Abstract
Functional near-infrared spectroscopy (fNIRS) is a noninvasive optical imaging technique that indirectly assesses neuronal activity by measuring changes in oxygenated and deoxygenated hemoglobin in tissues using near-infrared light. fNIRS has been used not only to investigate cortical activity in healthy human subjects and animals but also to reveal abnormalities in brain function in patients suffering from neurological and psychiatric disorders and in animals that exhibit disease conditions. Because of its safety, quietness, resistance to motion artifacts, and portability, fNIRS has become a tool to complement conventional imaging techniques in measuring hemodynamic responses while a subject performs diverse cognitive and behavioral tasks in test settings that are more ecologically relevant and involve social interaction. In this review, we introduce the basic principles of fNIRS and discuss the application of this technique in human and animal studies.
Keywords: brain recording, functional neuroimaging, fNIRS, functional near-infrared spectroscopy, neurovascular coupling
INTRODUCTION
In response to external stimuli or changes in the environment, the brain undergoes various electrophysiological and neurochemical reactions that are the result of the harmonious interactions of neurons and non-neuronal cells in the brain. The increase in local neuronal activity is usually accompanied by the consumption of glucose and oxygen (Villringer and Chance, 1997), which in turn leads to an increase in local blood flow and blood volume through the distension of the capillaries, eventually leading to the entry of oxygen-bound hemoglobin into the region (Roy and Sherrington, 1890). During this neurovascular coupling, the amount of oxygen supplied is typically greater than that consumed locally, resulting in a substantial increase in oxygenated hemoglobin and a slight reduction in deoxygenated hemoglobin in the region (Fox et al., 1988; Villringer and Dirnagl, 1995). Since the magnitute and location of both oxy- and deoxy-hemoglobin are tightly linked to the extent and location of neuronal activity, the hemodynamic response is often measured as an alternative marker of neuronal activity (Chance et al., 1993; Ogawa et al., 1990; Villringer and Chance, 1997).
Over the last several decades, a number of non-invasive or minimally invasive methods have been developed to measure neuronal activity in the brain. Neurophysiological techniques such as magnetoencephalography (MEG), electroencephalography (EEG), and event-related brain potentials offer the ability to measure the overall change in the electromagnetic field with an excellent time resolution of a thousandth of a second, but with very limited spatial resolution. On the other hand, brain imaging techniques such as positron emission tomography (PET), single-positron emission computed tomography, and functional magnetic resonance imaging (fMRI) allow indirect measurement of neuronal activity by monitoring local hemodynamic and metabolic changes. These techniques have excellent spatial resolution but limited temporal resolution (Irani et al., 2007).
FUNCTIONAL NEAR-INFRARED SPECTROSCOPY
Functional near-infrared spectroscopy (fNIRS) is an emerging optical imaging technique that measures the hemodynamic response in the brain using light in the near-infrared wavelength range (~700–900 nm), which is known to be permeable to biological samples (Fig. 1) (Fox and Raichle, 1986; Jobsis, 1977). The fNIRS system consists of two main components: a light source, such as a light emitting diode or laser, which irradiates near-infrared light at the surface of the subject’s head; and the nearby photodetector, which captures photons returning to the surface of the head after being scattered, reflected, and absorbed in the tissue (Bunce et al., 2006; Villringer and Chance, 1997). Some photons that leave the light source can reach the photodetector by typically following a banana-shaped path in the tissue, but the remaining photons are either dispersed away from the photodetector or absorbed by chromophores present in the tissue, including oxy- and deoxy-hemoglobin and cytochrome c oxidase (Bonner et al., 1987; Gratton et al., 1994). Because oxy- and deoxy-hemoglobin are the main absorbers of near-infrared light with distinct absorption spectra, the relative changes in these chromophores in a target region can be assessed by measuring the absorption of light at two wavelengths, one of which is more sensitive to oxyhemoglobin and the other to deoxy-hemoglobin (Ferrari and Quaresima, 2012). In other words, the local changes in the hemodynamic response can be determined by using a modified version of the Beer-Lambert law, with the light intensities at the source and detector (León-Carrión and León-Domínguez, 2012). In order to measure the absolute amount of these chromophores in the tissue, rather than the relative changes, more sophisticated fNIRS approaches such as a time-resolved system are required. This technique has been validated by the strong correlation observed between the hemodynamic responses measured by fNIRS and by other standard imaging techniques such as fMRI (Ferrari and Quaresima, 2012; Kleinschmidt et al., 1996).
Fig. 1.
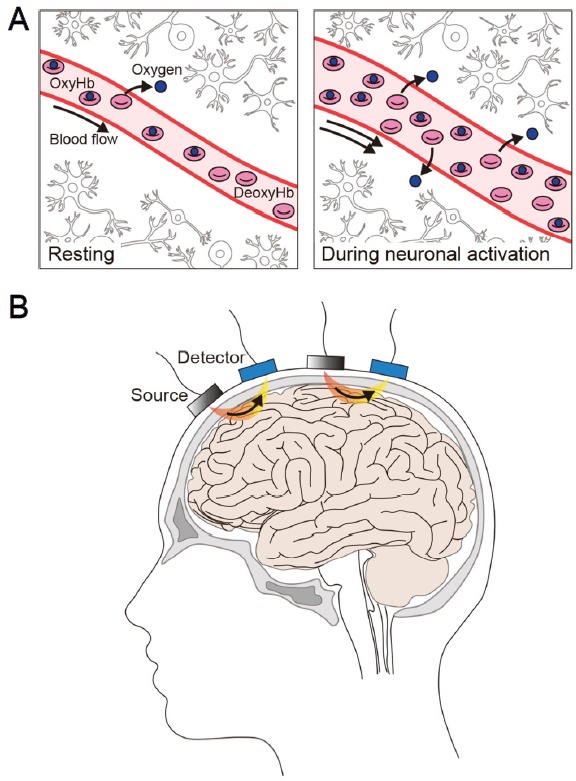
fNIRS imaging of cortical regions in humans.
(A) fNIRS uses near-infrared light to measures the relative changes in oxygenated (Oxy-Hb) and deoxygenated (Deoxy-Hb) hemoglobin in a brain region at baseline (“Resting”) and during neuronal activation. Oxy-Hb and deoxy-Hb are the main chromophores absorbing near-infrared light, and they exhibit distinct absorption spectra. (B) The surface of the head is irradiated with a combination of near-infrared wavelengths of light generated by the light source (“Source”). Photons returning to the surface of the head after traveling a banana-shaped path (in orange and yellow) in the tissues are captured by the photodetector (“Detector”) on the scalp. The number and array of the light source and photodetector on the head vary between studies.
fNIRS has distinct strengths and limitations when compared to other neuroimaging techniques. The temporal resolution of this technique is better than that of fMRI and PET, whereas the spatial resolution is better than that of MEG and EEG, but lower than that of fMRI and PET. One of the major limitations of fNIRS is its shallow imaging depth, which is the result of an exponential attenuation of light intensity as it travels through the scalp and skull and then penetrates through the brain tissue (Bunce et al., 2006; Irani et al., 2007). It has been shown that the imaging depth is influenced by certain factors such as the wavelength and intensity of light, the optical properties of the tissue (such as skin color), and the distance between the light source and the photodetector (Ferrari and Quaresima, 2012; León-Carrión and León-Domínguez, 2012). In general, the more intense the light and/or the further the distance between the light source and the photodetector, the deeper the light can penetrate into the tissue (Ferrari and Quaresima, 2012). However, there are realistic constraints to these parameters, in that the light intensity should be kept much lower than the safety limit in order to avoid skin damage caused by the associated heat, and the distance between the light source and photodetector should be close enough to transmit and receive signals efficiently (Villringer and Chance, 1997). Thus, the imaging depth of fNIRS is generally limited to the surface of the cortex in the case of humans (Sakudo, 2016).
Despite these few limitations, fNIRS offers many advantages. Because it uses a safe, low-energy near-infrared light, fNIRS can continuously and repeatedly measure the hemodynamic response; this type of measurement is not possible with other imaging techniques that require the use of radioisotopes or contrast reagents. Its quietness and tolerance of motion artifacts make fNIRS ideal for use in infants, children, and patients who are uncomfortable with conventional imaging techniques that require confinement in an fMRI magnet, physical constraints, or exposure to loud noise. In addition, other features of fNIRS, including its compact, portable, and wireless design, allow researchers to conduct more physiologically and clinically relevant studies under natural and socially interactive settings. It is also notable that the cost of the fNIRS device and of each test session are much lower than those of other functional neuroimaging methods. Finally, fNIRS can be readily used in conjunction with implanted devices such as cochlear implants and other recording and stimulating equipment, including EEG and the transcranial magnetic stimulator (Boas et al., 2014; Bunce et al., 2006; Irani et al., 2007; León-Carrión and León-Domínguez, 2012; Lloyd-Fox et al., 2010).
APPLICATIONS OF fNIRS TO HUMAN SUBJECTS
Over the years, the fNIRS technique has received increasing attention as a new approach to complement other standard imaging techniques such as fMRI. Given its unique strengths, combining fNIRS with a variety of behavioral tests that assess cognitive, motor, and emotional functions offers great advantages not only in studying basic brain function in normal human subjects but also in identifying abnormalities in neuronal activity in the brain of patients suffering from neurological and psychiatric disorders.
