Abstract
Purpose
Many women with major depressive disorder (MDD) respond inadequately to standard treatments. Augmentation of conventional antidepressants with creatine monohydrate and 5-hydroxytryptophan (5-HTP) could correct deficits in serotonin production and brain bioenergetics associated with depression in women, yielding synergistic benefit. We describe an open-label study of 5-HTP and creatine augmentation in women with MDD who had failed selective serotonin reuptake inhibitor (SSRI) or serotonin-norepinephrine reuptake inhibitor (SNRI) monotherapy.
Methods
15 women who were adequately adherent to an SSRI or SNRI and currently suffering from MDD, with a 17-item Hamilton Depression Rating Scale (HAM-D) score ≥ 16, were treated with 5g of creatine monohydrate daily and 100mg of 5-HTP twice daily for 8 weeks, with 4 weeks of post-treatment follow-up. The primary outcome was change in mean HAM-D scores.
Results
Mean HAM-D scores declined from 18.9 ± 2.5 at pretreatment visits to 7.5 ±4.4 (p < 0.00001), a decrease of 60%. Participants did not experience any serious treatment-related adverse events.
Conclusions
Combination treatment with creatine and 5-HTP may represent an effective augmentation strategy for women with SSRI- or SNRI-resistant depression. Given the limitations of this small, open-label trial, future study in randomized, placebo-controlled trials is warranted.
Keywords: major depressive disorder, altitude, hypobaric hypoxia, serotonin, brain bioenergetics
1. Introduction
Major depressive disorder (MDD) is a chronic illness with a lifetime prevalence of over 16%.1 It is associated with significant personal and social costs,2 diminished quality of life, and disability.3,4 This burden falls disproportionately on women. They are twice as likely as men to develop depression,5 are more likely to attempt suicide6 and to take antidepressants,7 and are overrepresented in clinical trials of treatment-resistant depression.8 Accordingly, studies addressing the treatment of depression in women and the development of treatment-resistance are urgently needed.
The mechanisms underpinning the higher rates of depression in women are not known and many possibilities have been suggested. One possibility involves alterations in serotoninergic pathways, which are often associated with depression and suicide.9 Women may be more vulnerable to processes that impair serotonin production, since women exhibit lower baseline serotonin levels.10 Conversely, women may be more responsive to serotonin replacement strategies. Although selective serotonin reuptake inhibitors (SSRIs) are widely prescribed for MDD, animal studies suggest that they may be less effective in the setting of reduced serotonin synthesis, as SSRI non-response is observed in several serotonin depletion paradigms.11–13 Deficits in serotonin synthesis could be corrected by supplementation with 5-hydroxytryptophan (5-HTP),14 which is metabolized directly into serotonin by l-aromatic acid decarboxylase.15 5-HTP has previously been investigated as an antidepressant, increases brain serotonin stores when administered orally,15,16 and increases extracellular serotonin levels in mice deficient in tryptophan hydroxylase or receiving a serotonin reuptake inhibitor.17 To our knowledge, 5-HTP has not been investigated as an augmenting agent for use with SSRIs or SNRIs in humans, probably because of concerns about serotonin syndrome,18 though it has been successfully combined with tricyclic antidepressants19–21 and monoamine oxidase inhibitors.22
Inadequate response to traditional antidepressants could also result from alterations in brain bioenergetics. In depressed adults, phosphorus magnetic resonance spectroscopy demonstrates reduced total nucleotide triphosphate concentrations and higher phosphocreatine concentrations when compared to healthy volunteers, and this pattern is more common in women.23–25 These changes can be rectified by administration of oral creatine monohydrate, which is also emerging as an effective augmenting agent for depression. In human trials involving women, oral creatine alters brain creatine, phosphocreatine, and total nucleotide triphosphate levels26,27 in tandem with improvements in mood.28 In a large placebo-controlled study, creatine augmentation of escitalopram produced significant improvements in depression scores compared to placebo in treatment-naïve women with MDD.29
Because of the increased vulnerability of women to depression, and because women may be more responsive to both creatine supplementation and correction of serotonin deficits than men, we undertook a pilot open-label study to examine the combined use of creatine and 5-HTP as augmenting agents for the treatment of depression in women with incomplete responses to SSRIs or serotonin-norepinephrine reuptake inhibitors (SNRIs). Combined treatment was utilized in the hope that the two supplements would have synergistic efficacy.
