Abstract
Emerging evidence has shown that dioxin causes dysregulation of microRNAs (miRs) in a variety of tissues or cells. However, little is known about dioxin effects on neuronal miRs expression. In the present study, 277 differentially expressed miRs were identified by miRs microarray analysis in 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD, at 10−10 M) treated SK-N-SH neuroblastoma cells. Among them, 53 miRs exhibited changes of more than 0.4-fold. Consistent with the microarray data, we verified the induction effect of TCDD on hsa-miR-608 expression, which is a primate-specific miR associated with brain functions. Bioinformatics analysis showed involvement of hsa-miR-608 in cytoskeleton organization, in which one of the hsa-miR-608 target genes, Cell Division Cycle 42 (CDC42), might play a role. We also confirmed induction of CDC42 expression by TCDD in SK-N-SH cells. TCDD induced the expression of CDC42 mRNA in hsa-miR-608 inhibitor transfected cells more obviously than in control cells, suggesting involvement of both transcriptional and post-transcriptional mechanisms in the TCDD-induced CDC42 regulation. Furthermore, CH223191, an antagonist of the aryl hydrocarbon receptor (AhR), counteracted TCDD-induced hsa-miR-608 and CDC42 expression. These results indicated that AhR not only mediates transcriptional induction of CDC42, but also hsa-miR-608-induced post-transcriptional regulation of CDC42 in dioxin treated neuroblastoma cells.
Introduction
Exposure to dioxins or dioxin-like compounds (DLCs) has been associated with impaired development of the nervous system and loss of brain functions (e.g. motor coordination, emotion, cognition and psychological disorders) in experimental animals and humans1–5. Dioxins cause alterations in expression of various genes, including neurotransmission6, 7, neurodevelopment8, 9 and cytotoxicity related genes10, 11, which are considered as important mechanisms for dioxin effects. The aryl hydrocarbon receptor (AhR) is known as a key mediator of transcriptional regulation of the altered gene expression caused by dioxin exposure6, 8, in which dioxin binds to the AhR to form the dioxin-receptor complex, undergoes nuclear translocation and coupling with AhR nuclear translocator, and finally interacts with the dioxin responsive elements (DREs) in promoters of target genes to manipulate gene expression12. With the recent rise of epigenetics, emerging evidence suggested possible involvement of dioxins or DLCs in epigenetic regulation13. However, whether this epigenetic regulation by dioxins could be present and participate in the adverse effects on the nervous system remains unclear.
As one of the major epigenetic mechanisms, microRNAs (miRs), a new class of small non-coding RNAs, post-transcriptionally regulate gene expression by inhibiting mRNA translation or inducing mRNA degradation14. The miRs participate in diverse aspects of neuronal development, function and plasticity14, 15. Their dysfunction may lead to neurodevelopmental abnormalities and neurodegenerative disorders15. Brain-enriched miR-124 increases neuron formation through the repression of SRY-box transcription factor Sox9 during neurogenesis in adult mice, and promotes neurite elongation through the inhibition of ROCK1 expression in M17 cells16, 17. A primate specific miR, miR-608, inhibits the migration and invasion of glioma stem cells by targeting macrophage migration inhibitory factor18, and altering its expression in the brain could affect anxiety19.
Dioxin has been demonstrated to cause dysregulation of miRs in a variety of tissues or cells, such as miR-191-5p in HepG2 cells20, miR-335-5p in MDA-MB-231 and BT474 cells21, miR-212-3p and miR-132-3p in MDA-MB-231 and BT474 cells22, miR-25-3p and miR-92a-3p in multiple myeloma cells23. However, little is known about the effect of dioxins on miR expression in the human nervous system. Given the important function of miRs in the nervous system and the above correlation between dioxin and miRs, we hypothesize that gene alterations caused by dioxins in the nervous system may be due to both transcriptional and post-transcriptional mechanisms involving miRs.
A human neuroblastoma cell line, SK-N-SH cell, exhibits numerous biochemical characteristics of neurons, and has been employed as a model of human neurons to investigate the toxicity of dioxins6, 24. In the current study, we adopted miR microarray analyses to perform global analysis of differentially expressed miRs caused by dioxins. To make the study relevant to real environmental pollution, an average concentration of dioxin in human serum was used as the experimental treatment concentration6. Aberrant expression profiling of miRs was obtained in dioxin-treated SK-N-SH cells. Among them, a primate-specific miR was selected for further validation and function study. A target gene of this identified miR was chosen to reveal the effect of dioxin on its expression and underling mechanism for the dysregulation of the gene, including the role of AhR in regulation.
Results
Alteration in miRs expression profile in response to TCDD in SK-N-SH cells
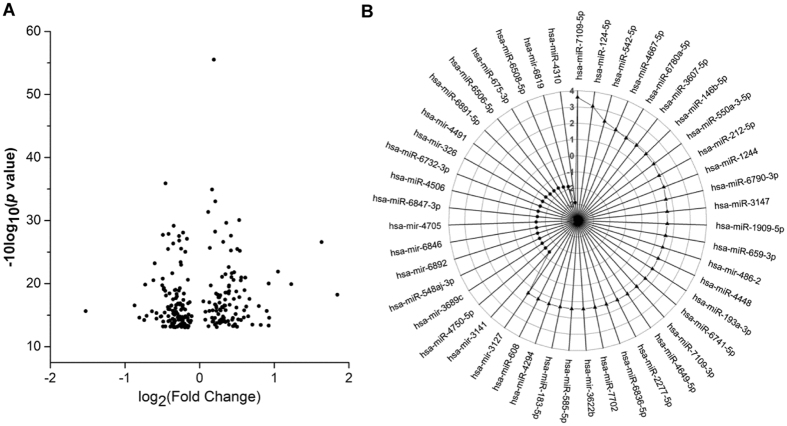
For an overview of the effects of dioxin on the expression of neuronal miRs, we analyzed the miR expression profiles using Affymetrix GeneChip miR 4.0 Arrays in SK-N-SH cells treated with 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) (at 10−10 M) or DMSO. The clustered heat map showed discrimination among all the treatment groups (Supplementary Fig. 1). In addition, a dendrogram of the cluster analysis of miRs was generated based on the similarity between different treatment groups (Supplementary Fig. 1). The biological replicates were clustered tightly together and separated clearly from the other treatment suggesting that the differences between the two groups were mainly due to the different treatments (Supplementary Fig. 1). A total of 277 miRs were differentially expressed in SK-N-SH cells treated with 10−10 M TCDD as compared with DMSO-treated cells with a cut-off of p < 0.05 (Fig. 1A and Supplementary Table 1). Among these differentially expressed miRs, 53 miRs exhibited more than 0.4-fold of changes, including 32 up-regulated miRs and 21 down-regulated miRs (Fig. 1B). According to previously published studies, 13 of these 53 miRs are related to the nervous system, including the primate specific hsa-miR-608 and human specific hsa-miR-183-5p (Table. 1). To our knowledge, dioxin-induced alterations in the expression of hsa-miR-608 or hsa-miR-183-5p have not been documented in human-derived neuronal systems.
Figure 1.
