Abstract
Purpose
To determine magnetic resonance elastography (MRE)-derived stiffness of pancreas in healthy volunteers with emphasis on: 1) short term and midterm repeatability; and 2) variance as a function of age.
Methods
Pancreatic MRE was performed on 22 healthy volunteers (age range:20-64years) in a 3T-scanner. For evaluation of reproducibility of stiffness estimates, the scans were repeated per volunteer on the same day (short term) and one month apart (midterm). MRE wave images were analyzed using 3D inversion to estimate the stiffness of overall pancreas and different anatomic regions (i.e., head, neck, body, and tail). Concordance and Spearman correlation tests were performed to determine reproducibility of stiffness measurements and relationship to age.
Results
A strong concordance (ρc=0.99;p-value<0.001) was found between short term and midterm repeatability pancreatic stiffness measurements. Additionally, the pancreatic stiffness significantly increased with age with good Spearman correlation coefficient (all ρ>0.81;p<0.001). The older age group (>45yrs) had significantly higher stiffness compared to the younger group (≤45yrs) (p<0.001). No significant difference (p>0.05) in stiffness measurements was observed between different anatomical regions of pancreas, except neck stiffness was slightly lower (p<0.012) compared to head and overall pancreas at month 1.
Conclusion
MRE-derived pancreatic stiffness measurements are highly reproducible in the short and midterm and increase linearly with age in healthy volunteers. Further studies are needed to examine these effects in patients with various pancreatic diseases to understand potential clinical applications.
Keywords: Pancreatic MRE, MR Elastography of Pancreas, Pancreas Stiffness
Introduction
Advances in non-invasive imaging of the pancreas are needed to address the increasing burden of pancreatic disorders. Pancreatic cancer is the third leading cause of cancer-related death in the United States, and has one of the worst 5 year survival rates of all cancers (1,2). One important factor for this poor survival is difficulty detecting cancers early with currently available diagnostic methods (3). Similarly, the early diagnosis of chronic pancreatitis (CP) is challenging, and patients often develop irreversible pancreatic insufficiency prior to clinical diagnosis (4). The current imaging modalities for CP do not permit an accurate diagnosis at an early stage, during which interventions can be provided to retard disease progression (5).
There are currently multiple options for pancreatic imaging, however each has limitations (4). Computed tomography (CT) can provide high spatial resolution, but is unable to detect early CP with high sensitivity (4,5). Endoscopic retrograde pancreatography (ERCP) was previously utilized due to sensitive imaging of the pancreatic duct, however due to risks of post-ERCP pancreatitis (along with increased access to MRCP) this test is no longer recommended for diagnostic purposes alone (4,5). Similarly, magnetic resonance cholangiopancreatography (MRCP) provides information of the main pancreatic and branch ducts, but does not provide reliable evidence regarding pancreatic fibrosis (4). Lastly, endoscopic ultrasonography (EUS) provides sensitive imaging of the pancreatic parenchyma and pancreatic ducts, but these findings are subject to inter-observer variability and overlap with changes seen during normal aging and secondary to cigarette smoking and diabetes mellitus (6). In summary, the current imaging modalities are complimentary for the diagnosis of CP, but the accuracy for early CP remains limited.
Both CP and pancreatic ductal adenocarcinoma (PDAC) cause inflammation and fibrosis leading to an increase in pancreatic stiffness compared to a healthy state (5,7). Additionally, stiffness of the pancreas also changes with age due to atrophy (8). Therefore, it is important to understand mechanical properties of the pancreas that can provide valuable information other than morphological changes in detecting early stage pancreatic diseases.
Magnetic resonance elastography (MRE) is a noninvasive technique to estimate stiffness of soft tissues (9-15) and is currently a clinical diagnostic tool to stage liver fibrosis (16-20). To date there are few studies to evaluate the feasibility of in-vivo pancreatic MRE (21-24). An early study by Shi et al (22) determined the stiffness of pancreas at multiple frequencies and subsequent studies (21,23) used small cohorts to determine variation in stiffness between normal and focal pancreatic disease. The aim of the study was to evaluate the short term and midterm reproducibility and age-related variance of MRE-derived pancreatic stiffness in healthy volunteers.
Methods
Pancreatic MRE was performed on 22 healthy subjects (n=66 repeated measurements; age range of 20-64 years) with a mean body mass index (BMI) of 24.5 kg/m2 and range of 20.6 -34.55 kg/m2, after obtaining written informed consent under the approval of the Institutional Review Board. Subjects with no known prior history of pancreatic diseases were included in the study. Only two subjects reported BMI ≥ 30 kg/m2
Experimental Setup
All imaging was performed in a 3T MRI scanner (Tim Trio, Siemens Healthcare, Erlangen, Germany). Subjects were positioned supine head first in the scanner. A pneumatic driver system (Resoundant Inc, Rochester, MN) was used to induce 60Hz vibrations (22) into the pancreas as shown in Figure 1, with output power in the range of 20%-80% depending on the body habitus of the volunteer. The passive driver was securely positioned using an elastic Velcro strap centered at the epigastrium and connected via a plastic tube to the active driver, which was outside the scan room to induce vibrations. Mechanical vibration with a frequency of 60Hz has provided adequate penetration depth into the pancreas because of the rigid passive driver used in this study compared to a soft passive driver used earlier (21,22); and also 60Hz has shorter wavelength compared to 40Hz frequency. Subjects underwent a baseline scan, then returned one month later for two additional scans during the same study visit. During the subsequent study visit subjects underwent an initial scan, then left the scanning room and returned for another scan to permit assessment of short term repeatability. The short term and midterm repeatability measurements were performed to determine the robustness of MRE technique. Only in the advent of severe physiological changes such as effects of hydration (24) (i.e. with increase in hydration pancreas stiffness decreased), we anticipate changes in stiffness values during midterm repeatability.