2. Materials and Methods
2.1 Subject Selection
Adult women with MDD confirmed by the Structured Clinical Interview for DSM-IV were recruited. Subjects were required to have continued depression of at least moderate severity at both the screening and baseline visits, with 17-item Hamilton Depression Rating Scale (HAM-D) scores ≥ 16, despite adequate adherence to at least 8 weeks of any Food and Drug Administration-approved SSRI or SNRI at standard doses. Eligible dose ranges for antidepressants represented in the study included citalopram 20mg-40mg daily,30 escitalopram 10mg-20mg daily,31 fluoxetine 20mg-80mg daily,32 fluvoxamine 100mg-300mg daily,33 sertraline 50mg-200mg daily,34 paroxetine 20mg-50mg daily,35 venlafaxine 75mg-375mg daily,36,37 and duloxetine 40mg-120mg daily.38 We assessed whether subjects had received an adequate trial of their current antidepressant based on Antidepressant Treatment Response Questionnaire (ATRQ) dosage guidelines.39 Adherence was assessed by participant report and required completion of at least 75% of scheduled doses. Exclusion criteria included histories of bipolar disorder, psychotic illness, renal disease, diabetes, colitis or diverticulitis, pulmonary disease, cardiac disease, corrected QT interval > 500ms, seizure disorder, current serious suicide risk identified by the Columbia Suicide Severity Rating Scale (C-SSRS), current treatment with an antipsychotic, mood stabilizer, neurostimulation, or any non-SSRI/SNRI antidepressant, and pregnancy. Because of the possible increased risk of serotonin syndrome in subjects taking an SSRI/SNRI and 5-HTP, subjects with a history of serotonin syndrome were excluded. Because of an association between tryptophan and eosinophilia-myalgia syndrome (EMS), subjects with rheumatologic conditions, a history of tryptophan intolerance, or pre-existing eosinophilia were excluded. The study was approved by the local institutional review board, and informed consent was obtained from all participants. The trial was registered with ClinicalTrials.gov (identifier: NCT02356107).
2.2 Study Measures and Intervention
This open-label study of 5-HTP and creatine as combined augmentation treatment included a 2-visit screening phase to assure eligibility, an 8-week treatment phase with visits at weeks 1, 2, 4, 6, and 8, and two post-treatment visits at weeks 10 and 12. Participants were given fixed-doses of 5g of Creapure® brand of Creatine (AlzChem LLC, Trostberg, Germany) and 100 mg twice daily of 5-HTP derived from Griffonia seed extract (Fuller Enterprise USA Inc, Ontario, Canada) by mouth daily for 8 weeks, in addition to their usual dose of SSRI or SNRI. Vital signs and a screening physical were performed at each visit. Adverse events were assessed at each visit, and subjects were asked about 5-HTP-specific effects such as nausea, vomiting, diarrhea, insomnia, palpitations, headache, and weight gain.40 No specific inquiries were made regarding adverse effects of creatine, as generally these are negligible.41,42 Study measures included the HAM-D, Montgomery-Asberg Depression Rating Scale (MADRS), Clinical Global Impression severity (CGI-S), and Beck Anxiety Inventory (BAI) for study outcomes, as well as C-SSRS, and Young Mania Rating Scale (YMRS) for assessment of adverse effects. Subjects were screened for serotonin syndrome utilizing the Hunter criteria43 and for EMS44,45 by clinician assessment at each visit. The Discontinuation Emergent Signs and Symptoms Checklist (DESS)46 was administered during follow up visits at weeks 10 and 12 to monitor for symptoms related to discontinuation of the study medications. Safety labs (complete blood count, creatinine, blood urea nitrogen) were administered at the screening visit and at follow-up visits if indicated.
2.3 Statistical Analysis
Our primary outcome was change in HAM-D scores over 8 weeks of 5-HTP and creatine augmentation of subjects’ baseline SSRI or SNRI. Change from mean HAM-D score from the screening and baseline visits over time was analyzed using repeated-measures linear mixed models.47 HAM-D scores from the screening and baseline period were averaged and compared with the mean HAM-D scores from the treatment period, using last observation carried forward. MADRS and BAI scores were analyzed in a similar way. Each clinical score was considered the dependent variable in the linear mixed model. Week was included as a fixed factor and subject was treated as a random factor. Sidak correction was used to compensate for multiple comparisons, controlling for Type I error. Statistical significance was defined at an alpha level of corrected p = 0.05, two-tailed.
3. Results
3.1 Demographics and Baseline Symptoms
Fifteen women with a mean age of 34 ± 11.6 years were enrolled (Table 1). Twelve subjects completed the study. The subjects were moderately to severely ill at baseline, with a mean HAM-D score of 18.9 ± 2.5, a mean MADRS score of 25.4 ± 3.6, and a mean CGI-S score of 4.1 ± 0.3. Twelve patients were treated with an SSRI (fluoxetine, sertraline, escitalopram, citalopram, paroxetine), while 3 patients were treated with an SNRI (venlafaxine, duloxetine). All subjects had been taking at least the minimum recommended dose of their respective antidepressant according to the ATRQ for at least 8 weeks.
Table 1.
