Abstract
Background
With increasing age, estimated glomerular filtration rate (eGFR) decline is a frequent manifestation and is strongly associated with other preclinical target organ damage (TOD). In literature, many equations exist in assessing patients’ eGFR. However, these equations were mainly derived and validated in the population from Western countries, which equation should be used for risk stratification in the Chinese population remains unclear, as well as their comparison. Considering that TOD is a good marker for risk stratification in the elderly, in this analysis, we aimed to investigate whether the recent eGFR equations derived from Asian and Chinese are better associated with preclinical TOD than the other equations in elderly Chinese.
Methods
A total of 1,599 community-dwelling elderly participants (age >65 years) in northern Shanghai were prospectively recruited from June 2014 to August 2015. Conventional cardiovascular risk factors were assessed, and hypertensive TOD including left ventricular mass index (LVMI), carotid–femoral pulse wave velocity (cf-PWV), carotid intima-media thickness (IMT), ankle–brachial index (ABI) and urine albumin to creatinine ratio (UACR) was evaluated for each participant. Participant’s eGFR was calculated from the Modification of Diet in Renal Disease (MDRD), Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI), Chinese-abbreviated MDRD (c-aMDRD), Asian-modified CKD-EPI (aCKD-EPI) equation and Chinese-modified CKD-EPI (cCKD-EPI) equation.
Results
In multivariate regression analysis, only eGFRs from aCKD-EPI were significantly and inversely associated with carotid IMT (P=0.005). In multivariate logistic models, decreased eGFR from all the equations were significantly associated with lower ABI (P<0.001), microalbuminuria (P=0.02 to P<0.001) and increased cf-PWV (P<0.001). Only decreased eGFRs from aCKD-EPI and cCKD-EPI equations were significantly associated with increased IMT (both crude P<0.05). In the receiver operator characteristic (ROC) analysis, only aCKD-EPI and cCKD-EPI equations presented significant associations with all the listed preclinical TODs (P-value from <0.05 to <0.001).
Conclusion
In community-dwelling elderly Chinese, eGFRs from aCKD-EPI and cCKD-EPI equations are better associated with preclinical TOD. aCKD-EPI and cCKD-EPI equations should be preferred when making risk assessment.
Keywords: elderly Chinese, eGFR equation, preclinical target organ damage, eGFR
Background
In terms of epidemiological transition, end-stage cardiovascular disease, including atrial fibrillation, renal disease and heart failure, is becoming more and more frequent.1 This condition grows even worse for the aging population, as the incidence and prevalence of most known cardiovascular disorders increase with age.2 However in China, we are lacking in evidence for the management of chronic diseases in elderly. Therefore, studies based on elderly Chinese are warranted.
Estimated glomerular filtration rate (eGFR) calculated from serum–creatinine concentration is a convenient approach to assess patients’ renal function, which is strongly associated with the occurrence and development of hypertensive target organ damage (TOD).3–6 Current hypertension guidelines including the European Society of Hypertension/European Society of Cardiology guidelines advocate the estimation of glomerular filtration rate (GFR) from serum–creatinine concentrations as an effective approach to enhancing risk prediction.6,7 For decades, the most commonly used eGFR equations were mainly established and validated in Caucasians, such as Cockcroft–Gault, Modification of Diet in Renal Disease (MDRD) and recently emerging Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI).8–10 It was reported that Cockcroft–Gault equation is less accurate than the MDRD study equation in elderly people.11–13 However, the MDRD equation underestimated the gold standard GFR in Asian population.14 The CKD-EPI equation is the most recent equation established for estimating GFR, and the CKD-EPI creatinine equation appears to be more accurate than the MDRD equation, as the accuracy of the CKD-EPI equation is validated in various study populations and clinical conditions.10 Moreover, in recent years, some investigators proposed and validated some Asian- or Chinese-modified eGFR equations, which might be more appropriate for Chinese.15,16
Nevertheless, which eGFR equation should be used for risk prediction in elderly Chinese is still under debate.17 We have documented in the protocol that TOD, as an intermediate state between conventional risk factors (age, smoking, diabetes mellitus [DM]) and clinical events, may be a good marker for risk stratification in the elderly.18,19 It might better represent exposure to risk factors than the risk factor itself.20 To make risk assessment in an elderly participant, examining the associations between eGFR and preclinical TOD is essential, as well as dictating which equation to choose. A unique eGFR equation is essential to make the risk prediction and risk score calculation.
To date, there are no studies comparing the eGFR equations in their associations with preclinical TOD in elderly Chinese.13 Therefore, based on the existing eGFR equations, we used data from the Northern Shanghai Study to investigate: whether eGFR calculated from newly established eGFR equations associates more strongly with the preclinical hypertensive TOD in a Chinese community-based elderly cohort.
Methods
Study design and participants
This analysis was nested in the Northern Shanghai Study (ClinicalTrials.gov Identifier: NCT02368938) under the registered name “Prognosis Factors of Mortality and Cardiovascular Diseases in the Elderly Chinese: the Northern Shanghai Study”, which was a prospective, ongoing and multistage community-based study.19 This study was approved by the Shanghai Tenth People’s Hospital Institutional Review Board.
The study design was described in the protocol already.19 Between June 2014 and August 2015, 1,721 subjects were invited, of whom 1,599 (92.9%) were enrolled. In 1,599 participants, 1,586 participants’ serum creatinine concentrations were measured. Among these 1,586 participants, 1,579 (99%) had intima-media thickness (IMT), 1,553 (98%) had ankle–brachial index (ABI), 1,577 (99%) had left ventricular mass index (LVMI), 1,537 (97%) had urine albumin to creatinine ratio (UACR) and 1,533 (97%) had pulse wave velocity. All these 1,586 participants had basic anthropometric data, including body weight, body height and office blood pressure. Thus, the final sample size in our analysis was 1,586.
Social, anthropometric and biological parameters
As it was described already, information including sex, age, weight, and height, smoking habits, history of DM, hypertension, cardio- or cerebrovascular diseases and usage of medications was obtained from the questionnaire.19 The venous blood and the urine sample were obtained in a fasting state in the morning. Blood creatinine was measured by enzymatic method.
Estimation of GFR
To estimate GFR, the MDRD study equation, the derived Chinese-abbreviated MDRD (c-aMDRD) equation, the CKD-EPI equation, the Asian-modified CKD-EPI (aCKD-EPI) equation and the Chinese-modified CKD-EPI (cCKD-EPI) equation were used.8,10,13,15–17,21
Measurements of hypertensive TOD
As previously described, carotid artery IMT was measured using the MyLab 30 Gold Cardiovascular System (Esaote SpA, Genoa, Italy) with a 7.5 MHz transducer.19,22,23 Four-limb blood pressure of participants was measured with VP-1000 (Omron Healthcare, Kyoto, Japan). Carotid–femoral pulse wave velocity (cf-PWV), as a golden standard of arterial stiffness, was performed with applanation tonometry (SphygmoCor, AtCor Medical, Sydney, Australia) according to the European Expert Consensus on Arterial Stiffness.24–26 The urine microalbumin to creatinine ratio was calculated from the urine sample test.
Statistical analysis
Baseline demographic and clinical characteristics were presented as mean ± standard deviation (SD) or median (interquartile range) for continuous variables and count (percentage) for categorical variables. Relations between eGFR and hypertensive TOD indicators were performed using multivariate regression analysis. Logistic regression analysis was performed to determine the independent relations between different equation-derived eGFRs and the hypertensive TOD. The SD of eGFR values was calculated respectively (Table S1). To adjust confounders, systolic blood pressure, smoking, body mass index (BMI), DM, low-density lipoprotein cholesterol (LDL-c), urinary albumin, serum calcium, serum phosphorus and hypertension duration were forced into the model. On the other hand, use of age and sex as adjustors for relations between eGFR and TOD, in which eGFR is also calculated from age and sex, would have resulted in adjustments for age and sex effects twice. Thus, we exclude age and sex from the confounders to avoid excessive computation. Maximum likelihood parameter estimates of odds ratios with 95% Wald confidence intervals were calculated. C-statistics were used to assess the discrimination abilities of adjusted logistic models (Table S2). Akaike information criterion was used to compare the goodness-of-fit among the different models (Table S3). The area under the curve (AUC) of the receiver operator characteristic (ROC) curves was used to indicate the detection value of eGFR equations. Z-test was used to compare the AUCs. Statistical analyses were performed using the SAS software version 9.4 (SAS Institute, Inc., Cary, NC, USA). The two-sided significant level of 5% was considered as the level of statistical significance.
