Abstract
Study Design
Systematic literature review and expert survey
Objective
Determination of factors associated with neurologic improvement in patients with neurologic deficits secondary to MESCC. Clear understanding of these factors will guide surgical decision making by helping to elucidate which patients are more likely to benefit from surgery and how surgeons can increase the probability of neurologic and functional restoration.
Summary of Background Data
Surgical spinal cord decompression has been shown to improve neurologic function in patients with symptomatic MESCC. However, prognostication of neurologic improvement after surgery remains challenging, due to sparse data and complexity of these patients.
Methods
PubMed and Embase databases were searched for relevant publications. PRISMA Statement guided publication selection and data reporting. GRADE guidelines were used for evidence quality evaluation and recommendation formulation.
Results
Low-quality evidence supports the use of the duration and severity of neurologic deficit as predictors of neurological recovery in patients with MESCC. Low-quality evidence supports the use of thoracic level of compression and prior irradiation as adverse predictors of neurological recovery. Nearly all of the AOSpine Knowledge Forum Tumor members who responded to the survey agreed that ambulation with assistance represented a successful surgical result and that duration of ambulation loss and the severity of weakness should be considered when trying to predict whether surgery would result in restoration of ambulation.
Conclusions
Review of literature and expert opinion support the importance of duration of ambulation loss and the severity of neurologic deficit (muscle strength, bladder function) in prediction of neurologic recovery among patients with symptomatic MESCC. Efforts to reduce the duration of ambulation loss and to prevent progression of neurologic deficits should be made in order to improve the probability of neurologic recovery.
Keywords: metastasis, spine, neurologic outcome, spinal cord compression, ambulation, bladder, tumor, prognosis, systematic review, MESCC, recovery, surgery
Introduction
Spinal surgery remains an integral treatment component in the continuously shifting landscape of managing patients with metastatic cancer.1–4 The goals of surgery in patients with MESCC include restoration and preservation of neurologic function, and spinal stability and pain relief. The functional status of cancer patients correlates with quality of life and survival, with ambulatory patients surviving longer than patients who have lost the ability to ambulate.3,5 Ambulation also serves as a requisite for further systemic therapy and inclusion in clinical trials, further supporting the significance of neurologic preservation and restoration.
It has been observed that preoperative ambulation status is one of the strongest and most consistent predictors of postoperative ambulation and that surgery is a valuable intervention in preservation of ambulation among patients without profound neurologic deficits.6,7 However, the challenge lies in identifying the most appropriate patient for surgery. The variety of neurologic deficits that can occur secondary to MESCC, variation in the severity of spinal cord dysfunction and the heterogeneity of the patient population has resulted in a lack of clear understanding of what are the most important predictive factors specific to neurologic recovery. The objective of the current study is to determine which factors are associated with neurologic improvement in patients with neurologic deficits secondary to MESCC and to improve patient selection for surgery.
Methods
Question Definition
Patient population
MESCC causing neurologic impairment
Intervention
Surgery
Outcome
Neurological recovery
Subgroup Analysis
Preoperative prognostic indicators associated with postoperative neurologic improvement
Study Question
In patients with metastatic spine disease causing neurological impairment what are the preoperative prognostic indicators for neurological recovery after surgery?
PubMed and Embase databases were searched using the following search strategy: metastatic spine AND prognosis AND neurological outcome OR “metastatic spinal cord compression” AND (ambulation or continence or spinal cord injury or ASIA). Initial search results were screened based on publication titles and abstracts and restricted to English-language publications. Subsequently full-text articles of interest were assessed for inclusion of appropriate patient population and reporting of information that addressed the intervention, outcome and subgroup analysis stated in the question definition above. Bibliographies were also screened for relevant publications that were not found during the database searches. Data included in the current publication was extracted and reconciled by two authors (IL and SZ). The PRISMA Statement checklist and information flow diagram were used in order to ensure clear description of the publication selection and data reporting process.8
GRADE guidelines were used for evaluation of evidence quality, recommendation formulation and determination of recommendation strength as seen in Box 1.9,10 Observational publications included in the current review were all initially classified as low quality of evidence. GRADE criteria for lowering or raising the rating of evidence quality were used to evaluate the association between individual preoperative variables and neurologic recovery.