Baseline characteristics of women receiving SSRI/SNRI augmentation with 5-HTP and creatine
| Characteristic | Value | |
|---|---|---|
| N | 15 | |
| Age (years) | 34 ± 11.6 | |
| Race | ||
| Caucasian | 15 (100%) | |
| Other | 0 (0%) | |
| Mean Weeks Ill | 395.2 ± 286.0 | |
|
| ||
| Antidepressant Types and Doses | N (mean dose) | |
|
| ||
| SSRIs | Escitalopram | 3 (16.7mg) |
| Citalopram | 2 (35mg) | |
| Fluoxetine | 2 (40mg) | |
| Paroxetine | 1 (20mg) | |
| Sertraline | 4 (62.5mg) | |
| SNRIs | Duloxetine | 1 (40mg) |
| Venlafaxine | 2 (150mg) | |
3.2 Effects of 5-HTP and creatine on depression and anxiety
Mean HAM-D scores improved significantly over the 8 weeks of treatment (18.9 ± 2.5 to 7.5 ± 4.4, p < 0.00001), representing an average 60.3% reduction from the baseline/screening visits (Table 2 and Figure 1). Ten patients (66.7%) met response criteria of at least a 50% reduction in HAM-D score. Seven patients (46.7%) met HAM-D remission criteria (HAM-D score ≤ 7).48 There was no significant difference in HAM-D rating improvement with respect to baseline SSRI or SNRI use (60.4% vs 57.4%, p =0.88).
Table 2.
Efficacy and safety data for women receiving SSRI/SNRI augmentation with 5-HTP and creatine
| Outcomes | Pretreatment | Week 8 |
|---|---|---|
| Measure | Mean ± SD | Mean ± SD |
| HAM-D | 18.9 ± 2.5 | 7.5 ± 4.4, p < 0.00001 |
| MADRS | 25.4 ± 3.6 | 9.0 ± 6.6, p < 0.00001 |
| BAI | 22.7 ± 9.2 | 9.3 ± 6.4, p < 0.00001 |
| CGI | 4.1 ± 0.4 | 1.9 ± 1.0, p < 0.00001 |
| YMRS | 1.2 ± 0.9 | 0.4 ± 1.1, p = 0.011 |
| Weight | 71.4 ± 19.3kg | 72.5 ± 19.8kg, p = 0.051 |
Figure 1.
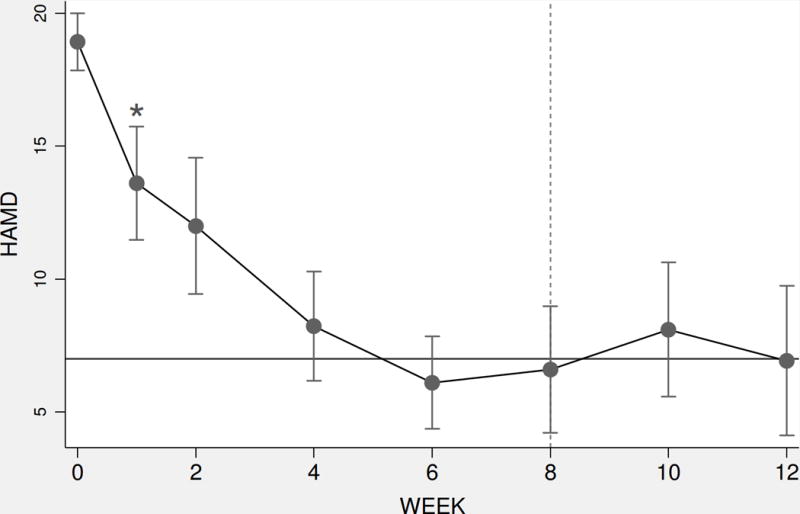
HAM-D scores over 8 weeks in women receiving SSRI/SNRI augmentation with 5-HTP and creatine. Active treatment provided through week 8; weeks 10 and 12 represent post-treatment follow-up. Error bars represent 95% confidence interval of the mean. Solid horizontal line indicates potential remission score (HAM-D=7). Asterisk (*) represents beginning of statistical significance compared to baseline (Sidak corrected p < 0.05). Dashed vertical line indicates the finish of 8 weeks of treatments.
Mean MADRS scores over 8 weeks were significantly reduced (25.4 ± 3.6 to 9.0 ± 6.6, p < 0.00001), representing an average 65% reduction (Figure 2). Twelve patients (80%) met MADRS response criteria (at least 50% reduction in MADRS score), and 8 patients (53%) met MADRS remission criteria (MADRS score < 10).49 Improvements in anxiety were also significant, with BAI scores significantly reduced (22.7 ± 9.2 to 9.3 ± 6.4, p <0.00001), representing a mean 59.9% decrease (Figure 3). CGI-S scores were also significantly improved (4.1 ± 0.4 to 1.9 ± 1.0, p < 0.00001)
Figure 2.