Figure 1.
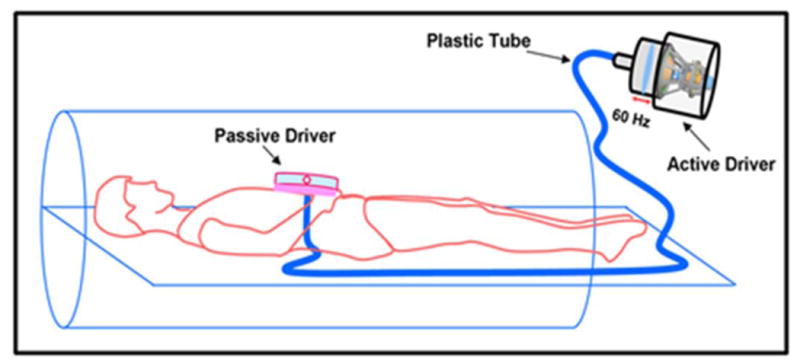
Schematic diagram of driver setup in the MRI scanner. The passive driver is placed on the epigastric region and connected to the active driver to induce the required frequency of vibrations.
Image Acquisition
Balanced steady state free precession (bSSFP) axial scout images were acquired with the same resolution as MRE images. A GRE MRE (20,25) sequence was performed to obtain axial slices including the entire pancreas. Imaging parameters included: FOV: 380×380mm; flip angle: 120; matrix size: 128×64; TR/TE: 25/21.1ms; motion encoding gradient (MEG): 60Hz; MRE phase offsets: 4; #slices: 3-5; slice thickness: 5mm. MEGs were applied to spatially encode in-plane and through plane components of motion in separate breathholds. The acquisition time for a single slice and each encoding direction was an 8sec breathhold.
Image Analysis
First, bSSFP images were masked to segment pancreas and were copied onto MRE data for further processing. The wave images were inspected by an experienced user (AK, ∼12yrs of experience in MRE) for an adequate image quality (i.e. motion artifacts due to improper breathhold and any other artifacts) and the displacement field. Curl processing was performed on the MRE wave data to remove longitudinal motion and directionally filtered to remove reflected waves. Then, a 3D local frequency estimation algorithm was performed to obtain a weighted stiffness map of pancreas using MRE-Lab software (Mayo Clinic, Rochester, MN) as described earlier (14). Finally, bSSFP scout images were masked by a reader (SS, who was trained by an experienced abdominal radiologist with 15yrs experience, ZS) to segment different regions of pancreas using in-house custom built software in Matlab (MathWorks, Natick, MA). Appropriate scout images were used to exclude the regions of fat. These masks were copied onto MRE stiffness maps to determine the regions of interest (i.e., head, neck, body and tail) to report the mean stiffness values of each region and an overall mean stiffness value.
Statistical Analysis
For the assessment of short term and midterm reproducibility, a concordance correlation analysis was performed between two repeat measures obtained during the subsequent study visit. Additionally, for midterm reproducibility, paired t-tests were performed to determine the difference between stiffness measurements obtained during the initial study visit and the first scan obtained during the subsequent study visit. Furthermore, to compare the difference among different anatomical regions of pancreas, analysis of variance with repeated measures followed by pairwise comparison was performed and the multiplicity of measurements was adjusted by using Holm's procedure. Spearman correlation method was used to determine relationship between BMI and age and pancreatic stiffness. Furthermore, spearman correlation method was used to determine the relationship between pancreatic stiffness and age of the subjects. Additionally, the subjects were categorized into binary groups based on age (i.e., young (≤ 45 years) and old (>45 years)) at the time of the first study visit (26,27). An interaction contrasts in a mixed effect model was used, incorporating repeated measures for each subject (28) to test the difference in pancreatic stiffness at short term measures between age groups. A p-value <0.05 was considered to be statistically significant.
Results
Discernible waves were observed in the pancreas at 60Hz vibrational frequency. Figure 2 shows magnitude image with red contour delineating pancreas and a snapshot of wave propagation after curl processing in all three spatial encoding directions (i.e., x, y, and z) of the pancreas and the corresponding stiffness map with an overall mean stiffness value of 1.42 kPa. No significant correlation (p>0.7) was observed between BMI and age and as well as with different anatomical and overall pancreatic stiffness.
Figure 2.
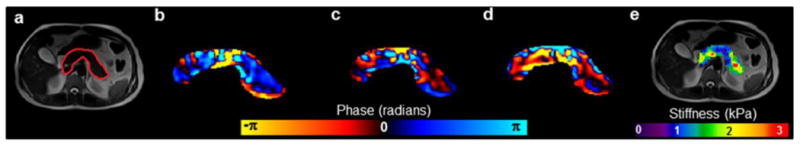
(a) Magnitude image with red contour delineating the pancreas, (b-d) snapshot of curl displacement field in the three spatial encoding directions (i.e., x, y, z), respectively, and (e) the corresponding overall weighted stiffness map.