Survey of AOSpine Knowledge Forum Tumor members was conducted (Supplemental Table 2). The purpose of the survey was to define the specific outcomes of interest when discussing post-operative neurologic function and to determine which preoperative patient variables affect surgical decision-making in expert clinical practice. Questions about the neurologic outcomes of interest were derived based on the data endpoints reported in the MESCC treatment literature.
Results
Literature Search
Initial search produced 312 publications and 310 remained after removing duplicate records. The abstracts of these publications were screened and 204 publications were excluded. One hundred and six full-text articles were assessed for eligibility and seven articles met the inclusion criteria. Reasons for exclusion are provided in Figure 1. All of the included publications reported retrospectively collected patient data (Supplementary Supplement Table 1). Putz et al.,11 and Furstenberg et al.,12 studied the same patient population.
Figure 1.
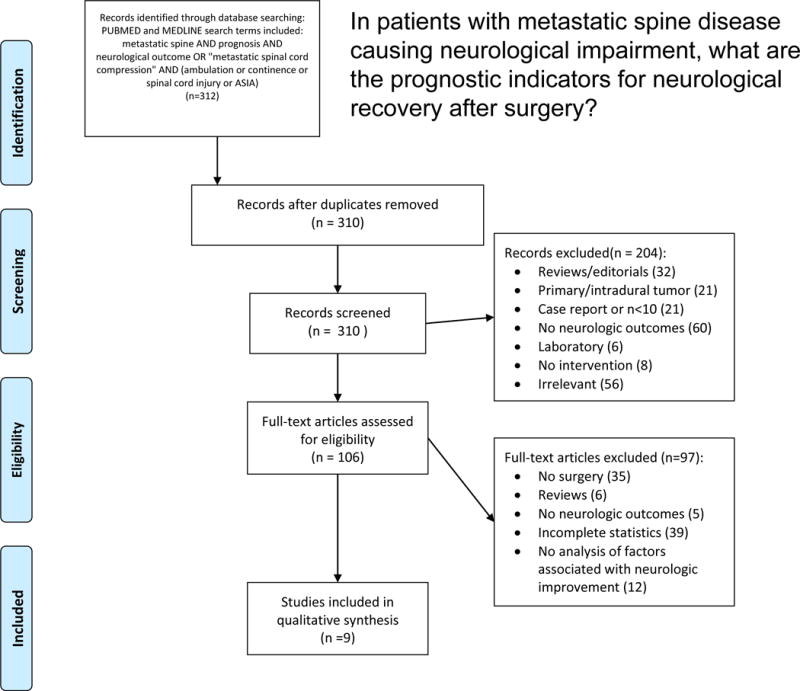
PRISMA flow diagram demonstrating included/excluded articles.
