Abstract
Myelodysplastic syndromes (MDS) are a group of heterogeneous hematologic malignancies. This study aims to identify latent chromosomal abnormalities relevant to MDS, which may optimize the current diagnosis of MDS. Affymetrix CytoScan 750 K microarray platform was utilized to perform a genome-wide detection of chromosomal aberrations in the bone marrow cells of the patients. The findings were compared with the results from traditional karyotypic analysis and FISH to reveal latent chromosomal aberrations. Chromosomal gain, loss, and UPD, and complex karyotypes were identified in those samples. In addition to established cytogenetic aberrations detected by karyotypic analysis, CytoScan 750 K microarray also detected cryptic chromosomal lesions in MDS. Those latent defects underlying multiple gene mutations may construe the clinical variability of MDS. In Conclusion, Affymetrix CytoScan 750 K microarray is efficient in identifying latent chromosomal aberrations in MDS.
Introduction
Myelodysplastic syndromes (MDS) constitutes a group of heterogeneous premalignant disorder of clonal hematopoietic stem cells (HSC), typically characterized by hypercellular bone marrow with immature blood cell lineages, leading to ineffective hemopoiesis, dysplasia, peripheral blood cytopenia, and frequent evolution to acute myeloid leukemia (AML)1. MDS often affects the elderly patients with a mean age of 70 years, with an incidence of 3-5/100000 persons2. Chromosomal abnormalities are frequently found in the bone marrow cells of about 50–60% primary MDS and in 80% of secondary MDS patients3. Since chromosomal lesions have a great influence on the diagnosis and prognosis of MDS, it has become clear that precise cytogenetic analysis is vital for an accurate diagnosis of MDS4. Common chromosomal aberrations include copy number variation (CNV), acquired uniparental disomy (UPD), and complex karyotypes. Chromosomal gain may engender the amplification of oncogenes. On the contrary, chromosomal loss may lead to deletion of tumor suppressor genes (TSGs)5. UPD results from mitotic recombination when segments of homologous chromosomes are exchanged, hence both copies of a chromosome pair that are inherited from one parent. Identification of UPD has important significance for investigating the pathogenesis of MDS6. Complex chromosomal aberrations (≥3 aberrations) are found in about 20% of MDS patients and are related to an increased risk of progress into AML with unfavorable prognosis7. More importantly, additional previously cryptic chromosomal lesions may affect the phenotypes of well-established aberrations. Those small cryptic aberrations may have diagnostic significance.
Currently, traditional metaphase cytogenetics (MC) still remains a gold standard in karyotype analysis of MDS. However, 40–50% of myelodysplastic syndromes (MDS) patients do not exhibit karyotypic abnormalities that can be detected by classical cytogenetic techniques8. Especially, UPD is not recognizable by MC because the chromosome banding patterns remain preserved6. Fluorescence in situ hybridization (FISH) may complement MC analysis, but its application is confined to identify particular chromosomal lesions by the probes utilized. The genetic complexity of malignant cells implores more precise genome-wide techniques, in order to identify some cryptic chromosomal aberrations in mixed cell lines9. The advent of high-resolution single nucleotide polymorphism array (SNP-A) technique has enabled a genome-wide scanning of specific chromosomal abnormalities previously undetectable by conventional MC or FISH10.
In this study, we have demonstrated the feasibility of applying Affymetrix Cytoscan 750 K Microarray to identify chromosomal CNV, UPD, and complex karyotypes in MDS patients. We postulate that Affymetrix Cytoscan 750 K Microarray would not only identify established chromosomal defects, but also reveal previously subliminal chromosomal lesions in MDS. Identification of those latent chromosomal aberrations may contribute to the stratification of subtypes in MDS, assign appropriate phenotypes, and design individualized treatment.
Results
Clinical features of the patients
Conventional metaphase cytogenetic assay, FISH and Affymetrix Cytoscan 750 K Microarray were utilized to detect the common and latent chromosomal lesions for the patients. We selected 25 representative patients for our study, including 17 male and 8 female. Their ages range from 4-86 years old. 10 patients with CNV, 10 patients with UPD, and 5 patients with complex karyotypes were typically presented. The cohort comprises patients with RA(n = 4), RARS(n = 3), RCMD(n = 3), RCMD-RS(n = 1), RAEB-1(n = 2), RAEB-2(n = 2), 5q- syndrome(n = 1), MDS-U (n = 1), sAML(n = 8).