MADRS scores over 8 weeks in women receiving SSRI/SNRI augmentation with 5-HTP and creatine. Active treatment provided through week 8; weeks 10 and 12 represent post-treatment follow-up. Error bars represent 95% confidence interval of the mean. Solid horizontal line indicates potential remission score (MADRS=10). Asterisk (*) represents beginning of statistical significance compared to baseline (Sidak corrected p < 0.05). Dashed vertical line indicates the finish of 8 weeks of treatments.
Figure 3.

BAI scores over 8 weeks in women receiving SSRI/SNRI augmentation with 5-HTP and creatine. Active treatment provided through week 8; weeks 10 and 12 represent post-treatment follow-up. Error bars represent 95% confidence interval of the mean. Solid horizontal line indicates potential remission score (BAI=10). Asterisk (*) represents beginning of statistical significance compared to baseline (Sidak corrected p < 0.05). Dashed vertical line indicates the finish of 8 weeks of treatments.
3.3 Safety and adverse events
Combination treatment with adjunctive 5-HTP and creatine was safe and well tolerated. Two participants withdrew from the study and were lost to follow-up. One subject described symptoms (increased anxiety, tremor) that raised concern for serotonin syndrome, but did not exhibit vital sign changes, physical exam abnormalities, or meet Hunter criteria for that condition. This subject was, however, withdrawn because of relapse of anorexia nervosa, which was deemed unrelated to study participation. A total of seven adverse events were recorded during the treatment period. These include cold/flu symptoms (n=1), kidney stones (n=1), urinary tract infection (n=1), diagnosis of medullary sponge kidney (n=1), injury/fall (n=2), night sweats/increased sweating (n=1). The only severe adverse event was the diagnosis of medullary sponge kidney disease, which was not attributed to study participation, as the condition is familial.50 There was a trend toward mild weight gain during treatment (71.4 ± 19.3kg to 72.5 ± 19.8kg, p = 0.051), with the mean change being 1.1 ± 1.9kg. There were no significant changes in heart rate or blood pressure. There were no cases of serotonin syndrome, EMS, or treatment-emergent mania/hypomania based on YMRS scores. YMRS scores improved significantly from baseline by week 8 (1.2 ± 0.9 to 0.4 ± 1.1, p = 0.011). There was no treatment-emergent suicidal ideation based on C-SSRS, and there were no suicide attempts.
3.4 Discontinuation symptoms
Seven of the 12 study completers (58.3%) decided to continue treatment with creatine, 5-HTP, or both after the 8-week treatment period, though this required purchasing them over the counter. These subjects believed the treatment had been helpful and were reluctant to discontinue it. Accordingly, we were unable to collect adequate data regarding the effect of study drug cessation on discontinuation symptoms. There was no significant difference in DESS scores averaged over weeks 10 and 12 for subjects who elected to continue treatment versus those who did not (193.4 ± 13.2 vs 184.9 ± 10.3, p = 0.30).
4. Discussion
This study provides preliminary evidence that combination treatment with 5-HTP and creatine monohydrate may represent effective adjunctive treatment for women with MDD who have responded inadequately to SSRIs or SNRIs for at least 8 weeks. The treatment had few adverse effects, apart from a trend toward mild (~1kg) weight gain. Together, these factors suggest combination treatment with 5-HTP and creatine is a promising approach to antidepressant treatment and deserving of further study. The wide availability of both compounds is a further merit of the study, as it contributes to formalized understanding of the antidepressant effect of two nutritional supplements that are already in widespread use. Caution is still necessary, however, if only because of historical problems in guaranteeing the potency and safety of many nutritional supplements.
The study had obvious limitations. It is a small, open-label study without a placebo control. Likewise, combination treatment with 5-HTP and creatine makes it impossible to discern what proportion of patients’ response was due to each medication, though comparison with historical controls treated with creatine or 5-HTP alone as augmentation might partially rectify this limitation. In Lyoo et al.,29 subjects received escitalopram with adjunctive creatine or placebo. There, the change in mean HAM-D score at 8 weeks was 79.7%, while in the current study combining 5-HTP with creatine, it was only 60.3%. Nardini et al.19 compared adjunctive 5-HTP 300mg daily or placebo in patients treated with clomipramine 50mg daily. At the end of four weeks, HAM-D scores had fallen by 57.7% in the 5-HTP group and by 40.7% in the placebo group, a significant difference. These studies would seem to indicate no synergistic benefit of combination treatment. But comparisons between response rates in the current and historical studies is limited by their intrinsic differences. In Lyoo et al., the mean reduction in HAM-D scores with placebo at 8 weeks was high, at 62.5%, subjects were more depressed at baseline (mean HAM-D score 26.9 vs 18.9), they had been free of psychotropic medication for ≥ 8 weeks, and 78.8% were antidepressant naïve. This is in stark contrast to the more treatment-resistant subjects studied here, all of whom were depressed despite ≥ 8 weeks of antidepressant monotherapy. In Nardini et al., the patients were hospitalized, were somewhat more ill at baseline, were treated for only four weeks, and were started on 5-HTP or placebo at the same time as clomipramine. Accordingly, historical controls do not exclude the possibility of synergistic benefit.