Duration of Symptoms
Five articles reported an association between the duration of symptom onset and neurologic improvement after surgery. Four of these publications dichotomized time and used 48 hours as the cut point. Chaichana et al., studied 23 non-ambulatory patients and reported a relative risk of ambulation recovery of 2.9 (1.13–2.93, p<0.02) in patients with less than 48 hours between symptom onset and surgery. The mean duration of symptoms was 18 days (SD 18 days) among patients who regained ambulation and 48 days (SD 54 days) among patients who did not recover.7 Crnalic et al., studied patients with MESCC secondary to prostate cancer and found that patients with less than 48 hours between loss of ambulation and surgery were more likely to be ambulatory 4 weeks after surgery.13 Among patients who regained ambulation, the median non-ambulatory duration was one day (range 1–3 days) and it was two days (range 0–7) in the group without ambulation restoration (p<0.002). Sixty –four percent of patients (18/28) with less than 48 hours between loss of ambulation and surgery recovered ambulation, while only eighteen percent (3/17) regained ambulation after a longer time to surgery (p<0.005). Furstenberg et al., studied the effect of the timing of surgical decompression in 35 patients with neurologic deficits due to MESCC.12 Among 21 patients who underwent surgical decompression within 48 hours of symptom onset, 71% experienced improvement of their ASIA score and 29% remained stable. However in the group that underwent surgery more than 48 hours after symptom onset, only 29% improved, with 43% remaining the same and 29% deteriorating (p<0.01). Quraishi et al., reported that among 121 patients with neurologic deficits secondary to MESCC, there was a negative correlation between the time from deficit presentation to surgery and the change in Frankel grade (Kendall tau rank correlation coefficient, r=−0.176).14 They also reported that patients who underwent surgery within 48 hours of symptom onset experienced greater improvement in the Frankel grade, compared to patients who underwent surgery more than 48 hours after symptom onset (p<0.05). Seven percent of patients suffered neurologic deterioration in the former group, while 13% deteriorated in the latter group. Hessler et al., operated on 194 patients with neurologic deficits due to spinal metastases and evaluated pre-operative and post-operative Frankel scores that were measured five to seven days after surgery.15 They reported a significant correlation between the pre-operative duration of symptoms and the post-operative neurologic outcome (p<0.01, Kendall-τ b = −0.331), and patients with symptom duration of <10 days and <3 days having higher probability of neurologic improvement compared to patients with >15 days symptom duration. The duration of symptoms did not appear to correlate with the probability of neurologic deterioration.
Severity of Pre-Operative Neurologic Deficit
Two neurologic findings associated with neurologic recovery were reported. Furstenberg et al., reported an association between preoperative bladder function and ASIA score improvement.12 Among twenty patients with normal bladder function, 45% showed improvement in their ASIA score, 45% remained the same and 10% deteriorated. However, among 15 patients with bladder dysfunction, 93% of patients remained the same and 7% deteriorated. Park et al., treated 102 patients with MESCC at thoracic level causing lower extremity weakness.16 Patients with Grade III or better muscle strength were much more likely to ambulate 48 hours after surgery compared to patients with more profound weakness (Odds ratio 49.2, CI 18.4–167.8, p<0.001). Among non-ambulatory patients, 86% of patients recovered ability to ambulate if their muscle strength was Grade III or better, while only 22% recovered if their muscle strength was I or II. Furthermore, the data reported by Hessler et al., indirectly support the association between the severity of pre-operative neurologic deficit and post-operative recovery.15 They reported that 40.2% of patients neurologically improved after surgery, with 37.6% improving by one Frankel grade and 2.6% improving by 2 grades. Therefore, patients with Frankel scores C or D were much more likely to ambulate post-operatively.
Radiographic Parameters
Two radiographic finding associated with ambulation recovery were reported. Chaichana et al., reported that there was a trend toward significance in the association between the presence of vertebral compression fracture (VCF) and ambulation recovery.17 Patients with VCF were possibly less likely to regain ambulation (OR 2.5, CI 0.85–9.94, p 0.14), with 50% of non-ambulatory patients without VCF regaining ambulation compared to 35% of patients with VCF (p<0.09). In a separate publication, Chaichana et al., reported a trend toward a significant association between the level of compression and ambulation recovery, with patients with thoracic levels of compression less likely to regain ambulation (RR 0.003, CI 0.001–1.113, p<0.07).7
Local and systemic oncologic considerations
The aggressiveness of the tumor causing MESCC and of systemic disease may also be associated with ambulation recovery after surgery. Tumor progression after radiation therapy may be adversely associated with recovery of ambulation after surgery. Chaichana et al., reported that pre-operative radiotherapy serves as a negative predictor of post-operative ambulation recovery (RR 0.41, p<0.03).7 Crnalic et al., reported that among patients with MESCC due to prostate cancer, the severity of systemic disease was associated with ambulation recovery after surgery.13 Seventy-nine percent of patients with hormone-naïve prostate cancer recovered ambulation compared to 46% of patients with hormone resistance (p<0.037). Furthermore, 64% of patients with prostate-specific antigen (PSA)level less than 200 ng/ml recovered ambulation, compared to 29% with PSA ≥ 200 ng/ml (p<0.036). Finally, Putz et al., reported an association between the pre-operative Tokuhashi score and the change in ASIA Impairment Scale (AIS).11 Patients with AIS improvement had mean Tokuhashi score of 9, the group with no change had mean score of 8 and the group with AIS deterioration had a score of 7 (r=0.33, p<0.048). However, a larger series from the same institution did not support the association between the preoperative Tokuhashi score and neurologic improvement (Putz 2014).
