeutic clinical trials.
Trial Design
In this open-label phase I trial with standard 3+3 design, study patients received RO4929097 orally for 3 days on/4 days off each week in 4 week cycles (Days 1–3, 8–10, 15–17, and 22–24) and bevacizumab 10 mg/kg every two weeks by intravenous infusion in an outpatient setting on Days 1 and 15. Patients received brain MRIs with contrast every 8 weeks (2 cycles of RO4929097 and 4 doses of IV bevacizumab). Three pre-specified RO4929097 doses (5, 10, and 20 mg) were evaluated. Three patients per cohort were treated at each RO4929097 dose level and the dose escalation occurred in a stepwise fashion with an expansion to a total of 6 patients at the putative MTD/or the highest dose (20mg). A cycle of therapy was 28 days. To accommodate pharmacokinetic sampling, the first infusion of bevacizumab for study patients was on Day 2 or 3 of Cycle 1. The second infusion was on Day 15 of Cycle 1 and had to have been completed prior to administration of first weekly dose of RO4929097. Treatment cycles were repeated until disease progression or until development of significant toxicities.
The recommended phase II dose of the combination therapy was defined as the maximum administered dose of RO4929097 at which 0 or 1 of 6 study patients experience a DLT. If dose escalation reached the highest dose level with ≤ 1 DLT in 6 patients, then that dose was declared the MTD.
Toxicity Assessment
Safety and toxicity assessments were obtained at each study visit. Adverse events and other symptoms were graded according to the NCI Common Terminology Criteria of Adverse Events 4.0 (CTCAE version 4.0). A standard definition of dose limiting toxicity (DLT) was used. For study patients who experienced a DLT, RO4929097 and/or bevacizumab was held. Patients who experienced a grade 4 non-hematological DLT was taken off treatment. If the trial patient recovered (≤ grade 1 [or tolerable grade 2 for non-hematologic toxicity] or ≤ baseline) a dose reduction was required for subsequent doses, with a maximum of 2 dose reductions. Hematological toxicities were considered dose limiting if any of the following occurred: ANC of ≤ 500/μL or second occurrence of ANC < 1,000/μL (grade 3), PLTs ≤ 25,000/μL or second occurrence of platelets < 50,000/μL (grade 3), febrile neutropenia, or thrombocytopenic bleeding (defined as platelets < 50,000/μL and associated with clinically significant bleeding).
Response Assessment
Although response was not the primary endpoint of this trial, patients with measurable disease were assessed by the RANO (Response Assessment in Neuro-Oncology) criteria every 8 weeks with a contrast-enhanced brain MRI scan and characterized into the following categories: complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD).
Tissue Biomarker Analysis
NICD staining of subject tissues
Indirect immunofluorescence assays of paraffin embedded MG tissues were performed as previously described on pretreated tumor tissue of 9 of 13 enrolled subjects.[15, 16] After deparaffinization, slides were incubated with primary antibodies against activated Notch1 (Abcam, ab8925) at 1:200 concentration overnight at 4°C. This antibody is specific for the human N-terminal sequence of the cleaved Notch. Slides were subsequently probed with Alexa Fluor 488 secondary antibody (Invitrogen, 1:1000). Cell nuclei were counterstained with DAPI (1μg/ml). All slides were photographed with Leica SP5 confocal microscope.
Pharmacokinetic Studies
The objective of the pharmacokinetic studies was to evaluate the effect of bevacizumab on the plasma pharmacokinetics of RO4929097. Sampling was performed to define the time course of the concentration of RO4929097 in plasma during the 24 h dosing interval for the doses given on days 1 and 15 of cycle 1 to all patients enrolled in the Phase I part of the clinical trial. The first dose of bevacizumab was not given until day 2 or 3 after all pharmacokinetic samples for the initial dose of RO4929097 were collected. The RO4929097 dose on day 15 was given shortly after completing bevacizumab infusion. Blood was drawn into tubes containing sodium heparin before and at 0.25, 0.5, 1, 2, 4, 6, 8 and 24 h after taking RO4929097 on days 1 and 15. The sample collection tubes were mixed with the anticoagulant and placed over ice until centrifuged (1,300 g, 10-min, 4°C). Plasma was removed and stored in tubes at −80°C until assayed. Concentration of total RO4929097 in plasma (i.e. free and protein bound drug) was determined by reversed-phase high performance liquid chromatography with mass spectrometric detection as previously described, with minor modifications [17]. The analytical method was validated according to current recommendations [18]. RO4929097 was determined with an interday accuracy of 101.2% and a precision of 2.1% at the 2.5-ng/mL lower limit of quantitation.