Another limitation is that, although the subjects were persistently depressed, with an average symptom duration of 395.2 ± 286.0 weeks, we did not obtain complete medication histories to ascertain whether they met standard criteria for treatment-resistance (failure of at least 2 antidepressant trials). Additionally, baseline antidepressant doses for some subjects were in the low therapeutic range, and participants were taking a variety of antidepressants, creating sample heterogeneity. The study also focused on adult women, limiting its generalizability.
Finally, these data were collected at the moderately high altitude of Salt Lake City, Utah, so that our efficacy, tolerability and safety results may not generalize to women residing at altitudes nearer sea level. This is because alterations in serotonin metabolism and brain bioenergetics have been linked to hypoxia, and increased altitude of residence can produce chronic hypobaric hypoxia (reduced blood oxygen because of reduced atmospheric pressure).51,52 Hypoxia may reduce the efficiency of serotonin synthesis, as the activity of tryptophan hydroxylase is oxygen-dependent, and the conversion of tryptophan to 5-HTP by tryptophan hydroxylase is the rate-limiting step in serotonin production.53 Indeed, simulated high altitude decreases levels of serotonin54 and reduces response to fluoxetine55 in rodent models. Altitude and hypobaric hypoxia can also affect brain bioenergetics. Healthy individuals residing at 1370m (Salt Lake City, UT) show reduced inorganic phosphate levels and increased brain pH compared to those residing near sea level (Belmont, MA or Charleston, SC).56 In rodents, hypobaric hypoxia increases the ratios of cellular metabolites like inositol to total creatine in the frontal cortex,57 which is compatible with reductions in total creatine level. Accordingly, the women participating in this study may have been more likely than women residing at sea level to respond to the intervention, and, because of reduced basal serotonin synthesis, less likely to develop serotonin syndrome.
This small, open-label study of combined 5-HTP and creatine monohydrate as adjunctive treatment for major depressive disorder in women not responsive to an SSRI or SNRI demonstrated robust symptomatic improvement and good safety and tolerability, although its short duration of 12 weeks prevents us from reaching conclusions about the interventions’ long term adverse effects. Further research on this treatment is merited, including definitive randomized placebo-controlled trials, trials including men, multi-arm trials comparing 5-HTP to creatine to the combination, and trials involving the incorporation of biomarkers of treatment response, including neuroimaging modalities and measurements of serotonin synthesis.
Acknowledgments
This work was supported by a grant from the R. Harold Burton Foundation (Salt Lake City, Utah) and the Utah Science, Technology, and Research (USTAR) Initiative (Salt Lake City, Utah).
Funding Source: This work was supported by a grant from the R. Harold Burton Foundation (Salt Lake City, Utah) and the Utah Science, Technology, and Research (USTAR) Initiative (Salt Lake City, Utah).
Footnotes
Address for Reprints: Not applicable
Trial Registration: ClinicalTrials.gov identifier: NCT02356107
References
- 1.Kessler RC, Berglund P, Demler OJ, et al. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62:593–602. doi: 10.1001/archpsyc.62.6.593. [DOI] [PubMed] [Google Scholar]
- 2.Birnbaum HG, Kessler RC, Kelley D, et al. Employer burden of mild, moderate, and severe major depressive disorder: mental health services utilization and costs, and work performance. Depress Anxiety. 2010;27:78–89. doi: 10.1002/da.20580. [DOI] [PubMed] [Google Scholar]
- 3.van der Voort TY, Seldenrijk A, van Meijel B, et al. Functional versus syndromal recovery in patients with major depressive disorder and bipolar disorder. J Clin Psychiatry. 2015;76:809–814. doi: 10.4088/JCP.14m09548. [DOI] [PubMed] [Google Scholar]
- 4.Kessler RC, Barber C, Birnbaum HG, et al. Depression in the workplace: effects on short-term disability. Health Aff (Millwood) 1999;18:163–171. doi: 10.1377/hlthaff.18.5.163. [DOI] [PubMed] [Google Scholar]
