Abstract
With the current commercial foot-and-mouth disease vaccine, inoculating twice increases the formation of denatured meat due to granuloma or residual adjuvant at the injection site in pigs, resulting in economic loss. Therefore, we investigated protective antibody levels after reducing the amount of adjuvant in the vaccine. Field applicability of the experimental vaccine, made with a new adjuvant ISA 201, was tested by vaccinating farm animals with half-volume doses (1 mL/animal) of commercial vaccine and monitoring their immunogenicity. Among pigs, the group that received a half-volume dose showed similar or higher titers of structural protein antibody and neutralizing antibody than those receiving the standard dose (2 mL). In pigs, the durable effects of antibody titer of the reduced vaccine volume did not diminish up to the time of slaughter. Among cattle, boosting with a second 1 mL vaccine increased virus neutralizing antibody for the protective effects. The boosting effects were more marked in cattle than in pigs. The immune responses differed between species with the effect of the half-volume vaccination being lower in cattle than in pigs. In conclusion, the immune response to the half-volume vaccine was similar to that from the standard volume vaccine in pigs, but not in cattle.
Keywords: foot-and-mouth disease, immunity, vaccination methods, vaccine
Introduction
Foot-and-mouth disease (FMD) is a highly contagious disease in domestic animals that forms vesicles in the mouths and on the hooves of cloven-hooved animals, resulting in decreased livestock productivity. The disease has high lethality among young animals [9], therefore, it can produce large-scale economic losses. The FMD virus (FMDV) can be spread through the respiratory system or via contact, and the virus spreads very rapidly within a herd once an animal is infected. Once FMD occurs, to prevent the spread of the disease destroying the affected animal is inevitable. Hence, vaccination is important for the prevention of FMD spread. Following a large-scale occurrence of FMD in November 2010, all farm animals susceptible to FMD were vaccinated in Korea [11]. Occurrence of the disease then appeared to end, but FMD recurred in July, August, and December of 2014, and again from January to March in 2016 [12]. From 2000 onwards, both type A and type O FMDVs have occurred in Korea. A trivalent vaccine [O1 Manisa, A May97 (or A22 Iraq), and Asia1 Shamir] was used to prevent three serotypes that have occurred in Korea. The O/Jincheon/SKR/2014 FMDV occurred in Korea in 2014 and 2015. The type O, Mya98 lineage FMDV occurred in 2016, whereas, in 2017, type O, Ind2001d lineage and type A, sea-97 lineage FMDVs were circulating in East Asian countries (unpublished data). Since 2015, the O1 Manisa + O 3039 vaccine has been used for pigs, and, with the addition of A22 Iraq, for cattle. There is a contention within the Korean pig farming industry that economic losses occur as a result of delayed absorption of the oil-based vaccine. It is thought that residual adjuvant can cause the formation of denatured meat and granuloma in the animal's neck region after vaccination.
Hence, we recognized the need to develop a new locally available vaccine that would increase the matching rate for domestically occurring FMDV strains and would improve protective effects. In addition, we investigated whether using a new vaccine adjuvant could improve protective effects and decrease the occurrence of denatured meat in pigs. In a previous study, we had observed that a new experimental vaccine made with the ISA 201 adjuvant improved protective effects and immunogenicity in experimental animals, including pigs [17]. To determine whether that previous study's result could be similarly achieved with animals in the field, we investigated protective effects in cattle and pigs by monitoring the neutralizing antibody level after inoculation with the experimental ISA 201-adjuvant FMD vaccine.
For the experimental vaccine, an antigen was produced and purified from a BHK-21 cell culture of the O/Andong/SKR/2010 FMDV strain, which occurred domestically in Korea in 2010. To estimate its protective capabilities, we tested antibody production and neutralization effects on a homologous FMDV of the O/Andong/SKR/2010 strain and on a heterologous virus of the O/Jincheon/SKR/2014 strain that was recently isolated in Korea.
With regard to the current commonly used FMD vaccine, an initial vaccine is administered to 8-week-old pigs and cattle and an additional boosting inoculation is administered after another 4 weeks. The additional inoculation not only increases the possibility of denatured meat formation such as granulomatous lesions at the injection site, but it also causes economic problems in a practical sense as it increases the demand for labor on farms for application of the second vaccine [2]. Therefore, we conducted an analysis of the difference between the protective antibody levels obtained from a standard vaccine and a decreased dose volume vaccine, which resulted in a reduced amount of in vivo oil adjuvant administration. In addition, we analyzed the differences between protective effects in single- and double-vaccination groups.