RO4929097 plasma concentration-time curves were analyzed by standard noncompartmental methods using WinNonlin Professional 5.0 software (Pharsight Corp., Cary, NC).[19] The maximum concentration of drug in plasma (Cmax) was the observed maximum value for the set of samples obtained for each dosing interval. Area under the plasma concentration-time curve from time zero to 24 h after dosing (AUC24) was estimated by the logarithmic-linear trapezoidal algorithm. Pharmacokinetic parameters are reported as the geometric mean ± SD of values for individual patients at each dose level.[20, 21]
Statistical Analyses
The primary objective of this study was to define the MTD of R04929097 in combination with bevacizumab in patients with recurrent malignant gliomas using a standard 3+3 design. The study also intended to assess over all safety of the treatment and assess the pharmacokinetics of the combination of R04929097 with bevacizumab of the phase I part of the study. Data are presented with standard descriptive summaries. Observed toxicities are summarized based on CTCAE v4.0 with relationship of possible, probable, and definitely to the combination regimen. Response rate is presented as percentage along with 95% confidence interval (exact method). Survival probability was estimated using the Kaplan-Meier method. The confidence interval of median time survival was constructed by the method of Brookmeyer-Crowley. All analyses were conducted using the SAS software (version 9.3, SAS Institute).
Results
Subject Characteristics
A total of 13 recurrent malignant glioma patients were treated with RO4929097 in combination with bevacizumab. One treated patient was ineligible for the trial. The analysis data for toxicity included all 13 treated patients, while the analytical dataset for the trial results other than toxicity included the 12 eligible patients only.
The median age of the study patients was 56 years (range: 42–62). Seven patients were male (58%) and five were female. Eleven of the 12 patients (92%) had a KPS ≥ 80. Eight patients had 1 prior relapse, while 1 patient had 3 prior relapses. Eleven patients had at least a subtotal resection, and ten patients had glioblastomas.
Toxicities
There were three grade 3 toxicities (one with fatigue, two with hypertension) and three grade 4 toxicities (colonic perforation, thrombocytopenia, and sigmoidectomy) that were possibly, probably, or definitely related to either RO4929097, bevacizumab, or both. The most common grade 2 toxicities were fatigue (23%), headache (15%), hypertension (15%), and hypophosphatemia (15%). The most common grade 1 toxicities included neutropenia (38%), thrombocytopenia (31%), elevated ALT (31%), elevated AST (23%), and fatigue (23%) (Table 1).
Table 1.
Overall toxicities that was possible, probable, or definitely attributable to either RO4929097 or Bevacizumab or both.
| Toxicities | Grade 1 |
Grade 2 |
Grade 3 |
Grade 4 |
Total |
|---|---|---|---|---|---|
| No. Of patients (%) | |||||
| Alanine aminotransferase increased | 4(31) | 4(31) | |||
| Alkaline 28hosphatise increased | 1(8) | 1(8) | |||
| Alopecia | 2(15) | 2(15) | |||
| Anemia | 2(15) | 2(15) | |||
| Arthralgia | 1(8) | 1(8) | |||
| Aspartate aminotransferase increased | 3(23) | 3(23) | |||
| Blood bilirubin increased | 1(8) | 1(8) | |||
| Colonic perforation | 1(8) | 1(8) | |||
| Constipation | 2(15) | 2(15) | |||
| Cough | 2(15) | 2(15) | |||
| Dyspepsia | 2(15) | 2(15) | |||
| Dysphagia | 1(8) | 1(8) | |||
| Dysphasia | 1(8) | 1(8) | |||
| Edema limbs | 1(8) | 1(8) | |||
| Epistaxis | 2(15) | 2(15) | |||
| Fall | 1(8) | 1(8) | |||
| Fatigue | 3(23) | 3(23) | 1(8) | 7(54) | |
| Headache | 1(8) | 2(15) | 3(23) | ||
| Hematoma | 1(8) | 1(8) | |||
| Hyperglycemia | 1(8) | 1(8) | |||