Ethics approval
This study was approved by the Shanghai Tenth People’s Hospital Institutional Review Board. All participants signed a consent form.
Results
Characteristics of the participants and investigated eGFR equations
Basic anthropometric data and associated disease conditions were available in 1,586 participants (Table 1), including conventional cardiovascular risk factors, asymptomatic hypertensive TOD, eGFR, diseases and treatment. The present elderly population had an average age of 72.7±6.0 years. Among these community-dwelling elderly, 44.58% (n=707) were male and 22.8% (n=362) were smokers, with an average BMI of 23.9±3.5 kg/m2 and an average blood pressure of 134±18/79±9 mmHg (Table 1). Following the population sample condition, the overall hypertensive TOD conditions are listed (Table 1). The preliminary values of eGFRs (n=1,586) are listed in Table 1, and a different profile of eGFRs is also listed. A large proportions of participants with hypertension (52.6%, n=834) and stage 2 CKD (57%, n=904) are also described (Table 1). The original studies from which the eGFR equation was derived are listed in Table S4.
Table 1.
Characteristics of the community-based participants
| Sample number (male), n (%) | 1,586 (44.58) |
| Age (years) | 72.7±6.0 |
| BMI (kg/m2) | 23.9±3.5 |
| Brachial SBP/DBP (mmHg) | 134±18/79±9 |
| Smoking, n (%) | 362 (22.8) |
| TODs | |
| LVMI (g/m2) | 89.96±28.55 |
| LVH % | 27.5 |
| Aortic PWV (m/s) | 9.41±2.31 |
| Increased aortic PWV % | 32.4 |
| IMT (mm) | 0.61±0.15 |
| Increased IMT % | 4.1 |
| LABI | 1.04±0.13 |
| Low ABI % | 18.9 |
| UACR (mg/g) | 54.9±181.9 |
| Microalbuminuria % | 38.7 |
| eGFR, n=1,586 | |
| MDRD | 84.02±20.56 |
| c-aMDRD | 92.45±24.49 |
| CKD-EPI | 78.18±14.26 |
| aCKD-EPI | 93.04±18.03 |
| cCKD-EPI | 76.31±14.92 |
| Diseases and treatments, n=1,586 | |
| DM, n (%) | 310 (19.6) |
| Hypertensive patients, n (%) | 834 (52.6) |
| Antihypertensive treatment, n (%) | 791 (49.9) |
| Stage 2 CKD, n (%) | 904 (57.0) |
| Stage 3 or more CKD, n (%) | 134 (8.5) |
Notes: Continuous variables are shown as mean ± SD or median (interquartile range) as appropriate. Stage 2 CKD: eGFR 60–89 mL/min/1.73 m2 (MDRD) and stage 3 or more CKD: eGFR <60 mL/min/1.73 m2 (MDRD).
Abbreviations: BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; TOD, target organ damage; LVMI, left ventricular mass index; LVH, left ventricular hypertrophy; PWV, pulse wave velocity; IMT, intima–media thickness; LABI, left ankle brachial index; ABI, ankle–brachial index; UACR, urine albumin to creatinine ratio; eGFR, estimated glomerular filtration rate; MDRD, Modification of Diet in Renal Disease; c-aMDRD, Chinese-abbreviated MDRD; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; DM, diabetes mellitus; SD, standard deviation; CKD, chronic kidney disease.
Relations between equation-derived eGFRs and preclinical hypertensive TOD
As shown in Table 2, eGFRs calculated from all the equations were inversely associated with cf-PWV and positively associated with ABI after adjusting confounders (P<0.001). eGFRs from CKD-EPI, aCKD-EPI and cCKD-EPI equations were inversely associated with LVMI (P<0.05). eGFRs from the CKD-EPI equation were significantly associated with UACR (β=−0.06, P=0.01). As for IMT, after adjustment, only eGFRs calculated from the aCKD-EPI equation were significantly associated with IMT (β=−0.07, P=0.005; Table 2). Crude linear regression results were consistent with it and are shown in Table S5 (β=−0.10, P<0.001 and β=−0.08, P=0.002 for aCKD-EPI and cCKD-EPI, respectively).
Table 2.
Relations between equation-eGFR and cardiovascular TOD indicators
| Equations | IMT (n=1,579)
|
ABI (n=1,553)
|
LVMI (n=1,577)
|
UACR (n=1,537)
|
Aortic PWV (n=1,533)
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| β | P-value | β | P-value | β | P-value | β | P-value | β | P-value | |
| MDRD | 0.001 | 0.95 | 0.12 | <0.001 | −0.05 | 0.07 | −0.04 | 0.09 | −0.14 | <0.001 |
| CKD-EPI | −0.02 | 0.42 | 0.16 | <0.001 | −0.06 | 0.01 | −0.06 | 0.01 | −0.20 | <0.001 |
| aCKD-EPI | −0.07 | 0.005 | 0.12 | <0.001 | −0.06 | 0.02 | −0.03 | 0.30 | −0.17 | <0.001 |
| cCKD-EPI | −0.05 | 0.07 | 0.11 | <0.001 | −0.06 | 0.02 | −0.03 | 0.28 | −0.16 | <0.001 |
| c-aMDRD | −0.01 | 0.78 | 0.11 | <0.001 | −0.05 | 0.06 | −0.03 | 0.17 | −0.13 | <0.001 |
Note: Adjusted confounders: SBP, smoking, BMI, DM, LDL-c, urinary albumin, serum calcium, serum phosphorus and hypertension duration.
Abbreviations: eGFR, estimated glomerular filtration rate; TOD, target organ damage; IMT, intima–media thickness; ABI, ankle–brachial index; LVMI, left ventricular mass index; UACR, urine albumin to creatinine ratio; PWV, pulse wave velocity; MDRD, Modification of Diet in Renal Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD; SBP, systolic blood pressure; BMI, body mass index; DM, diabetes mellitus; LDL-c, low-density lipoprotein cholesterol.
eGFRs and occurrence of carotid hypertrophy
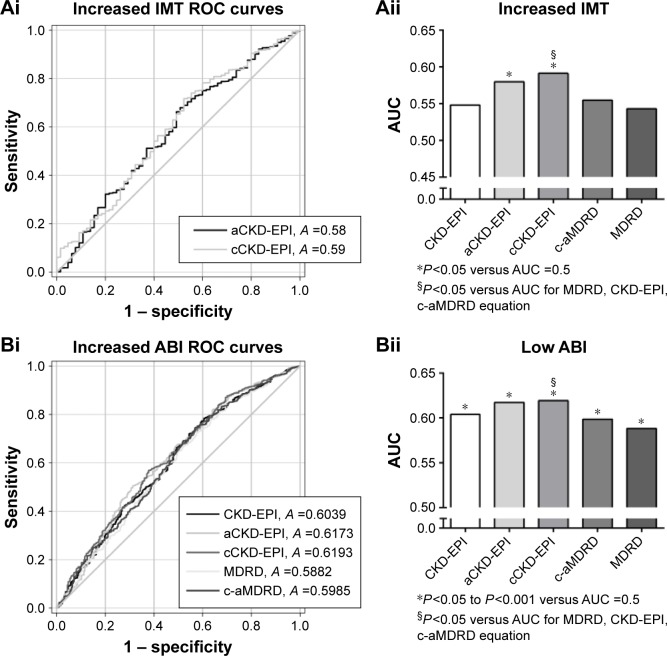
For carotid hypertrophy (IMT ≥0.9 mm), decreased eGFRs from all the equations failed to show an independent association with carotid hypertrophy (all P>0.05; Table 3). However, eGFRs calculated from aCKD-EPI and cCKD-EPI equations showed a good performance in associations with increased IMT (both P<0.05 versus AUC =0.5, Figure 1Ai). Especially for the eGFR from cCKD-EPI, it had a better performance than the other equations (except for aCKD-EPI) in distinguishing carotid hypertrophy (P<0.05 versus AUC of other equations except for aCKD-EPI; Figure 1Ai and Aii).
