Abstract
The Cassia auriculata herb has been traditionally used in India for medicinal purposes to treat hyperglycemia, diabetes, rheumatism, asthma, and skin diseases. In the present study, ethanolic extract of Cassia auriculata flower (Et-CAF) depicted anti-hyperlipidemic effect in the budding yeast cells. The hyperlipidemic conditions were induced in the yeast cells with oleic acid which showed an increase in triacylglycerol (TAG) and sterol esters (SE), and was supported by the mRNA expression of LRO1 and DGA1 (involved in TAG formation); as well as ARE1 and ARE2 (involved in SE formation). The anti-hyperlipidemic effect by the Et-CAF was compared with the commercial drug Atorvastatin. The lipid droplets were increased in the hyperlipidemic yeast cell that was observed under the confocal microscope with BODIPY staining; Atorvastatin and Et-CAF reduced the lipid droplets. This study revealed that the anti-hyperlipidemic effect in Et-CAF has gained importance and might be used to fill the gap created by the allopathic drugs.
Electronic supplementary material
The online version of this article (doi:10.1007/s13197-017-2735-0) contains supplementary material, which is available to authorized users.
Keywords: Cassiaauriculata flower, Hyperlipidemia, Neutral lipids, Saccharomyces cerevisiae, Triacylglycerol, Sterol esters, Lipid droplets
Introduction
Cassia auriculata (CA) belongs to the family of Caesalpiniaceae and is used in Asian beverages (Nadkarni 2002). Herbal teas are often consumed concurrently with conventional medications. In the traditional Ayurvedic medicine, CA is widely used for treating diabetes, rheumatism, asthma, and skin diseases (Kirtikar and Basu 2006). It has also been used in the treatment of hyperlipidemia and various other diseases. The tea prepared from the leaves of CA is used in chronic fever and fruits are antihelminthic (Joshi 2000). It was observed that South Indian tribals prepare a paste from CA leaves in vinegar, which is applied on the skin for various skin diseases (Jeeva et al. 2007: Kingston et al. 2009). Dried powder of CA flowers mixed with goat milk is taken orally to prevent white discharge in Tamilnadu (Muthu et al. 2006) and is also used in spermatorrhoea (Joshi 2000). CA is considered to be beneficial for individuals suffering from constipation, and diseases of the urinary tract. In Ayurvedic medicine, CA is used to treat diabetes, conjunctivitis, joint and muscle pain (rheumatism), ophthalmia, jaundice, liver disease, and urinary tract disorders (Joshi 2000). A mixture called ‘‘avarai panchaga choornam” prepared from dried powdered plant parts (equal amount of leaves, roots, flowers, bark and unripe fruits) is used to treat diabetes (Latha and Pari 2003; Pari and Latha 2002). Some constituents such as flavonoids, polysaccharides, anthracene derivatives, dimeric procyanidins, and β-sitosterol have been reported in various parts of CA (Chattopadhyaya et al. 1996). Phenolic compounds such as phenolic acid, flavonoids, tannins, catechins, and anthocyanins are the secondary plant metabolites that have metal chelating properties and serve as natural antioxidants (Halliwell et al. 2005). Hyperlipidemia (high cholesterol and triglyceride levels), and dyslipidemia are common in the modern society and are known to increase the risk of heart attack, stroke and other cardiovascular diseases like atherosclerosis and coronary heart diseases (Harikumar et al. 2013). The increasing morbidity and mortality from the hyperlipidemic conditions are the biggest challenge to nutritionists and medical scientists all over the world. The conventional drugs are usually toxic because they are highly active. Lipid lowering drugs like fibrates, Atorvastatin, and bile acid sequestrants are used in treating hyperlipidemia, but they are known to cause side effects (Chattopadhyaya et al. 1996), including diarrhea, nausea, myositis and abnormal liver function severely handicapping their application. In contrast to conventional medicines, herbal medicines are often perceived as ‘natural.’ Thus new lipid-lowering agents with high therapeutic value and minimum tolerable side effects from natural sources are under exploration. One such traditional plant is CA commonly called Tanner’s Cassia in English and as “Avarai” in Tamil (family: Cesalpinaceae). It is an evergreen shrub that grows in India. The flowers, leaves, stem, root and unripe fruit are profoundly used in Ayurvedic medicine.