| Hypertension | 1(8) | 2(15) | 2(15) | 5(38) | |
| Hypoalbuminemia | 2(15) | 2(15) | |||
| Hypokalemia | 1(8) | 1(8) | |||
| Hypomagnesemia | 1(8) | 1(8) | |||
| Hypophosphatemia | 2(15) | 2(15) | |||
| Irregular Menstruation | 1(8) | 1(8) | |||
| Mucositis oral | 1(8) | 1(8) | 2(15) | ||
| Nausea | 2(15) | 2(15) | |||
| Neutrophil count decreased | 2(15) | 2(15) | |||
| Non-cardiac chest pain | 1(8) | 1(8) | |||
| Platelet count decreased | 4(31) | 1(8) | 5(38) | ||
| Sigmoidectomy | 1(8) | 1(8) | |||
| Vascular disorders | 1(8) | 1(8) | |||
| Voice alteration | 2(15) | 2(15) | |||
| Vomiting | 1(8) | 1(8) | |||
| White blood cell decreased | 5(38) | 5(38) |
Four patients were enrolled at the lowest (5 mg/kg) dose level of RO4929097. There was no grade 4 toxicity at this dose level. One patient had grade 3 fatigue and one had grade 3 hypertension. The remaining toxicities were grades 1 and 2. Initially three additional patients were enrolled in the next dose cohort (10 mg/kg). There was no grade 3 toxicity. One patient developed grade 4 colonic perforation and had a subsequent sigmoidectomy. This adverse event was a known side effect of bevacizumab and was felt to definitely be related to bevacizumab alone, and not secondary to RO4929097. Three more patients were subsequently enrolled at this dose level without additional grade 3 or 4 toxicity. Thus, the trial proceeded to the 3rd and highest dose level (20 mg/kg). At this dose level, there was one grade 3 toxicity (hypertension) and one grade 2 toxicity (headache); the remaining toxicities at this cohort were grade 1. An additional three study patients were planned to enroll at this dose cohort in order to confirm this dose to be the MTD. However, Roche decided to halt patient enrollment in clinical trials involving RO4929097 and cease production of the drug as well as all further clinical development of RO4929097. Thus, the three confirmatory patients could not be enrolled at 20 mg/kg. The recommended phase II dose was 10 mg/kg of RO4929097 orally for 3 days on/4 days off each week in combination with bevacizumab. However, it is possible that had three additional trial patients been enrolled at the highest cohort, 20 mg/kg of RO4929097 may have been the recommended phase II dose.
Pharmacokinetics
The plasma pharmacokinetics of RO4929097 for a single 24 h dosing interval was characterized when given alone and together with bevacizumab in a total of 13 patients during treatment with daily oral doses of 5, 10, or 20 mg. Mean values of the Cmax and AUC24 determined at each dose level are presented in Table 2. The mean Cmax increased less than proportionately with escalation of the dose from 5 to 20 mg. The mean AUC24 for patients receiving the 10 mg dose was approximately 2-fold greater than achieved with the 5 mg dose and essentially the same as the 20 mg dose. Differences between the mean values of Cmax and AUC24 when given alone and following the second biweekly dose of bevacizumab were less than 15% at each dose level evaluated.
Table 2.
RO4929097 pharmacokinetic parametersa
| Dose | Concurrent | No. of | Cmax | AUC24 | |||||
|---|---|---|---|---|---|---|---|---|---|
| (mg) | Day | bevacizumab | patients | (ng/mL) | (ng●h/mL) | ||||
| 5 | 1 | − | 4 | 129 | ± | 41 | 1,215 | ± | 361 |
| 15 | + | 4 | 117 | ± | 37 | 1,212 | ± | 355 | |
| Δ% | −9.7 | −0.2 | |||||||
| 10 | 1 | − | 6 | 177 | ± | 32 | 2,385 | ± | 606 |
| 15 | + | 5b | 189 | ± | 49 | 2,351 | ± | 1,071 | |
| Δ% | 7.0 | −1.5 | |||||||
| 20 | 1 | − | 3 | 241 | ± | 172 | 2,232 | ± | 781 |
| 15 | + | 3 | 245 | ± | 192 | 2,555 | ± | 1,363 | |
| Δ % | 1.9 | 14.5 | |||||||
Values are reported as the geometric mean ± SD.
The AUC24 could only be determined for four patients.