Differentially expressed miRs in response to dioxin treatment in SK-N-SH cells. Cells were treated with 10−10 M TCDD or 0.1% DMSO for 24 hr. (A) The volcano plot shows the −10log10 (p-value) on the y-axis and the fold of change (log2) on the x-axis. (B) A radar chart presenting 53 miRs exhibiting more than 0.4-fold of changes, including 32 up-regulated miRs and 21 down-regulated miRs.
Table 1.
Association of 13 differentially expressed miRs with pathological conditions in the nervous system.
| Disease/cell lines | miRBase: Accession | miR | Reference |
|---|---|---|---|
| Brain tumors | MIMAT0004762 | hsa-miR-486-3p | 31 |
| MIMAT0004591 | hsa-miR-124-5p | 32 | |
| MIMAT0000756 | hsa-miR-326 | 33–36 | |
| MIMAT0000459 | hsa-miR-193a-3p | 37 | |
| MIMAT0007882 | hsa-miR-1909-5p | 38 | |
| MIMAT0003276 | hsa-miR-608& | 18 | |
| MIMAT0002809 | hsa-miR-146b-5p | 39–41 | |
| MIMAT0000261 | hsa-miR-183-5p# | 42, 43 | |
| Neurodegenerative diseases | MIMAT0002177 | hsa-miR-486-5p | 44 |
| MIMAT0002809 | hsa-miR-146b-5p | 45–48 | |
| MIMAT0000459 | hsa-miR-193a-3p | 47, 48 | |
| MIMAT0018006 | hsa-miR-3622b-3p | 49 | |
| MIMAT0019711 | hsa-miR-4649-5p | 50 | |
| Tumors cell lines | MIMAT0004591 | hsa-miR-124-5p | 32 |
| MIMAT0002809 | hsa-miR-146b-5p | 39, 40, 51 | |
| MIMAT0000756 | hsa-miR-326 | 36, 52–54 | |
| MIMAT0003276 | hsa-miR-608 | 18, 55 | |
| MIMAT0003340 | hsa-miR-542-5p | 56 | |
| MIMAT0003337 | hsa-miR-659-3p | 57 | |
| MIMAT0000261 | hsa-miR-183-5p | 42 |
Note: & represents primate specific miR, # represents human specific miR.
Dioxin induces the expression of hsa-miR-608
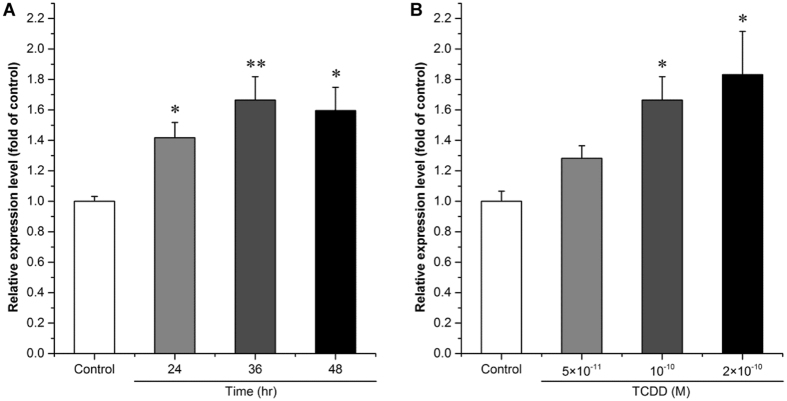
The effect of TCDD on hsa-miR-608 expression was further validated by quantitative PCR (qPCR) analysis25. We found the expression of hsa-miR-608 was significantly increased by approximately 42%, 67% and 50% after treatment with 10−10 M TCDD for 24, 36, 48 hr, respectively (Fig. 2A, p = 1.82E-02, p = 8.93E-06 and p = 2.85E-02, respectively). However, there were no significant differences between the effects at the different time points. Because 36 hr was the time point showing the greatest change in hsa-miR-608 expression, 36 hr was used to reveal the response of hsa-miR-608 expression to different concentrations of TCDD. Results showed that the expression of hsa-miR-608 was significantly increased by approximately 83% after 36 hr treatment with 2 × 10−10 M TCDD compared with DMSO treated controls (Fig. 2B, p = 1.59E-02). However, the lowest testing concentration of TCDD (5 × 10−11 M) showed no effect on hsa-miR-608 expression (Fig. 2B, p = 1.000) and there was no change in the expression of the human-specific miR, hsa-miR-183-5p, upon TCDD treatment (data not shown).
Figure 2.
Effect of TCDD on the expression of hsa-miR-608 in cultured SK-N-SH cells. SK-N-SH cells were treated with 10−10 M TCDD or 0.1% DMSO for 24 hr, 36 hr or 48 hr (A), or with 5 × 10−11, 10−10, or 2 × 10−10 M TCDD or 0.1% DMSO for 36 hr (B). Total miRs was extracted for determination of the expression level of hsa-miR-608. Quantitative PCR analyses were performed as mentioned in M & M. U6 rRNA was used as an internal control. Values were expressed as mean ± S.E. (n = 3) and each independent sample was detected in triplicate. Statistical analysis was done by t-test (A) or by one-way ANOVA with Bonferroni test (B), and *p < 0.05, **p < 0.01, compared with control (DMSO treated cells).
Predicted biological function of hsa-miR-608
Target gene searching is important for functional predictions of miRs14. From the literature, 17 genes have been reported as target genes of hsa-miR-608 (Supplementary Table 2). Because of the limitation of experimentally verified target genes of hsa-miR-608, computational prediction was included in the study26. There were 557 communal target genes predicted for hsa-miR-608 employing two widely used online software programs (Supplementary Table 3). After removing duplicate genes, 571 unique target genes were identified and subjected to functional clustering. In total, 25 Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways and 220 Gene ontology (GO) terms (including 164 Biological Processes (BP), 25 Molecular Functions (MF) and 31 Cellular Components (CC)) were found to be related to these genes using DAVID online software (Supplementary Tables 4 and 5). Based on KEGG analyses, the target genes of hsa-miR-608 were associated with several well-known neuronal signaling pathways, including hsa05214: glioma (p = 1.53E-02), hsa04720: long-term potentiation (p = 5.92E-03), hsa04360: axon guidance (p = 8.38E-04) and hsa04722: neurotrophin signaling pathway (p = 5.87E-04) (Fig. 3A). Gene Ontology analysis showed that hsa-miR-608 target genes are involved in a series of important cellular and molecular events such as GO: 0030036 ~actin cytoskeleton organization (p = 1.68E-02), GO: 0030029 ~actin filament-based process (p = 1.32E-02), GO: 0007264 ~small GTPase mediated signal transduction (p = 2.32E-02), GO: 0031175 ~neuron projection development (p = 2.96E-04) and GO: 0030030 ~cell projection organization (p = 4.16E-06) (Fig. 3B). Among the genes associated with these neural biological functions, Cell Division Cycle 42 (CDC42) was the one present with the highest frequency and selected for further study.
Figure 3.