Materials and Methods
Virus purification and inactivation
The FMDV O/Andong/SKR/2010 (AD virus) and BHK-21 cells were used for antigen preparation. For viral infection, the culture medium was replaced with serum-free Dulbecco's modified Eagle's medium (DMEM; Cellgro, USA) and the cells were inoculated with the virus. After 1 h of incubation at 37℃ in an atmosphere of 5% CO2, the extracellular viruses were removed. Twenty-four hours postinfection, the viruses were inactivated twice by treating with 0.003 N binary ethylenimine for 24 h in a shaking incubator and were concentrated with polyethylene glycol (PEG) 6000 (81260; Sigma-Aldrich, USA). The virus concentrate was layered on 15%–45% sucrose density gradients and centrifuged [13]. After ultracentrifugation, the bottom of the centrifuge tube was punctured and 1 mL fractions were collected. The presence of FMDV particles in a sample of each fraction was determined by using a lateral flow device (BioSign FMDV Ag; Princeton BioMeditech, USA). Prior to its use in the field experiment, the pre-PEG treatment supernatant was passed through a ZZ-R cell line at least twice to check that no cytopathic effect (CPE) had occurred, thus confirming the absence of live virus in the supernatant. Moreover, 100 µL of purified antigen was inoculated into C57BL/6 mice via the intraperitoneal (IP) route [15]. We confirmed the absence of viremia in mouse serum by performing real-time polymerase chain reaction at 3 day postchallenge.
Preparation of the experimental vaccines
The concentrated O/Andong/SKR/2010 FMDV antigens were diluted with Tris-NaCl buffer (pH 7.6) and then added to Montanide ISA 201 VG (ISA 201; Seppic, France). The ratio of adjuvant volume to total volume was 50:50. The mixture was stirred at 300 rpm/min for 10 min at 30℃ in a water incubator in order to form a water-in-oil-in-water blend. The stability of the vaccines was tested by the using the dropping method [10].
Immunization of the animals
For the field experiment, 8-week-old cattle and pigs in three farmhouses were divided into three groups, each group containing 20 pigs or 5 cows (Table 1). Groups A for pigs and D for cattle received a single 10 µg/2 mL vaccine. Groups B for pigs and E for cattle received a single 10 µg/1 mL vaccine. Group C for pigs and F for cattle received a second 10 µg/1 mL vaccine 4 weeks after the initial 10 µg/1 mL vaccination. Pig serum was collected before the initial vaccination and at the 2nd, 3rd, 4th, 8th, and 12th week post vaccination (wpv). Bovine serum was collected before the initial vaccination, and at the 2nd, 4th, 8th, and 12th wpv. Animal experiments were performed in strict accordance with recommendations in the Guide for the Care and Use of Laboratory Animals of the Animal and Plant Quarantine Agency. All animal procedures were approved by the Institutional Animal Care and Use Committee of the Animal and Plant Quarantine Agency of Korea (IACUC No. 2016-343). All possible efforts were made to minimize animal suffering.
Table 1. Strategy for immunization with different vaccination methods in pigs and cattle.
ELISA for the detection of structural protein antibodies
For the detection of structural protein (SP) antibodies in sera, PrioCHECK FMDV type O (Prionics, Switzerland) was used. The absorbance of the enzyme-linked immunosorbent assay (ELISA) plate was converted to a percent inhibition (PI) value. When the PI value was 50% or above, the animals were regarded as antibody positive.
Virus neutralization test
Titers of neutralizing antibodies in the serum were measured by applying a virus neutralization test (VNT). Serum samples were collected from animals after vaccination and were heat-inactivated at 56℃ for 30 min. The cell density was adjusted to form a 70% monolayer, and the test was conducted by using a two-fold dilution of sera. Following incubation of the test serum with FMDV (O/Andong/SKR/2010 [=O/SKR(KOR)/Nov/2010, AD] or O/Jinchon/SKR/2014 [=O/SKR(KOR)/Dec/2014, JC]) 100 tissue culture infectious dose (TCID)50/0.05 mL for 1 h [17], LF-BK (bovine kidney) cells were added to the plate and incubated for 2–3 days [6]. The CPE was checked to determine the titers, which were calculated as the log10 of the reciprocal antibody dilution to neutralize 100 TCID50 of virus [5]. When the neutralizing antibody was 1.2 or above, the sera were regarded as antibody positive.