- 5.Bebbington PDG, Jenkins R, Lewis G, et al. The influence of age and sex on the prevalence of depressive conditions: report from the National Survey of Psychiatric Morbidity. Int Rev Psychiatry. 2003;15:74–83. doi: 10.1080/0954026021000045976. [DOI] [PubMed] [Google Scholar]
- 6.Mościcki EK, O’Carroll P, Rae DS, et al. Suicide attempts in the Epidemiologic Catchment Area Study. Yale J Biol Med. 1988;61:259–268. [PMC free article] [PubMed] [Google Scholar]
- 7.Pratt LA, Brody DJ, Gu Q. Antidepressant use in persons aged 12 and over: United States, 2005–2008. National Center for Health Statistics Data Brief. 2011:1–8. [PubMed] [Google Scholar]
- 8.Mrazek DA, Hornberger JC, Altar CA, Degtiar I. A review of the clinical, economic, and societal burden of treatment-resistant depression: 1996–2013. Psychiatr Serv. 2014;65:977–987. doi: 10.1176/appi.ps.201300059. [DOI] [PubMed] [Google Scholar]
- 9.van Heeringen K, Mann JJ. The neurobiology of suicide. Lancet Psychiatry. 2014;1:63–72. doi: 10.1016/S2215-0366(14)70220-2. [DOI] [PubMed] [Google Scholar]
- 10.Nishizawa S, Benkelfat C, Young SN, et al. Differences between males and females in rates of serotonin synthesis in human brain. Proc Natl Acad Sci USA. 1997;94:5308–5313. doi: 10.1073/pnas.94.10.5308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kulikov AV, Tikhonova MA, Osipova DV, et al. Association between tryptophan hydroxylase-2 genotype and the antidepressant effect of citalopram and paroxetine on immobility time in the forced swim test in mice. Pharmacol Biochem Behav. 2011;99:683–687. doi: 10.1016/j.pbb.2011.06.020. [DOI] [PubMed] [Google Scholar]
- 12.Sachs BD, Ni JR, Caron MG. Brain 5-HT deficiency increases stress vulnerability and impairs antidepressant responses following psychosocial stress. Proc Natl Acad Sci USA. 2015;112:2557–2562. doi: 10.1073/pnas.1416866112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Siesser WB, Sachs BD, Ramsey AJ, et al. Chronic SSRI treatment exacerbates serotonin deficiency in humanized tph2 mutant mice. ACS Chem Neurosci. 2013;4:84–88. doi: 10.1021/cn300127h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Boadle-Biber MC. Regulation of serotonin synthesis. Prog Biophys Mol Biol. 1993;60:1–15. doi: 10.1016/0079-6107(93)90009-9. [DOI] [PubMed] [Google Scholar]
- 15.Birdsall TC. 5-Hydroxytryptophan: a clinically-effective serotonin precursor. Altern Med Rev. 1998;3:271–80. [PubMed] [Google Scholar]
- 16.Turner EH, Loftis JM, Blackwell AD. Serotonin a la carte: supplementation with the serotonin precursor 5-hydroxytryptophan. Pharmacol Ther. 2006;109:325–338. doi: 10.1016/j.pharmthera.2005.06.004. [DOI] [PubMed] [Google Scholar]
- 17.Jacobsen JP, Rudder ML, Roberts W, et al. SSRI Augmentation by 5-Hydroxytryptophan slow release: mouse pharmacodynamic proof of concept. Neuropsychopharmacology. 2016;412:324–334. doi: 10.1038/npp.2016.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148:705–713. doi: 10.1176/ajp.148.6.705. [DOI] [PubMed] [Google Scholar]
- 19.Nardini M, de Stefano R, Iannuccelli M, et al. Treatment of depression with L-5-hydroxytryptophan combined with chlorimipramine, a double-blind study. Int J Clin Pharmacol Res. 1983;3:239–250. [PubMed] [Google Scholar]
- 20.van Praag HM. Serotonin precursors in the treatment of depression. Adv Biochem Psychopharmacol. 1982;34:259–286. [PubMed] [Google Scholar]
- 21.Van Hiele LJ. l-5-Hydroxytryptophan in depression: The first substitution therapy in psychiatry? Neuropsychobiology. 1980;6:230–240. doi: 10.1159/000117757. [DOI] [PubMed] [Google Scholar]
- 22.Alino JJ, Gutierrez JL, Iglesias ML. 5-Hydroxytryptophan (5-HTP) and a MAOI (nialamide) in the treatment of depressions. A double-blind controlled study. Int Pharmacopsychiatry. 1976;11:8–15. doi: 10.1159/000468207. [DOI] [PubMed] [Google Scholar]
- 23.Iosifescu DV, Bolo NR, Nierenberg AA, et al. Brain bioenergetics and response to triiodothyronine augmentation in major depressive disorder. Biol Psychiatry. 2008;63:1127–1134. doi: 10.1016/j.biopsych.2007.11.020. [DOI] [PubMed] [Google Scholar]