The main features of fNIRS, such as its portability and lower susceptibility to motion artifacts, allow researchers to measure changes in cortical activity during diverse motor tasks, ranging from moving the fingers and hands in clicking a mouse or keyboard during a Stroop task or computer game (Carrieri et al., 2016; Harmat et al., 2015; Kashou et al., 2016; Shortz et al., 2015) to those body movements requiring coordination, such as juggling (Carius et al., 2016) and balancing on a board (Herold et al., 2017), and even to those requiring fine motor skills, such as simulating surgery (Andreu-Perez et al., 2016) and flight (Choe et al., 2016; Gateau et al., 2015). In addition, several recent studies have investigated the effects of bodily and road conditions, such as fatigue (Xu et al., 2017), age (Foy et al., 2016), and road curve (Oka et al., 2015), on the hemodynamic responses of subjects during a driving simulation test. These studies found an activation of a number of brain regions that are known to be involved in cognitive and motor functions, including the prefrontal cortex (PFC), motor cortex, premotor cortex (PMC), and supplementary motor area (SMA).
By the same token, fNIRS has become an excellent tool for investigating conditions such as stroke and Parkinson’s disease (PD) that are primarily characterized by impaired movements. It has been shown that a significant increase in asymmetrical hemodynamic response occurs in a number of regions mediating motor functions, including the sensorimotor cortex (SMC), SMA, PMC, and PFC, when patients suffering from either stroke or PD perform motor-related tasks, including treadmill walking and postural perturbation tasks (Al-Yahya et al., 2016; Fujimoto et al., 2014; Maidan et al., 2015; 2016; Mihara et al., 2007; Miyai et al., 2002; Nieuwhof et al., 2016).
Because it is safe and flexible, fNIRS is well suited to studies of infants and children. For example, recent studies have demonstrated that lateral activation of the cerebral cortex occurs during language development (Altvater-Mackensen and Grossmann, 2016; Vannasing et al., 2016). In addition, it has been reported that the activity of the medial PFC (Urakawa et al., 2015) and inferior frontal and temporal regions (Lloyd-Fox et al., 2015) increases when infants interact with their parents and other infants, respectively. fNIRS has also been used to study neurodevelopmental disorders, such as attention deficit hyperactivity disorder (ADHD) and autism spectrum disorder (ASD), that occur mainly in infants and children. ADHD is characterized by the persistent symptoms of inattention, hyperactivity, and impulsivity (Faraone et al., 2005). By using behavioral tasks requiring attentiveness and concentration, fNIRS imaging has demonstrated that these ADHD-related symptoms are associated with low cerebral cortical activity in a number of regions, including the PFC, inferior prefrontal gyrus, middle prefrontal gyrus, supra-marginal gyrus, and angular gyrus (Araki et al., 2015; Ichikawa et al., 2014; Inoue et al., 2012; Ishii-Takahashi et al., 2015; Kochel et al., 2015; Monden et al., 2015; Moser et al., 2009; Nagashima et al., 2014a; 2014b; 2014c; Negoro et al., 2010; Schecklmann et al., 2011b; Tsujimoto et al., 2013; Weber et al., 2005; Xiao et al., 2012; Yasumura et al., 2014). Similarly, frontal hypoactivity has been observed in ASD patients during behavioral tasks designed to assess social relations and communicative abilities that are dysfunctional or inappropriate in these patients (Iwanaga et al., 2013; Kajiume et al., 2013; Kita et al., 2011; Kuwabara et al., 2006; Minagawa-Kawai et al., 2009; Tamura et al., 2012; Xiao et al., 2012; Yasumura et al., 2014). Furthermore, the effect of neurofeedback training to improve facial processing in ASD patients has been assessed using fNIRS (Liu et al., 2017).
fNIRS imaging has also been used in patients suffering from schizophrenia and affective disorders such as depression, panic disorder, and posttraumatic stress disorder (PTSD) in order to explore cortical regions showing abnormal activity that may account for the inappropriate emotional responses found in patients with these disorders. Exposure to extreme stress can have long-lasting effects that often lead to psychological symptoms of PTSD, including memory deficits and poor health. It has been reported that the PFC of patients suffering from PTSD, for example, victims of the Tokyo subway sarin gas attack, exhibit complex changes in brain response, in that cortical activity increases dramatically when the person encounters an object associated with tragic and traumatic events (Matsuo et al., 2003a), but decreases when the person performs tasks requiring cognitive functions (Matsuo et al., 2003b). Consistent with earlier findings using other imaging techniques, a majority of fNIRS studies have found that patients suffering from major depression disorder show hypoactivation in PFC during cognitive tasks, suggesting the importance of the prefrontal function in symptoms associated with this disorder (Herrmann et al., 2004; Matsuo et al., 2002; Noda et al., 2012; Ohta et al., 2008; Pu et al., 2011; 2012; Schecklmann et al., 2011a). Similarly, hypoactivation of the frontal brain region has been found in panic disorder patients during cognitive tasks as well as a word fluency test (Akiyoshi et al., 2003; Nishimura et al., 2007; 2009; Ohta et al., 2008).
Finally, fNIRS scans of schizophrenic patients also indicate the presence of brain abnormalities, primarily in the bilateral PFC and frontal and temporal cortex in the left hemisphere, during various cognitive tasks, such as the verbal fluency and emotional go/no-go tasks (Azechi et al., 2010; Chou et al., 2015; Egashira et al., 2015; Ehlis et al., 2007; Ikezawa et al., 2009; Koike et al., 2011; Kubota et al., 2005; Marumo et al., 2014; Quan et al., 2015; Quaresima et al., 2009; Shimodera et al., 2012; Takeshi et al., 2010; Takizawa et al., 2008; Watanabe and Kato, 2004).
Most fNIRS experiments carried out thus far have been performed on individual subjects. However, with the development of the hyperscanning system that enables simultaneous interpersonal brain scanning, it is now possible to measure the synchronization of the hemodynamic responses from multiple subjects (Jiang et al., 2012). Recent studies have shown that a simultaneous increase in activity in the left inferior frontal cortex and the right temporo-parietal junction is more common in subjects who are singing or playing computer games while facing each other than in subjects who perform the same task while viewing the walls (Osaka et al., 2015; Tang et al., 2016). Furthermore, the hyperscanning system has been used to compare the differences in the cortical responses of subjects according to the gender of the collaborators (Baker et al., 2016). Finally, a recent study has been able to measure cortical activities from four subjects participating in a conversation at the same time (Nozawa et al., 2016). If the hyperscanning system were combined with recently developed wireless or wearable fNIRS techniques, it might be possible to measure brain activity from multiple subjects who participate in group or outdoor activities, which has thus far been impossible with the wired fNIRS system or other neuroimaging techniques (Ayaz et al., 2013; Cutini and Brigadoi, 2014; Di and Zhang, 2017; Dommer et al., 2012; Holper and Wolf, 2011; Holper et al., 2010; 2011; 2012a; 2012b; 2012c; 2013; 2014; Macnab and Shadgan, 2012; McKendrick et al., 2016; 2017; Piper et al., 2014; von Luhmann et al., 2015; 2017). In the conventional wired system, a subject is connected to fiber-optic bundles that may restrict the subject’s head position, limit the range of action to the length of the fiber-optic bundles, and cause discomfort, particularly in young subjects and patients.
In summary, fNIRS is a non-invasive, safe, less restrictive, and low-cost alternative that has successfully measured changes in brain activity in healthy human subjects and patients while they are performing a variety of behavioral tasks under ecologically relevant conditions.
APPLICATIONS OF FNIRS TO MODEL ANIMALS
The fNIRS technique has been used in a variety of model animals, including non-human primates, sheep, dogs, pigs, and rodents. The advantages of model animals include the ready availability of invasive techniques, diverse disease models, genetically engineered models, and advanced molecular genetic tools (Franceschini et al., 2008). The number of fNIRS studies performed using animals is still far lower than that of human studies, probably because the clinical applications of this technique has been the primary consideration, and there are many other non-invasive and invasive methods already available for animals. However, recent studies have shown a potential for utilizing fNIRS in nonhumans, with broad applicability to model animals.
fNIRS studies on non-human primates have focused on customizing and cross-validating this technique with other well-established methods, such as electrophysiological recordings (Zaidi et al., 2015). Previous studies have found a strong correlation between visual stimulus-elicited responses in the primary visual cortex and PFC of monkeys when measured by fNIRS and via electrodes in the same experiment (Wakita et al., 2010; Zaidi et al., 2015). Interestingly, one study has shown that the activity of the frontal region of monkeys increases in response to various visual stimuli and that the magnitude of the response varies depending on the type of visual stimuli, such as a flower, monkey, snake, or food (Lee et al., 2017). One study has shown a significant correlation between the hemodynamic response measured by fNIRS and the cortical field potentials detected by the epidural electrodes during a delayed working memory task in monkeys (Fuster et al., 2005). However, another study using the same behavioral paradigm has demonstrated that electrophysiological responses measured by EEG exhibit characteristics of complexity and fragmentation of the electrical frequencies, whereas the hemodynamic response obtained with fNIRS increases homogenously and cumulatively, suggesting that the two methods measure slightly different aspects of the neural responses (Ardestani et al., 2016).