Table 3.
The performance (adjusted logistic regression) of different eGFRs to detect the cardiovascular TODs with a specific number of each assessment
| Total (n=1,586)
|
Carotid hypertrophy (n=1,579)
|
Lower limb atherosclerosis (n=1,553)
|
Cardiac hypertrophy (n=1,577)
|
Macro-albuminuria (n=1,537)
|
Arterial stiffening (n=1,533)
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| Equations | OR* | P-value | OR* | P-value | OR* | P-value | OR* | P-value | OR* | P-value |
| MDRD | 1.15 (0.86–1.55) | 0.35 | 1.52 (1.30–1.78) | <0.001 | 1.09 (0.96–1.24) | 0.19 | 1.83 (1.26–2.67) | 0.002 | 1.49 (1.30–1.71) | <0.001 |
| CKD-EPI | 1.12 (0.87–1.43) | 0.38 | 1.51 (1.33–1.71) | <0.001 | 1.07 (0.95–1.20) | 0.27 | 1.71 (1.29–2.27) | <0.001 | 1.55 (1.38–1.75) | <0.001 |
| aCKD-EPI | 1.19 (0.91–1.55) | 0.20 | 1.52 (1.32–1.74) | <0.001 | 1.17 (1.03–1.32) | 0.016 | 1.59 (1.18–2.16) | 0.003 | 1.48 (1.30–1.68) | <0.001 |
| cCKD-EPI | 1.26 (0.94–1.69) | 0.13 | 1.56 (1.34–1.82) | <0.001 | 1.13 (1.00–1.28) | 0.045 | 1.53 (1.07–2.19) | 0.02 | 1.52 (1.34–1.73) | <0.001 |
| c-aMDRD | 1.17 (0.87–1.59) | 0.30 | 1.56 (1.32–1.83) | <0.001 | 1.02 (0.90–1.16) | 0.74 | 1.77 (1.21–2.60) | 0.003 | 1.47 (1.28–1.69) | <0.001 |
Notes: Odds ratio is presented as OR (95%CI).
Indicating per 1 SD decrease in eGFR. Adjusted confounders: systolic blood pressure, smoking, BMI, DM, LDL-c, urinary albumin, serum calcium, serum phosphorus and hypertension duration. Considering an increased IMT (IMT ≥0.9 mm) as carotid hypertrophy, a lower ABI (≤0.9) as lower limb atherosclerosis, left ventricular hypertrophy (>115 g/m2 for male and >95 g/m2 for female) as cardiac hypertrophy, an increased UACR (>30 mg/g) as micro-albuminuria and an increased PWV (>10 m/s) as arterial stiffening.
Abbreviations: BMI, body mass index; TOD, target organ damage; PWV, pulse wave velocity; IMT, intima–media thickness; ABI, ankle–brachial index; UACR, urine albumin to creatinine ratio; eGFR, estimated glomerular filtration rate; MDRD, Modification of Diet in Renal Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; c-aMDRD, Chinese-abbreviated MDRD; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; DM, diabetes mellitus; SD, standard deviation; LDL-c, low-density lipoprotein cholesterol.
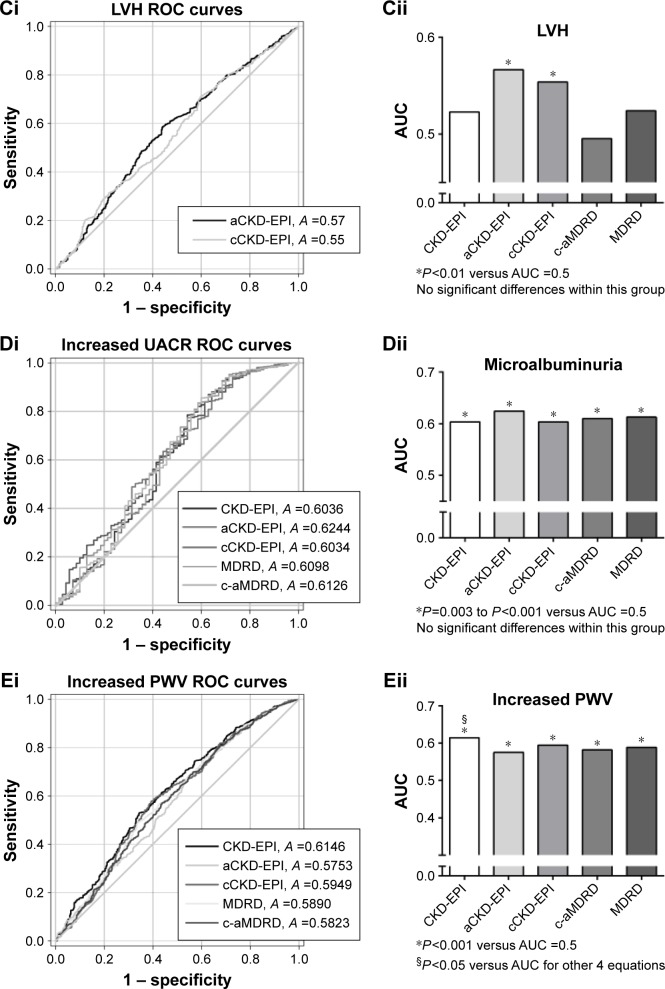
Figure 1.
The performance (AUC of the ROC curve) of different eGFRs to detect specific cardiovascular TOD.
Notes: Only P<0.05 versus AUC =0.5 of the eGFR equations are shown in the ROC curves. On the left panel (Ai, Bi, Ci, Di, Ei), only eGFR equations with P<0.05 versus AUC =0.5 are presented in the ROC curves. On the right panel (Aii, Bii, Cii, Dii, Eii), AUCs of different eGFR equations are presented as histograms of different gray levels. The abbreviation for each equation is under the x-axis. The significance is presented at bottom.
Abbreviations: AUC, area under the curve; ROC, receiver operator characteristic; eGFR, estimated glomerular filtration rate; TOD, target organ damage; IMT, intima-media thickness; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; c-aMDRD, Chinese-abbreviated MDRD; MDRD, Modification of Diet in Renal Disease; ABI, ankle–brachial index; LVH, left ventricular hypertrophy; UACR, urine albumin to creatinine ratio; PWV, pulse wave velocity.
eGFRs and occurrence of lower limb atherosclerosis
For lower limb atherosclerosis, which was characterized with a lower ABI (≤0.9), decreased eGFRs calculated from all the equations were independently associated with lower limb atherosclerosis (all P<0.001; Table 3). Moreover, eGFR from all the equations showed a good performance in associations with lower limb atherosclerosis (P<0.05 to P<0.001 versus AUC =0.5; Figure 1Bi). Especially, the eGFR from cCKD-EPI showed a better association with lower limb atherosclerosis than eGFRs from other equations except for aCKD-EPI (P<0.05 versus AUC of other equations except for aCKD-EPI; Figure 1Bi and Bii).
eGFRs and occurrence of cardiac hypertrophy
For left ventricular hypertrophy, the increased eGFRs from aCKD-EPI and cCKD-EPI equations were independently associated with cardiac hypertrophy (P=0.016 and P=0.045 for aCKD-EPI and cCKD-EPI, respectively). Moreover, only eGFRs from aCKD-EPI and cCKD-EPI equations showed a good performance for the detection of LVH (both P=0.002 versus AUC =0.05; Figure 1Ci and Cii).
eGFRs and occurrence of microalbuminuria
For microalbuminuria, which was defined as an increased UACR (>30 mg/g), decrease in eGFRs calculated from all the equations was independently associated with the occurrence of microalbuminuria after confounders were adjusted (P=0.02 to P<0.001; Table 3). The eGFRs from all the equations showed a good performance in distinguishing the occurrence of microalbuminuria (P<0.001 versus AUC =0.5; Figure 1Di). However, no significant differences in detection value were found within these equations (Figure 1Di and Dii).
eGFRs and occurrence of arterial stiffening
For the arterial stiffening, which was defined as pulse wave velocity (PWV) >10 m/s, decrease in eGFRs calculated from all the equations was independently associated with arterial stiffening (P<0.001; Table 3). The eGFRs from all the equations had a good performance in detecting increased PWV (P<0.001 versus AUC =0.5; Figure 1Ei), especially eGFRs from the CKD-EPI equation (P<0.05 versus AUC of other four equations; Figure 1Ei and Eii).