MRE-derived pancreatic stiffness measurements were reproducible. A high concordance correlation coefficient (ρc = 0.99 (95% confidence interval (CI): 0.992-0.996; p<0.001)) was obtained when all anatomic regions of the pancreas were evaluated for short term reproducibility (Figure 3). Similarly, Figure 4 shows a high concordance correlation coefficient (ρc = 0.99 (95% confidence interval (CI): 0.986-0.993; p<0.001)) was obtained when all anatomic regions of the pancreas were evaluated for midterm reproducibility. Also, stiffness estimates for all regions and overall pancreas were similar when comparing scans one month apart (i.e., midterm repeatability) (Table 1). Figure 5 shows the stiffness plots comparing estimates for different regions and overall pancreas at baseline and the one month (M1) subsequent study visit. Furthermore, no significant difference (p>0.05) in stiffness measurements was observed between most of the anatomical regions of the pancreas, except stiffness of neck region was slightly lower than that of the head (difference = 0.056 kPa, p=0.009) and overall pancreas (difference = 0.058, p = 0.012) at M1.
Figure 3.
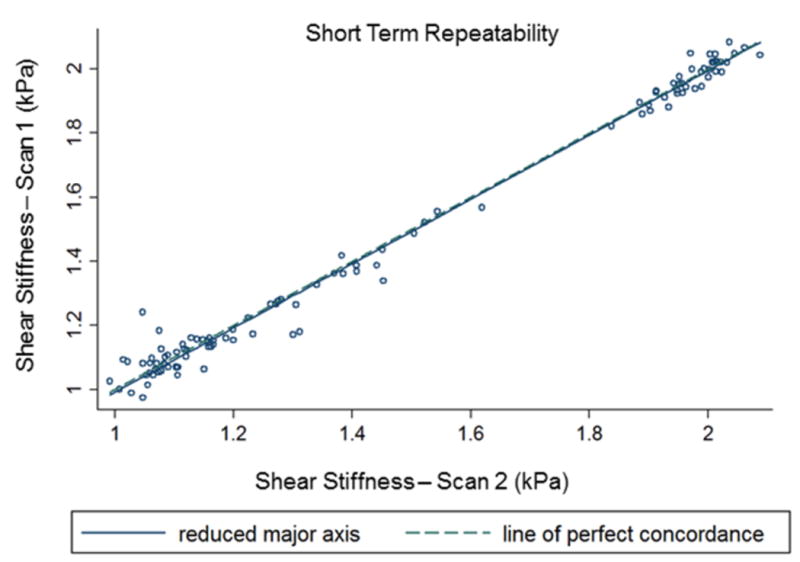
Concordance correlation plot comparing the overall (all anatomical regions of pancreas) MRE-derived pancreatic stiffness measurements obtained during short term reproducibility. The correlation coefficient is 0.994 (p-value <0.001).
Figure 4.
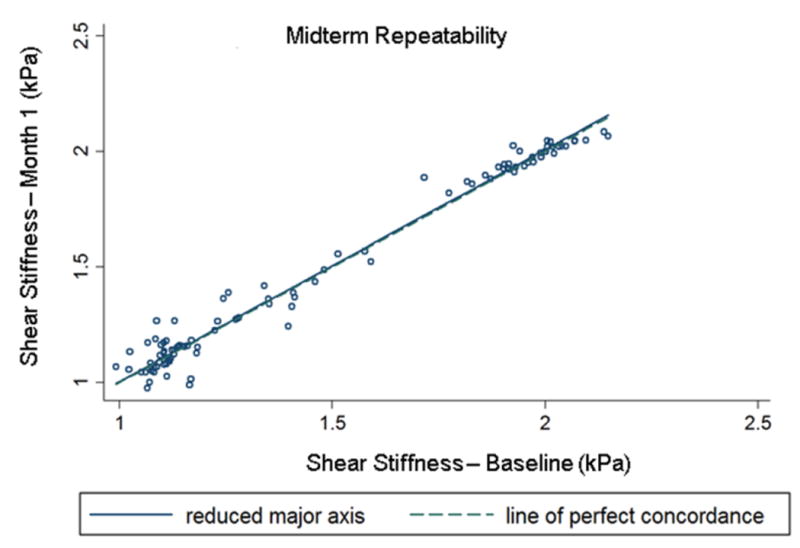
Concordance correlation plot comparing the overall (all anatomical regions of pancreas) MRE-derived pancreatic stiffness measurements obtained during midterm reproducibility. The correlation coefficient is 0.99 (p-value <0.001).
Table 1.
Mean differences in MRE-derived stiffness measurements for different anatomic regions and overall pancreas obtained one month apart.
| Part | Difference (Baseline-M1) (kPa) | 95% CI: Lower (kPa) | 95% CI: Upper (kPa) | p-value* |
|---|---|---|---|---|
| Head | -0.0156 | -0.0390 | 0.00785 | 0.179 |
| Neck | 0.00676 | -0.0338 | 0.0473 | 0.730 |
| Body | -0.00329 | -0.0323 | 0.0257 | 0.816 |
| Tail | 0.00627 | -0.0131 | 0.0256 | 0.508 |
| Overall | -0.0206 | -0.0390 | -0.00234 | 0.141 |
P-values represent results from a paired t-test comparison.