Unlike the fNIRS system used for primates, a mobile, miniaturized 8-channel wireless fNIRS system has been typically used to study sheep (Guldimann et al., 2015; Vogeli et al., 2014), goats (Gygax et al., 2013), and dogs (Gygax et al., 2015). To investigate cortical activities representing the affective state, fNIRS imaging has been performed on animals exposed to rich and stable versus poor and unpredictable housing conditions, hot and cold stimuli (Vogeli et al., 2015a), gentle grooming (Muehlemann et al., 2011), or video clips showing positive versus negative social interactions (Vogeli et al., 2015b). Although the effects of these stimuli on the emotional responses of the animals were inconsistent among studies, fNIRS imaging was able to monitor changes in hemodynamic activity in response to such stimuli in the same animals.
In rodents, a set of miniaturized optodes consisting of a light source and photodetector was placed on either the surface of the skull or brain using variable interoptode distances (approximately 400 μm-1.5 cm) and diverse array formations, depending on the brain region being targeted. A previous study using an adapted electroptode (a combination of optode and electrode) has demonstrated a strong correlation between the signals measured by EEG and by fNIRS in the somatosensory cortex and auditory cortex when the rats were stimulated with an electric shock on the forepaw and by the speech sounds of humans, respectively, validating the efficacy of fNIRS in this model animal (Mahmoudzadeh et al., 2017). fNIRS was also shown to be able to detect the sequential activation of the primary and secondary somatosensory cortices and the motor cortex when an electrical stimulus was applied to the whiskers of rats, indicating that fNIRS is capable of detecting a subtle difference in the time sequence, thanks to its high sampling rate (Im et al., 2010). In rats, fNIRS has been used in a wide range of experiments to investigate the effect of diverse stimuli on brain activity, such as electrical stimulation of the somatosensory cortex (Lee et al., 2012), subcutaneous injections of amphetamine and nicotine (Crespi et al., 2005), and the transcranial direct-current stimulation of the barrel cortex (Han et al., 2014). Furthermore, fNIRS imaging has been applied to rodents experiencing epilepsy (Hoshi et al., 1985; Lee et al., 2010; Roche-Labarbe et al., 2010), stroke (Chang et al., 2007; Wolf et al., 1997), brain damage (Abookasis et al., 2013), or pain (He et al., 2012). For example, fNIRS imaging is capable of measuring an increased activity in various cortical and subcortical regions that have previously been shown to mediate pain when the animals are treated with noxious stimuli (e.g., subcutaneous formalin or a noxious pinch on the hindpaw).
In mice, fNIRS has been shown to be capable not only of identifying the correlated activity change in multiple cortical regions during seizures but also of distinguishing between different types of seizures that are classified according to their EEG signal patterns (Lee et al., 2010). Typically, brain activity in mice is measured by non-invasive methods such as fMRI and intrinsic signal optical imaging (Cui et al., 2014), as well as by invasive methods such as multi-photon microscopy and microendoscopy. Because fMRI is susceptible to motion artifacts, it is mainly used for anesthetized or movement-restricted animals (Grandjean et al., 2014; Jonckers et al., 2011; Martin et al., 2013). Multi-photon microscopy and microendoscopy, on the other hand, measure the fluorescent signal emitted by an activity marker exogenously expressed in neurons, such as a genetically encoded calcium indicator GCaMP, at the cellular level by imaging a target region in head-fixed or freely moving animals (Ghosh et al., 2011; Helmchen, 2009). Fiber photometry is another technique measuring the collective changes in fluorescence of an activity marker expressed in a target region using a fiber-optic cable installed just above the target region in freely moving animals (Kim et al., 2016; Packer et al., 2015). These recording techniques offer the ability to directly measure neuronal activity at high resolution in an awake animal, but they have the disadvantage of requiring expensive equipment and the expression of a neuroactivity marker in a target region (Girven and Sparta, 2017). In contrast, fNIRS offers a low-cost alternative for non-invasively or invasively measuring changes in activity in the cortical and subcortical regions of awake, behaving animals by using a set of fiber-optic cables that either transmit or capture photons in the target region (Fig. 2). If fNIRS could be integrated with optogenetics, a method enabling the manipulation of neuronal activity using a light-responsive protein called an opsin, it might be possible to monitor and manipulate neuronal activity using the same fiber-optic cables installed in a target region; this approach would permit the examination of correlations and causal relationships between regional activity and physiological or behavioral changes in the same animal (Fig. 2). If the fNIRS and optogenetic equipment were designed to control each other, it would be possible to maintain regional activity by continuously manipulating the activity using optogenetics according to the change in regional activity measured by fNIRS; such an approach might be useful for patients with brain disorders who often suffer from a sudden activity change in the brain.
Fig. 2.
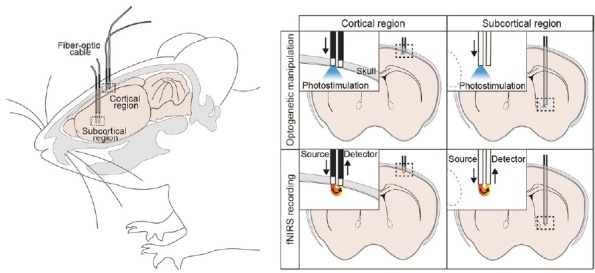
Potential integration of optogenetics and fNIRS.
Activity of neurons in a cortical (left panels) or a subcortical (right panels) region can be manipulated by expressing a light-responsive protein called an opsin (such as channelrhodopsin-2) and delivering photostimulation using a fiber-optic cable installed just above the target region (top panels) in the rodent brain. Using the same fiber-optic cable, a combination of wavelengths of near-infrared light can be delivered to the target brain region alternately (bottom panels). The neighboring fiber-optic cable, which is connected to the photodetector, captures the returning photons. Thus, it would be possible to perform targeted optogenetic manipulation and fNIRS imaging simultaneously in the same animal.
CONCLUSIONS
Since fNIRS was first developed in 1985, its usage has been increasing gradually. Most studies have focused on establishing and improving fNIRS technology, developing data analysis methods, and confirming the validity of the technique by reproducing the results obtained via other imaging techniques. Taking advantage of the strengths of fNIRS allows researchers to perform experiments that are either impossible or limited with other imaging techniques. With the new fNIRS technologies such as wireless and wearable devices and the hyperscanning system, fNIRS has great potential to provide novel insights into brain function by allowing the use of the technique under more ecologically relevant testing conditions. Finally, fNIRS can be developed into a low-cost option that would permit us to diagnose brain disorders using a more objective method based on brain activity and also to monitor the efficacy of therapeutic approaches during the course of treatment.
ACKNOWLEDGMENTS
This work was supported by the Basic Science Research Program (2017R1A2B4003351, H.L.) and the Brain Research Program (2016M3C7A1947307, H.L., U.L., and H.J.J.) of the National Research Foundation of Korea (NRF), funded by the Ministry of Sciences, ICT, and Future Planning; and the KBRI basic research program through Korea Brain Research Institute, funded by the Ministry of Sciences, ICT, and Future Planning (17-BR-04, H.L.). We thank Dr. Deborah McClellan for editorial assistance.
