Abstract
Remotely triggered drug delivery devices have recently been realized as injectable or implantable formulations that enable the patient or physician to control the timing and dose of drug release over an extended period of time. Such devices could increase patient compliance, maximize therapeutic effectiveness, and minimize side effects. They can be triggered by near-infrared (NIR) light, which can harmlessly and painlessly pass through tissue at controlled doses and exposure times. We will discuss the use of NIR-triggered devices and materials, with examples. Safety issues and future prospects will also be addressed.
Introduction
There has recently been a great deal of interest in remotely triggerable drug delivery systems in which a depot of drug is administered once, then repeatedly activated via a painless external trigger such as an electromagnetic field or ultrasound. Such systems could give patients control over their therapeutic regimen.
Near-infrared (NIR) light is emerging as a useful tool in clinical care and is a promising method of triggering drug delivery systems. One major reason for the appeal of NIR light is that it can penetrate up to 10 cm into soft tissue due to hemoglobin and water having their lowest absorption coefficients in that regime1. NIR systems have already been used clinically for monitoring blood oxygenation levels within the body2, deep-tissue fluorescent imaging3 or cancer therapy by hyperthermia.4 Preclinical NIR-activated drug delivery systems have been widely studied, particularly those that function by heating a thermosensitive component of that system through inductive coupling to gold nanomaterials5. Unlike electronic devices, those systems do not require an onboard power supply, and so they can be realized as injectable materials and/or fully bioresorbable formulations.
NIR-triggered devices of that type have been demonstrated. For example, we developed a drug filled NIR-triggered capsule capped with a nanocomopsite membrane containing gold nanoshells and an interconnected network of pNIPAm nanoparticles.6 NIR irradiation heated the nanoshells causing shrinkage of the pNIPAm, which increased the permeability of the membrane and resulted in drug release (Fig. 1A). Devices loaded with aspart, a fast-acting analog of insulin could provide sustained or pulsatile drug release, depending on the timing of the NIR irradiation (Fig. 1B and 1C). They were designed for in vivo use by engineering the maximal release rate – achieved with pNIPAm in the fully collapsed state – to approximately 2 U/hr by tuning the geometry of the membrane and the concentration of nanomaterials within. These devices were implanted subcutaneously in diabetic rats and triggered with 30-min NIR laser pulses, which achieved glycemic reduction with a magnitude controlled by the intensity of the light6.
Figure 1.
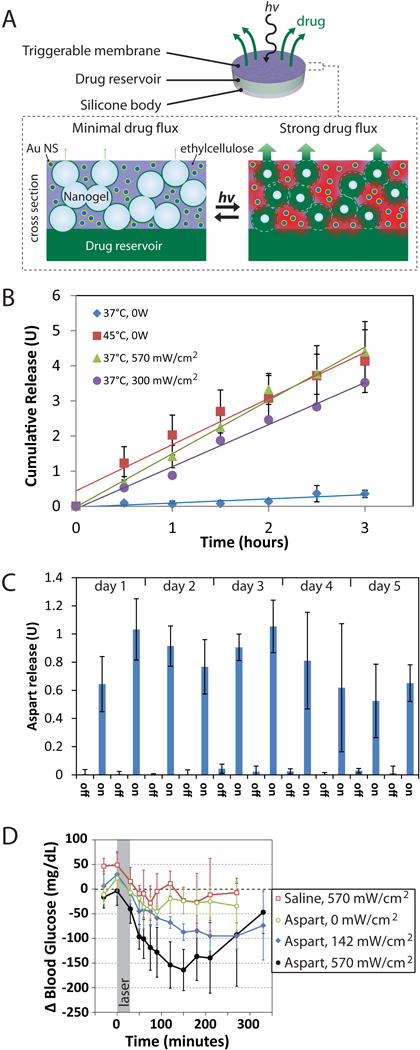
(A) Schematic of a device and cross-section of the membrane portion. (B) Sustained release of aspart from a typical device with triggering by applying heat or by laser irradiation. (C) Pulsatile release from the same type of device using 30-minute laser pulses (continuous wave, 808 nm, 570 mW/cm2). (D) Effect of irradiation on blood glucose levels in diabetic Sprague-Dawley rats. Adapted from ref. 6.