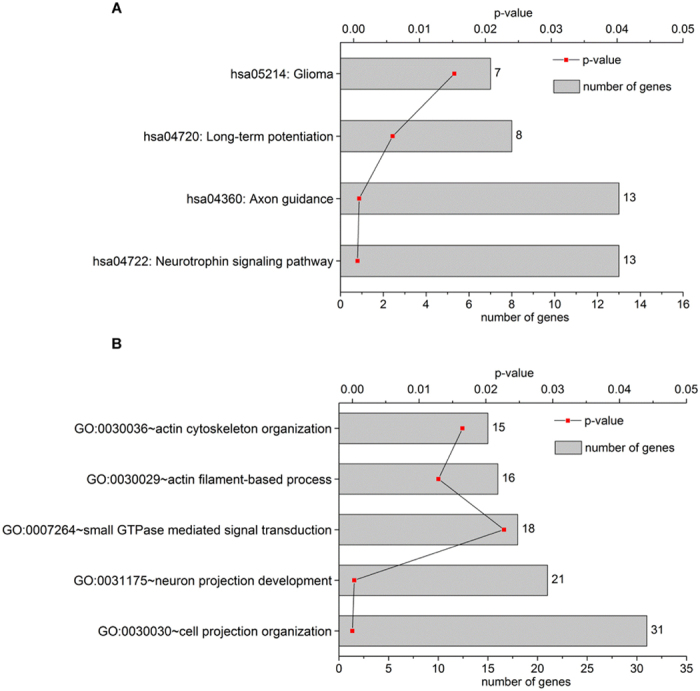
Functional annotations related to the nervous system for hsa-miR-608 target genes. KEGG pathway (A) and GO (B) cluster analyses were performed via DAVID website software. The vertical axis shows the annotated functions of the target genes. The horizontal axis shows p-value and the gene number of each cluster.
Involvement of hsa-miR-608 in gene regulation of CDC42 by dioxin
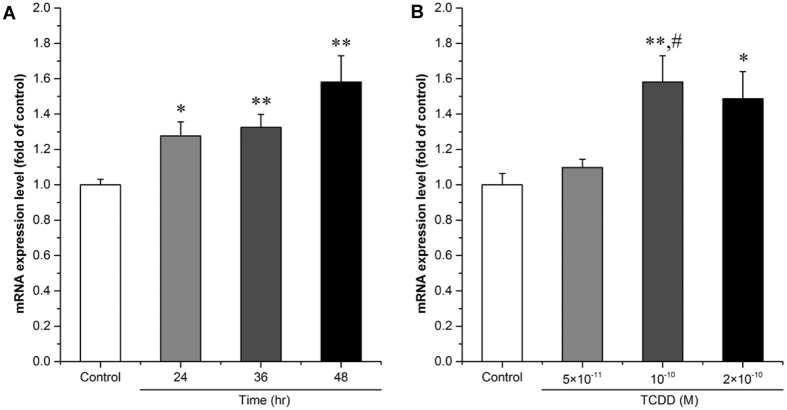
The effects of TCDD on the expression of CDC42 were verified in SK-N-SH cells. The expression of CDC42 gradually increased with time increase and showed significant induction by approximately 28%, 32% and 58% upon TCDD (10−10 M) treatment at different time points compared with the DMSO controls, respectively (Fig. 4A, p = 2.39E-02, p = 6.43E-03 and p = 2.44E-03, respectively). Because 48 hr was the time point showing the greatest change in CDC42 expression, this treatment time was used for the following experiments. The expression of CDC42 was significantly increased by approximately 49% after treatment with 2 × 10−10 M TCDD compared with DMSO treated controls (Fig. 4B, p = 3.58E-02). Significant change (approximately 44%) was found in the 10−10 M TCDD treated group compared with the DMSO treated control (Fig. 4B, p = 3.72E-02), however, the lowest concentration of TCDD (5 × 10−11 M) showed no effect on CDC42 expression (Fig. 4B, p = 1.000).
Figure 4.
Effect of TCDD on the expression of CDC42 in cultured SK-N-SH cells. Cells were treated with 10−10 M TCDD or 0.1% DMSO for 24 hr, 36 hr or 48 hr (A), or with 5 × 10−11, 10−10, or 2 × 10−10 M TCDD or 0.1% DMSO for 48 hr (B). Expression level of the CDC42 mRNAs was determined by qPCR analysis. 18 S rRNA was used as an internal control. Values are expressed as mean ± S.E. from triplicate samples in three independent experiments. Statistical analysis was done by one-way ANOVA with Bonferroni test. *p < 0.05 and **p < 0.01, compared with control (DMSO treated cells). #p < 0.01 compared with 5 × 10−11 M TCDD.
In line with the finding in human breast cancer cells, the mRNA expression of CDC42 was suppressed in SK-N-SH neuroblastoma cells transfected with an hsa-miR-608 mimic (Supplementary Fig. 2, p = 3.36E-02). Moreover, the mRNA expression of CDC42 was increased in hsa-miR-608 inhibitor transfected cells compared to a negative control (NC), further supporting the notion that hsa-miR-608 can suppress gene expression of neuronal CDC42 (Fig. 5, p = 1.26E-03). To investigate the role of hsa-miR-608 in neuronal CDC42 gene regulation by dioxin, SK-N-SH cells transfected with the hsa-miR-608 inhibitor or NC were treated with TCDD or solvent. We found TCDD induced the mRNA expression of CDC42 in hsa-miR-608 inhibitor transfected cells more obviously than in NC transfected cells, suggesting involvement of hsa-miR-608 mediated suppression in the dysregulation of CDC42 by dioxin (Fig. 5, approximately 118% versus 30% of induction, p = 9.12E-10). On the other hand, in hsa-miR-608 inhibitor transfected cells, the TCDD treatment group had a significant induction effect on CDC42 mRNA expression compared to the solvent treated group, suggesting that a mechanism other than hsa-miR-608 induced suppression is involved in CDC42 regulation upon TCDD treatment (Fig. 5, approximately 118% versus 47% of induction, p = 3.23E-05). This mechanism may involve transcriptional induction of CDC42 by TCDD.
Figure 5.