REFERENCES
- Abookasis D, Shochat A, Mathews MS. Monitoring hemodynamic and morphologic responses to closed head injury in a mouse model using orthogonal diffuse near-infrared light reflectance spectroscopy. J Biomed Optics. 2013;18:045003. doi: 10.1117/1.JBO.18.4.045003. [DOI] [PubMed] [Google Scholar]
- Akiyoshi J, Hieda K, Aoki Y, Nagayama H. Frontal brain hypoactivity as a biological substrate of anxiety in patients with panic disorders. Neuropsychobiology. 2003;47:165–170. doi: 10.1159/000070587. [DOI] [PubMed] [Google Scholar]
- Al-Yahya E, Johansen-Berg H, Kischka U, Zarei M, Cockburn J, Dawes H. Prefrontal cortex activation while walking under dual-task conditions in stroke: a multimodal imaging study. neurorehabil Neural Repair. 2016;30:591–599. doi: 10.1177/1545968315613864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altvater-Mackensen N, Grossmann T. The role of left inferior frontal cortex during audiovisual speech perception in infants. Neuroimage. 2016;133:14–20. doi: 10.1016/j.neuroimage.2016.02.061. [DOI] [PubMed] [Google Scholar]
- Andreu-Perez J, Leff DR, Shetty K, Darzi A, Yang G-Z. Disparity in frontal lobe connectivity on a complex bimanual motor task aids in classification of operator skill level. Brain Connectivity. 2016;6:375–388. doi: 10.1089/brain.2015.0350. [DOI] [PubMed] [Google Scholar]
- Araki A, Ikegami M, Okayama A, Matsumoto N, Takahashi S, Azuma H, Takahashi M. Improved prefrontal activity in AD/HD children treated with atomoxetine: a NIRS study. Brain Dev. 2015;37:76–87. doi: 10.1016/j.braindev.2014.03.011. [DOI] [PubMed] [Google Scholar]
- Ardestani A, Shen W, Darvas F, Toga AW, Fuster JM. Modulation of frontoparietal neurovascular dynamics in working memory. J Cogn Neurosci. 2016;28:379–401. doi: 10.1162/jocn_a_00903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ayaz H, Onaral B, Izzetoglu K, Shewokis PA, McKendrick R, Parasuraman R. Continuous monitoring of brain dynamics with functional near infrared spectroscopy as a tool for neuroergonomic research: empirical examples and a technological development. Front Hum Neurosci. 2013;7:871. doi: 10.3389/fnhum.2013.00871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azechi M, Iwase M, Ikezawa K, Takahashi H, Canuet L, Kurimoto R, Nakahachi T, Ishii R, Fukumoto M, Ohi K, et al. Discriminant analysis in schizophrenia and healthy subjects using prefrontal activation during frontal lobe tasks: a near-infrared spectroscopy. Schizophr Res. 2010;117:52–60. doi: 10.1016/j.schres.2009.10.003. [DOI] [PubMed] [Google Scholar]
- Baker JM, Liu N, Cui X, Vrticka P, Saggar M, Hosseini SMH, Reiss AL. Sex differences in neural and behavioral signatures of cooperation revealed by fNIRS hyperscanning. Sci Rep. 2016;6:26492. doi: 10.1038/srep26492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boas DA, Elwell CE, Ferrari M, Taga G. Twenty years of functional near-infrared spectroscopy: introduction for the special issue. Neuroimage. 2014;85:1–5. doi: 10.1016/j.neuroimage.2013.11.033. [DOI] [PubMed] [Google Scholar]
- Bonner R, Nossal R, Havlin S, Weiss G. Model for photon migration in turbid biological media. JOSA A. 1987;4:423–432. doi: 10.1364/josaa.4.000423. [DOI] [PubMed] [Google Scholar]
- Bunce SC, Izzetoglu M, Izzetoglu K, Onaral B, Pourrezaei K. Functional near-infrared spectroscopy. IEEE Eng Med Biol Mag. 2006;25:54–62. doi: 10.1109/memb.2006.1657788. [DOI] [PubMed] [Google Scholar]
- Carius D, Andra C, Clauss M, Ragert P, Bunk M, Mehnert J. Hemodynamic response alteration as a function of task complexity and expertise - an fNIRS study in jugglers. Front Hum Neurosci. 2016;10:126. doi: 10.3389/fnhum.2016.00126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrieri M, Petracca A, Lancia S, Moro SB, Brigadoi S, Spezialetti M, Ferrari M, Placidi G, Quaresima V. Prefrontal cortex activation upon a demanding virtual hand-controlled task: a new frontier for neuroergonomics. Front Hum Neurosci. 2016;10:53. doi: 10.3389/fnhum.2016.00053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chance B, Zhuang Z, UnAh C, Alter C, Lipton L. Cognition-activated low-frequency modulation of light absorption in human brain. Proc Natl Acad Sci USA. 1993;90:3770–3774. doi: 10.1073/pnas.90.8.3770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang G, Wang K, Hsu C, Chen J. Development of functional near infrared spectroscopy system for assessing cerebral hemodynamics of rats with ischemic stroke. J Med Biol Eng. 2007;27:207. [Google Scholar]
- Choe J, Coffman BA, Bergstedt DT, Ziegler MD, Phillips ME. Transcranial direct current stimulation modulates neuronal activity and learning in pilot training. Front Hum Neurosci. 2016;10:34. doi: 10.3389/fnhum.2016.00034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou PH, Lin WH, Lin CC, Hou PH, Li WR, Hung CC, Lin CP, Lan TH, Chan CH. Duration of untreated psychosis and brain function during verbal fluency testing in first-episode schizophrenia: a near-infrared spectroscopy study. Sci Rep. 2015;5:18069. doi: 10.1038/srep18069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crespi F, Bandera A, Donini M, Heidbreder C, Rovati L. Non-invasive in vivo infrared laser spectroscopy to analyse endogenous oxy-haemoglobin, deoxy-haemoglobin, and blood volume in the rat CNS. J Neurosci Methods. 2005;145:11–22. doi: 10.1016/j.jneumeth.2004.11.016. [DOI] [PubMed] [Google Scholar]
- Cui G, Jun SB, Jin X, Luo G, Pham MD, Lovinger DM, Vogel SS, Costa RM. Deep brain optical measurements of cell type–specific neural activity in behaving mice. Nat Protocols. 2014;9:1213. doi: 10.1038/nprot.2014.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cutini S, Brigadoi S. Unleashing the future potential of functional near-infrared spectroscopy in brain sciences. J Neurosci Methods. 2014;232:152–156. doi: 10.1016/j.jneumeth.2014.05.024. [DOI] [PubMed] [Google Scholar]
- Di H, Zhang X. Deception detection by hybrid-pair wireless fNIRS system. Int J Dig Crime Forensic (IJDCF) 2017;9:15–24. [Google Scholar]
- Dommer L, Jager N, Scholkmann F, Wolf M, Holper L. Between-brain coherence during joint n-back task performance: a two-person functional near-infrared spectroscopy study. Behav Brain Res. 2012;234:212–222. doi: 10.1016/j.bbr.2012.06.024. [DOI] [PubMed] [Google Scholar]
- Egashira K, Matsuo K, Nakashima M, Watanuki T, Harada K, Nakano M, Matsubara T, Takahashi K, Watanabe Y. Blunted brain activation in patients with schizophrenia in response to emotional cognitive inhibition: a functional near-infrared spectroscopy study. Schizophr Res. 2015;162:196–204. doi: 10.1016/j.schres.2014.12.038. [DOI] [PubMed] [Google Scholar]
- Ehlis AC, Herrmann MJ, Plichta MM, Fallgatter AJ. Cortical activation during two verbal fluency tasks in schizophrenic patients and healthy controls as assessed by multi-channel near-infrared spectroscopy. Psychiatry Res Neuroimaging. 2007;156:1–13. doi: 10.1016/j.pscychresns.2006.11.007. [DOI] [PubMed] [Google Scholar]
- Faraone SV, Perlis RH, Doyle AE, Smoller JW, Goralnick JJ, Holmgren MA, Sklar P. Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatry. 2005;57:1313–1323. doi: 10.1016/j.biopsych.2004.11.024. [DOI] [PubMed] [Google Scholar]
- Ferrari M, Quaresima V. A brief review on the history of human functional near-infrared spectroscopy (fNIRS) development and fields of application. Neuroimage. 2012;63:921–935. doi: 10.1016/j.neuroimage.2012.03.049. [DOI] [PubMed] [Google Scholar]
- Fox PT, Raichle ME. Focal physiological uncoupling of cerebral blood flow and oxidative metabolism during somatosensory stimulation in human subjects. Proc Natl Acad Sci USA. 1986;83:1140–1144. doi: 10.1073/pnas.83.4.1140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox PT, Raichle ME, Mintun MA, Dence C. Nonoxidative glucose consumption during focal physiologic neural activity. Science. 1988;241:462–464. doi: 10.1126/science.3260686. [DOI] [PubMed] [Google Scholar]
- Foy HJ, Runham P, Chapman P. Prefrontal cortex activation and young driver behaviour: a fNIRS study. PLoS One. 2016;11:e0156512. doi: 10.1371/journal.pone.0156512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franceschini MA, Nissilä I, Wu W, Diamond SG, Bonmassar G, Boas DA. Coupling between somatosensory evoked potentials and hemodynamic response in the rat. Neuroimage. 2008;41:189–203. doi: 10.1016/j.neuroimage.2008.02.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujimoto H, Mihara M, Hattori N, Hatakenaka M, Kawano T, Yagura H, Miyai I, Mochizuki H. Cortical changes underlying balance recovery in patients with hemiplegic stroke. Neuroimage. 2014;85:547–554. doi: 10.1016/j.neuroimage.2013.05.014. [DOI] [PubMed] [Google Scholar]