Survey
Forty-one members of the AOSpine Knowledge Forum Tumor received invitations to complete the survey with 32 people responding (78% response rate). Among the respondents 94% were surgeons and 6% were radiation oncologists, with median practice duration of 8 years (1–38 range). Survey questions and response distribution are included in Supplemental Table 2.
Survey results indicated that satisfactory outcomes of surgery included improvement in muscle strength or preservation of bowel/bladder function even without recovery of ambulation (69% and 90% of respondents, respectively). Ninety-seven percent of respondents agreed that ambulation with assistance or walking aids represented a successful surgical result. While the responses regarding minimal desired distance of ambulation lacked consensus, 53% indicated that at least 4 meters of ambulation were required in order to qualify as meaningful ambulation.
The panel unanimously agreed that the duration of ambulation loss should be considered when trying to predict whether surgery would result in restoration of ambulation. Forty-one percent of participants did not think that prolonged duration of ambulation loss should exclude the patient from consideration for surgery, while 13% responded that they would exclude patients from surgery after 24 hours of ambulation loss. Sixty-nine percent of participants stated that patients were less likely to recover ambulation after 48 hours of ambulation loss.
Ninety-four percent of the respondents agreed that the severity of weakness should be considered when trying to predict whether patients would recover ambulation after surgery. Forty percent of the panel stated that MRC grade 0 in bilateral lower extremities should exclude patients from undergoing surgery, and additional 23% used MRC grade of 1 as exclusion criterion.
Questions about the importance of age, level of spinal cord compression, the number of contiguous compressed spinal levels, presence of VCF, and radiation therapy history did not result in consensus about the value of these variables in prediction of postoperative neurologic recovery.
Discussion
Primary goals of treatment in patients with MESCC include preservation or restoration of neurologic function and spinal stability, local tumor control and pain relief. Clear understanding of treatment goals and which variables are associated with attainment of these goals can significantly facilitate therapeutic decision making.
Variables Associated with Neurologic Recovery
Duration of Symptoms
Data consistently indicate that the duration of time between the onset of neurologic deficit and surgery adversely affects the probability of neurologic improvement and possibly places the patients at risk of neurologic deterioration and the survey of AOSpine Knowledge Forum Tumor members unanimously confirmed the importance of symptom duration in prognostication of postoperative neurologic recovery. Low quality of evidence supports the association between neurologic recovery and the duration of neurologic symptoms before surgery.
Severity of Pre-Operative Neurologic Deficit
Two studies demonstrated a strong association between the severity of pre-operative neurologic deficit and post-operative neurologic recovery. These data suggest that pre-operative bladder dysfunction serves as a negative prognostic indicator of post-operative neurologic improvement and that MRC muscle strength ≥ III serves as a strong predictor of ambulation recovery. Low quality of evidence supports the association of preoperative bladder dysfunction and motor deficit with postoperative neurologic improvement. This association is further supported by the data from numerous publications reporting the association between pre-operative ambulation and post-operative ambulation.
Radiographic Parameters
Trends toward adverse association between the presence of VCF and the thoracic level of compression were reported, however it did not reach statistical significance (p<0.09) resulting the very low quality of evidence in support of this association. While the probability of ambulation recovery was dramatically lower in patients with thoracic MESCC compared to the cervical and lumbar levels (RR 0.003 compared to cervical level), the association failed to reach statistical significance on multivariate analysis (p<0.07). The large reported magnitude of effect, may support the upgrading of evidence quality to low from very low.