In the preceding example, the device was used for systemic drug delivery. Similar devices could be used for local drug delivery, e.g. for chronic pain. Sustained release formulations containing local anesthetics and adjuvant molecules have been used to achieve prolonged duration nerve blockade lasting days or weeks in vivo7. Those formulations have the limitation that once initiated, nerve blockade proceed relatively monotonically until the drug content of the device is depleted. The timing and intensity cannot be modulated to account for changes in daily activity or level of pain. In contrast, remotely triggered formulations allow the patient to adjust the timing and magnitude of the dose of local anesthetic delivered.
Nanoparticle-based systems can be useful for triggered localized delivery even when delivered systemically, since they can be targeted to a particular region of the body8, or selectively activated by aiming the light at the anatomical region of interest9. Moreover, systemically administered nanoparticles with appropriate dimensions can accumulate in tumors due to the enhanced permeability and retention effect (EPR)8, where they can be activated after they have reached their destination (Fig. 2A). Gold nanocages coated with poly(n-isopropyl methacrylamide) (pNIPAm) represent one such system; drug was loaded into the interior of the cage, and released when NIR collapsed the polymer shell (Fig. 2B). The release rate of drug was correlated to the intensity of irradiation (Fig. 2C)10.
Figure 2.
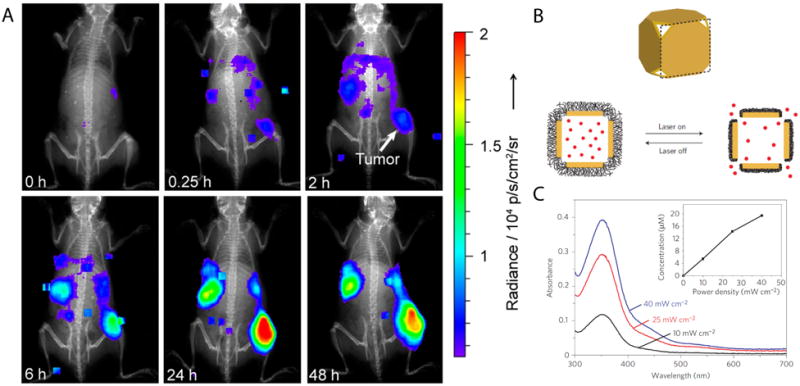
(A) Luminescence imaging of whole mice showing accumulation of PEG-coated radioluminescent 198Au nanoshells at tumor sites. (B) Schematic of gold nanocage controlled-release system. Upon NIR irradiation, the pNIPAm shell (black) collapses so that the drug (red) is released. (C) Release of model drug after triggering for 2 minutes at various irradiances. Adapted from refs. 8 and 10.
Materials for NIR-triggered release
NIR light cannot directly achieve many of the photo-initiated reactions (e.g. isomerization, bond cleavage, caging/uncaging) that can be performed by light with shorter wavelengths and higher energies, such as UV light. Nonetheless, it can be used to trigger drug delivery systems by a variety of approaches.
One commonly used means of rendering systems responsive to NIR light is to incorporate gold nanoparticles (AuNPs), which inductively couple with light – either from a pulsed or continuous wave source – to produce heat. That heat can be used to release a surface-bound ligand, or to trigger another physical process, such as disruption of a temperature-sensitive liposome or collapse of a polymer. As in the example above, AuNPs have been used in conjunction with pNIPAm-based hydrogels that become hydrophobic when heated beyond a critical temperature, and collapse reversibly. This approach was first demonstrated by incorporating the gold nanoshells directly within the pNIPAm, which also behaved as a reservoir for small molecules or proteins. NIR light caused the nanocomposite to collapse, expelling the biomolecules contained within. That system could also be triggered over multiple cycles, although most of the drug was released after the first activation11.
AuNPs can be rationally synthesized with absorption maxima within the NIR-range, and so can be designed to match the wavelength of a given triggering light source for maximum efficiency. Such tunable optical properties also enable the design of drug delivery systems where the release of two or more drugs is controlled independently by irradiation with different wavelengths of light corresponding to the non-overlapping spectra of distinct populations of AuNPs6.