Statistical analysis
Student's t-tests were performed with GraphPad Instat (ver. 3.05; GraphPad Software, USA) to compare the vaccines' immunogenicity and protective effects between the various groups.
Results
Immune responses in pigs immunized with the experimental FMD vaccine
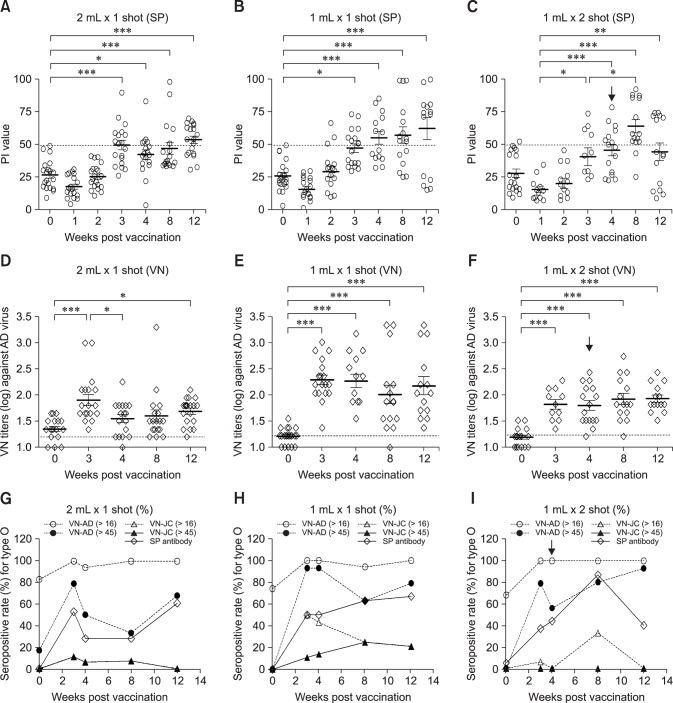
After vaccinating pigs and cattle, serum SP and virus neutralizing (VN) antibodies to the FMDV were observed (Figs. 1, 2, 3, 4). Group A (panels A, D, and G in Fig. 1), which received the single 10 µg/2 mL vaccination, had an SP antibody-positive rate of 52.6% at the 3rd wpv. The mean titer decreased at the 4th and 8th wpv, but the antibody-positive rate recovered (61.1%) at the 12th wpv. Similarly, group B (panels B, E, and H in Fig. 1), which had received the single 10 µg/1 mL vaccination, showed an SP antibody-positive rate of 50.0% at the 3rd wpv, and the rate continued to increase until the 12th wpv. Group C (panels C, F, and I in Fig. 1), which received an additional 10 µg/1 mL booster vaccination at the 4th week after the initial 10 µg/1 mL vaccination had the highest SP antibody-positive rate at the 8th wpv, indicating the effectiveness of the additional vaccination. However, the effect did not last and the SP antibody-positive rate had decreased by the 12th wpv.
Fig. 1. Antibody responses in pigs vaccinated with the experimental foot-and-mouth disease vaccine with an ISA 201 oil adjuvant. (A) PI values of type O SP-enzyme-linked immunosorbent assay (ELISA) from sera of pigs vaccinated with 2 mL of vaccine. (B) PI values of type O SP-ELISA from sera of pigs vaccinated with 1 mL of vaccine. (C) PI values of type O SP-ELISA from sera of pigs vaccinated twice with 1 mL of vaccine. (D) VN titers to O/Andong/SKR/2010 in sera of pigs vaccinated with 2 mL of vaccine. (E) VN titers to O/Andong/SKR/2010 in sera of pigs vaccinated with 1 mL of vaccine. (F) VN titers to O/Andong/SKR/2010 in sera of pigs vaccinated twice with 1 mL of vaccine. (G) Antibody-positive rate (%) for type O in pigs vaccinated with 2 mL of vaccine. (H) Antibody-positive rate (%) for type O in pigs vaccinated with 1 mL of vaccine. (I) Antibody-positive rate (%) for type O in pigs vaccinated twice with 1 mL of vaccine. The antibody-positive rate was applied based on positive cutoff with O-SP ELISA (PI > 50) and VNT (> 1:16 for positive baseline and 1:45 for protectable level). Twenty pigs were included in this study. Black arrows indicate second vaccination (boosting). PI, percent inhibition value in SP-ELISA; VN, virus neutralizing; SP antibody, antibody against structural protein in SP-ELISA; AD, O/Andong/SKR/2010; JC, O/Jincheon/SKR/2014; VN-AD, virus neutralizing titer against O/Andong/SKR/2010; VN-JC, virus neutralizing titer against O/Jincheon/SKR/2014. *p < 0.05, **p < 0.01, ***p < 0.001.