Figure 5.
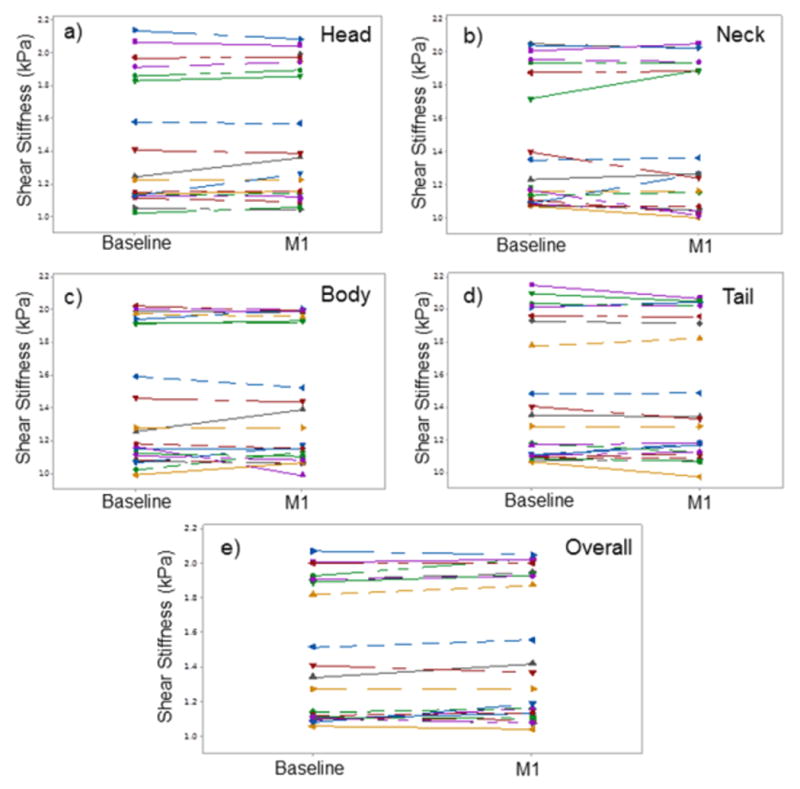
Line plots of shear stiffness measurements obtained during midterm reproducibility scans at each of the anatomic regions (a-d) and overall pancreas (e) were similar with no significant difference (p>0.1).
MRE-derived stiffness of the pancreas increased as a function of age. A good Spearman correlation coefficient (ρ > 0.82) was found as a function of age to different regions and overall stiffness of the pancreas as shown in Figure 6 (all p<0.001). Similarly, there was a significant increase (p<0.001; 95% CI: 0.64-0.93) in stiffness in the older age group (compared to younger) for all regions and overall pancreas (Table 2). Figure 7 shows boxplots showing significant difference stiffness measurements obtained between two age groups for all regions and overall pancreas at baseline and M1. The mean stiffness values for older age group for all regions and overall pancreas was in the range of 1.94-2kPa and for the young age group was 1.13-1.2kPa.
Figure 6.
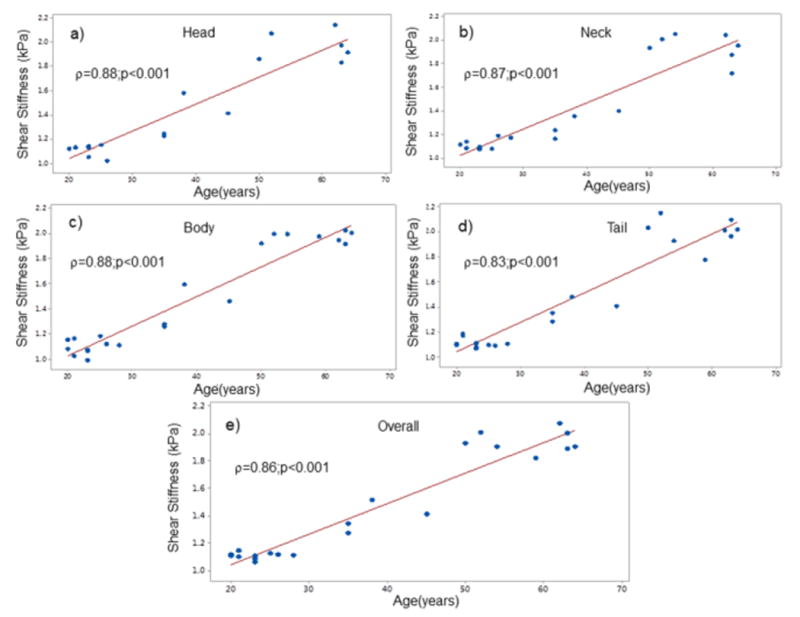
Spearman correlation plots of shear stiffness demonstrated significant correlation with age for each of the anatomic regions (a-d) and overall pancreas (e).
Table 2.