NIR-triggered drug delivery systems based on AuNPs must contain a heat-sensitive element – either a polymer, the drug itself, or a linker that tethers the drug to the surface of the particle5. An alternative approach involves upconverting nanoparticles, which emit light at a shorter wavelength (higher energy) than the excitation wavelength (anti-Stokes emission). The energy levels of these particles, which are typically composed of NaYF4 doped with trivalent lanthanides (e.g. Yb3+, Tm3+, Er3+, Ho3+), can be tuned so that they absorb NIR and emit visible or UV light. The UV light might be used to activate a protein caged by a photo-labile ligand9, to photoisomerize a polymer so that it deforms and releases a drug, or to convert a pro-drug into its active form12. In one recent study, NIR light was used in vivo to target doxorubicin-loaded upconversion nanoparticles to the site of a tumor. The particles were functionalized with caged folic acid ligands; the NIR light was upconverted to UV emission, releasing the cages and thus allowing the particles to bind to folate receptors on the surface of the tumor.9
Polymer-based systems have been developed that respond to NIR without the need for gold or upconverting nanoparticles. PLGA microparticles released drug when irradiated with 980 nm light, presumably because that wavelength heated nanodomains of water within the polymer, causing a thermally-induced phase change13. Molecules have also been developed that undergo chemical changes by two-photon absorption, where energies of two simultaneously-absorbed NIR photons are combined. Triggered delivery by that process has been achieved by encapsulating molecules with NIR-sensitive polymers that are either disrupted or dissolved with irradiated with NIR12.
Safety
The safety of the NIR light itself is of concern, as it may cause burns at sufficiently high powers and/or irradiation times6. This concern is particularly apposite in patient-controlled devices, where the patient may choose to activate the device repeatedly. Materials for drug delivery should be designed so that the irradiance required to fully activate the device is minimized, particularly in the case of systems that will be placed deep within tissues, which will absorb or scatter a substantial portion of the light.
As with all drug delivery systems14, the biocompatibility of NIR-triggered systems is important. Many such systems may cause localized heating of photoresponsive materials and perhaps of surrounding tissues, which may be harmful. Moreover, the biocompatibility, biodistribution, and other biological parameters of some materials – including nanomaterials – may be ill-defined.
Expert Opinion
NIR triggered drug delivery systems could enhance efficacy, reduce side effects, and increase patient compliance. They can be realized as either implantable devices or injectable formulations, and could therefore find use throughout the body provided that a light source can reach them without being so attenuated by interposing tissue that it is ineffective or so powerful that tissue is injured. Since NIR-triggered devices can in principle release drugs in any temporal profile – pulsatile, sustained, crescendo, etc. – they enable dosing regiments that are not achievable by conventional means. They could also enable localized release, which may be useful for indications requiring drugs that are effective at high regional concentrations, but are systemically toxic.
NIR sources will need to be engineered for point-of-care use. Pulsed laser systems can deliver a large amount of energy to the device while causing minimal tissue damage, and are generally required in order to activate polymers by 2-photon absorption12. However, those systems will most likely be confined to the clinic because of their bulk, sophistication and cost. Continuous wave diode lasers can be purchased for <$1,000, are small enough to be hand-held, and are safe so long as the exposure time and irradiance are maintained below established limits15. NIR-range LEDs are an alternative, and are potentially safer than lasers since the light that they produce is not collimated, although adequate heat dissipation is an issue that would need to be addressed3. LEDs might eventually be incorporated into wearable devices that produce irradiation sequences controlled by the patient, a preprogrammed regimen, or sensors that enable closed-loop feedback control.
Acknowledgments
This research was funded by National Institutes of Health (NIH) Grant GM073626 (to D.S.K.), a Sanofi Biomedical Innovation Funding Award through the MIT Center for Biomedical Innovation (to D.S.K.), and a Ruth L. Kirschstein National Research Service Award fellowship, NIH Award F32GM096546 (to B.P.T.).