Performances of the model from a specific eGFR equation to detect different TODs
The discriminatory properties of the adjusted logistic model depended on the choice of TOD and the equation to calculate eGFR (Table S2). To detect the listed cardiovascular TOD, all the logistic models in which the eGFRs were calculated from different equations showed moderate predictive ability for the discrimination of TOD (C-statistics from 0.613 [95% CI, 0.536–0.690] to 0.747 [95% CI, 0.667–0.828]). There is a trend that the model had a better performance in detecting microalbuminuria and arterial stiffening (C statistics from 0.707 [95% CI, 0.680–0.734] to 0.747 [95% CI, 0.667–0.828]). Under most conditions, the goodness-of-fit among the different models showed no significant differences. In particular, eGFRs from the CKD-EPI equation showed the best goodness-of-fit among the different models compared with eGFRs from other equations (Table S3).
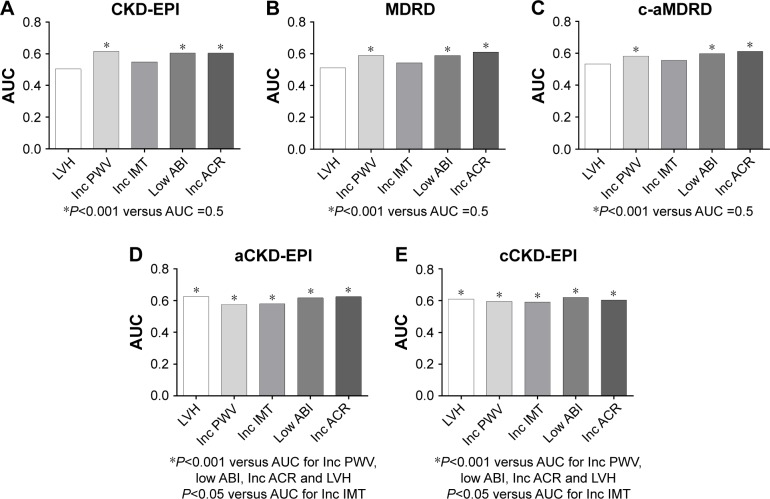
Figure 2 shows a preliminary performance summary of eGFRs to detect TOD, classified by a specific equation. The eGFRs from aCKD-EPI and cCKD-EPI equations have a good performance for the detection of all the listed hypertensive TOD (Figure 2D and E). The eGFRs from other three equations failed to distinguish cardiac hypertrophy and carotid hypertrophy (Figure 2A–C).
Figure 2.
The performance (AUC of the ROC curve) of eGFRs from a specific equation to detect a series of cardiovascular TOD.
Notes: (A–C) eGFRs (calculated from CKD-EPI, MDRD and c-aMDRD equations) versus AUC =0.5 have statistical differences with PWV, ABI and UACR. (D and E) The AUCs of eGFR (calculated from aCKD-EPI and cCKD-EPI equations) versus AUC =0.5 have statistical differences with all the listed hypertensive TOD. AUCs were classified by each eGFR equation. AUCs of different TODs are presented as histograms of different colors. The abbreviation for each TOD is under the x-axis. The significance is presented at bottom.
Abbreviations: AUC, area under the curve; ROC, receiver operator characteristic; eGFR, estimated glomerular filtration rate; TOD, target organ damage; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; MDRD, Modification of Diet in Renal Disease; c-aMDRD, Chinese-abbreviated MDRD; PWV, pulse wave velocity; ABI, ankle–brachial index; UACR, urine albumin to creatinine ratio; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; LVH, left ventricular hypertrophy; IMT, intima-media thickness; ACR, albumin to creatinine ratio.
Discussion
Summary of findings
The Northern Shanghai Study is one of the largest ongoing prospective population study in 1,599 community-dwelling elderly Chinese, and it was authorized and municipal funded with prior registration. In the present analysis, we identified that eGFRs from aCKD-EPI and cCKD-EPI have a better linear dependence with IMT. By ROC analysis, the aCKD-EPI and cCKD-EPI showed a better performance for the detection of the listed hypertensive TOD. Therefore, eGFRs from aCKD-EPI and cCKD-EPI equations were better associated with hypertensive TOD compared with other eGFR equations. aCKD-EPI and cCKD-EPI equations may be more helpful for risk stratification.
eGFRs and hypertensive TOD in the previous studies
As mentioned earlier, CKD affects a large part of the aging population, which has a considerable prevalence of TOD. The associations between eGFR and other TOD have been widely investigated in previous studies and in our analysis, including cf-PWV, UACR, ABI, IMT and LVH.
In our analysis, decreased eGFRs calculated from all the equations were significantly associated with arterial stiffening (increased cf-PWV). This result was consistent with the Health Aging and Body Composition (ABC) study. In the Health ABC study, which consisted of 2,129 elderly adults, PWV was associated with a rapid decline in renal function over a median of 8.9 years (incident rate ratio [IRR], 95% CI, 1.39, 1.09–1.77). ABC investigators emphasized the adjustment for blood pressure (BP), and we also adjusted BP in our analysis.27
As for UACR, the association of reduced eGFR and increased albumin to creatinine ratio (ACR) was consistent with previous studies. Reduced eGFR and increased ACR are always accompanied by each other and are also consistent in a trend of risk prediction.28,29
Previous studies also proved that low ABI was associated with the decline in eGFR calculated from CKD-EPI equation. In a prospective cohort study consisting of 2,592 participants from the Framingham Offspring cohort, investigators found that, compared to normal ABI, low ABI was associated with 5.73-fold increased odds of rapid eGFR decline (95% CI, 2.77–11.85; P<0.001).30 This trend was also consistent with our results.
On the other hand, investigators revealed that for a general community population of 3,629 participants, reduced eGFR calculated from CKD-EPI-derived equations was independently associated with carotid artery thickening. The odds ratio for increased IMT was 1.299 (95% CI, 1.062–1.588).31 This result was in line with our finding when taking the community elderly population of our analysis into account.
As for LVH, it was positively associated with renal function decline. Furthermore, in a retrospective study consisting of 300 in- and outpatients, Chinese investigators found that renal function decline (calculated from CKD-EPI) would contribute to pathological LVH in the elderly.3 Their finding suggests that LVH is associated with reduced eGFR calculated from the CKD-EPI equation. This conclusion was in accordance with the trend in our analysis.
Possible reasons for the better associations between equations and TOD
Our results suggested that the aCKD-EPI equation and cCKD-EPI equation were associated better with hypertensive TOD than the other equations in the elderly Chinese community-dwelling population. Such associations have some underlying reasons.
The superiority of CKD-EPI equation-derived eGFR in detecting an increased PWV and left ventricular hypertrophy has been validated. The investigators argued more on the associations between the MDRD equation and the CKD-EPI equation.32 Especially, the four-level ethnicity CKD-EPI equation significantly decreased bias in Asians, including Chinese, although the original CKD-EPI equation may not be accurate for Asians.15,33 Thus, in the four-level ethnicity CKD-EPI equation, the aCKD-EPI equation may be more accurate for elderly Chinese.
In terms of the detection value of eGFR, age and race are important factors that should be considered, as age and race played a vital role in the eGFR calculation. The superiority of cCKD-EPI to detect the elderly’s TOD may come from the influence of the population’s age profile. The population from which the cCKD-EPI was derived had an average age of 61±12.7 years, and our target population had an average age of 72.7±6.0 years, both of which were >60 years old. Although the populations of other equations also had elderly participants, the average age was relatively younger. Thus, from the age point of view, the population of the cCKD-EPI study was the most adjacent one to our investigated population (Table S4).