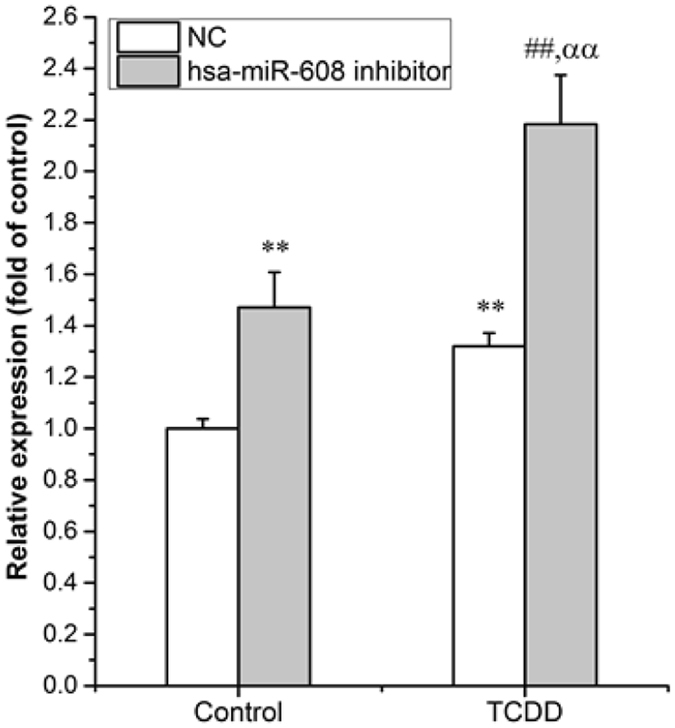
Involvement of hsa-miR-608 in gene regulation of CDC42 by dioxin. Cells were transfected with hsa-miR-608 inhibitors or NC for 24 hr, followed by 10−10 M TCDD treatment for an additional 36 hr. Total mRNAs was extracted for determination of the expression level of CDC42. Quantitative PCR analyses were performed as mentioned in M & M. 18 S rRNA was used as an internal control. Values are expressed as mean ± S.E. from triplicate samples in three independent experiments. Statistical analysis was done by one-way ANOVA with Bonferroni test. **p < 0.01 compared with Control (DMSO treated cells) & NC (NC transfected cells) group, ##p < 0.01 compared with Control (DMSO treated cells) & hsa-miR-608 inhibitor (Inhibitor transfected cells) group, ααp < 0.01 compared with TCDD (TCDD treated cells) & NC (NC transfected cells) group.
Involvement of AhR in regulation of CDC42 by dioxin
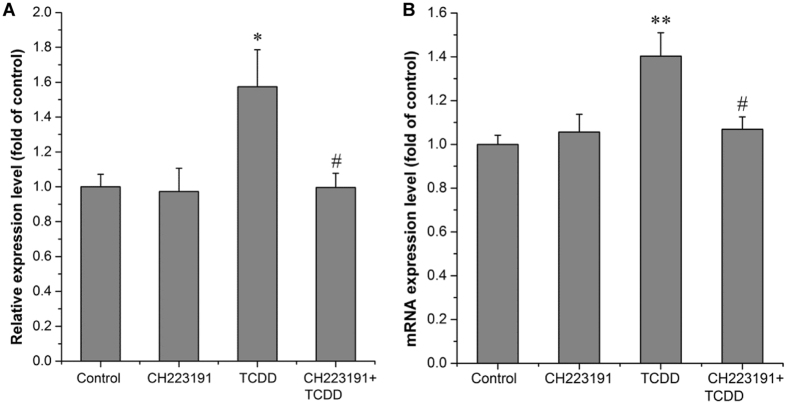
We further explored the role of AhR in the regulation of CDC42 by dioxin. Cells were treated with CH223191, a ligand-selective antagonist of the AhR, before TCDD or solvent treatment. We found that the induction of hsa-miR-608 by TCDD was totally reversed by CH223191 treatment (Fig. 6A). Moreover, the pretreatment with CH223191 could eliminate the effect of TCDD on CDC42 expression as well (Fig. 6B). These results suggested that AhR might be a communal upstream molecule for both the transcriptional induction of CDC42 and the post-transcriptional suppression involving hsa-miR-608.
Figure 6.
TCDD induces hsa-miR-608 and CDC42 expression via AhR-dependent pathway. Cells were treated with 10−6 M CH223191 (AhR antagonist) or 0.01% DMSO for 3 hr before treatment with 10−10 M TCDD or 0.01% DMSO for 36 hr (A) and 24 hr (B). Total miRs/mRNA was extracted for determination of the expression level of hsa-miR-608 (A) or CDC42 (B). Quantitative PCR analyses were performed as mentioned in M & M. U6 rRNA was used as an internal control for hsa-miR-608 and 18 S rRNA was used as an internal control for CDC42. Values were expressed as mean ± S.E. (n = 3) and each independent sample was detected in triplicate. Statistical analysis was done by one-way ANOVA with Bonferroni test. *p < 0.05 and **p < 0.01compared with control (DMSO treated cells). #p < 0.05 compared with TCDD treatment alone.
Discussion
Dioxin is not only involved in the regulation of gene expression at the transcriptional level, but also at the post-transcriptional level, such as through miRs20–23. The expression of miRs can be influenced by various external environment conditions including dioxin exposure27. MiR microarray technology is a powerful high throughput tool capable of revealing alterations in miR expression profiles, which benefits global understanding of the post-transcriptional molecular bases of dioxin effects or toxicities28, 29. However, to our knowledge, there is no systematic study on dioxin induced miR alterations in models of the human nervous system. In the present study, 277 differentially expressed miRs were found in dioxin treated SK-N-SH cells. Among these differentially expressed miRs, 53 miRs exhibited more than 0.4-fold of changes in TCDD treated groups (0.1 nM). Other studies looking at TCDD treatments have observed even larger changes in miR expression (upwards to 2.5-fold), but they also used higher concentrations of TCDD, which could partially explain a stronger response in expression levels20–23. The relatively low TCDD concentration used in the present study was close to the average serum concentration of TCDD (0.01–0.1 nM) among individuals exposed to dioxins in Vietnam, Seveso Italy and Taiwan6. Thus, the overall changes in neuronal miR expression profiles could model environmentally relevant outcomes, which makes us more confident to further discuss the health impact association of the dioxin-induced miRs alterations.
Emerging evidence shows that miRs play important roles in human disease and cancer30. According to the literature, 13 miRs with more than 0.4-fold changes in the present microarray dataset are involved in neurodegenerative diseases or brain tumors, suggesting that dioxin may cause these health impacts through miRs31–57. Primate and human specific miRs have been considered as powerful sources for cancer biomarkers and novel therapeutic target studies58. Thus, primate or human-specific miRs from the present dataset might provide specific clues for further toxicological relevance analysis. The primate-specific miR, hsa-miR-608 was identified from the dataset and verified to be induced by TCDD treatment in human neuroblastoma cells. In the functional prediction of hsa-miR-608, association with glioma and/or cytoskeleton organization was found, which is consistent with the reported correlation of hsa-miR-608 with the migration of glioma stem cells18. Therefore, we hypothesize that glioma and/or cytoskeleton related defects might be a potential outcome of dioxin exposure in the nervous system, in which dioxin-induced dysregulation of hsa-miR-608 and/or its target genes might play roles.
CDC42, a known target gene of hse-miR-608 in a human macrophage cell line U937 and a human breast cancer cell line MT-119, 59, is a small GTPase of the Rho family involved in cytoskeleton reorganization and cell migration and motility60. In this study, by using hsa-miR-608 mimic and inhibitor, we confirmed that hsa-miR-608 could target CDC42 and down-regulate its expression in SK-N-SH neuroblastoma cells. More importantly, we first reported that TCDD treatment could induce the expression of hsa-miR-608, which subsequently could enhance one of the post-transcriptional down-regulation processes of CDC42. However, the overall effect of TCDD treatment on CDC42 was the up-regulation of its mRNA level, suggesting that apart from the post-transcriptional down-regulation via hsa-miR-608, another up-regulatory mechanism for CDC42 expression could be involved in the dioxin effect. The presence of DRE core sequences (5′- GCGTG- 3′) in the promoter region is a common feature of classical dioxin-responsive genes12, 61. According to human CDC42 promoter sequence analysis (−2500 to + 50 bp)62, five putative DREs consensus sequences were found, suggesting a possible capability of TCDD to control CDC42 expression at the transcriptional level. This is supported by the finding that CDC42 expression was increased through over-expression of AhR in mouse Neuro2a neuroblastoma cells63. Therefore, we hypothesize that transcriptional up-regulation via AhR pathway might participate the dioxin-mediated CDC42 induction in the present human neuroblastoma cells. This multilevel regulation of TCDD on CDC42 expression could explain our present finding that TCDD treatment in hsa-miR-608 inhibitor transfected cells could induce CDC42 mRNA expression more obviously than that of NC transfected cells.