Response Rates
Two (both GBM) of 12 patients (17%, 95%CI: 2–48%) had radiographic responses according to the RANO criteria. One patient had a CR (Figure 1), and another patient had a PR.
Figure 1.

Complete response was observed in one subject.
Magnetic resonance images obtained prior to study initiation (left) and after 5 months of study participation (right), show improvement in both contrast enhanced and FLAIR sequences.
Survival Outcomes
At the last follow-up 10 of the 12 patients had died while 2 were still alive. Median OS was 10.9 months (95%CI: 4–13.3 months). The median PFS was 3.7 months (95%CI: 1.9–5.7 months). Figure 2 demonstrates the Kaplan-Meier plot for OS and PFS of the study cohort. Two of 12 patients (17%, 95%CI: 2–48%) were free of disease progression at 6 months from treatment initiation.
Figure 2.
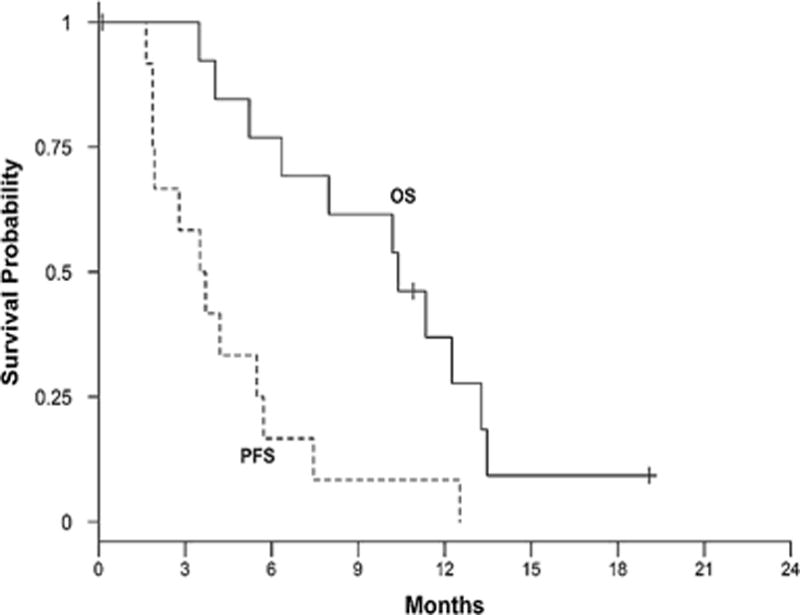
OS and PFS curves for the study cohort.
Tissue Biomarker Analysis
NICD staining of subject tissues
Pretreated tumor tissues were available for 9 of 13 enrolled subjects. Glioma tissues from all nine subjects demonstrated expression of NICD, indicative of cellular notch activation (Figure 3). Staining intensity was categorized into three groups: Strong, Moderate, and Weak.
Figure 3.
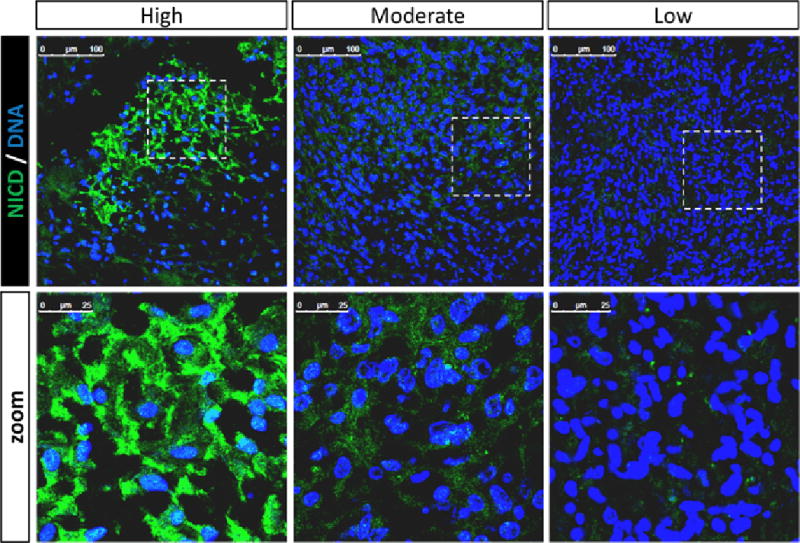
NICD expression in pre-treated GBM tissues. All tested specimens demonstrated expression of the notch intracellular domain. Staining intensities were divided into high, moderate, and low expression. Nuclear counter staining (Dapi) is shown in middle row.