These associations may also come from the influence of the population’s race. Our target population is community-based elderly Chinese. The cCKD-EPI study has a population of Chinese. The validation cohort of aCKD-EPI equation consisted of 675 Chinese and 248 Japanese, which means that the majority of the cohort is Chinese. Therefore, from the race point of view, the populations from which the aCKD-EPI and cCKD-EPI were derived were adjacent to our community-dwelling population (Table S4). Furthermore, both coefficients of these two equations were calculated from the local population. A previous study revealed that adjusting a coefficient based on the local population would improve their accuracy.33 Thus, these associations of these two equations in detecting hypertensive TOD may come from the adjusted coefficients based on the local population.
Strengths and limitations
The present analysis has several strengths. First, this study is the first study comparing the eGFR equations in their associations with preclinical TOD in elderly Chinese. Second, we had a good population consistency of various measurements. We analyzed five major hypertensive TOD in >97% (97%–99%) of the final 1,586 participants in our analysis. There are also limitations in this study. First, in this study, the serum creatinine concentrations were measured by the enzymatic method, so that we excluded the Cockcroft–Gault equation, which is based on the Jaffe method. Second, our target was to compare the associations between equation-derived eGFRs and TOD; we did not have the data of GFR from inulin clearance. Thus, the superiority is only for the associations with preclinical TOD. Finally, in this study, we should avoid overinterpretation of the present data as the present analysis was cross-sectional in nature; the ROC curves were not particularly impressive. Additionally, a limited number of participants had clinically significant increases in IMT (n=65, 4.12% of the total 1,586 participants). Therefore, future longitudinal analysis and a clinical sample with a high prevalence of TOD are required.
Conclusion
Considering that TOD is a good marker for risk stratification in the elderly, by showing that Chinese specific CKD-EPI equation derived eGFRs were associated better with 5 major preclinical TOD, cCKD-EPI equation and aCKD-EPI equation are more helpful for risk stratification. Future studies may give attention to the detection value of eGFR for future cardiovascular events. And prior interventions and managements for renal diseases in the elderly should also be considered.
Supplementary materials
Table S1.
The respectively calculated SDs of eGFR values for the logistic regression
| Total (n=1,586)
|
IMT (n=1,579)
|
ABI (n=1,553)
|
LVMI (n=1,577)
|
UACR (n=1,537)
|
Aortic PWV (n=1,533)
|
|---|---|---|---|---|---|
| Equations | SD | SD | SD | SD | SD |
| MDRD | 20.5683668 | 20.5557993 | 20.5548362 | 20.4855702 | 20.5961544 |
| CKD-EPI | 14.2426876 | 14.2090751 | 14.2406751 | 14.1873433 | 14.1307789 |
| aCKD-EPI | 18.0208154 | 18.0347565 | 18.0355388 | 17.9665128 | 17.9131654 |
| cCKD-EPI | 14.9272658 | 14.9582379 | 14.9289286 | 14.8616948 | 14.9174066 |
| c-aMDRD | 24.4979976 | 24.5138248 | 24.4881662 | 24.3729941 | 24.5445129 |
Abbreviations: SD, standard deviation; eGFR, estimated glomerular filtration rate; IMT, intima–media thickness; ABI, ankle–brachial index; LVMI, left ventricular mass index; UACR, urine albumin to creatinine ratio; PWV, pulse wave velocity; MDRD, Modification of Diet in Renal Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD.
Table S2.
Assessment of the discrimination abilities of adjusted logistic models to detect the cardiovascular TOD
| Total (n=1,586)
|
Carotid hypertrophy (n=1,579)
|
Lower limb atherosclerosis (n=1,553)
|
Cardiac hypertrophy (n=1,577)
|
Microalbuminuria (n=1,537)
|
Arterial stiffening (n=1,533)
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| Equations | C-statistic (95% CI) | P-value | C-statistic (95% CI) | P-value | C-statistic (95% CI) | P-value | C-statistic (95% CI) | P-value | C-statistic (95% CI) | P-value |
| MDRD | 0.618 (0.543, 0.693) |
<0.001 | 0.640 (0.604, 0.676) |
<0.001 | 0.685 (0.656, 0.713) |
<0.001 | 0.738 (0.657, 0.819) |
<0.001 | 0.709 (0.682, 0.736) |
<0.001 |
| CKD-EPI | 0.613 (0.536, 0.690) |
0.002 | 0.647 (0.611, 0.682) |
<0.001 | 0.686 (0.657, 0.714) |
<0.001 | 0.747 (0.667, 0.828) |
<0.001 | 0.723 (0.696, 0.749) |
<0.001 |
| aCKD-EPI | 0.615 (0.538, 0.692) |
0.002 | 0.647 (0.612, 0.681) |
<0.001 | 0.685 (0.656, 0.713) |
<0.001 | 0.729 (0.646, 0.812) |
<0.001 | 0.707 (0.680, 0.734) |
<0.001 |
| cCKD-EPI | 0.624 (0.550, 0.699) |
<0.001 | 0.649 (0.614, 0.684) |
<0.001 | 0.685 (0.656, 0.714) |
<0.001 | 0.724 (0.643, 0.805) |
<0.001 | 0.713 (0.685, 0.740) |
<0.001 |
| c-aMDRD | 0.620 (0.545, 0.694) |
<0.001 | 0.644 (0.608, 0.680) |
<0.001 | 0.685 (0.656, 0.713) |
<0.001 | 0.736 (0.654, 0.817) |
<0.001 | 0.707 (0.680, 0.734) |
<0.001 |
Notes: Adjusted confounders: SBP, smoking, BMI and DM for carotid hypertrophy, lower limb atherosclerosis, microalbuminuria and arterial stiffening and sex, SBP, smoking, BMI and DM for cardiac hypertrophy. Considering an increased IMT (IMT ≥0.9 mm) as carotid hypertrophy, a lower ABI (≤0.9) as lower limb atherosclerosis, left ventricular hypertrophy (>115 g/m2 for males and >95 g/m2 for females) as cardiac hypertrophy, an increased UACR (>30 mg/g) as microalbuminuria and an increased PWV (>10 m/s) as arterial stiffening.
Abbreviations: TOD, target organ damage; MDRD, Modification of Diet in Renal Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD; SBP, systolic blood pressure; BMI, body mass index; DM, diabetes mellitus; IMT, intima–media thickness; ABI, ankle–brachial index; UACR, urine albumin to creatinine ratio; PWV, pulse wave velocity.
Table S3.
Goodness-of-fit among the different models
| Total (n=1,586)
|
Carotid hypertrophy (n=1,579)
|
Lower limb atherosclerosis (n=1,553)
|
Cardiac hypertrophy (n=1,577)
|
Microalbuminuria (n=1,537)
|
Arterial stiffening (n=1,533)
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| Equations | AIC | P-value | AIC | P-value | AIC | P-value | AIC | P-value | AIC | P-value |
| MDRD | 521.247 | ns | 1,421.704 | <0.01 | 1,736.528 | ns | 302.080 | ns | 1,711.694 | <0.01 |
| CKD-EPI | 521.392 | ns | 1,411.528 | Reference | 1,737.047 | ns | 299.038 | Reference | 1,690.964 | Reference |
| aCKD-EPI | 520.530 | ns | 1,415.310 | ns | 1,732.407 | Reference | 303.961 | ns | 1,707.235 | <0.01 |
| cCKD-EPI | 519.782 | Reference | 1,415.070 | ns | 1,734.270 | ns | 306.967 | 0.02 | 1,701.476 | <0.01 |
| c-aMDRD | 521.030 | ns | 1,419.735 | 0.02 | 1,738.172 | ns | 303.390 | ns | 1,713.643 | <0.01 |
Abbreviations: AIC, Akaike information criterion; MDRD, Modification of Diet in Renal Disease; ns, not significant; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD.
Table S4.