It is well-known that AhR mediates dioxin-induced transcriptional regulation of a series of genes. Emerging evidence has shown that these dioxin-responsive genes include not only protein-coding genes, but also non-coding genes, such as miRs20–23. In this study, by using the AhR antagonist, we demonstrated hsa-miR-608 as a novel dioxin-responsive non-coding gene regulated by the AhR-dependent pathway. Hsa-miR-608, located within the intron region of its host gene SEMA4G, has been hypothesized to share a common promoter with the host64. Through bioinformatics analysis, three DREs consensus sequences were found in the putative promoter region (chr10:100970529-100972528, the current human genome GRCh38 assembly) of hsa-miR-608, further supporting the involvement of AhR-dependent signaling in the regulation. Thus, AhR might be a communal upstream signaling molecule in the transcriptional induction and the post-transcriptional suppression of CDC42 by dioxin. In line with this notion, the alteration of CDC42 induced by TCDD was almost completely reversed by AhR angonist treatment. Moreover, the list of miRs involved in the AhR-mediated multilevel regulation of CDC42 by dioxin is still open. Based on the present miR array dataset, another up-regulated miR, has-miR-6836, is predicted to target to CDC42. It shares promoter with its host gene SNX8, which contains 10 putative consensus DRE sequences. However, the role of has-miR-6836 in dioxin-induced CDC42 dysregulation needs further investigation. Given the association of CDC42 and AhR with cytoskeleton organization65, this AhR mediated multilevel regulation of CDC42 by dioxin might result in interference of cytoskeleton-related cellular processes of the neurons, which merits further investigation.
In conclusion, we showed that dioxin could cause alterations in expression of a considerable fraction of miRs in human SK-N-SH neuroblastoma cells. Some differentially expressed miRs, including primate-specific hsa-miR-608, are known to be correlated with pathological conditions in the nervous system. Hsa-miR-608 was validated to be transcriptionally up-regulated upon TCDD treatment, which might be related to several cellular events in neurons, such as neuron differentiation, neuron projection, and actin cytoskeleton organization. CDC42, a cytoskeleton organization related gene, was demonstrated as a target gene of hsa-miR-608 in SK-N-SH cells, which could be significantly up-regulated upon TCDD treatment. Both transcriptional and hsa-miR-608 related post-transcriptional mechanisms are involved in the regulation of CDC42 caused by dioxin, in which AhR is an important upstream mediator.
Materials and Methods
Cell culture
SK-N-SH, a cell line derived from human neuroblastoma cells, was purchased from the cell resource center of the Chinese Academy of Medical Sciences (Beijing, China). Cells were maintained in Dulbecco′s modified Eagle’s medium (DMEM, Gibco, Paisley, Scotland, UK), supplemented with 10% fetal bovine serum (FBS, Corning, NY, USA), and 1% penicillin–streptomycin (Gibco, Paisley, Scotland, UK). Cells were cultured at 37 °C in a 5% CO2 humidified incubator.
Exposure experiments
2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), the most potent congener of dioxins, was purchased from Wellington Laboratories Inc. (Ontario, Canada) and dissolved in dimethyl sulfoxide (DMSO). Cells were seeded in culture dishes at an appropriate density for 24–48 hr. At 70% confluence, cells were treated with environmentally relevant low concentrations of TCDD6. DMSO was present at 0.1% or lower for all treatments. CH223191, an antagonist of AhR, was obtained from Sigma (St. Louis, MO, USA) and used at a concentration of 10−6 M. After treatments, cells were washed with phosphate-buffered saline (PBS, pH 7.4) twice and prepared for miR microarray and other experimental processes.
MiRs isolation and miR microarray analysis
Total RNA containing miRs was extracted from SK-N-SH cells treated with 0.1% DMSO or 10−10 M TCDD for 24 hr. Trizol reagent (Invitrogen, Carlsbad, USA) followed by miRNeasy kit (Qiagen, Germany) were used for enrichment of miRs. This is a common method of sample processing for the miR microarray66. In brief, cells were lysed in Trizol/chloroform reagent according to the manufacturer’s instruction. The mixture was centrifuged at 12,000 g for 15 min at 4 °C, and the upper aqueous phase was mixed with 1.5 volume of 100% ethanol. This mixture was then processed using an miRNeasy kit according to the manufacturer’s instruction. The total RNAs of three control groups and three TCDD-treated groups were separately hybridized to Affymetrix GeneChip miR 4.0 Arrays each containing 2578 human mature miR probe sets. The arrays were scanned by means of a Affymetrix GeneChip Scanner 3000 7 G system (Affymetrix, USA). Scanned images (CEL format) were imported into Affymetrix Expression Console software (version 1.4.1; Affymetrix, USA) for normalization by Robust Multichip Average (RMA) algorithm, and the generated CHP files were imported into Affymetrix Transcriptome Analysis Console software (version 3.0; Affymetrix, USA) for further analysis. miRs expression data were statistically analysed using one-way ANOVA, and p value < 0.05 was considered as a significant change. The miRs with more than 0.4-fold of changes were further investigated.
A review on association of candidate miRs with the nervous system
Published literature in English until February 2017 was searched for relevant studies. Articles were primarily collected from database searches in PubMed, Web of Science and Google Scholar. The search string referenced the miRs (more than 0.4-fold of changes) and neurodevelopmental abnormalities or neurodegenerative disorders (e.g., cognition, memory, neurobehavior, glioma, glioblastoma, glioblastoma multiforme, medulloblastoma, brain tumors, Alzheimer’s Disease, Parkinson Disease, Amyotrophic Lateral Sclerosis Disease, Myotonic Dystrophy Type 2 Disease, Huntington’s Disease, Neurofibromatosis Type 1 Disease). All terms were searched using both controlled vocabulary and free text words in titles and abstracts. In addition, studies that did not contain original data, such as reviews, editorials or conference abstracts, were excluded.