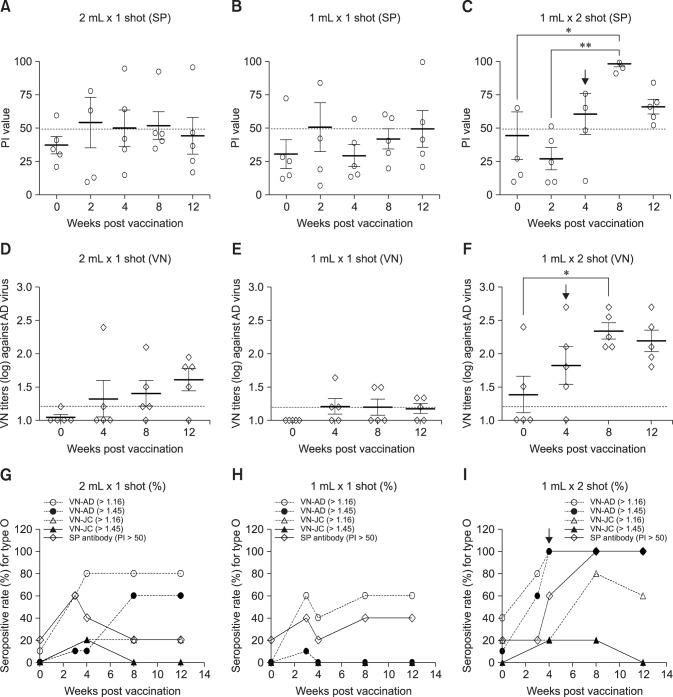
Fig. 2. Antibody responses in cattle vaccinated with the experimental foot-and-mouth disease vaccine with an ISA 201 oil adjuvant. (A) PI values of type O SP-enzyme-linked immunosorbent assay (ELISA) from sera of cattle vaccinated with 2 mL of vaccine. (B) PI values of type O SP-ELISA from sera of cattle vaccinated with 1 mL of vaccine. (C) PI values of type O SP-ELISA from sera of cattle vaccinated twice with 1 mL of vaccine. (D) VN titers to O/Andong/SKR/2010 in sera of cattle vaccinated with 2 mL of vaccine. (E) VN titers to O Andong/SKR/2010 in sera of cattle vaccinated with 1 mL of vaccine. (F) VN titers to O/Andong/SKR/2010 in sera of cattle vaccinated twice with 1 mL of vaccine. (G) Antibody-positive rate (%) of type O in cattle vaccinated with 2 mL of vaccine. (H) Antibody-positive rate (%) of type O in cattle vaccinated twice with 1 mL of vaccine. (I) Antibody-positive rate (%) of type O in cattle vaccinated twice with 1 mL of vaccine. The antibody-positive rate was applied by positive cutoff with O-SP ELISA (PI > 50) and VNT (> 1:16 for positive baseline and 1:45 for predicted protective level). Black arrows indicate second vaccination (boosting). PI, percent inhibition value in SP-ELISA; VN, virus neutralizing; SP antibody, antibody against structural protein in SP-ELISA; AD, O/Andong/SKR/2010; JC, O/Jincheon/SKR/2014; VN-AD, virus neutralizing titer against O/Andong/SKR/2010; VN-JC, virus neutralizing titer against O/Jincheon/SKR/2014. *p < 0.05, **p < 0.01, ***p < 0.001.