- 24.Moore CM, Christensen JD, Lafer B, et al. Lower levels of nucleoside triphosphate in the basal ganglia of depressed subjects: a phosphorous-31 magnetic resonance spectroscopy study. Am J Psychiatry. 1997;154:116–118. doi: 10.1176/ajp.154.1.116. [DOI] [PubMed] [Google Scholar]
- 25.Volz HP, Rzanny R, Riehemann S, et al. 31P magnetic resonance spectroscopy in the frontal lobe of major depressed patients. Eur Arch Psychiatry Clin Neurosci. 1998;248:289–295. doi: 10.1007/s004060050052. [DOI] [PubMed] [Google Scholar]
- 26.Lyoo IK, Kong SW, Sung SM, et al. Multinuclear magnetic resonance spectroscopy of high-energy phosphate metabolites in human brain following oral supplementation of creatine-monohydrate. Psychiatry Res. 2003;123:87–100. doi: 10.1016/s0925-4927(03)00046-5. [DOI] [PubMed] [Google Scholar]
- 27.Kondo DG, Sung YH, Hellem TL, et al. Open-label adjunctive creatine for female adolescents with SSRI-resistant major depressive disorder: a 31-phosphorus magnetic resonance spectroscopy study. J Affect Disord. 2011;135:354–361. doi: 10.1016/j.jad.2011.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kondo DG, Forrest LN, Shi X, et al. Creatine target engagement with brain bioenergetics: a dose-ranging phosphorus-31 magnetic resonance spectroscopy study of adolescent females with SSRI-resistant depression. Amino Acids. 2016;48:1941–1954. doi: 10.1007/s00726-016-2194-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lyoo IK, Yoon S, Kim TS, et al. A randomized, double-blind placebo-controlled trial of oral creatine monohydrate augmentation for enhanced response to a selective serotonin reuptake inhibitor in women with major depressive disorder. Am J Psychiatry. 2012;169:937–945. doi: 10.1176/appi.ajp.2012.12010009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Montgomery S, Pedersen V, Tanghøj P, et al. Int Clin Psychopharmacol. 1994;9:35–40. doi: 10.1097/00004850-199403001-00006. [DOI] [PubMed] [Google Scholar]
- 31.Kennedy SH, Andersen HF, Lam RW. Efficacy of escitalopram in the treatment of major depressive disorder compared with conventional selective serotonin reuptake inhibitors and venlafaxine XR: a meta-analysis. J Psychiatry Neurosci. 2006;31:122–131. [PMC free article] [PubMed] [Google Scholar]
- 32.Zahajszky J, Rosenbaum J, Tollefson G. Fluoxetine. In: Schatzberg AF, Nemeroff CB, editors. American Psychiatric Publishing Textbook of Psychopharmacology. 4th. Arlington: American Psychiatric Publishing; [online] Available at: http://dx.doi.org/10.1176/appi.books.9781585623860.as13. [Google Scholar]
- 33.Aboujaoude E, Koran L. Fluvoxamine. In: Schatzberg AF, Nemeroff CB, editors. American Psychiatric Publishing Textbook of Psychopharmacology. 4th. Arlington: American Psychiatric Publishing; [online] Available at: http://dx.doi.org/10.1176/appi.books.9781585623860.as16. [Google Scholar]
- 34.Block D, Yonkers K, Carpenter L. Sertraline. In: Schatzberg AF, Nemeroff CB, editors. American Psychiatric Publishing Textbook of Psychopharmacology. 4th. Arlington: American Psychiatric Publishing; [online] Available at: http://dx.doi.org/10.1176/appi.books.9781585623860.as14. [Google Scholar]
- 35.Ehmke C, Nemeroff C. Paroxetine. In: Schatzberg AF, Nemeroff CB, editors. American Psychiatric Publishing Textbook of Psychopharmacology. 4th. Arlington: American Psychiatric Publishing; [online] Available at: http://dx.doi.org/10.1176/appi.books.9781585623860.as15. [Google Scholar]
- 36.Rudolph RL, Fabre LF, Feighner JP, et al. A randomized, placebo-controlled, dose-response trial of venlafaxine hydrochloride in the treatment of major depression. J Clin Psychiatry. 1998;59:116–122. doi: 10.4088/jcp.v59n0305. [DOI] [PubMed] [Google Scholar]
- 37.Rudolph RL, Feiger AD. A double-blind, randomized, placebo-controlled trial of once-daily venlafaxine extended release (XR) and fluoxetine for the treatment of depression. J Affect Disord. 1999;56:171–181. doi: 10.1016/s0165-0327(99)00067-1. [DOI] [PubMed] [Google Scholar]
- 38.Mallinckrodt CH, Prakash A, Andorn AC, et al. Duloxetine for the treatment of major depressive disorder: a closer look at efficacy and safety data across the approved dose range. J Psychiatr Res. 2006;40:337–348. doi: 10.1016/j.jpsychires.2005.08.010. [DOI] [PubMed] [Google Scholar]
