Abstract
Sterosomes are a recently developed type of non-phospholipid liposomes formed from single-chain amphiphiles and high content of sterols. Although sterosomes presented significantly increased stability compared to conventional phospholipid liposomes, current sterosome biomaterials are not truly bioactive and have no intrinsic therapeutic effects. The purpose of this study was to develop a sterosome formulation with osteoinductive properties by an effective selection of sterol, one of the sterosome components. Oxysterols are oxidized derivatives of cholesterol and are known to stimulate osteogenesis and bone formation. Thus, 20S-hydroxycholesterol (Oxy), one of the most potent oxysterols for bone regeneration was examined as a promising candidate molecule to form fluid lamellar phases with a single-chain amphiphile, namely stearylamine (SA). First, the optimal composition was identified by investigating the phase behavior of SA/Oxy mixtures. Next, the capacity of the optimized SA/Oxy sterosomes to promote osteogenic differentiation of bone marrow stromal cells was assessed in vitro in a hydrogel environment. Furthermore, we explored the effects of osteogenic oxysterol sterosomes in vivo with the mouse critical-sized calvarial defect model. Our results showed that SA/Oxy sterosomes induced osteogenic differentiation in vitro and enhanced calvarial healing without delivery of additional therapeutic agents, indicating their intrinsic bone forming potential. This study suggests a promising non-phospholipid liposomal platform with osteoinductive properties for delivery of small molecular drugs and/or other therapeutic genes for enhanced bone formation.
Keywords: Oxysterol, Stearylamine, Sterosomes, Mesenchymal stem cells, Osteogenic differentiation
TOC Image
Over the years, numerous efforts have been devoted to design and create new functional materials to formulate drug delivery vehicles, including organic, inorganic, composite and polymers. Among various delivery vectors, including liposomes,1, 2 polymersomes,3, 4 niosomes,5 micelles,6 hydrogels,7 dendrimers,8, 9 etc., liposomes are one of the most successful10 in the commercial market, because of their simplicity, biocompatibility, biodegradability, the enhancement on drug efficacy, etc. There are 12 liposomal formulations that have been approved for clinical applications as of 2012, including Doxil®, Caelyx®, Myocet®, DaunoXome®, Visudyne®, Ambisome®, DepoDur®, loading therapeutic agents, such as doxorubicin, daunorubicin, amphotericin B, verteporfin, or morphine, to enhance their delivery to the targeted tissue.
Liposomal nanotechnology has significantly evolved since the first discovery of liposomes in 1964 by Alec D. Bangham.11 Liposomes have been widely used in various fields including pharmaceutical,1 cosmetic,12 food,13 and textile14 industries. Naturally occurring lipids such as phospholipids are the most widely used molecules that self-assemble into bilayers in the presence or absence of cholesterol (Chol). However, more extensive use of conventional phospholipid liposomes is limited due to their poor stability. Recently, a novel class of liposomes formed from non-phospholipid molecules, named sterosomes, was formulated with single-chain amphiphiles and high content of sterols.15 The mixtures of these single-chain amphiphile and sterol molecules can lead to highly stable liquid ordered (lo) bilayers, which may not occur if the molecules are hydrated individually. For example, a cationic amphiphile such as stearylamine (SA) mixed with Chol at equimolar ratio forms stable unilamellar vesicles with high positive surface charges and very low permeability.16 These sterosomes show a great potential for delivery of drugs or therapeutic genes because of their increased stability. Moreover, they show a superior gene knockdown efficiency compared to conventional phospholipid liposomes, as shown in our recent study.17
Although traditional liposomes are formed with lipids that are inherently biocompatible, very few liposomal biomaterials are truly bioactive and have no intrinsic therapeutic effects (e.g., osteoinductive). Thus, there is a need to develop advanced liposomes that would be more than nanocontainers and also would display targeted therapeutic functionalities. This path has been explored by selecting a sterol component that has osteoinductive properties. Chol was found to have positive effects on osteogenic differentiation in murine mesenchymal stem cells.18, 19 However, the potency of its effects is limited. Oxysterols are naturally occurring Chol oxidation products,20, 21 and have been implicated in many physiologic and pathological processes including Chol metabolism,22 apoptosis,23 inflammation and immunosuppression,24 lipid trafficking,25 and atherosclerosis.21 Possible role of oxysterols in cellular differentiation has been well documented. For instance, the oxysterols 7α, 27- and 7β, 27-dihydroxycholesterol promote the differentiation of mouse and human immune cells;26 25-hydroxycholesterol not only induces the differentiation of human keratinocytes,27 but also inhibits enveloped viruses in cultured cells;28 and 20S-hydroxycholesterol (Oxy) drives the differentiation of osteogenic cells in culture and in animals.29, 30 In addition, it was reported that oxysterols, such as Oxy, activated the Hedgehog signaling to enhance osteogenesis through smoothened binding,31, 32 while inhibiting peroxisome proliferator activated receptor-γ (PPARγ) expression and adipogenic differentiation.33 These oxysterols are good candidate sterols for the formation of nanovectors based on lo bilayers made with single chain amphiphiles.34
In this present work, we report a formulation of liposomes with osteoinductive properties using Oxy and SA, a primary amine with an 18-carbon chain, for potential applications in bone regeneration. This sterol component, an analogue of Chol with an extra hydroxyl group on carbon 20, would not only increase the stability of the nanovectors, but also confer a functional surface of the drug delivery vehicle promoting anabolic mineralization (Figure 1A). SA was chosen because it provides a good hydrophobic match with the sterol, and a charged interface, two prerequisites to form lo phase from mixtures of single-chain amphiphile and sterol.15 First, we determined the optimal composition of SA/Oxy mixtures by investigating their phase behavior. Next, the ability of the optimized liposome formulation to enhance osteogenic differentiation of bone marrow stromal cells (BMSCs) was assessed in hydrogels. Lastly, we determined that the SA/Oxy liposomes could contribute to the healing of critical-sized mouse calvarial defects. Herein we demonstrate a simple promising liposomal formulation tailored for applications toward treating bone defects.
Figure 1.