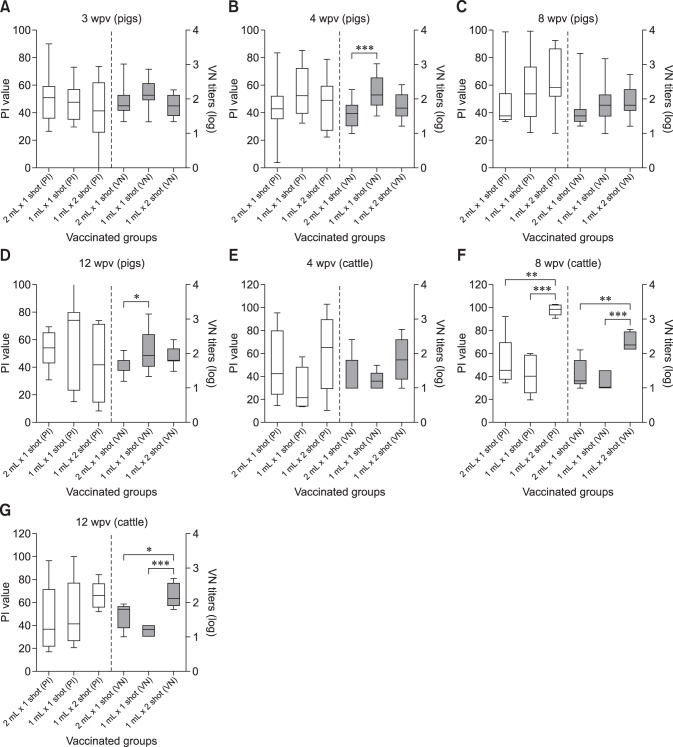
Fig. 3. Comparison of antibody responses in pig and cattle vaccinated with the experimental foot-and-mouth disease vaccine with an ISA 201 oil adjuvant. (A) Antibody responses (ELISA and VNT) of pigs at the 3rd week post vaccination (wpv). (B) Antibody responses (ELISA and VNT) of pigs at the 4th wpv. (C) Antibody responses (ELISA and VNT) of pigs at the 8th wpv. (D) Antibody responses (ELISA and VNT) of pigs at the 12th wpv. (E) Antibody responses (ELISA and VNT) of cattle at 4th wpv. (F) Antibody responses (ELISA and VNT) of cattle at the 8th wpv. (G) Antibody responses (ELISA and VNT) of cattle at the 12th wpv. PI, percent inhibition value in SP-ELISA; VN, virus neutralizing; ELISA, enzyme-linked immunosorbent assay; VNT, virus neutralization test. *p < 0.05, **p < 0.01, ***p < 0.001.
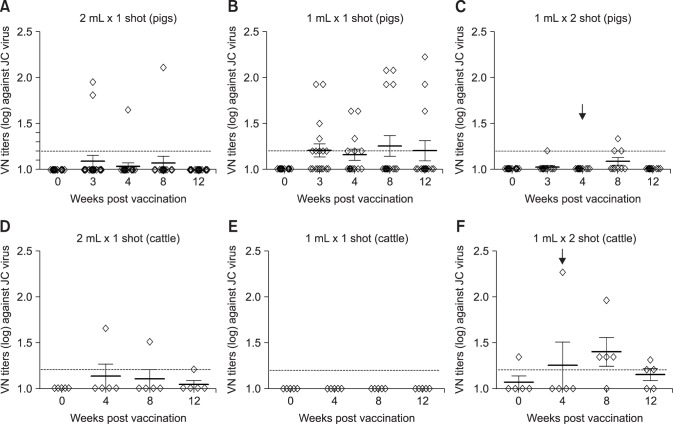
Fig. 4. Comparison of neutralizing titers in response to a heterologous virus in pig and cattle vaccinated with experimental foot-and-mouth disease vaccine. (A) Virus neutralizing (VN) titers to O/Jincheon/SKR/2014 in sera of pigs vaccinated with 2 mL of vaccine. (B) VN titers to O/Jincheon/SKR/2014 in sera of pigs vaccinated with 1 mL of vaccine. (C) VN titers to O/Jincheon/SKR/2014 in sera of pigs vaccinated twice with 1 mL of vaccine. (D) VN titers to O/Jincheon/SKR/2014 in sera of cattle vaccinated with 2 mL of vaccine. (E) VN titers in sera of cattle to O/Jincheon/SKR/2014 vaccinated with 1 mL of vaccine. (F) VN titers to O/Jincheon/SKR/2014 in sera of cattle vaccinated twice with 1 mL of vaccine. Black arrows indicate second vaccination (boosting).