The yeast Saccharomyces cerevisiae is a valuable model system and used in the current preliminary work to study the metabolic pathways and cellular mechanisms of hyperlipidemia since it is genetically tractable and shares many similarities with the humans (Botstein et al. 1997). Comparative genomic studies have shown that 40% of yeast proteins have at least one human homolog and 30% of genes involved in human disease have orthologs in yeast (Sturgeon et al. 2006). The major neutral lipids are triacylglycerol (TAG) and sterol esters (SE) (Connerth et al. 2009; Leber et al. 1994; Zinser et al. 1991) and stored as lipid droplets (LDs) in yeast. In yeast, the diacylglycerol acyltransferase, Dga1p, is the major enzyme for TAG synthesis (Sorger and Daum 2002). The yeast also contains Lro1p (homologous to mammalian lecithin cholesterol acyltransferase- LCAT), also involved in TAG biosynthesis (Dahlqvist et al. 2000; McLean et al. 1986). In S. cerevisiae, esterification of sterols is carried out by the acyl-CoA dependent reaction using ARE1, and ARE2 (SE syntheses) and these are as well homologous to human acyl-CoA cholesterol acyltransferase [ACAT] (CzarneckaH 1993; Yang et al. 1996). They are 49% identical to each other and exhibit approximately 24% identity to human ACAT. A high degree of conservation of the cellular lipid metabolism from yeast to mammalian cells provides an excellent possibility to use yeast S. cerevisiae for studying the general principles of lipid-related metabolic processes. In the present investigation, hyperlipidemia was induced by using oleic acid; and the anti-hyperlipidemic activity of C. auriculata flowers is being evaluated.
Materials and methods
Fresh Cassia auriculata flowers (CAF) were collected from Bharathidasan University Campus, Tiruchirappalli, Tamilnadu, India. The plant was identified and authenticated by Dr. S. John Britto, The Director, The Rapinat Herbarium and Centre for Molecular Systematics at St Joseph’s College Tiruchirappalli, Tamilnadu India. A voucher specimen (No. RVK 001). The flowers were dried under shade thoroughly and powdered.
Extraction and fractionation
The CAF powder was suspended in 0.4% DMSO and successively fractionated in various organic solvents such as chloroform, acetone, ethyl acetate, and ethanol. The fractions were dried in a vacuum drier, and the equal amount of the vacuum dried CAF extracts, and the standard drug Atorvastatin were dissolved in 1000 µl of DMSO (0.4%). Give details.
Yeast strains and growth condition
Wild-type S. cerevisiae strain BY4741 [MATa his3Δ1; leu2Δ0; met15Δ0; ura3Δ0] was grown in 5 ml of YPD (1% yeast extract, 2% peptone, and 2% dextrose) medium (pH 7.0) with aeration at 30 °C for 16 h. Then0.1 OD (A600nm) of cells was harvested and transferred to 25 ml of YPD media, and simultaneously various concentrations of oleic acid (0.125, 0.25, 0.5 mM) was added to the yeast cells to induce hyperlipidemia and cells were again grown for 16 h. The concentration of oleic acid at which the yeast cells showed maximum lipid accumulation was picked up and chosen for the further studies.
To the oleic acid-induced hyperlipidemic yeast cells (A600nm of 0.1 OD), various concentrations of CAF- extract (100, 250, 500 µg) were simultaneously added, and the cells were continued to grow for 16 h. The wild-type yeast cells were simultaneously treated with oleic acid and various concentrations (1, 2, 3, 4 and 5 mM) of Atorvastatin and grown for 16 h at 180 rpm. Similarly, the cells were simultaneously treated with oleic acid and various concentrations (100–500 µg) of CAF treated with different solvent extract (chloroform, acetone, ethyl acetate or ethanol). Simultaneously the wild-type cells were also treated with (0.4%) DMSO alone, or 500 µg of CAF in DMSO (0.4%), or 5 mM of Atorvastatin in DMSO (0.4%) and cells were grown for 16 h.
Lipid extraction and separation by TLC
An equal amount of cells from the above groups were taken, and total lipids from yeast cells were extracted by the method of Bligh and Dyer (Bligh and Dyer 1959). The total lipids were separated using silica gel thin layer chromatography (TLC), and the neutral lipid was separated using the following solvents: petroleum ether/diethyl ether/acetic acid (70:30:1, v/v). Individual neutral lipids were identified by comparing the Rf values of the unknown with the Rf values of the standard, and the TLC was quantified using Image J software.