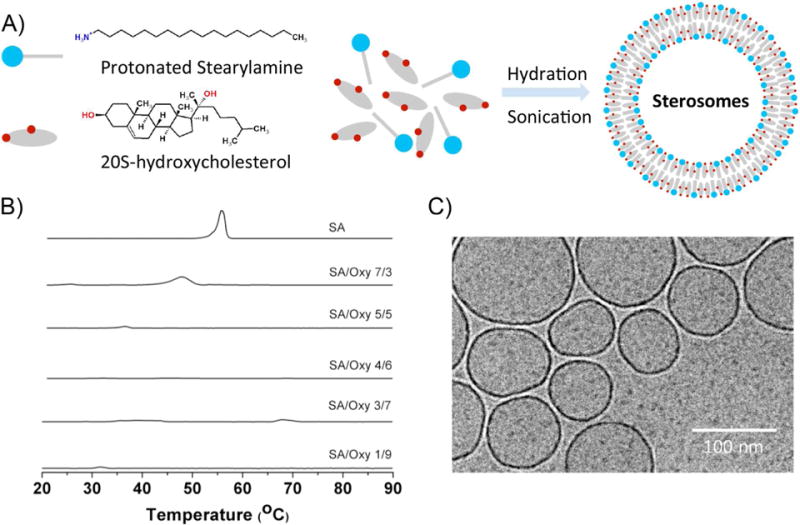
Characterization of SA/Oxy mixtures and their derived liposomes. (A) Schematic illustration of SA and osteoinductive oxysterol molecules and their self-assembled sterosome. (B) Thermograms of hydrated pure SA and of SA/Oxy mixtures of various molar ratios, pH 7.4. (C) Cryo-TEM image of freshly prepared SA/Oxy 4/6 liposomes.
RESULTS AND DISCUSSION
Characterization of SA/Oxy mixtures
SA mixed in equimolar proportion with Chol forms stable bilayers when pH is below the pKa of its amine group, i.e. when the polar head group carries a positive charge.16 It was established that the molecular details of sterols have a strong impact on their propensities to form fluid bilayers with single-chain amphiphiles.15, 34 Therefore, the first step of the work was to assess the nature of the self-assemblies formed by mixtures of SA and Oxy. The thermal behavior of various SA/Oxy mixtures was examined with differential scanning calorimetry (DSC). The thermograms of these mixtures are presented in Figure 1B. For pure SA, a sharp peak appeared at around 55°C, corresponding to the melting of the amphiphile. A shift of the peak towards low temperatures and an increase of its width were observed in the presence of up to 50 mol% Oxy. When SA/Oxy molar ratio was 4/6, no peaks could be observed in the thermogram, suggesting the absence of transition over the temperature range investigated (20 – 90°C). With increasing the proportion of Oxy, small endothermic peaks reappeared in the thermograms. They could correspond to solid-solid transitions of an excess amount of Oxy, as previously observed for Chol.35 Such thermal behavior is reminiscent of that observed for several single-chain amphiphile/sterol systems, including SA/Chol mixtures.15, 16 In order to provide a description of the chain conformational order of SA in these self-assemblies, infrared spectra were recorded (Figure S1). The position of the C–H symmetric stretching mode ν(CH) is mainly influenced by the trans-gauche isomerization along the alkyl chain.36, 37 The ν(CH) band for pure SA appeared at 2850 cm−1 below 50°C, a position indicative of ordered chains, and upshifted to 2853 cm−1 at 55°C, reflecting the disordering of the chain occurring upon SA melting.16 The addition of Oxy (30 and 60 mol%) led to the disappearance of the abrupt shift of the ν(CH) band position; instead there was a small and progressive shift of the band that was around 2851, and 2850 cm−1 for SA/Oxy molar ratios of 7/3 and 4/6, respectively. It was inferred that the SA alkyl chain in these mixtures remained ordered over the whole temperature range, a behavior similar to that of SA/Chol system.16 This behavior is consistent with the DSC results. It was therefore hypothesized that SA/Oxy 4/6 mixture formed a lo phase, without excess of SA or Oxy; similar-single chain amphiphile/sterol molar ratios also led to lo phase bilayers in several analogous sterosome systems.15 This SA/Oxy 4/6 suspension was therefore submitted to sonication to determine whether it was possible to form liposomes. Dynamic light scattering and Cryo-TEM confirmed the formation of liposomes. Their dynamic diameter was measured as 120 ± 3 nm with a narrow distribution (PDI 0.22 ± 0.02). The results were consistent with the observation on the TEM image (Figure 1C). Their zeta potential was +39.0 ± 2 mV, a result consistent with the protonated form of the primary amine at physiological pH. No significant changes were observed in size (Figure S2A), distribution (data not shown) and zeta potential over 14 days (Figure S2B), indicating a stable system, a feature that could facilitate the future translation to clinic development. Therefore, we have successfully created sterosomes that include one potent osteogenic component. Next, we further tested the osteogenic ability of these SA/Oxy sterosomes in vitro and in vivo.
Cytotoxicity of SA/Oxy sterosomes
Cationic liposomes tend to be cytotoxic, generating toxic reactive oxygen intermediates and disrupting the function of cellular and sub-cellular membranes.17 The protonation state of SA confers to the sterosomes a high positive surface charge, leading to a stable colloidal system38 but potentially toxic to living cells. The cytotoxicity of SA/Oxy sterosomes was evaluated on MSCs in hydrogels with Alamar blue assay and Live/Dead staining (Figure 2 & S3). No cytotoxicity on cell proliferation was observed up to 500 μg/mL of SA/Oxy vesicles.
Figure 2.
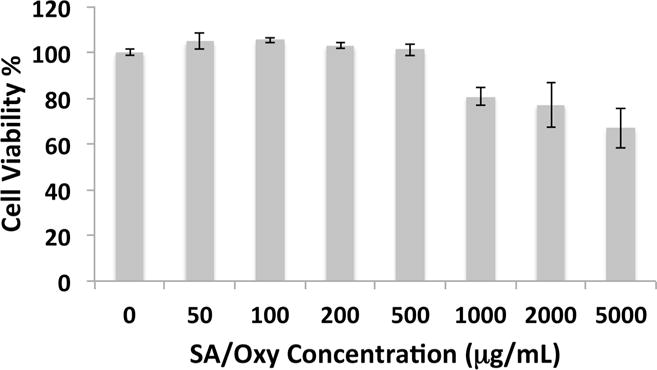
Cytotoxicity of SA/Oxy sterosomes evaluated with Alamar blue assay after 24 h incubation.