Discussion
There is a critical need to identify more effective therapies for recurrent MG patients. The rationale for combining bevacizumab with a GSI, a notch signaling inhibitor, was based on the hypothesis that notch inhibition may offset development of aggressive cellular behavior seen in bevacizumab refractory cases. Notch signaling has been reported to: 1) Stimulate migration of cancer cells [22, 23]; 2) Play a key role in the activation of cellular hypoxic response by stabilizing HIF-1α [24, 25]; 3) Directly regulate stemness of neural stem cells.[26] Considering that hypoxia in a HIF-1α dependent manner reprograms cells to a stem-like phenotype, inhibition of notch signaling may promote cellular differentiation.[27, 28] Also, the addition of a notch signaling inhibitor to bevacizumab may allow for sustained inhibition of VEGF signaling with attenuation or delay in the development of infiltrative glioma response.[29] A recent study showing hypoxia can enhance γ-secretase activity through a HIF-1α mediated mechanism further validates our strategy of combined inhibition of notch and VEGF.[30]
The aim of this study was to determine a potential MTD dose of the gamma-secretase inhibitor RO4929097 when given concurrently with bevacizumab. Although there was one DLT (grade 4 colonic perforation) at the second dose cohort (10 mg/kg), this was most likely a bevacizumab-related effect, as this is a well described complication of bevacizumab. Additional study patients enrolled at this dose level did not have any grade 3 or 4 toxicity. There was one grade 3 toxicity and no grade 4 toxicity at the highest dose level (20 mg/kg). However, since the study sponsor decided to discontinue production of RO4929097, the final recommended phase II dose of RO4929097 with bevacizumab was 10 mg/kg.
A secondary objective of this clinical trial was to determine if the plasma pharmacokinetics of RO4929097 was affected when given in combination with bevacizumab. Hepatic metabolism mediated by CYP3A4 appears to be a major route of elimination for RO4929097. RO4929097 induces CYP3A4 at doses as low as 10 to 20 mg in cancer patients and it has been shown to increase its own elimination upon repeated daily administration at doses greater than 24 mg.[31, 32] Bevacizumab is not a substrate or modulator of the hepatic CYP450 enzyme system and an effect on the hepatic metabolism of RO4929097 was not anticipated.[33]
Information on the plasma pharmacokinetics of total RO4929097 when given orally as a single agent was described in reports of two phase I clinical trials in patients with advanced solid tumors.[31, 34] The mean AUC24 for the initial 10 mg dose of RO4929097 given to patients with malignant gliomas (2,385 ± 606 ng·h/mL) in the present investigation was in very good agreement with the results when the 10 mg dose was combined with cediranib (38). However, unlike in these two previously reported studies, escalating the daily dose to 20 mg did not result in a significant increase in the mean AUC24 (2,232 ± 781 ng·h/mL). Factors that could account for the apparent decrease in the bioavailability of RO4929097 in patients at the 20 mg dose level were not evident. The pathophysiological characteristics of the patients and their use of concurrent supporting medications were comparable at each dose level in this study. Nevertheless, the results of these studies served to conclusively demonstrate that bevacizumab did not have a clinically significant effect on the pharmacokinetics of RO4929097.
The median OS and PFS results were reported. Definitive conclusions regarding efficacy, however, cannot be made with such few study subjects. Furthermore, radiologic response observed in two subjects may merely reflect bevacizumab effect. Recurrent malignant glioma patients do not have effective therapies and as such, treatment combinations that have even modest efficacy would be highly desirable in this patient population. The combination of RO4929097 and bevacizumab was well-tolerated. Although this particular gamma-secretase inhibitor is no longer being produced, given the compelling scientific rationale and experience of targeting this pathway by other groups, additional studies combining antiangiogenic agents with Notch pathway inhibitors should be further explored.[35]
Supplementary Material
Table 3.