Mean differences in MRE-derived stiffness measurements between young (≤ 45 years) and old (>45 years) age groups for different regions and overall pancreas.
| Part | Time point | Difference (Old vs Young) (kPa) | 95% CI: Lower (kPa) | 95% CI: Upper (kPa) | p-value |
|---|---|---|---|---|---|
| Head | Baseline | 0.7886 | 0.6612 | 0.9161 | <.0001 |
| M1 | 0.7726 | 0.6473 | 0.8979 | <.0001 | |
| Neck | Baseline | 0.7782 | 0.6741 | 0.8823 | <.0001 |
| M1 | 0.8292 | 0.7251 | 0.9333 | <.0001 | |
| Body | Baseline | 0.7857 | 0.6624 | 0.9091 | <.0001 |
| M1 | 0.7851 | 0.6617 | 0.9084 | <.0001 | |
| Tail | Baseline | 0.8103 | 0.6952 | 0.9253 | <.0001 |
| M1 | 0.8038 | 0.6888 | 0.9188 | <.0001 | |
| Overall | Baseline | 0.7536 | 0.6385 | 0.8687 | <.0001 |
| M1 | 0.7696 | 0.6545 | 0.8847 | <.0001 |
Figure 7.
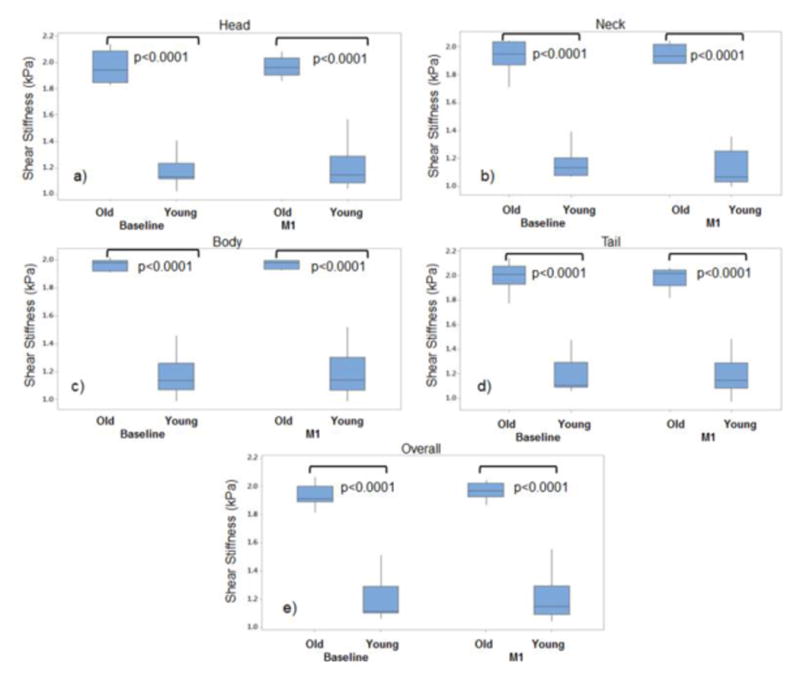
Boxplots showing significant stiffness differences between young and old volunteers both at baseline and month 1 for a) head, b) neck, c) body, d) tail and e) overall pancreas.
Discussion
This study of healthy volunteers demonstrated excellent short and midterm reproducibility of MRE-derived stiffness estimates of the pancreas. Additionally, the MRE-derived stiffness of the pancreas increased with age when compared as a continuous or categorical variable.
The pancreatic MRE technique in this study is reproducible. It is important to demonstrate robustness of MRE-derived shear stiffness of pancreas, as the pancreas is an elongated organ located deep in the abdomen before its application in future patient studies. Standardizing the MRE technique including passive driver placement at a frequency of excitation is critical to demonstrate the reproducible wave penetration in the pancreas to obtain robust stiffness estimates. Additionally, 3D inversion is also important to process the multislice data with 3D wave field as the wave propagation is complex. An earlier study had demonstrated the feasibility of using MRE to estimate pancreatic stiffness at multiple frequencies (22) and short term reproducibility on 5 normal volunteers (21). However, neither extensively studied both short and midterm reproducibility. This study demonstrated that MRE-derived shear stiffness estimates of the pancreas are reproducible both during midterm and short term repeatability with a driver placement for 60Hz frequency of excitation.
MRE-derived shear stiffness of the pancreas increased with age. It is known that during aging soft tissues undergo atrophy, which also occurs in the pancreas. It has already been shown that in various tissues such as the aorta and the heart, the stiffness increases with age (9,14,25). Histologically, Detlefsen et al (29) showed that the amount of fibrotic tissue in the pancreas increases during the course of aging which reflects an increased stiffness. Additionally, the stiffness of the pancreas also depends on the structural component apart from amount of fibrosis (8,29). It has been shown that in a study both fat and fibrosis increased (30), whereas in others there was no correlation (31) or negative correlation of fat to fibrosis (32), where fibrosis is associated to increase in stiffness. However, in the group of subjects, appropriate care was taken to avoid the regions of fat (SS, who was trained by an experienced abdominal radiologist with 15yrs experience, ZS) when reporting the stiffness values. A previous study using EUS-determined elastography measurements (8) has shown that stiffness of the pancreas increase with age, which is consistent with the current study.