Osteogenic ability of SA/Oxy sterosome in vitro
We investigated the correlation between the concentrations of SA/Oxy sterosomes and their osteogenic capacity on MSCs in hydrogels, a system that provides a 3D mimicking environment. MeGC hydrogels are prepared from a naturally occurring polysaccharide under visible light irradiation with a blue light initiator, riboflavin (Vitamin B).39–42 This hydrogel system was devised to deliver stem cells or bioactive molecules in the defect site in a safe manner, minimizing potential adverse effects of UV exposure and toxic initiators. Figure 3 presents the ALP (day 7) and mineralization (day 28) staining, and corresponding quantification inferred from colorimetric assays. A gel without any encapsulated cells was used as a control to eliminate nonspecific staining. At day 7, the ALP staining intensified with increasing concentration of SA/Oxy sterosomes, up to 200 μg/mL. At 500 μg/mL, the staining intensity decreased. Similar trends were observed for mineralization staining on day 28. Progressively stronger red color appeared up to the dosage of 200 μg/mL sterosomes, followed by a decrease of intensity for 500 μg/mL of SA/Oxy sterosomes. The quantification of ALP expression and calcium deposition showed results consistent with the corresponding staining. The Pearson’s correlation test was performed to determine the correlation between SA/Oxy sterosome concentration and MSC ALP activity or mineralization in the range of 0–200 μg/mL. The correlation coefficients (r) were 0.952 and 0.989 for day 3 and 7 ALP activity, respectively. The r values were 0.908 and 0.947 for day 14 and 28 mineralization, respectively. These values demonstrated a strong positive correlation between the concentrations of SA/Oxy sterosomes and their osteogenic capacity on MSCs in hydrogels. Oxy is a known agonist of the nuclear hormone receptor liver X receptor (LXR). Activation of LXR does not induce but actually inhibits osteogenesis.43 Higher concentrations of SA/Oxy sterosomes may activate LXR, leading to decreased staining of ALP and calcium. The most potent concentration for osteogenesis in hydrogels was 200 μg/mL. This concentration reached the threshold of osteogenic ability of Oxy on MSCs in this setting.
Figure 3.
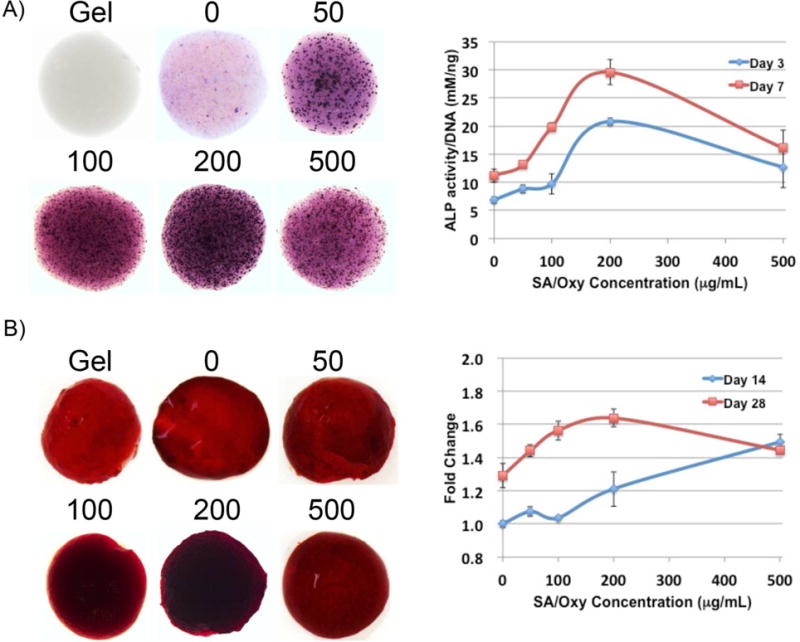
Bioactivity evaluation of SA/Oxy vesicles in hydrogels. (A) Alkaline phosphatase (ALP) expression of MSCs in hydrogels on day 7 and colorimetric quantification of ALP activity for day 3 blue, day 7 red. (B) Mineralization in hydrogels stained with Alizarin red S on day 28 and colorimetric quantification of mineralization for day 14 blue, day 28 red. (n = 4)
The expressions of three specific osteogenic genes were quantified with qRT PCR (Figure 4A). Since Chol exhibited limited osteogenic effects on MSCs,18 similar vesicles prepared from Chol (SA/Chol 5/5) optimized in our previous study16 were used as a positive control and gels encapsulating MSCs only set as a negative control. Three particular markers (ALP, an osteogenic marker in the early stage, Runx2, an osteoblast specific marker, and OCN, an osteogenic marker in the later stage) were monitored to assess the osteogenic ability of SA/Oxy sterosomes at a gene level. On day 7, ALP gene expression was significantly increased for the SA/Oxy group: it corresponded to ~1.8 fold increase compared to the SA/Chol group and ~3.3 fold increase in comparison with the negative control group. Runx2 gene level exhibited significant upregulation for the SA/Oxy group, ~1.6 to ~2.8 fold augmentation compared to the SA/Chol and control groups. On day 14, significant increase of OCN gene expression was observed for the SA/Oxy group as well, ~1.6 to ~2.5 fold increase compared to the SA/Chol and control groups. Taken altogether, SA/Oxy sterosomes presented considerable effects on osteogenic differentiation of MSCs.
Figure 4.
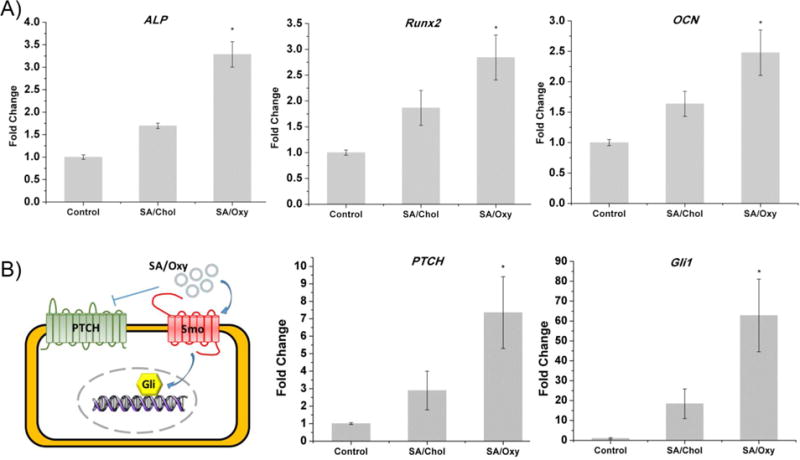
Gene expressions of MSCs in hydrogel culture with no liposomes (Control, negative control group) or in the presence of SA/Chol (positive control group) or SA/Oxy (experimental group) liposomes. Gene markers (A) ALP, Runx2 were evaluated on day 7 and OCN was evaluated on day 14. (B) Schematic illustration of hedgehog (Hh) signaling is presented, and PTCH, Gli1 were evaluated after 24 h incubation. (n = 5, *p < 0.05)
Oxysterol is known to induce osteogenesis through activation of the Hedgehog (Hh) pathway.31,32 Binding of Hh ligands to Patched (PTCH) or addition of small molecule agonists of Smoothened (Smo) such as Oxy promotes Smo activity, leading to activation of the Gli transcription factors. An illustration scheme was presented in Figure 4B. We have further investigated whether the SA/Oxy sterosome function for mediating osteogenesis was through activating Hh signaling by monitoring expression of PTCH1 and Gli1, Hh transcriptional target genes (Figure 4B). Both SA/Chol and SA/Oxy sterosomes increased the levels of PTCH and Gli1 mRNA in MSCs, indicating upregulation of Hh signaling activity. In particular, SA/Oxy sterosomes dramatically increased the gene expression.