- Fuster J, Guiou M, Ardestani A, Cannestra A, Sheth S, Zhou YD, Toga A, Bodner M. Near-infrared spectroscopy (NIRS) in cognitive neuroscience of the primate brain. Neuroimage. 2005;26:215–220. doi: 10.1016/j.neuroimage.2005.01.055. [DOI] [PubMed] [Google Scholar]
- Gateau T, Durantin G, Lancelot F, Scannella S, Dehais F. Real-time state estimation in a flight simulator using fNIRS. PLoS One. 2015;10:e0121279. doi: 10.1371/journal.pone.0121279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh KK, Burns LD, Cocker ED, Nimmerjahn A, Ziv Y, El Gamal A, Schnitzer MJ. Miniaturized integration of a fluorescence microscope. Nat Methods. 2011;8:871–878. doi: 10.1038/nmeth.1694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Girven KS, Sparta DR. Probing deep brain circuitry: new advances in in vivo calcium measurement strategies. ACS Chem Neurosci. 2017;8:243–251. doi: 10.1021/acschemneuro.6b00307. [DOI] [PubMed] [Google Scholar]
- Grandjean J, Schroeter A, Batata I, Rudin M. Optimization of anesthesia protocol for resting-state fMRI in mice based on differential effects of anesthetics on functional connectivity patterns. Neuroimage. 2014;102:838–847. doi: 10.1016/j.neuroimage.2014.08.043. [DOI] [PubMed] [Google Scholar]
- Gratton G, Maier JS, Fabiani M, Mantulin WW, Gratton E. Feasibility of intracranial near-infrared optical scanning. Psychophysiology. 1994;31:211–215. doi: 10.1111/j.1469-8986.1994.tb01043.x. [DOI] [PubMed] [Google Scholar]
- Guldimann K, Vogeli S, Wolf M, Wechsler B, Gygax L. Frontal brain deactivation during a non-verbal cognitive judgement bias test in sheep. Brain Cogn. 2015;93:35–41. doi: 10.1016/j.bandc.2014.11.004. [DOI] [PubMed] [Google Scholar]
- Gygax L, Reefmann N, Pilheden T, Scholkmann F, Keeling L. Dog behavior but not frontal brain reaction changes in repeated positive interactions with a human: a non-invasive pilot study using functional near-infrared spectroscopy (fNIRS) Behav Brain Res. 2015;281:172–176. doi: 10.1016/j.bbr.2014.11.044. [DOI] [PubMed] [Google Scholar]
- Gygax L, Reefmann N, Wolf M, Langbein J. Prefrontal cortex activity, sympatho-vagal reaction and behaviour distinguish between situations of feed reward and frustration in dwarf goats. Behav Brain Res. 2013;239:104–114. doi: 10.1016/j.bbr.2012.10.052. [DOI] [PubMed] [Google Scholar]
- Han C-H, Song H, Kang Y-G, Kim B-M, Im C-H. Hemodynamic responses in rat brain during transcranial direct current stimulation: a functional near-infrared spectroscopy study. Biomed Optics Exp. 2014;5:1812–1821. doi: 10.1364/BOE.5.001812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harmat L, de Manzano O, Theorell T, Hogman L, Fischer H, Ullen F. Physiological correlates of the flow experience during computer game playing. Int J Psychophysiol. 2015;97:1–7. doi: 10.1016/j.ijpsycho.2015.05.001. [DOI] [PubMed] [Google Scholar]
- He J-W, Tian F, Liu H, Peng YB. Cerebrovascular responses of the rat brain to noxious stimuli as examined by functional near-infrared whole brain imaging. J Neurophysiol. 2012;107:2853–2865. doi: 10.1152/jn.00050.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helmchen F. Two-photon functional imaging of neuronal activity. In: Frostig RD, editor. In vivo optical imaging of brain function. 2nd ed. Chapter 2. Boca Raton (FL): CRC Press/Taylor & Francis; 2009. [PubMed] [Google Scholar]
- Herold F, Orlowski K, Bormel S, Muller NG. Cortical activation during balancing on a balance board. Hum Mov Sci. 2017;51:51–58. doi: 10.1016/j.humov.2016.11.002. [DOI] [PubMed] [Google Scholar]
- Herrmann MJ, Ehlis AC, Fallgatter AJ. Bilaterally reduced frontal activation during a verbal fluency task in depressed patients as measured by near-infrared spectroscopy. J Neuropsychiatr Clin Neurosci. 2004;16:170–175. doi: 10.1176/jnp.16.2.170. [DOI] [PubMed] [Google Scholar]
- Holper L, Wolf M. Single-trial classification of motor imagery differing in task complexity: a functional near-infrared spectroscopy study. J Neuroeng Rehabil. 2011;8:34. doi: 10.1186/1743-0003-8-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holper L, Muehlemann T, Scholkmann F, Eng K, Kiper D, Wolf M. Testing the potential of a virtual reality neurorehabilitation system during performance of observation, imagery and imitation of motor actions recorded by wireless functional near-infrared spectroscopy (fNIRS) J Neuroeng Rehabil. 2010;7:57. doi: 10.1186/1743-0003-7-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holper L, Shalom DE, Wolf M, Sigman M. Understanding inverse oxygenation responses during motor imagery: a functional near-infrared spectroscopy study. Eur J Neurosci. 2011;33:2318–2328. doi: 10.1111/j.1460-9568.2011.07720.x. [DOI] [PubMed] [Google Scholar]
- Holper L, Kobashi N, Kiper D, Scholkmann F, Wolf M, Eng K. Trial-to-trial variability differentiates motor imagery during observation between low versus high responders: a functional near-infrared spectroscopy study. Behav Brain Res. 2012a;229:29–40. doi: 10.1016/j.bbr.2011.12.038. [DOI] [PubMed] [Google Scholar]
- Holper L, Scholkmann F, Shalom DE, Wolf M. Extension of mental preparation positively affects motor imagery as compared to motor execution: a functional near-infrared spectroscopy study. Cortex. 2012b;48:593–603. doi: 10.1016/j.cortex.2011.02.001. [DOI] [PubMed] [Google Scholar]
- Holper L, Scholkmann F, Wolf M. Between-brain connectivity during imitation measured by fNIRS. Neuroimage. 2012c;63:212–222. doi: 10.1016/j.neuroimage.2012.06.028. [DOI] [PubMed] [Google Scholar]
- Holper L, Goldin AP, Shalóm DE, Battro AM, Wolf M, Sigman M. The teaching and the learning brain: a cortical hemodynamic marker of teacher–student interactions in the Socratic dialog. Int J Edu Res. 2013;59:1–10. [Google Scholar]
- Holper L, Wolf M, Tobler PN. Comparison of functional near-infrared spectroscopy and electrodermal activity in assessing objective versus subjective risk during risky financial decisions. Neuroimage. 2014;84:833–842. doi: 10.1016/j.neuroimage.2013.09.047. [DOI] [PubMed] [Google Scholar]
- Hoshi Y, Kobayashi N, Tamura M. Interpretation of near-infrared spectroscopy signals: a study with a newly developed perfused rat brain model. J Appl Physiol. 1985;90:1657–1662. doi: 10.1152/jappl.2001.90.5.1657. [DOI] [PubMed] [Google Scholar]
- Ichikawa H, Nakato E, Kanazawa S, Shimamura K, Sakuta Y, Sakuta R, Yamaguchi MK, Kakigi R. Hemodynamic response of children with attention-deficit and hyperactive disorder (ADHD) to emotional facial expressions. Neuropsychologia. 2014;63:51–58. doi: 10.1016/j.neuropsychologia.2014.08.010. [DOI] [PubMed] [Google Scholar]
- Ikezawa K, Iwase M, Ishii R, Azechi M, Canuet L, Ohi K, Yasuda Y, Iike N, Kurimoto R, Takahashi H, et al. Impaired regional hemodynamic response in schizophrenia during multiple prefrontal activation tasks: a two-channel near-infrared spectroscopy study. Schizophr Res. 2009;108:93–103. doi: 10.1016/j.schres.2008.12.010. [DOI] [PubMed] [Google Scholar]
- Im C-H, Jung Y-J, Lee S, Koh D, Kim D-W, Kim B-M. Estimation of directional coupling between cortical areas using near-Infrared spectroscopy (NIRS) Opt Express. 2010;18:5730–5739. doi: 10.1364/OE.18.005730. [DOI] [PubMed] [Google Scholar]
- Inoue Y, Sakihara K, Gunji A, Ozawa H, Kimiya S, Shinoda H, Kaga M, Inagaki M. Reduced prefrontal hemodynamic response in children with ADHD during the go/nogo task: a NIRS study. Neuroreport. 2012;23:55–60. doi: 10.1097/WNR.0b013e32834e664c. [DOI] [PubMed] [Google Scholar]
- Irani F, Platek SM, Bunce S, Ruocco AC, Chute D. Functional near infrared spectroscopy (fNIRS): an emerging neuroimaging technology with important applications for the study of brain disorders. Clin Neuropsychol. 2007;21:9–37. doi: 10.1080/13854040600910018. [DOI] [PubMed] [Google Scholar]
- Ishii-Takahashi A, Takizawa R, Nishimura Y, Kawakubo Y, Hamada K, Okuhata S, Kawasaki S, Kuwabara H, Shimada T, Todokoro A, et al. Neuroimaging-aided prediction of the effect of methylphenidate in children with attention-deficit hyperactivity disorder: a randomized controlled trial. Neuropsychopharmacology. 2015;40:2676–2685. doi: 10.1038/npp.2015.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwanaga R, Tanaka G, Nakane H, Honda S, Imamura A, Ozawa H. Usefulness of near-infrared spectroscopy to detect brain dysfunction in children with autism spectrum disorder when inferring the mental state of others. Psychiatry Clin Neurosci. 2013;67:203–209. doi: 10.1111/pcn.12052. [DOI] [PubMed] [Google Scholar]