List of the eGFR equations in our study
| Equations | eGFR (mL/min/1.73 m2) | Participant | Race | Reference |
|---|---|---|---|---|
| MDRD | 186× Scr−1.154× age−0.203×0.742 (if female) ×1.212 (if black) | Middle aged CKD patients Age =50.6±12.7 years1 |
Black and White | Group TMoDiRDS2 |
| c-aMDRD | 175× Scr−1.234× age−0.179×0.79 (if female) | Middle aged CKD patients Age =49.9±15.8 years |
Chinese | Collaboration CeI3 |
| CKD-EPI | Female & Scr ≤0.7: 144 (166 if black) × (Scr/0.7)−0.329× (0.993)age Female & Scr >0.7: 144 (166 if black) × (Scr/0.7)−1.209× (0.993)age Male & Scr ≤0.9: 141 (163 if black) × (Scr/0.9)−0.411× (0.993)age Male & Scr >0.9: 141 (163 if black) × (Scr/0.9)−1.209× (0.993)age |
Middle aged CKD patients Age =47±15 years |
Black and White or other | Levey et al4 |
| aCKD-EPI | Female & Scr ≤0.7: 151× (Scr/0.7)−0.328× (0.993)age Female & Scr >0.7: 151× (Scr/0.7)−1.210× (0.993)age Male & Scr ≤0.9: 149× (Scr/0.9)−0.412× (0.993)age Male & Scr >0.9: 149× (Scr/0.9)−1.210× (0.993)age |
Middle aged CKD patients Age =49±15 years |
Asian (675 Chinese and 248 Japanese) | Stevens et al5 |
| cCKD-EPI | Female & Scr ≤0.7: 94× (Scr/0.7)−0.511× (0.998)age Female & Scr >0.7: 128× (Scr/0.7)−0.543× (0.992)age Male & Scr ≤0.9: 117× (Scr/0.9)−0.277× (0.994)age Male & Scr >0.9: 102× (Scr/0.9)−0.558× (0.994)age |
T2DM CKD patients Age =61.0±12.7 years |
Chinese | Liu et al6 |
Abbreviations: eGFR, estimated glomerular filtration rate; MDRD, Modification of Diet in Renal Disease; CKD, chronic kidney disease; c-aMDRD, Chinese-abbreviated MDRD; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI.
Table S5.
Crude relations between equation-eGFR and cardiovascular TOD indicators
| Equations | IMT (n=1,579)
|
ABI (n=1,553)
|
LVMI (n=1,577)
|
UACR (n=1,537)
|
Aortic PWV (n=1,533)
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| β | P-value | β | P-value | β | P-value | β | P-value | β | P-value | |
| MDRD | −0.005 | 0.84 | 0.11 | <0.001 | −0.06 | 0.01 | −0.07 | 0.004 | −0.15 | <0.001 |
| CKD-EPI | −0.03 | 0.19 | 0.15 | <0.001 | −0.09 | <0.001 | −0.11 | <0.001 | −0.22 | <0.001 |
| aCKD-EPI | −0.10 | <0.001 | 0.12 | <0.001 | −0.09 | <0.001 | −0.07 | 0.005 | −0.17 | <0.001 |
| cCKD-EPI | −0.08 | 0.002 | 0.10 | <0.001 | −0.07 | 0.003 | −0.06 | 0.03 | −0.15 | <0.001 |
| c-aMDRD | −0.02 | 0.41 | 0.10 | <0.001 | −0.06 | 0.01 | −0.07 | 0.009 | −0.13 | <0.001 |
Note: Crude linear regression was performed without adjustment.
Abbreviations: eGFR, estimated glomerular filtration rate; TOD, target organ damage; IMT, intima-media thickness; ABI, ankle–brachial index; LVMI, left ventricular mass index; UACR, urine albumin to creatinine ratio; PWV, pulse wave velocity; MDRD, Modification of Diet in Renal Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD.
References
- 1.Levey AS, Coresh J, Greene T, et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med. 2006;145(4):247–254. doi: 10.7326/0003-4819-145-4-200608150-00004. [DOI] [PubMed] [Google Scholar]
- 2.Group TMoDiRDS Design and statistical issues of the modification of diet in renal disease trial. Control Clin Trials. 1991;12(5):566–586. doi: 10.1016/0197-2456(91)90069-x. [DOI] [PubMed] [Google Scholar]
- 3.Collaboration CeI Modification and evaluation of MDRD estimating equation for Chinese patients with chronic kidney disease. Chin J Nephrol. 2006;22(10):589–595. [Google Scholar]
- 4.Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stevens LA, Claybon MA, Schmid CH, et al. Evaluation of the chronic kidney disease epidemiology collaboration equation for estimating the glomerular filtration rate in multiple ethnicities. Kidney Int. 2011;79(5):555–562. doi: 10.1038/ki.2010.462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liu X, Gan X, Chen J, Lv L, Li M, Lou T. A new modified CKD-EPI equation for Chinese patients with type 2 diabetes. PloS One. 2014;9(10):e109743. doi: 10.1371/journal.pone.0109743. [DOI] [PMC free article] [PubMed] [Google Scholar]
Acknowledgments
We would like to express our great gratitude to all the people who participated in the Northern Shanghai Study. We also thank Yuyan Lu, Yiwu Zhou and Henry Xu for the dedication and precious advice they provided. This analysis is nested in the Northern Shanghai study which is under the financial support from the Shanghai municipal government (Grant IDs 2013ZYJB0902 and 15GWZK1002). Dr Yi Zhang was supported by the National Nature Science Foundation of China (Grant IDs 81300239 and 81670377).
Hongwei Ji, Han Zhang, Jing Xiong, Shikai Yu, Chen Chi and Jue Li acquired the original data for this study. Jacques Blacher, Yi Zhang and Yawei Xu formulated the methods and designed the protocol. Hongwei Ji and Han Zhang drafted the manuscript. All authors contributed toward data analysis, drafting and critically revising the paper and agree to be accountable for all aspects of the work.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Blacher J, Levy BI, Mourad JJ, Safar ME, Bakris G. From epidemiological transition to modern cardiovascular epidemiology: hypertension in the 21st century. Lancet. 2016;388(10043):530–532. doi: 10.1016/S0140-6736(16)00002-7. [DOI] [PubMed] [Google Scholar]
- 2.Rich MW, Chyun DA, Skolnick AH, et al. Knowledge gaps in cardiovascular care of the older adult population. J Am Coll Cardiol. 2016;67(20):2419–2440. doi: 10.1016/j.jacc.2016.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shi HT, Wang XJ, Li J, et al. Association of left ventricular hypertrophy with a faster rate of renal function decline in elderly patients with non-end-stage renal disease. J Am Heart Assoc. 2015;4(11):e002213. doi: 10.1161/JAHA.115.002213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tomiyama H, Townsend RR, Matsumoto C, et al. Arterial stiffness/central hemodynamics, renal function, and development of hypertension over the short term. J Hypertens. 2014;32(1):90–99. doi: 10.1097/HJH.0b013e3283658e7d. [DOI] [PubMed] [Google Scholar]
- 5.Salles GF, Cardoso CR, Pereira VS, Fiszman R, Muxfeldt ES. Prognostic significance of a reduced glomerular filtration rate and interaction with microalbuminuria in resistant hypertension: a cohort study. J Hypertens. 2011;29(10):2014–2023. doi: 10.1097/HJH.0b013e32834adb09. [DOI] [PubMed] [Google Scholar]
- 6.Matsushita K, Coresh J, Sang Y, et al. Estimated glomerular filtration rate and albuminuria for prediction of cardiovascular outcomes: a collaborative meta-analysis of individual participant data. Lancet Diabetes Endocrinol. 2015;3(7):514–525. doi: 10.1016/S2213-8587(15)00040-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mahmoodi BK, Matsushita K, Woodward M, et al. Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without hypertension: a meta-analysis. Lancet. 2012;380(9854):1649–1661. doi: 10.1016/S0140-6736(12)61272-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Group TMoDiRDS Design and statistical issues of the modification of diet in renal disease trial. Control Clin Trials. 1991;2(5):566–586. doi: 10.1016/0197-2456(91)90069-x. [DOI] [PubMed] [Google Scholar]
- 9.Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16(1):31–41. doi: 10.1159/000180580. [DOI] [PubMed] [Google Scholar]