- 39.Chandler GM, Iosifescu DV, Pollack MH, et al. Validation of the Massachusetts General Hospital Antidepressant Treatment History Questionnaire (ATRQ) CNS Neurosci Ther. 2010;16:322–325. doi: 10.1111/j.1755-5949.2009.00102.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Byerly WF, Judd LL, Reimherr FW, et al. 5-Hydroxytryptophan: A review of its antidepressant efficacy and adverse effects. J Clin Psychopharmacol. 1987;7:127–137. [PubMed] [Google Scholar]
- 41.Poortmans JR, Francaux M. Long-term oral creatine supplementation does not impair renal function in healthy athletes. Med Sci Sports Exerc. 1999;31:1108–1110. doi: 10.1097/00005768-199908000-00005. [DOI] [PubMed] [Google Scholar]
- 42.Shao A, Hathcock JN. Risk assessment for creatine monohydrate. Regul Toxicol Pharmacol. 2006;45:245–251. doi: 10.1016/j.yrtph.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 43.Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352:1112–1120. doi: 10.1056/NEJMra041867. [DOI] [PubMed] [Google Scholar]
- 44.Allen JA, Peterson A, Sufit R, et al. Post-epidemic eosinophilia–myalgia syndrome associated with L-tryptophan. Arthritis Rheum. 2011;63:3633–3639. doi: 10.1002/art.30514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.5-HTP. Natural Medicines Comprehensive Database Consumer Version 2014. (Accessed 08/23/2016, 2016 at https://medlineplus.gov/druginfo/natural/794.html#Safety.)
- 46.Baldwin DS, Montgomery SA, Nil R, et al. Discontinuation symptoms in depression and anxiety disorders. Int J Neuropsychopharmacol. 2007;10:73–84. doi: 10.1017/S1461145705006358. [DOI] [PubMed] [Google Scholar]
- 47.Cnaan A, Laird N, Slasor P. Tutorial in biostatistics: Using the general linear mixed model to analyse unbalanced repeated measures and longitudinal data. Stat Med. 1997;16:2349–2380. doi: 10.1002/(sici)1097-0258(19971030)16:20<2349::aid-sim667>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- 48.Frank E, Prien RF, Jarrett RB, et al. Conceptualization and rationale for consensus definitions of terms in major depressive disorder: remission, recovery, relapse, and recurrence. Arch Gen Psychiatry. 1991;48:851–855. doi: 10.1001/archpsyc.1991.01810330075011. [DOI] [PubMed] [Google Scholar]
- 49.Hawley C, Gale T, Sivakumaran T, et al. Defining remission by cut off score on the MADRS: selecting the optimal value. J Affect Disord. 2002;72:177–184. doi: 10.1016/s0165-0327(01)00451-7. [DOI] [PubMed] [Google Scholar]
- 50.Leung JC. Inherited renal diseases. Curr Pediatr Rev. 2014;10:95–100. doi: 10.2174/157339631002140513101755. [DOI] [PubMed] [Google Scholar]
- 51.Peacock AJ. Oxygen at high altitude. BMJ. 1998;317:1063–1066. doi: 10.1136/bmj.317.7165.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Grover RF, Weil JV, Reeves JT. Cardiovascular adaptation to exercise at high altitude. Exerc Sport Sci Rev. 1985;14:269–302. [PubMed] [Google Scholar]
- 53.Young SN. Elevated incidence of suicide in people living at altitude, smokers and patients with chronic obstructive pulmonary disease and asthma: possible role of hypoxia causing decreased serotonin synthesis. J Psychiatry Neurosci. 2013;38:423–426. doi: 10.1503/jpn.130002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ray K, Dutta A, Panjwani U, et al. Hypobaric hypoxia modulates brain biogenic amines and disturbs sleep architecture. Neurochem Int. 2011;58:112–118. doi: 10.1016/j.neuint.2010.11.003. [DOI] [PubMed] [Google Scholar]
- 55.Kanekar S, Bogdanova O, Olson P, et al. Antidepressant efficacy in a rodent model of hypoxia-related depression: do SSRIs lose efficacy at altitude? Neuropsychopharmacology 201. 40:S272–S442. Abstract, American College of Neuropsychopharmacology Annual Meeting. [Google Scholar]
- 56.Shi XF, Carlson PJ, Kim TS, et al. Effect of altitude on brain intracellular pH and inorganic phosphate levels. Psychiatry Res. 2014;222:149–156. doi: 10.1016/j.pscychresns.2014.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bogdanova OV, Abdullah O, Kanekar S, Bogdanov VB, Prescot AP, Renshaw PF. Neurochemical alterations in frontal cortex of the rat after one week of hypobaric hypoxia. Behav Brain Res. 2014;263:203–209. doi: 10.1016/j.bbr.2014.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]