RNA isolation and RT-PCR
The total RNA was isolated from yeast cells of the above- said groups using TRIzol reagent; the genomic DNA contamination was removed with the help of DNase. The extracted RNA was quantified using the Bio Photometer (Eppendorf, Hamburg, Germany). Two micrograms of total RNA was used for first-strand cDNA synthesis using a Prime Script RT reagent kit (Fermentas) following the manufacturer’s instructions. Two micrograms of cDNA were used as a template for the PCR amplification of target genes, LRO1, DGA1, ARE1, ARE2 and the internal control, ACT1. PCR for all the genes was performed with an initial incubation at 94 °C for 5 min, followed by 30 cycles at 94 °C for 30 s, 60 °C for 30 s, and 72 °C for 30 s. The reaction was terminated by a final incubation at 72 °C for 10 min. The mRNA expressions was analyzed in triplicates, and quantified. ACT1 was used as an endogenous control used for normalization. List of primer sequence is mentioned in Table 1.
Table 1.
Primer sequences and expected product sizes of the genes amplified
| S. No. | Gene | Primer | Size (Bp) |
|---|---|---|---|
| 1 | LRO1 | 5′AAATGGGTCGAGGCTGAAGG3′ 5′ATTCGACCAGTGCTTGTGGT3′ |
528 |
| 2 | DGA1 | 5′GGAGCGTTTGCAACAGAAGG3′ 5′AATTCTGCATCCGGTACCCC3′ |
632 |
| 3 | ARE1 | 5′TCGAGTTGGCTTTCATCCCC3′ 5′TGTCGAAGAGCAGGTGGAAC3′ |
558 |
| 4 | ARE2 | 5′GATCTGCGCCATCTTCGGTA3′ 5′GACACTTGGTCCCATGCAGA3′ |
589 |
| 5 | ACT1 | 5′ACGTCGCCTTGGACTTCGAA3′ 5′AGATGGAGCCAAAGCGGTGA3′ |
317 |
Lipid droplets staining
An Equal amount of cells from the above groups were resuspended in 2% para-formaldehyde for fixation. After washing with distilled water, BODIPY 493/503 (0.5 μg/ml) was added to the cell suspension and incubated for 20 min at room temperature. After two washes with PBS buffer, cells were resuspended in 50 μl of PBS and observed with a laser scanning fluorescence microscope (Zeiss LSM710, with 100× oil objective) directly after staining.
Results
Effect of oleic acid to induce lipid accumulation
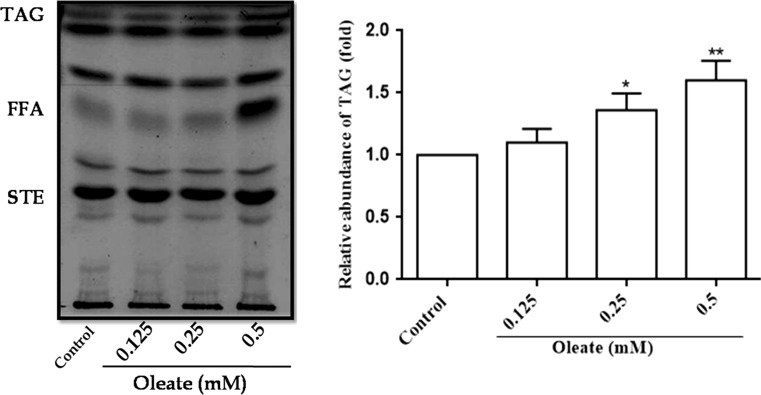
Oleic acid in a concentration (0.125, 0.25 and 0.5 mM) dependent manner accumulated TAG when compared with the wild-type control cells. Hence 0.5 mM was chosen as the optimum concentration for further experiments (Fig. 1).
Fig. 1.