- 10.Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Froissart M, Rossert J, Jacquot C, Paillard M, Houillier P. Predictive performance of the modification of diet in renal disease and Cockcroft-Gault equations for estimating renal function. J Am Soc Nephrol. 2005;16(3):763–773. doi: 10.1681/ASN.2004070549. [DOI] [PubMed] [Google Scholar]
- 12.Verhave JC, Fesler P, Ribstein J, du Cailar G, Mimran A. Estimation of renal function in subjects with normal serum creatinine levels: influence of age and body mass index. Am J Kidney Dis. 2005;46(2):233–241. doi: 10.1053/j.ajkd.2005.05.011. [DOI] [PubMed] [Google Scholar]
- 13.Stevens LA, Coresh J, Greene T, Levey AS. Assessing kidney function – measured and estimated glomerular filtration rate. N Engl J Med. 2006;354(23):2473–2483. doi: 10.1056/NEJMra054415. [DOI] [PubMed] [Google Scholar]
- 14.Imai E, Horio M, Nitta K, et al. Estimation of glomerular filtration rate by the MDRD study equation modified for Japanese patients with chronic kidney disease. Clin Exp Nephrol. 2007;11(1):41–50. doi: 10.1007/s10157-006-0453-4. [DOI] [PubMed] [Google Scholar]
- 15.Stevens LA, Claybon MA, Schmid CH, et al. Evaluation of the Chronic Kidney Disease Epidemiology Collaboration equation for estimating the glomerular filtration rate in multiple ethnicities. Kidney Int. 2011;79(5):555–562. doi: 10.1038/ki.2010.462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liu X, Gan X, Chen J, Lv L, Li M, Lou T. A new modified CKD-EPI equation for Chinese patients with type 2 diabetes. PLoS One. 2014;9(10):e109743. doi: 10.1371/journal.pone.0109743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lamb EJ, Stevens PE. Estimating and measuring glomerular filtration rate: methods of measurement and markers for estimation. Curr Opin Nephrol Hypertens. 2014;23(3):258–266. doi: 10.1097/01.mnh.0000444813.72626.88. [DOI] [PubMed] [Google Scholar]
- 18.Vernooij JW, van der Graaf Y, Nathoe HM, Bemelmans RH, Visseren FL, Spiering W. Hypertensive target organ damage and the risk for vascular events and all-cause mortality in patients with vascular disease. J Hypertens. 2013;31(3):492–499. doi: 10.1097/HJH.0b013e32835cd3cd. discussion 499–500. [DOI] [PubMed] [Google Scholar]
- 19.Ji H, Xiong J, Yu S, et al. Northern Shanghai Study: cardiovascular risk and its associated factors in the Chinese elderly – a study protocol of a prospective study design. BMJ Open. 2017;7(3):e013880. doi: 10.1136/bmjopen-2016-013880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.van der Veen PH, Geerlings MI, Visseren FL, et al. Hypertensive target organ damage and longitudinal changes in brain structure and function: the second manifestations of arterial Disease-Magnetic Resonance Study. Hypertension. 2015;66(6):1152–1158. doi: 10.1161/HYPERTENSIONAHA.115.06268. [DOI] [PubMed] [Google Scholar]
- 21.Collaboration CeI Modification and evaluation of MDRD estimating equation for Chinese patients with chronic kidney disease. Chin J Nephrol. 2006;22(10):589–595. [Google Scholar]
- 22.Zhang Y, Agnoletti D, Protogerou AD, et al. Characteristics of pulse wave velocity in elastic and muscular arteries: a mismatch beyond age. J Hypertens. 2013;31(3):554–559. doi: 10.1097/HJH.0b013e32835d4aec. discussion 559. [DOI] [PubMed] [Google Scholar]
- 23.Lu Y, Zhu M, Bai B, et al. Comparison of carotid-femoral and brachial-ankle pulse-wave velocity in association with target organ damage in the community-dwelling elderly Chinese: The Northern Shanghai Study. Journal of the American Heart Association. 2017;6(2):e004168. doi: 10.1161/JAHA.116.004168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chirinos JA. Arterial stiffness: basic concepts and measurement techniques. J Cardiovasc Transl Res. 2012;5(3):243–255. doi: 10.1007/s12265-012-9359-6. [DOI] [PubMed] [Google Scholar]
- 25.Laurent S, Cockcroft J, Van Bortel L, et al. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J. 2006;27(21):2588–2605. doi: 10.1093/eurheartj/ehl254. [DOI] [PubMed] [Google Scholar]
- 26.Van Bortel LM, Laurent S, Boutouyrie P, et al. Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J Hypertens. 2012;30(3):445–448. doi: 10.1097/HJH.0b013e32834fa8b0. [DOI] [PubMed] [Google Scholar]
- 27.Madero M, Peralta C, Katz R, et al. Association of arterial rigidity with incident kidney disease and kidney function decline: the Health ABC study. Clin J Am Soc Nephrol. 2013;8(3):424–433. doi: 10.2215/CJN.07900812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Grams ME, Sang Y, Ballew SH, et al. A meta-analysis of the association of estimated GFR, albuminuria, age, race, and sex with acute kidney injury. Am J Kidney Dis. 2015;66(4):591–601. doi: 10.1053/j.ajkd.2015.02.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Komenda P, Rigatto C, Tangri N. Estimated glomerular filtration rate and albuminuria: diagnosis, staging, and prognosis. Curr Opin Nephrol Hypertens. 2014;23(3):251–257. doi: 10.1097/01.mnh.0000444910.55665.e8. [DOI] [PubMed] [Google Scholar]
- 30.Foster MC, Ghuman N, Hwang SJ, Murabito JM, Fox CS. Low ankle-brachial index and the development of rapid estimated GFR decline and CKD. Am J Kidney Dis. 2013;61(2):204–210. doi: 10.1053/j.ajkd.2012.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wu Y, Hou J, Li J, Luo Y, Wu S. Correlation between carotid intima-media thickness and early-stage chronic kidney disease: results from asymptomatic polyvascular abnormalities in community study. J Stroke Cerebrovasc Dis. 2016;25(2):259–265. doi: 10.1016/j.jstrokecerebrovasdis.2015.09.026. [DOI] [PubMed] [Google Scholar]
- 32.Booysen HL, Woodiwiss AJ, Raymond A, et al. Chronic kidney disease epidemiology collaboration-derived glomerular filtration rate performs better at detecting preclinical end-organ changes than alternative equations in black Africans. J Hypertens. 2016;34(6):1178–1185. doi: 10.1097/HJH.0000000000000924. [DOI] [PubMed] [Google Scholar]
- 33.Earley A, Miskulin D, Lamb EJ, Levey AS, Uhlig K. Estimating equations for glomerular filtration rate in the era of creatinine standardization: a systematic review. Ann Intern Med. 2012;156(11):785–795. W–270, W–271, W–272, W–273, W–274, W–275, W–276, W–277, W–278. doi: 10.7326/0003-4819-156-11-201203200-00391. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1.
The respectively calculated SDs of eGFR values for the logistic regression
| Total (n=1,586)
|
IMT (n=1,579)
|
ABI (n=1,553)
|
LVMI (n=1,577)
|
UACR (n=1,537)
|
Aortic PWV (n=1,533)
|
|---|---|---|---|---|---|
| Equations | SD | SD | SD | SD | SD |
| MDRD | 20.5683668 | 20.5557993 | 20.5548362 | 20.4855702 | 20.5961544 |
| CKD-EPI | 14.2426876 | 14.2090751 | 14.2406751 | 14.1873433 | 14.1307789 |
| aCKD-EPI | 18.0208154 | 18.0347565 | 18.0355388 | 17.9665128 | 17.9131654 |
| cCKD-EPI | 14.9272658 | 14.9582379 | 14.9289286 | 14.8616948 | 14.9174066 |
| c-aMDRD | 24.4979976 | 24.5138248 | 24.4881662 | 24.3729941 | 24.5445129 |
Abbreviations: SD, standard deviation; eGFR, estimated glomerular filtration rate; IMT, intima–media thickness; ABI, ankle–brachial index; LVMI, left ventricular mass index; UACR, urine albumin to creatinine ratio; PWV, pulse wave velocity; MDRD, Modification of Diet in Renal Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD.
Table S2.