CNV
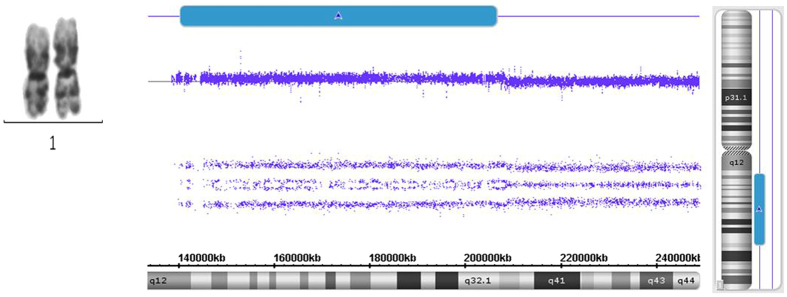
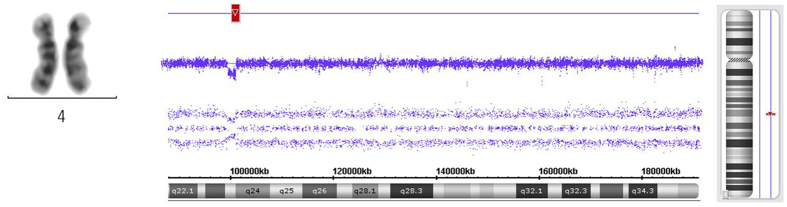
High resolution genome-wide Affymetrix Cytoscan 750 K Microarray is able to detect CNV larger than 100 Kb11. As for case 1#, 3#, 4#, 5#, MC presented concordant results with microarray, yet microarray provided more precise chromosomal lesions in the samples. For instance, in case 1#, MC only revealed trisomy 8. By contrast, Cytoscan 750 K Microarray exhibited additional cryptic gain(3q27.1-qter) and loss(6q23.2-qter). Specific lesions can be seen in Table 1, Supplementary Figure S1 and Table S1. In case 2#, MC revealed discordant result: t(3;21)(q26;q22). Cytoscan 750 K Microarray didn’t demonstrate this translocation, but unraveled cryptic gain(Yq11.222-pter) and loss (Yq11.222-qter) (detail in Supplementary Figure S2.1,2 and Table S2). From case 6# to case 10#, MC presented normal karyotypes. By contrast, Cytoscan 750 K Microarray disclosed loss(20q11.23-q13.13) in case 6#, loss(13q13.1-q21.33) in case 7#, loss(14q11.2) and gain(1q21.1-q32.2) in case 8# (Fig. 1), loss(5q15-q22.3) in case 9#, loss(4q24, 7q11.21) in case 10# (Fig. 2 and Table 1). The concrete size, location, and copy number state of these chromosomal lesions are available in our supplementary file. Those cryptic aberrations disclosed by Cytoscan 750 K Microarray are important for characterizing the patients with a diagnosis of MDS.
Table 1.
Comparison of CNV detected between Karyotypic analysis and Microarray.
| Patient NO. | Gender | Age (y) | Diagnosis | MC | Microarray | |
|---|---|---|---|---|---|---|
| gain | loss | |||||
| 1# | female | 71 | sAML | 47,XX, +8[20] | 3q27.1-qter +8 | 6q23.2-qter |
| 2# | male | 86 | sAML | 46,XY,t(3;21)(q26;q22) [19]/46,XY[1] | Yq11.222-pter | Yq11.222-qter |
| 3# | male | 69 | RAEB-2 | 46,XY, −20, +mar[16] /46,XY[4] | 20q13.2qter | 20p11.1-pter; 20q11.21-q13.2 |
| 4 # | male | 81 | RCMD | 46,X,-Y, +8[12]/46,XY[8] | +8 | Yp11.31-q11.23 |
| 5# | female | 60 | RCMD | 46,XX,del(20)(q11)[20] | 1q21.2; 20p11.1 | 20q11.23-q13.32 |
| 6# | male | 85 | RAEB-1 | 46,XY | 20q11.23-q13.13 | |
| 7# | female | 71 | RAEB-1 | 46,XX | 13q13.1-q21.33 | |
| 8# | female | 51 | RA | 46,XX | 1q21.1-q32.2 | 14q11.2 |
| 9# | male | 31 | 5q- syndrome | 46,XY | 5q15-q22.3 | |
| 10# | male | 78 | RARS | 46,XY | 4q24; 7q11.21 | |
Figure 1.
Comparison of chromosomal gain detected between MC and microarray. Although MC indicates normal chromosomal 1 in case 8#, Affymetrix Cytoscan 750 K Microarray still reveals cryptic gain(1q21.1-q32.2) with a large size.
Figure 2.
Comparison of chromosomal loss detected between MC and microarray. Although MC exhibits normal chromosomal 4 in case 10#, Affymetrix Cytoscan 750 K Microarray still discloses cryptic loss(4q24) with a small size.
UPD
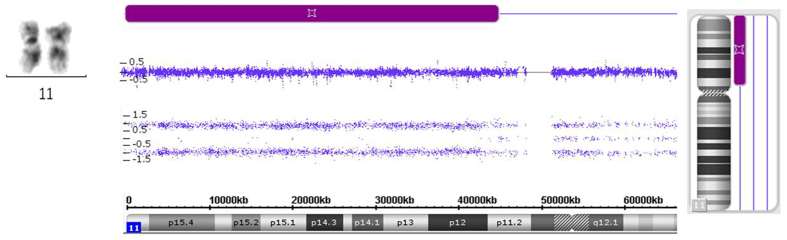
UPD still remains indiscernible by classical cytogenetic techniques, because it doesn’t change chromosomal banding patterns12. In this cohort, UPD in 10 MDS patients have not been detected by MC but by Cytoscan 750 K Microarray. For instance, in case 13#, although MC exhibits normal chromosome 11, Cytoscan 750 K Microarray still revealed UPD(11p11.2-pter) that approximately covered the entire short arm of chromosome 11 (Fig. 3). Additional recurrent UPDs relevant to MDS have also been identified by microarray, such as UPD(4q12-qter) in case 11#, UPD(13q11-qter) in case 12#, UPD(6p21.31-pter) in case 14#, UPD(11q13.1-qter) in case 15#, UPD(15q11.2-qter) in case 19#, and UPD(22q12.1-qter) in case 20#. Detailed aberrations of UPD have been listed in Table 2 and our supplementary file.
Figure 3.
UPD disclosed by microarray. Although MC indicates normal chromosome 11, cryptic UPD(11p11.2-pter) in the chromosomal short arm of case 13# is still disclosed by Affymetrix Cytoscan 750 K Microarray.
Table 2.