Lipid accumulation in yeast cells by oleic acid induction. An Equal amount of yeast cells (A600nm = 0.1 OD) were treated with different concentrations (0.1–0.5 mM) of oleic acid, and the cells were grown in YPD media for 16 h at 30 °C. Equal amount of cells were taken, and the lipids were extracted and neutral lipid separated on TLC plates, using the solvent system (petroleum ether: diethyl ether: acetic acid, 70:30:1, v/v). Individual neutral lipids were identified by comparing the Rf values of the unknown with the Rf values of the standard. Values are the mean ± SD of three separate experiments, each performed in triplicate. Control cells were compared with treated groups. Values are the mean ± SD of three separate experiments, each performed in triplicate. Control cells were compared with treated groups. Values are statistically significant at **p < 0.01, *p < 0.05
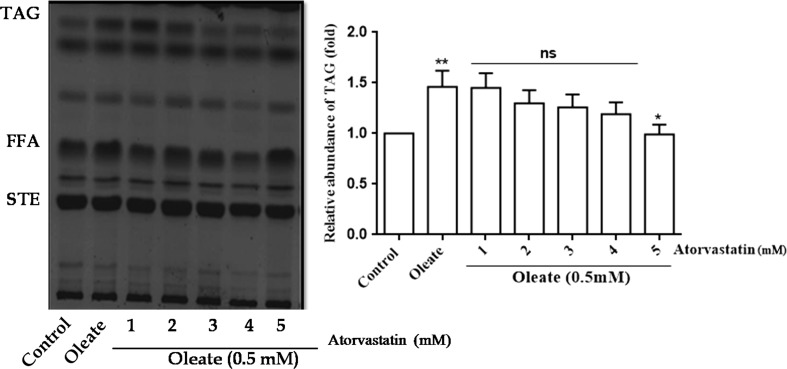
Effect of Atorvastatin on hyperlipidemia induced yeast cells
The drug Atorvastatin has been commercially used as an anti-hyperlipidemic drug. The hyperlipidemia induced by oleic acid in yeast cells were treated with different concentrations of Atorvastatin (1–5 mM). The TAG levels were restored to near normal levels with 5 mM Atorvastatin, and so this 5mMconcentration was chosen as the optimal concentration and used for all further experiments (Fig. 2).
Fig. 2.
Effect of Atorvastatin on neutral lipid pattern in hyperlipidemic yeast cells. An equal amount of yeast cells (A600nm = 0.1 OD) were treated with 0.5 mM of oleic acid and with different concentrations (1–5 mM) of Atorvastatin (standard anti-hyperlipidemic drug) and the cells were grown in YPD media for 16 h at 30 °C. Equal amount of cells were taken, and the lipids were extracted, and the neutral lipids separated and identified as described in Fig. 1. Values are the mean ± SD of three separate experiments, each performed in triplicate. Values are the mean ± SD of three separate experiments, each performed in triplicate. Control cells were compared with treated groups. Values are statistically significant at **p < 0.01
Effect of CAF (chloroform, acetone and ethyl acetate extracts) on hyperlipidemia induced yeast cells
The yeast cells were simultaneously treated with 0.5 mM oleic acid and with various concentrations of CAF extracts (from various extracts like chloroform, acetone and ethyl acetate) and the TAG levels were quantified. Yeast TAG level was not significantly reduced in the extracts(CAF- chloroform, acetone, ethyl acetate)treated cells even with the concentration up to 500 µg (Supplement Figs. 1, 2, 3).
Effect of CAF (ethanolic extract) on hyperlipidemia induced yeast cells
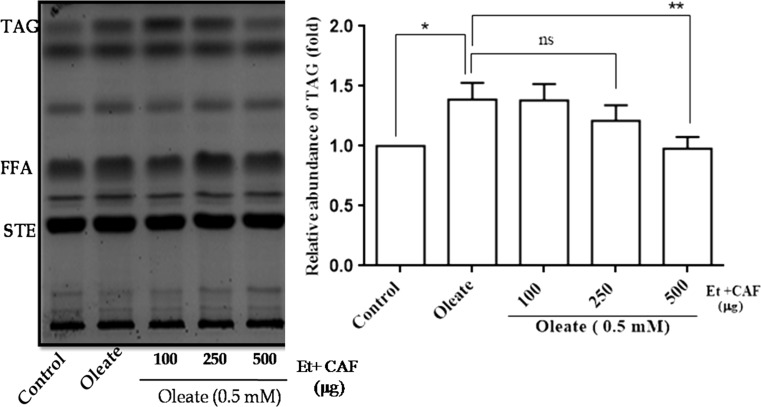
The yeast cells were simultaneously treated with 0.5 mM oleic acid and with various concentrations of CAF-ethanolic extract (100, 250, 500 µg) and the yeast TAG level quantified. Yeast TAG level was reduced significantly (Fig. 3), in CAF-ethanolic extract treated cells and the decrease was concentration dependent (up to 500 µg concentration).
Fig. 3.