Osteogenic ability of SA/Oxy sterosome in vivo
We further translated our osteogenic delivery vehicles to the in vivo calvarial defects model of mice for bone repair evaluation. Three groups (4 mice/group) with the 3-mm critical size defects were arranged. In the blank group, the created defect was left empty with no treatment. The other two groups were treated with hydrogels containing MSCs and SA/Chol or SA/Oxy sterosomes. Six weeks post-surgery, the size of the remaining defects was significantly smaller after treatment with MSCs and sterosomes compared to the blank control group. In Figure 5A, the SA/Oxy group led to the most effective bone healing. The relative new bone surface area, bone volume/tissue volume (BV/TV), and trabecular number (Tb. N. mm−1) were calculated from the μCT images. The normalization was conducted on the original 3 mm defect area (Figure 5B). The defects treated with SA/Chol and SA/Oxy sterosomes were covered by new bone at 31%, and 61%, respectively, whereas the defects without any treatment showed a minimal healing after 6 weeks (7%). The BV/TV and Tb. N. were up to 23% and 2.0 mm−1 for SA/Oxy group, significantly higher compared to the SA/Chol group (6% and 1.0) or the blank group (1% and 0.1).
Figure 5.
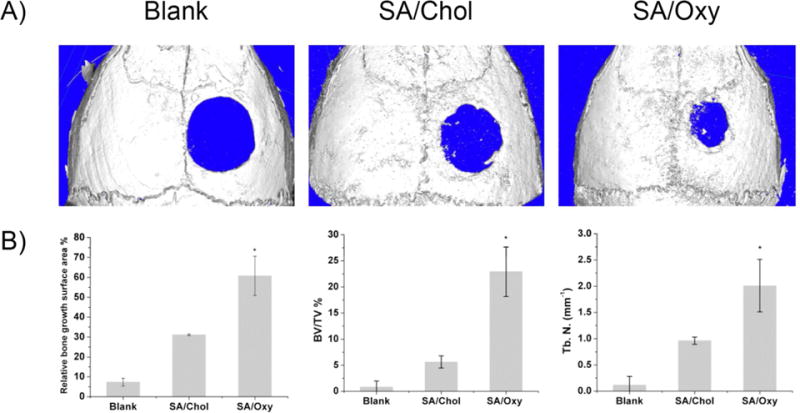
(A) Micro-computed tomography images of calvarial defects treated with hydrogels encapsulating MSCs and liposomes or left empty, six weeks post-surgery. (B) MicroCT quantification of bone regeneration in calvarial defects. Relative bone growth surface area, bone volume density (BV/TV%) and trabecular number (Tb. N. mm−1).
Histological evaluation was performed to further characterize the quality of new bone formation (Figure 6A). Hematoxylin-eosin (H&E) staining demonstrated that the edge of the defects treated with SA/Oxy sterosomes was filled with newly formed bone, and thick soft tissue connected the edges, 6 weeks post-surgery. The blank and SA/Chol groups exhibited very little bone tissue and very thin soft tissue connecting the defects. Masson trichrome staining demonstrated an osteoid matrix formed on the edge of defects treated with SA/Oxy sterosomes, while defects with the blank and SA/Chol groups were only filled with fibrous tissue with minimal bone regeneration. The distance of the defects became significantly shorter for the SA/Oxy group than that of the blank and SA/Chol groups. The relative length change was quantified with ImageJ (National Institutes of Health, Bethesda, MD). For SA/Chol and SA/Oxy groups, the distances of defects decreased by 10% and 50% in comparison with the blank group, respectively.
Figure 6.
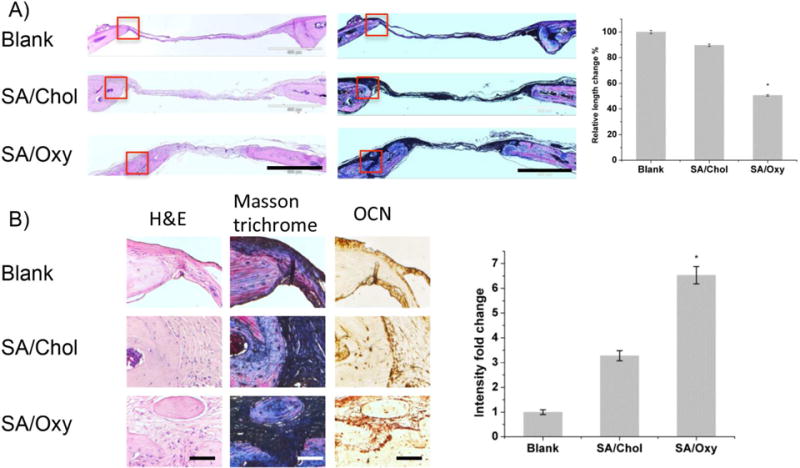
(A) Histological analysis of bone regeneration in calvarial defects, six weeks post-surgery. Hematoxylin-eosin (H&E) staining and Masson trichrome staining (scale bar = 500μm). The relative length change was measured and normalized to the blank group. (B) Magnified images of H&E and Masson trichrome staining (red boxes in A represent the magnified areas), and immunohistochemical staining of OCN (scale bar = 50 μm) and quantification of OCN staining with imageJ. (n = 4, *p < 0.05)
Moreover, we evaluated the OCN expression 6-week post-operative with immunohistochemical staining. The representative images are presented in Figure 6B. The results showed intensified staining for both SA/Chol and SA/Oxy groups. Highly intensified OCN staining (6.5-fold) was observed for SA/Oxy groups in comparison with the blank group, and 3.2-fold higher for SA/Chol group, after quantification with imageJ.