UPD exclusively identified by Cytoscan 750 K Microarray.
| Patient NO. | Gender | Age (y) | Diagnosis | MC | Microarray (UPD) |
|---|---|---|---|---|---|
| 11# | male | 86 | RCMD-RS | 46,XY | 4q12-qter; 6p22.2-p21.33 |
| 12# | female | 9 | RARS | 46,XX | 13q11-qter;Xq11.1-q13.1; Xq13.1-q21.1 |
| 13# | male | 4 | sAML | 46,XY | 11p11.2-pter; 2q11.1-q11.2 |
| 14# | male | 34 | RA | 46,XY | 6p21.31-pter |
| 15# | female | 76 | RAEB-2 | 46,XX | 11q13.1-qter |
| 16# | female | 51 | RCMD | 46,XX | 3p21.31-p21.1;17q22-qter |
| 17# | male | 43 | MDS-U | 46,XY | 3p21.31-p21.1;9p21.1-pter; 11p11.2-p11.12 |
| 18# | male | 77 | RARS | 46,XY | 11q12.3-q13.3; 14q24.1-qter |
| 19# | male | 75 | RA | 46,XY | 11p11.2-p11.12;15q11.2-qter |
| 20# | male | 85 | RA | 46,XY | 11p11.2-p11.12;22q12.1-qter |
Complex chromosomal lesions
Complex karyotypes in MDS often encompass three or more chromosomal abnormalities and are associated with unfavorable clinical outcomes13. Karyotypic analysis and Cytoscan 750 K Microarray have illustrated complex chromosomal lesions in 5 cases in point. Despite the fact that many large genomic aberrations were detected by metaphase cytogenetic assay, more hidden chromosomal loss and gain, especially UPD were still revealed by Cytoscan 750 K Microarray. In case 21#, MC indicates 40∼51, XY, ins(1)(p13p22p36), add(2)(q31), −5, −7, add(8)(p21), +9, add(9)(q34), −11, +mar1, +mar2, inc[cp6]. Comparatively, Cytoscan 750 K Microarray has revealed loss(1p22.3p21.2, 1p36.13p36.11, 5q14.3q21.3, 7q21.3q36.3, 12p13.31p12.1, 18q12.3qter), gain(8q11.1q24.3, 11p12q22.1, 13q11-q12.3), and UPD(7q22.1q31.32, 17p13.3p11.2) (Table 3, Supplementary Figure S21). As for case 22#, in spite of some “del” and “add” determined by karyotypic analysis, Cytoscan 750 K Microarray still disclosed a complex pattern of gains, losses and UPD in this case (Table 3, Supplementary Figure S22). Case 23#, 24#, and 25# have a series of complex chromosomal aberrations identified by Cytoscan 750 K Microarray that complement the results revealed by MC. By contrast, microarray analysis revealed cryptic UPD and a lot of smaller lesions, such as UPD(3p21.31-p21.1) and loss(3p14.1-q21.1, 5q13.3-q35.1, 9q21.11-q31.1) in case 25# (Table 3, Supplementary Figure S25). Generally, Cytoscan 750 K Microarray revealed complex rearrangements with multiple gains and losses. But MC indicated undefined materials exhibited as marker chromosomes (“+mar”) and chromosomal additions (“add”)13 in some cases. Detailed lesions of complex karyotypes have been listed in Table 3 and our Supplementary file.
Table 3.
Complex chromosomal lesions detected by MC and Microarray.
| Patient NO. | Gender | Age (y) | Diagnosis | Karyotypic analysis | Microarray | ||
|---|---|---|---|---|---|---|---|
| gain | loss | UPD | |||||
| 21# | male | 80 | sAML | 40∼51,XY,ins(1)(p13p22p36),add(2) (q31),-5,-7,add(8)(p21), + 9,add(9)(q34), -11, + mar1, + mar2,inc[cp6] | 8q11.1q24.3; 11p12q22.1; 13q11-q12.3 | 1p22.3p21.2; 1p36.13p36.11; 5q14.3q21.3; 7q21.3q36.3; 12p13.31p12.1; 18q12.3qter | 7q22.1q31.32; 17p13.3p11.2 |
| 22# | male | 71 | sAML | 41∼45,XY,del(2)(q33),-5,del(7)(p13),add(11)(q23), + 2∼3mar,inc[cp5]/46,XY[2] | 15q22.2-q23; 21q11.2q22.11 | 3p11.1-pter; 5q14.2-qter; Entire 7; 12p13.2p12.2; 15q24.1q25.1; 21q22.11q22.13 | 20q11.21-q11.23 |
| 23# | male | 28 | sAML | 42,X,add(Y)(p11),-5,-7,-11,-12,del(12)(p11),add(16)(q24),add(17)(q25),add(17)(p13),-22, + r[16]/46,XY[4] | 11q22.3-qter; 16q23.1-qter | 5q11.1-qter; Entire 7; 12p13.2-p11.23; 17p12-pter | |
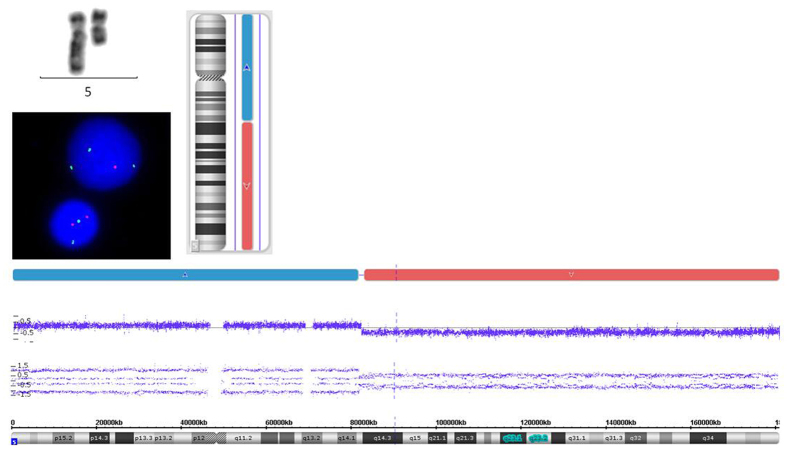
| 24# | male | 84 | sAML | 54∼56,XY, + 1, + 2,der(4;12)(q10;q10), + 5,del(5)(q13q31) × 2,add(7)(q32), + 8, + 11,del(12)(p11), + 17,i(17)(q10) × 2, + add(18)(q23), + 21, + 22[cp18]/46,XY[2] | Entire 1,2, 6, 8,10,11,18; 5q14.2pter; 9p23p21.2; 21q11.2qter; 22q11.1qter | 5q14.3qter; 12p13.31p11.21; 17q12 pter | 9p21.2qter |
| 25# | female | 53 | sAML | 41∼44,X,-X,-5,add(14)(p11),-16,-18,?del(20)(q11),add(21)(p11), + r, + mar,1dmin[cp9]/46,XX[1] | 5q12.3q13.2; 18q11.2-pter; 22q11.1-qter | 3p14.1-q21.1; 5q13.3-q35.1; 9q21.11-q31.1; Entire 16; 17p13.3p13.1; 18q11.2-qter; 20q11.21-qter; Xp21.3p11.21; Xq13.1-qter | 3p21.31-p21.1 |
Validation of microarray results