Local and systemic oncologic considerations
One publication reported pre-operative radiation therapy as an adverse prognostic indicator of ambulation recovery (RR 0.4). In patients with history of radiation therapy, the time interval shorter than a week between radiotherapy and surgery at the irradiated site has been shown to be associated with increased risk of post-operative wound complication and neurologic deterioration.18 Details about the timing and intent of pre-operative radiotherapy, along with the radiation dose and post-operative tumor control would likely play a role in better understanding of the relationship between preoperative radiotherapy and neurologic recovery. Low quality evidence supports preoperative radiotherapy as an adverse prognostic indicator of neurologic recovery.
The association between the Tokuhashi score and postoperative ASIA score improvement was reported in one publication with a small magnitude of effect. Furthermore, this association was not replicated in a larger series from the same institution, resulting in very low quality of evidence supporting the association between the Tokuhashi score and postoperative neurologic recovery. Finally, among patients with loss of ambulation due to prostate cancer MESCC, PSA level and hormone status served as a predictor of ambulation recovery. The severity and treatment responsiveness of cancer likely plays a role in the post-operative local control and the overall functional status of the patient, thereby affecting the ability to undergo rehabilitation therapy and to achieve neurologic recovery after surgery. Among patients with prostate cancer, low quality evidence supports the role of PSA level and hormone status as predictors of neurologic recovery. Considering the rapidly evolving options for systemic therapy in cancer patients and the changing histology-specific survival profiles, our ability to use cancer-specific predictors of neurologic recovery is limited.
Clinical Application of Prognostic Variables
The current systematic literature review and expert survey revealed only two factors partially amenable to physician control: duration and severity of neurologic deficit. The duration of preoperative deficit may be divided into two time intervals: time from deficit onset to physician evaluation and time between physician evaluation and surgery. Patient and physician education about early reporting and detection of neurologic deficits may result in earlier diagnosis and treatment of MESCC. Current data support making every effort to decrease the duration of neurologic deficit, indicating that once the decision is made to operate on a patient with neurologic dysfunction due to MESCC, surgery should take place as soon as possible under the safest possible circumstances. The value of expedited surgery probably decreases as the duration of deficit increases and we must rely on the judgment of the physicians in order to determine the optimal timing of surgery.
Low-quality evidence supports the association between the severity of neurologic deficit before surgery and postoperative neurologic recovery. In the overwhelming majority of cases, patients with MESCC present to medical attention with a static rather than progressive neurologic deficit. In the rare cases with progressive neurologic deficit, expedited surgical decompression may prevent progression of the neurologic deficit to loss of ambulation, bladder dysfunction and MRC grade ≤ II, thereby improving the probability of neurologic recovery. Therefore, current data support expedited surgery in patients with progressive neurologic deficit at the time of presentation.
The decision about the timing of surgery must be made after careful consideration of modifiable risk factors. The level of urgency of surgery is determined by the speed of neurologic deficit progression, the severity of deficit and the duration of deficit prior to presentation to medical attention. Currently available data fail to provide clear information about the hourly or even daily incremental effect on the neurologic recovery. Furthermore, survey of AOSpine Knowledge Forum Tumor members showed lack of consensus about any time points that would disqualify patients from surgery or that serve as concrete adverse indicators of recovery. Therefore, the urgency of surgical decompression must be weighed against the need to minimize the risk of perioperative morbidity and the decision about the timing of surgery must be made after consideration of individual and situation-specific safety and efficacy considerations. In patients at high risk of perioperative complication, palliative non-surgical interventions should be carefully considered.
Surgical decision making requires clear understanding of tumor biology. We must emphasize that the recommendations presented in the current review apply to solid tumor metastases and exclude tumors with high sensitivity to radiation therapy. Hematologic malignancies such as lymphoma and multiple myeloma and other exquisitely radiosensitive tumors such as small cell variants and seminomas rapidly decrease in volume after radiotherapy and generally do not require surgical decompression even in the setting of symptomatic MESCC. Among patients presenting with MESCC due to a tumor of unknown histology, neurologic preservation takes precedence over diagnosis, with surgical spinal cord decompression justified in order to halt progression of an evolving neurologic deficit and to shorten the duration of the deficit.