Effect of CAF (ethanolic extract) on neutral lipid pattern in hyperlipidemic yeast cells. An Equal amount of yeast cells (A600nm = 0.1 OD) were treated with 0.5 mM of oleic acid and with different concentrations (100, 250 and 500 µg) of CAF (ethanolic extract) and grown in YPD media for 16 h at 30 °C. Equal amount of cells were taken, and the lipids were extracted, and the neutral lipids separated and identified as described in Fig. 1. Values are the mean ± SD of three separate experiments, each performed in triplicate. Control cells were compared with treated groups. Values are statistically significant at **p < 0.01, *p < 0.05
In our study, we used oleic acid to induce hyperlipidemia and investigated the effect of different fractions of Cassia auriculata flowers (CAF). The CAF was extracted by four solvents (chloroform, acetone, ethyl acetate and ethanol). The ethanolic fraction alone depicted anti-hyperlipidemic effect, and the TAG levels were reduced in yeast cells in a concentration dependent manner (Fig. 3). Hence the CAF-ethanolic fraction (500 µg) was used for further studies.
Expression of neutral lipid genes in hyperlipidemia-induced yeast cells treated with CAF (ethanolic extract)
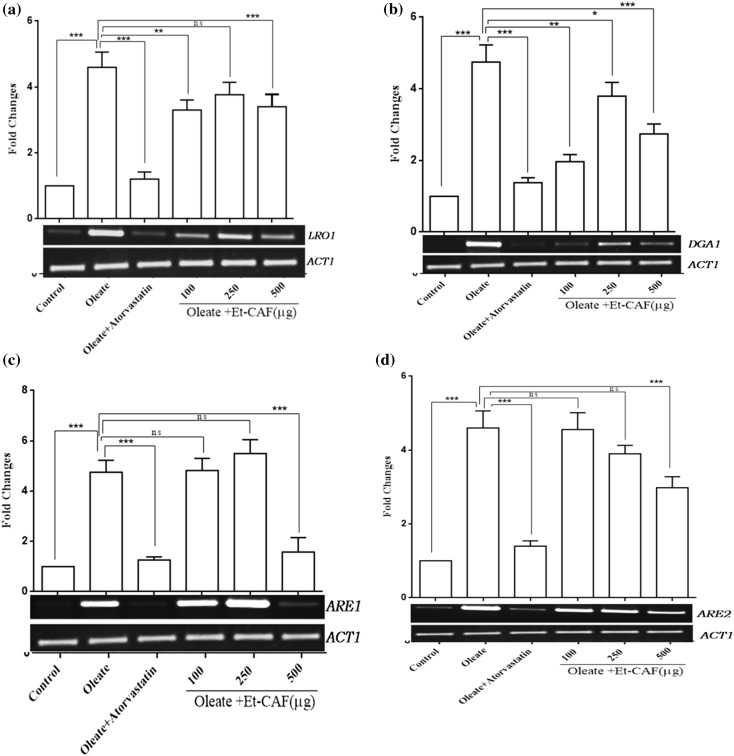
The lecithin cholesterol acyltransferase (LRO1) and diacylglycerol acyltransferase (DGA1) contribute to triacylglycerol synthesis. The LRO1 gene was ~4.2 fold up-regulated after oleic acid induction. Atorvastatin treatment brought the gene expression level back to control levels, and CAF (ethanolic extract) also reduced the LRO1 (in a concentration dependent manner) but to a lesser extent than Atorvastatin (Fig. 4a). The DGA1 gene was ~4.3 fold up-regulated after oleic acid induction. Atorvastatin treatment brought the gene expression level back to control levels, and CAF (ethanolic-extract) also reduced the DGA1 expression level back to control levels as with Atorvastatin. The CAF (ethanolic extract) even at 100 µg concentration reduced the DGA1 expression level back to control levels and remained constant even with the increase in CAF (ethanolic-extract) up to 500 µg (Fig. 4b).
Fig. 4.
Effect of CAF (ethanolic extract) on the expression of lipid droplet genes in hyperlipidemic yeast cells. An Equal amount of yeast cells (A600nm = 0.1 OD) were treated with 0.5 mM of oleic acid and with different concentrations (100, 250 and 500 µg) of CAF (ethanolic extract) and grown in YPD media for 16 h at 30 °C. The total RNA was isolated and converted to cDNA. The expression of a LRO1, b DGA1, c ARE1 and d ARE2 were quantified in wild-type control cells, and CAF-ethanolic extract-treated cells. Actin mRNA expression level was used as an internal control. Values are the mean ± SD of three separate experiments, each performed in triplicate. Control cells were compared with treated groups. Values are statistically significant ***p < 0.001, **p < 0.01, *p < 0.05
The sterol acyltransferases ARE1 andARE2 expression were up-regulated by 4.1 fold increased respectively with oleate exposure. Atorvastatin treatment brought the expression level of both the genes back to control levels. The CAF (ethanolic-extract) reduced the gene expression of ARE1in a concentration-dependent manner but to a lesser extent compared to Atorvastatin. The ARE2 gene was ~4.2 fold up-regulated after oleic acid induction, and Atorvastatin treatment brought the gene expression level back to control levels. However, the CAF (ethanolic extract) reduced the ARE2 expression to a lesser extent compared to Atorvastatin (Fig. 4c, d).