Bibliography
- 1.Weissleder R. A clearer vision for in vivo imaging. Nature Biotechnology. 2001 Apr;19(4):316–7. doi: 10.1038/86684. [DOI] [PubMed] [Google Scholar]
- 2.Boushel R, Langberg H, Olesen J, Gonzales-Alonzo J, Bulow J, Kjaer M. Monitoring tissue oxygen availability with near infrared spectroscopy (NIRS) in health and disease. Scand J Med Sci Sports. 2001 Aug;11(4):213–22. doi: 10.1034/j.1600-0838.2001.110404.x. [DOI] [PubMed] [Google Scholar]
- 3.Gioux S, Choi HS, Frangioni JV. Image-Guided Surgery Using Invisible Near-Infrared Light: Fundamentals of Clinical Translation. Molecular Imaging. 2010 Sep-Oct;9(5):237–55. [PMC free article] [PubMed] [Google Scholar]
- 4.Kennedy LC, Bickford LR, Lewinski NA, Coughlin AJ, Hu Y, Day ES, et al. A New Era for Cancer Treatment: Gold-Nanoparticle-Mediated Thermal Therapies. Small. 2011 Jan 17;7(2):169–83. doi: 10.1002/smll.201000134. [DOI] [PubMed] [Google Scholar]
- 5.Timko BP, Dvir T, Kohane DS. Remotely Triggerable Drug Delivery Systems. Advanced Materials. 2010 Nov 24;22(44):4925–43. doi: 10.1002/adma.201002072. [DOI] [PubMed] [Google Scholar]
- 6.Timko BP, Arruebo M, Shankarappa SA, McAlvin JB, Okonkwo OS, Mizrahi B, et al. Near-infrared-actuated devices for remotely controlled drug delivery. Proceedings of the National Academy of Sciences of the United States of America. 2014 Jan 28;111(4):1349–54. doi: 10.1073/pnas.1322651111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McAlvin JB, Kohane DS. Prolonged Duration Local Anesthesia. In: Domb AJ, Khan W, editors. Focal Controlled Drug Delivery. New York: Springer; 2014. [Google Scholar]
- 8.Wang YC, Liu YJ, Luehmann H, Xia XH, Wan DH, Cutler C, et al. Radioluminescent Gold Nanocages with Controlled Radioactivity for Real-Time in Vivo Imaging. Nano Letters. 2013 Feb;13(2):581–5. doi: 10.1021/nl304111v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chien YH, Chou YL, Wang SW, Hung ST, Liau MC, Chao YJ, et al. Near-infrared light photocontrolled targeting, bioimaging, and chemotherapy with caged upconversion nanoparticles in vitro and in vivo. ACS Nano. 2013 Oct 22;7(10):8516–28. doi: 10.1021/nn402399m. [DOI] [PubMed] [Google Scholar]
- 10.Yavuz MS, Cheng YY, Chen JY, Cobley CM, Zhang Q, Rycenga M, et al. Gold nanocages covered by smart polymers for controlled release with near-infrared light. Nature Materials. 2009 Dec;8(12):935–9. doi: 10.1038/nmat2564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sershen SR, Westcott SL, Halas NJ, West JL. Temperature-sensitive polymer-nanoshell composites for photothermally modulated drug delivery. Journal of Biomedical Materials Research. 2000 Sep 5;51(3):293–8. doi: 10.1002/1097-4636(20000905)51:3<293::aid-jbm1>3.0.co;2-t. [DOI] [PubMed] [Google Scholar]
- 12.Tong R, Kohane DS. Shedding light on nanomedicine. Wiley Interdisciplinary Reviews-Nanomedicine and Nanobiotechnology. 2012 Nov-Dec;4(6):638–62. doi: 10.1002/wnan.1188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Viger ML, Sheng W, Dore K, Alhasan AH, Carling CJ, Lux J, et al. Near-Infrared-Induced Heating of Confined Water in Polymeric Particles for Efficient Payload Release. ACS Nano. 2014 Apr 10; doi: 10.1021/nn500702g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kohane DS, Langer R. Biocompatibility and drug delivery systems. Chemical Science. 2010 Oct 1;1(4):441–6. [Google Scholar]
- 15.America ANSIaLIo. American National Standard for Safe Use of Lasers. Orlando, FL: Laser Institute of America; 2000. [Google Scholar]