Survey of AOSpine Knowledge Forum Tumor members revealed that the majority of respondents considered incomplete neurologic recovery, such as independent ambulation with or without walking aids, or muscle strength improvement or intact bladder function without ambulation restoration as adequate goals of surgery for patients presenting with neurologic deficits due to MESCC. Therefore, while the current expert opinion suggests a fairly low threshold for justification of surgical intervention, the definition of favorable postoperative outcome requires patient-specific adjustment based on patient expectations, needs and overall medical condition.
Low- to very low-quality evidence supports consideration of the presence of VCF, level of spinal cord compression, prior radiation history, Tokuhashi score, and prostate-specific factors in prediction of postoperative neurologic deficit improvement. The predictive value of these variables is unclear at this time and current data does not support their utility in surgical decision making.
Limitations
Publications describing treatment outcomes in patients with MESCC employ heterogeneous methods of reporting of neurologic function and ambulation and the variability of outcome reporting confounds the ability to pool and compare results of these studies. Additionally, the timing of the postoperative assessment and the type of surgery conducted varied, further complicating combined interpretation of the data. Publication bias cannot be excluded as an additional limitation. Finally, we should not assume that the studies included in the current review exhausted the list of possible variables associated with neurologic recovery. For example, it has been postulated that the speed of deficit progression may serve as a predictor of neurologic recovery; however, currently available studies lack information about this variable. Future investigation should focus on finding new ways to predict and optimize neurologic recovery in patients with MESCC. The results of the current literature review and the survey provide guidance about the data points that should be included in future studies.
Recommendation
A patient who is experiencing neurologic deficit from solid tumor MESCC resulting in loss of ability to ambulate, in the absence of medical and oncologic contraindications, requires urgent surgical decompression. Expeditious diagnosis and prompt surgery are recommended in order to improve the probability of neurologic recovery.
Strength of Recommendation: Strong
Quality of Evidence: Low
Conclusions
In patients with metastatic spine disease causing neurological impairment the duration and severity of neurologic deficit serve as prognostic indicators for neurological recovery after surgery. Longer duration of preoperative deficit results in lower probability of neurological improvement. Patients with preserved bladder function and MRC grade ≥III preoperatively are have a higher probability of neurologic improvement. Effort should be made to shorten the duration of neurologic deficit and to prevent further neurologic deterioration. The decision about the timing of surgery must be made after consideration of modifiable risks of surgical morbidity. Clear predictive models with consideration of duration and severity of preoperative neurologic deficit are required in order to guide clinical practice and facilitate treatment decisions for patients with MESCC.
Supplementary Material
Acknowledgments
The manuscript submitted does not contain information about medical device(s)/drug(s).
AOSpine International funds were received in support of this work.
Relevant financial activities outside the submitted work: grants.