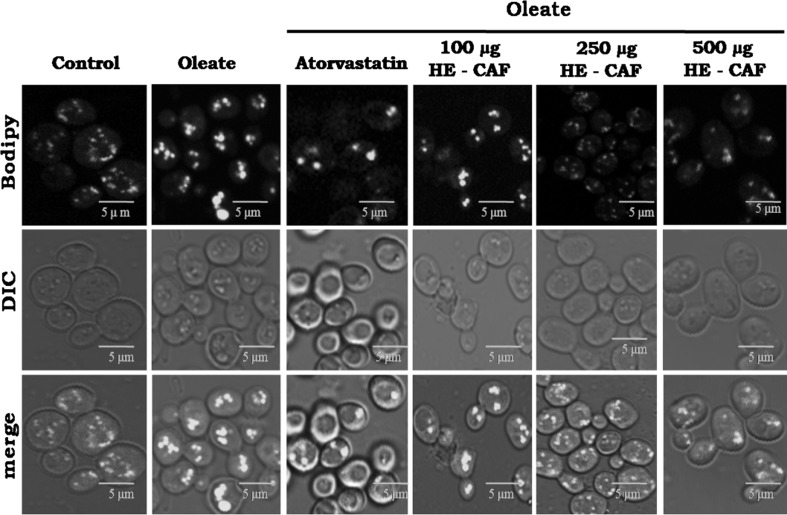
Lipid droplet staining by laser scanning fluorescent microscope
The lipid droplets (LDs), which has triacylglycerol and sterol esters were stained using BODIPY, a lipophilic dye. A significant increase in LD numbers was observed with oleic acid induction in yeast cells using laser scanning fluorescent microscopy. Atorvastatin treatment reduced the LD number to a great extent. Similarly, the CAF (ethanolic-extract) also significantly reduced the LD number in a concentration-dependent manner, and even 100 µg concentration (CAF-ethanolic extract) was able to reduce the LD number. Further with 250 and 500 µg of CAF (ethanolic-extract), the LD numbers were further reduced as in wild-type control cells (Fig. 5). The wild-type control cells were treated either with 20 µl of 0.4% DMSO, Et-CAF (500 µg) or with 5 mM of Atorvastatin. All the parameters mentioned above were measured in these groups, and the values were similar to the ones observed with the wild-type control cells, and hence these data were not shown.
Fig. 5.
Effect of CAF (ethanolic extract) on lipid droplet accumulation in hyperlipidemic yeast cells. An Equal amount of yeast cells (A600nm = 0.1 OD) were treated with 0.5 mM oleic acid and with different concentrations (100, 250 and 500 µg) of CAF (ethanolic extract) and the growth continued for 16 h at 30 °C. Cells were stained with BODIPY483/503, and the lipid droplets were viewed under laser scanning confocal microscopy
Identification of active component from the ethanolic extract of CAF
The ethanolic extract of CAF showed significant anti-hyperlipidemic effect. Hence the ethanolic CAF extract was subjected to HPLC, and we observed an active component Rutin that might account for the lipid-lowering effect (Supplementary material Fig. 4a, b). Hence for further experiments, we will focus analyzing the lipid-lowering activity of Rutin using animals as model system.