- Jiang J, Dai BH, Peng DL, Zhu CZ, Liu L, Lu CM. Neural Synchronization during face-to-face communication. J Neurosci. 2012;32:16064–16069. doi: 10.1523/JNEUROSCI.2926-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jobsis FF. Noninvasive, infrared monitoring of cerebral and myocardial oxygen sufficiency and circulatory parameters. Science. 1977;198:1264–1267. doi: 10.1126/science.929199. [DOI] [PubMed] [Google Scholar]
- Jonckers E, Van Audekerke J, De Visscher G, Van der Linden A, Verhoye M. Functional connectivity fMRI of the rodent brain: comparison of functional connectivity networks in rat and mouse. PLoS One. 2011;6:e18876. doi: 10.1371/journal.pone.0018876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kajiume A, Aoyama-Setoyama S, Saito-Hori Y, Ishikawa N, Kobayashi M. Reduced brain activation during imitation and observation of others in children with pervasive developmental disorder: a pilot study. Behav Brain Funct. 2013;9:21. doi: 10.1186/1744-9081-9-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashou NH, Giacherio BM, Nahhas RW, Jadcherla SR. Hand-grasping and finger tapping induced similar functional near-infrared spectroscopy cortical responses. Neurophotonics. 2016;3:025006. doi: 10.1117/1.NPh.3.2.025006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim CK, Yang SJ, Pichamoorthy N, Young NP, Kauvar I, Jennings JH, Lerner TN, Berndt A, Lee SY, Ramakrishnan C. Simultaneous fast measurement of circuit dynamics at multiple sites across the mammalian brain. Nat Methods. 2016;13:325–328. doi: 10.1038/nmeth.3770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kita Y, Gunji A, Inoue Y, Goto T, Sakihara K, Kaga M, Inagaki M, Hosokawa T. Self-face recognition in children with autism spectrum disorders: a near-infrared spectroscopy study. Brain Dev. 2011;33:494–503. doi: 10.1016/j.braindev.2010.11.007. [DOI] [PubMed] [Google Scholar]
- Kleinschmidt A, Obrig H, Requardt M, Merboldt K-D, Dirnagl U, Villringer A, Frahm J. Simultaneous recording of cerebral blood oxygenation changes during human brain activation by magnetic resonance imaging and near-infrared spectroscopy. J Cereb Blood Flow Metabol. 1996;16:817–826. doi: 10.1097/00004647-199609000-00006. [DOI] [PubMed] [Google Scholar]
- Kochel A, Schongassner F, Feierl-Gsodam S, Schienle A. Processing of affective prosody in boys suffering from attention deficit hyperactivity disorder: a near-infrared spectroscopy study. Soc Neurosci. 2015;10:583–591. doi: 10.1080/17470919.2015.1017111. [DOI] [PubMed] [Google Scholar]
- Koike S, Takizawa R, Nishimura Y, Takano Y, Takayanagi Y, Kinou M, Araki T, Harima H, Fukuda M, Okazaki Y, et al. Different hemodynamic response patterns in the prefrontal cortical sub-regions according to the clinical stages of psychosis. Schizophr Res. 2011;132:54–61. doi: 10.1016/j.schres.2011.07.014. [DOI] [PubMed] [Google Scholar]
- Kubota Y, Toichi M, Shimizu M, Mason RA, Coconcea CM, Findling RL, Yamamoto K, Calabrese JR. Prefrontal activation during verbal fluency tests in schizophrenia—a near-infrared spectroscopy (NIRS) study. Schizophr Res. 2005;77:65–73. doi: 10.1016/j.schres.2005.01.007. [DOI] [PubMed] [Google Scholar]
- Kuwabara H, Kasai K, Takizawa R, Kawakubo Y, Yamasue H, Rogers MA, Ishijima M, Watanabe K, Kato N. Decreased prefrontal activation during letter fluency task in adults with pervasive developmental disorders: a near-infrared spectroscopy study. Behav Brain Res. 2006;172:272–277. doi: 10.1016/j.bbr.2006.05.020. [DOI] [PubMed] [Google Scholar]
- Lee S, Lee M, Koh D, Kim B-M, Choi JH. Cerebral hemodynamic responses to seizure in the mouse brain: simultaneous near-infrared spectroscopy–electroencephalography study. J Biomed Opt. 2010;15:037010–037018. doi: 10.1117/1.3365952. [DOI] [PubMed] [Google Scholar]
- Lee S, Koh D, Jo A, Lim HY, Jung YJ, Kim CK, Seo Y, Im CH, Kim BM, Suh M. Depth-dependent cerebral hemodynamic responses following direct cortical electrical stimulation (DCES) revealed by in vivo dual-optical imaging techniques. Opt Express. 2012;20:6932–6943. doi: 10.1364/OE.20.006932. [DOI] [PubMed] [Google Scholar]
- Lee YA, Pollet V, Kato A, Goto Y. Prefrontal cortical activity associated with visual stimulus categorization in non-human primates measured with near-infrared spectroscopy. Behav Brain Res. 2017;317:327–331. doi: 10.1016/j.bbr.2016.09.068. [DOI] [PubMed] [Google Scholar]
- León-Carrión J, León-Domínguez U. Functional nearinfrared spectroscopy (fNIRS): principles and neuroscientific applications. Neuroimaging-Methods (InTech) 2012:47–74. [Google Scholar]
- Liu N, Cliffer S, Pradhan AH, Lightbody A, Hall SS, Reiss AL. Optical-imaging-based neurofeedback to enhance therapeutic intervention in adolescents with autism: methodology and initial data. Neurophotonics. 2017;4:011003. doi: 10.1117/1.NPh.4.1.011003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lloyd-Fox S, Blasi A, Elwell C. Illuminating the developing brain: the past, present and future of functional near infrared spectroscopy. Neurosci Biobehav Rev. 2010;34:269–284. doi: 10.1016/j.neubiorev.2009.07.008. [DOI] [PubMed] [Google Scholar]
- Lloyd-Fox S, Szeplaki-Kollod B, Yin J, Csibra G. Are you talking to me? Neural activations in 6-month-old infants in response to being addressed during natural interactions. Cortex. 2015;70:35–48. doi: 10.1016/j.cortex.2015.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macnab A, Shadgan B. Biomedical applications of wireless continuous wave near infrared spectroscopy. Biomed Spectroscopy and Imaging. 2012;1:205–222. [Google Scholar]
- Mahmoudzadeh M, Dehaene-Lambertz G, Wallois F. Electrophysiological and hemodynamic mismatch responses in rats listening to human speech syllables. PLoS One. 2017;12:e0173801. doi: 10.1371/journal.pone.0173801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maidan I, Bernad-Elazari H, Gazit E, Giladi N, Hausdorff JM, Mirelman A. Changes in oxygenated hemoglobin link freezing of gait to frontal activation in patients with Parkinson disease: an fNIRS study of transient motor-cognitive failures. J Neurol. 2015;262:899–908. doi: 10.1007/s00415-015-7650-6. [DOI] [PubMed] [Google Scholar]
- Maidan I, Nieuwhof F, Bernad-Elazari H, Reelick MF, Bloem BR, Giladi N, Deutsch JE, Hausdorff JM, Claassen JAH, Mirelman A. The role of the frontal lobe in complex walking among patients with Parkinson’s disease and healthy older adults: an fNIRS study. Neurorehabil Neural Repair. 2016;30:963–971. doi: 10.1177/1545968316650426. [DOI] [PubMed] [Google Scholar]
- Martin C, Zheng Y, Sibson NR, Mayhew JE, Berwick J. Complex spatiotemporal haemodynamic response following sensory stimulation in the awake rat. Neuroimage. 2013;66:1–8. doi: 10.1016/j.neuroimage.2012.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marumo K, Takizawa R, Kinou M, Kawasaki S, Kawakubo Y, Fukuda M, Kasai K. Functional abnormalities in the left ventrolateral prefrontal cortex during a semantic fluency task, and their association with thought disorder in patients with schizophrenia. Neuroimage. 2014;85:518–526. doi: 10.1016/j.neuroimage.2013.04.050. [DOI] [PubMed] [Google Scholar]
- Matsuo K, Kato N, Kato T. Decreased cerebral haemodynamic response to cognitive and physiological tasks in mood disorders as shown by near-infrared spectroscopy. Psychol Med. 2002;32:1029–1037. doi: 10.1017/s0033291702005974. [DOI] [PubMed] [Google Scholar]
- Matsuo K, Kato T, Taneichi K, Matsumoto A, Ohtani T, Hamamoto T, Yamasue H, Sakano Y, Sasaki T, Sadamatsu M, et al. Activation of the prefrontal cortex to trauma-related stimuli measured by near-infrared spectroscopy in posttraumatic stress disorder due to terrorism. Psychophysiology. 2003a;40:492–500. doi: 10.1111/1469-8986.00051. [DOI] [PubMed] [Google Scholar]
- Matsuo K, Taneichi K, Matsumoto A, Ohtani T, Yamasue H, Sakano Y, Sasaki T, Sadamatsu M, Kasai K, Iwanami A, et al. Hypoactivation of the prefrontal cortex during verbal fluency test in PTSD: a near-infrared spectroscopy study. Psychiatry Res Neuroimaging. 2003b;124:1–10. doi: 10.1016/s0925-4927(03)00093-3. [DOI] [PubMed] [Google Scholar]