References
- 1.Choi D, Fox Z, Albert T, et al. Rapid improvements in pain and quality of life are sustained after surgery for spinal metastases in a large prospective cohort. British journal of neurosurgery. 2016:1–8. doi: 10.3109/02688697.2015.1133802. [DOI] [PubMed] [Google Scholar]
- 2.Fehlings MG, Nater A, Tetreault L, et al. Survival and Clinical Outcomes in Surgically Treated Patients With Metastatic Epidural Spinal Cord Compression: Results of the Prospective Multicenter AOSpine Study. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2016;34:268–76. doi: 10.1200/JCO.2015.61.9338. [DOI] [PubMed] [Google Scholar]
- 3.Patchell RA, Tibbs PA, Regine WF, et al. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: a randomised trial. Lancet. 2005;366:643–8. doi: 10.1016/S0140-6736(05)66954-1. [DOI] [PubMed] [Google Scholar]
- 4.Bilsky M, Laufer I, Burch S. Shifting paradigms in the treatment of metastatic spine disease. Spine (Philadelphia, Pa 1976) 2009;34:S101–S7. doi: 10.1097/BRS.0b013e3181bac4b2. [DOI] [PubMed] [Google Scholar]
- 5.Falicov A, Fisher CG, Sparkes J, et al. Impact of surgical intervention on quality of life in patients with spinal metastases. Spine. 2006;31:2849–56. doi: 10.1097/01.brs.0000245838.37817.40. [DOI] [PubMed] [Google Scholar]
- 6.Cho W, Chang UK. Neurological and survival outcomes after surgical management of subaxial cervical spine metastases. Spine (Phila Pa 1976) 2012;37:E969–77. doi: 10.1097/BRS.0b013e31824ee1c2. [DOI] [PubMed] [Google Scholar]
- 7.Chaichana KL, Woodworth GF, Sciubba DM, et al. Predictors of ambulatory function after decompressive surgery for metastatic epidural spinal cord compression. Neurosurgery. 2008;62:683–92. doi: 10.1227/01.neu.0000317317.33365.15. discussion -92. [DOI] [PubMed] [Google Scholar]
- 8.Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS medicine. 2009;6:e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Guyatt G, Oxman AD, Akl EA, et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. Journal of clinical epidemiology. 2011;64:383–94. doi: 10.1016/j.jclinepi.2010.04.026. [DOI] [PubMed] [Google Scholar]
- 10.Neumann I, Santesso N, Akl EA, et al. A guide for health professionals to interpret and use recommendations in guidelines developed with the GRADE approach. J Clin Epidemiol. 2016;72:45–55. doi: 10.1016/j.jclinepi.2015.11.017. [DOI] [PubMed] [Google Scholar]
- 11.Putz C, Wiedenhofer B, Gerner HJ, et al. Tokuhashi prognosis score: an important tool in prediction of the neurological outcome in metastatic spinal cord compression: a retrospective clinical study. Spine (Phila Pa 1976) 2008;33:2669–74. doi: 10.1097/BRS.0b013e318188b98f. [DOI] [PubMed] [Google Scholar]
- 12.Furstenberg CH, Wiedenhofer B, Gerner HJ, et al. The effect of early surgical treatment on recovery in patients with metastatic compression of the spinal cord. J Bone Joint Surg Br. 2009;91:240–4. doi: 10.1302/0301-620X.91B2.20894. [DOI] [PubMed] [Google Scholar]
- 13.Crnalic S, Hildingsson C, Bergh A, et al. Early diagnosis and treatment is crucial for neurological recovery after surgery for metastatic spinal cord compression in prostate cancer. Acta Oncol. 2013;52:809–15. doi: 10.3109/0284186X.2012.705437. [DOI] [PubMed] [Google Scholar]
- 14.Quraishi NA, Rajagopal TS, Manoharan SR, et al. Effect of timing of surgery on neurological outcome and survival in metastatic spinal cord compression. Eur Spine J. 2013;22:1383–8. doi: 10.1007/s00586-012-2635-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hessler C, Burkhardt T, Raimund F, et al. Dynamics of neurological deficit after surgical decompression of symptomatic vertebral metastases. Spine. 2009;34:566–71. doi: 10.1097/BRS.0b013e31819a825d. [DOI] [PubMed] [Google Scholar]
- 16.Park JH, Jeon SR. Pre- and postoperative lower extremity motor power and ambulatory status of patients with spinal cord compression due to a metastatic spinal tumor. Spine (Phila Pa 1976) 2013;38:E798–802. doi: 10.1097/BRS.0b013e3182927559. [DOI] [PubMed] [Google Scholar]
- 17.Chaichana KL, Pendleton C, Wolinsky JP, et al. Vertebral compression fractures in patients presenting with metastatic epidural spinal cord compression. Neurosurgery. 2009;65:267–74. doi: 10.1227/01.NEU.0000349919.31636.05. discussion 74–5. [DOI] [PubMed] [Google Scholar]
- 18.Ghogawala Z. Spinal radiation before surgical decompression adversely affects outcomes of surgery for symptomatic metastatic spinal cord compression. Spine (Philadelphia, Pa 1976) 2001;26:818–24. doi: 10.1097/00007632-200104010-00025. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