The resolution of conventional metaphase cytogenetic analysis is approximately 5 Mb14. In order to validate those lesions identified by microarray, we also initiated FISH to detect the chromosomal aberrations in some of those cases. For instance, in case 17#, UPD(3p21.31p21.1), UPD(9p21.1pter), and UPD(11p11.2p11.12) haven’t been detected by MC but by microarray. FISH also exhibit normal chromosome 5, 7, 8, 20 (Supplementary Figure S17, Table S17). In contrast, MC revealed +5 and del(5)(q13q31) in case 24#, while Cytoscan 750 K Microarray disclosed gain(5q14.2pter) and loss(5q14.3qter). FISH indicated loss of CSF1R signal, representing loss(5q33-34). So genomic loss in chromosomal 5 has been confirmed by FISH (Fig. 4). In addition, trisomy 8 in case 24# has also been validated by FISH (Supplementary Figure S24).
Figure 4.
Validation of genomic aberrations in chromosomal 5. As for 24#, MC revealed del(5)(q13q31), while microarray disclosed gain(5q14.2pter) and loss(5q14.3qter). FISH has validated loss(5q33-34).
Discussion
Chromosomal abnormalities are frequent in MDS and have many clinical implications. However, a great amount of patients don’t indicate cytogenetic abnormalities. MDS patients with the same chromosome lesions may have heterogeneous phenotypes, implying that some hidden genomic changes may exist among the patients15. In the present study, we utilized traditional MC and Affymetrix Cytoscan 750 K Microarray to identify latent chromosomal changes in a cohort of 25 patients. Our results indicated that high-resolution Affymetrix Cytoscan 750 K Microarray improves the identification of chromosomal aberrations by karyotypic analysis in MDS.
Gain and loss of gene copies may result in gene over-expression, absence of any functional transcript, or modest changes in gene expression16. We have unveiled chromosomal aberrations in regions defined as CNV of contiguous clones undetectable by classical karyotypic analysis. For instance, we identified cryptic gain (3q27.1-qter) and loss (6q23.2-qter) in case 1#. In this region, Loss(6q23.2-q23.3) has been reported to involve in sAML17. Furthermore, cryptic chromosomal aberrations detected by microarray have been useful for a comprehensive analysis of gene mutations in MDS. The copy number status of TET2 (on 4q24), IRF1 (5q31.1), NPM1 (5q35.1), LAMB4 (7q31.1), EZH2 (7q36.1), ETV6 (12p13.2), TP53 (17p13.1), NF1 (17q11.2), ASXL1 (20q11.21), RUNX1 (21q22.12), and STAG2 (Xq25) have been investigated in a large cohort of MDS patients18. In our cohort, we have identified a 1.48 Mb loss at 4q24 in case 10#. TET2 gene in this region is a tumor-suppressor gene19. The deletion or mutation of TET2 often predict inferior prognosis in patients with chronic myelomonocytic leukemia20. Although aberration in chromosome 5 has been considered as the most prevalent chromosomal lesion in MDS, some cryptic defects which affect additional key genes have not been clarified comprehensively. The major commonly deleted region (CDR) has been delineated at band 5q31.1. We have identified a 97.71 Mb loss(5q14.3qter) in case 24#. Deletion of 5q may engender haploinsufficiency of many critical genes, including ribosomal protein S14 (RPS14), casein kinase 1 α1 (CSNK1A1), adenomatous polyposis coli (APC), heat shock protein family A (HSP70) member 9 (HSPA9), early growth response 1 (EGR1), DEAD-box helicase 41 (DDX41), NPM1, TRAF-interacting protein with forkhead- associated domain B (TIFAB), Diaphanous-related formin 1 (DIAPH1), microRNA (miR)-145 and miR-146a21. Haploinsufficiency for the ribosomal gene RPS14 has been reported to impede erythroid differentiation in the 5q- syndrome22. Heterozygous deletion of CSNK1A1 may upregulate WNT signaling and stimulate stem cell expansion23, 24. Loss(5q14.3qter) in case 24# has been validated by FISH. We have also detected loss(7q11.21) in case 10# and monosomy 7 in case 22#, 23#. Deletion of 7q and monosomy 7 are also prevalent in MDS and often portend unfavorable outcome25. These chromosomal alterations can also cause haploinsufficiency of some key genes implicated in MDS. These genes comprise EZH2, CUX1 and MLL326–28. MLL3 haploinsufficiency cooperates with RAS mutation and Trp53 to exacerbate leukemia26. In addition, we have found gain(20q13.2qter) in case 3#. MacKinnon et al. have investigated AML and MDS patients with 20q amplification. They identified a 250 kb common region which subsumed HCK, TM9SF4, PLAGL2, and POFUT1 gene. These patients often had a higher proportion of erythroblasts. The amplification of 20q portends the existence of oncogene29. Generally, these latent chromosomal aberrations contain a lot of key genes which are tightly associated with the pathogenesis of MDS. CNV identified by Affymetrix Cytoscan 750 K Microarray can contribute to the differential diagnosis of subtypes in MDS.