- McKendrick R, Parasuraman R, Murtza R, Formwalt A, Baccus W, Paczynski M, Ayaz H. Into the wild: neuroergonomic differentiation of hand-held and augmented reality wearable displays during outdoor navigation with functional near infrared spectroscopy. Front Hum Neurosci. 2016;10:216. doi: 10.3389/fnhum.2016.00216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKendrick R, Mehta R, Ayaz H, Scheldrup M, Parasuraman R. Prefrontal hemodynamics of physical activity and environmental complexity during cognitive work. Hum Factors. 2017;59:147–162. doi: 10.1177/0018720816675053. [DOI] [PubMed] [Google Scholar]
- Mihara M, Miyai I, Hatakenaka M, Kubota K, Sakoda S. Sustained prefrontal activation during ataxic gait: a compensatory mechanism for ataxic stroke? Neuroimage. 2007;37:1338–1345. doi: 10.1016/j.neuroimage.2007.06.014. [DOI] [PubMed] [Google Scholar]
- Minagawa-Kawai Y, Naoi N, Kikuchi N, Yamamoto J, Nakamura K, Kojima S. Cerebral laterality for phonemic and prosodic cue decoding in children with autism. Neuroreport. 2009;20:1219–1224. doi: 10.1097/WNR.0b013e32832fa65f. [DOI] [PubMed] [Google Scholar]
- Miyai I, Yagura H, Oda I, Konishi I, Eda H, Suzuki T, Kubota K. Premotor cortex is involved in restoration of gait in stroke. Ann Neurol. 2002;52:188–194. doi: 10.1002/ana.10274. [DOI] [PubMed] [Google Scholar]
- Monden Y, Dan I, Nagashima M, Dan H, Uga M, Ikeda T, Tsuzuki D, Kyutoku Y, Gunji Y, Hirano D, et al. Individual classification of ADHD children by right prefrontal hemodynamic responses during a go/no-go task as assessed by fNIRS. NeuroImage-Clin. 2015;9:1–12. doi: 10.1016/j.nicl.2015.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moser SJ, Cutini S, Weber P, Schroeter ML. Right prefrontal brain activation due to Stroop interference is altered in attention-deficit hyperactivity disorder - a functional near-infrared spectroscopy study. Psychiatry Res Neuroimaging. 2009;173:190–195. doi: 10.1016/j.pscychresns.2008.10.003. [DOI] [PubMed] [Google Scholar]
- Muehlemann T, Reefmann N, Wechsler B, Wolf M, Gygax L. In vivo functional near-infrared spectroscopy measures mood-modulated cerebral responses to a positive emotional stimulus in sheep. Neuroimage. 2011;54:1625–1633. doi: 10.1016/j.neuroimage.2010.08.079. [DOI] [PubMed] [Google Scholar]
- Nagashima M, Monden Y, Dan I, Dan H, Mizutani T, Tsuzuki D, Kyutoku Y, Gunji Y, Hirano D, Taniguchi T, et al. Neuropharmacological effect of atomoxetine on attention network in children with attention deficit hyperactivity disorder during oddball paradigms as assessed using functional near-infrared spectroscopy. Neurophotonics. 2014a;1:025007. doi: 10.1117/1.NPh.1.2.025007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagashima M, Monden Y, Dan I, Dan H, Tsuzuki D, Mizutani T, Kyutoku Y, Gunji Y, Hirano D, Taniguchi T, et al. Acute neuropharmacological effects of atomoxetine on inhibitory control in ADHD children: a fNIRS study. NeuroImage-Clin. 2014b;6:192–201. doi: 10.1016/j.nicl.2014.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagashima M, Monden Y, Dan I, Dan H, Tsuzuki D, Mizutani T, Kyutoku Y, Gunji Y, Momoi MY, Watanabe E, et al. Neuropharmacological effect of methylphenidate on attention network in children with attention deficit hyperactivity disorder during oddball paradigms as assessed using functional near-infrared spectroscopy. Neurophotonics. 2014c;1:015001. doi: 10.1117/1.NPh.1.1.015001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negoro H, Sawada M, Iida J, Ota T, Tanaka S, Kishimoto T. Prefrontal dysfunction in attention-deficit/hyperactivity disorder as measured by near-infrared spectroscopy. Child Psychiatry & Hum Dev. 2010;41:193–203. doi: 10.1007/s10578-009-0160-y. [DOI] [PubMed] [Google Scholar]
- Nieuwhof F, Reelick MF, Maidan I, Mirelman A, Hausdorff JM, Rikkert MGO, Bloem BR, Muthalib M, Claassen JA. Measuring prefrontal cortical activity during dual task walking in patients with Parkinson’s disease: feasibility of using a new portable fNIRS device. Pilot and feasibility studies. 2016;2:59. doi: 10.1186/s40814-016-0099-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishimura Y, Tanii H, Fukuda M, Kajiki N, Inoue K, Kaiya H, Nishida A, Okada M, Okazaki Y. Frontal dysfunction during a cognitive task in drug-naive patients with panic disorder as investigated by multi-channel near-infrared spectroscopy imaging. Neurosci Res. 2007;59:107–112. doi: 10.1016/j.neures.2007.05.016. [DOI] [PubMed] [Google Scholar]
- Nishimura Y, Tanii H, Hara N, Inoue K, Kaiya H, Nishida A, Okada M, Okazaki Y. Relationship between the prefrontal function during a cognitive task and the severity of the symptoms in patients with panic disorder: a multi-channel NIRS study. Psychiatry Res Neuroimaging. 2009;172:168–172. doi: 10.1016/j.pscychresns.2009.01.001. [DOI] [PubMed] [Google Scholar]
- Noda T, Yoshida S, Matsuda T, Okamoto N, Sakamoto K, Koseki S, Numachi Y, Matsushima E, Kunugi H, Higuchi T. Frontal and right temporal activations correlate negatively with depression severity during verbal fluency task: a multi-channel near-infrared spectroscopy study. J Psychiatr Res. 2012;46:905–912. doi: 10.1016/j.jpsychires.2012.04.001. [DOI] [PubMed] [Google Scholar]
- Nozawa T, Sasaki Y, Sakaki K, Yokoyama R, Kawashima R. Interpersonal frontopolar neural synchronization in group communication: an exploration toward fNIRS hyperscanning of natural interactions. Neuroimage. 2016;133:484–497. doi: 10.1016/j.neuroimage.2016.03.059. [DOI] [PubMed] [Google Scholar]
- Ogawa S, Lee T-M, Kay AR, Tank DW. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc Natl Acad Sci USA. 1990;87:9868–9872. doi: 10.1073/pnas.87.24.9868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohta H, Yamagata B, Tomioka H, Takahashi T, Yano M, Nakagome K, Mimura M. Hypofrontality in panic disorder and major depressive disorder assessed by multi-channel near-infrared spectroscopy. Depress Anxiety. 2008;25:1053–1059. doi: 10.1002/da.20463. [DOI] [PubMed] [Google Scholar]
- Oka N, Yoshino K, Yamamoto K, Takahashi H, Li S, Sugimachi T, Nakano K, Suda Y, Kato T. Greater activity in the frontal cortex on left curves: a vector-based fNIRS study of left and right curve driving. PLoS One. 2015;10:e0127594. doi: 10.1371/journal.pone.0127594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osaka N, Minamoto T, Yaoi K, Azuma M, Shimada YM, Osaka M. How two brains make one synchronized mind in the inferior frontal cortex: fNIRS-based hyperscanning during cooperative singing. Front Psychol. 2015;6:1811. doi: 10.3389/fpsyg.2015.01811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Packer AM, Russell LE, Dalgleish HW, Häusser M. Simultaneous all-optical manipulation and recording of neural circuit activity with cellular resolution in vivo. Nat Methods. 2015;12:140–146. doi: 10.1038/nmeth.3217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piper SK, Krueger A, Koch SP, Mehnert J, Habermehl C, Steinbrink J, Obrig H, Schmitz CH. A wearable multi-channel fNIRS system for brain imaging in freely moving subjects. Neuroimage. 2014;85:64–71. doi: 10.1016/j.neuroimage.2013.06.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pu S, Yamada T, Yokoyama K, Matsumura H, Kobayashi H, Sasaki N, Mitani H, Adachi A, Kaneko K, Nakagome K. A multi-channel near-infrared spectroscopy study of prefrontal cortex activation during working memory task in major depressive disorder. Neurosci Res. 2011;70:91–97. doi: 10.1016/j.neures.2011.01.001. [DOI] [PubMed] [Google Scholar]
- Pu SH, Nakagome K, Yamada T, Yokoyama K, Matsumura H, Mitani H, Adachi A, Nagata I, Kaneko K. The relationship between the prefrontal activation during a verbal fluency task and stress-coping style in major depressive disorder: a nearinfrared spectroscopy study. J Psychiatr Res. 2012;46:1427–1434. doi: 10.1016/j.jpsychires.2012.08.001. [DOI] [PubMed] [Google Scholar]
- Quan WX, Wu TN, Li ZH, Wang YD, Dong WT, Lv B. Reduced prefrontal activation during a verbal fluency task in Chinese-speaking patients with schizophrenia as measured by nearinfrared spectroscopy. Prog Neuro-Psychopharmacol Biol Psychiatry. 2015;58:51–58. doi: 10.1016/j.pnpbp.2014.12.005. [DOI] [PubMed] [Google Scholar]
- Quaresima V, Giosue P, Roncone R, Casacchia M, Ferrari M. Prefrontal cortex dysfunction during cognitive tests evidenced by functional near-infrared spectroscopy. Psychiatry Res Neuroimaging. 2009;171:252–257. doi: 10.1016/j.pscychresns.2008.02.002. [DOI] [PubMed] [Google Scholar]