Study Patient Demographic Characteristics
| ABTC 1002 | PHASE I RO4929097+BEV |
|---|---|
| Characteristics | N=12 |
| Age - year | |
| Median (range) | 56 (42–62) |
| Sex - no. (%) | |
| Male | 7(58) |
| Female | 5 (42) |
| Race - no. (%) | |
| African American | 2 (17) |
| White | 10 (83) |
| Karnofsky Performance Status- no. (%) | |
| 90 | 6(50) |
| 80 | 5 (42) |
| 70 | |
| 60 | 1 (8) |
| Mini Mental Score | |
| Median (range) | 29 (21–30) |
| Prior Relapses - no. (%) | |
| 1 | 8 (67) |
| 2 | 3 (25) |
| 3 | 1 (8) |
| Initial Procedure - no. (%) | |
| Resection | 11 (92) |
| Biopsy | 1 (8) |
| Initial Histological Diagnosis no. (%) | |
| Glioblastoma | 10 (83) |
| Other | 2 (17) |
Acknowledgments
Funding: CA UM1 137443 awarded to ABTC
Footnotes
Conflict of Interest:
Timothy F. Cloughesy has been compensated by Roche/Genentech for consulting work and expert testimony. No other authors report conflict of interest.
References
- 1.Wen PY, Kesari S. Malignant gliomas in adults. The New England journal of medicine. 2008;359:492–507. doi: 10.1056/NEJMra0708126. [DOI] [PubMed] [Google Scholar]
- 2.Bolos V, Grego-Bessa J, de la Pompa JL. Notch signaling in development and cancer. Endocr Rev. 2007;28:339–363. doi: 10.1210/er.2006-0046. [DOI] [PubMed] [Google Scholar]
- 3.Bray SJ. Notch signalling: a simple pathway becomes complex. Nat Rev Mol Cell Biol. 2006;7:678–689. doi: 10.1038/nrm2009. [DOI] [PubMed] [Google Scholar]
- 4.Nefedova Y, Gabrilovich D. Mechanisms and clinical prospects of Notch inhibitors in the therapy of hematological malignancies. Drug Resist Updat. 2008;11:210–218. doi: 10.1016/j.drup.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fan X, Matsui W, Khaki L, Stearns D, Chun J, Li YM, Eberhart CG. Notch pathway inhibition depletes stem-like cells and blocks engraftment in embryonal brain tumors. Cancer research. 2006;66:7445–7452. doi: 10.1158/0008-5472.CAN-06-0858. [DOI] [PubMed] [Google Scholar]
- 6.Aster JC, Pear WS, Blacklow SC. Notch signaling in leukemia. Annu Rev Pathol. 2008;3:587–613. doi: 10.1146/annurev.pathmechdis.3.121806.154300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Staal FJ, Langerak AW. Signaling pathways involved in the development of T-cell acute lymphoblastic leukemia. Haematologica. 2008;93:493–497. doi: 10.3324/haematol.12917. [DOI] [PubMed] [Google Scholar]
- 8.Chen Y, De Marco MA, Graziani I, Gazdar AF, Strack PR, Miele L, Bocchetta M. Oxygen concentration determines the biological effects of NOTCH-1 signaling in adenocarcinoma of the lung. Cancer research. 2007;67:7954–7959. doi: 10.1158/0008-5472.CAN-07-1229. [DOI] [PubMed] [Google Scholar]
- 9.Konishi J, Kawaguchi KS, Vo H, Haruki N, Gonzalez A, Carbone DP, Dang TP. Gamma-secretase inhibitor prevents Notch3 activation and reduces proliferation in human lung cancers. Cancer research. 2007;67:8051–8057. doi: 10.1158/0008-5472.CAN-07-1022. [DOI] [PubMed] [Google Scholar]
- 10.Peacock CD, Watkins DN. Cancer stem cells and the ontogeny of lung cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2008;26:2883–2889. doi: 10.1200/JCO.2007.15.2702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kanamori M, Kawaguchi T, Nigro JM, Feuerstein BG, Berger MS, Miele L, Pieper RO. Contribution of Notch signaling activation to human glioblastoma multiforme. J Neurosurg. 2007;106:417–427. doi: 10.3171/jns.2007.106.3.417. [DOI] [PubMed] [Google Scholar]
- 12.Shih AH, Holland EC. Notch signaling enhances nestin expression in gliomas. Neoplasia. 2006;8:1072–1082. doi: 10.1593/neo.06526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zeng Q, Li S, Chepeha DB, Giordano TJ, Li J, Zhang H, Polverini PJ, Nor J, Kitajewski J, Wang CY. Crosstalk between tumor and endothelial cells promotes tumor angiogenesis by MAPK activation of Notch signaling. Cancer cell. 2005;8:13–23. doi: 10.1016/j.ccr.2005.06.004. [DOI] [PubMed] [Google Scholar]