Recent investigations have indicated that UPD can be responsible for homozygosity of mutations of critical genes within chromosomal regions30. Reduction to homozygosity as a result of UPD was preliminarily considered to be a mechanism for the inactivation of tumor suppressor genes30. We have demonstrated UPD in 10 cases with normal karyotypes. For example, we detected UPD(13q11-qter) in case 12#. FLT3 gene in 13q12 encodes class III receptor tyrosine kinase that regulates hematopoiesis31. FLT3-ITD internal tandem duplications have been observed during disease progression and confers an unfavorable prognosis32. We also identified UPD(11p11.2-pter) in case 13#. WT1 gene mutation has been reported in UPD 11p which is related to the pathogenesis of AML33. Furthermore, we have also found UPD (11q13.1-qter) in case 15#. The c-CBL gene is located in 11q23.37. Clonal selection of UPD 11q and CBL gene mutation often reflected the progression of MDS to AML34. It is noteworthy that in RCMD case 16#, the UPD of region 17q22-qter harbored the ETV4 gene, which encodes an ETS transcription factor indispensable for hematopoiesis35. Additionally, we have found UPD(9p21.1-pter) in case 17#. UPD 9p is tightly associated with a homozygous activating JAK2(V617F) gene mutation, implying serious prognosis7. Consequently, nonrandom segmental UPDs identified in this cohort may contribute to the investigation of the pathogenesis of MDS underlying large deletions.
MDS with complex chromosomal aberrations often herald short survival and an increased risk of evolution to AML36. Complex karyotypes with multiple chromosomal changes are found in about 20% of newly diagnosed MDS patients and are relevant to a poor prognosis3. We exhibited 5 cases with complex chromosomal lesions, which have been listed in our supplementary file. A combination of traditional karyotypic analysis with Affymetrix Cytoscan 750 K Microarray may well provide a more comprehensive detection of complex chromosomal aberrations in MDS.
Given the recent discovery of many recurrent gene mutations in MDS, it’s still urgent to validate prior mutational correlative data. The temporal order of mutation acquisition has reflected the importance of subclonal genetic events in MDS. For instance, mutations impacting RNA splicing and DNA methylation occur early in disease progression, while kinase activating mutations (such as KIT and NRAS) occur even later in disease progression37. Early detection of subclonal mutations may reflect significant prognostic variables in MDS38. In our cohort, we have found many discrepancies between MC and Cytoscan 750 K Microarray. Microarray analysis has a variable ability to detect mosaicism that FISH and karyotyping may not accurately detect the level of mosaicism13. The most likely reason for these discrepancies is probably that some aberrations are subclonal.
On the other hand, one criticism of microarray for detecting chromosomal aberrations in MDS is the possibility of “false positive” results or findings of unclear clinical significance. A proportion of alterations identified in the patients may reflect normal age-related chromosomal changes. For instance, we have found loss(Yq11.222-qter) in case 2#. Loss of the Y-chromosome (LOY) is described as both a normal age-related event and a marker of a neoplastic clone in hematologic diseases39. Paired normal DNA from the same MDS patient may reduce the number of false positives generated by microarray40.
Additionally, some patients with clonal cytopenia of undetermined significance (CCUS) that do not meet the criteria for MDS may also benefit from SNP-A. A recent study has applied combined comparative genomic hybridization and SNP-A to detect cryptic chromosomal lesions in both MDS and cytopenias of undetermined significance. Based on the combined array findings, 42% of patients with indeterminate morphologic findings were categorized as CCUS. Cryptic array findings among those patients comprised large-scale UPD (up to 118 Mb) and genomic deletion of loci implicated in MDS pathogenesis (eg, TET2 (4q22) and NUP98 (11p15)). The latent chromosomal lesions revealed by SNP-A helped to indicate clonal hematopoiesis and prompted classification as CCUS41. Hence microarray analysis significantly improves the detection rate of clinically significant findings.
In conclusion, Affymetrix Cytoscan 750 K Microarray have identified many cryptic chromosomal abnormalities relevant to MDS, which may interpret the clinical variability and enhance our understanding of the pathogenesis of MDS.
Material and Methods
Patients and Specimen
The cohort of this study comprises patients whose bone marrow aspirates were recruited in Kingstar Global company for pathologic diagnosis of MDS from December 2014 to July 2015. All specimens were acquired with patients’ approval, under the protocols permitted by Institutional Ethics Committee of Wuhan university, in comply with Helsinki Declaration. Informed consent was signed for each patient. And any publication of identifying information was also approved by the participants.