- Roche-Labarbe N, Zaaimi B, Mahmoudzadeh M, Osharina V, Wallois A, Nehlig A, Grebe R, Wallois F. NIRS-measured oxy-and deoxyhemoglobin changes associated with EEG spike-and-wave discharges in a genetic model of absence epilepsy: the GAERS. Epilepsia. 2010;51:1374–1384. doi: 10.1111/j.1528-1167.2010.02574.x. [DOI] [PubMed] [Google Scholar]
- Roy CS, Sherrington CS. On the regulation of the blood-supply of the brain. J Physiol. 1890;11:85–158. doi: 10.1113/jphysiol.1890.sp000321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakudo A. Near-infrared spectroscopy for medical applications: current status and future perspectives. Clin Chim Acta. 2016;455:181–188. doi: 10.1016/j.cca.2016.02.009. [DOI] [PubMed] [Google Scholar]
- Schecklmann M, Dresler T, Beck S, Jay JT, Febres R, Haeusler J, Jarczok TA, Reif A, Plichta MM, Ehlis AC, et al. Reduced prefrontal oxygenation during object and spatial visual working memory in unpolar and bipolar depression. Psychiatry Res Neuroimaging. 2011a;194:378–384. doi: 10.1016/j.pscychresns.2011.01.016. [DOI] [PubMed] [Google Scholar]
- Schecklmann M, Schaldecker M, Aucktor S, Brast J, Kirchgassner K, Muhlberger A, Warnke A, Gerlach M, Fallgatter AJ, Romanos M. Effects of methylphenidate on olfaction and frontal and temporal brain oxygenation in children with ADHD. J Psychiatr Res. 2011b;45:1463–1470. doi: 10.1016/j.jpsychires.2011.05.011. [DOI] [PubMed] [Google Scholar]
- Shimodera S, Imai Y, Kamimura N, Morokuma I, Fujita H, Inoue S, Furukawa TA. Mapping hypofrontality during letter fluency task in schizophrenia: a multi-channel near-infrared spectroscopy study. Schizophr Res. 2012;136:63–69. doi: 10.1016/j.schres.2012.01.039. [DOI] [PubMed] [Google Scholar]
- Shortz AE, Pickens A, Zheng Q, Mehta RK. The effect of cognitive fatigue on prefrontal cortex correlates of neuromuscular fatigue in older women. J Neuroeng Rehab. 2015;12:115. doi: 10.1186/s12984-015-0108-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeshi K, Nemoto T, Fumoto M, Arita H, Mizuno M. Reduced prefrontal cortex activation during divergent thinking in schizophrenia: a multi-channel NIRS study. Prog Neuro-Psychopharmacol Biol Psychiatry. 2010;34:1327–1332. doi: 10.1016/j.pnpbp.2010.07.021. [DOI] [PubMed] [Google Scholar]
- Takizawa R, Kasai K, Kawakubo Y, Marumo K, Kawasaki S, Yamasue H, Fukuda M. Reduced frontopolar activation during verbal fluency task in schizophrenia: a multi-channel nearinfrared spectroscopy study. Schizophr Res. 2008;99:250–262. doi: 10.1016/j.schres.2007.10.025. [DOI] [PubMed] [Google Scholar]
- Tamura R, Kitamura H, Endo T, Abe R, Someya T. Decreased leftward bias of prefrontal activity in autism spectrum disorder revealed by functional near-infrared spectroscopy. Psychiatry Res Neuroimaging. 2012;203:237–240. doi: 10.1016/j.pscychresns.2011.12.008. [DOI] [PubMed] [Google Scholar]
- Tang HH, Mai XQ, Wang S, Zhu CZ, Krueger F, Liu C. Interpersonal brain synchronization in the right temporoparietal junction during face-to-face economic exchange. Soc Cogn Affect Neurosci. 2016;11:23–32. doi: 10.1093/scan/nsv092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsujimoto S, Yasumura A, Yamashita Y, Torii M, Kaga M, Inagaki M. Increased prefrontal oxygenation related to distractor-resistant working memory in children with attention-deficit/hyperactivity disorder (ADHD) Child Psychiatry Hum Dev. 2013;44:678–688. doi: 10.1007/s10578-013-0361-2. [DOI] [PubMed] [Google Scholar]
- Urakawa S, Takamoto K, Ishikawa A, Ono T, Nishijo H. Selective medial prefrontal cortex responses during live mutual gaze interactions in human infants: an fNIRS study. Brain Topogr. 2015;28:691–701. doi: 10.1007/s10548-014-0414-2. [DOI] [PubMed] [Google Scholar]
- Vannasing P, Florea O, Gonzalez-Frankenberger B, Tremblay J, Paquette N, Safi D, Wallois F, Lepore F, Beland R, Lassonde M, et al. Distinct hemispheric specializations for native and nonnative languages in one-day-old newborns identified by fNIRS. Neuropsychologia. 2016;84:63–69. doi: 10.1016/j.neuropsychologia.2016.01.038. [DOI] [PubMed] [Google Scholar]
- Villringer A, Dirnagl U. Coupling of brain activity and cerebral blood flow: basis of functional neuroimaging. Cereb Brain Metabol Rev. 1995;7:240–276. [PubMed] [Google Scholar]
- Villringer A, Chance B. Non-invasive optical spectroscopy and imaging of human brain function. Trends Neurosci. 1997;20:435–442. doi: 10.1016/s0166-2236(97)01132-6. [DOI] [PubMed] [Google Scholar]
- Vogeli S, Lutz J, Wolf M, Wechsler B, Gygax L. Valence of physical stimuli, not housing conditions, affects behaviour and frontal cortical brain activity in sheep. Behav Brain Res. 2014;267:144–155. doi: 10.1016/j.bbr.2014.03.036. [DOI] [PubMed] [Google Scholar]
- Vogeli S, Wolf M, Wechsler B, Gygax L. Frontal brain activity and behavioral indicators of affective states are weakly affected by thermal stimuli in sheep living in different housing conditions. Front Vet Sci. 2015a;2:9. doi: 10.3389/fvets.2015.00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogeli S, Wolf M, Wechsler B, Gygax L. Housing conditions influence cortical and behavioural reactions of sheep in response to videos showing social interactions of different valence. Behav Brain Res. 2015b;284:69–76. doi: 10.1016/j.bbr.2015.02.007. [DOI] [PubMed] [Google Scholar]
- von Luhmann A, Herff C, Heger D, Schultz T. Toward a wireless open source instrument: functional near-infrared spectroscopy in mobile neuroergonomics and BCI applications. Front Hum Neurosci. 2015;9:617. doi: 10.3389/fnhum.2015.00617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Luhmann A, Wabnitz H, Sander T, Muller KR. M3BA: a mobile, modular, multimodal biosignal acquisition architecture for miniaturized EEG-NIRS-based hybrid BCI and monitoring. IEEE Trans Biomed Eng. 2017;64:1199–1210. doi: 10.1109/TBME.2016.2594127. [DOI] [PubMed] [Google Scholar]
- Wakita M, Shibasaki M, Ishizuka T, Schnackenberg J, Fujiawara M, Masataka N. Measurement of neuronal activity in a macaque monkey in response to animate images using near-infrared spectroscopy. Front Behav Neurosci. 2010;4:31. doi: 10.3389/fnbeh.2010.00031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe A, Kato T. Cerebrovascular response to cognitive tasks in patients with schizophrenia measured by nearinfrared spectroscopy. Schizophr Bull. 2004;30:435–444. doi: 10.1093/oxfordjournals.schbul.a007090. [DOI] [PubMed] [Google Scholar]
- Weber P, Lutschg J, Fahnenstich H. Cerebral hemodynamic changes in response to an executive function task in children with attention-deficit hyperactivity disorder measured by near-infrared spectroscopy. J Dev Behav Pediatr. 2005;26:105–111. doi: 10.1097/00004703-200504000-00005. [DOI] [PubMed] [Google Scholar]
- Wolf T, Lindauer U, Reuter U, Back T, Villringer A, Einhaupl K, Dirnagl U. Noninvasive near infrared spectroscopy monitoring of regional cerebral blood oxygenation changes during peri-infarct depolarizations in focal cerebral ischemia in the rat. J Cereb Blood Flow Metab. 1997;17:950–954. doi: 10.1097/00004647-199709000-00004. [DOI] [PubMed] [Google Scholar]
- Xiao T, Xiao Z, Ke XY, Hong SS, Yang HY, Su YL, Chu KK, Xiao X, Shen JY, Liu YJ. Response inhibition impairment in high functioning autism and attention deficit hyperactivity disorder: evidence from near-infrared spectroscopy Data. PLoS One. 2012;7:e46569. doi: 10.1371/journal.pone.0046569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu LW, Wang BT, Xu GC, Wang W, Liu ZA, Li ZY. Functional connectivity analysis using fNIRS in healthy subjects during prolonged simulated driving. Neurosci Lett. 2017;640:21–28. doi: 10.1016/j.neulet.2017.01.018. [DOI] [PubMed] [Google Scholar]
- Yasumura A, Kokubo N, Yamamoto H, Yasumura Y, Nakagawa E, Kaga M, Hiraki K, Inagaki M. Neurobehavioral and hemodynamic evaluation of Stroop and reverse Stroop interference in children with attention-deficit/hyperactivity disorder. Brain Dev. 2014;36:97–106. doi: 10.1016/j.braindev.2013.01.005. [DOI] [PubMed] [Google Scholar]
- Zaidi AD, Munk MH, Schmidt A, Risueno-Segovia C, Bernard R, Fetz E, Logothetis N, Birbaumer N, Sitaram R. Simultaneous epidural functional near-infrared spectroscopy and cortical electrophysiology as a tool for studying local neurovascular coupling in primates. Neuroimage. 2015;120:394–399. doi: 10.1016/j.neuroimage.2015.07.019. [DOI] [PubMed] [Google Scholar]