Quantitative analysis of candidate miRs
MiRs from SK-N-SH cells were isolated by miRcute miR isolation kit (Tiangen Biotech, Beijing, China) according to the manufacturer’s instruction. In brief, cells were lysed with Buffer MZ/chloroform reagent (the similar function as Trizol/chloroform reagent). The mixture was centrifuged at 12,000 g for 15 min at 4 °C, and the upper aqueous phase was transferred to an absorption column in the miR extraction kit. The obtained mixture was then processed according to the manufacturer’s instruction. The extracted products were reverse transcribed to cDNA using the miRcute miR First-Strand cDNA synthesis kit (Tiangen Biotech). cDNAs were stored at −20 °C. All procedures were carried out according to the instructions provided by the manufacturer. Expression levels of the mature miRs were quantified by qPCR analysis with small nuclear RNA U6 (snRNA) as an internal reference for normalization. The qPCR analysis was performed on a LightCycler Roche 480 instrument (LC-480II, Roche, USA) using miRcute miR qPCR detection kit (SYBR Green) (Tiangen Biotech). Forward and reverse primers were provided by Tiangen Biotech. The qPCR analyses were performed under the following conditions: 94 °C 2 min, 1 cycle; 94 °C 20 s, 60 °C 34 s, 40 cycles. The data were analyzed using a ∆∆Ct method67.
Bioinformatics analysis
Target genes of miRs were predicted by updated versions of TargetScan v7.0 (updated: Aug 2015, http://www.targetscan.org/vert_71/) and miRDB (updated: Mar 2015, http://www.mirdb.org/miRDB/). Communal predicted target genes of the miRs from the two tools were taken into account. The predicted target genes were then subjected to KEGG enrichment analyses and GO analysis using DAVID (the Database for Annotation, Visualization and Integrated Discovery, https://david.ncifcrf.gov/home.jsp) with the total human genome information as the background.
Cell transfection
The cells were seeded in 12-well plates at a concentration of 2 × 105 cell/well one day before transfection. The NC, hsa-miR-608 mimics or inhibitor (GenePharma, China) at 50 nM for each well were introduced into the cells using Lipofectamine RNAiMAX transfection reagent (Invitrogen) according to the manufacturer’s instruction. Twenty-four hours after transfection, cells were subjected directly to qPCR analyses or to treatment with TCDD at 10−10 M for 36 hr followed by the gene expression testing.
Quantitative analysis of the selected gene
For revealing mRNA levels of the selected gene, CDC42, total RNAs were extracted from SK-N-SH cells using RNAprep pure Cell/Bacteria Kit (Tiangen Biotech) according to manufacturer’s instructions. Total RNA (2.5 µg) was reverse transcribed using Thermo Scientific RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific) according to the manufacturer’s instructions. The expression level of CDC42 was quantified by qPCR using GoTaq® qPCR Master Mix kit according to the manufacturer’s instructions (Promega, Madison, WI, USA). The SYBR green signal was detected by the LightCycler 480 Instrument (LC-480II, Roche). The conditions of the qPCR were as follows: 5 min at 95 °C, followed by 45 cycles each consisting of 10 s at 95 °C, 20 s at 60 °C and 30 s at 72 °C, and a cooling step of 16 s at 42 °C. The data were analyzed using a ∆∆Ct method67. The sequences of mRNA primer pairs used were as follows: CDC42 (NM_001791.3), forward primer 5′-GATGGTGCTGTTGGTAAA-3′ and reverse primer 5′-TAACTCAGCGGTCGTAAT-3′; 18 S (NR_003286), forward primer 5′-CGCCGCTAGAGGTGAAATTC-3′ and reverse primer 5′-TTGGCAAATGCTTTCGCTC-3′.
Statistical analysis
Data are expressed as mean ± S.E. from at least three independent experiments. The differences were assessed using one-way ANOVA test with Bonferroni correction using IBM SPSS19.0 software (IBM Corp., Armonk, NY) and p < 0.05 (*) or p < 0.01 (**) was considered to be statistically significant. The charts were drawn by Origin 9.0 software (OriginLab, MA).
Electronic supplementary material
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21377160, 21525730), the Strategic Priority Research Program of the Chinese Academy of Sciences (No. XDB14030401, XDB14030402), and we thank Dr. Marjorie A. Phillips from UC Davis for comments during the manuscript preparation.
Author Contributions
H.Q.X. and B.Z. conceived the study. T.X. and H.Q.X. designed the experiments. T.X., Y.P.L., Y.J.X., Y.S.C. and L.Y.W. performed the experiments. T.X., H.Q.X., L.X. and B.Z. analyzed and interpreted the data. H.Q.X. and T.X. wrote the manuscript. All authors reviewed the manuscript and approved the final version.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Tuan Xu and Heidi Q. Xie contributed equally to this work.
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-10311-3
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hill A, Howard CV, Strahle U, Cossins A. Neurodevelopmental defects in zebrafish (Danio rerio) at environmentally relevant dioxin (TCDD) concentrations. Toxicological sciences: an official journal of the Society of Toxicology. 2003;76:392–399. doi: 10.1093/toxsci/kfg241. [DOI] [PubMed] [Google Scholar]
- 2.Kakeyama M, Endo T, Zhang Y, Miyazaki W, Tohyama C. Disruption of paired-associate learning in rat offspring perinatally exposed to dioxins. Archives of toxicology. 2014;88:789–798. doi: 10.1007/s00204-013-1161-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Boucher O, Muckle G, Bastien CH. Prenatal exposure to polychlorinated biphenyls: a neuropsychologic analysis. Environmental health perspectives. 2009;117:7–16. doi: 10.1289/ehp.11294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tran NN, et al. Impacts of Perinatal Dioxin Exposure on Motor Coordination and Higher Cognitive Development in Vietnamese Preschool Children: A Five-Year Follow-Up. PloS one. 2016;11 doi: 10.1371/journal.pone.0147655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bouchard MF, Oulhote Y, Sagiv SK, Saint-Amour D, Weuve J. Polychlorinated biphenyl exposures and cognition in older U.S. adults: NHANES (1999-2002) Environmental health perspectives. 2014;122:73–78. doi: 10.1289/ehp.1306532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xie, H. Q. et al. AhR-mediated effects of dioxin on neuronal acetylcholinesterase expression in vitro. (2013). [DOI] [PMC free article] [PubMed]
- 7.Sans JDP, et al. Developmental exposure to 2,3,7,8-tetrachlorodibenzo- p -dioxin may alter LH release patterns by abolishing sex differences in GABA/glutamate cell number and modifying the transcriptome of the male anteroventral periventricular nucleus. Neuroscience. 2016;329:239–253. doi: 10.1016/j.neuroscience.2016.04.051. [DOI] [PubMed] [Google Scholar]
- 8.Gohlke JM, Stockton PS, Sieber S, Foley J, Portier CJ. AhR-mediated gene expression in the developing mouse telencephalon. Reproductive toxicology. 2009;28:321–328. doi: 10.1016/j.reprotox.2009.05.067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kimura E, et al. In utero and lactational dioxin exposure induces Sema3b and Sema3g gene expression in the developing mouse brain. Biochemical & Biophysical Research Communications. 2016;476:108–113. doi: 10.1016/j.bbrc.2016.05.048. [DOI] [PubMed] [Google Scholar]