Cytogenetic analysis
Traditional G-banding Karyotypic analysis was initiated on bone marrow aspirates by trypsin and Giemsa dye. Short-term cell cultures were carried out in medium supplemented with GM-CSF or conditioned medium III. Then the cells were harvested and metaphase preparations were performed according to standard procedures. Karyotypes were depicted in the light of International System for Human Cytogenetic Nomenclature 201642. At least 20 metaphases per sample should be analyzed whenever possible.
FISH Fluorescence in situ hybridization (FISH) was performed according to the manufacturer’s protocols, in order to validate chromosomal aberrations detected by Affymetrix Cytoscan 750 K Microarray. A total of 400 interphase nuclei were evaluated by two independent pathologists under fluorescent microscope. The locus-specific probes were displayed in Table 4.
Table 4.
The probes and targets of FISH.
| Probe | Target |
|---|---|
| D5S23,D5S721/CSF1R | 5p15.2/5q33-34 |
| D7Z1/D7S486 | 7p11.1-q11.1/7q31 |
| D8Z2 | 8p11.1-q11.1 |
| D20S108 | 20q12 |
DNA preparation
DNA was extracted from bone marrow of individual patients using the QIAamp DNA Blood Mini Kit according to the manufacturer’s instructions. The concentration and quality of DNA samples were evaluated by Nanodrop 2000 spectrophotometer (Thermo Scientific). DNA integrity was assessed by 1% agarose gel electrophoresis. The quality controls (QC) of Affymetrix CytoScan 750 K microarray required that DNA concentration should be no less than 50 ng/µL, OD260/280 is about 1.9, OD260/230 is about 2.0.
Cytoscan 750 K Microarray Assay
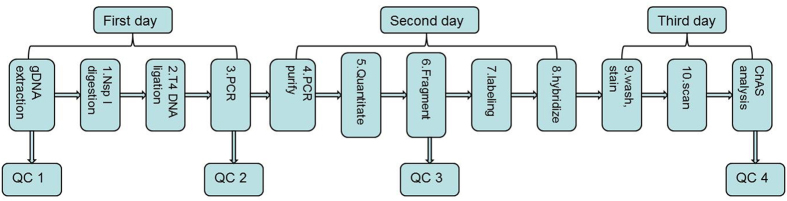
Affymetrix Cytoscan 750 K Microarray provides a genome-wide coverage with focus on cytogenetic relevant regions, including 550,000 markers for detecting copy number variation and 200,000 high performing SNP probes with genotype accuracy >99%. All probes are empirically selected for exceptional performance. The Affymetrix® CytoScan™ Assay protocol is optimized for processing 8 to 24 samples at a time to obtain whole genome copy number and SNP information. The Workflow of CytoScanTM Assay can be briefed as follows (Fig. 5):
Digestion of gDNA with Nsp I restriction endonuclease.
Ligation with Adaptor and T4 DNA Ligase.
Polymerase chain reaction (PCR) to amplify Ligated Samples and PCR Product Check.
PCR Product Purification with magnetic beads.
Quantitation of purified samples.
Fragmentation of Purified PCR Products and QC Gel Analysis.
Labeling the Fragmented DNA with TdT enzyme.
Hybridization with CytoScan 750 K Microarray at 50 °C oven for 16 to 18 hours.
Wash and Stain the genechips on Fluidics Station.
Scan the arrays in optic GeneChip Scanner 3000.
Figure 5.
The Workflow of Affymetrix Cytoscan 750 K Microarray. QC1 defines DNA concentration ≥ 50ng/µL, OD260/280≈1.9, OD260/230 ≈ 2.0. QC2 defines PCR products on 1% gel electrophoresis should be 150 bp-2000 bp, Purified PCR products ≥ 300 ng/µL, OD260/280 ≈ 1.9, OD260/230≈2.0. QC3 defines fragmentation products on 1% gel electrophoresis should be 25 bp-125 bp. QC4 defines: SNPQC ≥ 15.0; MAPD ≤ 0.25; Waviness SD ≤ 0.12.
All cases have followed the protocols and QC guidelines provided by the manufacturer.
Data analysis
The data of Cytoscan 750 K Microarray were analyzed using Chromosome Analysis Suite Version 2.0 (Affymetrix). The QC thresholds were: SNPQC ≥ 15.0; MAPD ≤ 0.25; Waviness SD ≤ 0.12. These QC metrics can evaluate the overall quality of SNP array data. Median Absolute Pairwise Difference (MAPD) represents the typical distance between marker pairs with respect to log2 ratios. SNPQC measures the degree of separation between genotype clusters aggregated across multiple markers. Waviness-SD gauges the differences between probe sets. The microarray data were interpreted according to the annotations of genome version GRCh37 (hg19). Only the samples which complied with QC criteria and identified CNV with over 100 Kb and at least 10 aberrant probes were chosen for further analysis. Identified CNVs were contrasted with the Database of Genomic Variants (http://dgv.tcag.ca/dgv/app/home) to exclude the polymorphic variations in healthy population. As for UPD, we used an algorithm that regards both location and size of >5 Mb aberrations in order to preclude nonclonal regions. To reckon the size of the affected genome in each patient, we recognized the total size of alterations in chromosomes, including CNV and UPD.
Electronic supplementary material
Acknowledgements
This study was supported by National Natural Science Foundation of China (NO. 81372407) and (NO. 81670123).