Discussion
Hyperlipidemia and lipid droplet (LD) formation was induced by oleic acid in the yeast (Marr et al. 2012; Binns et al. 2006) and same method was followed by us in the current study. During the induction of hyperlipidemia, the neutral lipid TAG was significantly increased. The medicinal plant Cassia auriculata has been reported to show diverse biological activities; pharmacological functions including anti-diabetic, and antioxidant activities (Pari and Latha 2002; Kumaran 2007; Surana et al. 2008). The anti-hyperlipidemic effect of CAF was tested in the four different solvent fractions (chloroform, acetone, ethyl acetate and ethanol). The CAF-ethanol fraction alone showed maximum anti-hyperlipidemic activity (Fig. 3). Fluvastatin and Atorvastatin are widely used anti-hypercholesterolemic drugs and we used Atorvastatin as a positive control in testing the anti-hyperlipidemic activity in the current study. Excess fatty acids and sterols in yeast are stored as TAG and SE in the form of lipid droplets (LDs). LDs are located in the cytosol and endoplasmic reticulum, and the LDs were significantly increased in both the numbers and size in the current study in the oleic acid induced yeast cells (Fig. 5). The treatment with Atorvastatin or CAF-ethanolic fraction significantly reduced the lipid droplets number and size (Fig. 5). The genes involved in the formation of storage lipid (LRO1, DGA1, ARE1, and ARE2) were all up-regulated in hyperlipidemic yeast cells and the treatment with Et-CAF fraction or Atorvastatin significantly reduced their mRNA as in wild-type control cells (Fig. 4). The pathways of lipid synthesis, storage, and mobilization, as well as their related regulatory processes, in the yeast model, are similar to the mammalian system, and so this model system can be used to contribute to the development of therapeutics for diseases related to dysfunctions of the lipid metabolism in humans. The recombinant yeast has been used to screen the anti-hyperlipidemic effect of CAF, and this study will replace animal work in the preliminary studies.
Conclusion
Our study showed that the ethanolic extract of Cassia auriclulata (Et-CAF) has anti-hyperlipidemic activity. Similar to the commonly used lipid lowering drug Atorvastatin, Et-CAF also attenuates the accumulated neutral lipids as well as lipid droplets in oleic acid induced hyperlipidemic yeast cells. The gene expression pattern also supports the observed pattern. In the future work, we would like to elucidate if the novel compound rutin found in Et-CAF possess anti-hyperlipidemic activity.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgement
The Junior Research Fellowship and instrumentation facility provided by the National Medicinal Plant Board (NMPB), Ministry of Ayush, DST-PURSE, Department of Science and Technology, India, and infrastructure facilities from the DST-FIST of Biochemistry Department, Life Sciences facilities of Bharathidasan University are gratefully acknowledged. We thank Prof. Ram Rajasekharan, (CFTRI, Mysore, India) for providing the yeast strain and molecular reagents for this study.
Compliance with ethical standards
Conflict of interest
The authors have no conflicts of interest to disclose.
Footnotes
Electronic supplementary material
The online version of this article (doi:10.1007/s13197-017-2735-0) contains supplementary material, which is available to authorized users.
References
- Binns D, Januszewski T, Chen Y, Hill J, Markin VS, Zhao Y, Gilpin C, Chapman KD, Anderson RG, Goodman JM. An intimate collaboration between peroxisomes and lipid bodies. J Cell Biol. 2006;173:719–731. doi: 10.1083/jcb.200511125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37:911–917. doi: 10.1139/o59-099. [DOI] [PubMed] [Google Scholar]
- Botstein D, Chervitz S.A, Cherry J.M (1997) Yeast as a model organism. Science New York, N.Y 277: 1259–1260 [DOI] [PMC free article] [PubMed]
- Chattopadhyaya R, Pathak D, Jindal DP. Antihyperlipidemic agents. A review. Indian Drugs. 1996;33:85–97. [Google Scholar]
- Connerth M, Grillitsch K, Kofeler H, Daum G. Analysis of lipid particles from yeast. J Bacteriol. 2009;173:2026–2034. doi: 10.1007/978-1-60761-322-0_18. [DOI] [PubMed] [Google Scholar]
- CzarneckaH Yokoyama S. Regulation of lecithin-cholesterol acyltransferase reaction by acyl acceptors and demonstration of its “idling” reaction. J Biol Chem. 1993;268:19334–19349. [PubMed] [Google Scholar]