MRE-derived pancreatic stiffness measurements reported in this study correspond with previous studies. Earlier study by Shi et al (22) reported stiffness values at 40Hz and 60Hz using a soft passive driver to induce the vibrations. However, in the current study we have used 60Hz vibrations with a hard passive driver that was designed for liver studies as used by Itoh et al (23); and was able to generate required displacement field with good penetration in the pancreas and also have shorter wavelength when compared to 40Hz vibration. Furthermore, Dittmann et al. (24) used pressurized pads powered by medical compressed to induce vibrations in the pancreas ranging from 3060Hz in increments of 10Hz. Usually, hard paddles have more energy generated for a given frequency. Therefore, 60Hz frequency might be more appropriate for application in pancreas because of its geometry. Additionally, Shi et al (22) used direct inversion compared to LFE used in this study to evaluate the stiffness measurements. It is known that direct inversion is an image based inversion technique that requires kernel to obtain derivatives of first harmonic displacement field for estimating stiffness; and whereas LFE is frequency domain based approach using log normal filters without the need for kernels to estimate stiffness and is slightly robust to noise compared to direct inversion (33). Whereas, Dittmann et al. (24) used 2D high resolution tomoelastography inversion to estimate the weighted wavenumbers in determining wave speed/shear stiffness in the pancreas (34). It was claimed that the tomoelastography is more robust to noise and also generates high resolution 2D stiffness maps. The reported stiffness values at 60Hz by Shi et al (22) are in the range reported in our study. Furthermore, Itoh et al (23) reported only 2D in-plane stiffness values with only one encoding direction which produces higher stiffness values compared to 3D stiffness values (9,14) reported in our study. Whereas, Dittmann et al. (24) reported stiffness values of 1.45kPa at 40Hz compared to 1.2kPa at 60Hz in young volunteers in this study. This difference in stiffness can be attributed to the frequency used as well as different inversion strategy applied.
This study reports the stiffness values across ages for different regions and overall pancreas. These values can be potentially used to compare against different disease states for various age groups. The overall stiffness values can be potentially used to compare against diffuse disease states such as CP, whereas region specific stiffness values are relevant for localized diseases, such as pancreatic cancer. Therefore, the present study provides approximate ranges for normal stiffness values of pancreas.
There are a few considerations to accurately interpret these results. First, due to the statistically small sample size we were unable to assess for gender differences in the stiffness measurements and there is equal spread of subjects between two different age groups. Second, these studies were only performed in healthy volunteers, so future studies are needed to develop disease-specific reference ranges. Third, erosion of two pixels was performed to avoid the edge effects stemming from inversion to report the mean stiffness of different pancreatic regions. Fourth, regions of interest were manually drawn to derive the mean stiffness values for the anatomic regions; however, regions were outlined by the same investigator to minimize bias from inter-observers. Fifth, a few slices with a 5 mm slice thickness, the curl processing on the data might be challenging; however, because of the shear wavelengths observed (figure 2) in the pancreas (at the current driving frequency) and finite difference operator used by curl is adequate to obtain the shear displacements for stiffness estimation. Finally, we did not control the hydration status of the subjects, which might alter the stiffness estimates. However, we did not observe significant difference in stiffness estimates during midterm repeatability.
Conclusion
In summary, this study demonstrated excellent short term and midterm reproducibility of MRE-derived stiffness values of the pancreas. Stiffness of the pancreas positively correlated with increased age. These data provide additional evidence showing potential for the future clinical application of MRE technology for imaging of pancreatic diseases and warrants additional investigations.
Acknowledgments
Funding Source: This manuscript has been supported by Grant sponsor: American Heart Association; Grant number: 13SDG14690027; Grant sponsor: Center for Clinical and Translational Sciences; Grant number: UL1TR000090; Grant Sponsor: NIH – NHLBI; Grant number: NIH-R01HL124096. Research reported in this publication was also supported by NCI and NIDDK of the NIH under award number U01DK108327 (PH and DC).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Zhu L, Pickle LW, Ghosh K, Naishadham D, Portier K, Chen HS, Kim HJ, Zou ZH, Cucinelli J, Kohler B, Edwards BK, King J, Feuer EJ, Jemal A. Predicting US- and state-level cancer counts for the current calendar year. Cancer. 2012;118(4):1100–1109. doi: 10.1002/cncr.27405. [DOI] [PubMed] [Google Scholar]