Author Contributions
Qibin Song, Yi Yao, Min Peng designed this study; Yuxin Chu wrote this manuscript; Weihong Yang and Xiaoqing Li performed experiments and analyzed the data; Shiang Huang managed patients and collected clinical samples.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-10551-3
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Lukackova R, Gerykova, Bujalkova M, Majerova L, Mladosievicova B. Molecular genetic methods in the diagnosis of myelodysplastic syndromes. A review. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158:339–345. doi: 10.5507/bp.2013.084. [DOI] [PubMed] [Google Scholar]
- 2.Schlegelberger B, et al. Update on cytogenetic and molecular changes in myelodysplastic syndromes. Leuk Lymphoma. 2012;53:525–536. doi: 10.3109/10428194.2011.618235. [DOI] [PubMed] [Google Scholar]
- 3.Zemanova Z, et al. Involvement of deleted chromosome 5 in complex chromosomal aberrations in newly diagnosed myelodysplastic syndromes (MDS) is correlated with extremely adverse prognosis. Leuk Res. 2014;38:537–544. doi: 10.1016/j.leukres.2014.01.012. [DOI] [PubMed] [Google Scholar]
- 4.Platzbecker U, et al. Update on developments in the diagnosis and prognostic evaluation of patients with myelodysplastic syndromes (MDS): consensus statements and report from an expert workshop. Leuk Res. 2012;36:264–270. doi: 10.1016/j.leukres.2011.11.005. [DOI] [PubMed] [Google Scholar]
- 5.Mitelman, F., Johansson, B. & Mertens, F. Mitelman database of chromosome aberrations and gene fusions in cancer. Available from: http://cgap.nci.nih.gov/Chromosomes/Mitelman (2016)
- 6.Gondek LP, et al. Single nucleotide polymorphism arrays complement metaphase cytogenetics in detection of new chromosomal lesions in MDS. Leukemia. 2007;21:2058–2061. doi: 10.1038/sj.leu.2404745. [DOI] [PubMed] [Google Scholar]
- 7.Svobodova K, et al. Copy number neutral loss of heterozygosity at 17p and homozygous mutations of TP53 are associated with complex chromosomal aberrations in patients newly diagnosed with myelodysplastic syndromes. Leuk Res. 2016;42:7–12. doi: 10.1016/j.leukres.2016.01.009. [DOI] [PubMed] [Google Scholar]
- 8.Thiel A, et al. Comprehensive array CGH of normal karyotype myelodysplastic syndromes reveals hidden recurrent and individual genomic copy number alterations with prognostic relevance. Leukemia. 2011;25:387–399. doi: 10.1038/leu.2010.293. [DOI] [PubMed] [Google Scholar]
- 9.Simons A, et al. Genome-wide arrays in routine diagnostics of hematological malignancies. Hum Mutat. 2012;33:941–948. doi: 10.1002/humu.22057. [DOI] [PubMed] [Google Scholar]
- 10.Huh J, et al. Different characteristics identified by single nucleotide polymorphism array analysis in leukemia suggest the need for different application strategies depending on disease category. Genes Chromosomes Cancer. 2013;52:44–55. doi: 10.1002/gcc.22005. [DOI] [PubMed] [Google Scholar]
- 11.Shin S, et al. Routine Chromosomal Microarray Analysis is Necessary in Korean Patients With Unexplained Developmental Delay/Mental Retardation/Autism Spectrum Disorder. Ann Lab Med. 2015;35:510–518. doi: 10.3343/alm.2015.35.5.510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hu Q, et al. The prevalence of chromosomal aberrations associated with myelodysplastic syndromes in China. Ann Hematol. 2016;95:1241–1248. doi: 10.1007/s00277-016-2698-7. [DOI] [PubMed] [Google Scholar]
- 13.Kolquist KA, et al. Microarray-based comparative genomic hybridization of cancer targets reveals novel, recurrent genetic aberrations in the myelodysplastic syndromes. Cancer Genet. 2011;204:603–628. doi: 10.1016/j.cancergen.2011.10.004. [DOI] [PubMed] [Google Scholar]
- 14.Iafrate AJ, et al. Detection of large-scale variation in the human genome. Nat Genet. 2004;36:949–951. doi: 10.1038/ng1416. [DOI] [PubMed] [Google Scholar]
- 15.Gondek LP, et al. Detection of cryptic chromosomal lesions including acquired segmental uniparental disomy in advanced and low-risk myelodysplastic syndromes. Exp Hematol. 2007;35:1728–1738. doi: 10.1016/j.exphem.2007.08.009. [DOI] [PubMed] [Google Scholar]
- 16.Mayrhofer M, Viklund B, Isaksson A. Rawcopy: Improved copy number analysis with Affymetrix arrays. Sci Rep. 2016;6 doi: 10.1038/srep36158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gondek LP, et al. Chromosomal lesions and uniparental disomy detected by SNP arrays in MDS, MDS/MPD, and MDS-derived AML. Blood. 2008;111:1534–1542. doi: 10.1182/blood-2007-05-092304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Haferlach T, et al. Landscape of genetic lesions in 944 patients with myelodysplastic syndromes. Leukemia. 2014;28:241–247. doi: 10.1038/leu.2013.336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ganguly BB, Kadam NN. Mutations of myelodysplastic syndromes (MDS): An update. Mutat Res Rev Mutat Res. 2016;769:47–62. doi: 10.1016/j.mrrev.2016.04.009. [DOI] [PubMed] [Google Scholar]