The SA/Oxy sterosomes demonstrated as a proof of concept for liposome itself being therapeutic. In a more general view, single-chain amphiphile/sterol liposomes formed a versatile platform as several therapeutic or targeting components can be included. For example, SA was chosen in the present work to confer the positive surface charges and maintain the stability of the vesicles; however a single-chain glucosamine may be a better amphiphile candidate to promote bone regeneration.44 Meanwhile, SA might be a good candidate to create a liposome surface that targets cancerous legions. It is reported that many types of cancer cells display an elevated amount of phosphatidylserine on the luminal surface.45 The addition of SA in phosphatidylcholine liposomes was shown to induce apoptosis in cancer cell lines with this single agent targeting phosphatidylserine. Therefore, without any additional targeting ligands, SA can target certain cancer cells by itself. Certainly, more efforts should be devoted to further understand this aspect. Liposomes that integrate intrinsic therapeutic benefits should play a key role in the development of the next generations of drug delivery nanocarriers. Sustained drug release profiles were observed with SA/Oxy liposomes by encapsulating sulforhodamine B (SRB) as a model drug (Figure S4). Potent drugs and therapeutic genes can be loaded into those liposomes to achieve synergistic effects in combined treatments in the future. In addition, the vesicle size can be readily controlled by liposome extrusion through filters with defined pore sizes.
CONCLUSIONS
In this work, we have developed functional liposomes with osteoinductive properties by including an osteogenic sterol, Oxy, as one of the liposomal molecules. As shown in the presented results, this formulation can induce osteogenic differentiation of MSCs to promote bone regeneration even without additional therapeutic agents, demonstrating its intrinsic bone-forming potential. The additional knowledge gained from this study may suggest nanocarrier design strategies encapsulating drugs into functional non-phospholipid bilayers to improve clinical efficacy of current therapeutic agents.
EXPERIMENTAL SECTION
Materials
Oxy (>95%) was supplied from R&D system Inc. (Minneapolis, MN). SA (99%), Chol (>99%), sodium chloride (>99%), 5-bromo-4-chloro-3-indoxylphosphate (BCIP), Nitro Blue Tetrazolium (NBT), Alizarin red S, L-ascorbic acid, p-nitrophenol phosphate, β-glycerophosphate, dexamethasone, methanol (spectrograde), benzene (high purity), Tween-20, SRB tris(hydroxymethyl)-aminomethane (TRIS) (99%), and EDTA were ordered from Sigma-Aldrich (St. Louis, MO). Trizol reagents and cDNA transcription kit, Fetal Bovine Serum (FBS), high Glucose DMEM, and penicillin/streptomycin (100 U/mL) (P/S) were purchased from Life Technologies (Grand Island, NY). RNeasy Mini kit was provided by Qiagen (Valencia, CA). C57/BL nude mice were purchased from Charles River Laboratories (Wilmington, MA). All solvents and products were used as received.
Sterosomes preparation
SA and sterols powders were mixed and dissolved in benzene/methanol (9:1 volume ratio). The mixtures were frozen in liquid N2 and lyophilized for 16 h to completely remove the organic solvent.17 The lyophilized lipid mixtures were hydrated in a TRIS buffer, containing 50 mM TRIS and 140 mM NaCl at pH 7.4. Five temperature cycles from liquid N2 temperature to ~70 °C with vortex in between, were carried out for ensuring good hydration. Afterwards, the pH was readjusted, if necessary, with diluted HCl or NaOH solution. Liposomes were prepared by sonication using a high power 500 watts sonic dismembrator (20 s on, 5 s off, 20% amplitude, 25 watts/cm2 power intensity) for 20 min. Liquid chromatography–mass spectrometry (LC-MS) was explored as quality control to detect the SA/Oxy ratios before and after liposome formation (Detailed method in SI). The size and the zeta potential of prepared liposomes were measured on a Malvern Zetasizer.
Thermal behavior characterization
Differential Scanning Calorimetry (DSC) analysis was conducted with a VP-DSC microcalorimeter (MicroCal, Northampton, MA). The TRIS buffer served as control. The thermograms were recorded from 20 to 90 °C, at a heating rate of 40 °C/h. Data acquisition and analysis were conducted with the Origin software (MicroCal software, Northampton, MA).
Infrared spectra were acquired on a Thermo Nicolet 4700 spectrometer, equipped with a DTGS alanine detector. An aliquot of the hydrated mixture was placed between a pair of CaF2 windows separated by a Teflon spacer that was 5 μm thick. This assembly was mounted in a brass sample holder and the temperature was controlled by Peltier elements. The spectra were recorded as a function of increasing temperature, with an equilibration period of 6 minutes. The nominal spectral resolution was 0.5 cm−1 and each spectrum was the results of 32 co-added interferograms.
Cryogenic transmission electron microscopy (Cryo-TEM)
An aliquot (2.5 μL) of the liposome sample was dripped on a Quantifoil grid (Quantifoil R2/1 100 holey carbon films grids, Cu 200 mesh), blotted for 10 s with an FEI vitrobot in 100% relative humidity, and then plunged directly into liquid ethane. The image was collected on a FEI Tecnai TF20 at an accelerating voltage of 200 kV using TVIPS EM-Menu program (defocus:−6.5 μm, dose: 3400 e/nm2). The instrument is equipped with a 16-megapixel CCD camera.
Cell culture in 3D hydrogels
Photocrosslinkable methacrylated glycol chitosan (MeGC) was prepared in accordance with an established protocol.39 The mouse bone marrow stromal cell line (BMSCs, D1 ORL UVA [D1], D1 cell, CRL-12424) was supplied from American Type Culture Collection (Manassas, VA). Sterosomes and BMSCs at a density of 2 × 106 cells/mL were mixed in 2% w/v MeGC solution. The suspension (40 μL) was mixed with a riboflavin photoinitiator (final concentration 6 μM) and exposed to blue light (400 ~ 500 nm and 500 ~ 600 mW/cm2, Bisco Inc., Schaumburg, IL). The resulting hydrogels were incubated in 1 mL of proper media.
Cytotoxicity
Cytotoxicity of SA/Oxy sterosomes was evaluated using Alamar blue assay and the Live/Dead staining kit (Invitrogen, Carlsbad, CA). Hydrogels with encapsulated BMSCs and SA/Oxy sterosomes in various concentrations were incubated in freshly prepared growth medium (DMEM, 10% FBS, 1% P/S) for 24 h. The medium was then replaced with Alamar blue solution (10% v/v) in growth medium. 3-h incubation later, the fluorescence intensity of Alamar blue was measured at 570/585 nm (excitation/emission). Hydrogels containing only BMSCs served as positive control for 100% viability. The 10% (v/v) Alamar blue solution was added in an empty well with a plain hydrogel and incubated at the same time as a blank group for quality control. The relative cell viability % was evaluated according to equation (1):
| (1) |
, where Fb, Fc, and Fs are the fluorescence intensity of the blank, the positive control, and the sample, respectively.