- 10.Li Y, et al. 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) induces microglial nitric oxide production and subsequent rat primary cortical neuron apoptosis through p38/JNK MAPK pathway. Toxicology. 2013;312:132–141. doi: 10.1016/j.tox.2013.08.008. [DOI] [PubMed] [Google Scholar]
- 11.Xu G, et al. 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) induces expression of p27kip1 and FoxO3a in female rat cerebral cortex and PC12 cells. Toxicology letters. 2014;226:294–302. doi: 10.1016/j.toxlet.2014.02.019. [DOI] [PubMed] [Google Scholar]
- 12.Beischlag, T. V., Morales, J. L., Hollingshead, B. D. & Perdew, G. H. The aryl hydrocarbon receptor complex and the control of gene expression. Critical Reviews™ in Eukaryotic Gene Expression 18 (2008). [DOI] [PMC free article] [PubMed]
- 13.Xie HQ, et al. New perspectives for multi-level regulations of neuronal acetylcholinesterase by dioxins. Chemico-biological interactions. 2016 doi: 10.1016/j.cbi.2016.06.030. [DOI] [PubMed] [Google Scholar]
- 14.Selbach M, et al. Widespread changes in protein synthesis induced by microRNAs. Nature. 2008;455:58–63. doi: 10.1038/nature07228. [DOI] [PubMed] [Google Scholar]
- 15.Fiore R, Siegel G, Schratt G. MicroRNA function in neuronal development, plasticity and disease. Biochimica et biophysica acta. 2008;1779:471–478. doi: 10.1016/j.bbagrm.2007.12.006. [DOI] [PubMed] [Google Scholar]
- 16.Cheng LC, Pastrana E, Tavazoie M, Doetsch F. miR-124 regulates adult neurogenesis in the subventricular zone stem cell niche. Nat Neurosci. 2009;12:399–408. doi: 10.1038/nn.2294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gu X, et al. miR-124 represses ROCK1 expression to promote neurite elongation through activation of the PI3K/Akt signal pathway. Journal of molecular neuroscience: MN. 2014;52:156–165. doi: 10.1007/s12031-013-0190-6. [DOI] [PubMed] [Google Scholar]
- 18.Wang Z, Xue Y, Wang P, Zhu J, Ma J. MiR-608 inhibits the migration and invasion of glioma stem cells by targeting macrophage migration inhibitory factor. Oncology reports. 2016;35:2733–2742. doi: 10.3892/or.2016.4652. [DOI] [PubMed] [Google Scholar]
- 19.Hanin G, et al. Competing targets of microRNA-608 affect anxiety and hypertension. Human molecular genetics. 2014;23:4569–4580. doi: 10.1093/hmg/ddu170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Elyakim E, et al. hsa-miR-191 is a candidate oncogene target for hepatocellular carcinoma therapy. Cancer research. 2010;70:8077–8087. doi: 10.1158/0008-5472.CAN-10-1313. [DOI] [PubMed] [Google Scholar]
- 21.Zhang S, et al. Aryl hydrocarbon receptor agonists induce microRNA-335 expression and inhibit lung metastasis of estrogen receptor negative breast cancer cells. Molecular cancer therapeutics. 2012;11:108–118. doi: 10.1158/1535-7163.MCT-11-0548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hanieh H. Aryl hydrocarbon receptor-microRNA-212/132 axis in human breast cancer suppresses metastasis by targeting SOX4. Mol Cancer. 2015;14 doi: 10.1186/s12943-015-0443-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gordon MW, et al. Regulation of p53-targeting microRNAs by polycyclic aromatic hydrocarbons: Implications in the etiology of multiple myeloma. Molecular carcinogenesis. 2014 doi: 10.1002/mc.22175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Xu HM, et al. Dioxin and dioxin-like compounds suppress acetylcholinesterase activity via transcriptional downregulations in vitro. Journal of molecular neuroscience: MN. 2014;53:417–423. doi: 10.1007/s12031-013-0167-5. [DOI] [PubMed] [Google Scholar]
- 25.Backes C, et al. Bias in High-Throughput Analysis of miRNAs and Implications for Biomarker Studies. Analytical chemistry. 2016;88:2088–2095. doi: 10.1021/acs.analchem.5b03376. [DOI] [PubMed] [Google Scholar]
- 26.Agarwal V, Bell GW, Nam JW, Bartel DP. Predicting effective microRNA target sites in mammalian mRNAs. Elife 4. 2015 doi: 10.7554/eLife.05005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Inukai S, Slack F. MicroRNAs and the Genetic Network in Aging. Journal of molecular biology. 2013;425:3601–3608. doi: 10.1016/j.jmb.2013.01.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Singh NP, Singh UP, Guan H, Nagarkatti P, Nagarkatti M. Prenatal exposure to TCDD triggers significant modulation of microRNA expression profile in the thymus that affects consequent gene expression. PloS one. 2012;7 doi: 10.1371/journal.pone.0045054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Moffat ID, et al. microRNAs in adult rodent liver are refractory to dioxin treatment. Toxicological sciences: an official journal of the Society of Toxicology. 2007;99:470–487. doi: 10.1093/toxsci/kfm189. [DOI] [PubMed] [Google Scholar]
- 30.Di Leva G, Garofalo M, Croce CM. MicroRNAs in cancer. Annual review of pathology. 2014;9 doi: 10.1146/annurev-pathol-012513-104715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Masliah-Planchon J, et al. MicroRNAome profiling in benign and malignant neurofibromatosis type 1-associated nerve sheath tumors: evidences of PTEN pathway alterations in early NF1 tumorigenesis. BMC genomics. 2013;14 doi: 10.1186/1471-2164-14-473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen Q, et al. MiR-124-5p inhibits the growth of high-grade gliomas through posttranscriptional regulation of LAMB1. Neuro-oncology. 2014;16:637–651. doi: 10.1093/neuonc/not300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Qiu S, et al. Interactions of miR-323/miR-326/miR-329 and miR-130a/miR-155/miR-210 as prognostic indicators for clinical outcome of glioblastoma patients. Journal of translational medicine. 2013;11 doi: 10.1186/1479-5876-11-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang S, et al. Expression and clinical significance of microRNA-326 in human glioma miR-326 expression in glioma. Medical oncology. 2013;30:1–6. doi: 10.1007/s12032-012-0373-y. [DOI] [PubMed] [Google Scholar]
- 35.Ferretti E, et al. Concerted microRNA control of Hedgehog signalling in cerebellar neuronal progenitor and tumour cells. The EMBO journal. 2008;27:2616–2627. doi: 10.1038/emboj.2008.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Du W, et al. Targeting the SMO oncogene by miR-326 inhibits glioma biological behaviors and stemness. Neuro-oncology. 2015;17:243–253. doi: 10.1093/neuonc/nou217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kunder R, et al. Real-time PCR assay based on the differential expression of microRNAs and protein-coding genes for molecular classification of formalin-fixed paraffin embedded medulloblastomas. Neuro-oncology. 2013;15:1644–1651. doi: 10.1093/neuonc/not123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Braoudaki M, et al. MicroRNA expression profiles in pediatric dysembryoplastic neuroepithelial tumors. Medical oncology. 2015;33:1–7. doi: 10.1007/s12032-015-0719-3. [DOI] [PubMed] [Google Scholar]