Assessment of the discrimination abilities of adjusted logistic models to detect the cardiovascular TOD
| Total (n=1,586)
|
Carotid hypertrophy (n=1,579)
|
Lower limb atherosclerosis (n=1,553)
|
Cardiac hypertrophy (n=1,577)
|
Microalbuminuria (n=1,537)
|
Arterial stiffening (n=1,533)
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| Equations | C-statistic (95% CI) | P-value | C-statistic (95% CI) | P-value | C-statistic (95% CI) | P-value | C-statistic (95% CI) | P-value | C-statistic (95% CI) | P-value |
| MDRD | 0.618 (0.543, 0.693) |
<0.001 | 0.640 (0.604, 0.676) |
<0.001 | 0.685 (0.656, 0.713) |
<0.001 | 0.738 (0.657, 0.819) |
<0.001 | 0.709 (0.682, 0.736) |
<0.001 |
| CKD-EPI | 0.613 (0.536, 0.690) |
0.002 | 0.647 (0.611, 0.682) |
<0.001 | 0.686 (0.657, 0.714) |
<0.001 | 0.747 (0.667, 0.828) |
<0.001 | 0.723 (0.696, 0.749) |
<0.001 |
| aCKD-EPI | 0.615 (0.538, 0.692) |
0.002 | 0.647 (0.612, 0.681) |
<0.001 | 0.685 (0.656, 0.713) |
<0.001 | 0.729 (0.646, 0.812) |
<0.001 | 0.707 (0.680, 0.734) |
<0.001 |
| cCKD-EPI | 0.624 (0.550, 0.699) |
<0.001 | 0.649 (0.614, 0.684) |
<0.001 | 0.685 (0.656, 0.714) |
<0.001 | 0.724 (0.643, 0.805) |
<0.001 | 0.713 (0.685, 0.740) |
<0.001 |
| c-aMDRD | 0.620 (0.545, 0.694) |
<0.001 | 0.644 (0.608, 0.680) |
<0.001 | 0.685 (0.656, 0.713) |
<0.001 | 0.736 (0.654, 0.817) |
<0.001 | 0.707 (0.680, 0.734) |
<0.001 |
Notes: Adjusted confounders: SBP, smoking, BMI and DM for carotid hypertrophy, lower limb atherosclerosis, microalbuminuria and arterial stiffening and sex, SBP, smoking, BMI and DM for cardiac hypertrophy. Considering an increased IMT (IMT ≥0.9 mm) as carotid hypertrophy, a lower ABI (≤0.9) as lower limb atherosclerosis, left ventricular hypertrophy (>115 g/m2 for males and >95 g/m2 for females) as cardiac hypertrophy, an increased UACR (>30 mg/g) as microalbuminuria and an increased PWV (>10 m/s) as arterial stiffening.
Abbreviations: TOD, target organ damage; MDRD, Modification of Diet in Renal Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD; SBP, systolic blood pressure; BMI, body mass index; DM, diabetes mellitus; IMT, intima–media thickness; ABI, ankle–brachial index; UACR, urine albumin to creatinine ratio; PWV, pulse wave velocity.
Table S3.
Goodness-of-fit among the different models
| Total (n=1,586)
|
Carotid hypertrophy (n=1,579)
|
Lower limb atherosclerosis (n=1,553)
|
Cardiac hypertrophy (n=1,577)
|
Microalbuminuria (n=1,537)
|
Arterial stiffening (n=1,533)
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| Equations | AIC | P-value | AIC | P-value | AIC | P-value | AIC | P-value | AIC | P-value |
| MDRD | 521.247 | ns | 1,421.704 | <0.01 | 1,736.528 | ns | 302.080 | ns | 1,711.694 | <0.01 |
| CKD-EPI | 521.392 | ns | 1,411.528 | Reference | 1,737.047 | ns | 299.038 | Reference | 1,690.964 | Reference |
| aCKD-EPI | 520.530 | ns | 1,415.310 | ns | 1,732.407 | Reference | 303.961 | ns | 1,707.235 | <0.01 |
| cCKD-EPI | 519.782 | Reference | 1,415.070 | ns | 1,734.270 | ns | 306.967 | 0.02 | 1,701.476 | <0.01 |
| c-aMDRD | 521.030 | ns | 1,419.735 | 0.02 | 1,738.172 | ns | 303.390 | ns | 1,713.643 | <0.01 |
Abbreviations: AIC, Akaike information criterion; MDRD, Modification of Diet in Renal Disease; ns, not significant; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD.
Table S4.
List of the eGFR equations in our study
| Equations | eGFR (mL/min/1.73 m2) | Participant | Race | Reference |
|---|---|---|---|---|
| MDRD | 186× Scr−1.154× age−0.203×0.742 (if female) ×1.212 (if black) | Middle aged CKD patients Age =50.6±12.7 years1 |
Black and White | Group TMoDiRDS2 |
| c-aMDRD | 175× Scr−1.234× age−0.179×0.79 (if female) | Middle aged CKD patients Age =49.9±15.8 years |
Chinese | Collaboration CeI3 |
| CKD-EPI | Female & Scr ≤0.7: 144 (166 if black) × (Scr/0.7)−0.329× (0.993)age Female & Scr >0.7: 144 (166 if black) × (Scr/0.7)−1.209× (0.993)age Male & Scr ≤0.9: 141 (163 if black) × (Scr/0.9)−0.411× (0.993)age Male & Scr >0.9: 141 (163 if black) × (Scr/0.9)−1.209× (0.993)age |
Middle aged CKD patients Age =47±15 years |
Black and White or other | Levey et al4 |
| aCKD-EPI | Female & Scr ≤0.7: 151× (Scr/0.7)−0.328× (0.993)age Female & Scr >0.7: 151× (Scr/0.7)−1.210× (0.993)age Male & Scr ≤0.9: 149× (Scr/0.9)−0.412× (0.993)age Male & Scr >0.9: 149× (Scr/0.9)−1.210× (0.993)age |
Middle aged CKD patients Age =49±15 years |
Asian (675 Chinese and 248 Japanese) | Stevens et al5 |
| cCKD-EPI | Female & Scr ≤0.7: 94× (Scr/0.7)−0.511× (0.998)age Female & Scr >0.7: 128× (Scr/0.7)−0.543× (0.992)age Male & Scr ≤0.9: 117× (Scr/0.9)−0.277× (0.994)age Male & Scr >0.9: 102× (Scr/0.9)−0.558× (0.994)age |
T2DM CKD patients Age =61.0±12.7 years |
Chinese | Liu et al6 |
Abbreviations: eGFR, estimated glomerular filtration rate; MDRD, Modification of Diet in Renal Disease; CKD, chronic kidney disease; c-aMDRD, Chinese-abbreviated MDRD; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI.
Table S5.
Crude relations between equation-eGFR and cardiovascular TOD indicators
| Equations | IMT (n=1,579)
|
ABI (n=1,553)
|
LVMI (n=1,577)
|
UACR (n=1,537)
|
Aortic PWV (n=1,533)
|
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| β | P-value | β | P-value | β | P-value | β | P-value | β | P-value | |
| MDRD | −0.005 | 0.84 | 0.11 | <0.001 | −0.06 | 0.01 | −0.07 | 0.004 | −0.15 | <0.001 |
| CKD-EPI | −0.03 | 0.19 | 0.15 | <0.001 | −0.09 | <0.001 | −0.11 | <0.001 | −0.22 | <0.001 |
| aCKD-EPI | −0.10 | <0.001 | 0.12 | <0.001 | −0.09 | <0.001 | −0.07 | 0.005 | −0.17 | <0.001 |
| cCKD-EPI | −0.08 | 0.002 | 0.10 | <0.001 | −0.07 | 0.003 | −0.06 | 0.03 | −0.15 | <0.001 |
| c-aMDRD | −0.02 | 0.41 | 0.10 | <0.001 | −0.06 | 0.01 | −0.07 | 0.009 | −0.13 | <0.001 |
Note: Crude linear regression was performed without adjustment.
Abbreviations: eGFR, estimated glomerular filtration rate; TOD, target organ damage; IMT, intima-media thickness; ABI, ankle–brachial index; LVMI, left ventricular mass index; UACR, urine albumin to creatinine ratio; PWV, pulse wave velocity; MDRD, Modification of Diet in Renal Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; aCKD-EPI, Asian-modified CKD-EPI; cCKD-EPI, Chinese-modified CKD-EPI; c-aMDRD, Chinese-abbreviated MDRD.