Staining and quantification of ALP and Alizarin red S
All hydrogels were incubated in osteogenic media (DMEM, 10% FBS, 50 μg/mL L-ascorbic acid, 10 mM β-glycerophosphate, 1% P/S, and 100 nM dexamethasone). On day 3 and 7, gels were fixed for 20 min in 10% formalin, thoroughly rinsed with PBS, and stained for 2 h in a BCIP and NBT solution buffered with 100 mM Tris, 100 mM NaCl, 50 mM MgCl2, pH 8.5. The images were acquired on an Olympus SZX16 Stereomicroscope (Tokyo, Japan). The expressed ALP was stained in blue. For the ALP activity assay, gels were rinsed with PBS, and incubated for 5 min at 4 °C in a lysis buffer (0.1% Tween 20 in PBS). ALP activity was determined colorimetrically using p-nitrophenyl phosphate as a substrate and measured at 405 nm. The ALP activity was calculated based on total DNA contents determined by the picogreen assay (Thermo Scientific, Rockford, IL).
For the calcium staining, gels were immersed in 10% formalin for 20 min, rinsed with PBS, and stained in Alizarin red S solution (2%) for 5 min. Afterwards, the gels were rinsed three times in PBS for 16 h with gentle shaking. The images were next taken on the Olympus SZX16 Stereomicroscope. Deposited calcium was stained red. The semi quantification of deposited calcium was performed with acetic acid extraction and neutralization with ammonium hydroxide. The colorimetric measurement was carried out at 405 nm, and the results were normalized to the weight of stained gels.
RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA was extracted from MSCs encapsulated with sterosomes (200 μg/mL) in hydrogels using Trizol reagent and RNeasy Mini kit. Reversely transcribed cDNA from 500 ng of total RNA with a cDNA transcription kit (Invitrogen) was measured on a LightCycler 480 PCR (Indianapolis, IN) in the presence of 20 μL SYBR Green for 45 cycles. The relative gene levels were calculated with the reference to the housekeeping gene (GAPDH). The sequences of primers were listed in Table S1.
Calvarial defect model
All surgical experiments strictly abided by the guidelines of the Chancellor’s Animal Research Committee (ARC) at the University of California, Los Angeles. All full-thickness craniotomy defects (3 mm) were drilled on 8~12 week-old male CD-1 nude mice on the right parietal bone. Each defect was cleaned and then treated with MeGC hydrogels containing sterosomes (200μg/mL) and BMSCs or left empty. Following the surgery, all animals were monitored until they regain sternal recumbency and transported to the vivarium for postoperative care. Buprenorphine was injected subcutaneously in all animals with a concentration of 0.1 mg/kg for 3 days for pain management. All animals had free access to water including trimethoprim-sulfamethoxazole to avoid infection for 7 days. Detailed information is descried elsewhere.17
MicroCT scanning and analysis
The harvested calvarial tissues from all experimental animals 6 weeks post implantation, were immersed in formaldehyde solution (4%) at 25 °C for 48 h under gentle shaking. High–resolution RCT images were acquired for all the samples on SkyScan 1172 (Kontich, Belgium) with 0.5 mm Al filtration (184 μA, 57 kVp). The exposure time was 190 ms, and 475 projections were acquired at the angle of 190°. The resolution was 10 μm for both of the pixel size of projections and the voxel size of the reconstructed dataset. Visualization and reconstruction of the data were carried out with the OsiriX MD imaging software. The new bone surface area of all samples was determined using ImageJ (National Institutes of Health, Bethesda, MD), and normalized to the original defect surface area (3 mm in diameter). Bone volume density (BV/TV%) and trabecular number (Tb. N. mm−1) were measured with the SkyScan CT-Analyzer program (Bruker microCT).
Histological evaluation
After μCT scanning, all fixed tissues were incubated in EDTA solution (10%) for 7 days with gentle shaking, with one time refresh of the EDTA solution on day 3. After decalcification, samples were embedded into paraffin and cut at a thickness of 5 μm. Hematoxyline-eosin (H&E) and Masson trichrome staining were performed to evaluate new bone formation. The blue color, indicative of regenerated or mature bone, was observed with an Olympus IX71 microscope (Tokyo Japan). The size of the remaining defect was determined with ImageJ, compared to the blank control group.
Additional sections were undergone immunohistochemical analysis. The deparaffinized sections were processed with citric acid for antigen retrieval, thereafter incubated with the primary antibody osteocalcin (OCN), and were detected by the HRP/DAB kit (Abcam). The sections were further counterstained with Mayer’s hematoxylin. Each stained image was further quantified with ImageJ.
Statistical analysis
All values presented in this work were the average of at least three independent experiments and the error bars represented the standard deviations. The analysis of variances (ANOVA) followed by Tukey’s post hoc test was employed in this work, and p < 0.05 was used as the cutoff for significance. The Pearson’s correlation test was performed to estimate the correlation between sterosome concentration and osteogenic capacity.
Supplementary Material
Acknowledgments
This work was supported by the National Institutes of Health grants R01 AR060213 and R21 DE021819. We thank A. P. Ramos for his efficient help with the IR measurements.