- 39.Li Y, et al. miR-146b-5p inhibits glioma migration and invasion by targeting MMP16. Cancer letters. 2013;339:260–269. doi: 10.1016/j.canlet.2013.06.018. [DOI] [PubMed] [Google Scholar]
- 40.Liu J, et al. miR-146b-5p functions as a tumor suppressor by targeting TRAF6 and predicts the prognosis of human gliomas. Oncotarget. 2015;6:29129–29142. doi: 10.18632/oncotarget.4895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Srinivasan, S., Patric, I. R. P. & Somasundaram, K. A Ten-microRNA Expression Signature Predicts Survival in Glioblastoma. PloS one6, doi:10.1371/journal.pone.0017438 (2011). [DOI] [PMC free article] [PubMed]
- 42.Tanaka H, et al. MicroRNA-183 upregulates HIF-1α by targeting isocitrate dehydrogenase 2 (IDH2) in glioma cells. Journal of neuro-oncology. 2013;111:273–283. doi: 10.1007/s11060-012-1027-9. [DOI] [PubMed] [Google Scholar]
- 43.Ye Z, et al. Upregulation of miR-183 expression and its clinical significance in human brain glioma. Neurological Sciences. 2016;37:1341–1347. doi: 10.1007/s10072-016-2599-5. [DOI] [PubMed] [Google Scholar]
- 44.Hoss AG, et al. Study of plasma-derived miRNAs mimic differences in Huntington’s disease brain. Movement disorders: official journal of the Movement Disorder Society. 2015;30:1961–1964. doi: 10.1002/mds.26457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cogswell JP, et al. Identification of miRNA changes in Alzheimer’s disease brain and CSF yields putative biomarkers and insights into disease pathways. Journal of Alzheimer’s disease. 2008;14:27–41. doi: 10.3233/JAD-2008-14103. [DOI] [PubMed] [Google Scholar]
- 46.Wang WX, Huang Q, Hu Y, Stromberg AJ, Nelson PT. Patterns of microRNA expression in normal and early Alzheimer’s disease human temporal cortex: white matter versus gray matter. Acta neuropathologica. 2011;121:193–205. doi: 10.1007/s00401-010-0756-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Dong H, et al. A panel of four decreased serum microRNAs as a novel biomarker for early Parkinson’s disease. Biomarkers: biochemical indicators of exposure, response, and susceptibility to chemicals. 2016;21:129–137. doi: 10.3109/1354750X.2015.1118544. [DOI] [PubMed] [Google Scholar]
- 48.Greco S, et al. Deregulated MicroRNAs in Myotonic Dystrophy Type 2. PloS one. 2012;7 doi: 10.1371/journal.pone.0039732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Denk J, et al. MicroRNA Profiling of CSF Reveals Potential Biomarkers to Detect Alzheimer’s Disease. PloS one. 2015;10 doi: 10.1371/journal.pone.0126423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Takahashi I, et al. Identification of plasma microRNAs as a biomarker of sporadic Amyotrophic Lateral Sclerosis. Molecular Brain. 2015;8:1–9. doi: 10.1186/s13041-015-0161-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yang W, et al. MiR-146b-5p overexpression attenuates stemness and radioresistance of glioma stem cells by targeting HuR/lincRNA-p21/beta-catenin pathway. Oncotarget. 2016 doi: 10.18632/oncotarget.9214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhou J, et al. MicroRNA-326 Functions as a Tumor Suppressor in Glioma by Targeting the Nin One Binding Protein (NOB1) PloS one. 2013;8 doi: 10.1371/journal.pone.0068469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wolter M, Werner T, Malzkorn B, Reifenberger G. Role of microRNAs Located on Chromosome Arm 10q in Malignant Gliomas. Brain pathology. 2016;26:344–358. doi: 10.1111/bpa.12294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kefas B, et al. Pyruvate kinase M2 is a target of the tumor-suppressive microRNA-326 and regulates the survival of glioma cells. Neuro-oncology. 2010;12:1102–1112. doi: 10.1093/neuonc/noq080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhang Y, Schiff D, Park D, Abounader R. MicroRNA-608 and microRNA-34a regulate chordoma malignancy by targeting EGFR, Bcl-xL and MET. PloS one. 2014;9 doi: 10.1371/journal.pone.0091546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bray I, et al. MicroRNA-542-5p as a novel tumor suppressor in neuroblastoma. Cancer letters. 2011;303:56–64. doi: 10.1016/j.canlet.2011.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Piscopo P, et al. Reduced miR-659-3p Levels Correlate with Progranulin Increase in Hypoxic Conditions: Implications for Frontotemporal Dementia. Frontiers in molecular neuroscience. 2016;9 doi: 10.3389/fnmol.2016.00031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Koufaris, C. Human and primate‐specific microRNAs in cancer: Evolution, and significance in comparison with more distantly‐related research models. BioEssays: news and reviews in molecular, cellular and developmental biology (2016). [DOI] [PubMed]
- 59.Jeyapalan Z, et al. Expression of CD44 3′-untranslated region regulates endogenous microRNA functions in tumorigenesis and angiogenesis. Nucleic Acids Res. 2011;39:3026–3041. doi: 10.1093/nar/gkq1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Liu Y, et al. Inactivation of Cdc42 in neural crest cells causes craniofacial and cardiovascular morphogenesis defects. Developmental biology. 2013;383:239–252. doi: 10.1016/j.ydbio.2013.09.013. [DOI] [PubMed] [Google Scholar]
- 61.Manabu Nukaya SM, Christopher A. Bradfield. The role of the dioxin-responsive element cluster between the Cyp1a1 and Cyp1a2 loci in aryl hydrocarbon receptor biology. Proceedings of the National Academy of Sciences. 2009;106:4923–4928. doi: 10.1073/pnas.0809613106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yang F, et al. Repression of the long noncoding RNA-LET by histone deacetylase 3 contributes to hypoxia-mediated metastasis. Molecular cell. 2013;49:1083–1096. doi: 10.1016/j.molcel.2013.01.010. [DOI] [PubMed] [Google Scholar]
- 63.Akahoshi E, Yoshimura S, Ishihara-Sugano M. Over-expression of AhR (aryl hydrocarbon receptor) induces neural differentiation of Neuro2a cells: neurotoxicology study. Environmental health: a global access science source. 2006;5 doi: 10.1186/1476-069X-5-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Yang PW, et al. Association of miRNA-related genetic polymorphisms and prognosis in patients with esophageal squamous cell carcinoma. Annals of surgical oncology. 2014;21(Suppl 4):S601–609. doi: 10.1245/s10434-014-3709-3. [DOI] [PubMed] [Google Scholar]
- 65.Angeles-Floriano T, et al. Absence of aryl hydrocarbon receptor alters CDC42 expression and prevents actin polymerization during capacitation. Molecular reproduction and development. 2016 doi: 10.1002/mrd.22736. [DOI] [PubMed] [Google Scholar]
- 66.Raitoharju E, et al. miR-21, miR-210, miR-34a, and miR-146a/b are up-regulated in human atherosclerotic plaques in the Tampere Vascular Study. Atherosclerosis. 2011;219:211–217. doi: 10.1016/j.atherosclerosis.2011.07.020. [DOI] [PubMed] [Google Scholar]
- 67.Livak KJ, Schmittgen TD. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔ C T Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.