- 20.Cui Y, et al. TET2 mutations were predictive of inferior prognosis in the presence of ASXL1 mutations in patients with chronic myelomonocytic leukemia. Stem Cell Investig. 2016;3 doi: 10.21037/sci.2016.09.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sperling AS, Gibson CJ, Ebert BL. The genetics of myelodysplastic syndrome: from clonal haematopoiesis to secondary leukaemia. Nat Rev Cancer. 2017;17:5–19. doi: 10.1038/nrc.2016.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schneider RK, et al. Rps14 haploinsufficiency causes a block in erythroid differentiation mediated by S100A8 and S100A9. Nat Med. 2016;22:288–297. doi: 10.1038/nm.4047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schneider RK, et al. Role of casein kinase 1A1 in the biology and targeted therapy of del(5q) MDS. Cancer Cell. 2014;26:509–520. doi: 10.1016/j.ccr.2014.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kronke J, et al. Lenalidomide induces ubiquitination and degradation of CK1α in del(5q) MDS. Nature. 2015;523:183–188. doi: 10.1038/nature14610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hasle H. Myelodysplastic and myeloproliferative disorders of childhood. Hematology Am Soc Hematol Educ Program. 2016;2016:598–604. doi: 10.1182/asheducation-2016.1.598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen C, et al. MLL3 is a haploinsufficient 7q tumor suppressor in acute myeloid leukemia. Cancer Cell. 2014;25:652–665. doi: 10.1016/j.ccr.2014.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McNerney ME, et al. CUX1 is a haploinsufficient tumor suppressor gene on chromosome 7 frequently inactivated in acute myeloid leukemia. Blood. 2013;121:975–983. doi: 10.1182/blood-2012-04-426965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nikoloski G, et al. Somatic mutations of the histone methyltransferase gene EZH2 in myelodysplastic syndromes. Nat Genet. 2010;42:665–667. doi: 10.1038/ng.620. [DOI] [PubMed] [Google Scholar]
- 29.Mackinnon RN, et al. The paradox of 20q11.21 amplification in a subset of cases of myeloid malignancy with chromosome 20 deletion. Genes Chromosomes Cancer. 2010;49 doi: 10.1002/gcc.20806. [DOI] [PubMed] [Google Scholar]
- 30.Hemmat M, et al. Submicroscopic deletion of 5q involving tumor suppressor genes (CTNNA1, HSPA9) and copy neutral loss of heterozygosity associated with TET2 and EZH2 mutations in a case of MDS with normal chromosome and FISH results. Mol Cytogenet. 2014;7 doi: 10.1186/1755-8166-7-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pellagatti A, Boultwood J. The molecular pathogenesis of the myelodysplastic syndromes. Eur J Haematol. 2015;95:3–15. doi: 10.1111/ejh.12515. [DOI] [PubMed] [Google Scholar]
- 32.Tsitsipatis D, et al. Synergistic killing of FLT3ITD-positive AML cells by combined inhibition of tyrosine-kinase activity and N-glycosylation. Oncotarget. 2017;8:26613–26624. doi: 10.18632/oncotarget.15772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.O’Keefe C, et al. Copy neutral loss of heterozygosity: a novel chromosomal lesion in myeloid malignancies. Blood. 2010;115:2731–2739. doi: 10.1182/blood-2009-10-201848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Barresi V, et al. Clonal selection of 11q CN-LOH and CBL gene mutation in a serially studied patient during MDS progression to AML. Leuk Res. 2010;34:1539–1542. doi: 10.1016/j.leukres.2010.07.004. [DOI] [PubMed] [Google Scholar]
- 35.Merkerova MD, et al. From cryptic chromosomal lesions to pathologically relevant genes: integration of SNP-array with gene expression profiling in myelodysplastic syndrome with normal karyotype. Genes Chromosomes Cancer. 2012;51:419–428. doi: 10.1002/gcc.21927. [DOI] [PubMed] [Google Scholar]
- 36.Wafa. A, et al. A new complex karyotype in a unique de novo myelodysplastic syndrome case involving ten chromosomes and monoallelic loss of TP53. Gene Reports. 2016;4:208–212. doi: 10.1016/j.genrep.2016.05.005. [DOI] [Google Scholar]
- 37.Murphy DM, et al. NRAS mutations with low allele burden have independent prognostic significance for patients with lower risk myelodysplastic syndromes. Leukemia. 2013;27:2077–2081. doi: 10.1038/leu.2013.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Papaemmanuil E, et al. Clinical and biological implications of driver mutations in myelodysplastic syndromes. Blood. 2013;122:3616–3627. doi: 10.1182/blood-2013-08-518886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ganster C, et al. New data shed light on Y-loss-related pathogenesis in myelodysplastic syndromes. Genes Chromosomes Cancer. 2015;54:717–724. doi: 10.1002/gcc.22282. [DOI] [PubMed] [Google Scholar]
- 40.Heinrichs S, et al. Accurate detection of uniparental disomy and microdeletions by SNP array analysis in myelodysplastic syndromes with normal cytogenetics. Leukemia. 2009;23:1605–1613. doi: 10.1038/leu.2009.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Evans AG, Ahmad A, Burack WR, Iqbal MA. Combined comparative genomic hybridization and single-nucleotide polymorphism array detects cryptic chromosomal lesions in both myelodysplastic syndromes and cytopenias of undetermined significance. Mod Pathol. 2016;29:1183–1199. doi: 10.1038/modpathol.2016.104. [DOI] [PubMed] [Google Scholar]
- 42.Stevens-Kroef M, Simons A, Rack K, Hastings RJ. Cytogenetic Nomenclature and Reporting. Methods Mol Biol. 2017;1541:303–309. doi: 10.1007/978-1-4939-6703-2_24. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.