- Dahlqvist A, Ståhl U, Lenman M, Banas A, Lee M, Sandager L, Ronne H, Stymne S. Phospholipid:diacylglycerol acyltransferase: an enzyme that catalyzes the acyl-CoA independent formation of triacylglycerol in yeast and plants. Proc Natl Acad Sci USA. 2000;97:6487–6492. doi: 10.1073/pnas.120067297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halliwell B, Rafter J, Jenner A. Health promotion by flavonoids, tocopherols, tocotrienols and other phenols: direct and indirect effects? Antioxidant or not ? Clin. Nutr. 2005;81:268S–2276. doi: 10.1093/ajcn/81.1.268S. [DOI] [PubMed] [Google Scholar]
- Harikumar K, Abdul Althaf S, Kishore KB, RamunaikM Suvarna CS. A Review on Hyperlipidemic. Int. J. Novel Trends Pharm. 2013;3:59. [Google Scholar]
- Jeeva GM, Jeeva S, Kingston C. Traditional treatment of skin diseases in south Travancore, Southern Peninsular India. Indian J. Traditional Knowledge. 2007;6:498–501. [Google Scholar]
- Joshi SG (2000) Cesalpinaceae—Cassia auriculata. Textbook of Medicinal Plants. IBH Publishing Oxford, pp : 119
- Kingston C, JeevaS Jeeva GM, Kiruba S, Mishra BP, Kannan D. Indigenous knowledge of various medicinal plants in treating skin diseases in Kanyakumari district. Southern India. Indian J. Traditional Knowledge. 2009;8:196–200. [Google Scholar]
- Kirtikar KR, Basu BD. Indian medicinal plants. 2. Dehradun: International Book Distributors; 2006. p. 868. [Google Scholar]
- KumaranA Karunakaran RJ. Antioxidant activity of Cassia auriculata flowers. Fitoterapia. 2007;78:46–47. doi: 10.1016/j.fitote.2006.09.031. [DOI] [PubMed] [Google Scholar]
- Latha M, Pari L. Preventive effects of Cassia auriculata L. flowers on brain lipid peroxidation in rats treated with streptozotocin. Mol Cell Biochem. 2003;243:23–28. doi: 10.1023/A:1021697311150. [DOI] [PubMed] [Google Scholar]
- Leber R, Zinser E, Zellnig G, Paltauf F, Daum G. Characterization of lipid particles of the yeast, Saccharomyces cerevisiae. Yeast. 1994;10:1421–1428. doi: 10.1002/yea.320101105. [DOI] [PubMed] [Google Scholar]
- Marr N, Foglia J, Terebiznik M, AthenstaedtK, .Zarem- bergV(2012) Controlling lipid fluxes at glycerol-3-phosphateacyltransferase step in yeast: unique contribution of Gat1p to oleic acid-induced lipid particle formation. J. Biol. Chem., 287:10251–1026 [DOI] [PMC free article] [PubMed]
- McLean J, Fielding C, Drayna D, Dieplinger H, Baer B, Kohr W, Henzel W, Lawn R. Cloning and expression of human lecithin-cholesterol acyltransferase cDNA. Proc. Natl. Acad. Sci. U. S. A. 1986;83:2335–2339. doi: 10.1073/pnas.83.8.2335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muthu C, Ayyanar M, RajaN Ignacimuthu S. Medicinal plants used by traditional healers in Kancheepuram district of Tamilnadu. India: J. Ethanobiol. Ethanomed. Vol; 2006. p. 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nadkarni KM. The Indian materiamedica. Mumbai: Popular Prakashan; 2002. p. 284. [Google Scholar]
- Pari L, Latha M. Antidiabetic activity of cassia auriculata flowers: effect on lipid peroxidation in streptozotocin diabetes rats. Pharmaceutical Biology. 2002;40:512–517. doi: 10.1076/phbi.40.7.512.14683. [DOI] [Google Scholar]
- Sorger D, Daum G. Synthesis of triacylglycerols by the acyl-coenzyme A:diacyl glycerol acyltransferase Dga1p in lipid particles of the yeast Saccharomyces cerevisiae. J Bacteriol. 2002;184:519–524. doi: 10.1128/JB.184.2.519-524.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sturgeon CM, Kemmer D, Anderson HJ, Roberge M. Yeast as a tool to uncover the Cellular targets of drugs. Biotechnol J. 2006;1:289–298. doi: 10.1002/biot.200500039. [DOI] [PubMed] [Google Scholar]
- Surana SJ, Gokhale SB, Jadhav RB, Sawant JB, Wadekar RL. Antihyperglycemic Activity of Various Fractions of Cassia auriculata Linn. in Alloxan Diabetic Rats. Indian Journal of Pharmaceutical Sciences. 2008;70:227–229. doi: 10.4103/0250-474X.41461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang H, Bard M, Bruner DA, Gleeson A, Deckelbaum RJ, Aljinovic G, Pohl TM, Rothstein R, Sturley SL. Sterol esterification in yeast: a two-gene process. Science. 1996;272(5266):1353–1356. doi: 10.1126/science.272.5266.1353. [DOI] [PubMed] [Google Scholar]
- Zinser E, Sperka-Gottlieb CD, Fasch EV, Kohlwein SD, Paltauf F, Daum G. Phospholipid synthesis and lipid composition of sub cellular membranes in the unicellular eukaryote Saccharomyces cerevisiae. J Bacteriol. 1991;173:2026–2034. doi: 10.1128/jb.173.6.2026-2034.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.