The neutralizing antibody decreased in the single vaccination groups after the 3rd wpv, showing results that were similar to those obtained via ELISA (panels A–D in Fig. 3). Overall, group C, which was inoculated twice with the 10 µg/1 mL vaccine, had a higher antibody titer level (antibody-positive rate by VNT) than that in the 10 µg/1 mL single-dose group. However, the antibody titers in the 10 µg/1 mL-vaccinated groups showed a downward trend after the 4th wpv. Following the additional vaccination at the 4th wpv, the double 10 µg/1 mL-vaccinated group showed a protective antibody level (> 1:45 VNT, 60%–90%) that lasted until the 12th wpv.
Immune responses in cattle immunized with the experimental FMD vaccine
Two-month-old cows were vaccinated by using the same vaccine levels and doses as those used for the pigs (Figs. 2 and 3). The cows in group D (panels A, D, and G in Fig. 2), which received the single 10 µg/2 mL vaccination, had an SP antibody-positive rate of 60% at the 2nd wpv. However, the proportion of animals with positive reactions gradually decreased afterward. The group E cows (panels B, E, and H in Fig. 2), which received the single 10 µg/1 mL vaccination, had an SP antibody-positive rate of 20%–40% over the full observation period. In group F cows (panels C, F, and I in Fig. 2), which had received two 10 µg/1 mL vaccinations, the antibody-positive rate increased after the additional vaccination, and all animals in the group had positive PI values after the 8th wpv.
The neutralizing antibody titer in cow serum was measured before and after vaccination. In group D cows (10 µg/2 mL), the number of animals with had a positive neutralizing antibody gradually increased with time post vaccination. However, group E cows (single 10 µg/1 mL vaccination) did not exhibit an upward trend. For the twice-vaccinated group F cows, the neutralizing antibody titer increased after the additional vaccination, and all animals in the group had protective antibody titer levels (> 1:45) that persisted until the 12th wpv.
Immune responses against the domestically occurring heterologous FMDV
When measuring the neutralizing antibody titers against the FMDV (O/Jincheon/SKR/2014, JC virus) that occurred in Jincheon City in December 2014, the antibody titer was low in most groups (Fig. 4). Similar results were observed in the single- and twice-vaccinated groups. Among the pig groups, the 10 µg/1 mL once-vaccinated group (group B) contained animals that had developed some neutralizing antibodies, but most pigs exhibited a low neutralizing antibody level. In the twice-vaccinated cattle, the neutralizing antibody level increased slightly after the second vaccination and only decreased at 12th wpv. The results reveal a low level of antibody formation against the heterologous virus, even though it was of the same topotype that had occurred domestically in December 2014.
Discussion
Adjuvants increase the effects of a vaccine by stimulating an immune reaction. Oil, alum salts, and virosomes are mainly used as adjuvants because they increase the immune reaction by acting with lipopolysaccharides, double-stranded RNA, and single-stranded RNA. Hence, the choice of adjuvant is important in determining the effectiveness of a vaccine [7,8]. However, in the case of the oil adjuvant that is widely used in animals, local lesions at the injection site such as edemas, inflammatory reactions, and necrosis have been reported [1,18]. On farms, there are further issues related to the presence of denatured meat. For other virus vaccines, reducing vaccine volume to a minimum amount has been proposed as a method to decrease the side effects arising from the adjuvant. In particular, a previous study reported that inoculating with a reduced dose of a smallpox vaccine could maintain the vaccine's effectiveness and control morbidity at the same time [3]. With respect to the FMD vaccine, discarding denatured meat such as granuloma lesions resulting from residual adjuvant has been reported to result in a huge economic loss every year in the pig farming industry. There is also the problem that pigs show low immunogenicity and protective effects compared to those in cattle. Hence, the currently used FMD vaccine requires an improvement in its effectiveness; such an improvement can involve changing the vaccine components and/or amounts. Moreover, these problems should be solved in order to prevent the occurrence and spread of FMD. We previously confirmed the possibility of using an effective new vaccine formulation through an experiment involving specific adjuvant mixtures [14]. In the present study, we tested the field applicability of an experimental vaccine made with the adjuvant ISA 201 by vaccinating field animals (pigs and cows) and monitoring their immunogenicity. The FMD vaccine with ISA 201 adjuvant has been shown to provide a rapid cellular immune response in vaccinated animals [7], and in our previous study, we suggested that the ISA 201-based vaccine was more effective than the standard adjuvant FMD vaccine [14].