- 2.Chen HS, Portier K, Ghosh K, Naishadham D, Kim HJ, Zhu L, Pickle LW, Krapcho M, Scoppa S, Jemal A, Feuer EJ. Predicting US- and state-level cancer counts for the current calendar year. Cancer. 2012;118(4):1091–1099. doi: 10.1002/cncr.27404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Camara SN, Yin T, Yang M, Li X, Gong Q, Zhou J, Zhao G, Yang ZY, Aroun T, Kuete M, Ramdany S, Camara AK, Diallo AT, Feng Z, Ning X, Xiong JX, Tao J, Qin Q, Zhou W, Cui J, Huang M, Guo Y, Gou SM, Wang B, Liu T, Olivier OE, Conde T, Cisse M, Magassouba AS, Ballah S, Keita NL, Souare IS, Toure A, Traore S, Balde AK, Keita N, Camara ND, Emmanuel D, Wu HS, Wang CY. High risk factors of pancreatic carcinoma. J Huazhong Univ Sci Technolog Med Sci. 2016;36(3):295–304. doi: 10.1007/s11596-016-1583-x. [DOI] [PubMed] [Google Scholar]
- 4.Conwell DL, Lee LS, Yadav D, Longnecker DS, Miller FH, Mortele KJ, Levy MJ, Kwon R, Lieb JG, Stevens T, Toskes PP, Gardner TB, Gelrud A, Wu BU, Forsmark CE, Vege SS. American Pancreatic Association Practice Guidelines in Chronic Pancreatitis Evidence-Based Report on Diagnostic Guidelines. Pancreas. 2014;43(8):1143–1162. doi: 10.1097/MPA.0000000000000237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Janssen J, Schlorer E, Greiner L. EUS elastography of the pancreas: feasibility and pattern description of the normal pancreas, chronic pancreatitis, and focal pancreatic lesions. Gastrointest Endosc. 2007;65(7):971–978. doi: 10.1016/j.gie.2006.12.057. [DOI] [PubMed] [Google Scholar]
- 6.Iglesias-Garcia J, Lindkvist B, Larino-Noia J, Dominguez-Munoz JE. The role of endoscopic ultrasound (EUS) in relation to other imaging modalities in the differential diagnosis between mass forming chronic pancreatitis, autoimmune pancreatitis and ductal pancreatic adenocarcinoma. Revista Espanola De Enfermedades Digestivas. 2012;104(6):315–321. doi: 10.4321/s1130-01082012000600006. [DOI] [PubMed] [Google Scholar]
- 7.Witt H, Apte MV, Keim V, Wilson JS. Chronic pancreatitis: challenges and advances in pathogenesis, genetics, diagnosis, and therapy. Gastroenterology. 2007;132(4):1557–1573. doi: 10.1053/j.gastro.2007.03.001. [DOI] [PubMed] [Google Scholar]
- 8.Janssen J, Papavassiliou I. Effect of Aging and Diffuse Chronic Pancreatitis on Pancreas Elasticity Evaluated using Semiquantitative EUS Elastography. Ultraschall in Der Medizin. 2014;35(3):253–258. doi: 10.1055/s-0033-1355767. [DOI] [PubMed] [Google Scholar]
- 9.Kenyhercz WE, Raterman B, Illapani VS, Dowell J, Mo X, White RD, Kolipaka A. Quantification of aortic stiffness using magnetic resonance elastography: Measurement reproducibility, pulse wave velocity comparison, changes over cardiac cycle, and relationship with age. Magn Reson Med. 2015 doi: 10.1002/mrm.25719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kolipaka A, Araoz PA, Mcgee KP, Manduca A, Ehman RL. Magnetic Resonance Elastography as a Method for the Assessment of Effective Myocardial Stiffness Throughout the Cardiac Cycle. Magnetic Resonance in Medicine. 2010;64(3):862–870. doi: 10.1002/mrm.22467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kolipaka A, Woodrum D, Araoz PA, Ehman RL. MR elastography of the in vivo abdominal aorta: a feasibility study for comparing aortic stiffness between hypertensives and normotensives. J Magn Reson Imaging. 2012;35(3):582–586. doi: 10.1002/jmri.22866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sack I, Beierbach B, Hamhaber U, Klatt D, Braun J. Non-invasive measurement of brain viscoelasticity using magnetic resonance elastography. NMR Biomed. 2008;21(3):265–271. doi: 10.1002/nbm.1189. [DOI] [PubMed] [Google Scholar]
- 13.Sinkus R, Siegmann K, Xydeas T, Tanter M, Claussen C, Fink M. MR elastography of breast lesions: understanding the solid/liquid duality can improve the specificity of contrast-enhanced MR mammography. Magn Reson Med. 2007;58:1135–1144. doi: 10.1002/mrm.21404. [DOI] [PubMed] [Google Scholar]
- 14.Wassenaar PA, Eleswarpu CN, Schroeder SA, Mo X, Raterman BD, White RD, Kolipaka A. Measuring age-dependent myocardial stiffness across the cardiac cycle using MR elastography: A reproducibility study. Magn Reson Med. 2016;75(4):1586–1593. doi: 10.1002/mrm.25760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.da Silveira JS, Scansen BA, Wassenaar PA, Raterman B, Eleswarpu C, Jin N, Mo X, White RD, Bonagura JD, Kolipaka A. Quantification of myocardial stiffness using magnetic resonance elastography in right ventricular hypertrophy: initial feasibility in dogs. Magn Reson Imaging. 2016;34(1):26–34. doi: 10.1016/j.mri.2015.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Klatt D, Asbach P, Rump J, Papazoglou S, Somasundaram R, Modrow J, Braun J, Sack I. In vivo determination of hepatic stiffness using steady-state free precession magnetic resonance elastography. Invest Radiol. 2006;41(12):841–848. doi: 10.1097/01.rli.0000244341.16372.08. [DOI] [PubMed] [Google Scholar]