- 14.Li JL, Sainson RC, Oon CE, Turley H, Leek R, Sheldon H, Bridges E, Shi W, Snell C, Bowden ET, Wu H, Chowdhury PS, Russell AJ, Montgomery CP, Poulsom R, Harris AL. DLL4-Notch signaling mediates tumor resistance to anti-VEGF therapy in vivo. Cancer research. 2011;71:6073–6083. doi: 10.1158/0008-5472.CAN-11-1704. [DOI] [PubMed] [Google Scholar]
- 15.Soeda A, Hara A, Kunisada T, Yoshimura S, Iwama T, Park DM. The evidence of glioblastoma heterogeneity. Scientific reports. 2015;5:7979 d. doi: 10.1038/srep07979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Park DM, Li J, Okamoto H, Akeju O, Kim SH, Lubensky I, Vortmeyer A, Dambrosia J, Weil RJ, Oldfield EH, Park JK, Zhuang Z. N-CoR pathway targeting induces glioblastoma derived cancer stem cell differentiation. Cell cycle. 2007;6:467–470. doi: 10.4161/cc.6.4.3856. [DOI] [PubMed] [Google Scholar]
- 17.Wu J, Wiegand R, LoRusso P, Li J. Validation and implementation of a liquid chromatography/tandem mass spectrometry assay for quantitation of the total and unbound RO4929097, a gamma-secretase inhibitor targeting Notch signaling, in human plasma. Journal of chromatography B, Analytical technologies in the biomedical and life sciences. 2011;879:1537–1543. doi: 10.1016/j.jchromb.2011.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shah VP, Midha KK, Findlay JW, Hill HM, Hulse JD, McGilveray IJ, McKay G, Miller KJ, Patnaik RN, Powell ML, Tonelli A, Viswanathan CT, Yacobi A. Bioanalytical method validation–a revisit with a decade of progress. Pharmaceutical research. 2000;17:1551–1557. doi: 10.1023/a:1007669411738. [DOI] [PubMed] [Google Scholar]
- 19.Gabrielsson J. Pharmacokinetic/Pharmacodynamic Data Analysis: Concepts and Applications. Swedish Pharmaceutical Press; Uppsala, Sweden: 1994. [Google Scholar]
- 20.Lacey LF, Keene ON, Pritchard JF, Bye A. Common noncompartmental pharmacokinetic variables: are they normally or log-normally distributed? Journal of biopharmaceutical statistics. 1997;7:171–178. doi: 10.1080/10543409708835177. [DOI] [PubMed] [Google Scholar]
- 21.Mizuta E, Tsubotani A. Preparation of mean drug concentration–time curves in plasma. A study on the frequency distribution of pharmacokinetic parameters. Chemical & pharmaceutical bulletin. 1985;33:1620–1632. doi: 10.1248/cpb.33.1620. [DOI] [PubMed] [Google Scholar]
- 22.Sahlgren C, Gustafsson MV, Jin S, Poellinger L, Lendahl U. Notch signaling mediates hypoxia-induced tumor cell migration and invasion. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:6392–6397. doi: 10.1073/pnas.0802047105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bolos V, Mira E, Martinez-Poveda B, Luxan G, Canamero M, Martinez AC, Manes S, de la Pompa JL. Notch activation stimulates migration of breast cancer cells and promotes tumor growth. Breast cancer research: BCR. 2013;15:R54. doi: 10.1186/bcr3447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gustafsson MV, Zheng X, Pereira T, Gradin K, Jin S, Lundkvist J, Ruas JL, Poellinger L, Lendahl U, Bondesson M. Hypoxia requires notch signaling to maintain the undifferentiated cell state. Developmental cell. 2005;9:617–628. doi: 10.1016/j.devcel.2005.09.010. [DOI] [PubMed] [Google Scholar]
- 25.Qiang L, Wu T, Zhang HW, Lu N, Hu R, Wang YJ, Zhao L, Chen FH, Wang XT, You QD, Guo QL. HIF-1alpha is critical for hypoxia-mediated maintenance of glioblastoma stem cells by activating Notch signaling pathway. Cell death and differentiation. 2012;19:284–294. doi: 10.1038/cdd.2011.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Androutsellis-Theotokis A, Leker RR, Soldner F, Hoeppner DJ, Ravin R, Poser SW, Rueger MA, Bae SK, Kittappa R, McKay RD. Notch signalling regulates stem cell numbers in vitro and in vivo. Nature. 2006;442:823–826. doi: 10.1038/nature04940. [DOI] [PubMed] [Google Scholar]