In the field experiment on pigs, the groups that received half (i.e., 10 µg/1 mL) of the original dose volume showed similar or higher values of SP antibody in ELISA results. Similar results were observed for the neutralizing antibody. It is conjectured that, although there were identical amounts of antigen injected, a larger amount of antigen was more promptly exposed in the 10 µg/1 mL inoculation group than in the 10 µg/2 mL inoculation group because less oil adjuvant would have entered the animal in the former group. In addition, we were able to confirm that the effects in the half-volume dose pigs did not diminish up to the time of slaughter, even though we reduced the dose volume but maintained the previously recommended amount of vaccine antigen.
However, no noticeable boosting effect from the second vaccination was observed in the SP antibody and neutralizing antibody levels for pigs. In contrast, in the field experiment with cattle, the SP antibody titer developed faster than in pigs and the boosting effects also greatly increased after the second vaccination. A similar phenomenon was observed in the neutralizing antibody levels. The results indicate that the effects of the second vaccination were greater in cattle than in pigs. Since serological reactivity was observed after the second vaccination, secondary vaccination will be required in cattle.
We expect to improve further the experimental FMD vaccine made with ISA 201 adjuvant for a solution of field problem. As we only checked the injection site with unaided eyes (data not shown), follow-up studies involving pathological examination might be needed. In a previous study [14], immunization in pigs and goats with the experimental vaccine using the same antigen and adjuvant was observed to protect against a homologous challenge virus. Follow-up studies to determine in vivo field protection will be required in the presence of an actual FMD infection, which can provide information on interference phenomena arising from diverse vaccines being used to inoculate young animals soon after birth and on maternal transfer of the antibody [4,16].
Acknowledgments
We thank the staff of the Foot-and-Mouth Disease Division and the Center for FMD Vaccine Research at the Animal and Plant Quarantine Agency. We thank Seppic, France and Intercare, Korea for supplying the adjuvant. This work was supported by the Animal and Plant Quarantine Agency, Gimcheon City, Korea.
Footnotes
Conflict of Interest: The authors declare no conflicts of interest.
References
- 1.Barnett PV, Pullen L, Williams L, Doel TR. International bank for foot-and-mouth disease vaccine: assessment of Montanide ISA 25 and ISA 206, two commercially available oil adjuvants. Vaccine. 1996;14:1187–1198. doi: 10.1016/s0264-410x(96)00055-2. [DOI] [PubMed] [Google Scholar]
- 2.Choi SH, Park SI. Economic burden of foot-and-mouth disease vaccination-induced injection site lesions in slaughtered pigs and its causal relationship. J Prev Vet Med. 2015;39:153–156. [Google Scholar]
- 3.Couch RB, Winokur P, Edwards KM, Black S, Atmar RL, Stapleton JT, Kissner JM, Shinefield H, Denny TN, Bybel MJ, Newman FK, Yan L National Institute of Allergy and Infectious Diseases Smallpox Vaccine Study Group. Reducing the dose of smallpox vaccine reduces vaccine-associated morbidity without reducing vaccination success rates or immune responses. J Infect Dis. 2007;195:826–832. doi: 10.1086/511828. [DOI] [PubMed] [Google Scholar]
- 4.Dekker A, Chénard G, Stockhofe N, Eblé PL. Proper timing of foot-and-mouth disease vaccination of piglets with maternally derived antibodies will maximize expected protection levels. Front Vet Sci. 2016;3:52. doi: 10.3389/fvets.2016.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fowler VL, Knowles NJ, Paton DJ, Barnett PV. Marker vaccine potential of a foot-and-mouth disease virus with a partial VP1 G-H loop deletion. Vaccine. 2010;28:3428–3434. doi: 10.1016/j.vaccine.2010.02.074. [DOI] [PubMed] [Google Scholar]