Footnotes
Supporting Information Available
This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Torchilin VP. Recent Advances with Liposomes as Pharmaceutical Carriers. Nat Rev Drug Discovery. 2005;4:145–160. doi: 10.1038/nrd1632. [DOI] [PubMed] [Google Scholar]
- 2.Immordino ML, Dosio F, Cattel L. Stealth Liposomes: Review of the Basic Science, Rationale, and Clinical Applications, Existing and Potential. Int J Nanosci. 2006;2006:297–315. [PMC free article] [PubMed] [Google Scholar]
- 3.Discher DE, Ortiz V, Srinivas G, Klein ML, Kim Y, Christian D, Cai S, Photos P, Ahmed F. Emerging Applications of Polymersomes in Delivery: from Molecular Dynamics to Shrinkage of Tumors. Prog Polym Sci. 2007;32:838–857. doi: 10.1016/j.progpolymsci.2007.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Christian DA, Cai S, Bowen DM, Kim Y, Pajerowski D, Discher DE. Polymersome Carriers: from Self Assembly to siRNA and Protein Therapeutics. Eur J Pharm Biopharm. 2009;71:463–474. doi: 10.1016/j.ejpb.2008.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Marianecci C, Di Marzio L, Rinaldi F, Celia C, Paolino D, Alhaique F, Esposito S, Carafa M. Niosomes from 80s to Present: the State of the Art. Adv Colloid Interface Sci. 2014;205:187–206. doi: 10.1016/j.cis.2013.11.018. [DOI] [PubMed] [Google Scholar]
- 6.Husseini GA, Pitt WG. Micelles and Nanoparticles for Ultrasonic Drug and Gene Delivery. Adv Drug Deliv Rev. 2008;60:1137–1152. doi: 10.1016/j.addr.2008.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fakhari A, Subramony JA. Engineered in-Situ Depot Forming Hydrogels for Intratumoral Drug Delivery. J Control Release. 2015;220:465–475. doi: 10.1016/j.jconrel.2015.11.014. [DOI] [PubMed] [Google Scholar]
- 8.Esfand R, Tomalia DA. Poly(Amidoamine) (Pamam) Dendrimers: from Biomimicry to Drug Delivery and Biomedical Applications. Drug Discov Today. 2001;6:427–436. doi: 10.1016/s1359-6446(01)01757-3. [DOI] [PubMed] [Google Scholar]
- 9.Menjoge AR, Kannan RM, Tomalia DA. Dendrimer Based Drug and Imaging Conjugates: Design Considerations for Nanomedical Applications. Drug Discov Today. 2010;15:171–185. doi: 10.1016/j.drudis.2010.01.009. [DOI] [PubMed] [Google Scholar]
- 10.Arias JL. Liposomes in Drug Delivery: a Patent Review (2007-Present) Expert Opin Ther Pat. 2013;23:1399–1414. doi: 10.1517/13543776.2013.828035. [DOI] [PubMed] [Google Scholar]
- 11.Bangham AD, Horne RW. Negative Staining of Phospholipids and Their Structural Modification by Surface-Active Agents as Observed in the Electron Microscope. J Mol Biol. 1964;8:660–668. doi: 10.1016/s0022-2836(64)80115-7. [DOI] [PubMed] [Google Scholar]
- 12.Papakostas D, Rancan F, Sterry W, Blume-Peytavi U, Vogt A. Nanoparticles in Dermatology. Arch Dermatol Res. 2011;303:533–550. doi: 10.1007/s00403-011-1163-7. [DOI] [PubMed] [Google Scholar]
- 13.Fathi M, Mozafari MR, Mohebbi M. Nanoencapsulation of Food Ingredients Using Lipid Based Delivery Systems. Trends Food Sci Technol. 2012;23:13–27. [Google Scholar]
- 14.Barani H, Montazer M. A Review on Applications of Liposomes in Textile Processing. J Liposome Res. 2008;18:249–262. doi: 10.1080/08982100802354665. [DOI] [PubMed] [Google Scholar]
- 15.Cui Z-K, Lafleur M. Lamellar Self-Assemblies of Single-Chain Amphiphiles and Sterols and Their Derived Liposomes: Distinct Compositions and Distinct Properties. Colloid Surf B-Biointerfaces. 2014;114:177–185. doi: 10.1016/j.colsurfb.2013.09.042. [DOI] [PubMed] [Google Scholar]
- 16.Cui Z-K, Bouisse A, Cottenye N, Lafleur M. Formation of pH-Sensitive Cationic Liposomes from a Binary Mixture of Monoalkylated Primary Amine and Cholesterol. Langmuir. 2012;28:13668–13674. doi: 10.1021/la302278q. [DOI] [PubMed] [Google Scholar]
- 17.Cui Z-K, Fan J, Kim S, Bezouglaia O, Fartash A, Wu BM, Aghaloo T, Lee M. Delivery of siRNA via Cationic Sterosomes to Enhance Osteogenic Differentiation of Mesenchymal Stem Cells. J Control Release. 2015;217:42–52. doi: 10.1016/j.jconrel.2015.08.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li HF, Guo HJ, Li H. Cholesterol Loading Affects Osteoblastic Differentiation in Mouse Mesenchymal Stem Cells. Steroids. 2013;78:426–433. doi: 10.1016/j.steroids.2013.01.007. [DOI] [PubMed] [Google Scholar]
- 19.Luchetti G, Sircar R, Kong JH, Nachtergaele S, Sagner A, Byrne EFX, Covey DF, Siebold C, Rohatgi R. Cholesterol Activates the G-Protein Coupled Receptor Smoothened to Promote Hedgehog Signaling. eLife. 2016;5:22. doi: 10.7554/eLife.20304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bjorkhem I, Meaney S, Diczfalusy U. Oxysterols in Human Circulation: Which Role Do They Have? Curr Opin Lipidol. 2002;13:247–253. doi: 10.1097/00041433-200206000-00003. [DOI] [PubMed] [Google Scholar]
- 21.Bjorkhem I, Diczfalusy U. Oxysterols - Friends, Foes, or Just Fellow Passengers? Arterioscler Thromb Vasc Biol. 2002;22:734–742. doi: 10.1161/01.atv.0000013312.32196.49. [DOI] [PubMed] [Google Scholar]
- 22.Radhakrishnan A, Ikeda Y, Kwon HJ, Brown MS, Goldstein JL. Sterol-Regulated Transport of Srebps from Endoplasmic Reticulum to Golgi: Oxysterols Block Transport by Binding to Insig. Proc Natl Acad Sci U S A. 2007;104:6511–6518. doi: 10.1073/pnas.0700899104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Panini SR, Sinensky MS. Mechanisms of Oxysterol-Induced Apoptosis. Curr Opin Lipidol. 2001;12:529–533. doi: 10.1097/00041433-200110000-00008. [DOI] [PubMed] [Google Scholar]