- 27.Soeda A, Park M, Lee D, Mintz A, Androutsellis-Theotokis A, McKay R, Engh J, Iwama T, Kunisada T, Kassam A. Hypoxia promotes expansion of the CD133-positive glioma stem cells through activation of HIF-1α. Oncogene. 2009;28:3949–3959. doi: 10.1038/onc.2009.252. [DOI] [PubMed] [Google Scholar]
- 28.Bar EE, Lin A, Mahairaki V, Matsui W, Eberhart CG. Hypoxia increases the expression of stem-cell markers and promotes clonogenicity in glioblastoma neurospheres. The American journal of pathology. 2010;177:1491–1502. doi: 10.2353/ajpath.2010.091021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hovinga KE, Shimizu F, Wang R, Panagiotakos G, Van Der Heijden M, Moayedpardazi H, Correia AS, Soulet D, Major T, Menon J, Tabar V. Inhibition of notch signaling in glioblastoma targets cancer stem cells via an endothelial cell intermediate. Stem cells. 2010;28:1019–1029. doi: 10.1002/stem.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Villa JC, Chiu D, Brandes AH, Escorcia FE, Villa CH, Maguire WF, Hu CJ, de Stanchina E, Simon MC, Sisodia SS, Scheinberg DA, Li YM. Nontranscriptional role of Hif-1alpha in activation of gamma-secretase and notch signaling in breast cancer. Cell reports. 2014;8:1077–1092. doi: 10.1016/j.celrep.2014.07.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Diaz-Padilla I, Hirte H, Oza AM, Clarke BA, Cohen B, Reedjik M, Zhang T, Kamel-Reid S, Ivy SP, Hotte SJ, Razak AA, Chen EX, Brana I, Wizemann M, Wang L, Siu LL, Bedard PL. A phase Ib combination study of RO4929097, a gamma-secretase inhibitor, and temsirolimus in patients with advanced solid tumors. Investigational new drugs. 2013;31:1182–1191. doi: 10.1007/s10637-013-0001-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tolcher AW, Messersmith WA, Mikulski SM, Papadopoulos KP, Kwak EL, Gibbon DG, Patnaik A, Falchook GS, Dasari A, Shapiro GI, Boylan JF, Xu ZX, Wang K, Koehler A, Song J, Middleton SA, Deutsch J, Demario M, Kurzrock R, Wheler JJ. Phase I study of RO4929097, a gamma secretase inhibitor of Notch signaling, in patients with refractory metastatic or locally advanced solid tumors. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2012;30:2348–2353. doi: 10.1200/JCO.2011.36.8282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shih T, Lindley C. Bevacizumab: an angiogenesis inhibitor for the treatment of solid malignancies. Clinical therapeutics. 2006;28:1779–1802. doi: 10.1016/j.clinthera.2006.11.015. [DOI] [PubMed] [Google Scholar]
- 34.Sahebjam S, Bedard PL, Castonguay V, Chen Z, Reedijk M, Liu G, Cohen B, Zhang WJ, Clarke B, Zhang T, Kamel-Reid S, Chen H, Ivy SP, Razak AR, Oza AM, Chen EX, Hirte HW, McGarrity A, Wang L, Siu LL, Hotte SJ. A phase I study of the combination of ro4929097 and cediranib in patients with advanced solid tumours (PJC-004/NCI 8503) British journal of cancer. 2013;109:943–949. doi: 10.1038/bjc.2013.380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xu R, Shimizu F, Hovinga K, Beal K, Karimi S, Droms L, Peck KK, Gutin P, Iorgulescu JB, Kaley T, DeAngelis L, Pentsova E, Nolan C, Grommes C, Chan T, Bobrow D, Hormigo A, Cross JR, Wu N, Takebe N, Panageas K, Ivy P, Supko JG, Tabar V, Omuro A. Molecular and Clinical Effects of Notch Inhibition in Glioma Patients: A Phase 0/I Trial. Clinical cancer research: an official journal of the American Association for Cancer Research. 2016 doi: 10.1158/1078-0432.CCR-16-0048. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