- 6.Fukai K, Morioka K, Yamada M, Nishi T, Yoshida K, Kitano R, Yamazoe R, Kanno T. Comparative performance of fetal goat tongue cell line ZZ-R 127 and fetal porcine kidney cell line LFBK-αvβ6 for Foot-and-mouth disease virus isolation. J Vet Diagn Invest. 2015;27:516–521. doi: 10.1177/1040638715584156. [DOI] [PubMed] [Google Scholar]
- 7.Ibrahim Eel-S, Gamal WM, Hassan AI, Mahdy Sel-D, Hegazy AZ, Abdel-Atty MM. Comparative study on the immunopotentiator effect of ISA 201, ISA 61, ISA 50, ISA 206 used in trivalent foot and mouth disease vaccine. Vet World. 2015;8:1189–1198. doi: 10.14202/vetworld.2015.1189-1198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Khorasani A, Madadgar O, Soleimanjahi H, Keyvanfar H, Mahravani H. Evaluation of the efficacy of a new oil-based adjuvant ISA 61 VG FMD vaccine as a potential vaccine for cattle. Iran J Vet Res. 2016;17:8–12. [PMC free article] [PubMed] [Google Scholar]
- 9.Koh WM, Bogich T, Siegel K, Jin J, Chong EY, Tan CY, Chen MI, Horby P, Cook AR. The epidemiology of hand, foot and mouth disease in Asia: a systematic review and analysis. Pediatr Infect Dis J. 2016;35:e285–e300. doi: 10.1097/INF.0000000000001242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McKercher PD, Graves JH. A review of the current status of oil adjuvants in foot-and-mouth disease vaccines. Dev Biol Stand. 1976;35:107–112. [PubMed] [Google Scholar]
- 11.Park JH, Lee KN, Kim SM, Lee HS, Ko YJ, Tark DS, Shin YK, Seo MG, Kim B. Reemergence of foot-and-mouth disease, South Korea, 2000-2011. Emerg Infect Dis. 2014;20:2158–2161. doi: 10.3201/eid2012.130518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Park JH, Tark D, Lee KN, Lee SY, Ko MK, Lee HS, Kim SM, Ko YJ, Seo MG, Chun JE, Lee MH, Kim B. Novel foot-and-mouth disease virus in Korea, July-August 2014. Clin Exp Vaccine Res. 2016;5:83–87. doi: 10.7774/cevr.2016.5.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Park JN, Lee SY, Chu JQ, Lee YJ, Kim RH, Lee KN, Kim SM, Tark DS, Kim B, Park JH. Protection to homologous and heterologous challenge in pigs immunized with vaccine against foot-and-mouth disease type O caused an epidemic in East Asia during 2010/2011. Vaccine. 2014;32:1882–1889. doi: 10.1016/j.vaccine.2014.01.067. [DOI] [PubMed] [Google Scholar]
- 14.Park ME, Lee SY, Kim RH, Ko MK, Lee KN, Kim SM, Kim BK, Lee JS, Kim B, Park JH. Enhanced immune responses of foot-and-mouth disease vaccine using new oil/gel adjuvant mixtures in pigs and goats. Vaccine. 2014;32:5221–5227. doi: 10.1016/j.vaccine.2014.07.040. [DOI] [PubMed] [Google Scholar]
- 15.Park ME, Lee SY, Kim RH, Ko MK, Park JN, Lee KN, Kim SM, Choi JH, You SH, Kim B, Lee JS, Park JH. Altered adjuvant of foot-and-mouth disease vaccine improves immune response and protection from virus challenge. Trial Vaccinol. 2016;5:97–104. [Google Scholar]
- 16.Patil PK, Sajjanar CM, Natarajan C, Bayry J. Neutralizing antibody responses to foot-and-mouth disease quadrivalent (type O, A, C and Asia 1) vaccines in growing calves with pre-existing maternal antibodies. Vet Microbiol. 2014;169:233–235. doi: 10.1016/j.vetmic.2014.01.005. [DOI] [PubMed] [Google Scholar]
- 17.Xu M, Zuest R, Velumani S, Tukijan F, Toh YX, Appanna R, Tan EY, Cerny D, MacAry P, W CI, Fink K. A potent neutralizing antibody with therapeutic potential against all four serotypes of dengue virus. NPJ Vaccines. 2017;2:2. doi: 10.1038/s41541-016-0003-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yamanaka M, Hiramatsu K, Hirahara T, Okabe T, Nakai M, Sasaki N, Goto N. Pathological studies on local tissue reactions in guinea pigs and rats caused by four different adjuvants. J Vet Med Sci. 1992;54:685–692. doi: 10.1292/jvms.54.685. [DOI] [PubMed] [Google Scholar]