- 17.Venkatesh SK, Yin M, Ehman RL. Magnetic resonance elastography of liver: technique, analysis, and clinical applications. J Magn Reson Imaging. 2013;37(3):544–555. doi: 10.1002/jmri.23731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yin M, Talwalkar JA, Glaser KJ, Manduca A, Grimm RC, Rossman PJ, Fidler JL, Ehman RL. Assessment of hepatic fibrosis with magnetic resonance elastography. Clinical Gastroenterology and Hepatology. 2007;5(10):1207–1213 e1202. doi: 10.1016/j.cgh.2007.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yin M, Glaser KJ, Talwalkar JA, Chen J, Manduca A, Ehman RL. Hepatic MR Elastography: Clinical Performance in a Series of 1377 Consecutive Examinations. Radiology. 2016;278(1):114–124. doi: 10.1148/radiol.2015142141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chamarthi SK, Raterman B, Mazumder R, Michaels A, Oza VM, Hanje J, Bolster B, Jin N, White RD, Kolipaka A. Rapid acquisition technique for MR elastography of the liver. Magn Reson Imaging. 2014;32(6):679–683. doi: 10.1016/j.mri.2014.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.An H, Shi Y, Guo Q, Liu Y. Test-retest reliability of 3D EPI MR elastography of the pancreas. Clin Radiol. 2016 doi: 10.1016/j.crad.2016.03.014. [DOI] [PubMed] [Google Scholar]
- 22.Shi Y, Glaser KJ, Venkatesh SK, Ben-Abraham EI, Ehman RL. Feasibility of Using 3D MR Elastography to Determine Pancreatic Stiffness in Healthy Volunteers. Journal of Magnetic Resonance Imaging. 2015;41(2):369–375. doi: 10.1002/jmri.24572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Itoh Y, Takehara Y, Kawase T, Terashima K, Ohkawa Y, Hirose Y, Koda A, Hyodo N, Ushio T, Hirai Y, Yoshizawa N, Yamashita S, Nasu H, Ohishi N, Sakahara H. Feasibility of Magnetic Resonance Elastography for the Pancreas at 3T. Journal of Magnetic Resonance Imaging. 2016;43(2):384–390. doi: 10.1002/jmri.24995. [DOI] [PubMed] [Google Scholar]
- 24.Dittmann F, Tzschatzsch H, Hirsch S, Barnhill E, Braun J, Sack I, Guo J. Tomoelastography of the abdomen: Tissue mechanical properties of the liver, spleen, kidney, and pancreas from single MR elastography scans at different hydration states. Magn Reson Med. 2016 doi: 10.1002/mrm.26484. [DOI] [PubMed] [Google Scholar]
- 25.Damughatla AR, Raterman B, Sharkey-Toppen T, Jin N, Simonetti OP, White RD, Kolipaka A. Quantification of aortic stiffness using MR elastography and its comparison to MRI-based pulse wave velocity. J Magn Reson Imaging. 2015;41(1):44–51. doi: 10.1002/jmri.24506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gruessner RW, Sutherland DE, Gruessner AC. Mortality assessment for pancreas transplants. Am J Transplant. 2004;4(12):2018–2026. doi: 10.1111/j.1600-6143.2004.00667.x. [DOI] [PubMed] [Google Scholar]
- 27.Schenker P, Wunsch A, Ertas N, Schaeffer M, Rump LC, Viebahn R, Vonend O. Long-term results after simultaneous pancreas-kidney transplantation using donors aged 45 years or older. Transpl P. 2008;40(4):923–926. doi: 10.1016/j.transproceed.2008.03.078. [DOI] [PubMed] [Google Scholar]
- 28.Verbeke G, Molenberghs G. Linear mixed models for longitudinal data. New York ; London: Springer; 2000. [Google Scholar]
- 29.Detlefsen S, Sipos B, Feyerabend B, Kloppel G. Pancreatic fibrosis associated with age and ductal papillary hyperplasia. Virchows Archiv. 2005;447(5):800–805. doi: 10.1007/s00428-005-0032-1. [DOI] [PubMed] [Google Scholar]
- 30.Catanzaro R, Cuffari B, Italia A, Marotta F. Exploring the metabolic syndrome: Nonalcoholic fatty pancreas disease. World J Gastroenterol. 2016;22(34):7660–7675. doi: 10.3748/wjg.v22.i34.7660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.van Geenen EJM, Smits MM, Schreuder TCMA, van der Peet DL, Bloemena E, Mulder CJJ. Smoking Is Related to Pancreatic Fibrosis in Humans. American Journal of Gastroenterology. 2011;106(6):1161–1166. doi: 10.1038/ajg.2011.43. [DOI] [PubMed] [Google Scholar]
- 32.Mathur A, Pitt HA, Marine M, Saxena R, Schmidt CM, Howard TJ, Nakeeb A, Zyromski NJ, Lillemoe KD. Fatty pancreas: a factor in postoperative pancreatic fistula. Ann Surg. 2007;246(6):1058–1064. doi: 10.1097/SLA.0b013e31814a6906. [DOI] [PubMed] [Google Scholar]
- 33.Manduca A, Oliphant TE, Dresner MA, Mahowald JL, Kruse SA, Amromin E, Felmlee JP, Greenleaf JF, Ehman RL. Magnetic resonance elastography: non-invasive mapping of tissue elasticity. Med Image Anal. 2001;5(4):237–254. doi: 10.1016/s1361-8415(00)00039-6. [DOI] [PubMed] [Google Scholar]
- 34.Tzschatzsch H, Guo J, Dittmann F, Hirsch S, Barnhill E, Johrens K, Braun J, Sack I. Tomoelastography by multifrequency wave number recovery from time-harmonic propagating shear waves. Med Image Anal. 2016;30:1–10. doi: 10.1016/j.media.2016.01.001. [DOI] [PubMed] [Google Scholar]