- 24.Park K, Scott AL. Cholesterol 25-Hydroxylase Production by Dendritic Cells and Macrophages Is Regulated by Type I Interferons. J Leukoc Biol. 2010;88:1081–1087. doi: 10.1189/jlb.0610318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.LeBlanc MA, McMaster CR. Lipid Binding Requirements for Oxysterol-Binding Protein Kes1 Inhibition of Autophagy and Endosome-Trans-Golgi Trafficking Pathways. J Biol Chem. 2010;285:33875–33884. doi: 10.1074/jbc.M110.147264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Soroosh P, Wu JJ, Xue XH, Song J, Sutton SW, Sablad M, Yu JX, Nelen MI, Liu XJ, Castro G, Luna R, Crawford S, Banie H, Dandridge RA, Deng X, Bittner A, Kuei C, Tootoonchi M, Rozenkrants N, Herman K, et al. Oxysterols Are Agonist Ligands of Rorγt and Drive Th17 Cell Differentiation. Proc Natl Acad Sci U S A. 2014;111:12163–12168. doi: 10.1073/pnas.1322807111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Olivier E, Dutot M, Regazzetti A, Dargère D, Auzeil N, Laprévote O, Rat P. Lipid Deregulation in UV Irradiated Skin Cells: Role of 25-Hydroxycholesterol in Keratinocyte Differentiation During Photoaging. J Steroid Biochem Mol Biol. 2016;169:189–197. doi: 10.1016/j.jsbmb.2016.05.015. [DOI] [PubMed] [Google Scholar]
- 28.Liu SY, Aliyari R, Chikere K, Li GM, Marsden MD, Smith JK, Pernet O, Guo HT, Nusbaum R, Zack JA, Freiberg AN, Su L, Lee B, Cheng G. Interferon-Inducible Cholesterol-25-Hydroxylase Broadly Inhibits Viral Entry by Production of 25-Hydroxycholesterol. Immunity. 2013;38:92–105. doi: 10.1016/j.immuni.2012.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hokugo A, Saito T, Li A, Sato K, Tabata Y, Jarrahy R. Stimulation of Bone Regeneration Following the Controlled Release of Water-Insoluble Oxysterol from Biodegradable Hydrogel. Biomaterials. 2014;35:5565–5571. doi: 10.1016/j.biomaterials.2014.03.018. [DOI] [PubMed] [Google Scholar]
- 30.Kha HT, Basseri B, Shouhed D, Richardson J, Tetradis S, Hahn TJ, Parhami F. Oxysterols Regulate Differentiation of Mesenchymal Stem Cells: Pro-Bone and Anti-Fat. J Bone Miner Res. 2004;19:830–840. doi: 10.1359/JBMR.040115. [DOI] [PubMed] [Google Scholar]
- 31.Montgomery SR, Nargizyan T, Meliton V, Nachtergaele S, Rohatgi R, Stappenbeck F, Jung ME, Johnson JS, Aghdasi B, Tian HJ, Weintraub G, Inoue H, Atti E, Tetradis S, Pereira RC, Hokugo A, Alobaidaan R, Tan Y, Hahn TJ, Wang JC, et al. A Novel Osteogenic Oxysterol Compound for Therapeutic Development to Promote Bone Growth: Activation of Hedgehog Signaling and Osteogenesis through Smoothened Binding. J Bone Miner Res. 2014;29:1872–1885. doi: 10.1002/jbmr.2213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nachtergaele S, Mydock LK, Krishnan K, Rammohan J, Schlesinger PH, Covey DF, Rohatgi R. Oxysterols Are Allosteric Activators of the Oncoprotein Smoothened. Nat Chem Biol. 2012;8:211–220. doi: 10.1038/nchembio.765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kim WK, Meliton V, Amantea CM, Hahn TJ, Parhami F. 20(S)-Hydroxycholesterol Inhibits PPAR Gamma Expression and Adipogenic Differentiation of Bone Marrow Stromal Cells through a Hedgehog-Dependent Mechanism. J Bone Miner Res. 2007;22:1711–1719. doi: 10.1359/jbmr.070710. [DOI] [PubMed] [Google Scholar]
- 34.Cui Z-K, Bastiat G, Jin C, Keyvanloo A, Lafleur M. Influence of the Nature of the Sterol on the Behavior of Palmitic Acid/Sterol Mixtures and Their Derived Liposomes. Biochim Biophys Acta. 2010;1798:1144–1152. doi: 10.1016/j.bbamem.2010.02.008. [DOI] [PubMed] [Google Scholar]
- 35.Epand RM, Bach D, Borochov N, Wachtel E. Cholesterol Crystalline Polymorphism and the Solubility of Cholesterol in Phosphatidylserine. Biophys J. 2000;78:866–873. doi: 10.1016/S0006-3495(00)76644-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mantsch HH, McElhaney RN. Phospholipid Phase Transitions in Model and Biological Membranes as Studied by Infrared Spectroscopy. Chem Phys Lipids. 1991;57:213–226. doi: 10.1016/0009-3084(91)90077-o. [DOI] [PubMed] [Google Scholar]
- 37.Kodati RV, Lafleur M. Comparaison between Orientational and Conformational Orders in Fluid Lipid Bilayers. Biophys J. 1993;64:163–170. doi: 10.1016/S0006-3495(93)81351-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Muller RH, Jacobs C, Kayser O. Nanosuspensions as Particulate Drug Formulations in Therapy Rationale for Development and What We Can Expect for the Future. Adv Drug Deliv Rev. 2001;47:3–19. doi: 10.1016/s0169-409x(00)00118-6. [DOI] [PubMed] [Google Scholar]
- 39.Hu J, Hou Y, Park H, Choi B, Hou S, Chung A, Lee M. Visible Light Crosslinkable Chitosan Hydrogels for Tissue Engineering. Acta Biomater. 2012;8:1730–8. doi: 10.1016/j.actbio.2012.01.029. [DOI] [PubMed] [Google Scholar]
- 40.Choi B, Kim S, Lin B, Li K, Bezouglaia O, Kim J, Evseenko D, Aghaloo T, Lee M. Visible-Light-Initiated Hydrogels Preserving Cartilage Extracellular Signaling for Inducing Chondrogenesis of Mesenchymal Stem Cells. Acta Biomater. 2015;12:30–41. doi: 10.1016/j.actbio.2014.10.013. [DOI] [PubMed] [Google Scholar]
- 41.Kim S, Cui Z-K, Fan J, Fartash A, Aghaloo TL, Lee M. Photocrosslinkable Chitosan Hydrogels Functionalized with the RGD Peptide and Phosphoserine to Enhance Osteogenesis. J Mater Chem B. 2016;4:5289–5298. doi: 10.1039/C6TB01154C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Arakawa C, Ng R, Tan S, Kim S, Wu B, Lee M. Photopolymerizable Chitosan-Collagen Hydrogels for Bone Tissue Engineering. J Tissue Eng Regen Med. 2017;4:164–174. doi: 10.1002/term.1896. [DOI] [PubMed] [Google Scholar]
- 43.Dwyer JR, Sever N, Carlson M, Nelson SF, Beachy PA, Parhami F. Oxysterols Are Novel Activators of the Hedgehog Signaling Pathway in Pluripotent Mesenchymal Cells. J Biol Chem. 2007;282:8959–8968. doi: 10.1074/jbc.M611741200. [DOI] [PubMed] [Google Scholar]
- 44.Muise-Helmericks RC, Demcheva M, Vournakis JN, Seth A. Poly-N-Acetyl Glucosamine Fibers Activate Bone Regeneration in a Rabbit Femur Injury Model. J Trauma-Injury Infect Crit Care. 2011;71:S194–S196. doi: 10.1097/TA.0b013e3182258799. [DOI] [PubMed] [Google Scholar]
- 45.Utsugi T, Schroit AJ, Connor J, Bucana CD, Fidler IJ. Elevated Expression of Phosphatidylserine in the Outer-Membrane Leaflet of Human Tumor-Cells and Recognition by Activated Human Blood Monocytes. Cancer Res. 1991;51:3062–3066. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


